
DEPARTMENT OF HEALTH AND HUMAN SERVICES
Centers for Disease Control and Prevention
National Center for Health Statistics
01-0304 (10/01)
Copyright Information
Permission has been obtained from the
copyright holders to reproduce certain
quoted material in this report. Further
reproduction of this material is
prohibited without specific permission
of the copyright holder. All other
material contained in this report is in the
public domain and may be used and
reprinted without special permission;
citation as to source, however, is
appreciated.
Suggested Citation
National Center for Health Statistics.
Healthy People 2000 Final Review.
Hyattsville, Maryland: Public Health
Service. 2001.
Library of Congress Catalog Card
Number 76–641496

DEPARTMENT OF HEALTH AND HUMAN SERVICES
Centers for Disease Control and Prevention
National Center for Health Statistics
Hyattsville, Maryland
October 2001
DHHS Publication No. 01-0256
Department of Health and Human Services
Tommy G. Thompson
Secretary
Centers for Disease Control and Prevention
Jeffrey P. Koplan, M.D., M.P.H.
Director
National Center for Health Statistics
Edward J. Sondik, Ph.D.
Director

Preface
The Healthy People 2000 Final
Review is the seventh and last in a
series of profiles tracking the year 2000
objectives. This report was compiled by
the National Center for Health Statistics,
Centers for Disease Control and
Prevention (CDC), with considerable
input from the Department of Health
and Human Service’s lead agencies for
the year 2000 objectives, the Office of
Disease Prevention and Health
Promotion and the Office on Minority
Health. The Healthy People 2000
Steering Committee and the National
Committee on Vital and Health Statistics
served in a review capacity.
The Healthy People 2000 Review,
which replaced the Prevention Profiles
that monitored the 1990 national health
objectives, continued the series of
profiles of the Nation’s health objectives
as an integral part of the Department’s
disease prevention and health promotion
initiative for the year 2000. This
initiative was unveiled in September
1990 by the Secretary of the Department
of Health and Human Services with the
release of Healthy People 2000:
National Health Promotion and Disease
Prevention Objectives. This publication,
which incorporates the 1995 midcourse
review modifications to the objectives,
provides the latest available tracking
data for objectives and subobjectives in
all priority areas throughout the decade.
iii

Foreword
Since 1990, Healthy People 2000
has provided the national health
promotion agenda that guided
Americans toward living longer and
healthier lives. Healthy People 2000 not
only identified objectives to improve the
Nation’s health, but also set measurable
targets to monitor progress toward its
goals. The ability to quantify and assess
progress on health objectives is at the
heart of the Healthy People initiative.
This Healthy People 2000 Final Review
completes the series of Healthy People
2000 Reviews published to monitor and
evaluate the Nation’s progress toward
the year 2000 targets. As such, it
presents the final assessments of our
progress in the decade.
Healthy People 2000 was an
ambitious effort and invaluable in
setting 319 specific objectives for the
Nation. In fact, progress was achieved
on over 60 percent of the objectives.
Some of the major accomplishments
include surpassing the target for
reducing deaths from coronary heart
disease and cancer. The Nation also met
its targets for AIDS incidence, primary
and secondary syphilis cases,
mammography exams, and violent
(homicide, suicide, and firearm-related)
deaths. The tobacco-related mortality
targets were also met. Both infant
mortality and the number of children
with elevated blood lead levels nearly
met their targets as well. The Nation
also made progress toward the goal of
reducing health disparities for more than
one-half of the special population
objectives identified to be at increased
risk by Healthy People 2000. We can all
take pride in the fact that the past
decade saw substantial improvements in
the quality of life for many Americans.
Underlying and contributing to the
progress in preventing disease and
improving health are the advances in
health information. Healthy People 2000
was a catalyst at all levels of
government for the creation and
dissemination of quality, comparable
health data.
This final edition of the Healthy
People 2000 Review contains several
new data assessments that should prove
valuable for many of our constituents.
The first is an assessment of the
progress toward the second goal of
Healthy People 2000, reducing health
disparities. The assessment is based on
data collected for nine special
populations that are at increased health
risk: four race/ethnicity groups, women,
two age groupings, disability, and
socioeconomic status. Another is the
presentation of data for the 47 ‘‘sentinel
objectives’’ developed to represent the
scope and magnitude of Healthy People
2000. This includes data for each
sentinel objective at the national level
and State data for those objectives
where sources were available. Finally, in
addition to data that update the trends
shown in previous reports for the 18
Health Status Indicators, this report
shows data for the first time for the 16
Priority Data Needs developed at the
same time as the Health Status
Indicators but which lacked data for
assessment below the national level.
Although many gains in health
promotion and disease prevention have
been made, much remains to be done.
For 15 percent of the Healthy People
2000 objectives, the movement was
actually away from the targets. In
January 2000, Healthy People 2010 was
released, ushering the Healthy People
initiative into the new millennium. The
monitoring and assessment goals of
Healthy People 2010 are even more
ambitious than those that have directed
our efforts over the past decade. For
example, the Healthy People 2000 goal
to reduce health disparities has been
strengthened in Healthy People 2010 to
focus on eliminating health disparities.
Another important development for
the next decade is the publication
Tracking Healthy People 2010.Asa
guidebook on the statistics used for
Healthy People, it will assist programs
at the local, State, and national levels in
producing and using the requisite data
and, thereby, measurably advance the
information base available to enhance
the public’s health. Thanks to the solid
foundation laid by the accomplishments
of Healthy People 2000, Healthy People
2010 will be even stronger, as it builds
on the experiences of the last decade,
identifies our current health
opportunities and challenges, and
furthers the development of partnerships
among public and private organizations
to improve the health of the American
people.
Edward J. Sondik, Ph.D. Director,
National Center for Health Statistics
iv

Acknowledgments
Overall responsibility for planning
and coordinating the content of the
Healthy People 2000 Final Review
rested with the Division of Health
Promotion Statistics (DHPS), Office of
Analysis, Epidemiology, and Health
Promotion (OAEHP), National Center
for Health Statistics (NCHS), under the
general direction of Diane K. Wagener
and Richard J. Klein.
Production coordination was shared
among DHPS analysts: John
Aberle-Grasse, Elizabeth W. Jackson,
Colleen M. Ryan, and Kathleen M.
Turczyn.
Production was accomplished by
several DHPS working teams, which
included John Aberle-Grasse, Manon
Boudreault, Lillie Featherstone, Tracy
Fenwick, Elizabeth W. Jackson, Kenneth
Keppel, Insun Kim, Richard J. Klein,
Michael Molla, Jeffrey N. Pearcy,
Suzanne E. Proctor, Cheryl V. Willis,
Colleen M. Ryan, Thomas C. Socey,
Kathleen M. Turczyn, Diane K.
Wagener, and Jean F. Williams.
The sections discussing the Healthy
People 2000 goals were developed with
substantial input by the Office of
Minority Health (OMH)—particularly
Olivia D. Carter-Pokras and Tuei
Doong, the Office of Disease Prevention
and Health Promotion (ODPHP)—
particularly Omar Passons, the Agency
for Healthcare Research and Quality
(AHRQ), and the Health Resources and
Services Administration (HRSA)—
particularly David Atkins, Melissa
Clarke, Paul Nannis, Kathryn Rickard,
and Lyman Van Nostrand.
The priority area chapters were
coordinated by DHPS analysts and
written by working teams as follows:
Priority Area 1: Carol Macera
(CDC) and Christine Spain (OPHS)
Priority Area 2: Carter Blakely
(ODPHP), Nancy T. Crane (FDA), and
Pamela E Starke-Reed (NIH)
Priority Area 3: Karil Bialostosky
(CDC)
Priority Area 4: James Colliver
(NIH), Wendy Davis (SAMHSA), Susan
Farrell (NIH), Ann Mahony (SAMSHA),
Beatrice Rouse (SAMSHA), and Dorita
Sewell (SAMHSA)
Priority Area 5: Evelyn Kappeler
(OPHS)
Priority Area 6: Michele Edwards
(SAMHSA), Doreen Koretz (NIMH),
Ann Maney (NIMH), and Dorita Sewell
(SAMHSA)
Priority Area 7: Mark Anderson
(CDC), Lee Annest (CDC), Alex Crosby
(CDC), Martha Highsmith (CDC), Tom
Simon (CDC), and Tim Thornton (CDC)
Priority Area 8: Melissa Clarke
(HRSA), Catherine Hutsell (CDC), and
Brick Lancaster (CDC)
Priority Area 9: Tim Groza (CDC)
Priority Area 10: Bob Castellan
(CDC), Dawn Castillo (CDC), Regina
Pana-Cryan (CDC), Bill Eschenbacher
(CDC), John Franks (CDC), Joe Hurrell
(CDC), Lynn Jenkins (CDC), Jay Jones
(CDC), Boris Lushniak (CDC), Mark
Mendell (CDC), Bob Roscoe (CDC),
Lee Sanderson (CDC), John Sestito
(CDC), and Rosie Sokas (CDC)
Priority Area 11: David Evans
(ATSDR), Paul Garbe (CDC), and John
Schelp (NIH)
Priority Area 12: Linda S. Brophy
(FDA) and Elisa Elliot (FDA)
Priority Area 13: Alice M. Horowitz
(NIH), Candace M. Jones (HRSA), and
Stuart Lockwood (CDC)
Priority Area 14: Charlotte
Dickinson (CDC), Renee Schwalberg
(CDC), Myra Tucker (CDC), and Stella
M. Yu (HRSA)
Priority Area 15: Matilde Alvarado
(NIH), James Cleeman (NIH), Janet B.
Croft (CDC), Karen Donato (NIH),
Robinson Fulwood (NIH), Wayne H.
Giles (CDC), Kurt Greenlund (CDC),
Jeanette Guyton-Krishnan (NIH), Mary
M. Hand (NIH), Greg Heath (CDC),
Keith Hewitt (NIH), Darwin Labarthe
(CDC), Terry Long (NIH), George A.
Mensah (CDC), Greg Morosco (NIH),
Linda Redman (CDC), Edward J.
Roccella (NIH), Nancy Watkins (CDC),
and Zhi-Jie Zheng (CDC)
Priority Area 16: Barry Portnoy
(NIH) and Karen Richard (CDC)
Priority Area 17: Stephanie
Benjamin (CDC), Ben Burton (NIH),
Bill Foster (NIH), Linda Geiss (CDC),
Ed Tierney (CDC), and Frank Vinicor
(CDC)
Priority Area 18: Dave Brownell
(CDC), Brenda Garza (CDC), Shelley
Gordon (HRSA), Stacie Greby (CDC),
Gena Hill (CDC), Joyce Neal (CDC),
Ida Onorato (CDC), and Eva Seiler
(CDC)
Priority Area 19: Susan C. Clark
(CDC), Melinda Flock (CDC), Joel R.
Greenspan (CDC), Dana M. Shelton
(CDC), and Jack N. Spencer (CDC)
Priority Area 20: Jennifer Brooks
(CDC), Sarah Foster (CDC), John
Seggerson (CDC), and Nicole Smith
(CDC)
Priority Area 21: David Atkins
(AHRQ), Melissa Clarke (HRSA), Paul
Nannis (HRSA), Kathryn Rickard
(AHRQ), and Lyman Van Nostrand
(HRSA)
Priority Area 22: Diane Wagener
(CDC)
Technical review was provided by
Jennifer Madans, NCHS Associate
Director for Science. Publications
management and editorial review were
provided by Gail V. Johnson and
Rolfe W. Larson. The designer was
Sarah M. Hinkle. Graphics were
produced by Dorothy Day and
supervised by Stephen L. Sloan.
Production was done by Annette F.
Holman. Electronic publishing was done
by Annette F. Holman, Christine Brown,
and Margaret Avery. Printing was
managed by Patty L Wilson and
Joan Burton.
Publication of Healthy People 2000
Final Review would not have been
possible without the contributions of
many staff members throughout NCHS
and numerous other agencies,
particularly Carter Blakey, Office of
Disease Prevention and Health
Promotion. These people gave
generously of their time and knowledge;
their cooperation and assistance are
gratefully acknowledged.
NOTE: Centers for Disease Control and
Prevention (CDC), Office of Public Health Service
(OPHS), Office of Disease Prevention and Health
Promotion (ODPHP), Food and Drug
Administration (FDA), National Institute of Health
(NIH), Substance Abuse and Mental Health
Services Administration (SAMHSA), National
Institute of Mental Health (NIMH), Health
Resources and Services Administration (HRSA),
Agency for Toxic Substances and Disease Registry
(ATSDR), Agency for Healthcare Research and
Quality (AHRQ).
v

Contents
Preface iii
Foreword
iv
Acknowledgments v
List of Figures
ix
List of Tables
xi
Introduction 1
Background
1
Summary of Progress 2
Organization and Scope of this Review
4
Healthy People 2000 Goals
7
Goal 1: Increase the Span of Healthy Life 7
Life Expectancy at Birth
7
Fair or Poor Health 7
Healthy and Unhealthy Life
7
Goal 2: Reducing Health Disparities 8
Reducing Disparities: American Indian/Alaska Native
9
Reducing Disparities: Asian or Pacific Islander
10
Reducing Disparities: Black or African American 10
Reducing Disparities: Hispanic or Latino 10
Reducing Disparities: Women
10
Reducing Disparities: Adolescents and Young Adults 10
Reducing Disparities: Older Adults
11
Reducing Disparities: People with Disabilities
11
Reducing Disparities: People with Low Socioeconomic Status 11
Goal 3: Achieving Access to Preventive Services
11
Health Status Indicators and Priority Data Needs 19
Health Status Indicators
19
Priority Data Needs 19
Sentinel Objectives
27
National Data
27
State Data 30
Healthy People 2000 Priority Areas
1. Physical Activity and Fitness
65
2. Nutrition 76
3. Tobacco
93
4. Substance Abuse: Alcohol and Other Drugs 108
5. Family Planning 117
6. Mental Health and Mental Disorders
127
7. Violent and Abusive Behavior
137
8. Educational and Community-Based Programs 146
9. Unintentional Injuries
155
10. Occupational Safety and Health 167
11. Environmental Health
176
12. Food and Drug Safety 187
13. Oral Health
195
14. Maternal and Infant Health 206
15. Heart Disease and Stroke
217
16. Cancer
228
17. Diabetes and Chronic Disabling Conditions 238
18. HIV Infection
254
19. Sexually Transmitted Diseases 267
20. Immunization and Infectious Diseases
279
21. Clinical Preventive Services 294
22. Surveillance and Data Systems
305
Appendix 315
vii

List of Figures
A. Healthy People 2000 objectives: Summary of progress by priority area 3
B. Life expectancy at birth: United States, 1988–98
8
C. Percent of people with fair or poor health: United States, 1990–98 9
D. Expected years of healthy and unhealthy life: United States, 1998
10
E. Assessment of disparity for American Indian/Alaska Native objectives 11
F. Assessment of disparity for Asian or Pacific Islander objectives
11
G. Assessment of disparity for black or African American objectives 12
H. Assessment of disparity for Hispanic or Latino objectives
12
J. Assessment of disparity for women objectives 13
K. Assessment of disparity for adolescents and young adults objectives
13
L. Assessment of disparity for older adults objectives 14
M. Assessment of disparity for people with disabilities objectives
14
N. Assessment of disparity for people with low socioeconomic status objectives
15
O. Access to care: United States, 1988–99 15
P. Percent of people under 65 years without health insurance coverage: United States, 1998 16
Q. Objective 1.3–Percent of adults who participated in regular physical activity (5 or more times per week):
United States and each State, 1996 and 1998
32
R. Objective 1.5–Percent of adults who reported no leisure-time physical activity: United States and each State, 1996
and 1998
33
S. Objective 2.3–Percent of persons 18 years and older who were overweight: United States and each State, 1998
and 1999
34
T. Objective 3.4–Percent of persons 18 years and older who reported current cigarette smoking: United States and each
State, 1998 and 1999 35
U. Objective 4.1–Alcohol-related motor vehicle crash deaths per 100,000 population: United States and each State,
1997 and 1998
36
W. Objective 4.6–Percent of youth who currently use marijuana: United States and each State, 1999 37
Y. Objective 4.6–Percent of youth who currently smoke cigarettes: United States and each State, 1999
38
Z. Objective 5.1–Live birth rates for females 15–17 years old: United States and each State, 1997 39
AA. Objective 6.1–Age-adjusted death rates for suicide: United States and each State, 1997 and 1998
40
BB. Objective 7.1–Age-adjusted death rates for homicide: United States and each State, 1997 and 1998 41
CC. Objective 9.1–Age-adjusted death rates for unintentional injuries: United States and each State, 1997 and 1998
42
DD. Objective 9.12–Percent of persons wearing a safety belt while driving or riding in a car: United States and each
State, 1995 and 1997
43
EE. Objective 10.1–Work-related injury death rates: United States and each State, 1998 and 1999 44
FF. Objective 10.2–Nonfatal work-related injuries per 100 full-time workers in private industry: United States and
reporting States, 1997 and 1998
45
GG. Objective 11.5–Percent of persons in counties that have not exceeded the Environmental Protection Agency (EPA)
standards for air quality: United States and each State, 1997 and 1998
46
HH. Objective 14.1–Infant mortality rates: United States and each State, 1997 and 1998
47
JJ. Objective 14.5–Percent of live births of low birthweight: United States and each State, 1997 and 1998 48
KK. Objective 14.11–Percent of mothers who began prenatal care in the first trimester: United States and each State,
1997 and 1998
49
LL. Objective 15.1–Age-adjusted death rates for coronary heart disease: United States and each State, 1997 and 1998
50
MM. Objective 15.2–Age-adjusted death rates for stroke: United States and each State, 1997 and 1998 51
NN. Objective 16.1–Age-adjusted death rates for all cancers: United States and each State, 1997 and 1998
52
OO. Objective 16.11–Percent of women 50 years of age and older who received a mammogram and breast exam within
the last 2 years: United States and each State, 1998 and 1999
53
PP. Objective 16.12–Percent of women 18 years of age and older who had a Pap test during the last 3 years:
United States and each State, 1998 and 1999
54
QQ. Objective 16.13–Percent of persons 50 years of age and older who have had a fecal occult blood test during the last
2 years: United States and each State, 1997 and 1999
55
RR. Objective 16.13–Percent of persons 50 years of age and older who have ever had a sigmoidoscopy or proctoscopic
exam: United States and each State, 1997 and 1999
56
SS. Objective 17.9–Age-adjusted death rates for diabetes-related deaths: United States and each State, 1997 and 1998
57
TT. Objective 19.1–Reported incidence of gonorrhea per 100,000 population: United States and each State, 1998 and
1999
58
UU. Objective 19.3–Reported cases of primary and secondary syphilis per 100,000 population: United States and each
State, 1998 and 1999
59
WW. Objective 20.1–Reported cases of measles: United States and each State, 1998 and 1999 60
viii

YY. Objective 20.11–Estimated vaccination coverage with the 4DTP/3polio/1MMR series among children ages
19–35 months: United States and each State, 1998 and 1999
61
ZZ. Objective 21.4–Percent of persons under 65 years of age with no health insurance coverage: United States and each
State, 1998 and 1999
62
Priority Area Progress Figures
1. Final status of Physical Activity and Fitness objectives 66
2. Final status of Nutrition objectives
77
3. Final status of Tobacco objectives 94
4. Final status of Substance Abuse: Alcohol and Other Drugs objectives 109
5. Final status of Family Planning objectives
118
6. Final status of Mental Health and Mental Disorders objectives 128
7. Final status of Violent and Abusive Behavior objectives
138
8. Final status of Educational and Community-Based Programs objectives
150
9. Final status of Unintentional Injuries objectives 156
10. Final status of Occupational Safety and Health objectives 168
11. Final status of Environmental Health objectives
177
12. Final status of Food and Drug Safety objectives 188
13. Final status of Oral Health objectives
196
14. Final status of Maternal and Infant Health objectives
207
15. Final status of Heart Disease and Stroke objectives 218
16. Final status of Cancer objectives
229
17. Final status of Diabetes and Chronic Disabling Conditions objectives 239
18. Final status of HIV Infection objectives
255
19. Final status of Sexually Transmitted Diseases objectives 268
20. Final status of Immunization and Infectious Diseases objectives
280
21. Final status of Clinical Preventive Services objectives 295
22. Final status of Surveillance and Data Systems objectives
304
ix

List of Tables
A. Life-stage objectives: United States, 1987–98 2
B. Health Status Indicators: United States, 1996–98
20
C. Health Status Indicators by race and Hispanic origin: United States, 1998
21
D. Priority Data Needs 22
E. An Assessment of Target Attainment for the Healthy People 2000 Sentinel Health Objectives for the United States
and Individual States
28
Priority Area Summary Tables
1. Physical Activity and Fitness objectives 70
2. Nutrition objectives
83
3. Tobacco objectives
100
4. Substance Abuse: Alcohol and Other Drugs objectives 112
5. Family Planning objectives
121
6. Mental Health and Mental Disorders objectives 132
7. Violent and Abusive Behavior objectives 142
8. Educational and Community-Based Programs objectives
150
9. Unintentional Injuries objectives
160
10. Occupational Safety and Health objectives 171
11. Environmental Health objectives
181
12. Food and Drug Safety objectives 192
13. Oral Health objectives
199
14. Maternal and Infant Health objectives 211
15. Heart Disease and Stroke objectives
221
16. Cancer objectives
231
17. Diabetes and Chronic Disabling Conditions objectives 244
18. HIV Infection objectives
259
19. Sexually Transmitted Diseases objectives 272
20. Immunization and Infectious Diseases objectives 285
21. Clinical Preventive Services objectives
298
22. Surveillance and Data Systems objectives
309
x

Introduction
Background
History of the Healthy People
Initiative
The Healthy People process began
in 1979 with the release of Healthy
People: The Surgeon General’s Report
on Health Promotion and Disease
Prevention (1), which provided national
goals for reducing premature deaths and
for preserving independence for older
adults. The five primary goals of the
1979 Surgeon General’s report were to
enhance the health of the U.S.
population in five major life stages
(infants, children, adolescents and young
adults, adults, and older adults).
Different focal points were targeted for
each age group, such as low birth-
weight and birth defects for infants, and
functional independence, influenza, and
pneumonia for older adults. Fifteen
priority areas were also identified as
keys to achievement of the overall
health status goals. In 1980, Promoting
Health/Preventing Disease: Objectives
for the Nation set forth 226 targeted
measurable health objectives for the
Nation to achieve over 10 years in 15
priority areas (2). This national agenda
for health proved its merit when, from
1979 to 1990, the infant mortality rate
decreased by nearly 35 percent and the
motor vehicle fatality rate for children
fell 28 percent. Promoting
Health/Preventing Disease: Objectives
for the Nation served as a model for the
development of Healthy People 2000
(3), which subsequently provided the
starting point for Healthy People 2010
(4).
Life-Stage Objectives
The five major life stages theme has
continued through the decades as part of
the Healthy People initiative. Healthy
People 2000 included targets for
reducing deaths among people under age
65, and for reducing the proportion of
people 70 years and over who have
difficulty performing two or more
activities of daily living. Healthy People
2000 contained four age-related
objectives.
Reduce the death rate for children
by 15 percent to no more than 28 per
100,000 children 1–14 years of age, and
for infants by approximately 30 percent
to no more than 7 per 1,000 live births.
Reduce the death rate for
adolescents and young adults by
15 percent to no more than 85 per
100,000 people 15–24 years.
Reduce the death rate for adults by
20 percent to no more than 340 per
100,000 people 25–64 years of age.
Reduce to no more than 90 per
1,000 people the proportion of all
people age 70 years and over who have
difficulty in performing two or more
personal care activities (a reduction of
about 19 percent), thereby preserving
independence.
As shown in table A, data for 1998
indicate that the targets for children and
adolescents and young adults have been
met. The 1998 infant mortality rates of
7.2 per 1,000 live births was very close
to meeting the year 2000 target.
However, the rate for older adults who
had difficulty performing two or more
personal care activities (bathing, eating,
dressing, using the toilet, or getting
in/out of chair/bed) moved away from
the target during the Healthy People
2000 tracking period.
Healthy People 2000
Healthy People 2000: National
Health Promotion and Disease
Prevention Objectives presented the
Nation’s health improvement agenda for
the last decade of the 20th century (3).
Released in 1990, it articulated goals
and objectives aimed at significantly
improving the health of all Americans
by the year 2000.
In developing its own strategy to
improve the health of all Americans,
Healthy People 2000 drew on the
experiences and knowledge gained
during the earlier Healthy People
endeavors. The Healthy People 2000
process was guided by three broad
goals: (1) increase the span of healthy
life, (2) reduce health disparities, and
(3) achieve access to preventive
services. To help meet these overarching
goals, it identified more than 300
national objectives addressing a broad
array of health issues. Forty-seven of
these objectives that were representative
of the scope and magnitude of Healthy
People 2000 were identified as
‘‘sentinel’’ objectives (see Sentinel
Objectives section).
The Healthy People 2000
objectives, each assigned a specific
target, were organized into 22 priority
areas. The activities of each priority area
were coordinated by at least one agency
of the Public Health Service. Addressing
special population groups at high risk of
poor health, Healthy People 2000 set
specific targets to narrow the gap
between the total population and those
groups with higher than average rates of
death, disease, and disability. In
addition, it included priority areas such
as HIV infection and cancer that were
not included in the 1990 objectives.
Underscoring the vital role of
partnerships, Healthy People 2000 was
the product of a cooperative effort
among government agencies, businesses,
nonprofit organizations, and the
scientific community. It was developed
in collaboration with 22 work groups of
experts, agencies of the Federal
government, the National Academy of
Sciences’ Institute of Medicine, and a
consortium of over 375 members
representing national voluntary
organizations and all the State health
departments. Regional and national
meetings provided input from a broad
cross section of citizens, families, and
communities. Moreover, following
extensive public review of and comment
on a draft document, the Healthy People
2000 objectives were revised and
refined.
Midcourse Modifications
Throughout the 1990s, a series of
reports tracked the progress of the
Nation in achieving the Healthy People
2000 objectives. Halfway through the
decade, the national commitment to
disease prevention and health promotion
was reaffirmed during the midcourse
review process, which allowed for the
modification and addition of objectives.
The midcourse review was a 2-year
process, culminating in the publication
of the Healthy People 2000 Midcourse
Review and 1995 Revisions (5) in 1995.
During the midcourse review, work
groups of the Public Health Service met
to consider new data, new information,
and new science that had become
available since the release of Healthy
People 2000 in 1990 (3). As a result, 19
new objectives were added to the
original 300 unduplicated main
objectives, bringing the total number of
objectives to 319. Additional data that
showed increased health risk or disparity
between the total population and people
in age, sex, racial, or ethnic minority
groups resulted in the addition of 111
new special population subobjectives
(bringing the total number of
subobjectives to 319). Including the
Healthy People 2000 Final Review 1

midcourse changes, Healthy People
2000 contained a total of 638 objectives
and subobjectives; because some priority
areas share identical objectives, the
number of objectives and subobjectives
including duplicates is 805. No changes
were made to the three broad goals of
Healthy People 2000 or to the
organization of the 22 priority areas.
The midcourse review also included
58 target revisions (29 objectives and 29
subobjectives), in almost all cases to
make the target more challenging. Text
changes were made to 75 existing
objectives, in some cases considerably
modifying the objective. All midcourse
review modifications are detailed in
Healthy People Statistical Note No. 13
(6).
The midcourse modifications
established baselines for all Healthy
People 2000 objectives for which data
were available. Most of these baselines
are the same as those established in the
original Healthy People 2000 report (3);
others were changed to reflect revisions
to the original baselines or were newly
created.
Healthy People 2010
Healthy People 2010: Objectives for
Improving Health (4), released in
January 2000, carries the Healthy People
initiative into the next decade as well as
into a new millennium. As the third
generation of 10–year goals for the
Nation, it builds on initiatives pursued
over the past two decades. Central to
Healthy People 2010 are its two broad
goals, which challenge the Nation to (1)
increase quality and years of healthy life
and (2) eliminate health disparities.
Providing a framework to achieve
these goals, Healthy People 2010
identifies 467 specific health promotion
and disease prevention objectives in 28
focus areas. Like its predecessors,
Healthy People 2010 is the product of
an extensive cooperative national
process involving both the public and
private sectors, including the Healthy
People Consortium, which by the end of
the 20th century had grown to include
some 350 national organizations and
250 State public health, mental health,
substance abuse, and environmental
agencies.
To facilitate tracking progress
toward the second goal of Healthy
People 2010—eliminating health
disparities—objectives that utilize
population-based measures display the
baseline status of multiple population
groups. The minimum breakout set of
groups includes race and ethnicity,
gender, and measures of socioeconomic
status. However, many objectives
include additional population groups
such as age, geographic location, health
insurance status, disability status, sexual
orientation, or people with a specific
health status or condition. Of the 396
measures in Healthy People 2010 that
listed population groups, baseline data
were available for nearly three quarters
of the over 6,000 data items for the
minimum set of groups (4).
Leading Health Indicators
Setting itself apart from previous
Healthy People efforts, Healthy People
2010 introduces for the first time a set
of 10 Leading Health Indicators (LHIs).
Intended to serve as a gauge of the
Nation’s well-being, the LHIs reflect
major public health priorities in the
United States at the beginning of the
21st century. They were selected
because of their importance as public
health issues, their ability to motivate
action, and the availability of data to
measure their progress. The 10 LHIs are
physical activity, overweight and
obesity, tobacco use, substance abuse,
responsible sexual behavior, mental
health, injury and violence,
environmental quality, immunization,
and access to health care. Each LHI will
be monitored using one or more of the
467 specific objectives, thus serving as a
link to the entire Heathy People 2010
initiative.
Through the LHIs, and through the
467 objectives, Healthy People 2010
continues an impressive tradition of
improving the Nation’s health through a
comprehensive process of setting goals
and measuring results. For more
information on Healthy People 2010,
visit the Web site at
http://www.health.gov/healthypeople.
Summary of Progress
Healthy People 2000 identifies 319
unduplicated main objectives. Because
some priority areas share identical
objectives, certain objectives are
presented in more than one priority area,
which increases the total number of
objectives to 376 including the
duplicates. Subobjectives for racial and
ethnic minorities and other special
populations were established to address
increased health risks or disparities
compared with the total population.
There are 319 subobjectives; with
duplicates, there are 429 (5).
Movement of an objective either
toward or away from the target was
assessed by the direction of the change
between the baseline and the most
recent data point for the measure(s) used
to track the objective. In most cases,
only measures with baselines and targets
were used for assessment; supplemental
data (no baseline or target) were used if
Table A. Life stage objectives: United States, 1987–98
Objectives
Baseline
1987 1991 1992 1993 1994 1995 1996 1997 1998
2000
Target
Infant mortality (per 1,000 live births) .......... 10.1 8.9 8.5 8.4 8.0 7.6 7.3 7.2 7.2 7
Children 1–14 years (total deaths per 100,000) . . . 33.7 30.7 28.8 29.8 28.5 27.8 26.5 25.1 24.0 28
Adolescents and young adults 15–24 years (total
deaths per 100,000) ...................... 97.8 100.1 95.6 98.5 98.0 95.3 89.6 86.2 82.3 85
Adults 25–64 years (total deaths per 100,000) . . . 426.9 400.7 394.7 400.1 398.6 397.3 382.0 368.1 364.0 340
People 70 years and over (difficulty in performing
two or more personal care activities per 1,000) . .
1
141 --- --- --- ---
2
163 --- --- --- 90
- - - Data not available.
1
1984–85 data.
2
1994–95 data
SOURCES: National Vital Statistics System, CDC, NCHS.
For people 65 years and over: National Health Interview Survey, CDC, NCHS; National Nursing Home Survey, CDC, NCHS.
2 Healthy People 2000 Final Review

measures with baseline and targets were
not available. Some of the changes
observed were relatively small and may
be within what could be expected on the
basis of sampling or random variation.
For objectives with more than one
measure (compound objectives), if data
showed movement in different
directions, progress was labeled as
‘‘mixed.’’ For compound objectives with
data not available for all measures,
progress was determined by the
direction of the measure(s) with data.
For example, objective 12.3 is tracked
using three measures. However,
sufficient tracking data are only
available for two of the three measures:
Refrigeration of perishable foods and
washing cutting boards with soap.
Progress for this objective is assessed
using these two measures. All measures
of a compound objective had to be met
for an objective to be considered met;
for example, a compound objective with
three measures meeting the target and
one measure progressing toward the
target would be considered moving
toward the target (objective 1.10). A few
objectives were very broad in scope and
tracking data were not available; in
these cases, the subobjectives were used
to track progress (for example, objective
17.14).
The following summary of progress,
based on the 319 unduplicated main
objectives, presents the final status for
the Healthy People 2000 objectives. The
most recent data indicate that 68
objectives (21 percent) met the year
2000 targets and an additional 129
(41 percent) showed movement toward
the targets. Data for 35 objectives
(11 percent) showed mixed results and 7
(2 percent) showed no change from the
baseline. Only 47 objectives (15 percent)
showed movement away from the
targets. The status of 32 objectives
(10 percent) could not be assessed.
Among these unassessed objectives, 23
have baseline data but no additional data
with which to evaluate progress (several
objectives in this category have
supplemental data that cannot be used
for determination of progress), and 9
objectives lacked baselines (see Priority
Area 22). Figure A shows the progress
of the objectives by priority area.
Progress for each individual objective is
shown in the priority area summary
tables (tables 1–22).
A number of Healthy People 2000
targets were revised during the
midcourse review process, in almost all
cases to make the target more
challenging (see section on Midcourse
Modifications). As a result, eight
objectives and nine subobjectives that
did not meet the targets in the final
assessment would have met their targets
if the original targets had not been
revised during the midcourse review.
0
1234567891011
Healthy People 2000
Priority Areas
12 13 14 15 16 17 18 19 20 21 22
5
10
15
20
25
35
30
Number of objectives
Cannot assess
Moved away from target
No change
Mixed progress
Moved toward target
Met
Healthy People 2000 Final Review 3
Figure A. Healthy People 2000 objectives: Summary of progress by
priority area
Area Met
Moved
toward
target
Mixed
progress
No
change
Moved
away
from
target
Cannot
assess
1............ 1 6 0 1 4 1
2............ 6 13 2 0 6 0
3............ 7 10 5 1 1 2
4............ 3 7 4 0 2 4
5............ 0 8 3 0 0 1
6............ 5 1 1 0 7 1
7............ 7 4 0 0 5 3
8............ 5 2 2 1 1 3
9............ 7 11 2 2 2 2
10........... 7 7 1 1 4 0
11........... 4 9 2 0 1 1
12........... 2 5 0 0 1 0
13........... 1 12 1 1 2 0
14........... 1 8 2 1 3 2
15........... 3 12 0 0 2 0
16........... 7 8 2 0 0 0
17........... 2 6 3 0 11 1
18........... 5 5 1 0 3 3
19........... 4 7 2 0 1 3
20........... 3 7 5 0 2 2
21........... 1 2 2 0 1 2
22........... 2 4 1 0 0 0
Total ......... 83 154 41 8 59 31
Organization and Scope of this
Review
This Review is organized into three
major sections and presents the final
data collected for the 376 objectives that
comprise Healthy People 2000 (3).
While the data for these individual
objectives are the basis for this Review,
the additional analyses presented
provide further insight into the Nation’s
progress in health improvement over the
last decade.
The Introduction provides a brief
discussion of the history of the Healthy
People initiative, the current effort, and
the transition to the next decade in
Healthy People 2010 (4). There is also a
summary of progress for the past decade
and a description of the organization
and scope of this Review.
The first section presents
information that cuts across the 22
Healthy People 2000 priority areas. The
first part of this section summarizes
progress regarding the three broad goals
set in Healthy People 2000: Goal
1—Increase the span of healthy life,
Goal 2—Reduce health disparities, and
Goal 3—Achieve access to preventive
services. The second part of this section
presents data for three summary lists:
the Health Status Indicators (a set of
indicators of community health status
relevant to public health practice); the
Priority Data Needs (important
indicators for evaluating the health of a
population but not necessarily available
at all levels of government); and the
Sentinel Objectives (objectives
representative of the scope and
magnitude of the changes envisioned by
Healthy People 2000). Each of these
parts provide information through text,
charts, and tables describing progress
over the last decade by focusing on
particular issues that generally address
the 22 priority areas.
The second section consists of 22
chapters providing a progress report for
each of the Healthy People 2000
priority areas. Each chapter contains a
progress quotient chart, presenting a
summary of progress for each objective;
a discussion of the objectives that
addresses the area’s public health
significance, data highlights, progress
for the objectives, major results,
transition to Healthy People 2010, and
data issues; a summary data table; and
the full text of the objectives.
Technical notes and tables comprise
the Appendix, the third section. The
technical notes present and discuss data
and analysis issues involved in
monitoring the objectives and
subobjectives. The appendix provides
additional information that may be
useful in understanding and interpreting
the data. Additional sources of
information on Healthy People 2000 are
also presented.
This report is available on the
Internet at http://www.cdc.gov/nchs/.
References
1. Department of Health, Education, and Welfare.
Healthy People: The Surgeon General’s Report on
Health Promotion and Disease Prevention. Public
Health Service. U.S. Government Printing Office.
Washington. 1979.
2. Department of Health and Human Services.
Promoting health/preventing disease: Objectives
for the nation. Washington: Public Health Service.
1980.
3. Department of Health and Human Services.
Healthy people: National health promotion and
disease prevention objectives. Washington: Public
Health Service. 1991.
4. Department of Health and Human Services.
Healthy People 2010. 2d ed. With understanding
and improving health and objectives for improving
health. 2 vols. Washington: U.S. Government
Printing Office. 2000.
5. Department of Health and Human Services.
Healthy People 2000 midcourse review and 1995
revisions. Washington: Public Health Service.
1995.
6. Turczyn KM and Ryan CM. Healthy People
2000 midcourse revisions: A compendium. Healthy
people statistical notes; no 13. Hyattsville,
Maryland: National Center for Health Statistics.
1997.
4 Healthy People 2000 Final Review

