
The contents of this document do not have the force and effect of law and are not meant to bind the public
in any way, unless specifically incorporated into a contract. This document is intended only to provide
clarity to the public regarding existing requirements under the law.
Medicare Administrative Contractor (MAC) COVID-19 Test Pricing
January 25, 2021
On March 5 and February 13, 2020, CMS announced new Healthcare Common Procedure Coding
System (HCPCS) codes for healthcare providers and laboratories to use to bill Medicare for SARS-
CoV2 tests. Starting in April 2020, laboratories performing SARS-CoV2/2019-nCoV (COVID-19)
tests developed by the Centers for Disease Control and Prevention (CDC) could bill Medicare and
other health insurers for services that occurred after February 4, 2020 using the newly created
HCPCS code U0001, and laboratories performing non-CDC laboratory tests for SARS-CoV-2/2019-
nCoV (COVID-19) could bill for them using HCPCS code U0002. Additionally, the American
Medical Association (AMA) created CPT code 87635 for infectious agent detection by nucleic acid
tests on March 13, 2020, as well as CPT codes 86769 and 86328 for serology tests on April 10,
2020. Laboratories performing these tests may bill Medicare for services that occurred after the
codes’ respective effective dates.
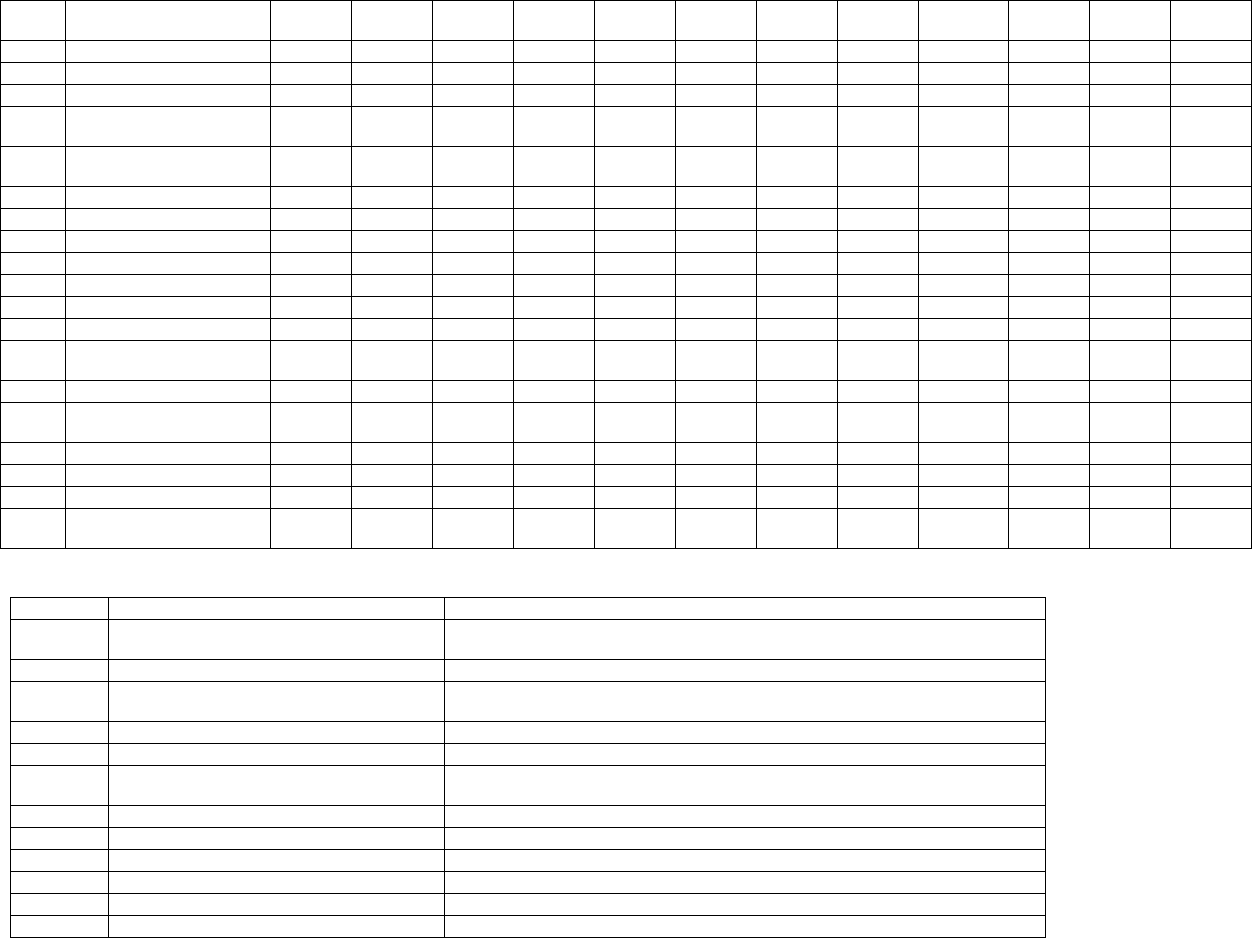
Local Medicare Administrative Contractors (MACs) are responsible for developing the payment
amount for claims they receive for newly created HCPCS codes in their respective jurisdictions
until Medicare establishes national payment rates. The MAC-developed payment amounts are
identified below. Since the last update to this table on May 19, 2020, a number of additional CPT
codes have been created to describe types of COVID-19 laboratory testing.