Healthy People 2000 Goals

Healthy People 2000
Goals
The purpose of Healthy People
2000 was to commit the Nation to the
attainment of three broad goals. These
goals were to increase the span of
healthy life, to reduce health disparities
among Americans, and to achieve access
to preventive services for all Americans.
Attaining these broad goals would bring
the Nation’s health to its full potential.
Goal 1: Increase the Span of
Healthy Life
In the 20th century, the population
of the United States underwent a
continuous period of health
improvement that is unparalleled in
history. At the turn of the century, the
population was characterized by a
mostly low standard of living, poor
hygiene, poor nutrition, and a high
prevalence of communicable and acute
diseases typical of a population with
high mortality and high fertility. It
finished the century with a mostly high
standard of living, good hygiene, good
nutrition, and health-related habits
typical of populations with low
mortality and low fertility. In addition,
there was a low frequency of
communicable diseases and acute
conditions that were major causes of
most of the premature deaths in the
earlier part of the century. As a
consequence of the dramatic fall in
mortality, the average expectation of life
at birth for the total population
increased from 47.3 years in 1900 to
76.8 years in 1998 (1), an increase of
62 percent or 3.6 months per year for
each of the 98 years.
The same population and health
transitions that have jointly propelled
the average life expectancy by nearly 30
years, however, are also responsible for
ushering an era of new challenges. The
population is aging at an unprecedented
rate, resulting in an increased prevalence
of diseases that are noncommunicable
and not always fatal, but degenerative
and chronic. This adds a new dimension
to measuring the health of the
population. Expectation of life, which is
a traditional measure of longevity, has
now effectively become a ‘‘partial’’
measure of health for an aging
population that expects to live a long
life with degenerative diseases and
chronic conditions.
For an aging population fast
approaching the theoretical maximum
life span, the fundamental issues involve
not only longevity, but quality of life as
well. Quality of life (QOL) is a
subjective appraisal of life (2), relating
to the ‘‘goodness’’ of life as assessed by
external conditions (3). It is a
multidimensional concept that can, in
the broadest sense, include material
comfort, work, health, and active
recreation (4). The World Health
Organization’s concept of QOL on the
other hand, is based on the assessment
of the six broad domains that are related
to physical, psychological,
environmental, spiritual, level of
independence, and social relationship
(5). Some of these domains are directly
health related and others are not.
Healthy People 2000 is primarily
concerned with health-related QOL (6).
The formulation of the concept of
health-related QOL began in the 1970s
with a limited focus on domains of
physical function for measuring
activities such as activities of daily
living (5). Since then, the concept has
been expanded to include other domains,
such as general health perception,
limitations in usual roles, activity
restrictions, symptoms, and cognitive
psychological function (7).
The first goal of Healthy People
2000, to increase the span of healthy
life, is monitored using three statistics.
Life expectancy at birth (a measure of
longevity), the percent of people
reporting that their general health status
is fair or poor (a measure of
health-related QOL), and expected years
of healthy and unhealthy life (a
summary measure incorporating both
mortality and morbidity, known as years
of healthy life or YHL).
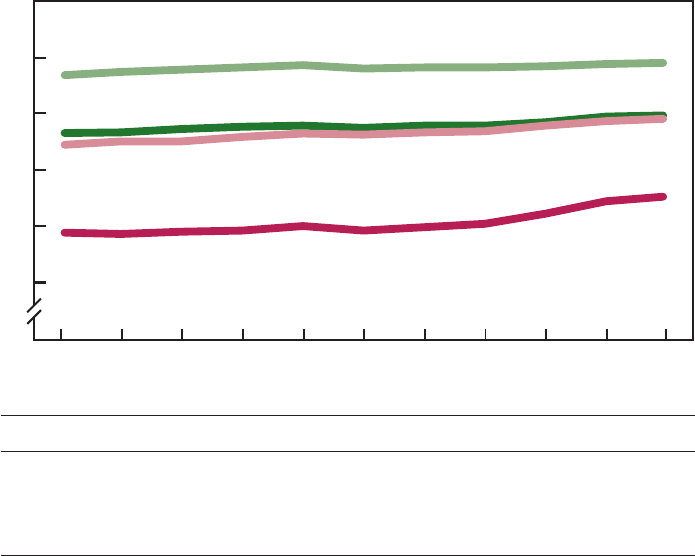
Life Expectancy at Birth
The expectation of life at birth is
the number of years a newborn would
expect to live if that person experienced
the mortality schedule existing at the
time of birth. This measure is one of the
most commonly used measures to
summarize mortality and study trends in
the span of human life. Figure B
presents the life expectancy at birth for
the U.S. population by sex and race for
the decade monitored by Healthy People
2000. Between 1988 and 1998 the
expectation of life increased from 74.9
to 76.7 years, an increase of nearly 2
years (2.4 percent). Life expectancy has
increased for all four population
subgroups shown. The biggest gain,
however, was for black males (3.2 years
or 5.0 percent), the population subgroup
with the lowest life expectancy.
Consequently, the difference between
the highest (white females) and lowest
(black males) life expectancy declined
over the decade from 10.5 years to 9.1
years, an indication that the disparity in
expectation of life among the subgroups
decreased in the 10-year period.
Fair or Poor Health
Between 1990 and 1996, the
percent of people reporting fair or poor
health increased from 9.9 to 10.4 percent
(figure C). Beginning in 1997, there was
a change in the methodology used to
measure this statistic in the National
Health Interview Survey so that data
collected in 1997 and subsequent years
are not directly comparable to those for
1996 and prior years. Between 1997 and
1998, the percent decreased slightly,
from 9.2 to 9.1 percent. Figure C also
displays the percent of people reporting
fair or poor health by socioeconomic
status (SES): poor (family income less
than poverty level), near poor (100 to
199 percent of poverty level), and
middle/high income (at least 200 percent
of poverty level). There is a clear
association between poor health and
poverty. In 1990, 22 percent of poor
people reported fair or poor health, three
times that reported by middle or high
income people (7 percent). In 1998, the
difference was slightly greater
(22 percent compared with 6 percent).
During the 1997–98 period, the trend in
the percent of people reporting fair or
poor health increased for the poor
population but decreased for the middle
or high-income population, widening the
disparity.
Healthy and Unhealthy Life
The broad purpose of the 319
Healthy People 2000 objectives was to
improve the health of the population.
Consequently, all objectives, at least
indirectly, involve increasing the span of
healthy life. However, one specific
objective (8.1, also duplicated as 17.1
and 21.1), targets a single measure of
the span of healthy life. The objective
measures health-related quality of life
using the summary measure of health
known as YHL. This measure estimates
the average number of years expected to
Healthy People 2000 Final Review 7
be lived in a healthy state for people of
a specified population. As a summary
measure of population health, this
measure incorporates mortality and
morbidity into a single statistic ( 8,9).
The mortality component utilizes total
mortality, while the morbidity
component is evaluated using two
measures: respondent-assessed health
and activity and functional limitations
due to chronic conditions. Individuals
are classified into 30 groups according
to both measures and these groups are
then assigned a quality-of-life score
(based on a mathematical model)
indicating the quality-of-life for that
group relative to a healthy state (10).
The YHL is calculated using a life table
approach that incorporates the
age-specific mortality with the
age-specific health related quality-
of-life score. The resulting ‘‘average
years of healthy life’’ is equivalent to
the average years a person would live if
he or she lived in a healthy state.
In 1990 the YHL was 64.0 years.
By 1998, this increased by 1.2 years to
65.2 years— a rise that was comparable
to the increase in expected years of life
(1.3 years between 1990 and 1998).
Consequently, average YHL as a percent
of total expected years of life was
largely unchanged. Figure D presents the
expected years of healthy life and
unhealthy life of the U.S. population by
race and ethnicity in 1998. The Hispanic
population has the greatest YHL.
However, Hispanics also have the
smallest proportion of life spent
equivalently in healthy states, that is
66.3 years of the total 82.1 expected life
years (81 percent). Blacks have the
smallest YHL and also a small
proportion of life spent in healthy states
(81 percent). Among whites, 86 percent
of the expected life years are
equivalently spent in healthy states.
Goal 2: Reducing Health
Disparities
The population of the United States
grew by 13 percent over the last decade,
and has increased in diversity at an even
greater rate (11). The aging of our
population is well documented and the
trend will continue for at least several
decades. Racial and ethnic minority
populations are among the fastest
growing of all communities in the
United States. Yet, these populations in
many respects, have poorer health and
remain chronically underserved by the
health care system. In many cases, the
health gaps initially identified in the
1985 Task Force Report on Black and
Minority Health (12) have grown.
Differences in accessibility, utilization,
quality of care, or benefits derived from
medical care are factors contributing to
inequality (13).
Healthy People 2000 included over
200 special population subobjectives to
address the overarching goal of reducing
health disparities in special populations
at higher risk than the total population
for death, disease, or disability (14).
Targets for these subobjectives were set
requiring a greater percent change with
the aim of reducing the gap between
these special populations and the total
population. A few subobjectives were
also included where, although the
special population was not at increased
risk, there was a disparate trend (for
example, 16.2b, lung cancer for
women). The populations highlighted in
Healthy People 2000 included American
Indian/Alaska Native, Asian or Pacific
Islander, black, Hispanic, women,
adolescents and young adults, older
adults, people with disabilities, and
people with low SES.
The Healthy People 2000
Midcourse Review and 1995 Revisions
resulted in the addition of many
subobjectives for these special
populations for a total of 49 objectives
or subobjectives for American
Indian/Alaska Native, 19 for Asian or
Pacific Islander, 97 for black, 84 for
Hispanic, 187 for women, 243 for
adolescents, 173 for older adults, 89 for
people with disabilities, and 65 for
people with low SES.
The objectives used for the
assessment of disparities include the
Healthy People 2000 subobjectives for
each of the nine special population
groups as well as other objectives
related to these groups. A list of the
objectives for each group can be found
in Appendix table VII. Some objectives
have measures for more than one group.
Progress toward the target can be
measured for all of these objectives;
however, not all the objectives can be
used to measure disparity. For groups
other than the racial/ethnic groups, a
0
1988 19901989 1991 1992 1993 1994 1995 1996 1997 1998
60
65
70
75
80
85
Years
White female
Black female
White male
Black male
1988 1989 1990 1991 1992 1993 1994 1995 1996 1997 1998
White female . 78.9 79.2 79.4 79.6 79.8 79.5 79.6 79.6 79.7 79.9 80.0
Black female. . 73.2 73.3 73.6 73.8 73.9 73.7 73.9 73.9 74.2 74.7 74.8
White male . . . 72.2 72.5 72.7 72.9 73.2 73.1 73.3 73.4 73.9 74.3 74.5
Black male . . . 64.4 64.3 64.5 64.6 65.0 64.6 64.9 65.2 66.1 67.2 67.6
SOURCE: CDC/NCHS, National Vital Statistics System.
8 Healthy People 2000 Final Review
Figure B. Life expectancy at birth: United States, 1988–98

relatively small proportion of the
objectives are applicable for assessment
of disparity. This is because, for these
groups, most objectives target the group
directly and do not also target a
reference population that could be used
to assess disparity status. Disparity was
assessed by comparing the percent
change between the baseline and last
data point for the special population
subobjective with the percent change
observed for the total population used to
track the objective. (See section on
Disparity Measurement in the
Appendix.)
The 1990s saw significant
improvements in the ability to track
progress for Healthy People 2000
objectives by special populations.
Leading into the decade, in 1989,
Hispanic origin was included for the
first time on standard birth and death
certificates recommended for use by all
States. By the end of the decade, the
number of States that publish vital
statistics data by race and ethnicity
increased from 19 to 27—surpassing the
Healthy People 2000 target. Despite
these improvements, data were still
missing at the end of the decade to
assess progress for 6 percent of the
objectives for American Indian/Alaska
Native, 5 percent for Asian or Pacific
Islander, 2 percent for black, 8 percent
for Hispanic, 10 percent for women,
26 percent for adolescents, 10 percent
for older adults, 7 percent for people
with disabilities, and 6 percent for
people with low SES.
At the conclusion of the decade,
more than one-half of the special
population subobjectives showed a
narrowing or elimination of disparities.
In addition, progress for special
populations was made in a number of
areas. However, only 16 percent of the
year 2000 targets for the special
population subobjectives were met
compared with 22 percent of all 318
unduplicated objectives and
subobjectives.
One of the two overarching goals
for Healthy People 2010, launched in
January 2000, extends this goal from
reducing disparities to actually
eliminating health disparities. As a
result, Healthy People 2010 has the
same target for all population groups,
including racial and ethnic groups. As
noted during the public comment period
for Healthy People 2010, ‘‘the ambitious
goal of eliminating health disparities
demands broader and more
interdisciplinary remedies than those
previously considered.’’
The following sections discuss
progress toward reducing disparities and
progress toward the year 2000 targets
for each of the nine special population
groups addressed by Healthy People
2000. A summary of progress is
graphically shown for each group along
with text discussion of some specific
examples. The specific examples are
shown for illustrative purposes only, and
do not infer priority or importance. A
complete assessment of progress toward
the target and progress toward reducing
disparity for the objectives in each of
the nine special population groups is
shown in Appendix table VII.
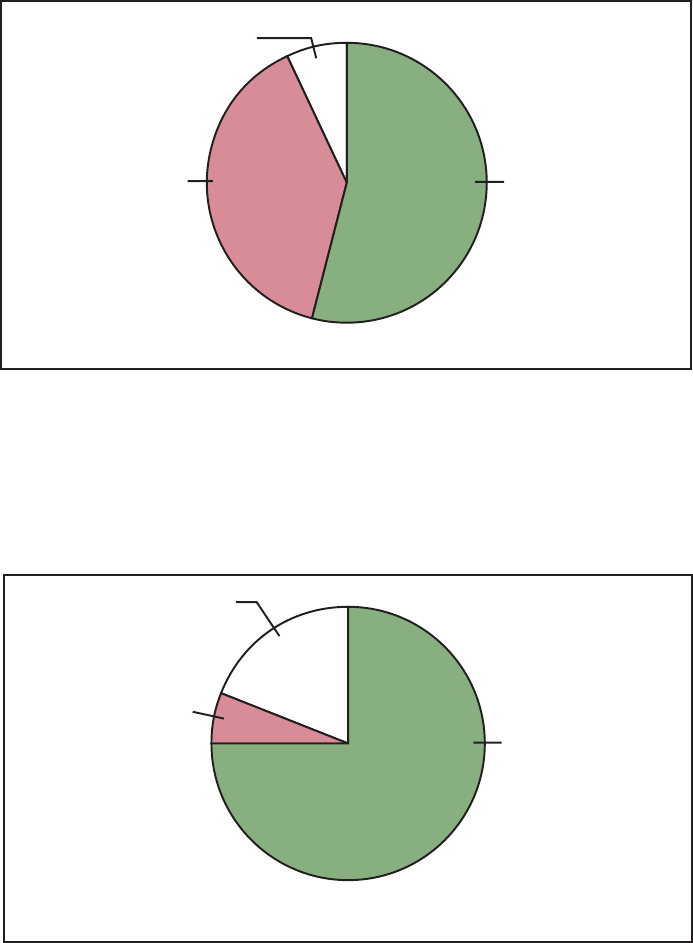
Reducing Disparities: American
Indian/Alaska Native
Figure E demonstrates progress
made in reducing disparities in the
health status of American Indians/Alaska
Natives. Twenty-four (55 percent) of the
forty-four American Indian/Alaska
Native subobjectives showed a reduction
or elimination of disparities in health
status between this population and the
total population. Of these 24 objectives,
20 met or moved toward their year 2000
targets.
Hepatitis A (20.3j) and bacterial
meningitis (20.7a) showed remarkable
improvement— surpassing year 2000
targets and narrowing the gap. Diabetes
prevalence for American Indians and
Alaska Natives, however, increased,
although the gap narrowed (17.11a)
because the prevalence for the total
population increased at an even greater
rate. Diabetes-related deaths (17.9b) and
end-stage renal disease rates (17.10b)
increased and the gaps widened.
0
1990 1991 1992 1993 1994 1995 1996 1997 1998
5
10
15
20
25
30
Below poverty level
100-199% of poverty level
200% or more of poverty level
Percent
1990 1991 1992 1993 1994 1995 1996 1997 1998
Below poverty
level ........ 22.1% 22.8% 23.8% 24.2% 23.7% 23.7% 23.8% 21.4% 22.2%
100–199% of
poverty level . . 14.5% 14.7% 15.0% 15.7% 15.0% 15.5% 15.2% 14.6% 15.6%
200% or more of
poverty level . . 6.8% 6.8% 7.0% 6.8% 6.9% 6.7% 6.6% 6.1% 5.7%
SOURCE: CDC/NCHS, National Health Interview Survey.
NOTE: The survey was redesigned in 1997. Data for 1997 and 1998 may not be directly
comparable with those for previous years.
Healthy People 2000 Final Review 9
Figure C. Percent of people with fair or poor health:
United States, 1990–98

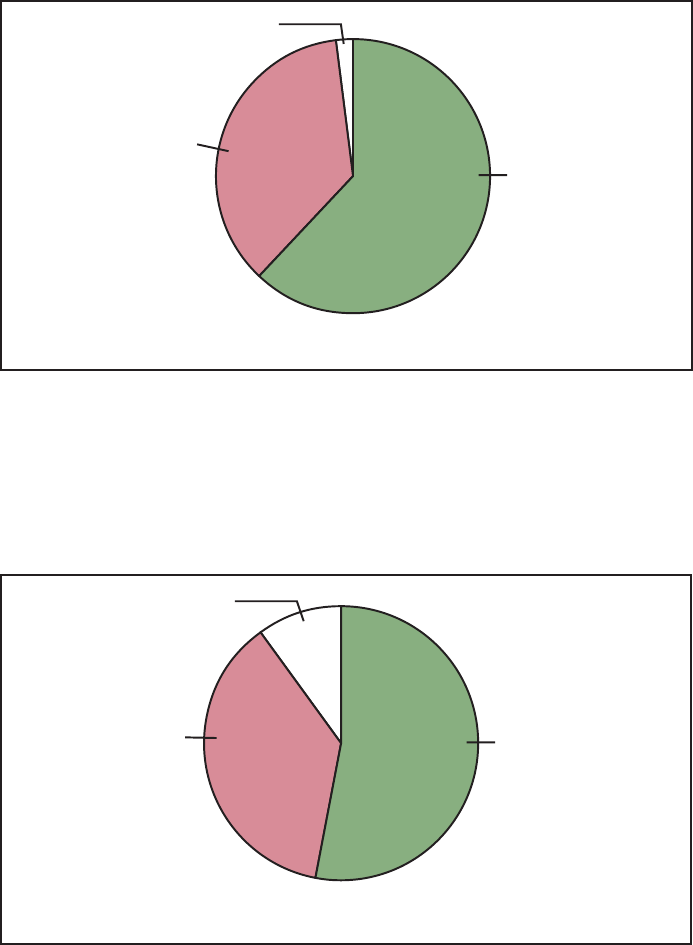
Reducing Disparities: Asian or
Pacific Islander
Figure F represents the disparity
status of the subobjectives for Asians or
Pacific Islanders. Twelve of the sixteen
subobjectives for this group showed a
reduction or elimination of disparities,
and all 12 subobjectives either met or
moved toward their targets.
For example, the year 2000 target
was met and disparity decreased for flu
vaccinations among the elderly (21.2).
Cigarette smoking decreased for
Southeast Asian males at the beginning
of the decade and the disparity narrowed
(3.4g). The number of Hepatitis B cases
among Asian or Pacific Islander children
dramatically decreased (20.3d) although
disparity was not assessed for this
objective because the measure for the
subobjective was different from that for
the main objective. One objective, new
tuberculosis cases (20.4a), showed a
widening of disparity.
Reducing Disparities: Black or
African American
Figure G depicts the status of
subobjectives to reduce disparities
between black Americans and the total
population. Elimination or reduction of
disparities occurred for 55 (62 percent)
of the 89 subobjectives. Of these 55
subobjectives, 50 either met or moved
toward their targets.
Objectives for which targets were
met and disparities were reduced
included cancer deaths (2.2a),
firearm-related deaths (7.3a),
unintentional injuries (9.2a), clinical
breast examination and mammography
(16.11e), and syphilis (19.3a).
Unfortunately, diabetes prevalence
(17.11e), diabetes-related deaths (17.9a),
lower extremity amputations (17.10c),
maternal mortality (14.3a) and fetal
alcohol syndrome (14.4b) increased and
the disparity gaps widened.
Reducing Disparities: Hispanic or
Latino
Figure H shows the status of the 76
subobjectives for Hispanic persons.
Forty (52 percent) of these subobjectives
showed a reduction in disparity between
Hispanics and the total population. It is
interesting to note that 10 of these 40
subobjectives moved away from their
targets but at a slower rate than the
objectives for total population.
Objectives for which targets were
met and disparities were reduced
included infant mortality for Puerto
Ricans (14.1c), use of clinical breast
examination and mammography among
Hispanic women over 50 years of age
(16.11c), and rates of congenital syphilis
(19.4b). Adolescent pregnancy (5.1b)
and high school completion rates (8.2a),
however, remained essentially the same.
Reducing Disparities: Women
Figure J depicts progress made in
reducing health disparities for women in
the United States. Of the 19 objectives
for which disparities between women
and the total population can be assessed,
9 objectives showed a reduction or
elimination of health status inequity. Of
these nine objectives, six either met or
moved toward their targets.
Progress was made in smoking
prevalence (3.4) and homicide rates
among African American women (7.1e).
Unfortunately, although new cases of
AIDS decreased in recent years, the rate
of decrease for women (18.1d) was less
than that for the total population.
Reducing Disparities: Adolescents
and Young Adults
The pie chart depicted in figure K
reflects changes in disparities between
adolescents and young adults and the
total population. Disparities in seven
subobjectives decreased or were
eliminated, while five widened. All
seven of the subobjectives that reduced
or eliminated the disparity either met or
moved toward their targets.
Motor vehicle crash death rates for
people 15–24 years (9.3b) and
drownings for males 15–34 years
decreased substantially and the
disparities gaps narrowed. However,
there were increases in disparities for
rape or attempted rape (7.7a) and
adolescent gonorrhea (19.1b).
0
15
30
45
60
75
90
Expected years of life
* Hispanic origin can be of any race.
Healthy
Total White Black Hispanic*
Unhealthy
Total White Black Hispanic*
Healthy life expectancy ........ 65.2 66.1 57.8 66.3
Unhealthy life expectancy ...... 11.5 11.2 13.5 15.8
Total life expectancy .......... 76.7 77.3 71.3 82.1
*Data are preliminary. Hispanic includes people of any race with Hispanic origin.
SOURCE: CDC/NCHS, National Vital Statistics System and National Health Interview Survey.
10 Healthy People 2000 Final Review
Figure D. Expected years of healthy and unhealthy life:
United States, 1998
Reducing Disparities: Older Adults
Figure L depicts progress on
subobjectives used to measure disparity
between older adults and the total
population. Eleven of the nineteen
subobjectives measured showed a
reduction or elimination in disparity, and
all of these either met or moved toward
their targets.
There was nearly a 20–percent
reduction in the rate of white male
suicide in the elderly population (6.1c),
narrowing the gap with the total
population. This reduction is reflected in
the increase in years of healthy life for
adults 65 years and over (8.1c). There
were, however, increases in the
disparities for death rates due to falls
(9.4a,b) and motor vehicle crashes
(9.3c).
Reducing Disparities: People with
Disabilities
Figure M shows progress made on
the six subobjectives among people with
disabilities for which disparity could be
measured. The graph indicates that
disparities have been reduced or
eliminated for one-half of the
subobjectives for people with
disabilities. One of these subobjectives
moved away from its target, although at
a slower rate than the total population,
thus reducing the disparity.
Data indicate that progress is being
made in reducing the percent of
overweight persons in the disabled
population (1.2e). In addition, disparities
in sedentary lifestyle between the
disabled and the total population are
being reduced (1.5b).
Reducing Disparities: People with
Low Socioeconomic Status
The chart shown in figure N
demonstrates the disparity status of the
36 subobjectives used to measure
disparities between people with low
socioeconomic status (SES) and the total
population. While nearly one-third of
the subobjectives showed a widening or
no change in disparity, 21 (58 percent)
improved. But of these 21 subobjectives,
3 moved away from their targets,
indicating that the total population
moved away from its target at a faster
rate than those with low SES.
Encouraging improvements were
seen in the areas of breast feeding
(2.11a) and the use of contraception
(5.12b,c). Conversely, there were
increases in the percent of overweight
persons (1.2a) and certain oral health
issues (for example, prevalence of
gingivitis, 13.5a).
Goal 3: Achieving Access to
Preventive Services
Access to health services—
including preventive care, primary care,
and tertiary care—often depends on two
independent factors: having health
insurance and having a regular source of
health care (15–18). Uninsured people
are less than half as likely as people
with health insurance to have a primary
care provider; to have received
appropriate preventive care, such as
recent mammograms or Pap tests; or to
have had any recent medical visits. Lack
of insurance also affects access to care
Disparity:
Eliminated/Narrowing
54% (N=24)
Cannot assess
7% (N=3)
Disparity:
Widening/No change
39% (N=17)
Number of objectives = 44
NOTE: Disparity is assessed by comparing the change for the special population with the change for
the total population. Not all objectives for the special population are applicable to disparity assessment.
(See Disparity Assessment section in the Appendix.)
Disparity:
Eliminated/Narrowing
75% (N=12)
Cannot assess
19% (N=3)
Disparity:
Widening/No change
6% (N=1)
Number of objectives = 16
NOTE: Disparity is assessed by comparing the change for the special population with the change for
the total population. Not all objectives for the special population are applicable to disparity assessment.
(See Disparity Assessment section in the Appendix.)
Healthy People 2000 Final Review 11
Figure E. Assessment of disparity for American Indian/Alaska
Native objectives
Figure F. Assessment of disparity for Asian or Pacific Islander
objectives
for relatively serious medical conditions.
Evidence suggests that lack of insurance
over an extended period significantly
increases the risk of premature death
and that death rates among hospitalized
patients without health insurance are
significantly higher than those with
insurance (19). Another study showed
that, among those without insurance,
chronically ill persons are even less
likely than those with acute conditions
to get health care services they need
(20). Beyond having health insurance,
access depends on the actual benefits
offered by the insurance plan. Many
plans do not offer adequate benefits for
some services such as mental health, eye
care, oral health, assistive devices, and
drugs. Medicaid expansions that increase
the proportion of a State’s population
eligible for Medicaid lead to increases
in enrollment, enhanced utilization of
medical services, and lower child death
rates (21).
More than 40 million persons in the
United States do not have a particular
doctor’s office, clinic, health center, or
other place where they usually go to
seek health care or health-related advice.
Even among privately insured persons, a
significant number lacked a usual source
of care or reported difficulty in
accessing needed care due to financial
constraints or insurance problems (15).
Barriers to obtaining health care can be
financial, structural, or personal.
Financial barriers include not having
health insurance, not having enough
health insurance to cover needed
services, or not having the financial
capacity to cover services outside a
health plan or insurance program.
Structural barriers include the lack of
primary care providers, medical
specialists, or other allied health care
professionals (including personal
assistants and caregivers) to meet
special needs; the lack of health care
facilities or exam equipment; or the lack
of transportation. Personal barriers
include cultural or spiritual differences,
language barriers (including sign
language), not knowing what to do or
when to seek care, or mistrust or
concerns about confidentiality or
discrimination.
The 1990s experienced a steady
increase in the delivery of certain
effective clinical preventive services,
including child and adult immunizations,
screening for cancer and risk factors for
heart disease, and counseling of smokers
to quit smoking. For the remaining
individual services, a variety of barriers
may have prevented most services from
achieving the Healthy People 2000
targets for delivery to the general
population. For most services, the
lowest delivery rates were observed
among specific racial and ethnic groups
and people with low income or less
education.
Two factors are consistently
associated with a greater likelihood of
receiving recommended preventive care:
health insurance and a usual source of
health care. Costs of preventive care can
be an important barrier, especially for
persons who may not perceive the
benefits of health care when they are
not sick. The substantial increase in the
coverage of preventive services by
insurers and health plans over the past
decade has reduced the costs of
prevention to individuals with insurance,
although deductibles and co-payments
may still deter some patients. Having a
Disparity:
Eliminated/Narrowing
62% (N=55)
Cannot assess
2% (N=2)
Disparity:
Widening/No change
36% (N=32)
Number of objectives = 89
NOTE: Disparity is assessed by comparing the change for the special population with the change for
the total population. Not all objectives for the special population are applicable to disparity assessment.
(See Disparity Assessment section in the Appendix.)
Disparity:
Eliminated/Narrowing
53% (N=40)
Cannot assess
10% (N=8)
Disparity:
Widening/No change
37% (N=28)
Number of objectives = 76
NOTE: Disparity is assessed by comparing the change for the special population with the change for
the total population. Not all objectives for the special population are applicable to disparity assessment.
(See Disparity Assessment section in the Appendix.)
12 Healthy People 2000 Final Review
Figure G. Assessment of disparity for black or African American
objectives
Figure H. Assessment of disparity for Hispanic or Latino
objectives

usual source of care, however, may be
as important as having health insurance.
Continuity of care makes it more likely
that persons in need of specific
preventive services will be identified,
that clinician recommendations will be
followed, and that patients will return
for services that need to be delivered
periodically.
Access to care, as measured by
having a usual source of health care
(21.3), increased from 78 percent in
1992 to 85 percent by 1998 for the
general population, although Hispanics,
American Indians/Alaska Natives, and
people below the poverty level lagged
behind (72–77 percent). First trimester
prenatal care also increased gradually,
but much more dramatic gains were
seen in the delivery of child
immunizations, where the proportion of
children who are fully immunized
climbed from 55 to 80 percent
(figure O).
Little progress, however, was
evident in reducing the number of
persons under 65 years without health
insurance (21.4)(figure P). The
population over 65 years in the United
States is assumed to be covered by
Medicare. Although rates of uninsured
people declined slightly from the middle
of the decade, they were higher for the
general population in 1998
(16.6 percent) than in 1989
(15.7 percent). Due to Medicaid
coverage of poor families and the State
Children’s Health Insurance Program
(SCHIP), children were less likely to be
uninsured than adults. Despite the
successes of these programs a
substantial proportion (12.7 percent)
continue to lack health insurance
coverage. Lack of health insurance is
most common in young adults, who are
less likely to have employer-based
insurance and more likely to decline
voluntary coverage.
References
1. National Center for Health Statistics. Health,
United States, 2000 With adolescent health
chartbook. Hyattsville, Maryland. 2000.
2. Kaplan RM, Andersen JP. The general health
policy model: An integral approach. In Spikler B
(ed). Quality of life assessments in clinical trials.
New York: Raven Press, Ltd. 1990.
3. Zautra A, Goodhart D. Quality of life
indicators: A review of the literature. Community
Ment Health Rev 4(1). 1979.
4. Freed MM. Quality of life: The physician’s
dilemma. Arch of Phys Med and Rehab
65:109–11.1984.
5. Murray CJL, Lopez AD. The global burden of
disease. World Health Organization: Harvard
University Press. 1996.
6. Torrance G. Utility approach to measuring
health-related quality of life. J Clin Dis
6:593–603. 1987.
7. Patrick DL, Erickson P. Health status and health
policy: Quality of life in health care evaluation
and resource allocation. New York: Oxford
University Press. 1993.
8. Fryback DG. Methodological issues in
measuring health status and health-related quality
of life for population health measures: A brief
overview of the ‘‘HALY’’ family of measures.
Washington: National Academy Press. 1998.
9. Institute of Medicine. Summarizing population
health: Directions for the development and
application of population metrics. Washington:
National Academy Press. 1998.
10. Erickson P, Wilson R, Shannon I. Years of
healthy life. Healthy people statistical notes; no 7.
Disparity:
Eliminated/Narrowing
47% (N=9)
Cannot assess
11% (N=2)
Disparity:
Widening/No change
42% (N=8)
Number of objectives = 19
NOTE: Disparity is assessed by comparing the change for the special population with the change for
the total population. Not all objectives for the special population are applicable to disparity assessment.
(See Disparity Assessment section in the Appendix.)
Disparity:
Eliminated/Narrowing
44% (N=7)
Cannot assess
25% (N=4)
Disparity:
Widening/No change
31% (N=5)
Number of objectives = 16
NOTE: Disparity is assessed by comparing the change for the special population with the change for
the total population. Not all objectives for the special population are applicable to disparity assessment.
(See Disparity Assessment section in the Appendix.)
Healthy People 2000 Final Review 13
Figure J. Assessment of disparity for women objectives
Figure K. Assessment of disparity for adolescents and young
adults objectives

Hyattsville, Maryland: National Center for Health
Statistics. 1995.
11. Census Bureau. Resident population of the 50
States, the District of Columbia, and Puerto Rico:
April 1, 2000 (Census 2000) and April 1, 1990
(1990 Census) and State Rank as of 2000 and
State Rank as of 1990.
http://www.census.gov/population/www/cen2000/
respop.html. 2001.
12. Department of Health and Human Services.
Report of the Secretary’s Task Force on Black and
Minority Health. Public Health Service. 1985.
13. National Center for Health Statistics. Health,
United States, 1998 with socioeconomic status and
health chartbook. Hyattsville, Maryland. 1998.
14. Department of Health and Human Services.
Healthy people 2000: National health promotion
and disease prevention objectives. Washington:
Public Health Service. 1991.
15. Department of Health and Human Services.
Healthy people 2010. 2d ed. With understanding
and improving health and objectives for improving
health. 2 vols. Washington: U.S. Government
Printing Office. November 2000.
16. CDC. Health insurance coverage and receipt of
preventive health services—United States, 1993.
MMWR 44:219–25. 1995.
17. Weissman JS, Epstein AM. The insurance gap:
Does it make a difference? Annual Review of
Public Health 14:243–70. 1993.
18. General Accounting Office. Health insurance:
Coverage leads to increased health care access for
children. GAO/HEHS–98–14. Washington:
General Accounting Office. 1998.
19. Reinhardt UE. Coverage and access in health
care reform. New Engl J of Med 330:1452–3.
1994.
20. Hafner-Eaton C. Physician utilization
disparities between the uninsured and insured:
Comparisons of the chronically ill, acutely ill, and
well nonelderly populations. JAMA 269:787–92.
1993.
21. Currie J, Gruber J. Health insurance eligibility,
utilization of medical care, and child health.
Quarterly J of Econ 111(2):431–66. 1996.
Disparity:
Eliminated/Narrowing
63% (N=11)
Cannot assess
5% (N=1)
Disparity:
Widening/No change
32% (N=7)
Number of objectives = 19
NOTE: Disparity is assessed by comparing the change for the special population with the change for
the total population. Not all objectives for the special population are applicable to disparity assessment.
(See Disparity Assessment section in the Appendix.)
Disparity:
Eliminated/Narrowing
50% (N=3)
Cannot assess
0% (N=0)
Disparity:
Widening/No change
50% (N=3)
Number of objectives = 6
NOTE: Disparity is assessed by comparing the change for the special population with the change for
the total population. Not all objectives for the special population are applicable to disparity assessment.
(See Disparity Assessment section in the Appendix.)
14 Healthy People 2000 Final Review
Figure L. Assessment of disparity for older adults objectives
Figure M. Assessment of disparity for people with disabilities
objectives

Disparity:
Eliminated/Narrowing
58% (N=21)
Cannot assess
11% (N=4)
Disparity:
Widening/No change
31% (N=11)
Number of objectives = 36
NOTE: Disparity is assessed by comparing the change for the special population with the change for
the total population. Not all objectives for the special population are applicable to disparity assessment.
(See Disparity Assessment section in the Appendix.)
0
1988 19901989 1991 1992 1993
Usual source of care
Year 2000 target
First trimester prenatal care
Childhood immunization
1994 1996 1997 19981995 1999
60
70
80
90
100
Percent
Healthy People 2000 Final Review 15
Figure N. Assessment of disparity for people with low
socioeconomic status objectives
Figure O. Access to care: United States, 1988–99
1988 1989 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Year
2000
target
First trimester prenatal
care ................. 75.9% 75.5% 75.8% 76.2% 77.7% 78.9% 80.2% 81.3% 81.9% 82.55% 82.8% 83.2% 90%
Childhood immunizations . . - - - - - - - - - - - - 55% 67% 68% 76% 78% 78% 81% 80% 90%
Usual source of care...... --- --- 80% --- 78% 83% 84% 85% 85% 84% 85% --- 90%
- - - Data not available.
Notes: Data for immunizations are for children 19–35 months receiving the 4 DTP:3 MMR:1 Polio series; data for usual source of care are for
people 18 years and over. 1999 data for early prenatal care are preliminary.
SOURCES: CDC/NCHS, National Vital Statistics System; CDC/NCHS, National Health Interview Survey; CDC/NCHS, CDC/NI, National
Immunization Survey.

0
10
20
30
40
50
Percent
All ages Under 18 18-24
Years
25-34 35-44 45-64
Year 2000 target = 0%
All ages
Under
18 years
18–24
years
25–34
years
35–44
years
45–64
years
People without health
insurance ............. 16.6% 12.7% 29.0% 22.2% 16.4% 12.2%
SOURCE: CDC/NCHS, National Health Interview Survey.
16 Healthy People 2000 Final Review
Figure P. Percent of people under 65 years without health
insurance coverage: United States, 1998

Health Status Indicators and Priority Data Needs

Health Status
Indicators and
Priority Data Needs
Health Status Indicators
Healthy People 2000 objective 22.1
called for the development of a set of
Health Status Indicators (HSIs)
appropriate for use by Federal, State,
and local health agencies (1). The
purpose of the HSIs was to make it
possible to compare health status
measures among national, State, and
local levels. Under the auspices of the
Centers for Disease Control and
Prevention, a group of public health
professionals, known as Committee
22.1, was convened, and through a
rigorous consensus process, a list of 18
Health Status Indicators was developed
(2). The HSI definitions are discussed in
detail in Healthy People Statistical
Notes, number 3, ‘‘Health Status
Indicators: Definitions and National
Data’’ (3).
Most of the HSIs are comparable to
specific Healthy People 2000 objectives.
However, several HSIs were defined
differently from corresponding
objectives in Healthy People 2000.
The International Classification of
Diseases, Ninth Revision (ICD–9) codes
used for the mortality indicators come
from established NCHS cause-of-death
lists. For three of the HSIs these codes
are different from ICD–9 codes used for
similar Healthy People 2000 objectives.
The HSI for homicide includes deaths
due to legal intervention (ICD–9:
E970–E978) while objective 7.1 is
based only on ICD–9 codes E960–E969.
The HSI for lung cancer deaths includes
cancer of the trachea (ICD–9: 162.0),
which is excluded from duplicate
objectives 3.2 and 16.2. The HSI for
heart disease deaths is based on ICD–9
codes 390–398, 402, and 404–429,
while duplicate objectives 1.1, 3.1, and
15.1 focus specifically on coronary heart
disease deaths (ICD–9: 402, 410–414,
and 429.2). With the exception of heart
disease, the differences in the death
rates between the HSIs and the
corresponding Healthy People 2000
objectives are relatively small.
The data source for the HSI and for
objective 10.1 for work-related injury
deaths is the Census of Fatal
Occupational Injuries, Bureau of Labor
Statistics, Department of Labor.
However, the HSI rate is for the total
population 16 years of age and over and
the denominator is obtained from the
Bureau of the Census. For objective
10.1, the rate is for full-time workers 16
years of age and over and the
denominator is provided by the Bureau
of Labor Statistics.
All of the HSIs were defined so that
the higher rate would indicate poorer
health status, contrary to several
corresponding Healthy People 2000
objectives. Prenatal care is measured by
the percent of mothers delivering live
infants who did not receive care during
the first trimester of pregnancy; air
quality is measured by the percent of
people living in counties exceeding U.S.
Environmental Protection Agency (EPA)
standards for air quality during the
previous year.
Three HSIs (total deaths, births to
adolescents, and childhood poverty)
have no corresponding Healthy People
2000 objective. Recent trend data and
data by race and Hispanic origin for the
18 HSIs are shown in tables B and C.
Two reports have been published in
the Statistical Notes series discussing
the national data for the Health Status
Indicators by race and Hispanic origin
(4) and an assessment of the HSIs for
each State (5).
Priority Data Needs
As part of their mandate to identify
a set of indicators of community health
status that would be relevant to public
health practice, Committee 22.1 also
produced a list of 16 Priority Data
Needs (PDNs) (5), for which State-level
data were not available at the time.
These measures included indicators of
processes affecting health, as well as
additional indicators of risk factors and
health outcomes. Most of the PDNs
generally correspond to objectives in
Healthy People 2000. However, because
of differences in the data systems used
at the national and State levels, there are
some differences in definition between
some of the PDNs and the
corresponding Healthy People 2000
objectives (6). These difference are
noted in table D. There were no specific
objectives for several PDN measures
(health care coverage for the population
under 18 years of age, regular source of
primary and dental care for children and
adolescents, and blood lead testing in
children under 5 years of age.)
National-level data for the
corresponding Healthy People 2000
objectives are also presented in table D.
As recommended by Committee
22.1, existing data collection systems
have been modified to address the
priority data needs, and State-level data
are now available for thirteen PDNs
from the data sources shown in table D.
There are still no sources of State-level
data on several PDNs (regular source of
primary care and dental services for
children and adolescents, overweight in
adolescents, blood lead levels in
children, incidence of Hepatitis B, and
prevalence of dental decay in children
and adolescents). These measures
remain on a list of data needs.
In the course of developing the
Healthy People 2010 objectives, a set of
Leading Health Indicators (LHIs) was
identified (see Introduction). The LHIs
focus on individual behaviors, physical
and social environmental factors, and
health system measures that are viewed
as determinants of health. Four of the
HSIs (motor vehicle crash deaths,
homicide, prenatal care, and air quality)
and seven of the PDNs (immunization
for children and elderly, health care
coverage, regular source of care,
smoking, alcohol misuse, and
overweight), for which both national and
State-level data are available, have been
incorporated in the LHIs.
References
1. U.S. Department of Health and Human
Services. Healthy people 2000: National health
promotion and disease prevention objectives.
Washington: Public Health Service. 1991.
2. Freedman, MA. Health Status Indicators for the
year 2000. Healthy people statistical notes; no 1.
Hyattsville, Maryland: National Center for Health
Statistics. 1991.
3. Klein, RJ, Hawk, SA. Health Status Indicators:
Definitions and national data. Healthy people
statistical notes; no 3. Hyattsville, Maryland:
National Center for Health Statistics. 1992.
4. Plepys C, Klein R. Health Status Indicators:
Differentials by race and Hispanic origin. Healthy
people statistical notes; no 10. Hyattsville,
Maryland: National Center for Health Statistics.
1995.
5. Keppel KG, Pearcy JN. Healthy people 2000:
An assessment based on the Health Status
Indicators for the United States and each State.
Healthy people statistical notes; no 19. Hyattsville,
Maryland: National Center for Health Statistics.
2000.
6. Kim I, Keppel KG. Priority Data Needs:
Sources of national, State, and local-level data and
data collection systems. Healthy people statistical
notes; no 15. Hyattsville, Maryland: National
Center for Health Statistics. 1997.
Healthy People 2000 Final Review 19

Table B. Health Status Indicators: United States, 1996–98
Health status indicators 1996 1997 1998
1 Race/ethnicity-specific infant mortality as measured by the rate (per 1,000 live births) of deaths among infants
under 1 year of age ................................................................... 7.3 7.2 7.2
White ............................................................................. 6.1 6.0 6.0
Black.............................................................................. 14.7 14.2 14.3
American Indian/Alaska Native
1
.......................................................... 10.0 8.7 9.3
Chinese
1
........................................................................... 3.2 3.1 4.0
Japanese
1
.......................................................................... *4.2 *5.3 *3.5
Filipino
1
............................................................................ 5.8 5.8 6.2
Hawaiian and part-Hawaiian
1
............................................................ * * *
Other Asian or Pacific Islander
1
.......................................................... 5.7 5.0 5.7
Hispanic origin
1,2
..................................................................... 6.1 6.0 5.8
2 Total deaths per 100,000 population (ICD–9 nos. 0–E999)
3
....................................... 491.6 479.1 471.7
3 Motor vehicle crash deaths per 100,000 population (ICD–9 nos. E810–E825)
3
........................ 16.2 15.9 15.6
4 Work-related injury deaths per 100,000 population
4
............................................. 3.1 3.0 2.9
5 Suicides per 100,000 population (ICD–9 nos. E950–E959)
3
...................................... 10.8 10.6 10.4
6 Homicides per 100,000 population (ICD–9 nos. E960–E978)
3
..................................... 8.5 8.0 7.3
7 Lung cancer deaths per 100,000 population (ICD–9 no. 162)
3
..................................... 37.9 37.4 37.0
8 Female breast cancer deaths per 100,000 females (ICD–9 no. 174)
3
............................... 20.2 19.4 18.8
9 Cardiovascular disease deaths per 100,000 population (ICD–9 nos. 390–448)
3
........................ 170.7 166.1 161.2
Heart disease deaths per 100,000 population (ICD–9 nos. 390–398, 402, 404–429)
3
................... 134.5 130.5 126.6
Stroke deaths per 100,000 population (ICD–9 nos. 430–438)
3
..................................... 26.4 25.9 25.1
10 Reported incidence (per 100,000 population) of acquired immunodeficiency syndrome
5,6
................ 27.8 23.1 19.5
11 Reported incidence (per 100,000 population) of measles......................................... 0.2 0.05 0.04
12 Reported incidence (per 100,000 population) of tuberculosis ...................................... 8.0 7.4 6.8
13 Reported incidence (per 100,000 population) of primary and secondary syphilis ....................... 4.3 3.2 2.6
14 Prevalence of low birth weight as measured by the percentage of live born infants weighing under 2,500
grams at birth ........................................................................ 7.4 7.5 7.6
15 Births to adolescents (10–17 years) as a percentage of total live births .............................. 5.1 4.9 4.6
16 Prenatal care as measured by the percentage of mothers delivering live infants who did not receive care
during the first trimester of pregnancy ...................................................... 18.1 17.5 17.2
17 Childhood poverty, as measured by the proportion of children under 15 years of age living in families at or
below the poverty level
Under 18 years ...................................................................... 20.5 19.9 18.9
Under 15 years ...................................................................... 21.1 20.5 19.5
5–17 years
7
......................................................................... 18.9 18.8 17.8
18 Proportion of persons living in counties exceeding U.S. Environmental Protection Agency standards for air
quality during the previous year
8
.......................................................... 18.7 21.1 23.5
*Infant mortality rates for groups with fewer than 10,000 births are considered unreliable. Infant mortality rates for groups with less than 7,500
births are considered highly unreliable and are not shown.
1
Rates based on a period-linked birth and infant death file using weighted data. See text for Priority Area 14.
2
Hispanic origin can be of any race.
3
Age adjusted to the 1940 U.S. standard population.
4
Data are for people 16 years and over.
5
By date of diagnosis. Adjusted for delays in reporting; not adjusted for underreporting.
6
Beginning with 1996, data are for people 13 years and over and methodology is changed. See text for Priority Area 18.
7
Related children in families.
8
Data based on 1990 county population estimates.
Health status indicators
Number Data source
1–3,5–9,14–16 National Vital Statistics System, CDC, NCHS.
4 Census of Fatal Occupational Injuries, DOL, BLS.
10 HIV/AIDS Surveillance System, CDC, NCHSTP.
11 National Notifiable Disease Surveillance System, CDC, EPO.
12 Tuberculosis Morbidity Data, CDC, NCHSTP.
13 Sexually Transmitted Disease Surveillance System, CDC, NCHSTP.
17 Current Population Survey, U.S. Bureau of the Census.
18 National Air Quality and Emission Trends Report, EPA, OAR.
20 Healthy People 2000 Final Review

Table C. Health Status Indicators by race and Hispanic origin: United States, 1998
Health status indictors
Race
Total White Black
American
Indian or
Alaska
Native
Asian or
Pacific
Islander
Hispanic
origin
1
1 Race/ethnicity-specific infant mortality as measured by the rate (per
1,000 live births) of deaths among infants under 1 year of age .... 7.2 6.0 14.3
2
9.3
2
5.5
2
5.8
2 Total deaths per 100,000 population (ICD–9 nos. 0–E999)
3
....... 471.7 450.3 690.9 458.1 264.6 342.8
3 Motor vehicle crash deaths per 100,000 population (ICD–9 nos.
E810–E825)
3
........................................ 15.7 15.7 16.6 31.8 8.6 14.9
4 Work-related injury deaths per 100,000 population
4
............. 2.9 2.9 2.4 1.7 1.9 3.4
5 Suicides per 100,000 population (ICD–9 nos. E950–E959)
3
...... 10.4 11.2 5.9 13.4 5.9 6.0
6 Homicides per 100,000 population (ICD–9 nos. E960–E978)
3
..... 7.3 4.3 25.2 9.9 3.7 9.9
7 Lung cancer deaths per 100,000 population (ICD–9 no. 162)
3
..... 37.0 36.8 44.6 25.1 17.2 13.6
8 Female breast cancer deaths per 100,000 females. (ICD–9 no.
174)
3
.............................................. 18.8 18.3 25.3 10.3 9.9 12.1
9 Cardiovascular disease deaths per 100,000 population (ICD–9
nos. 390–448)
3
....................................... 161.2 154.1 240.2 123.8 95.6 109.3
Heart disease deaths per 100,000 population (ICD–9 nos.
390–398, 402, 404–429)
3
............................... 126.7 122.0 183.3 97.1 67.5 84.2
Stroke deaths per 100,000 population (ICD–9 nos. 430–438)
3
..... 25.1 23.3 41.4 19.6 22.7 19.0
10 Reported incidence (per 100,000 population) of acquired
immunodeficiency syndrome
5
............................ 19.5
6
8.5
6
82.9 9.4 4.3 33.0
11 Reported incidence (per 100,000 population) of measles......... 0.04 - - - - - - - - - - - - - - -
12 Reported incidence (per 100,000 population) of tuberculosis ...... 6.8
6
2.3
6
17.8 12.6 36.6 13.6
13 Reported incidence (per 100,000 population) of primary and
secondary syphilis..................................... 2.6
6
0.5
6
17.1 2.8 0.4 1.5
14 Prevalence of low birth weight as measured by the percentage of
live born infants weighing under 2,500 grams at birth .......... 7.6 6.5 13.0 6.8 7.4 6.4
15 Births to adolescents (10–17 years) as a percentage of total live
births .............................................. 4.6 3.5 7.6 8.4 2.0 6.9
16 Prenatal care as measured by the percentage of mothers
delivering live infants who did not receive care during the first
trimester of pregnancy ................................. 17.2 15.2 26.7 31.2 16.9 25.7
17 Childhood poverty, as measured by the proportion of children
under 15 years of age living in families at or below the poverty
level
Under 18 years ...................................... 18.9 15.1 36.7 - - - - - - 34.4
Under 15 years ...................................... 19.5 15.5 38.4 - - - - - - 34.7
5–17 years
7
......................................... 17.8 - - - - - - - - - - - - - - -
18 Proportion of persons living in counties exceeding U.S.
Environmental Protection Agency standards for air quality during
the previous year
8
..................................... 23.5 22.6 24.9 18.9 44.9 43.8
- - - Data not available.
1
Hispanic origin can be of any race.
2
Rates based on a period linked birth and infant death file using weighted data. See text for Priority Area 14.
3
Age adjusted to the 1940 standard population.
4
Data are for people 16 years and over.
5
Beginning with 1996, data are for people 13 years and over and methodology is changed. See text for Priority Area 18.
6
Data are for the non-Hispanic population.
7
Related children in families.
8
Data based on 1990 county population estimates.
Health status indicators
Number Data source
1–3,5-9,14–16 National Vital Statistics System, CDC, NCHS.
4 Census of Fatal Occupational Injuries, DOL, BLS.
10 HIV/AIDS Surveillance System, CDC, NCHSTP.
11 National Notifiable Disease Surveillance System, CDC, EPO.
12 Tuberculosis Morbidity Data, CDC, NCHSTP.
13 Sexually Transmitted Disease Surveillance System, CDC, NCHSTP.
17 Current Population Survey, U.S. Bureau of the Census.
18 National Air Quality and Emission Trends Report, EPA, OAR.
Healthy People 2000 Final Review 21

Table D. Priority Data Needs
Priority Data Need
Objective
number
Duplicate
objective
number
1998
national
data
National
data
source
State data
source
Indicators of Processes:
1 Children 19–35 months of age who have been immunized
with 4-3-1 series ................................. 20.11
1
80% NIS NIS
2 Adults 65 years and over who have been immunized for
a. Pneumonia ................................. 20.11 46% NHIS BRFSS
b. Influenza................................... 20.11 63% NHIS BRFSS
3 Assessed rivers, lakes, and estuaries that support
a. Consumable fish
Rivers .................................... 11.10 88% NWQI NWQI
Lakes .................................... 11.10 59% NWQI NWQI
Estuaries.................................. 11.10 65% NWQI NWQI
b. Recreational activities
Rivers .................................... 11.10 72% NWQI NWQI
Lakes .................................... 11.10 80% NWQI NWQI
Estuaries.................................. 11.10 91% NWQI NWQI
4 Women 18 years and over receiving a Pap test in preceding
3 years ........................................ 16.12 79% NHIS BRFSS
5 Women 50 years and over receiving a mammogram in
preceding 2 years ................................
2
16.11
2
64% NHIS BRFSS
6 Population without health care coverage
a. Under 18 years .............................. ---
3
NHIS CPS
b. 18–64 years ................................ 21.4
4
16.6% NHIS BRFSS
7 Population with a regular source of
a. Primary care
5–17 years ................................ ---
3
NHIS - - -
18 years and over ........................... 21.3 85% NHIS BRFSS
b. Dental services (dental visit in past year)
5–17 years ................................ ---
3
MEPS - - -
18 years and over ........................... 13.14
5
65% NHIS BRFSS
Indicators of Risk Factors:
8 Cigarette smoking
a. Grades 9–12 ................................ 4.6 3.20
6
18.2% NHSDA YRBS
b. 18 years and over............................ 3.4 15.12, 16.6 24% NHIS BRFSS
9 Alcohol misuse
a. Grades 9–12 ................................ 4.7
1,7
30.8% MTF YRBS
b. 18 years and over............................ 4.7
1,8
40.0% MTF BRFSS
10 Overweight
a. 12–17 years ................................ 2.3 1.2, 15.10, 17.12
9,10
24% NHANES - - -
b. 18 years and over............................ 2.3 1.2, 15.10, 17.12
10,11
35% NHANES BRFSS
11 Hypertension awareness (18 years and over)............ 15.13
12
85% NHIS BRFSS
12 Hypercholesterolemia awareness (18 years and over) ..... 15.14
13
67% NHIS BRFSS
13 Confirmed abuse and neglect of children (per 1,000
children) ...................................... 7.4
14
41.9 NCANDS NCANDS
Indicators of Health Status Outcomes:
14 Children under 5 years of age who have been
a. Tested for blood lead levels..................... --- --- ---
b. Tested and have blood lead levels greater than
15 µg/dL .................................. 11.4
10,15
393,000 NHANES - - -
15 Incidence of hepatitis B (per 100,000 population) ......... 20.3
1
16.9 NNDSS NNDSS
16 Children with one or more decayed primary or permanent
teeth
a. 6–8 years .................................. 13.1
10
52% NHANES - - -
b. 15 years ................................... 13.1
10
61% NHANES - - -
- - - Not available.
1
1999 data.
2
Data are for clinical breast examination and mammogram.
3
Data are not shown. Not a Healthy People 2000 objective.
4
Preliminary data for persons under 65 years.
5
Data are for persons 35 years and over.
22 Healthy People 2000 Final Review
6
Data are for persons 12–17 years.
7
Data are for high school seniors.
8
Data are for college students.
9
Data are for persons 12–19 years.
10
1988–1994 data.
11
Data are for persons 20 years and over.
12
Data are for people who can state their blood pressure is high.
13
Data are for people who had their blood cholesterol checked in past 5 years.
14
1993 data.
15
Data are for persons 1–5 years of age.
Data source acronyms:
BRFSS Behavioral Risk Factor Surveillance System, CDC, NCCDPHP.
CPS Current Population Survey, U.S. Bureau of the Census.
MEPS Medical Expenditure Panel Survey, AHRQ.
MTF Monitoring the Future, NIH, NIDA
NHANES National Health and Nutrition Examination Survey, CDC, NCHS
NHIS National Health Interview Survey, CDC, NCHS
NHSDA National Household Survey on Drug Abuse, SAMHSA, OAS
NCANDS National Incidence of Child Abuse and Neglect Survey, Administration for Children and Families, NCCAN.
NIS National Immunization Survey, CDC, NIP and NCHS
NNDSS National Notifiable Disease Surveillance System, CDC, EPO
NWQI National Water Quality Inventory, EPA, Office of Water
YRBSS Youth Risk Behavior Surveillance System, CDC, NCCDPHP.
Healthy People 2000 Final Review 23

Sentinel Objectives

Sentinel Objectives
In the original Healthy People 2000
publication, objectives that were
representative of the scope and
magnitude of the improvements
envisioned in Healthy People 2000 were
identified as ‘‘sentinel objectives’’ (1).
Forty-seven objectives were selected
from the four categories of priority
areas—Health Promotion, Health
Protection, Preventive Services, and
Surveillance and Data Systems—and at
least one objective from each of the 22
priority areas was included. Sixteen
objectives were selected from the eight
Health Promotion Priority Areas, 10
objectives were selected from the 5
Health Protection Priority Areas, and 20
objectives were selected from the 8
Preventive Services Priority Areas. One
objective from the Surveillance and
Data Systems Priority Area, a
cross-cutting priority area that supports
each of the others, was also selected.
The sentinel objectives are listed in
table E.
A summary assessment of progress
toward achieving the objectives of
Healthy People 2000 based on the
sentinel objectives was published in
1995 (2). A similar summary based on
recent data is included here. Because the
success of Healthy People depends on
the combined efforts of governmental
agencies, professional associations, and
private and voluntary organizations
acting at the local level, this summary
includes data at the State level for those
objectives where sources of State data
are available.
National Data
A summary of target attainment
status for each of the sentinel objectives
in the United States is shown in table E.
For each objective, a progress quotient
of the targeted change achieved is
shown along with a color code
indicating the current status of progress
toward meeting the year 2000 target.
Calculation of the progress quotient is
described in the Measuring Progress
Toward the Healthy People 2000 Targets
section in the Appendix. One hundred
percent indicates that the target has been
met. Negative percentages indicate that
the change from the baseline to the most
recent value is moving away from the
year 2000 target. Dark green indicates
that the target has been met for 2 years
in a row or in two successive surveys.
Light green indicates that the target was
met in the most recent year or survey
cycle but not in the previous year or
survey cycle. Rose indicates that the
most recent value is between the
baseline and the target and, therefore,
progress has been made although the
target has not been met. Burgundy
indicates that the most recent value is
outside the baseline value, that is, the
most recent data are higher than the
baseline for objectives that targeted
reduction or lower than the baseline for
objectives that targeted an increase. In
these cases, the change from baseline to
most recent data is moving away from
the target.
Health Promotion Priority Areas
There are 19 measures associated
with the 16 sentinel objectives in the
Health Promotion Priority Areas
(table E). Data on the baselines and
most recent values for these objectives
are shown in the detailed tables for the
individual priority areas in the Healthy
People Priority Areas section of this
Review. Targets were met for 5 of the
19 measures or 5 of the 16 objectives in
the Health Promotion Priority Areas:
Increases in physical activity for adults,
reductions in suicide deaths, reductions
in people reporting stress, reductions in
homicide deaths, and increases in
workplaces with health promotion
programs. Movement toward the target
was evident for 10 measures associated
with 8 objectives. Percent changes
ranged from 7 percent for the objective
to increase the age of initiation of
smoking by youth to 93 percent for
reduction in alcohol-related motor
vehicle fatalities. Data for four measures
associated with four different objectives
were unchanged from the baseline or
moving in the wrong direction.
Compared with the baseline, the
proportion of the population with a
sedentary lifestyle did not change.
Change for three measures—reducing
overweight, reducing marijuana use by
youth, and reducing assault injuries—
was away from the target. Data are not
available to assess objective 8.4
concerning an increase in the proportion
of schools with health education
programs.
Health Protection Priority Areas
Eleven measures are associated with
the ten sentinel objectives in the Health
Protection Priority Areas. None of the
targets for these objectives were met.
On the other hand, all of these measures
moved toward the year 2000 targets.
Changes range from achieving
11 percent of the target to reduce the
proportion of children with dental caries
to reaching 97 percent of the target to
reduce the level of lead in children’s
blood.
Preventive Services Priority Areas
The Preventive Services Priority
Areas include 21 measures associated
with 20 sentinel objectives. Targets were
met for six objectives: reduction in
coronary heart disease deaths, reversal
in the rise in cancer deaths, increase in
breast exam and mammography,
reduction in HIV infection, reduction in
syphilis infection, and reduction in
epidemic-related pneumonia and
influenza deaths. Objective 16.1 targeted
a reduction in the age-adjusted cancer
death rate from 134 deaths per 100,000
population in 1987 to 130 per 100,000
in the year 2000. In 1998, an
age-adjusted cancer death rate of 124
per 100,000 was achieved. The
reduction of 10 deaths per 100,000 was
2½ times the targeted change of 4
deaths per 100,000. Compared with the
baseline, four objectives moved away
from the target: the percent of low
birthweight infants increased, the
percent of people limited in activity by
chronic conditions increased, the
diabetes-related death rate increased, and
the percent of the population with no
health insurance coverage increased.
Surveillance and Data Systems
Priority Area
Objective 22.1 called for the
development and implementation of a
set of common health status indicators
for use by Federal, State, and local
health agencies. A set of 18 indicators
was developed by consensus and
published in 1991 (3). These Health
Status Indicators have been broadly used
to monitor changes in health status over
time and to compare health status
among racial and ethnic groups (see
Health Status Indicators/Priority Data
Needs section). The Health Status
Indicators have also been used to
compare States (4) and to compare local
geographic areas within States (5).
Healthy People 2000 Final Review 27
Table E. An assessment of target attainment for the sentinel health objectivesHealthy People 2000
for the United States and individual States
United States Target status for States
Met Not met
Percent of
targeted
change
achieved
National
target
status
Between
national
baseline
and
target
"Worse"
than the
national
baseline
For 2
years or
2 surveys
Most
recent
year only
Priority areas and objectives
Met
1
2
3
4
Health Promotion Priority Areas
Physical Activity and Fitness
1.3 Moderate physical activity (5+ times a week)
100.0
Met
*
***
(7+ times a week)
28.6
NA NA NA NA
1.5 Sedentary lifestyle
0.0
001239
Nutrition
2.3 Overweight prevalence
-150.0
*
***
2.5 Dietary fat intake
33.3
NA NA NA NA
Tobacco
3.4 Cigarette smoking prevalence
35.7
1 0482
3.5 Smoking initiation
6.7
Substance Abuse: Alcohol and Other Drugs
4.1 Alcohol-related motor vehicle crash deaths
93.0
15 4266
4.6 Alcohol use by adolescents
68.8
NA NA NA NA
Marijuana use by adolescents
-131.8
*
***
Cocaine use by adolescents
66.7
NA NA NA NA
Cigarette use by adolescents
26.9
*
***
Family Planning
5.1 Adolescent pregnancy
40.6
NA NA NA NA
Live births to adolescents
15 32112
5.2 Unintended pregnancies
26.9
NA NA NA NA
Mental Health and Mental Disorders
6.1 Suicide
108.3
Met
12 71022
6.5 Adverse health effects from stress
114.1
Met
NA NA NA NA
Violent and Abusive Behavior
7.1 Homicide
107.7
Met
22 4614
7.6 Assault injuries
-50.0
NA NA NA NA
Educational and Community-Based Programs
8.4 Comprehensive school health education
CA CA
NA NA NA NA
8.6 Worksite health promotion activities
125.0
Met
NA NA NA NA
Health Protection Priority Areas
Unintentional Injuries
9.1 Unintentional injury deaths
85.2
19 1 9 22
9.12 Motor vehicle occupant protection systems
58.1
2 0 47 2
Occupational Safety and Health
10.1 Work-related injury deaths
75.0
15 2 17 17
10.2 Work-related injuries
88.2
10
5
5
5
20
5
6
5
Environmental Health
11.4 Blood lead levels in children
96.6
NA NA NA NA
11.5 Air quality
75.9
31 5 10 5
11.6 Homes with radon testing
34.3
NA NA NA NA
Food and Drug Safety
12.2 Salmonella enteriditis outbreaks
63.5
NA NA NA NA
Oral Health
13.1 Dental caries in children
10.5
NA NA NA NA
Adolescents 15 years
94.4
NA NA NA NA
13.4 Complete tooth loss in adults 65 years and over
43.8
NA NA NA NA
See objective 4.6 for cigarettes
28 Healthy People 2000 Final Review