As with other laboratory tests, there is generally no beneficiary cost sharing for COVID-19
laboratory tests under Medicare.
Note: Rates for HCPCS codes U0003, U0004, and U0005 established in CMS Ruling 2020-1-R and
CMS Ruling 2020-1-R2 do not appear in the table below.
The contents of this document do not have the force and effect of law and are not meant to bind the public in any way, unless specifically incorporated into a
contract. This document is intended only to provide clarity to the public regarding existing requirements under the law.
MAC COVID-19 Test Pricing
CPT
Code
Short Descriptor
JE
JF
JJ
JM
JH
JL
JN
J15
J6
JK
J5
J8
U0001
2019-Ncov Diagnostic P
$35.91
$35.91
$35.91
$35.91
$35.92
$35.92
$35.92
$35.92
$35.91
$35.91
$35.92
$35.92
U0002
Covid-19 Lab Test Non-CDC
$51.31
$51.31
$51.31
$51.31
$51.31
$51.31
$51.31
$51.31
$51.31
$51.31
$51.31
$51.31
87635
SarsCov2 Covid19 Amp Prb
$51.31
$51.31
$51.31
$51.31
$51.31
$51.31
$51.31
$51.31
$51.31
$51.31
$51.31
$51.31
87636
SarsCov2 & Inf A&B Amp
Prb
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
87637
SarsCov2 & Inf A&B&RSV
Amp Prb
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
0240U
Nfct DS Vir Resp RNA 3 Trgt
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
0241U
Nfct DS Vir Resp RNA 4 Trgt
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
$142.63
87426
SarsCov Coronavirus AG IA
$35.33
$35.33
$35.33
$35.33
$45.23
$45.23
$45.23
$35.33
$35.33
$35.33
$35.33
$35.33
87428
SarsCov & Inf Vir A&B AG IA
$63.59
$63.59
$63.59
$63.59
$73.49
$73.49
$73.49
$63.59
$63.59
$63.59
$63.59
$63.59
87811
SarsCov2 Covid19 W/Optic
$41.38
$41.38
$41.38
$41.38
$41.38
$41.38
$41.38
$41.38
$41.38
$41.38
$41.38
$41.38
86328
Ia Nfct A&B SarsCov2
Covid19
$45.23
$45.23
$45.23
$45.23
$45.23
$45.23
$45.23
$45.23
$45.23
$45.23
$45.23
$45.23
86408
Neutrlzg Antb SarsCov2 Scr
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
86409
Neutrlzg Antb SarsCov2
Titer
$79.61
$79.61
$79.61
$79.61
$105.33
$105.33
$105.33
$79.61
$105.33
$105.33
$79.61
$79.61
86413
SarsCov2 Antb Quantative
$51.43
$51.43
$51.43
$51.43
$42.13
$42.13
$42.13
$51.43
$51.43
$51.43
$51.43
$51.43
86769
SarsCov2 Covid19 Antibody
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
0224U
Antibody SarsCov2 Titer(s)
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
$42.13
0226U
Svnt SarsCov2 Elisa Plsm
Srm
$42.28
$42.28
$42.28
$42.28
$42.28
$42.28
$42.28
$42.48
$42.28
$42.28
$42.28
$42.28
Key
Jurisdiction
MAC
MAC States
J5
Wisconsin Physicians Service Government
Health Administrators
Iowa, Kansas, Missouri, Nebraska
J6
National Government Services, Inc.
Illinois, Minnesota, Wisconsin
J8
Wisconsin Physicians Service Government
Health Administrators
Indiana, Michigan
J15
Wyoming CGS Administrators, LLC
Kentucky, Ohio
JE
Noridian Healthcare Solutions, LLC
California, Hawaii, Nevada, American Samoa, Guam, Northern Mariana Islands
JF Noridian Healthcare Solutions, LLC
Alaska, Arizona, Idaho, Montana, North Dakota, Oregon, South Dakota, Utah,
Washington, Wyoming
JH
Novitas Solutions, Inc.
Arkansas, Colorado, New Mexico, Oklahoma, Texas, Louisiana, Mississippi
JJ
Palmetto GBA, LLC
Alabama, Georgia, Tennessee
JK
National Government Services, Inc.
Connecticut, New York, Maine, Massachusetts, New Hampshire, Rhode Island, Vermont
JL
Novitas Solutions, Inc.
Delaware, District of Columbia, Maryland, New Jersey, Pennsylvania
JM
Palmetto GBA, LLC
North Carolina, South Carolina, Virginia, West Virginia
JN
First Coast Service Options, Inc.
Florida, Puerto Rico, U.S. Virgin Islands