Preventive Services Priority Areas
Maternal and Infant Health
14.1
Infant mortality
93.5
17 3283
14.5
Low birth weight
-36.8
001635
14.11
Prenatal care in first trimester
48.6
00474
Heart Disease and Stroke
15.1
Coronary heart disease deaths
108.6 Met
28 5180
15.2
Stroke deaths
51.0
2 2407
15.4
Controlled high blood pressure
46.2
NA NA NA NA
15.6
Mean serum cholesterol level
76.9
NA NA NA NA
Cancer
16.1
Cancer deaths
250.0
Met 26
799
16.11
Breast examination and mammogram
111.4
Met 47
210
16.12
Pap test
40.0 23
8190
16.13
Fecal occult blood test
34.8
*
***
Proctosigmoidoscopy
80.0 *
***
Diabetes and Chronic Disabling Conditions
17.2
Limitation in major activity due to chronic conditions
-42.9
NA
NA NA NA
17.9
Diabetes-related deaths
-75.0
5
11629
HIV Infection
18.2
HIV infection prevalence
& Met
NA
NA
NA NA
Sexually Transmitted Diseases
19.1
Gonorrhea
83.5 26 0214
19.3
Primary and secondary syphilis
110.6 Met 38 112 0
Immunization and Infectious Diseases
20.1
Measles
97.1 20 12 19 0
20.2
Epidemic-related pneumonia and influenza deaths
102.5 Met
NA
NA NA NA
20.11
Childhood immunization
71.4 0 2 49 0
Clinical Preventive Services
21.4
Financial barriers to receipt of clinical preventive
-11.5 0 0 24 27
services
Surveillance and Data Systems Priority Area
22.1
Develop health status indicators
100.0 Met CA
CA CA CA
Year 2000 target met for two years in a row or in two successive surveys.
Target met in the most recent year or survey cycle but not in the previous year or survey cycle.
Most recent data indicates change in the direction of the target.
Most recent data "worse" than the baseline for the United States.
* Data for States are not sufficiently comparable with data for the United States to permit assessment of
target attainment.
CA Cannot assess progress for this objective.
0.0 Quantity more than zero but less than 0.05.
NA Data for States are not available.
& This objective has met its target. A progress quotient could not be calculated.
1
Number of States that met the target for the two most recent years or the two most recent surveys.
2
Number of States that met the target for the most recent year only.
3
Number of States with most recent rates between the national target and the national baseline.
4
Number of States with most recent rates equal to or greater than the national baseline for objectives
with targeted reductions, or equal to or less than the baseline for objectives with targeted increases.
5
Based on data for 41 States.
Table E. An assessment of target attainment for the sentinel health objectives
for the United States and individual States-Con.
Healthy People 2000
Healthy People 2000 Final Review 29
State Data
The national Healthy People 2000
targets generally were set based on data
available at the national level. In order
to assess the attainment of these national
targets at the State level, comparable
sources of data are required. There are
18 sentinel objectives for which State
and national data are comparable
because they rely on the same sources.
There are six additional objectives for
which national and State data are based
on different sources but they are
sufficiently comparable to determine
which States have met the national
target. Finally, data for States are shown
for four other sentinel objectives for
which national and State data are not
comparable; for these objectives the data
provide a basis for comparison among
States, and States in the quartile with
the most favorable rates are identified.
Since the data are not comparable, no
assessment of target attainment at the
State level is made. Altogether,
State-level data are shown for 29 of the
47 sentinel objectives. State-level data
are shown for 31 measures associated
with these 29 objectives (figures Q–ZZ).
Attainment of the national target for
an objective at the State level requires
that the target be met for two successive
calendar years or for two successive
survey cycles. Attainment of the target
in a single year could be primarily a
function of the variability in rates.
Attainment for two successive years or
survey cycles provides some assurance
that attainment was not just a function
of year-to-year variability in rates. In the
tables and maps that follow, dark green
is used to indicate that the target was
met for two successive years or survey
cycles. Light green indicates that the
target was met only in the most recent
year or the most recent survey cycle.
For objectives that called for reduction,
rose indicates that the rate for the most
recent year was greater than the national
target and less than the national
baseline. The national baseline
represents the rate for the United States
at the beginning of the monitoring
decade. For objectives that targeted an
increase in rates, rose indicates that the
rate for the most recent year was less
than the national target and greater than
the baseline for the United States.
Burgundy identifies States for which
rates in the most recent year were worse
than the national baseline, that is, the
rate was greater than the national
baseline for objectives that targeted
reductions or less than the baseline for
objectives that targeted increases. A
similar analysis of target attainment for
the 18 Health Status Indicators
associated with objective 22.1 (discussed
in Health Status Indicators/Priority Data
Needs section) has been published
elsewhere (4).
Many States developed objectives
and set targets for their own
populations. These State-specific
objectives and targets may differ from
the Healthy People 2000 objectives for
the Nation. Use of the national target is
convenient because it provides a single
standard against which all States can be
compared, some States, however, had
already achieved the national target at
the beginning of the period. Other States
may have had rates so much worse than
those of the United States at the
baseline that they could not realistically
expect to achieve the national target. In
this section, when sufficiently
comparable sources of national and
State data are available, States are
assessed in terms of whether or not they
met the national target.
A summary of target attainment for
the sentinel objectives among States is
also shown in table E. The numbers of
States that met or did not meet the
national target are shown on the right.
Health Promotion Priority Areas
In the Health Promotion Priority
Areas, State-level data for six objectives
are sufficiently comparable to national
data to determine whether national
targets were met. For example, objective
7.1 to reduce the homicide death rate
was met by 26 States. More States met
this objective than any of the other
objectives in the health promotion
priority areas. State-specific data are
shown in figures Q–BB for 9 of the 16
objectives (or 10 of the 19 measures) in
the health promotion priority areas. Data
for States are shown for four measures
for which national and State data are not
strictly comparable. These measures are
identified by asterisks (*) in table E.
Objectives for which data at the State
level were not available (NA) are
identified in table E. No State-level data
were available to monitor the sentinel
objectives in the Educational and
Community-Based Programs priority
area.
Health Protection Priority Areas
State-level data for five of the
objectives in the Health Protection
Priority Areas are sufficiently
comparable to national data to determine
whether national targets were met in
States. Objective 11.5 to increase the
proportion of people in areas with good
air quality was met by more States (36
States) than any other objective in these
priority areas. State-level data are shown
in figures CC–GG for 5 of the 10
objectives in the Health Protection
Priority Areas. No State-level data were
available to monitor sentinel objectives
in the Food and Drug Safety and Oral
Health priority areas.
Preventive Services Priority Areas
In the Preventive Services Priority
Areas, State-level data for 14 objectives
are sufficiently comparable to national
data to determine whether national
targets were met in each State. More
States (49 States) met objective 16.11 to
increase the proportion of women who
had a clinical breast exam and
mammogram within the last 2 years (47
States) than any of the other objectives
in the Preventive Services Priority
Areas. In fact, more States met the
target for this objective than any of the
other sentinel objectives. State-specific
data are shown in figures HH–ZZ for 15
of the 20 objectives (or 16 of the 21
measures) in the Preventive Services
Priority Areas. Data for States are
included for two measures for which
national and State data are not strictly
comparable (*).
No suitable measure of the extent to
which States made use of the Health
Status Indicators was developed in
conjunction with objective 22.1.
Anecdotal evidence suggests that States
used the Health Status Indicators in their
own efforts to disseminate data at both
the State and local level (5).
References
1. Department of Health and Human Services.
Healthy people 2000: National health promotion
and disease prevention objectives. Washington:
Public Health Service. 1991.
2. McGinnis MJ, Lee PR. Healthy People 2000 at
mid-decade. JAMA. 273(14). 1995.
3. Freedman, MA. Health Status Indicators for the
year 2000. Healthy people statistical notes; vol 1
no 1. Hyattsville, Maryland: National Center for
Health Statistics. 1991.
30 Healthy People 2000 Final Review
4. Keppel KG, Pearcy JN. Healthy people 2000:
An assessment based on the health status
indicators for the United States and each State.
Healthy people statistical notes; no 19. Hyattsville,
Maryland: National Center for Health Statistics.
2000.
5. National Center for Health Statistics. Health
Status Indicator reports: ‘‘State of the art.’’
Statistics and surveillance; no 8. Hyattsville,
Maryland: Public Health Service. 1996.
6. Office of Applied Statistics. Summary of
findings from the 1999 National Household
Survey on Drug Abuse. Rockville, Maryland:
Substance Abuse and Mental Health Services
Administration. 2000.
Healthy People 2000 Final Review 31

Figure Q. Objective 1.3--Percent of adults who participated in regular physical activity (5 or more times
per week): United States and each State, 1996 and 1998
Percent of adults
1996 1998
United States (NHIS)
23.0
1
30.0
United States --
In 1998, based on data from the National Health
Interview Survey (NHIS), 30 percent of the population 18 74 years of age
Median of States (BRFSS)
21.0
20.4
participated in light to moderate physical activity 5 or more times per
Alabama
17.6
21.4
week. The national target (30 percent) for Objective 1.3 was, therefore,
Alaska
23.8
24.8
attained. The baseline in 1985 was 22 percent for people 18-64 years
Arizona
21.0
13.5
of age.
Arkansas
18.1
15.4
California
21.4
24.7
In 1998, based on data from the Behavioral Risk Factor
Colorado
25.1
24.1
Surveillance System (BRFSS), the median percent of the population
Connecticut
21.4
20.4
18 years and older who participated in regular and sustained physical
Delaware
16.1
17.2
activity (5 or more times per week, 30 or more minutes per session,
District of Columbia
20.0
20.3
regardless of intensity) for the States was 20.4.
Florida
21.0
20.5
Georgia
11.6
18.0
Hawaii
28.1
30.3
Idaho
25.1
27.1
Illinois
21.1
21.9
Indiana
13.5
19.5
Iowa
18.9
19.8
Kansas
17.9
14.3
States --
It is not appropriate to assess attainment of the national
Kentucky
12.4
13.0
target based on the BRFSS data because the NHIS and BRFSS are not
Louisiana
15.9
16.1
comparable and the national target was set based on data from the NHIS.
Maine
21.0
24.2
The 12 States in the highest quartile for the percent of the population
Maryland
13.3
25.0
18 years and older participating in regular and sustained physical activity
Massachusetts
24.2
22.0
in 1998 are identified below.
Michigan
22.8
23.6
Minnesota
24.1
21.7
Hawaii 30.3
Mississippi
17.0
19.2
Wyoming 28.2
Missouri
17.8
19.1
Utah 27.2
Montana
24.2
22.5
Idaho 27.1
Nebraska
20.2
19.1
Oregon 26.7
Nevada
24.7
25.6
New Mexico 25.8
New Hampshire
22.5
23.7
Nevada 25.6
New Jersey
22.4
19.1
Vermont 25.5
New Mexico
23.9
25.8
Washington 25.5
New York
20.0
19.7
Maryland 25.0
North Carolina
13.6
18.5
Alaska 24.8
North Dakota
16.8
18.7
California 24.7
Ohio
10.2
19.0
Oklahoma
16.4
15.7
SOURCE: CDC, NCCDPHP, Behavioral Risk Factor Surveillance System.
Oregon
25.7
26.7
Pennsylvania
20.4
19.4
Rhode Island
23.4
20.4
South Carolina
14.3
17.8
South Dakota
18.6
18.2
Tennessee
14.3
16.6
Texas
21.5
20.8
Utah
27.2
27.2
Vermont
24.7
25.5
Virginia
21.4
21.3
Washington
26.3
25.5
West Virginia
12.8
13.5
Wisconsin
22.1
20.2
Wyoming
28.4
28.2
1
1995 data.
32 Healthy People 2000 Final Review

Figure R. Objective 1.5--Percent of adults who reported no leisure-time physical activity: United States
and each State, 1996 and 1998
Percent of adults
Target
18 years and older
status
1996 1998
(15.0)
United States (NHIS) 23.0
1
24.0
United States --
In 1998, based on data from the National Health
Interview Survey (NHIS), 24 percent of the population 18 years and
Median of States (BRFSS) 27.8 27.7
older reported no leisure-time physical activities during the last 2
Alabama 32.4 29.7
weeks. The national target for Objective 1.5 had not been attained
Alaska 25.4 23.5
(15 percent). The baseline in 1985 was 24 percent.
Arizona 33.3 51.3
Arkansas 37.4 35.9
In 1998, based on data from the Behavioral Risk Factor
California 23.6 25.5
Surveillance System (BRFSS), the median percent of the population
Colorado 20.2 21.3
18 years and older who did not participate in any leisure-time physical
Connecticut 25.6 27.1
activity during the previous month for the States was 27.7.
Delaware 36.1 35.4
District of Columbia 30.3 38.5
Florida 27.1 31.1
Georgia 51.4 29.6
Hawaii 21.0 18.0
Idaho 20.6 20.4
Illinois 24.9 27.1
Indiana 31.0 27.1
Iowa 26.9 26.7
Kansas 36.4 38.3
Kentucky 45.6 42.7
Louisiana 34.9 32.2
Maine 34.0 27.7
Maryland 33.8 20.3
Massachusetts 23.0 25.4
Michigan 23.3 21.4
Minnesota 23.6 25.5
Mississippi
39.5 33.8
Missouri 30.2 27.9
Montana 21.2 25.2
Nebraska 22.9 26.1
Nevada 22.7 24.1
New Hampshire
25.5 24.9
New Jersey 26.3 32.6
New Mexico 27.7 23.0
In 1998 there were 12 States with rates greater than
New York 30.4 31.0
the year 2000 target (15.0) and less than the 1985
North Carolina 40.7 27.7
baseline (24.0).
North Dakota 33.9 33.1
Ohio 42.6 29.8
In 1998 there were 39 States with rates greater than or
Oklahoma 38.2 43.0
equal to the 1985 baseline (24.0).
Oregon 19.6 18.9
Pennsylvania 26.3 32.7
SOURCE: CDC, NCCDPHP, Behavioral Risk Factor Surveillance System.
Rhode Island 26.7 29.9
South Carolina 29.7 33.7
South Dakota 34.8 33.3
Tennessee 40.8 35.8
Texas 27.9 27.9
Utah 17.1 17.1
Vermont 21.5 26.0
Virginia 29.2 24.8
Washington 19.1 17.6
West Virginia 42.7 43.7
Wisconsin 22.1 23.4
Wyoming
1995 data.
1
20.4 21.1
States
12
39
Healthy People 2000 Final Review 33

Figure S. Objective 2.3--Percent of persons 18 years and older who were overweight: United States and
each State, 1998 and 1999
Percent of persons
18 years and older
1998 1999
United States (NHANES)
--- 35.0
1
United States --
Based on data from the National Health and Nutrition
Examination Survey (NHANES) for 1988 94, 35 percent of the population
Median of States (BRFSS) 32.4 33.7
18 years and older were overweight based on height and weight obtained
Alabama 35.8 37.5
in a physical examination. The year 2000 target for Objective 2.3
Alaska 36.2 37.7
(20 percent) had not been attained for the period 1988 94. The baseline
Arizona 22.5 22.9
for Objective 2.3 was 26 percent for 1976 80.
Arkansas 33.4 38.1
California 31.0 33.5
In 1999, based on data from the Behavioral Risk Factor
Colorado 27.2 26.7
Surveillance System (BRFSS), the median percent of the population 18
Connecticut 27.4 28.6
years and older who were overweight based on a body mass index
Delaware 32.8 32.0
computed from self-reported height and weight was 33.7.
District of Columbia 31.8 30.2
Florida 32.3 33.6
Georgia 33.3 35.9
Hawaii 27.8 28.8
Idaho 29.9 33.5
Illinois 33.5 36.0
Indiana 34.5 34.1
Iowa 34.6 36.6
Kansas 31.7 33.4
States --
It is not appropriate to assess attainment of the national target
Kentucky 35.8 36.7
based on the BRFSS data because the NHANES and BRFSS are not
Louisiana 36.1 37.7
comparable and the national target was set based on data from the
Maine 31.2 32.7
NHANES. The 12 States in the lowest quartile for the percent of the
Maryland 34.5 32.7
population who were overweight in 1999 based on the BRFSS are
Massachusetts 26.9 28.3
identified below.
Michigan 34.6 36.8
Minnesota 32.5 29.2
Arizona 22.9
Mississippi 37.5 41.8
Colorado 26.7
Missouri 35.6 35.5
New Hampshire 27.9
Montana 29.1 29.8
Massachusetts 28.3
Nebraska 32.3 35.7
Connecticut 28.6
Nevada 27.0 28.9
Hawaii 28.8
New Hampshire 28.7 27.9
Nevada 28.9
New Jersey 29.7 31.4
Minnesota 29.2
New Mexico 29.9 33.0
Montana 29.8
New York 28.6 31.9
Utah 30.0
North Carolina 32.7 34.9
District of Columbia 30.2
North Dakota 33.5 36.3
Vermont 31.1
Ohio 33.6 35.9
Oklahoma 32.4 37.2
SOURCE: CDC, NCCDPHP, Behavioral Risk Factor Surveillance System.
1988-94 data.
1
---Data not available.
Oregon 32.8 32.8
Pennsylvania 34.8 35.5
Rhode Island 30.3 32.3
South Carolina 34.6 37.3
South Dakota 30.5 36.1
Tennessee 32.4 34.5
Texas 34.9 36.1
Utah 28.8 30.0
Vermont 27.4 31.1
Virginia 31.8 35.0
Washington 32.3 31.8
West Virginia 37.8 41.3
Wisconsin 34.0 33.8
Wyoming 28.1 31.8
34 Healthy People 2000 Final Review
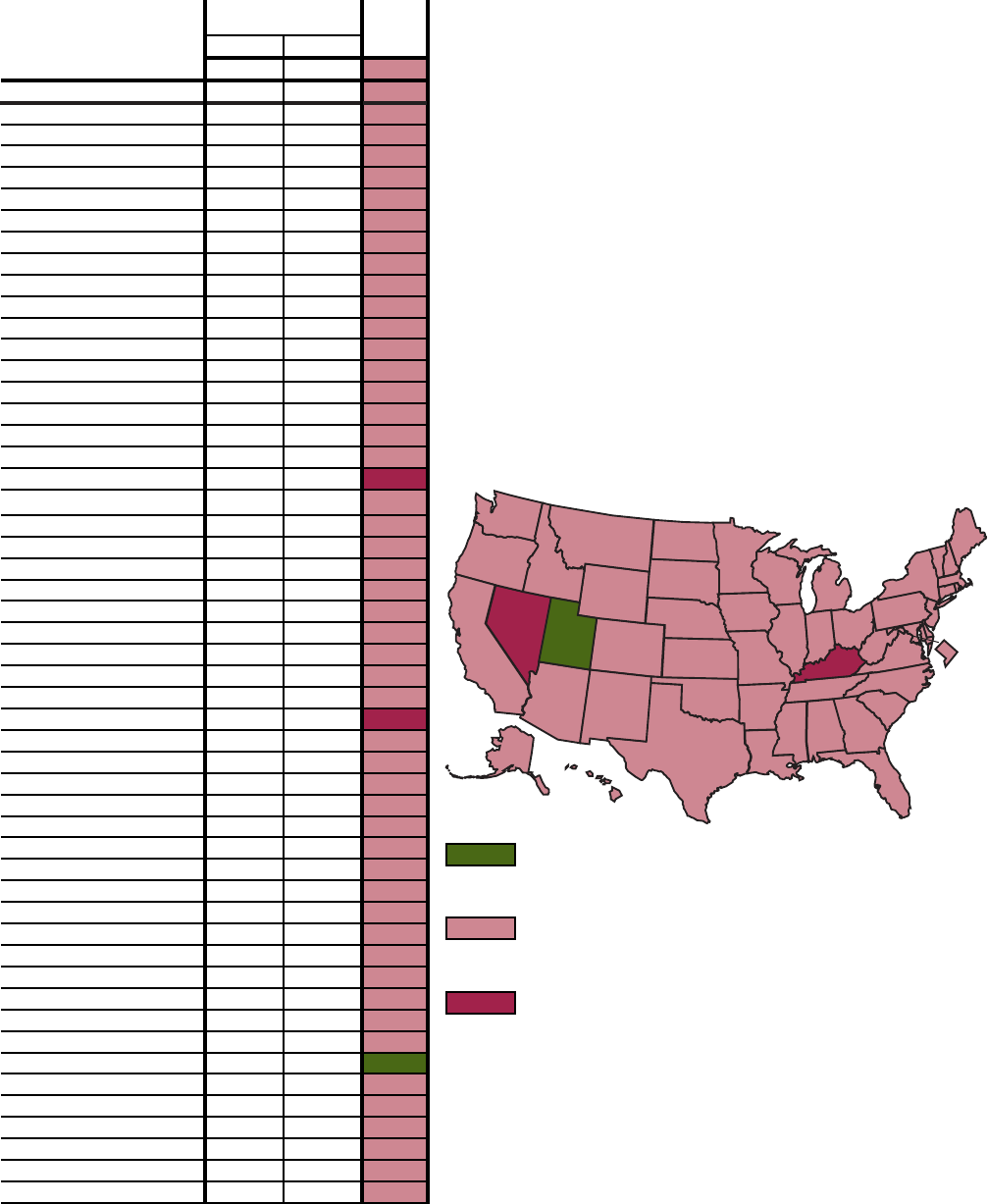
Figure T. Objective 3.4--Percent of persons 18 years and older who reported current cigarette smoking:
United States and each State, 1998 and 1999
Percent of persons
Target
18 years and older
status
1998 1999
(15.0)
United States (NHIS) 24.0 ---
United States
-- Based on data from the National Health Interview
Survey (NHIS), 24 percent of the population 18 years and older
Median of States (BRFSS) 22.9 22.8
reported current cigarette smoking. The baseline in 1987 was 29
Alabama 24.6 23.5
percent. Thirty-six percent of the change targeted by Objective 3.4
Alaska 26.1 27.3
had been achieved in 1998.
Arizona 21.9 20.1
Arkansas 25.9 27.2
Based on data from the Behavioral Risk Factor Surveillance
California 19.2 18.7
System (BRFSS), the median percent of persons 18 years and
Colorado 22.8 22.5
older who reported smoking every day or some days for the
Connecticut 20.9 22.8
States was 22.8.
Delaware 24.5 25.5
District of Columbia 21.6 20.6
Florida 22.0 20.6
Georgia 23.6 23.8
Hawaii 19.5 18.5
Idaho 20.3 21.5
Illinois 23.1 24.2
Indiana 26.0 27.0
Iowa 23.4 23.5
Kansas 21.1 21.0
Kentucky 30.8 29.7
Louisiana 25.5 23.5
Maine 22.4 23.3
Maryland 22.4 20.3
Massachusetts 20.9 19.3
Michigan 27.4 25.1
Minnesota 18.0 19.5
Mississippi 24.1 22.9
Missouri 26.4 27.1
Montana 21.5 20.2
Nebraska 22.0 23.2
Nevada 30.4 31.5
New Hampshire 23.3 22.3
New Jersey 19.1 20.6
States
New Mexico 22.6 22.5
1
Based on the BRFSS data, one State attained the
New York 24.1 21.8
national target for the year 2000 in both 1998 and
North Carolina 24.6 25.1
1999.
North Dakota 20.0 22.1
Ohio 26.1 27.6
48 In 1999, 48 States had rates greater than the year
Oklahoma 23.9 25.2
2000 target (15.0) and less than the 1987 baseline
Oregon 21.1 21.4
(29.0).
Pennsylvania 23.8 23.1
Rhode Island 22.6 22.3
2
In 1999 there were two States with rates greater than
South Carolina 24.7 23.6
or equal to the 1987 baseline (29.0).
South Dakota 27.2 22.5
Tennessee 26.1 24.8
SOURCE: CDC, NCCDPHP, Behavioral Risk Factor Surveillance System.
--- Data not available.
Texas 21.9 22.4
Utah 14.2 14.0
Vermont 22.3
21.7
Virginia 22.9 21.4
Washington 21.4 22.4
West Virginia 27.9 27.1
Wisconsin 23.4 23.7
Wyoming 22.8 23.9
Healthy People 2000 Final Review 35
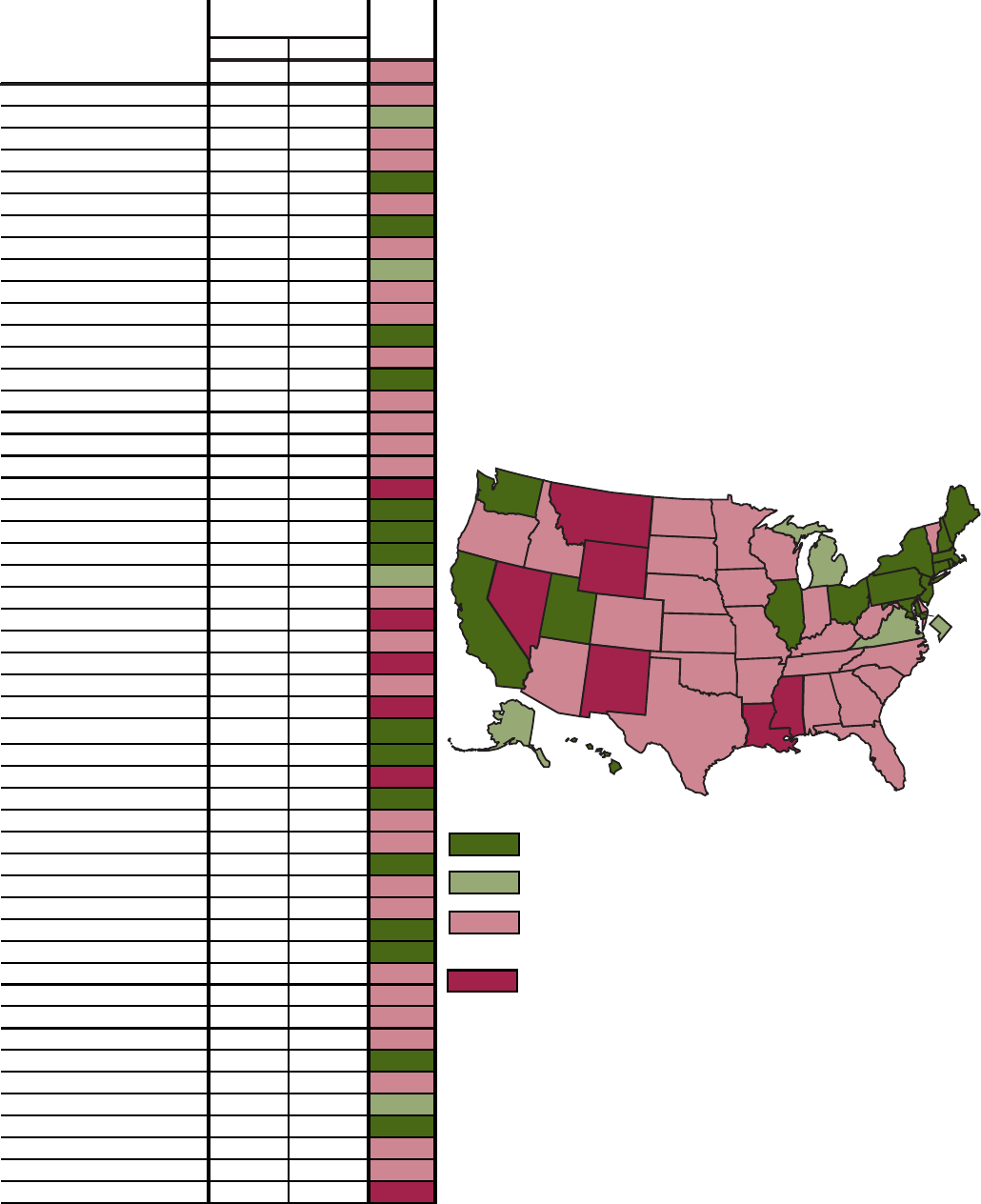
Figure U. Objective 4.1--Alcohol-related motor vehicle crash deaths per 100,000 population: United
States and each State, 1997 and 1998
Death rate
Target
per 100,000
status
1997 1998
(5.5)
United States 6.0 5.9
United States --
The baseline rate for alcohol-related motor
vehicle crash deaths in 1987 was 9.8. The rate in 1998 was 5.9.
Alabama 10.9 9.3
Ninety-three percent of the targeted change in rates was, therefore,
Alaska 6.7 5.0
achieved in 1998.
Arizona 9.5 9.1
Arkansas 7.6 7.6
California 4.1 4.1
Colorado 5.6 5.8
Connecticut 4.7 4.3
Delaware 8.3 6.1
District of Columbia 6.6 5.2
Florida 6.4 6.2
Georgia 7.7 6.7
Hawaii 4.9 4.7
Idaho 8.4 7.3
Illinois 4.9 5.0
Indiana 5.3 6.4
Iowa 6.1 5.7
Kansas 5.5 6.6
Kentucky 7.1 7.2
Louisiana 9.7 9.8
Maine 5.2 4.3
Maryland 4.3 4.0
Massachusetts 3.4 3.1
Michigan 5.7 5.5
Minnesota 4.1 5.9
Mississippi 12.6 12.8
Missouri 9.4 9.7
Montana 13.7 11.8
Nebraska 6.3 7.2
Nevada 9.5 10.1
New Hampshire 5.1 5.1
New Jersey 3.5 3.3
New Mexico 12.8 11.1
15
Fifteen States attained the target in both 1997 and
New York 2.5 2.0
1998.
North Carolina 7.1 6.8
North Dakota 7.8 6.9
4
Four States attained the target in 1998 but not in
Ohio 4.3 4.1
1997.
Oklahoma 9.1 7.5
Oregon 7.0 7.1
26
In 1998, 26 States had rates greater than the year
Pennsylvania 5.3 5.2
2000 target (5.5) and less than the 1987 baseline
Rhode Island 4.2 3.5
(9.8).
South Carolina 8.4 7.9
South Dakota 8.3 9.1
6
In 1998, there were six States with rates greater than
Tennessee 9.2 9.2
or equal to the 1988 baseline (9.8).
Texas 9.0 9.1
Utah 3.6 2.4
SOURCE: Department of Transportation, National Highway Traffic Safety Administration,
Vermont 5.8 6.4
Fatality Analysis Reporting System.
Virginia 5.7 5.1
Washington 5.3 5.4
West Virginia 8.0 8.0
Wisconsin 6.3 5.8
Wyoming 9.0 14.1
States
36 Healthy People 2000 Final Review
Figure W. Objective 4.6--Percent of youth who currently use marijuana: United States and each State, 1999
Percent
of youth
1999
United States 7.7
United States --
In 1999, based on data from the National Household Survey
on Drug Abuse (NHSDA), 7.7 percent of youth ages 12 17 used marijuana
Alabama 5.6
during the past month.
Alaska 10.5
Arizona 7.5
The NHSDA has been used since 1988 to monitor trends in substance use among
Arkansas 7.6
youth. A consistent methodology was used for monitoring national trends. The
California 8.4
national estimates in table 4, Substance Abuse: Alcohol and other drugs, are
Colorado 10.8
based on this methodology. In 1999, the old methodology would produce an estimate
Connecticut 9.3
of 7 percent of youth ages 12 17 using marijuana during the past month. The
Delaware 14.5
corresponding estimate based on the new methodology is 7.7 percent as shown here.
District of Columbia 10.3
Florida 6.6
Georgia 6.6
Hawaii 8.5
Idaho 6.3
Illinois 10.0
Indiana 9.6
Iowa 5.5
Kansas 7.1
States -- It is not appropriate to assess whether or not States attained
Kentucky 5.8
the national target. The national trend data are based on a methodology
Louisiana 7.0
that has been used since 1988 to monitor this objective. Data for States are
Maine 8.0
available from the NHSDA for the first time for 1999. The State data
Maryland 9.3
are based on a new methodology that will be used to monitor trends from
Massachusetts 12.9
1999 onward. Data for most States are derived through model-based estimation
Michigan 8.5
procedures (6). The 12 States with percentages of 12 17 year olds who
Minnesota 10.8
reported using marijuana during the past month in the lowest quartile are
Mississippi 7.4
shown below.
Missouri 7.4
Montana 11.5
Iowa 5.5
Nebraska 6.8
Alabama 5.6
Nevada 12.3
Kentucky 5.8
New Hampshire 11.7
Tennessee 5.8
New Jersey 8.2
Virginia 6.0
New Mexico 9.9
Texas 6.1
New York 7.4
Utah 6.2
North Carolina 7.3
Idaho 6.3
North Dakota 8.0
Oklahoma 6.5
Ohio 7.6
Florida 6.6
Oklahoma 6.5
Georgia 6.6
Oregon 10.0
Nebraska 6.8
Pennsylvania 7.3
Rhode Island 11.7
SOURCE: Substance Abuse and Mental Health Services Administration, National Household Survey on Drug Abuse.
South Carolina 7.8
South Dakota 7.2
Tennessee 5.8
Texas 6.1
Utah 6.2
Vermont 8.8
Virginia 6.0
Washington 10.3
West Virginia 7.1
Wisconsin 9.0
Wyoming 8.0
Healthy People 2000 Final Review 37

Figure Y. Objective 4.6--Percent of youth who currently smoke cigarettes: United States and each
State, 1999
Percent
of youth
1999
United States (NHSDA) 14.9
United States --
In 1999, based on data from the National Household
Survey on Drug Abuse (NHDSA), 14.9 percent of youth ages 12-17 smoked
Alabama 17.3
cigarettes during the past month.
Alaska 16.6
Arizona 14.5
Arkansas 20.2
California 9.0
The NHSDA has been used since 1988 to monitor trends in substance use among
Colorado 15.7
youth. A consistent methodology was used for monitoring national trends. The
Connecticut 16.0
national estimates in table 4, Substance Abuse: Alcohol and other drugs, are based
Delaware 19.5
on this methodology. In 1999, the old methodology would produce an estimate of
District of Columbia 10.7
15.9 percent of youth ages 12 17 smoking cigarettes during the past month. The
Florida 11.5
corresponding estimate based on the new methodology is 14.9 percent as shown here.
Georgia 14.5
Hawaii 10.2
Idaho 13.1
Illinois 17.6
Indiana 18.3
Iowa 18.2
Kansas 15.7
States --
It is not appropriate to assess whether or not States attained
Kentucky 23.9
the national target. The national trend data are based on a methodology
Louisiana 16.9
that has been used since 1988 to monitor this objective. The State data
Maine 16.7
are based on a new methodology that will be used to monitor trends from
Maryland 13.8
1999 onward. Data for most States are derived through model-based estimation
procedures (6). The 12 States with percentages of 12 17 year olds who
Massachusetts 16.7
reported smoking cigarettes during the past month in the lowest quartile
Michigan 16.2
are shown below.
Minnesota 20.8
Mississippi 18.8
California 9.0
Missouri 16.7
Hawaii 10.2
Montana 19.8
Utah 10.3
Nebraska 14.1
District of Columbia 10.7
Nevada 17.4
Florida 11.5
New Hampshire 15.1
New Jersey 11.9
New Jersey 11.9
New York 12.9
New Mexico 15.3
Idaho 13.1
New York 12.9
Texas 13.4
North Carolina 19.2
Maryland 13.8
North Dakota 22.4
Nebraska 14.1
Ohio 18.1
Washington 14.4
Oklahoma 17.4
Oregon 15.2
SOURCE: Substance Abuse and Mental Health Services Administration, National Household Survey
on Drug Abuse.
Pennsylvania 17.5
Rhode Island 14.8
South Carolina 19.5
South Dakota 18.9
Tennessee 17.2
Texas 13.4
Utah 10.3
Vermont 14.7
Virginia 14.7
Washington 14.4
West Virginia
22.5
Wisconsin 18.2
Wyoming 15.9
38 Healthy People 2000 Final Review

1
Objective 5.1 monitors the live birth rate for females 15 17 years old, however, there was no target specified. The target for the pregnancy rate for females 15 17 called for a 38 percent
reduction from a baseline of 80.3 to a target of 50 live births per 1,000 women in the year 2000. Assuming that a similar 38 percent reduction would apply to each of the three components of
the pregnancy rate (live births, fetal deaths, and abortions) a target was derived by reducing the live birth rate for females 15 17 in 1990 (37.5) by 38 percent. The resulting target is 23.3 live
births per 1,000 females ages 15 17 in the year 2000.
Figure Z. Objective 5.1--Live birth rates for females 15 17 years old: United States and each State, 1997
and 1998
Live birth rates
per 1,000 females
Target
ages 15 17
status
1997 1998
(23.3)
1
1
United States
32.1 30.4
United States --
The live birth rate for females 15 17 years old
was 37.5 in 1990. The rate in 1998 was 30.4. Fifty percent of the
Alabama 43.4 40.7
change required to attain the target for this part of Objective 5.1
Alaska 25.1 24.8
had been achieved in 1998.
Arizona 44.0 45.2
Arkansas 42.9 41.4
California 36.2 33.4
Colorado 29.9 29.0
Connecticut 22.5 21.4
Delaware 36.8 33.9
District of Columbia 65.9 65.5
Florida 35.1 33.3
Georgia 44.0 40.3
Hawaii 25.3 29.5
Idaho 23.3 24.5
Illinois 34.4 32.7
Indiana 32.1 28.9
Iowa 20.1 18.6
Kansas 27.5 24.8
Kentucky 35.4 31.5
Louisiana 42.1 40.4
Maine 15.4 14.9
Maryland 28.2 26.4
Massachusetts 19.1 18.2
Michigan 25.4 23.9
Minnesota 17.8 16.5
Mississippi 50.2 47.2
Missouri 29.6 28.6
Montana 20.1 19.8
Nebraska 21.3 20.5
Nevada 42.2 38.2
New Hampshire 14.0 13.1
New Jersey 21.3 20.2
States
New Mexico 44.4 44.2
15
Fifteen States attained the target in both 1997 and
New York 23.4 22.4
1998.
North Carolina 37.7 36.2
North Dakota 14.3 16.1
3
Three States attained the target in 1998 but not in
Ohio 28.6 26.7
1997.
Oklahoma 37.3 35.0
Oregon 27.0 26.3
21
In 1998, 21 States had rates greater than the year
Pennsylvania 21.9 21.8
2000 target (23.3) and less than the 1990 baseline
Rhode Island 27.6 24.4
(37.5).
South Carolina 40.0 39.6
South Dakota 21.8 19.6
12
In 1998, 12 States had rates greater than 37.5, the
Tennessee 38.5 37.7
baseline rate for the United States in 1990.
Texas 47.1 45.2
Utah 23.7 22.2
SOURCE: CDC, NCHS, National Vital Statistics System.
Vermont 12.1 11.4
Virginia 26.1 24.3
Washington 24.5 23.2
West Virginia 27.5 26.2
Wisconsin 21.4 19.6
Wyoming 23.3 22.8
Healthy People 2000 Final Review 39

Figure AA. Objective 6.1--Age-adjusted death rates for suicide: United States and each State, 1997 and 1998
Age-adjusted
death rate
Target
per 100,000
status
1997 1998
(10.5)
United States 10.6 10.4
United States --
The age-adjusted suicide death rate for the United
States was 11.7 per 100,000 population at baseline in 1987. In
Alabama 10.7 12.0
1998 the rate (10.4) was lower than the target for the year 2000 (10.5).
Alaska 21.3 22.1
The United States, therefore, attained the target for objective 6.1.
Arizona 15.7 16.0
Arkansas 13.4 13.1
California 9.9 9.6
Colorado 14.9 14.2
Connecticut 7.1 7.2
Delaware 10.8 8.0
District of Columbia 7.0 7.3
Florida 12.4 12.6
Georgia 11.2 10.0
Hawaii 11.0 9.2
Idaho 16.8 15.2
Illinois 7.1 8.1
Indiana 11.6 11.1
Iowa 11.6 10.7
Kansas 11.7 11.9
Kentucky 11.3 11.8
Louisiana 11.6 10.4
Maine 10.1 14.5
Maryland 9.3 8.9
Massachusetts 7.4 7.5
Michigan 9.5 9.1
Minnesota 9.4 9.4
Mississippi 11.7 11.5
Missouri 12.1 12.1
Montana 19.7 16.3
Nebraska 10.0 11.6
Nevada 22.0 21.2
New Hampshire 10.6 12.7
New Jersey 6.6 6.4
States
New Mexico 17.2 16.4
12
Twelve States attained the target in both 1997 and
New York 7.1 6.9
1998.
North Carolina 11.5 10.4
North Dakota 12.3 10.6
7
Seven States attained the target in 1998 but not in
Ohio 9.3 9.0
1997.
Oklahoma 14.3 13.1
Oregon 14.7 14.8
10
In 1998, 10 States had rates greater than the year
Pennsylvania 10.8 10.4
2000 target (10.5) and less than the 1987 baseline
Rhode Island 7.1 8.1
(11.7).
South Carolina 10.7 10.7
South Dakota 16.6 15.4
22
In 1998, 22 States had age-adjusted suicide death rates
Tennessee 12.4 12.6
greater than 11.7, the baseline rate for the United States
Texas 10.7 10.3
in 1987.
Utah 15.0 16.6
Vermont 10.9 12.9
SOURCE: CDC, NCHS, National Vital Statistics System.
Virginia 10.4 11.0
Washington 12.1 11.4
West Virginia 12.8 11.5
Wisconsin 10.3 10.7
Wyoming 18.4 16.8
40 Healthy People 2000 Final Review

Figure BB. Objective 7.1--Age-adjusted death rates for homicide: United States and each State, 1997
and 1998
Age-adjusted
death rate
Target
per 100,000
status
1997
1998
(7.2)
United States 7.8 7.1
United States --
At baseline in 1987 the age-adjusted homicide
death rate was 8.5 per 100,000. The age-adjusted homicide rate for
Alabama 12.2 10.8
the United States was 7.8 in 1997 and 7.1 in 1998. The target (7.2)
Alaska 8.6 7.8
was, therefore, attained for the first time in 1998.
Arizona 9.8 10.0
Arkansas 11.9 9.7
California 9.3 7.7
Colorado 4.6 5.1
Connecticut 4.6 5.0
Delaware 4.6 3.9
District of Columbia 61.5 44.6
Florida 8.4 7.9
Georgia 8.7 8.9
Hawaii 4.1 2.2
Idaho 3.5 2.7
Illinois 10.8 10.3
Indiana 7.7 7.2
Iowa 2.5 2.4
Kansas 6.5 6.5
Kentucky 6.8 6.4
Louisiana 17.0 14.4
Maine 2.1 2.3
Maryland 11.9 12.3
Massachusetts 2.5 2.3
Michigan 8.5 8.4
Minnesota 3.0 2.7
Mississippi 14.6 13.1
Missouri 8.4 8.4
Montana 4.6 3.2
Nebraska 3.9 3.6
Nevada 10.7 10.3
New Hampshire 2.2 *
New Jersey 5.1 4.4
States
New Mexico 9.7 9.9
22
Twenty-two States attained the target in both 1997
New York 6.8 5.8
and 1998.
North Carolina 9.3 9.3
North Dakota * *
4
Four States attained the target in 1998 but not in
Ohio 4.6 4.2
1997.
Oklahoma 9.1 6.7
Oregon 4.2 4.4
6
In 1998, six States had rates greater than 7.2 (the year
Pennsylvania 7.4 6.0
2000 target) and less than the 1987 baseline (8.5).
Rhode Island 3.1 2.6
South Carolina 9.5 9.2
14
Fourteen States had rates greater than 8.5, the baseline
South Dakota 3.2 *
rate for the United States in 1987.
Tennessee 10.7 9.5
Texas 7.7 7.4
5
Homicide rates were based on too few cases to be
Utah 2.9 2.9
considered reliable for these States in 1998.
Vermont * *
Virginia 7.8 6.5
SOURCE: CDC, NCHS, National Vital Statistics System.
Washington 4.8 4.3
West Virginia 5.5 4.9
Wisconsin 4.3 3.9
Wyoming 4.7 *
*Figure does not meet standards of reliability or precision.
Healthy People 2000 Final Review 41

Figure CC. Objective 9.1--Age-adjusted death rates for unintentional injuries: United States and each
State, 1997 and 1998
Age-adjusted
death rate
Target
per 100,000
status
1997 1998
(29.3)
United States 30.1 30.1
United States --
The age-adjusted death rate for unintentional
injuries was 34.7 at baseline in 1987. The United States had not
Alabama 45.9 43.2
attained the objective in 1998. However, 85 percent of the
Alaska 46.0 40.7
targeted change had been achieved.
Arizona
40.6 40.7
Arkansas
46.0 42.3
California 25.1 25.1
Colorado 31.6 32.8
Connecticut 23.9 24.5
Delaware 33.0 31.8
District of Columbia 24.6 31.5
Florida 32.1 33.3
Georgia 36.6 35.9
Hawaii 23.9 21.0
Idaho 40.3 39.4
Illinois
24.5 27.4
Indiana 29.8 30.7
Iowa 27.7 27.5
Kansas 31.9 34.9
Kentucky 38.4 36.9
Louisiana 39.3 39.9
Maine 25.8 27.1
Maryland 22.0 22.2
Massachusetts 15.0 15.5
Michigan 27.2 27.2
Minnesota 26.2 26.4
Mississippi 50.9 54.4
Missouri 36.4 35.8
Montana 45.4 42.1
Nebraska 31.0 31.6
Nevada 36.5 35.1
New Hampshire 21.1 23.3
New Jersey
23.9 21.7
States
New Mexico 52.4 51.2
19
Nineteen States attained the target in both 1997 and
New York 22.3 20.4
1998.
North Carolina 35.2 36.9
North Dakota 30.1 34.0
1
One State attained the target in 1998 but not in 1997.
Ohio 24.7 24.9
Oklahoma 40.6 39.5
9
In 1998, nine States had rates greater than the year
Oregon 33.6 34.9
2000 target (29.3) and less than the 1987 baseline
Pennsylvania 31.2 29.2
(34.7).
Rhode Island 19.0 17.3
South Carolina 40.2 41.1
22
In 1998, there were 22 States with rates greater than
South Dakota 34.4 39.8
or equal to the 1987 baseline (34.7).
Tennessee
39.8 41.0
Texas 33.5 33.7
SOURCE: CDC, NCHS, National Vital Statistics System.
Utah 30.4 30.6
Vermont 24.6 29.2
Virginia
28.2 27.7
Washington 28.6 28.5
West Virginia 35.4 37.1
Wisconsin
27.3
27.7
Wyoming 45.1 42.4
42 Healthy People 2000 Final Review

Figure DD. Objective 9.12--Percent of persons wearing a safety belt while driving or riding in a car:
United States and each State, 1995 and 1997
Percent of persons
Target
status
19971995
(85.0)
United States (NOPUS) 69.0
1
67.0
2
United States --
In 1999, based on the National Occupant
Protection Use Survey (NOPUS), 67.0 percent of persons were
Median of States (BRFSS) 66.7 69.3
observed to be using seat belts. At the baseline in 1988, 42
Alabama 67.4 66.2
percent of persons were observed to be using seat belts. The United
Alaska 66.3 65.3
States had, therefore, achieved 58 percent of the change required
Arizona 74.0 80.5
to meet the year 2000 target for objective 9.12 (85 percent).
Arkansas 67.2 65.5
California 85.1 87.2
In 1997, based on the Behavioral Risk Factor Surveillance System
Colorado 64.4 71.4
(BRFSS), the median percent of persons 18 and older who reported
Connecticut 69.0 69.1
that they always use seat belts when they drive or ride in a car for
Delaware 70.9 69.9
the States was 69.3. These data are limited to persons 18 and older--
District of Columbia --- 78.0
a part of the overall target population.
Florida 75.6 76.1
Georgia 63.9 75.4
Hawaii 87.3 87.1
Idaho 57.4 59.6
Illinois 69.1 68.1
Indiana 56.5 61.9
Iowa 62.7 67.2
Kansas 57.6 53.8
Kentucky 65.2 65.4
Louisiana 67.1 74.3
Maine 51.2 69.5
Maryland 74.5 76.1
Massachusetts 58.0 62.7
Michigan 70.3 72.3
Minnesota 58.7 59.7
Mississippi
58.1 56.5
Missouri
63.8 61.9
Montana 56.6 57.6
Nebraska 53.0 57.7
Nevada 71.5 73.7
New Hampshire 52.9 58.2
New Jersey 68.3 72.2
States
New Mexico 84.4 83.5
2
Based on BRFSS data, two States attained the
New York 73.4 73.8
national target in two consecutive surveys, 1995 and
North Carolina 85.9 84.7
1997.
North Dakota 41.5 40.2
Ohio 68.2 70.0
47
In 1997, 47 States had rates less than the year 2000
Oklahoma 54.0 63.1
target (85) and greater than the 1987 baseline (42).
Oregon 82.8 84.0
Pennsylvania 60.7 67.3
2
In 1997, there were two States with rates less than or
Rhode Island 50.0 56.8
equal to the 1988 baseline (42).
South Carolina
76.8 80.5
South Dakota 43.0 42.0
SOURCE: CDC, NCCDPHP, Behavioral Risk Factor Surveillance System.
1999 data.
2
1998 data.
1
---Data not available.
Tennessee 61.5 66.4
Texas 77.6 81.2
Utah 60.2 65.0
Vermont 71.1 73.7
Virginia 72.2 71.7
Washington 77.9 75.8
West Virginia
69.9 70.6
Wisconsin 56.1 61.3
Wyoming
47.7
50.2
Healthy People 2000 Final Review 43

Figure EE. Objective 10.1--Work-related injury death rates: United States and each State, 1998 and 1999
Death rate per
Target
100,000 workers
status
1998 1999
(4.0)
United States 4.5 4.5
United States -- The baseline for the work-related injury death rate
was 6.0 for 1983 87. The rate was 4.5 in both 1998 and 1999.
Seventy-five percent of the targeted change for the year 2000
had, therefore, been achieved.
Alabama 6.5 6.0
Alaska 14.4 14.2
Arizona 3.4 3.1
Arkansas 7.5 6.5
California 4.1 3.8
Colorado 3.6 4.5
Connecticut 3.5 2.3
Delaware 2.9 3.7
District of Columbia 5.3 5.3
Florida 5.5 4.9
Georgia 5.3 5.8
Hawaii 2.2 5.7
Idaho 8.2 6.9
Illinois 3.6 3.4
Indiana 5.2 5.7
Iowa 4.5 5.2
Kansas 7.2 5.8
Kentucky 6.4 6.4
Louisiana 8.2 7.2
Maine 4.2 5.0
Maryland 3.0 3.1
Massachusetts 1.4 2.6
Michigan 3.7 3.7
Minnesota 3.4 2.7
Mississippi 9.4 10.6
Missouri 5.3 6.0
Montana 13.2 10.9
Nebraska 6.3 7.3
Nevada 6.8 6.3
New Hampshire 3.6 2.2
New Jersey 2.6 2.6
States
New Mexico 6.2 5.1
Fifteen States attained the target in two consecutive years, 1998
New York 2.9 2.9
and 1999.
North Carolina 6.2 5.9
North Dakota 7.2 6.8
Two States attained the target only in 1999.
Ohio 3.4 4.0
Oklahoma 4.9 6.2
Seventeen States had rates in 1999 greater than the target and less
Oregon 4.3 4.2
than the baseline (6.0).
Pennsylvania 4.2 3.9
Rhode Island 2.5 2.3
In 1999, 17 States had rates greater than or equal to the baseline for
South Carolina 5.9 7.4
the United States in 1983 87 (6.0).
South Dakota 7.3 11.6
Tennessee 5.7 5.7
SOURCE: Department of Labor, Bureau of Labor Statistics, Census of Fatal Occupational Injuries.
Texas 5.4 4.8
Utah 6.6 5.2
Vermont 5.0 4.3
Virginia 5.2 4.5
Washington 3.9 3.0
West Virginia 7.7 7.5
Wisconsin 3.4 3.7
Wyoming 13.5 12.9
1
1
The rates for the United States were calculated based on denominators that were adjusted to include military personnel. The rates for States were calculated without any adjustment for
military personnel. The rates for States are, therefore, overestimates and comparisons among States may be affected by differences in the proportion of military personnel.
15
2
17
17
44 Healthy People 2000 Final Review
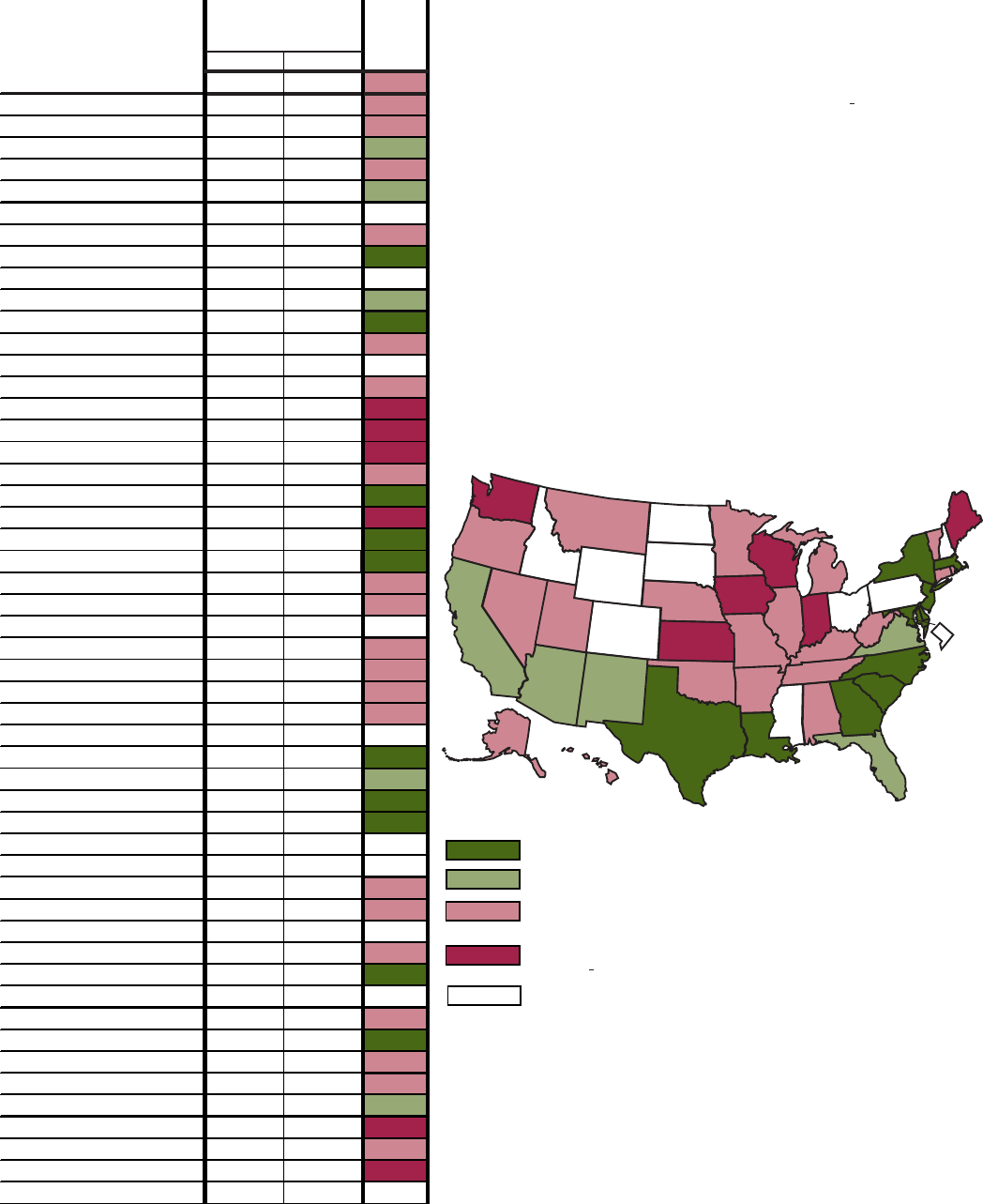
Figure FF. Objective 10.2--Nonfatal work-related injuries per 100 full-time workers in private industry:
United States and reporting States, 1997 and 1998
Nonfatal injury
rate per 100
Target
full-time workers
status
1997 1998
(6.0)
United States 6.6 6.2
United States --
The baseline for nonfatal work-related injuries
per 100 full-time workers in private industry was 7.7 for 1983 87.
Alabama 7.5 6.9
In 1998, the comparable rate was 6.2. The United States had
Alaska 7.9 7.2
achieved 88 percent of the change required to meet the year
2000 target.
Arizona 6.2 5.9
Arkansas 7.0 6.4
California 6.2 5.9
Colorado --- ---
Connecticut 6.2 6.2
Delaware 5.3 5.1
District of Columbia --- ---
Florida 6.3 5.7
Georgia 5.1 5.4
Hawaii 6.6 6.3
Idaho --- ---
Illinois --- 6.6
Indiana 8.4 7.8
Iowa 8.5 8.0
Kansas 7.8 7.7
Kentucky 8.5 7.6
Louisiana 4.7 4.9
Maine 7.1 7.8
Maryland 5.0 4.8
Massachusetts 5.4 5.5
Michigan 8.0 7.5
Minnesota 6.9 7.0
Mississippi --- ---
Missouri 7.1 7.0
Montana 7.6 7.4
Nebraska 8.3 7.6
Nevada 7.7 7.1
New Hampshire --- ---
New Jersey 5.7 4.7
New Mexico 6.2 6.0
Ten States attained the target in both 1997 and 1998.
New York 4.2 4.0
North Carolina 5.9 5.7
North Dakota --- ---
Five States attained the target only in 1998.
Ohio --- ---
Oklahoma 6.7 7.0
In 1998, 20 States had rates greater than the year 2000
Oregon 7.2 6.5
target (6.0) and less than the 1987 baseline (7.7).
Pennsylvania --- ---
Rhode Island 7.5 6.5
South Carolina 5.7 5.5
In 1998 there were six States with rates greater than or equal to
South Dakota --- ---
the 1983 87 baseline (7.7).
Tennessee 7.0 7.0
Texas 5.2 4.9
There were no data for 10 States.
Utah 7.8 7.2
Vermont 6.3 6.6
SOURCE: Department of Labor, Bureau of Labor Statistics, Annual Survey of Occupational
Virginia 6.1 5.4
Injuries and Illnesses.
---Data not available.
Washington 9.1 8.7
West Virginia --- 7.4
Wisconsin 9.0 8.5
Wyoming --- ---
States
10
5
10
20
6
Healthy People 2000 Final Review 45

Figure GG. Objective 11.5--Percent of persons in counties that have not exceeded the Environmental
Protection Agency (EPA) standards for air quality: United States and each State, 1997 and 1998
Percent in counties
not exceeding EPA
Target
air quality standards
status
1997 1998
(85.0)
United States
78.9 76.5
United States --
The percent of persons living in counties that
did not exceed EPA Air Quality Standards at baseline in 1988 was
Alabama 100.0 72.1
49.7. In 1998, the comparable percent was 76.5, although the
Alaska 85.9 85.9
percent had been higher in intervening years. Based on the
Arizona 42.1 42.1
estimate for 1998, the United States had achieved about 76 percent
Arkansas 100.0 100.0
of the targeted improvement in the percent of persons in counties
California 42.6 16.4
that have not exceeded EPA air quality standards. The air quality
Colorado 99.7 100.0
data should be interpreted with caution because they are subject
Connecticut 8.4 46.5
to the effects of weather patterns.
Delaware 16.7 17.0
District of Columbia 100.0 100.0
Florida 100.0 91.6
Georgia 80.8 70.3
Hawaii 100.0 100.0
Idaho 99.3 100.0
Illinois 97.8 97.8
Indiana 97.3 95.3
Iowa 100.0 88.2
Kansas 100.0 100.0
Kentucky 99.1 98.8
Louisiana 83.2 90.5
Maine 64.1 96.2
Maryland 38.3 72.1
Massachusetts 90.0 100.0
Michigan 100.0 90.6
Minnesota 100.0 100.0
Mississippi 100.0 100.0
Missouri 94.4 89.5
Montana 100.0 100.0
Nebraska 73.6 100.0
Nevada 38.1 38.1
New Hampshire 77.8 100.0
New Jersey 62.0 87.3
New Mexico 100.0 100.0
Thirty-one States attained the target in both 1997 and
New York 79.7 82.3
1998.
North Carolina 98.3 89.6
North Dakota 100.0 100.0
Five States attained the target only in 1998.
Ohio 100.0 98.1
Oklahoma 100.0 100.0
In 1998, there were 10 States with percents greater
Oregon 100.0 90.2
than the national baseline (49.7) and less than the
Pennsylvania 61.6 74.7
year 2000 target (85.0).
Rhode Island 100.0 100.0
South Carolina 100.0 91.0
In 1998, five States had percents less than or
South Dakota 100.0 100.0
equal to the baseline for the United States in 1988
Tennessee 87.2 66.9
(49.7).
Texas 54.6 69.2
Utah 100.0 100.0
SOURCE: Environmental Protection Agency, AIRS, OAR, National Air Quality and Emissions
Vermont 100.0 100.0
Trends Report.
Virginia 96.2 83.5
Washington 100.0 57.0
West Virginia 100.0 92.6
Wisconsin 76.8 74.2
Wyoming 100.0 100.0
States
31
5
10
5
46 Healthy People 2000 Final Review

Figure HH. Objective 14.1--Infant mortality rates: United States and each State, 1997 and 1998
Infant deaths
Target
per 1,000 live births
status
1997 1998
(7.0)
United States 7.2 7.2
United States --
The infant mortality rate at baseline in 1987 was
10.1 infant deaths per 1,000 live births. The rate in 1997 and 1998
Alabama 9.5 10.2
was 7.2. The United States has, therefore, achieved 94 percent of
Alaska 7.5 5.9
the targeted reduction in infant mortality rates.
Arizona 7.1 7.5
Arkansas 8.7 8.9
California 5.9 5.8
Colorado 7.0 6.7
Connecticut 7.2 7.0
Delaware 7.8 9.6
District of Columbia 13.2 12.5
Florida 7.1 7.2
Georgia 8.6 8.5
Hawaii 6.6 6.9
Idaho 6.8 7.2
Illinois 8.4 8.4
Indiana 8.2 7.6
Iowa 6.2 6.6
Kansas 7.4 7.0
Kentucky 7.3 7.5
Louisiana 9.5 9.1
Maine 5.1 6.3
Maryland 8.8 8.6
Massachusetts 5.2 5.1
Michigan 8.2 8.2
Minnesota 5.9 5.9
Mississippi 10.6 10.1
Missouri 7.6 7.7
Montana 6.9 7.4
Nebraska 7.4 7.3
Nevada 6.5 7.0
New Hampshire 4.3 4.4
New Jersey 6.3 6.4
New Mexico 6.1 7.2
Seventeen States attained the target in 1997 and 1998.
New York 6.7 6.3
North Carolina 9.2 9.3
Three States attained the target only in 1998.
North Dakota 6.2 8.6
Ohio 7.8 8.0
In 1998, 28 States had rates greater than the year 2000
Oklahoma 7.5 8.5
target (7.0) and less than the 1987 baseline (10.1).
Oregon 5.8 5.4
Pennsylvania 7.6 7.1
In 1998, there were three States with rates greater than or
Rhode Island 7.0 7.0
equal to the 1987 baseline (10.1).
South Carolina 9.6 9.6
South Dakota 7.7 9.1
SOURCE: CDC, NCHS, National Vital Statistics System.
Tennessee 8.6 8.2
Texas 6.4 6.4
Utah 5.8 5.6
Vermont 6.1 7.0
Virginia 7.8 7.7
Washington 5.6 5.7
West Virginia 9.6 8.0
Wisconsin 6.5 7.2
Wyoming 5.8 7.2
States
17
3
28
3
Healthy People 2000 Final Review 47

Figure JJ. Objective 14.5--Percent of live births of low birthweight: United States and each State,
1997 and 1998
Percent
Target
low birthweight
status
1997 1998
(5.0)
United States
7.5
7.6
United States --
The target for this objective has not been
attained. The percent low birthweight was 7.6 in 1998. The percent
Alabama
9.2
9.3
low birthweight has risen steadily since 1987 when the baseline was
Alaska
5.9
6.0
6.9 percent.
Arizona
6.9
6.8
Arkansas
8.4
8.9
California
6.2
6.2
Colorado
8.8
8.6
Connecticut
7.3
7.8
Delaware
8.7
8.4
District of Columbia
13.4
13.1
Florida
8.0
8.1
Georgia
8.8
8.5
Hawaii
7.2
7.5
Idaho
6.3
6.0
Illinois
7.9
8.0
Indiana
7.7
7.9
Iowa
6.4
6.4
Kansas
6.9
7.0
Kentucky
7.8
8.1
Louisiana
10.2
10.1
Maine
5.9
5.8
Maryland
8.8
8.7
Massachusetts
7.0
6.9
Michigan
7.7
7.8
Minnesota
5.9 5.8
Mississippi
10.1
10.1
Missouri
7.7
7.8
Montana
6.3
7.0
Nebraska
7.0
6.5
Nevada
7.6
7.6
New Hampshire
5.8
5.7
New Jersey 7.9 8.0
New Mexico 7.8 7.6
In 1998, 16 States had rates greater than the year
New York 7.8 7.8
2000 target (5.0) and less than the 1987 baseline (6.9).
North Carolina 8.8 8.8
North Dakota 6.2 6.5
In 1998, there were 35 States with rates greater than or
Ohio 7.7 7.7
equal to the 1987 baseline (6.9).
Oklahoma 7.3 7.2
Oregon 5.5 5.4
SOURCE: CDC, NCHS, National Vital Statistics System.
Pennsylvania 7.6 7.6
Rhode Island 7.4 7.6
South Carolina 9.2 9.5
South Dakota 5.5 5.8
Tennessee 8.8 9.1
Texas 7.3 7.4
Utah 6.6 6.7
Vermont 6.3 6.5
Virginia 7.7 7.9
Washington 5.6 5.7
West Virginia 8.3 8.0
Wisconsin 6.4 6.5
Wyoming 9.0 8.9
States
35
16
48 Healthy People 2000 Final Review
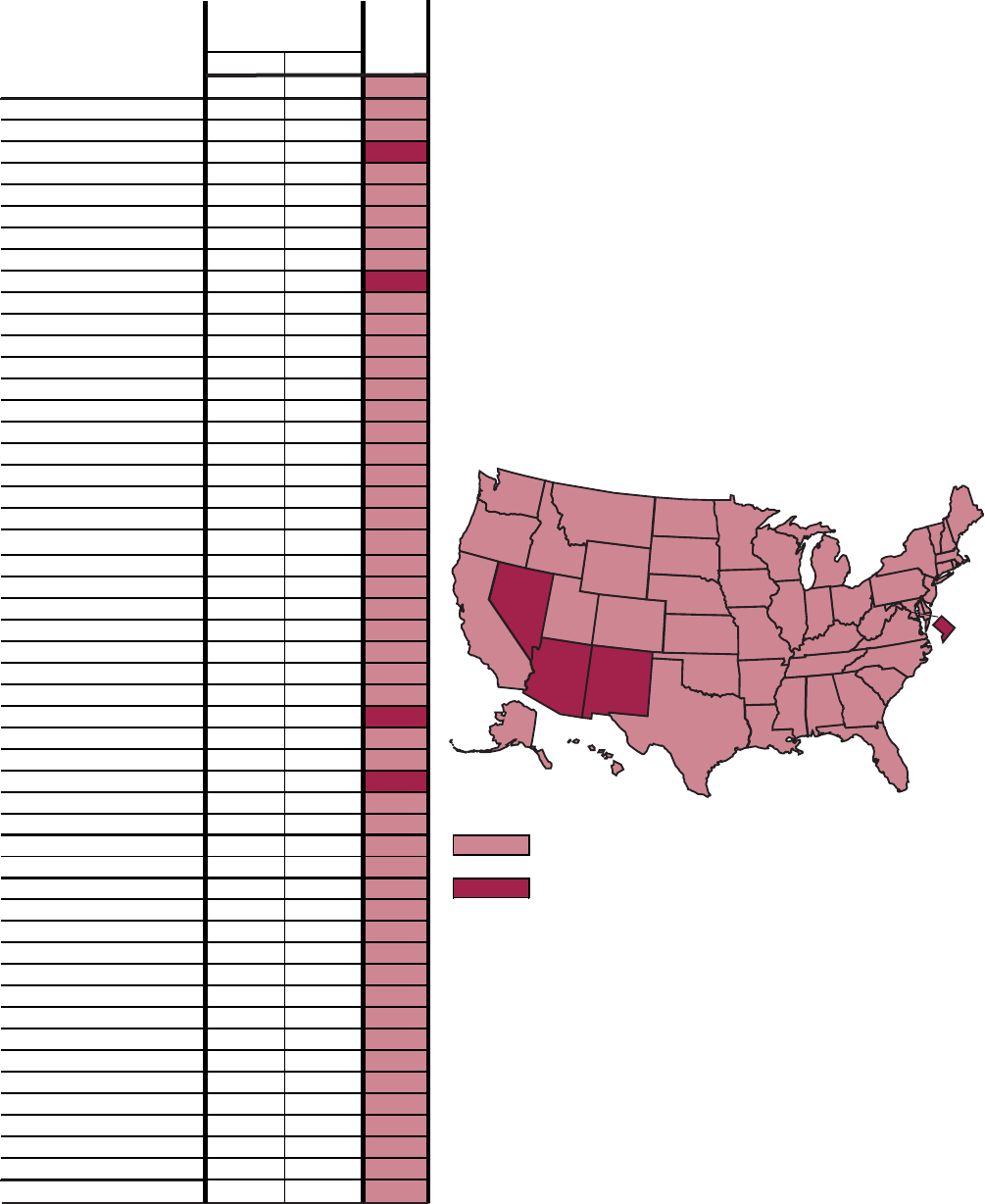
Figure KK. Objective 14.11--Percent of mothers who began prenatal care in the first trimester:
United States and each State, 1997 and 1998
Percent
beginning care
Target
in first trimester
status
1997 1998
(90.0)
United States 82.5 82.8
United States --
The United States has achieved about 49
percent of the targeted change in the percent of women beginning
Alabama 82.2 82.4
prenatal care in the first trimester of pregnancy. The baseline in
Alaska 80.4 81.4
1987 was 76 percent.
Arizona 75.4 75.1
Arkansas 75.7 77.8
California 81.8 82.4
Colorado 82.9 82.2
Connecticut 89.2 88.0
Delaware 82.5 83.4
District of Columbia 66.6 72.0
Florida 83.9 83.6
Georgia 85.8 86.4
Hawaii 83.4 85.4
Idaho 78.6 78.7
Illinois 82.4 82.7
Indiana 80.1 79.9
Iowa 87.4 87.3
Kansas 85.6 85.8
Kentucky 85.8 86.4
Louisiana 81.3 82.2
Maine 88.9 88.9
Maryland 88.8 87.8
Massachusetts 88.9 89.5
Michigan 84.2 84.3
Minnesota 84.1 84.5
Mississippi 80.1 80.6
Missouri 86.0 86.1
Montana 82.6 82.3
Nebraska 83.9 83.9
Nevada 76.1 74.6
New Hampshire 89.6 89.7
New Jersey 81.3 81.6
New Mexico 70.2 67.6
In 1998, 47 States had rates between the year 2000
New York 80.6 81.2
target (90.0) and the 1987 baseline (76.0).
North Carolina 83.9 84.5
North Dakota 84.8 85.6
In 1998, there were four States with rates lower than the
Ohio 85.1 85.5
1987 baseline (76.0).
Oklahoma 78.5 78.6
Oregon 81.1 80.2
Pennsylvania 83.8 84.8
Rhode Island 89.5 89.7
South Carolina 80.5 81.4
South Dakota 82.1 82.7
Tennessee 83.7 84.1
Texas 78.5 79.3
Utah 83.7 82.1
Vermont 88.0 87.4
Virginia 85.1 85.2
Washington
83.3 83.0
West Virginia 82.0 83.7
Wisconsin 84.6 84.3
Wyoming 82.5 81.3
SOURCE: CDC, NCHS, National Vital Statistics System.
States
4
47
Healthy People 2000 Final Review 49

Figure LL. Objective 15.1--Age-adjusted death rates for coronary heart disease: United States and each
State, 1997 and 1998
Age-adjusted
death rate
Target
per 100,000
status
1997 1998
(100.0)
United States 100.1 96.8
United States --
The United States attained the Year 2000 target
(an age-adjusted rate of 100 coronary heart disease deaths per
Alabama 92.3 88.8
100,000 population) for the first time in 1998. The baseline in 1987
Alaska 67.2 70.0
was an age-adjusted rate of 135 deaths per 100,000.
Arizona 87.1 86.3
Arkansas 108.4 106.4
California 95.4 94.3
Colorado 69.5 66.4
Connecticut 87.3 83.2
Delaware 109.5 98.0
District of Columbia 112.6 125.1
Florida 97.9 97.4
Georgia 97.8 95.5
Hawaii 65.2 62.6
Idaho 80.1 71.3
Illinois 103.1 101.3
Indiana 102.0 97.9
Iowa 101.7 97.5
Kansas 82.8 80.4
Kentucky 113.5 106.3
Louisiana 115.5 113.9
Maine 96.5 89.3
Maryland 100.1 97.1
Massachusetts 80.1 75.1
Michigan 111.6 109.1
Minnesota 67.3 63.8
Mississippi 129.4 128.2
Missouri 110.7 108.0
Montana 75.3 69.5
Nebraska 75.1 68.0
Nevada 101.8 101.9
New Hampshire 94.9 93.9
New Jersey 97.3 94.4
New Mexico 78.9 75.3
Twenty-eight States attained the target in 1997
New York 122.6 114.7
and 1998.
North Carolina 104.9 100.9
North Dakota 87.6 80.2
Ohio 106.5 103.4
Oklahoma 119.2 115.9
Oregon 77.0 72.3
Pennsylvania 101.6 96.0
Rhode Island 108.3 101.6
South Carolina 106.8 101.8
South Dakota 87.4 84.4
Tennessee 126.3 123.2
Texas 106.4 100.9
Utah 64.1 58.8
Vermont 95.2 82.9
Virginia 87.9 87.0
Washington 78.8 79.3
West Virginia 119.4 119.6
Wisconsin 85.1 84.3
Wyoming 83.0 82.0
States
Five States attained the target in 1998 but not in 1997.
In 1998, eighteen States had rates greater than the year
2000 target (100.0) and less than the 1987 baseline
(135.0).
SOURCE: CDC, NCHS, National Vital Statistics System.
28
5
18
50 Healthy People 2000 Final Review

Figure MM. Objective 15.2--Age-adjusted death rates for stroke: United States and each State,
1997 and 1998
Age-adjusted
death rate
Target
per 100,000
status
1997 1998
(20.0)
United States 25.8 25.1
United States --
The United States has achieved about half
(51 percent) of the targeted reduction in stroke deaths. The
Alabama 31.1 30.6
baseline in 1987 was an age-adjusted stroke death rate of 30.4
Alaska 22.7 25.2
and the comparable rate in 1998 was 25.1.
Arizona 23.3 22.5
Arkansas 38.2 35.3
California 25.9 25.0
Colorado 22.2 21.8
Connecticut 20.5 20.9
Delaware 22.0 23.2
District of Columbia 31.2 28.4
Florida 22.3 22.0
Georgia 34.0 31.6
Hawaii 28.2 24.1
Idaho 25.5 24.7
Illinois 26.2 25.4
Indiana 27.9 28.1
Iowa 23.1 23.1
Kansas 23.2 24.7
Kentucky 27.8 27.6
Louisiana 30.0 30.0
Maine 23.0 22.6
Maryland 25.2 25.1
Massachusetts 19.9 18.6
Michigan 26.2 25.9
Minnesota 24.4 21.9
Mississippi 35.1 33.4
Missouri 26.2 27.3
Montana 23.7 23.2
Nebraska 22.9 25.2
Nevada 26.0 25.3
New Hampshire 24.4 20.5
New Jersey 21.2 21.1
New Mexico 21.5 19.6
Two States attained the target in 1997 and 1998.
New York 19.2 18.3
North Carolina 32.0 33.1
Two States attained the target only in 1998.
North Dakota 23.6 22.5
Ohio 25.6 24.3
Forty States had rates between the year 2000 target
Oklahoma 28.5 27.7
(20.0) and the 1987 baseline (30.4) in 1998.
Oregon 28.9 29.0
Pennsylvania 24.4 23.5
In 1998, seven States had rates greater than the baseline
Rhode Island 23.1 20.0
for the United States in 1987 (30.4).
South Carolina 38.7 37.0
South Dakota 25.4 24.3
SOURCE: CDC, NCHS, National Vital Statistics System.
Tennessee 33.5 32.6
Texas 27.8 26.4
Utah 24.3 22.0
Vermont 23.7 23.2
Virginia 29.7 27.2
Washington 25.5 25.6
West Virginia 25.0 24.7
Wisconsin 26.3 26.0
Wyoming 21.4 26.2
States
2
2
40
7
Healthy People 2000 Final Review 51

Figure NN. Objective 16.1--Age-adjusted death rates for all cancers: United States and each State,
1997 and 1998
Age-adjusted
death rate
Target
per 100,000
status
1997 1998
(130.0)
United States 125.4
123.5
United States --
The age-adjusted death rate for all cancers in
1987 was 134 per 100,000. The rate in 1998 was 123.5. The United
Alabama 136.0 135.6
States has, therefore, achieved the target of 130 cancer deaths per
Alaska 116.4 116.6
100,000.
Arizona 113.1 110.6
Arkansas 138.6 133.2
California 114.3 110.6
Colorado 101.4 101.9
Connecticut 120.3 119.1
Delaware 141.5 138.9
District of Columbia 154.9 154.3
Florida 125.6 122.6
Georgia 131.8 129.8
Hawaii 95.0 100.5
Idaho 107.4 110.4
Illinois 129.6 128.4
Indiana 130.4 133.4
Iowa 116.5 117.3
Kansas 118.0 112.9
Kentucky 145.7 141.9
Louisiana 146.4 143.9
Maine 135.9 130.8
Maryland 133.8 131.3
Massachusetts 126.5 126.1
Michigan 127.2 124.7
Minnesota 115.3 112.6
Mississippi 141.1 141.3
Missouri 131.1 132.9
Montana 114.1 112.2
Nebraska 112.4 114.0
Nevada 128.5 133.9
New Hampshire 134.8 134.3
New Jersey 130.2 126.8
New Mexico 108.0 104.2
Twenty-six States attained the target in 1997 and 1998.
New York 122.4 119.7
North Carolina 130.6 128.6
Seven States attained the target only in 1998.
North Dakota 107.2 113.3
Ohio 131.8 131.5
Nine States had rates between the year 2000 target
Oklahoma 130.2 126.4
(130) and the 1987 baseline (134).
Oregon 121.2 123.4
Pennsylvania 131.4 127.7
Nine States had rates in 1998 higher than the baseline
Rhode Island 136.3 132.0
for the United States in 1987 (134).
South Carolina 133.4 130.5
South Dakota 116.5 116.7
SOURCE: CDC, NCHS, National Vital Statistics System.
Tennessee 139.9 138.9
Texas 122.7 120.2
Utah 84.6 93.5
Vermont 131.4 124.9
Virginia 130.2 126.8
Washington 117.9 117.8
West Virginia 140.3 139.1
Wisconsin 119.5 121.3
Wyoming 113.5 112.3
States
26
7
9
9
52 Healthy People 2000 Final Review

Figure OO. Objective 16.11--Percent of women 50 years of age and older who received a mammogram
and breast exam within the last 2 years: United States and each State, 1998 and 1999
Percent of women
Target
50 years and older
status
1998 1999
(60.0)
United States (NHIS) 64.0 ---
United States --
Based on data from the National Health
Interview Survey (NHIS) for 1998, 64 percent of women 50 and older
Median of States (BRFSS)
67.8 68.5
reported that they had a clinical breast exam and a mammogram
Alabama 69.9 65.3
within the last 2 years. The year 2000 target of 60 percent was,
Alaska 74.1 73.1
therefore, exceeded. The baseline in 1987 was 25 percent.
Arizona 81.0 76.9
Arkansas 57.0 61.1
In 1999, based on data from the Behavioral Risk Factor Surveillance
California 69.4 ---
System (BRFSS), the median percent of women 50 years of age and
Colorado 70.1 66.1
older who reported receiving both a mammogram and a clinical
Connecticut 69.4 77.8
breast exam within the last 2 years for the States was 68.5.
Delaware 77.1 77.2
District of Columbia 85.2 78.2
Florida 68.9 76.1
Georgia 64.1 66.2
Hawaii 69.7 69.5
Idaho 63.5 63.2
Illinois 62.1 67.9
Indiana 63.9 60.7
Iowa 64.0 68.7
Kansas 66.3 71.5
Kentucky 63.7 68.3
Louisiana 63.5 65.0
Maine 73.6 80.1
Maryland 70.7 75.6
Massachusetts 74.8 77.7
Michigan 69.8 76.5
Minnesota 60.0 69.0
Mississippi 60.1 59.3
Missouri 66.6 64.9
Montana 69.0 66.4
Nebraska 59.5 68.9
Nevada 62.4 64.7
New Hampshire 71.6 80.4
New Jersey 65.1 68.0
New Mexico 66.3 66.3
Based on BRFSS data, 47 States attained the target
New York 68.7 71.0
in both 1998 and 1999.
North Carolina 74.1 76.9
North Dakota 68.3 71.3
Two States attained the target in 1999 but not in 1998.
Ohio 68.1 71.0
Oklahoma 62.5 64.7
Mississippi attained the target in 1998 but not in 1999.
Oregon 67.8 69.3
Pennsylvania 66.5 67.9
1
Data for California were not available in 1999.
Rhode Island 70.2 74.8
South Carolina 66.9 72.6
SOURCE: CDC, NCCDPHP, Behavioral Risk Factor Surveillance System.
--- Data not available.
South Dakota 69.5 68.2
Tennessee 70.9 67.9
Texas 63.0 65.3
Utah 61.5 67.7
Vermont 67.1 70.0
Virginia 68.1 68.0
Washington 70.2 67.4
West Virginia 68.9 65.7
Wisconsin 66.8 68.8
Wyoming 60.5 64.1
States
47
2
1
Healthy People 2000 Final Review 53

Figure PP. Objective 16.12--Percent of women 18 years of age and older who had a Pap test during the
last 3 years: United States and each State, 1998 and 1999
Percent of women
Target
18 years and older
status
1998 1999
(85.0)
United States (NHIS)
79.0 ---
United States --
Based on data from the National Health
Interview Survey (NHIS) for 1998, 79 percent of women 18 years of
Median of States (BRFSS) 81.5 85.4
age and older reported that they had a Pap test within the last 3
Alabama 85.3 86.6
years. The baseline in 1987 was 75 percent. Forty percent of the
Alaska 89.5 90.9
change required to meet the year 2000 target was achieved.
Arizona 81.7 86.1
Arkansas 78.9 82.6
In 1999, based on data from the Behavioral Risk Factor Surveillance
California 82.3 ---
System (BRFSS), the median percent of women 18 years of age and
Colorado 87.0 87.4
older who reported having a Pap test during the last 3 years for
Connecticut 86.2 88.3
the States was 85.4.
Delaware 87.1 87.7
District of Columbia 93.9 90.8
Florida 85.9 85.3
Georgia 86.1 88.0
Hawaii 86.3 87.1
Idaho 82.5 79.3
Illinois 84.5 84.9
Indiana 80.9 82.4
Iowa 84.5 85.3
Kansas 85.6 87.4
Kentucky 84.3 83.4
Louisiana 84.8 85.4
Maine 85.0 89.5
Maryland 89.2 89.2
Massachusetts 87.7 86.2
Michigan 88.1 85.8
Minnesota 85.0 87.5
Mississippi 84.4 84.1
Missouri 85.4 84.6
Montana 80.6 85.2
Nebraska 82.4 84.8
Nevada 85.1 78.5
New Hampshire 84.2 88.4
New Jersey 81.4 84.3
New Mexico 82.6 83.7
Based on data from the BRFSS, 23 States attained
New York 83.8 84.2
the national target in both 1998 and in 1999.
North Carolina 87.7 90.9
North Dakota 85.5 86.6
Eight States attained the target in 1999 but not in 1998.
Ohio 84.2 85.5
Oklahoma 84.8 83.8
In 1999, there were 19 States where 78.5 to 84.9
Oregon 83.7 87.6
percent of women 18 and older reported having a Pap
Pennsylvania 83.8 83.6
test during the last 3 years.
Rhode Island 85.2 86.0
South Carolina 85.0 87.0
1
Data for California were not available in 1999.
South Dakota 86.3 85.4
Tennessee 86.1 85.1
SOURCE: CDC, NCCDPHP, Behavioral Risk Factor Surveillance System.
--- Data not available.
Texas 81.5 82.7
Utah 78.4 80.8
Vermont 86.0 88.4
Virginia 84.9 85.5
Washington 85.4 85.9
West Virginia 81.0 80.7
Wisconsin 83.4 84.7
Wyoming 82.0 83.8
States
23
8
19
54 Healthy People 2000 Final Review

Figure QQ. Objective 16.13--Percent of persons 50 years of age and older who have had a
fecal occult blood test during the last 2 years: United States and each State, 1997 and 1999
Percent of adults
50 years and older
1997 1999
United States (NHIS) --- 35.0
1
United States --
In 1998, based on data from the National Health
Interview Survey (NHIS), 35 percent of the population 50 years and
Median of States (BRFSS) 25.6 26.2
older had a fecal occult blood test during the last 2 years. The
Alabama 19.2 17.6
baseline in 1987 was 27 percent. Thirty-five percent of the change
Alaska 25.0 17.3
required to meet the year 2000 target was achieved.
Arizona 29.3 32.9
Arkansas 21.3 22.9
In 1999, based on data from the Behavioral Risk Factor Surveillance
California 24.8 23.0
System (BRFSS), the median percent of the population 50 years and
Colorado 32.2 29.7
older who had a fecal occult blood test during the last 2 years for
Connecticut 31.1 36.8
the States was 26.2.
Delaware 30.5 29.0
District of Columbia 37.8 43.0
Florida 30.1 32.4
Georgia 27.8 21.7
Hawaii 32.2 28.7
Idaho 25.4 23.1
Illinois 24.9 25.8
Indiana 22.2 22.4
Iowa 28.0 33.0
Kansas 31.5 26.1
States -- It is not appropriate to assess attainment of the national
Kentucky 26.1 23.9
target based on the BRFSS data because the NHIS and BRFSS are not
Louisiana 23.3 23.8
comparable and the national target was set based on data from the NHIS.
Maine 35.0 35.9
The twelve States in the highest quartile for the percent of the population
Maryland 32.1 37.8
50 years and older who reported having a fecal occult blood test during
Massachusetts 34.2 35.2
the last two years in 1999 are shown below.
Michigan 31.0 33.1
Minnesota 32.3 26.2
District of Columbia 43.0
Mississippi 13.5 17.8
New Hampshire 40.7
Missouri 25.1 25.0
North Carolina 38.5
Montana 24.0 26.4
Maryland 37.8
Nebraska 24.4 27.4
Vermont 37.4
Nevada 16.0 20.0
Connecticut 36.8
New Hampshire 33.9 40.7
Maine 35.9
New Jersey 28.5 35.4
Washington 35.7
New Mexico 21.7 23.9
New Jersey 35.4
New York 31.9 29.2
Massachusetts 35.2
North Carolina 36.5 38.5
Michigan 33.1
North Dakota 22.7 24.5
Iowa 33.0
Ohio 29.4 30.9
Oklahoma 14.3 23.3
SOURCE: CDC, NCCDPHP, Behavioral Risk Factor Surveillance System.
--- Data not available.
1998 data.
Oregon 34.6 32.2
Pennsylvania 28.8 31.0
Rhode Island 26.5 31.4
South Carolina 24.0 26.4
South Dakota 21.7 25.2
Tennessee 22.3 23.3
Texas 25.0 23.0
Utah 22.0 19.8
Vermont 35.1 37.4
Virginia 25.9 26.2
Washington 35.9 35.7
West Virginia 18.0 19.1
Wisconsin 25.4 21.3
Wyoming 19.3
1
18.4
Healthy People 2000 Final Review 55
Figure RR. Objective 16.13--Percent of persons 50 years of age and older who have ever had a
sigmoidoscopy or procotoscopic exam: United States and each State, 1997 and 1999
Percent of adults
50 and older
1997 1999
United States (NHIS) --- 37.0
1
United States --
In 1998, based on data from the National Health
Interview Survey (NHIS), 37 percent of the population 50 years and
Median of States (BRFSS) 40.8 43.6
older reported having ever had a proctosigmoidoscopy. The
Alabama 43.0 41.2
baseline in 1987 was 25 percent. Eighty percent of the change
Alaska 41.0 50.4
required to meet the year 2000 target was achieved.
Arizona 39.2 36.8
Arkansas 34.4 42.1
In 1999, based on data from the Behavioral Risk Factor Surveillance
California 46.6 48.2
System (BRFSS), the median percent of the population 50 years and
Colorado 43.5 44.2
older who ever had a sigmoidoscopy or proctoscopic exam for
Connecticut 45.0 48.7
the States was 43.6.
Delaware 47.8 56.0
District of Columbia 49.2 48.4
Florida 45.8 45.0
Georgia 48.1 47.4
Hawaii 51.6 44.2
Idaho 38.4 41.3
Illinois 40.3 43.9
Indiana 38.5 43.4
Iowa 40.7 46.4
Kansas 37.5 38.8
States --
It is not appropriate to assess attainment of the national
Kentucky 34.2 34.7
target based on the BRFSS data because the NHIS and BRFSS are not
Louisiana 37.0 36.7
comparable and the national target was set based on data from the NHIS.
Maine 42.4 42.4
The 12 States in the highest quartile for the percent of the population
Maryland 37.1 50.5
50 years and older who ever had a sigmoidoscopy or proctoscopic exam
Massachusetts 41.3 43.9
in 1999 are shown below.
Michigan 47.8 49.5
Minnesota 50.9 47.5
Delaware 56.0
Mississippi 35.8 37.4
Wisconsin 52.4
Missouri 41.1 38.0
Washington 50.8
Montana 39.7 43.2
Maryland 50.5
Nebraska 38.2 33.7
Alaska 50.4
Nevada 42.3 43.1
Michigan 49.5
New Hampshire 42.8 46.8
Connecticut 48.7
New Jersey 39.6 44.0
Rhode Island 48.7
New Mexico 39.4 43.1
District of Columbia 48.4
New York 43.7 42.2
California 48.2
North Carolina 39.2 39.1
Minnesota 47.5
North Dakota 44.9 42.0
Georgia
47.4
Ohio 37.5 40.7
Oklahoma 22.3 37.7
SOURCE: CDC, NCCDPHP, Behavioral Risk Factor Surveillance System.
---Data not available.
1998 data.
1
Oregon 46.1 47.1
Pennsylvania 40.2 39.7
Rhode Island 42.6 48.7
South Carolina 29.2 41.8
South Dakota 37.6 45.5
Tennessee 34.2 39.3
Texas 38.2 44.3
Utah 45.1 44.1
Vermont 40.2 42.2
Virginia 47.6 44.2
Washington 44.0 50.8
West Virginia 34.4 34.9
Wisconsin 50.3 52.4
Wyoming
45.3 46.5
56 Healthy People 2000 Final Review

Figure SS. Objective 17.9--Age-adjusted death rates for diabetes-related deaths: United States
and each State, 1997 and 1998
Age-adjusted
death rate
Target
per 100,000
status
1997 1998
(34.0)
United States 40.6 40.9
United States --
At baseline in 1986, the age-adjusted diabetes-
related death rate was 38 per 100,000 population. In 1998, the rate
Alabama 38.1 39.9
was 40.9. Instead of declining by 4 deaths per 100,000, the rate
Alaska 38.1 36.6
increased by 2.9 deaths per 100,000. In this case the rate moved
Arizona 29.3 30.6
away from the target.
Arkansas 33.2 34.5
California 44.9 45.8
Colorado 29.6 31.1
Connecticut 36.9 37.5
Delaware 48.4 48.5
District of Columbia 59.4 59.1
Florida 28.6 29.1
Georgia 38.7 39.1
Hawaii 41.4 42.2
Idaho 32.8 34.4
Illinois 40.5 40.6
Indiana 42.2 41.4
Iowa 36.7 37.1
Kansas 33.8 35.0
Kentucky 40.3 38.9
Louisiana 43.4 45.6
Maine 42.5 42.0
Maryland 54.3 54.2
Massachusetts 34.2 33.7
Michigan 43.3 43.8
Minnesota 34.4 34.8
Mississippi 51.0 51.3
Missouri 42.5 42.7
Montana 29.4 32.2
Nebraska 35.3 35.3
Nevada 31.9 33.6
New Hampshire 42.1 41.4
New Jersey 38.2 36.1
New Mexico 34.6 35.2
Five States attained the target in 1997 and 1998.
New York 35.5 35.2
North Carolina 49.7 52.0
One State attained the target only in 1998.
North Dakota 33.7 38.1
Ohio 52.3 51.2
In 1998, 16 States had rates between the year
Oklahoma 40.9 39.6
2000 target (34) and the 1987 baseline (38).
Oregon 39.3 42.8
Pennsylvania 46.1 45.3
In 1998, 29 States had rates greater than the baseline for
Rhode Island 45.3 43.7
the United States in 1987 (38).
South Carolina 49.0 49.0
South Dakota 32.4 36.4
SOURCE: CDC, NCHS, National Vital Statistics System.
Tennessee 42.2 44.4
Texas 46.1 45.9
Utah 33.8 36.1
Vermont 46.1 42.5
Virginia 39.6 38.7
Washington 35.4 36.1
West Virginia 54.3 56.1
Wisconsin 36.8 36.9
Wyoming 32.0 35.1
States
5
1
29
16
Healthy People 2000 Final Review 57

Figure TT. Objective 19.1--Reported incidence of gonorrhea per 100,000 population: United States
and each State, 1998 and 1999
Reported cases
per 100,000
Target
population
status
1998 1999
(100)
United States 131.6 132.0
United States --
In 1989, there were 300 gonorrhea cases per
100,000 population. The objective for the year 2000 was 100 cases
Alabama 292.7 249.2
per 100,000. In 1999 there were 133 cases per 100,000. Eighty-four
Alaska 53.9 48.7
percent of the target for the year 2000 had been achieved in 1999.
Arizona 90.2 89.8
Arkansas 155.7 126.4
California 59.7 56.3
Colorado 51.2 62.3
Connecticut 97.0 101.2
Delaware 209.3 220.6
District of Columbia 861.7 681.3
Florida 127.9 151.8
Georgia 270.4 272.8
Hawaii 42.4 39.1
Idaho 14.8 7.1
Illinois 180.4 191.7
Indiana 106.9 102.5
Iowa 56.5 47.6
Kansas 99.7 100.4
Kentucky 96.9 84.6
Louisiana 286.1 301.7
Maine 5.4 6.6
Maryland 219.2 201.7
Massachusetts 36.7 39.7
Michigan 166.6 161.3
Minnesota 57.3 59.3
Mississippi 388.4 376.0
Missouri 174.0 149.7
Montana 6.2 6.0
Nebraska 72.4 88.3
Nevada 82.7 72.0
New Hampshire 7.7 9.6
New Jersey 96.8 96.4
New Mexico 55.1 56.0
Twenty-six States attained the target in both 1998 and
New York 104.9 109.0
1999.
North Carolina 254.8 253.9
North Dakota 12.5 13.1
In 1999, 21 States had rates between the year 2000
Ohio 163.0 161.2
target (100) and the 1989 baseline (300).
Oklahoma 156.7 119.7
Oregon 26.8 27.2
In 1999, four States had rates greater than the baseline
Pennsylvania 97.6 110.8
for the United States in 1989 (300).
Rhode Island 43.5 60.7
South Carolina 301.7 387.0
SOURCE: CDC, NCHSTP, Sexually Transmitted Disease Surveillance System.
South Dakota 29.9 26.2
Tennessee 218.0 207.3
Texas 166.2 164.2
Utah 11.2 11.9
Vermont 6.4 8.8
Virginia 136.4 136.8
Washington 34.2 37.0
West Virginia 50.8 32.3
Wisconsin 121.6 126.9
Wyoming 7.5 9.0
States
26
21
4
58 Healthy People 2000 Final Review

Figure UU. Objective 19.3--Reported cases of primary and secondary syphilis per 100,000 population:
United States and each State, 1998 and 1999
Reported cases
per 100,000
Target
population
status
1998 1999
(4.0)
United States 2.6 2.4
United States --
In 1989, there were 18.1 cases of primary and
secondary syphilis per 100,000 population. The objective for the
Alabama 6.3 4.6
year 2000 was 4 cases per 100,000. In 1999, there were 2.5 cases
Alaska 0.2 0.2
per 100,000. More than 100 percent (111 percent) of the targeted
Arizona 4.0 4.4
change had been achieved in 1999.
Arkansas 4.3 3.4
California 0.9 0.9
Colorado 0.3 0.2
Connecticut 0.8 0.5
Delaware 2.8 1.3
District of Columbia 15.5 8.7
Florida 2.0 2.5
Georgia 4.4 5.5
Hawaii 0.3 0.3
Idaho 0.2 0.1
Illinois 3.5 3.5
Indiana 3.6 7.6
Iowa 0.2 0.3
Kansas 0.5 0.5
Kentucky 2.7 2.5
Louisiana 9.8 7.0
Maine 0.1 0.0
Maryland 12.6 6.6
Massachusetts 0.7 0.6
Michigan 2.1 2.5
Minnesota 0.2 0.2
Mississippi 9.5 7.0
Missouri 2.0 1.8
Montana 0.0 0.1
Nebraska 0.5 0.4
Nevada 0.9 0.3
New Hampshire 0.2 0.1
New Jersey 1.3 0.8
New Mexico 0.8 0.7
Thirty-eight States attained the target in both 1998 and 1999.
New York 0.7 0.8
North Carolina 9.6 6.1
North Dakota 0.0 0.0
One State attained the target only in 1999.
Ohio 1.2 0.8
Oklahoma 2.9 5.6
In 1999, 12 States had rates between the year 2000
Oregon 0.2 0.2
target (4.0) and the 1989 baseline (18.1).
Pennsylvania 0.8 0.7
Rhode Island 0.1 0.3
SOURCE: CDC, NCHSTP, Sexually Transmitted Disease Surveillance System.
South Carolina 7.1 6.9
South Dakota 0.1 0.0
Tennessee 10.4 11.7
Texas 2.2 2.4
Utah 0.2 0.1
Vermont 0.7 0.5
Virginia 2.2 2.2
Washington 0.8 1.3
West Virginia 0.2 0.3
Wisconsin 1.1 0.8
Wyoming
0.0 Quantity more than zero but less than 0.05.
0.2 0.0
States
38
1
12
Healthy People 2000 Final Review 59

Figure WW. Objective 20.1--Reported cases of measles: United States and each State, 1998 and 1999
Number of cases
1998 1999
United States 100 100
United States --
The United States did not attain the target of
zero cases of measles in 1998 or 1999. The number of measles
Alabama 1 0
cases was reduced by 97 percent from 3,396 in 1988 to 100 in both
Alaska 33 0
1998 and 1999.
Arizona 11 1
Arkansas 0 5
California 9 17
Colorado 0 0
Connecticut 0 2
Delaware 1 0
District of Columbia 0 0
Florida 2 2
Georgia 2 0
Hawaii 0 3
Idaho 0 0
Illinois 1 2
Indiana 3 2
Iowa 0 0
Kansas 0 0
Kentucky 0 2
Louisiana 0 0
Maine 0 0
Maryland 1 0
Massachusetts 2 8
Michigan 10 6
Minnesota 0 1
Mississippi 0 0
Missouri 0 0
Montana 0 0
Nebraska 0 0
Nevada 0 1
New Hampshire 0 1
New Jersey 8 0
New Mexico 0 0
Twenty States attained the target in both 1998 and 1999.
New York 4 5
North Carolina 1 0
Twelve States attained the target in 1999 but not in 1998.
North Dakota 0 0
Ohio 1 0
Nineteen States did not attain the target in 1998 or in 1999.
Oklahoma 0 0
Oregon 0 12
Pennsylvania 4 0
SOURCE: CDC, EPO, National Notifiable Disease Surveillance System.
Rhode Island 0 0
South Carolina 0 0
South Dakota 0 0
Tennessee 1 0
Texas 0 7
Utah 0 0
Vermont 1 0
Virginia 2 18
Washington 1 5
West Virginia 0 0
Wisconsin 1 0
Wyoming 0 0
States
20
12
19
Target
status
(0)
60 Healthy People 2000 Final Review

Figure YY. Objective 20.11--Estimated vaccination coverage with the 4DTP/3polio/1MMR series
among children ages 19-35 months: United States and each State, 1998 and 1999
Target
Percent
status
1998
1
1999
(90)
United States 80.6 79.9
United States --
In 1992, the vaccination coverage with the 4:3:1
series was 55 percent. In 1999, coverage had reached 80 percent,
Alabama 84.0 79.7
therefore, 71 percent of the targeted increase in immunization had
Alaska 82.7 82.2
been achieved in 1999.
Arizona 77.8 73.9
Arkansas 74.8 78.5
California 77.7 78.3
Colorado 78.2 77.2
Connecticut 90.7 87.1
Delaware 80.6 80.0
District of Columbia 73.7 78.5
Florida 80.9 82.0
Georgia 81.1 83.1
Hawaii 81.7 82.8
Idaho 76.4 70.0
Illinois 79.1 78.8
Indiana 78.9 75.4
Iowa 83.4 84.5
Kansas 83.6 79.7
Kentucky 83.0 88.6
Louisiana 79.7 76.9
Maine 89.0 84.1
Maryland 78.8 80.5
Massachusetts 87.4 87.3
Michigan 78.9 75.9
Minnesota 83.1 87.0
Mississippi 83.7 81.7
Missouri 85.8 75.5
Montana 82.8 84.8
Nebraska 78.0 83.7
Nevada 78.5 73.4
New Hampshire 85.1 84.5
New Jersey 85.0 80.9
New Mexico 73.3 75.6
Two States attained the target in 1999.
New York 85.7 83.4
North Carolina 84.1 81.8
Forty-nine States did not attain the target in 1998 and 1999
North Dakota 79.8 83.0
or in 1999.
Ohio 78.6 79.1
Oklahoma 78.5 74.0
SOURCE: CDC, NCHS and NIP, National Immunization Survey.
Oregon 75.5 73.2
Pennsylvania 84.0 86.6
Rhode Island 87.3 90.4
South Carolina 88.4 81.1
South Dakota 75.1 83.4
Tennessee 82.6 79.5
Texas 74.8 74.7
Utah 76.8 81.7
Vermont 87.3 90.7
Virginia 82.0 81.6
Washington 81.1 76.5
West Virginia 82.7 82.1
Wisconsin 79.3 85.4
Wyoming 80.4 83.5
1
4 or more doses of DTP/DT (diphtheria, tetanus, pertussis), 3 or more doses of polio, 1 or more doses of MMR (measles, mumps, rubella).
States
2
49
Healthy People 2000 Final Review 61

Figure ZZ. Objective 21.4--Percent of persons under 65 years of age with no health insurance coverage:
United States and each State, 1998 and 1999
Percent with no
health care
Target
coverage
status
1998 1999
(0)
United States (NHIS) 16.6 ---
United States --
In 1998, based on data from the National Health
Interview Survey (NHIS), 16.6 percent of the population under 65
United States (CPS) 18.4 17.4
years of age had no health insurance coverage (age adjusted). The
Alabama 19.5 16.2
baseline in 1989 was 15.7 percent. The proportion without health
Alaska 17.9 20.0
insurance coverage was greater in 1998 than it was in 1989.
Arizona 26.9 24.0
Arkansas 21.7 16.9
In 1999, based on data from the Current Population Survey (CPS),
California 24.4 22.3
17.4 percent of the population under 65 years of age had no health
Colorado 16.4 18.3
insurance coverage.
Connecticut 14.3 11.3
Delaware 17.1 12.8
District of Columbia 19.2 17.6
Florida 21.1 22.8
Georgia 19.4 17.9
Hawaii 11.3 12.3
Idaho 19.7 21.6
Illinois 16.6 15.7
Indiana 16.1 12.3
Iowa 10.9 9.5
Kansas 12.2 13.9
Kentucky 16.0 16.3
Louisiana 21.3 25.0
Maine 14.6 13.2
Maryland 18.9 13.5
Massachusetts 11.6 11.8
Michigan 14.9 12.4
Minnesota 10.3 8.9
Mississippi 22.9 18.9
Missouri 12.1 9.6
Montana 21.9 21.0
Nebraska 10.2 12.3
Nevada 23.7 22.8
New Hampshire 12.5 11.3
New Jersey 18.0 15.0
States
New Mexico 24.0 29.4
24
Based on the CPS data, 24 States had proportions of
New York 19.7 18.5
persons with no health insurance coverage greater
North Carolina 17.0 17.3
than the national target for the year 2000 (0) and
North Dakota 16.5 13.8
less than the national baseline in 1989 (15.7).
Ohio 11.7 12.5
Oklahoma 21.2 20.4
Oregon 16.0 16.4
27
In 1999, 27 States had proportions of persons
Pennsylvania 12.1 11.0
with no health insurance coverage greater than or equal
Rhode Island 7.6 8.1
to the national baseline in 1989 (15.7).
South Carolina 17.4 20.1
South Dakota 16.3 13.5
SOURCE: U.S. Census Bureau, Current Population Survey.
--Data not available.
Tennessee 14.3 12.6
Texas 26.9 25.7
NOTE: The NHIS data are age adjusted and refer to coverage at the time of the survey. The CPS
data are not age adjusted and refer to coverage during the previous calendar year. Despite these
differences, target attainment is assessed based on the CPS data because the objective was to
reduce the percent of the population without health insurance coverage to zero.
Utah 15.1 15.3
Vermont 11.0 13.6
Virginia 15.8 15.8
Washington 13.4 17.5
West Virginia 20.8 20.5
Wisconsin
Wyoming
13.2
18.8
12.2
17.8
62 Healthy People 2000 Final Review

Healthy People 2000 Priority Areas

Priority Area 1
Physical Activity and
Fitness
Background
The importance of physical activity,
fitness, and exercise spans a spectrum of
public health issues ranging from
disease prevention and enhancement of
a healthy lifestyle for all age groups to
the maintenance of an independent
lifestyle in the later stages of life (1).
Research has demonstrated that virtually
all individuals can benefit from regular
physical activity, whether they
participate in vigorous exercise or some
type of moderate health-enhancing
physical activity (2).
The importance of physical activity
in reducing morbidity and mortality
from chronic diseases has been well
established. A 1990 study showed that
14 percent of all deaths in the United
States were attributed to diet and
activity patterns (3). A more recent
study linked sedentary lifestyles to
23 percent of deaths from the major
chronic diseases (4). For example,
physical activity has been shown to
reduce the risk of developing or dying
from heart disease, diabetes, colon
cancer, and high blood pressure (2).
Some evidence suggests that physical
activity may also protect against lower
back pain and some forms of cancer (for
example, breast cancer) (5,6). On
average, people who are physically
active outlive those who are inactive (4,
7–10). However, most adults and many
children are not active enough to
achieve these health benefits, and the
prevalence of physical activity has
changed only slightly in the past decade
(2). Only 30 percent of adults spend
sufficient time in moderate activity and
only 14 percent are vigorously active at
the suggested levels.
The economic burden of physical
inactivity is substantial. One study
estimated that increasing regular
moderate physical activity among the
more than 88 million inactive Americans
over the age of 15 years might reduce
the annual national direct medical costs
by as much as $76.6 billion in 2000
dollars (11).
Data Summary
Highlights
More than one-half of the physical
activity and fitness objectives either
achieved or moved toward the year
2000 targets. The age-adjusted death
rate from coronary heart disease (1.1)
for the population as a whole declined
to a level that exceeded the year 2000
target, dropping from 156 deaths per
100,000 persons in 1987 to 87 per
100,000 in 1998. Among blacks, the
coronary heart disease death rate also
dropped but fell short of meeting the
year 2000 target.
Gains were made in increasing the
proportion of worksites that offer
physical fitness programs (1.10). By
1999, three of the four size groupings
for worksites (those with 50–99
employees, 100–249 employees, and
250–749 employees) had exceeded their
year 2000 targets, while worksites with
750 and more employees moved toward
but did not meet the target.
Participation in moderate (1.3)
physical activity five or more times per
week increased over the decade, meeting
the year 2000 target of 30 percent,
whereas the proportion of adults
participating in physical activity seven
or more times per week moved toward,
but did not meet the 30–percent target.
Regular participation in strengthening
and stretching exercises (1.6) among
students in grades 9–12 met the year
2000 target of 40 percent, while
participation among persons ages 18–64
years increased to 19 percent for weight
lifting and 32 percent for stretching. A
small improvement was also seen in the
proportion of persons participating in
vigorous (1.4) physical activity.
Some objectives moved away from
the year 2000 targets. The prevalence of
overweight (1.2) increased across all
population groups with the greatest
increase occurring among adolescents
ages 12–19 years. The proportion of
both male and female adults reporting
the use of sound weight loss practices
(1.7) decreased as did the proportion of
students participating in daily physical
education at school (1.8), and the
number of people 70 years and over
(1.13) needing help with self care.
Summary of Progress
Of the 13 objectives that address
physical activity and fitness, 1 objective
(1.1) surpassed the year 2000 target.
Another six objectives (1.3, 1.4, 1.6,
1.9, 1.10, and 1.12) moved toward their
targets, and four objectives (1.2, 1.7,
1.8, and 1.13) moved away from their
targets. One objective (1.5) showed no
change, and data are not available to
measure progress for the remaining
objective (1.11). See table 1 for the
tracking data for the objectives in this
priority area and figure 1 for a
quantitative assessment of progress.
Discussion
While 7 of the 13 objectives related
to physical activity and fitness showed
improvement by the end of the decade,
greater progress could be realized if
certain barriers are overcome. Moving
the Nation’s population into a more
active lifestyle is complicated and
requires changes at many levels
including individual behavior,
environmental features, policy issues,
social norms, and family supports. For
example, in spite of evidence that
suggests active children are likely to
become active adults, schools no longer
require daily physical education. School,
however, may be the best opportunity
for children to be active because most
children do not walk or ride a bike to
school and many spend the time after
school in sedentary activities (for
example, homework or watching
television). Similarly, worksites that
provide facilities and other supports for
an active lifestyle may offer the best
opportunities for adults.
Because physical inactivity is a risk
factor for many diseases and conditions,
making physical activity an integral part
of daily life is crucial. To achieve the
physical activity and fitness objectives,
public health programs will need to
develop comprehensive approaches that
provide for participation in physical
activities at many levels. Such
approaches should include
environmentally based strategies aimed
at increasing access to facilities as well
as efforts that promote family and
community activities (for example, at
schools, worksites, and communities).
Transition to Healthy People
2010
In Healthy People 2010, the focus
of the physical activity and fitness
objectives has been expanded to include
a broader range of activities than those
Healthy People 2000 Final Review 65
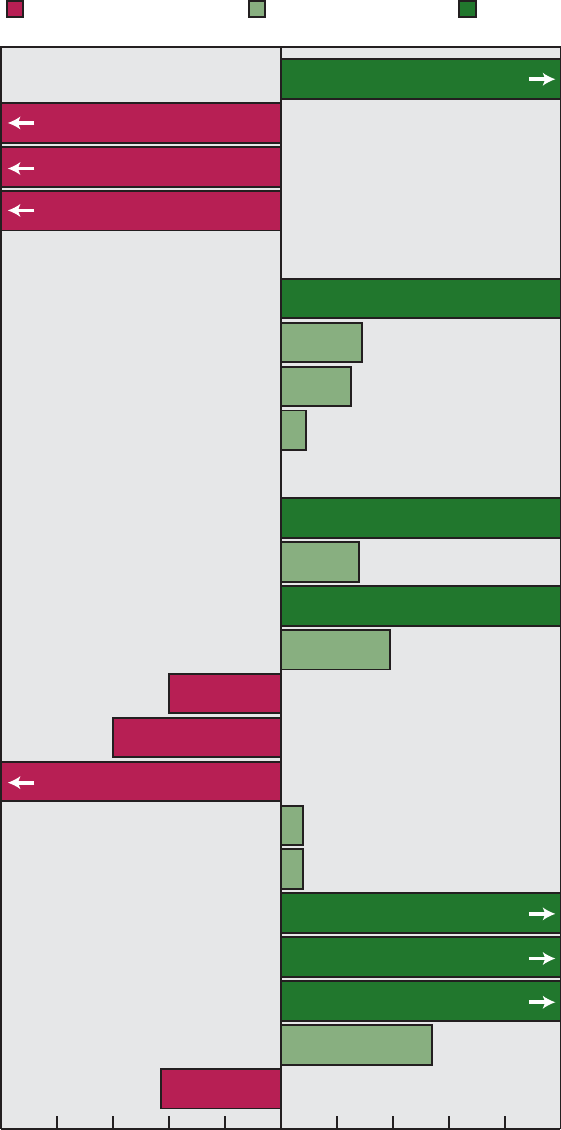
-100 -80 -60 -40 -20 0
Percent of target achieved
20 40 60 80 100
1.1 Coronary heart disease deaths
1.2 Overweight prevalence: 20-74 years
Male
Female
Adolescents 12-19 years
1.3 Light-to-moderate physical activity:
5 or more times per week
7 or more times per week
1.4 Vigorous physical activity 18 years and over
Students in grades 9-12
1.5 Sedentary lifestyle
1.6 Strengthening exercises: Grades 9-12
People 18-64 years
Stretching exercises: Grades 9-12
People 18-64 years
1.7 Weight loss practices: Overweight males
Overweight females
1.8 Daily school physical education: Grades 9-12
1.9 Active physical education: 21 or more minutes,
3-5 times per week
30 or more minutes, 1 time per week
1.10 Worksite fitness: 50-99 employees
100-249 employees
250-749 employees
750 or more employees
1.13 Difficulty with self-care: 70 years and over
Moved away from target Moved toward target Met target
* This objective has moved away from its target. A progress quotient could not be calculated.
NOTE: Complete tracking data are shown in table 1. Progress quotients are not calculated for objectives 1.11 and 1.12.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
109%
-150%
-250%
-143%
-40%
-43%
54%
8%
28%
29%
25%
Away*
No change
9%
39%
8%
-60%
-163%
100%
100%
100%
400%
158%
133%
66 Healthy People 2000 Final Review
Figure 1. Final status of Physical Activity and Fitness objectives
presented in Healthy People 2000.To
avoid duplication of objectives across
chapters, some topics previously
included in the Healthy People 2000
Physical Activity and Fitness chapter are
only in the Healthy People 2001 focus
areas that were considered their
‘‘primary homes’’ (for example, the
objectives for coronary heart disease
deaths and overweight prevalence are
located only in the Heart Disease and
Stroke and the Nutrition and Overweight
chapters, respectively).
The Healthy People 2010 Physical
Activity and Fitness chapter is organized
into four sections: physical activity in
adults, muscular strength/endurance and
flexibility, physical activity in children
and adolescents, and access. The
Healthy People 2010 objectives continue
to reflect the importance of moderate
activity in achieving and maintaining a
healthy lifestyle. For example, the 2010
objectives include measures of walking
and bicycling, and public access to local
school facilities after hours. These
objectives replace the unmeasured
Healthy People 2000 objective assessing
community fitness facilities (1.11)by
encouraging a combination of individual
behavior (walking or bicycling) and
community support for activity.
Attention on activity patterns among
children and adolescents is highlighted
in its own section, which includes
objectives on moderate and vigorous
physical activity, as well as school-based
policy objectives. A new objective
calling for a limit on television viewing
has been included in Healthy People
2010. This emphasis on children and
adolescents is particularly appropriate
considering that the health benefits of
physical activity are not limited to
adults and that patterns adopted early in
life may continue into adulthood.
The objective on physician
counseling regarding exercise (1.12) has
been combined with other counseling
behaviors to create a new objective in
the Healthy People 2010 chapter on
Access to Quality Health Services.
Likewise, a Healthy People 2010
objective on the importance of teaching
physical activity in health classes is in
the Educational and Community-Based
Programs chapter. Because physical
activity affects many other health
conditions, related objectives have been
identified from 23 other chapters.
Physical activity is the topic of 1 of
the 10 Leading Health Indicators (LHIs),
which Healthy People 2010 introduces
to serve as a barometer of the Nation’s
health. Two objectives from the Healthy
People 2010 Physical Activity and
Fitness focus area—the proportion of
adolescents who engage in vigorous
physical activity that promotes
cardiorespiratory fitness 3 or more days
per week for 20 or more minutes per
occasion and the proportion of adults
who engage regularly, preferably daily,
in moderate physical activity for at least
30 minutes per day—are used to
measure this LHI.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the changes between the
two decades of objectives, reflecting
new knowledge and direction for this
area.
Data Issues
Definitions
Operational definitions and data
collection specifications for all Healthy
People 2000 objectives in Priority Area
1 have been published in the National
Center for Health Statistics’ Healthy
People Statistical Notes series (12). Data
issues are discussed and references are
cited for expanded discussions of the
data systems that provide data for the
national objectives. Where appropriate,
the text of the questionnaire items used
to measure the objectives is also
provided. See the Appendix for further
information.
Coronary heart disease deaths (1.1)
are defined by ICD–9 codes 402,
410–414, and 429.2. These codes are
different from the codes used to define
the category ‘‘Diseases of heart,’’ which
often appears in published tables (see
the Appendix and Appendix table II).
When the Healthy People 2000
objectives were released in 1990 (13),
physical activity as a recognized benefit
for health outcomes was still a relatively
new concept; this newness contributed
to difficulties in tracking some
objectives with appropriate operational
definitions. Calculations vary from
simple counts (for example,
weight-training three or more times a
week) to complex formulas (for
example, calculating average kilocalories
expended per kilogram per day) (14).
Because of the growing appreciation of
the benefits of regular physical activity,
the intent of objective 1.3
(light-to-moderate physical activity) was
to generate calorie-burning activity by
emphasizing the importance of regular
physical activity that can be sustained
throughout the lifespan. The sum of all
physical activities performed at least 30
minutes per occasion five or more or
seven or more times a week regardless
of the intensity has been defined as
measuring this objective.
To measure the proportion of adults
performing vigorous physical activity
(1.4), the predicted maximum
cardiorespiratory capacity was estimated
using age- and sex-based regression
equations and then multiplying by
50 percent. Next, all the activities that
were performed for at least 20 minutes
that had a kilocalorie value that was
equal to or greater than that 50 percent
level were counted (15,16). The
estimated number of people who
exercise vigorously were respondents
who performed these activities three or
more times per week.
Overweight (1.2) for adults is
defined as a body mass index (BMI) at
or above the sex-specific 85th percentile
of the 1976–80 Second National Health
and Nutrition Examination Survey
(NHANES II) reference population
20–29 years of age. For adolescents,
overweight is the sex- and age-specific
85th percentile from NHANES II. BMI
cutoff points for adults are 27.8
kilograms per meter squared for males,
and 27.3 kilograms per meter squared
for females. Healthy People 2010
defines the upper limit of the healthy
weight range as a BMI of less than 25
because chronic disease risk increases in
most populations at or above this
cut-point (1). See the Nutrition chapter
(Priority Area 2) for additional
information.
The 1984–85 baseline figures for
objective 1.13 were derived by
combining estimates for the
noninstitutionalized population from the
1984 National Health Interview Survey
(NHIS) Supplement on Aging (SOA)
with data for those in nursing homes
from the 1985 National Nursing Home
Survey (NNHS). The 1984 SOA asked
about seven specific personal care
activities, also referred to as activities of
daily living (ADLs) for persons 65 years
and over. Because of the way the
questions were asked on the NNHS,
only five ADLs (bathing, dressing, using
the toilet, getting in and out of bed or
chair, and eating) were used for tracking
this objective. The numerator included
respondents to the SOA who said they
Healthy People 2000 Final Review 67
had ‘‘any difficulty’’ performing at least
two ADLs combined with patients for
whom administrators reported to the
NNHS as ‘‘receiving assistance’’ with at
least two ADLs. The denominator for
the baseline was the civilian,
noninstitutionalized population 65 years
and over plus the nursing home
population 65 years and over. The
update for this objective is derived from
combined data from the 1994 NHIS
Second Supplement on Aging (SOA II)
and data from the 1995 NNHS using the
same questions as were used for the
baseline. However, the 1994–95 update
is for persons 70 years and over. The
1984–85 data were also computed for
ages 70 years and over to provide a
comparison with the 1994–95 update.
Data Comparability
Overweight (1.2) was tracked with
two data sources. The primary data
source is the NHANES, which provided
baseline data for most of the overweight
objectives and the 1988–94 updates;
these data are derived from measured
height and weight. The second data
source is the NHIS. This survey
provides interim estimates shown in an
earlier publication (17), updates for
Hispanic females and American
Indians/Alaska Natives, and all data for
people with disabilities. NHIS estimates
are based on self-reported heights and
weights and are not comparable with the
actual measured data from NHANES:
Prevalence estimates of overweight from
self-reported height and weight are
lower. Trends from the NHIS self-report
measures, like those from NHANES,
show a steady increase in prevalence of
overweight; this increase, however, is
different in magnitude from that
observed in the data derived from
measured height and weight.
Objective 1.3 (light-to-moderate
physical activity) was tracked with the
NHIS. The list of activities asked on the
NHIS has not been identical from year
to year. The 1985 and 1990 surveys did
not ask about some activities for people
65 years and over; thus, the data shown
are for people 18–64 years of age. The
1991, 1995, and 1998 surveys asked
about some different activities than the
previous surveys, but people of all age
groups were asked the same questions.
Because of these differences, data from
1985 and 1990 are not comparable to
later data.
The 1985 and 1992 data for
objective 1.10 are from the National
Survey of Worksite Health Promotion
Activities, which were telephone surveys
of nongovernment worksites. Worksites
were sampled because different
worksites within the same company
could have different sets of health
promotion activities. Both active
methods (such as classes) and passive
methods (such as brochures) were
counted as worksite health promotion
activities. The 1995 update is from the
CDC-sponsored Worksite Benchmark
Survey, which used a methodology very
similar to the 1992 survey but did not
include passive methods of health
promotion (18,19). The 1998–99 data
are from the National Worksite Health
Promotion Survey, which randomly
sampled nongovernmental worksites of
50 or more employees in a particular
geographic location. This telephone
survey included questions regarding the
offering of some types of physical
activity or fitness facility for employees
at the workplace or through the
company’s health plans (20). The
1998–99 data are not comparable to the
data that were collected earlier, although
similar questions were asked.
Data for objective 1.12, clinician
counseling about physical activity, were
obtained from three different surveys,
making statements about trends
problematic. The 1988 baseline of
30 percent from the American College
of Physicians (ACP) survey was a
random stratified sample of ACP
members drawn from 21 geographic
regions yielding an initial sample of
1,251 internists. The sampling frame for
internists in the 1992 Primary Care
Provider Surveys (PCPS) also contained
a random stratified sample of ACP
members but was drawn from four
geographic regions with oversampling of
female members, yielding an initial
sample of 1,200 internists. Additional
provider groups sampled in the 1992
PCPS included pediatricians, nurse
practitioners, obstetricians/gynecologists,
and family physicians. Response rates
varied from 50 to 80 percent across the
groups. The data on inquiry for
objective 1.12 refer to the proportion of
health care providers who routinely
provided service to 81–100 percent of
their clients. Data on formulation of an
exercise plan represent the proportion of
providers who routinely provide this
service to 81–100 percent of their clients
who need this intervention. Because
inquiry and counseling services are not
reimbursable, documenting their
provision has proven to be difficult.
The Prevention in Primary Care
Study (PPCS) was conducted in
1997–98 to update data from the PCPS.
The design and items included in the
1997–98 study were similar to the
PCPS, but a slightly different sampling
frame was used, and some items
included in the 1992 surveys were not
included in the PPCS. The professionals
were sampled from listings of all
licensed, active practitioners in the
United States whose practices were at
least 50 percent primary care. Because
of low response rates from the other
provider groups, updates are available
only for nurse practitioners.
Proxy Data
Regular performance of physical
activities that enhance and maintain
muscular strength, muscular endurance,
and flexibility (1.6) generally requires
participation in a variety of physical
activities because not all activities will
satisfy all three factors. Because scoring
parameters for strength, endurance, and
flexibility are not yet available, this
objective has been tracked using data
from the NHIS on an activity that
increases muscular strength
(weightlifting) and an activity that
increases flexibility (stretching). The
data shown for students in grades 9–12
are from the Youth Risk Behavior
Surveillance System (YRBSS) and are
based on self-reported participation in
stretching exercises or strengthening
exercises that were done four or more
days per week.
Objective 1.7 is to increase to at
least 50 percent the proportion of
overweight people who use sound
dietary practices combined with regular
physical activity to attain appropriate
body weight. Respondents to the NHIS
identified as overweight based on their
self-reported heights and weights who
reported they were currently trying to
lose weight or control their weight by
eating fewer calories and exercising
more were defined as using sound
dietary practices for this objective.
However, an assessment of the quality
of dietary practices was not coupled
with a measure of regular physical
activity. The 1985 and 1990 NHIS
questionnaires asked respondents
specifically if they were eating fewer
calories to lose weight and if they were
increasing their physical activity to lose
weight. In 1991, 1993, 1995, and 1998,
68 Healthy People 2000 Final Review
eating fewer calories and exercising
more were among a list of 10 possible
methods of losing weight in response to
the question, ‘‘Are you currently doing
any of these things to control your
weight?’’ Respondents were asked this
question if they reported they were
trying to lose weight or stay about the
same.
Objective 1.9 targets time spent in
school physical education classes
devoted to activities that may be readily
carried into adulthood because their
performance requires only one or two
people (such as swimming, bicycling,
jogging, and racquet sports). The proxy
measure for this objective is the percent
of class time spent in actual physical
activity. The 1983 baseline data show
the percent of physical education class
time spent being physically active for all
students. The YRBSS data for students
in grades 9–12 show the percent who
exercised in physical education class 21
or more minutes 3–5 times a week and
30 or more minutes 1 or more times per
week. The 1994 data from the School
Health Policies and Programs Study
(SHPPS) show the proportion of
physical education teachers using class
time to involve students in actual
physical activities.
References
1. Department of Health and Human Services.
Healthy people 2010. 2d ed. With understanding
and improving health and objectives for improving
health. 2 vols. Washington: U.S. Government
Printing Office. 2000.
2. Department of Health and Human Services.
Physical activity and health: A report of the
Surgeon General. Atlanta, GA: Centers for Disease
Control and Prevention, National Center for
Chronic Disease Prevention and Health Promotion.
1996.
3. McGinnis JM, Foege WH. Actual causes of
death in the United States. JAMA 270(18):207–12.
1993.
4. Kujala UM, Kaprio J, Sarna S, et al.
Relationship of leisure-time physical activity and
mortality: The Finnish twin cohort. JAMA
279(6):440–4. 1998.
5. Frost H, Moffett JAK, Moser JS, et al.
Randomized controlled trial for evaluation of
fitness programme for patients with chronic low
back pain. British Med J 310:151–154. 1995.
6. McTiernan A, Stanford JL, Weiss NS, et al.
Occurrence of breast cancer in relation to
recreational exercise in women age 50–64 years.
Epidemiology 7(6):598–604. 1996.
7. Paffenbarger RS, Hyde RT, Wing AL, et al. The
association of changes in physical-activity level
and other lifestyle characteristics with mortality
among men. N Engl J Med 328(8):538–45. 1993.
8. Sherman SE, D’Agostino RB, Cobb JL, et al.
Physical activity and mortality in women in the
Framingham Heart Study. Am Heart J
128(5):879–84. 1994.
9. Kaplan GAA, Strawbridge WJ, Cohen RD, et
al. Natural history of leisure-time physical activity
and its correlates: Associations with mortality from
all causes and cardiovascular disease over 28
years. Am J Epid 144(8):793–97. 1996.
10. Kushi LH, Fee RM, Folsom AR, et al.
Physical activity and mortality in postmenopausal
women. JAMA 277:1287–92. 1997.
11. Pratt M, Macera CA, Wang G. Higher direct
medical costs associated with physical inactivity.
The Physician and Sportsmedicine 28:63–70.
2000.
12. Chong Y, Klein RJ, Plepys C, et al.
Operational definitions for year 2000 objectives:
Priority Area 1, physical activity and fitness.
Healthy people statistical notes; no 18. Hyattsville,
Maryland: National Center for Health Statistics.
1998.
13. Department of Health and Human Services.
Healthy people 2000: National health promotion
and disease prevention objectives. Washington:
Public Health Service. 1990.
14. Schoenborn CA. Health habits of U.S. adults,
1985: The ‘‘Alameda 7’’ revisited. Public Health
Rep 101:571–8. 1988.
15. Stephens T, Craig CL. Fitness and activity
measurement in the 1981 Canada Fitness Survey.
In Drury TF, ed. Assessing physical fitness and
physical activity in population based surveys.
Hyattsville, Maryland: National Center for Health
Statistics. Pp. 401–32. 1989.
16. Ainsworth BE, et al. Compendium of physical
activities: Classification of energy costs of human
physical activities. Med and Sci and Sports and
Exercise 25(1):71–80. 1993.
17. National Center for Health Statistics. Healthy
people 2000 review, 1992. Hyattsville, Maryland:
Public Health Service. 1993.
18. Department of Health and Human Services.
Business Responds to AIDS Benchmark Survey:
Technical report. Atlanta: Public Health Service,
Centers for Disease Control and Prevention. 1996.
19. Centers for Disease Control and Prevention.
Cancer screening offered by worksites—United
States, 1992 and 1995. MMWR 46(19):421–4.
1997.
20. Department of Health and Human Services.
Tracking healthy people 2010. Washington: U.S.
Government Printing Office. 2000.
Healthy People 2000 Final Review 69

Table 1. Physical Activity and Fitness Objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
1.1* Coronary heart disease deaths (age adjusted
per 100,000) ................................ 1987 135 122 118 114 114 110 108 105 100 97 - - - 100
a. Black .................................... 1987 168 158 156 151 154 147 147 140 136 133 - - - 115
1.2* Overweight prevalence (based on measured height
and weight unless otherwise indicated)
Adults 20–74 years ............................ 1976–80 26% - - - - - - - - - - - -
1,2
35% --- --- --- --- --- 20%
Male ..................................... 1976–80 24% - - - - - - - - - - - -
1,2
34% --- --- --- --- --- 20%
Female ................................... 1976–80 27% - - - - - - - - - - - -
1,2
37% --- --- --- --- --- 20%
Adolescents 12–19 years ....................... 1976–80 15% - - - - - - - - - - - -
1
24% --- --- --- --- --- 15%
a. Low-income female 20–74 years............... 1976–80 37% - - -
3
47% --- --- --- --- --- --- --- --- 25%
b. Black female 20–74 years.................... 1976–80 44% - - - - - - - - - - - -
1,2
52% --- --- --- --- --- 30%
c. Hispanic female 20–74 years ................. ... --- --- --- --- --- --- --- --- --- --- --- 25%
Hispanic female 20 years and over ........... ... --- 33% 32% 32% 33% 32% 35% ---
‡
44%
‡
46% --- ...
Mexican American female 20–74 years ........ 1982–84 39% - - - - - - - - - - - -
1,2
50% --- --- --- --- --- ...
Cuban female 20–74 years................. 1982–84 34% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Puerto Rican female 20–74 years ............ 1982–84 37% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
d. American Indian/Alaska Native 20 years and over. . 1984–88 29–75% - - -
4
40%
4
36%
4
48%
4
34%
4
43% - - -
‡,4
46%
‡,4
45% - - - 30%
e. People with disabilities 20 years and over
4
....... 1985 36% - - - 38% 37% 38% 38% 40% - - - - - - - - - - - - 25%
f. Females with high blood pressure 20–74 years . . . 1976–80 50% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 41%
g. Males with high blood pressure 20–74 years...... 1976–80 39% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 35%
h. Mexican American male 20–74 years ........... 1982–84 30% - - - - - - - - - - - -
1,2
37% --- --- --- --- --- 25%
1.3* Light-to-moderate physical activity
People 6 years and over........................ ... --- --- --- --- --- --- --- --- --- --- --- 30%
People 18 years and over
5 or more times per week ..................... 1985
5
22%
5
23%
6
24% --- --- --- 23% --- --- 30% --- 30%
7 or more times per week ..................... 1985
5
16%
5
16%
6
17% --- --- --- 16% --- --- 20% --- 30%
a. Hispanic 18 years and over
5 or more times per week................... 1991 20% . . . . . . - - - - - - - - - 22% - - - - - - 29% - - - 25%
1.4 Vigorous physical activity
Children and adolescents 6–17 years .............. ... --- --- --- --- --- --- --- --- --- --- --- 75%
Children and adolescents 10–17 years ............. 1984
§
59% --- --- --- --- --- --- --- --- --- --- 75%
Students in grades 9–12 ........................ ... --- --- 64% --- 66% --- 64% --- 64% --- 65% 75%
People 18 years and over ....................... 1985
5
12% - - -
6
16% --- --- --- 16% --- --- 14% --- 20%
a. Lower-income people 18 years and over......... 1985
5
7% ---
6
15% --- --- --- 14% --- --- 10% --- 12%
b. Black 18 years and over ..................... 1991
§
13% ... ... --- --- --- 13% --- --- 11% --- 17%
c. Hispanic 18 years and over................... 1991
§
14% ... ... --- --- --- 14% --- --- 11% --- 17%
1.5 Sedentary lifestyle
People 6 years and over........................ ... --- --- --- --- --- --- --- --- --- --- --- 15%
People 18 years and over ....................... 1985 24% 26% 24% - - - - - - - - - 23% - - - - - - 24% - - - 15%
a. People 65 years and over.................... 1985 43% - - - 29% - - - - - - - - - 27% - - - - - - 20% - - - 22%
b. People with disabilities ...................... 1985 35% 34% 30% - - - - - - - - - 29% - - - - - - - - - - - - 20%
c. Lower-income people ....................... 1985 32% 33% 32% - - - - - - - - - 28% - - - - - - 29% - - - 17%
d. Black 18 years and over ..................... 1991 28% . . . . . . - - - - - - - - - 28% - - - - - - 29% - - - 20%
See footnotes and key at end of table.
70 Healthy People 2000 Final Review

Table 1. Physical Activity and Fitness Objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
e. Hispanic 18 years and over .................. 1991 34% . . . . . . - - - - - - - - - 31% - - - - - - 36% - - - 25%
f. American Indian/Alaska Native 18 years and over . . 1991 29% . . . . . . - - - - - - - - - 23% - - - - - - 26% - - - 21%
1.6 Muscular strength, endurance, and flexibility
People 6 years and over........................ ... --- --- --- --- --- --- --- --- --- --- --- 40%
Students in grades 9–12
Strengthening 4 or more times per week .......... ... --- --- 37% --- 39% --- 38% --- 38% --- 40% ...
Stretching 4 or more times per week ............. ... --- --- 43% --- 44% --- 41% --- 40% --- --- ...
People 18–64 years
Weightlifting................................ 1990 11% . . . 16% - - - - - - - - - 18% - - - - - - 19% - - - 40%
Stretching ................................. ... --- --- 27% --- --- --- 32% --- --- 32% --- ...
1.7* Sound weight loss practices among overweight
people 12 years and over ..................... ... --- --- --- --- --- --- --- --- --- --- --- 50%
Overweight male 18 years and over ............... 1985 25% 22% 19% - - - 17% - - - 15% - - - - - - 15% - - - 50%
Overweight female 18 years and over .............. 1985 30% 29% 22% - - - 19% - - - 19% - - - - - - 18% - - - 50%
a. Overweight Hispanic male 18 years and over ..... 1991 15% . . . . . . - - - 11% - - - 13% - - - - - - 12% - - - 24%
b. Overweight Hispanic female 18 years and over.... 1991 13% . . . . . . - - - 16% - - - 16% - - - - - - 12% - - - 22%
1.8 Daily school physical education
Students in grades 1–12 ........................ 1984–86 36% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 50%
Students in grades 9–12 ........................ ... --- --- 42% --- 34% --- 25% --- 27% --- 29% ...
Students in middle/junior high schools
For 1 year ................................. ... --- --- --- --- --- 12% --- --- --- --- --- ...
For 2 or more years.......................... ... --- --- --- --- --- 30% --- --- --- --- --- ...
Students in senior high schools
For 1 year ................................. ... --- --- --- --- --- 29% --- --- --- --- --- ...
For 2 years ................................ ... --- --- --- --- --- 18% --- --- --- --- --- ...
For 3 years ................................ ... --- --- --- --- --- 9% --- --- --- --- --- ...
1.9 Active physical education class time
All students.................................. 1983 27% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 50%
Students in grades 9–12
21 or more minutes, 3–5 times per week .......... ... --- --- 37% --- 35% --- 33% --- 32% --- 38% ...
30 or more minutes, 1 or more times per week ..... ... --- --- 24% --- 25% --- 22% --- 21% --- 26% ...
Physical education teachers devoting more than one
class period to:
Jogging ................................... ... --- --- --- --- --- 47% --- --- --- --- --- ...
Tennis .................................... ... --- --- --- --- --- 30% --- --- --- --- --- ...
Aerobic dance .............................. ... --- --- --- --- --- 30% --- --- --- --- --- ...
Walking ................................... ... --- --- --- --- --- 15% --- --- --- --- --- ...
Swimming ................................. ... --- --- --- --- --- 14% --- --- --- --- --- ...
1.10 Worksite fitness programs
50–99 employees ............................. 1985 14% - - - - - - 33% - - - - - - - - - - - - - - - - - -
7
38% 20%
100–249 employees ........................... 1985 23% - - - - - - 47% - - - - - - - - - - - - - - - - - -
7
42% 35%
See footnotes and key at end of table.
Healthy People 2000 Final Review 71

Table 1. Physical Activity and Fitness Objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
250–749 employees ........................... 1985 32% - - - - - - 66% - - - - - - - - - - - - - - - - - -
7
56% 50%
750 and more employees ....................... 1985 54% - - - - - - 83% - - - - - - - - - - - - - - - - - -
7
68% 80%
Group classes, workshops, or lectures ............. ... ... --- --- 21% --- --- 19% --- --- --- --- 80%
1.11 Community fitness facilities
Hiking, biking, and fitness trail miles ............... 1986 1 per
71,000
people
--- --- --- --- --- --- --- --- --- --- 1per
10,000
people
Public swimming pools ......................... 1986 1 per
71,000
people
--- --- --- --- --- --- --- --- --- --- 1per
25,000
people
Acres of park and recreation open space ........... 1986 1.8 per
1,000
people
--- --- --- --- --- --- --- --- --- --- 4per
1,000
people
1.12 Clinician counseling about physical activity
Percent of sedentary patients .................... 1988 30% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 50%
Percent of clinicians routinely providing services
to 81–100% of patients
Inquiry about exercise habits
Pediatricians ............................... ... --- --- --- 16% --- --- --- ---
8,9
--- --- --- 50%
Nurse practitioners .......................... ... --- --- --- 30% --- --- --- ---
8
41% --- --- 50%
Obstetricians/gynecologists .................... ... --- --- --- 14% --- --- --- ---
8,9
--- --- --- 50%
Internists .................................. ... --- --- --- 40% --- --- --- ---
8,9
--- --- --- 50%
Family physicians ........................... ... --- --- --- 19% --- --- --- ---
8,9
--- --- --- 50%
Formulation of an exercise plan
Pediatricians ............................... ... --- --- --- 16% --- --- --- ---
8,9
--- --- --- 50%
Nurse practitioners .......................... ... --- --- --- 14% --- --- --- ---
8
27% --- --- 50%
Obstetricians/gynecologists .................... ... --- --- --- 13% --- --- --- ---
8,9
--- --- --- 50%
Internists .................................. ... --- --- --- 25% --- --- --- ---
8,9
--- --- --- 50%
Family physicians ........................... ... --- --- --- 18% --- --- --- ---
8,9
--- --- --- 50%
1.13* People with difficulty performing self-care activities
(per 1,000)
People 65 years and over ....................... 1984–85 111 - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 90
People 70 years and over ....................... 1984–85
§
141 --- --- --- --- ---
10
163 --- --- --- --- 90
a. People 85 years and over.................... 1984–85 371 - - - - - - - - - - - - - - -
10
471 --- --- --- --- 325
b. Black 65 years and over ..................... 1984–85
§
132 --- --- --- --- --- --- --- --- --- --- 98
Black 70 years and over ..................... 1984–85
§
166 --- --- --- --- ---
10
218 --- --- --- --- 98
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
‡
The NHIS was redesigned in 1997. Data may not be comparable with previous years; see Appendix.
1
1988–94 data.
2
Data are for persons 20 years and over.
3
1988–91 data.
4
Estimate derived from self-reported height and weight.
Met
Toward
Mixed/ no change
Away
Cannot assess
72 Healthy People 2000 Final Review

5
Data are for persons 18–64 years.
6
Operational definition changed from previous tracking data.
7
1998–99 data for percent of worksites that offered physical activity and/or fitness programs at the worksite or through their health plans.
8
1997–98 data.
9
Response rate for this group was too low to produce reliable estimates.
10
1994–95 data.
NOTE: Data include revisions and, therefore, may differ from data previously published in these reports and other publications.
Objective number Data source
1.1*,1.1a National Vital Statistics System, CDC, NCHS.
1.2*, 1.2a, b, f, g National Health and Nutrition Examination Survey, CDC, NCHS.
1.2c, h Data for Hispanic: National Health Interview Survey, CDC, NCHS.
Baselines for Mexican American, Cuban, Puerto Rican: Hispanic Health and Nutrition Examination Survey, CDC, NCHS.
Updates for Mexican American: National Health and Nutrition Examination Survey, CDC, NCHS.
1.2d Baseline: IHS, OPEL.
Updates: National Health Interview Survey, CDC, NCHS.
1.2e National Health Interview Survey, CDC, NCHS.
1.3*, 1.3a National Health Interview Survey, CDC, NCHS.
1.4 Baseline: For ages 10–17, National Children and Youth Fitness Study I, OPHS, ODPHP.
1991–99 for grades 9–12: Youth Risk Behavior Survey, CDC, NCCDPHP.
Baseline and updates for ages 18 and over: National Health Interview Survey, CDC, NCHS.
1.4a–c National Health Interview Survey, CDC, NCHS.
1.5, 1.5a–f National Health Interview Survey, CDC, NCHS.
1.6 For students in grades 9–12: Youth Risk Behavior Survey, CDC, NCCDPHP.
For people 18–64: National Health Interview Survey, CDC, NCHS.
1.7*, 1.7a–b National Health Interview Survey, CDC, NCHS.
1.8 Baseline for grades 5–12: National Children and Youth Fitness Study I, OPHS, ODPHP.
Baseline for grades 1–4: National Children and Youth Fitness Study II, OPHS, ODPHP.
Baseline and updates for grades 9–12: Youth Risk Behavior Survey, CDC, NCCDPHP.
For students in middle/junior and senior high schools: School Health Policies and Programs Study, CDC, NCCDPHP.
1.9 Baseline for all students: Siedentop D. Developing Teaching Skills in Physical Education. Palo Alto, CA Mayfield. 1983.
Data for grades 9–12: Youth Risk Behavior Survey, CDC, NCCDPHP.
For physical education teachers: School Health Policies and Programs Study, CDC, NCCDPHP.
1.10 National Survey of Worksite Health Promotion Activities, OPHS, ODPHP.
1995 data: Business Responds to AIDS Benchmark Survey, CDC, NCHSTP.
1998–99 data: National Worksite Health Promotion Survey, Association for Worksite Health Promotion.
1.11 Baseline: McDonald BL and Cordell HK. Local Opportunities for Americans: Final Report of the Municipal and County Park and Recreation Study,
Alexandria, VA: National Recreation and Park Association. 1988.
1.12 Baseline: American College of Physicians Membership Survey of Prevention Practices in Adult Medicine.
1992 data: Primary Care Provider Surveys, OPHS, ODPHP.
1998 data: Prevention in Primary Care Study, American College of Preventive Medicine.
1.13*, 1.13a–b National Health Interview Survey, CDC, NCHS; National Nursing Home Survey, CDC, NCHS.
* Duplicate objective. See full text of objective following this table.
Healthy People 2000 Final Review 73
Physical Activity and
Fitness Objectives
1.1*: Reduce coronary heart disease
deaths to no more than 100 per 100,000
people.
Duplicate objectives: 2.1, 3.1, and 15.1
1.1a*: Reduce coronary heart
disease deaths among blacks to no
more than 115 per 100,000.
Duplicate objectives: 2.1a, 3.1a, and
15.1a
1.2*: Reduce overweight to a prevalence
of no more than 20 percent among
people aged 20 and older and no more
than 15 percent among adolescents aged
12–19.
Duplicate objectives: 2.3, 15.10, and
17.12
1.2a*: Reduce overweight to a
prevalence of no more than
25 percent among low-income
women aged 20 and older.
Duplicate objectives: 2.3a, 15.10a,
and 17.12a
1.2b*: Reduce overweight to a
prevalence of no more than
30 percent among black women
aged 20 and older.
Duplicate objectives: 2.3b, 15.10b,
and 17.12b
1.2c*: Reduce overweight to a
prevalence of no more than
25 percent among Hispanic women
aged 20 and older.
Duplicate objectives: 2.3c, 15.10c,
and 17.12c
1.2d*: Reduce overweight to a
prevalence of no more than
30 percent among American Indians
and Alaska Natives.
Duplicate objectives: 2.3d, 15.10d,
and 17.12d
1.2e*: Reduce overweight to a
prevalence of no more than
25 percent among people with
disabilities.
Duplicate objectives: 2.3e, 15.10e,
and 17.12e
1.2f*: Reduce overweight to a
prevalence of no more than
41 percent among women with high
blood pressure.
Duplicate objectives: 2.3f, 15.10f,
and 17.12f
1.2g*: Reduce overweight to a
prevalence of no more than
35 percent among men with high
blood pressure.
Duplicate objectives: 2.3g, 15.10g,
and 17.12g
1.2h*: Reduce overweight to a
prevalence of no more than
25 percent among Mexican-
American men.
Duplicate objectives: 2.3h, 15.10h,
and 17.12h
1.3*: Increase to at least 30 percent the
proportion of people aged 6 and older
who engage regularly, preferably daily,
in light to moderate physical activity for
at least 30 minutes per day.
Duplicate objectives: 15.11 and 17.13
1.3a*: Increase to at least
25 percent the proportion of
Hispanics aged 18 and older who
engage in light to moderate physical
activity for at least 30 minutes per
day 5 or more times per week.
Duplicate objectives: 15.11a and
17.13a
1.4: Increase to at least 20 percent the
proportion of people aged 18 and older
and to at least 75 percent the proportion
of children and adolescents aged 6–17
who engage in vigorous physical
activity that promotes the development
and maintenance of cardiorespiratory
fitness 3 or more days per week for 20
or more minutes per occasion.
1.4a: Increase to at least 12 percent
the proportion of lower-income
people aged 18 and older (annual
family income less than $20,000)
who engage in vigorous physical
activity that promotes the
development and maintenance of
cardiorespiratory fitness 3 or more
days per week for 20 or more
minutes per occasion.
1.4b: Increase to at least 17 percent
the proportion of blacks aged 18
and older who engage in vigorous
physical activity that promotes the
development and maintenance of
cardiorespiratory fitness 3 or more
days per week for 20 or more
minutes per occasion.
1.4c: Increase to at least 17 percent
the proportion of Hispanics aged 18
and older who engage in vigorous
physical activity that promotes the
development and maintenance of
cardiorespiratory fitness 3 or more
days per week for 20 or more
minutes per occasion.
1.5: Reduce to no more than 15 percent
the proportion of people aged 6 and
older who engage in no leisure-time
physical activity.
1.5a: Reduce to no more than
22 percent the proportion of people
aged 65 and older who engage in
no leisure-time physical activity.
1.5b: Reduce to no more than
20 percent the proportion of people
with disabilities who engage in no
leisure-time physical activity.
1.5c: Reduce to no more than
17 percent the proportion of
lower-income people aged 18 and
older (annual family income less
than $20,000) who engage in no
leisure-time physical activity.
1.5d: Reduce to no more than
20 percent the proportion of blacks
aged 18 and older who engage in
no leisure-time physical activity.
1.5e: Reduce to no more than
25 percent the proportion of
Hispanics aged 18 and older who
engage in no leisure-time physical
activity.
1.5f: Reduce to no more than
21 percent the proportion of
American Indians/Alaska Natives
aged 18 and older who engage in
no leisure-time physical activity.
1.6: Increase to at least 40 percent the
proportion of people aged 6 and older
who regularly perform physical
activities that enhance and maintain
muscular strength, muscular endurance,
and flexibility.
1.7*: Increase to at least 50 percent the
proportion of overweight people aged 12
and older who have adopted sound
dietary practices combined with regular
physical activity to attain an appropriate
body weight.
Duplicate objective: 2.7
1.7a*: Increase to at least
24 percent the proportion of
overweight Hispanic males aged 18
and older who have adopted sound
dietary practices combined with
regular physical activity to attain an
appropriate body weight.
74 Healthy People 2000 Final Review
Duplicate objective: 2.7a
1.7b*: Increase to at least
22 percent the proportion of
overweight Hispanic females aged
18 and older who have adopted
sound dietary practices combined
with regular physical activity to
attain an appropriate body weight.
Duplicate objective: 2.7b
1.8: Increase to at least 50 percent the
proportion of children and adolescents
in 1st–12th grade who participate in
daily school physical education.
1.9: Increase to at least 50 percent the
proportion of school physical education
class time that students spend being
physically active, preferably engaged in
lifetime physical activities.
1.10: Increase the proportion of
worksites offering employer-sponsored
physical activity and fitness programs as
follows:
2000 target
Worksites with— (percent)
50–99 employees 20
100–249 employees 35
250–749 employees 50
750 or more employees 80
1.11: Increase community availability
and accessibility of physical activity and
fitness facilities as follows:
Hiking, biking, and fitness trail miles: 1
per 10,000 people
Public swimming pools: 1 per 25,000
people
Acres of park and recreation open
space: 4 per 1,000 people (250 people
per managed acre)
1.12: Increase to at least 50 percent the
proportion of primary care providers
who routinely assess and counsel their
patients regarding the frequency,
duration, type, and intensity of each
patient’s physical activity practices.
1.13*: Reduce to no more than 90 per
1,000 people the proportion of all
people aged 65 and older who have
difficulty in performing two or more
personal care activities, thereby
preserving independence.
Duplicate objective: 17.3 and age-related
objective for people aged 65 and older
1.13a*: Reduce to no more than
325 per 1,000 people the proportion
of all people aged 85 and older who
have difficulty in performing two or
more personal care activities,
thereby preserving independence.
Duplicate objective: 17.3a
1.13b*: Reduce to no more than 98
per 1,000 people the proportion of
blacks aged 65 and older who have
difficulty in performing two or more
personal care activities, thereby
preserving independence.
Duplicate objective: 17.3b
*Duplicate objective.
Healthy People 2000 Final Review 75

Priority Area 2
Nutrition
Background
Nutrition is essential for growth and
development, health, and well being.
Nutritional, or dietary, factors contribute
substantially to the burden of
preventable diseases and premature
deaths in the United States (1). Dietary
factors are associated with four of the
leading causes of death: coronary heart
disease, some types of cancer, stroke,
and type 2 diabetes (2). These health
conditions are estimated to cost society
more than $200 billion annually in
medical expenses and lost productivity
(3). Dietary factors also are associated
with osteoporosis, a condition that
affects more than 25 million persons in
the United States and is the major
underlying cause of bone fractures in
postmenopausal women and elderly
persons (4).
In general, excesses and imbalances
in some food components in the diet
have replaced once commonplace
nutrient deficiencies. The number of
overweight and obese persons has
increased substantially (5,6). Persons
who are overweight or obese are at
increased risk for high blood pressure,
type 2 diabetes, coronary heart disease,
stroke, gallbladder disease, osteoarthritis,
sleep apnea, respiratory problems, and
certain cancers. The total costs (medical
and lost productivity) attributable to
obesity alone amounted to $99 million
in 1995 (7). Overweight results when a
person eats more calories from food
(energy) than he or she expends, for
example, through physical activity.
Many factors influence this balance
between energy intake and output
including genetic, metabolic,
environmental, and cultural factors.
The relationship between nutrition
and health involves many dietary
components. A primary concern is that
many Americans consume too much
saturated fat, and too few vegetables,
fruits, and grain products that are high
in vitamins, minerals, carbohydrates
(starch and dietary fiber), and other
substances that are important to good
health. The Dietary Guidelines for
Americans also recommend that
consumers moderate their intake of total
fat, sodium, and sugars, while at the
same time obtain adequate amounts of
other nutrients such as iron and calcium
(8).
Data Summary
Highlights
The available data suggest that the
majority of the 27 nutrition objectives
either met or moved toward their year
2000 targets. However, for some
objectives the progress was modest and
for others there was movement away
from the targets.
Throughout the 1990s, the Nation
made steady gains in reducing deaths
from coronary heart disease (2.1), stroke
(2.22), all types of cancer (2.2), and
colorectal cancer (2.23). The prevalence
of high blood cholesterol (2.25) among
people ages 20–74 years decreased to a
level that met its target. Growth
retardation among low-income children
ages 5 years and under (2.4) exceeded
its target, declining from 11 percent in
1987 to 8 percent in 1999. The percent
of elementary and secondary schools
offering low-fat choices for breakfast
and lunch increased considerably (2.17),
although, by the end of the decade, only
about one in five schools offered
lunches that met goals for total fat and
saturated fat content.
Other nutrition objectives also
showed improvement during the 1990s.
The average fat intake among people
ages 2 years and over declined and the
proportion of the population who
consumed no more than 30 percent of
calories from fat increased (2.5). The
availability of reduced-fat processed
foods increased to such an extent that
the 2000 target was surpassed early in
the decade (2.15). Informative nutrition
labeling (2.14) was found on more
processed foods, fresh produce, and
fresh seafood. Similar labeling of fresh
meat and poultry, however, decreased.
Modest gains were also apparent in
promoting consumption of fruits,
vegetables, and grains. The average
number of servings of vegetables and
fruits consumed by the population
increased as did the proportion of
people who consumed at least five
servings (2.6). For grain products, the
year 2000 target was exceeded for both
the average number of servings
consumed and for the proportion of the
population who consumed at least six
servings (2.6). Despite this progress, a
majority of Americans still did not
consume the minimum recommended
number of vegetable and fruit servings
by the mid–1990s, nor did their
vegetable and grain choices reflect
sufficient variety.
Other objectives moved away from
the year 2000 targets. The prevalence of
overweight (2.3) increased substantially
between 1976–80 and 1988–94.
Preliminary analysis of data from the
1999 National Health and Nutrition
Examination Survey (NHANES)
suggests a continued increase in the
prevalence of overweight and obesity.
Additionally, the proportion of
self-reported overweight males and
females who report using exercise and
diet to lose weight (2.7) decreased. Both
the incidence and prevalence of diabetes
(2.24) increased for the population as a
whole and among the special population
groups for which there are
data—American Indians/Alaska Natives,
Mexican Americans, and blacks.
Iron deficiency (2.10) increased in
females of childbearing age (20–44
years) between 1976–80 and 1988–94.
Further, no progress was observed in
reducing the prevalence of iron
deficiency among young children (1–4
years) overall, although the prevalence
did decline for low-income children. An
objective to increase the population’s
consumption of calcium-rich foods (2.8)
also moved away from its target—only
about 1 in 10 females ages 11–24 years
consumed the recommended number of
servings in the mid–1990s.
Summary of Progress
Of the 27 objectives that address
nutrition, 6 objectives (2.1, 2.2, 2.4,
2.15, 2.23, and 2.25) met or exceeded
their year 2000 targets. Thirteen
objectives (2.5, 2.6, 2.11–2.13,
2.16–2.20, 2.22, 2.26, and 2.27) moved
toward the year 2000 targets. For two
objectives (2.9 and 2.14), progress was
mixed among the multiple measures
used for tracking. Six objectives (2.3,
2.7, 2.8, 2.10, 2.21, and 2.24) moved
away from the targets. Although the
objective status for 2.10 remained at
baseline for young children, the overall
status showed movement away from the
target, because iron deficiency increased
in females of childbearing age. The
status of objective 2.21 (nutrition
assessment, counseling, and referral) is
based on a very small decline reported
by nurse practitioners only. See table 2
for the tracking data for the objectives
in this priority area and figure 2 for a
quantitative assessment of progress.
76 Healthy People 2000 Final Review
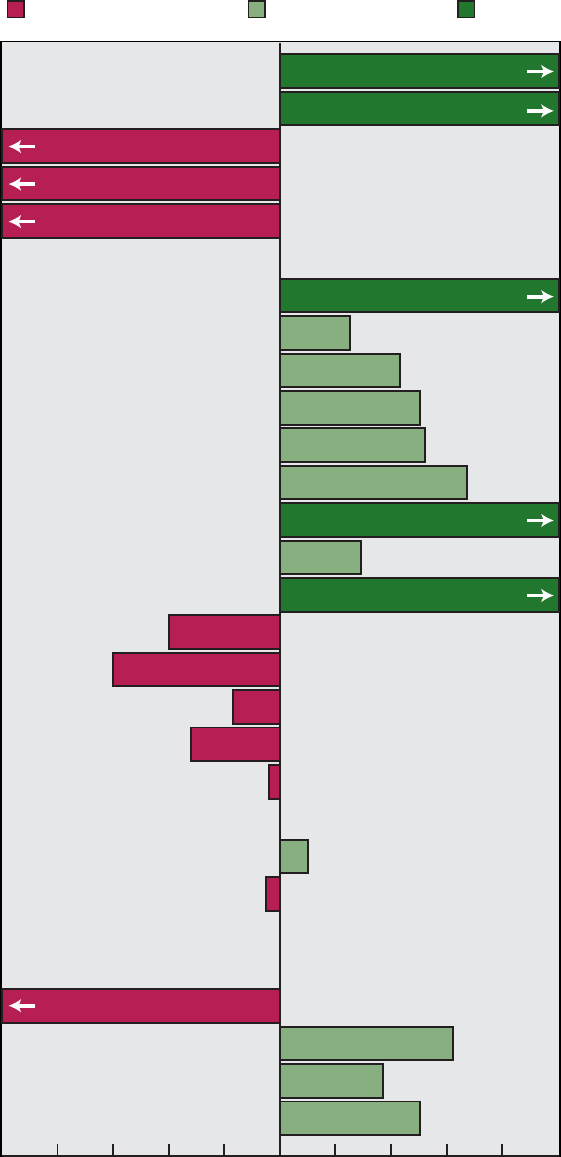
-100 -80 -60 -40 -20
0
20 40 60 80 100
2.1 Coronary heart disease deaths
2.2 Cancer deaths
2.3 Overweight prevalence: 20-74 years
Male
Female
Adolescents 12-19 years
2.4 Growth retardation: Low income children ≤5 years
2.5 Total dietary fat: Average percent of calories
Met goal for fat
Saturated fat: Average percent of calories
Met goal for saturated fat
2.6 Average servings: Vegetables/fruits
Grain products
2.7 Weight loss practices: Overweight males
Grain products
Met guidelines: Vegetables/fruits
Overweight females
2.8 Calcium: Three servings daily, 11-24 years
Pregnant or lactating females
Two servings daily, 2-10 years
25 years and over
2.9 Salt intake: Rarely or never use at table
Regularly buy reduced salt products
2.10 Iron deficiency: 1-2 years
Female 20-44 years
3-4 years
2.11 Breastfeeding: Early postpartum
At 6 months
2.12 Baby bottle tooth decay preventive practices
Moved away from target Moved toward target Met target
Chart continues onto next page – see notes at the end of the chart.
109%
550%
120%
300%
250%
150%
250%
143%
-17%
-60%
-40%
10%
62%
37%
50%
29%
25%
43%
50%
Away*
No change
No change
No change
52%
67%
-32%
-4%
-5%
150%
Percent of target achieved
Healthy People 2000 Final Review 77
Figure 2. Final status of Nutrition objectives

2.13 Use of food labels
2.14 Informative nutrition labeling: Processed foods
Fresh produce
Fresh seafood
Fresh meat/poultry
2.15 Availability of reduced-fat processed foods
2.16 Low-fat, low-calorie restaurant food choices
2.17 School lunches: 30% or less calories from fat
10% or less calories from saturated fat
School breakfasts: 30% or less calories from fat
10% or less calories from saturated fat
2.18 Receipt of home-delivered meals
2.20 Worksite: Nutrition education
2.19 States requiring nutrition education
Weight control
2.22 Stroke deaths
2.23 Colorectal cancer deaths
2.24 Diabetes: Incidence
Prevalence
2.25 High blood cholesterol prevalence: 20-74 years
Male
Female
2.26 Controlled high blood pressure
2.27 Mean serum cholesterol level: 20-74 years
Male
Female
Moved away from target Moved toward target Met target
*This objective has moved away from its target. A progress quotient could not be calculated.
NOTE: Complete tracking data are shown in table 2. Progress quotients are not calculated for objective 2.21.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
-52%
-50%
9%
81%
79%
25%
21%
16%
74%
63%
22%
60%
42%
26%
51%
31%
77%
82%
73%
Percent of target achieved
-100 -80 -60 -40 -20 0 20 40 60 80 100
93%
125%
180%
114%
140%
100%
-367%
78 Healthy People 2000 Final Review
Figure 2b. Final status of Nutrition objectives-Con.
Discussion
Potential factors influencing the
decline in deaths from heart disease and
stroke include the decline in the percent
of persons with high blood cholesterol,
and the increases in the proportion of
people with controlled high blood
pressure. Dietary factors and a number
of other factors including medical
treatment may have also influenced the
decline in these death rates.
Legislative and regulatory initiatives
also played a role in achieving progress
on some of the nutrition objectives. For
example, as a result of the passage of
the Nutrition Labeling and Education
Act of 1990 and the introduction of a
new food label in 1993, nutrition
information now appears on most
processed packaged foods, along with
credible health and nutrient content
claims and standardized serving sizes.
Further, at the start of the decade,
almost no retail food stores voluntarily
provided nutrition labeling for raw
produce and raw seafood, whereas the
available data suggest that the majority
do so now. While these efforts have
increased the availability of nutrition
information and healthful foods sold in
supermarkets, a significant challenge
remains on these fronts for
away-from-home foods purchased at
fast-food outlets, restaurants, cafeterias,
and other food service outlets.
Public-private sector partnerships
such as the 5-A-Day for Better Health
Program and the Dietary Guidelines
Alliance have also been instrumental in
achieving progress on the nutrition
objectives. However, despite many
efforts directed at healthful eating, the
prevalence of people who are
overweight has increased considerably,
which also affects the rising incidence
and prevalence of diabetes. Generous
portion sizes combined with limited
nutrition information for many foods
purchased away from home may be
partly responsible. Other factors, such as
the sedentary lifestyle of many people in
the United States, also likely influence
this alarming trend.
Transition to Healthy People
2010
The Healthy People 2000 nutrition
chapter has been renamed in Healthy
People 2010 to ‘‘Nutrition and
Overweight,’’ providing for an increased
focus on the achievement and
maintenance of healthy weight (9).
Overweight and obesity is the topic of
one of the 10 Leading Health Indicators
(LHIs), which Healthy People 2010
introduces to serve as a barometer of the
Nation’s health. Two objectives from the
Nutrition and Overweight focus
area—the proportion of children and
adolescents who are overweight or
obese and the proportion of adults who
are obese—are used to measure this
LHI. With the Healthy People 2010
initiative, the prevalence of overweight
among children will be tracked for the
first time. Body Mass Index (BMI) cut
points that will be used in Healthy
People 2010 to monitor prevalence of
overweight and obesity have been
changed to be consistent with recent
national and international
recommendations.
The Healthy People 2010 chapter
contains a section that lists objectives in
other chapters that are related to the
topics of nutrition and overweight.
Objectives that address coronary heart
disease, stroke, cancer, and diabetes, for
example, appear in the chapters for
Heart Disease and Stroke, Cancer, and
Diabetes, respectively.
Other modifications have been
made for consistency with the fifth
edition of the Dietary Guidelines for
Americans (8). For example, Healthy
People 2010 has set separate targets for
fruits, vegetables and grains and placed
increased emphasis on the quality of
vegetables and grains consumed. In
Healthy People 2010, there is a shift
from estimating intake of calcium-rich
foods (in the form of milk products) to
estimating total calcium intake from all
sources (including foods, dietary
supplements, and antacids). There is
also a shift from assessing salt use to
estimating total sodium intake from a
variety of sources (including foods,
dietary supplements, tap water, and salt
use at table). The iron deficiency and
anemia objectives have been expanded
to cover all pregnant women in addition
to other vulnerable groups such as
women of childbearing age and young
children.
A Healthy People 2010
developmental objective focuses on food
actually eaten at school, recognizing
increasing options available to children.
School nutrition education is addressed
along with broader school health
education issues in the chapter on
educational and community-based
programs. Healthy People 2010 also has
two objectives that aim to promote
nutrition education and weight
management initiatives at the worksite
and nutrition counseling in the health
care setting.
Additionally, disparities in health
status and risk factors for diet-related
disease are key themes of the Healthy
People 2010 Nutrition and Overweight
chapter. These are evident in many
segments of the population based on
gender, age, race and ethnicity, and
income. For example, overweight and
obesity are observed in all population
groups, but obesity is particularly
common among Hispanic and African
American women.
Several of the Healthy People 2000
objectives were dropped because they
were achieved, were considered to be
strategies for achieving other objectives,
or had unreliable data sources. These
objectives include weight loss practices,
baby bottle tooth decay, use of food
labels, nutrition labeling, restaurant/food
service choices, and availability of low-
or reduced-fat foods.
Finally, Healthy People 2010 has a
new objective aimed at increasing food
security among U.S. households. This
objective is a result of both increased
recognition of the problem of food
insecurity in the United States, and an
improved ability to measure it.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the changes between the
two decades of objectives, reflecting
new knowledge and direction for this
area.
Data Issues
Definitions
The death rates for coronary heart
disease (2.1), stroke (2.22), cancer (2.2),
and colorectal cancer (2.23) are age
adjusted to the 1940 U.S. population
(see Appendix for more information on
age-adjusted rates). The National Cancer
Institute age adjusts cancer deaths to the
1970 U.S. population. When the 1970
standard population is used, the
equivalent baseline, interim, and target
rates are all somewhat higher than those
generated using the 1940 population, but
the trends are very similar.
Coronary heart disease deaths (2.1)
are defined by ICD–9 codes 402,
410–414, and 429.2. These codes are
Healthy People 2000 Final Review 79
different from the codes used to define
the category ‘‘Diseases of heart,’’ which
often appears in published tables (see
Appendix and Appendix table II).
Overweight (2.3) for adults is
defined as a BMI at or above the
sex-specific 85th percentile of the
1976–80 National Health and Nutrition
Examination Survey (NHANES II)
reference population 20–29 years of age.
For adolescents, overweight is the sex-
and age-specific 85th percentile from
NHANES II. The cut points used to
define overweight approximate the
120 percent of desirable body weight
definition used in the 1990 objectives.
BMI cutoff points for adults are 27.8
kilograms per meter squared for males,
and 27.3 kilograms per meter squared
for females. Current international
research has indicated that a lower BMI
of 25 kilograms per meter squared may
be more clinically relevant to increased
risk of cardiovascular disease in adults
(5,6), and Healthy People 2010 defines
the upper limit of the healthy weight
range for adults as a BMI of less than
25. For adolescents, overweight and
obesity is defined as a BMI equal to or
greater than 23.0 for males 12–14 years,
24.3 for males 15–17 years, 25.8 for
males 18–19 years, 23.4 for females
12–14 years, 24.8 for females 15–17
years, and 25.7 for females 18–19 years.
Healthy People 2010 uses a BMI at or
above the gender- and age-specific 95th
percentile of BMI from the CDC growth
charts for children and adolescents (10).
Growth retardation for objective 2.4
is defined as height-for-age below the
fifth percentile of children in the
National Center for Health Statistics’
reference population derived from the
1971–74 National Health and Nutrition
Examination Survey (NHANES I).
Estimates for objective 2.6 (fruit,
vegetable, and grain intakes) exclude
fruits and vegetables eaten as part of
potato chips, condiments, fruit-flavored
candies, jellies, and jams.
Updates for iron deficiency (2.10)
from the 1988–94 NHANES III data
were made as comparable as possible to
the 1976–80 NHANES II estimates to
allow for trend comparisons. Three
methods are used to determine iron
deficiency: mean corpuscular volume
(MCV), erythrocyte protoporphyrin, and
transferrin saturation. Iron deficiency is
defined as having abnormal results for
two or more methods. In 1988–94,
MCV cutoff points were raised by one
unit to account for differences in MCV
values of the reference population at or
below the median between NHANES II
and NHANES III and known differences
in hematocrit measurements
(centrifugation in NHANES II versus
electronic measurements in NHANES
III) and possible methods differences in
red blood cell counts (both surveys used
electronic measurements but NHANES
III data showed counts that appeared
lower). Anemia is used as an index of
iron deficiency. Anemia among Alaska
Native children was defined as
hemoglobin less than 11 gm/dL or
hematocrit less than 34 percent. For
pregnant women in the third trimester,
anemia was defined according to CDC
criteria.
For objective 2.11, the definition
used for breastfeeding includes
exclusive use of human milk or the use
of human milk with a supplemental
bottle of formula or cow’s milk.
Objective 2.12 addresses feeding
practices that prevent baby bottle tooth
decay. The measure used to establish a
baseline for this objective for the total
population, caregivers with less than a
high school education (2.12a), blacks
(2.12c), and Hispanics (2.12d)isfor
children 6–23 months old. For this
objective, feeding practices to prevent
baby bottle tooth decay include child no
longer using a bottle, never used a
bottle, or if the child is still using a
bottle, that no bottle was given at
bedtime (excluding bottles with plain
water) during the past 2 weeks.
Objective 2.14, which targets
nutrition labeling of food products; and
labeling of fresh produce, fish, and meat
and poultry, is measured by the
percentage of retail food stores that
provide nutrition labeling. In 1990, the
Nutrition Labeling and Education Act
mandated the periodic assessment of
actions taken by food retailers to
provide consumers with nutrition
information for raw agricultural
commodities and raw fish, in particular
to determine whether food retailers
could achieve and maintain substantial
compliance with guidelines for a
voluntary nutrition labeling program.
Guidelines for the nutrition labeling of
these raw foods were issued in
November 1991. A baseline survey
conducted in August and September
1991, before the guidelines were issued,
determined that virtually no food
retailers provided complete nutrition
labeling for these foods. Baseline
estimates for objective 2.14 published
elsewhere (11,12) for fresh produce and
fresh seafood have been revised based
on this survey and a reinterpretation of
the available data. The first followup
survey to assess compliance with the
final rule was conducted in November
and December 1992. The dramatic
increase from 1991 to 1992 in the
percentage of retail food stores
providing nutrition labeling information
for raw produce and for raw fish
represents a highly significant response
to both the Nutrition Labeling and
Education Act and the Food and Drug
Administration’s implementation of
regulations.
High blood cholesterol (2.25)is
defined as serum cholesterol levels of at
least 240 mg/dL (13).
Objective 2.26 addresses the
proportion of people with hypertension
whose blood pressure is under control.
High blood pressure is defined as blood
pressure greater than or equal to 140
mm Hg systolic and/or 90 mm Hg
diastolic and/or taking antihypertensive
medication. The estimates used to track
this objective define control as
maintaining a blood pressure less than
140 mm Hg systolic and 90 mm Hg
diastolic through the use of
antihypertensive medication only and do
not include other nonpharmacologic
treatments such as weight loss, low
sodium diets, and restriction of alcohol.
Data Sources
Growth retardation among
low-income children (2.4) and
breastfeeding among American
Indian/Alaska Native mothers (2.11d)
are tracked by the Pediatric Nutrition
Surveillance System (PedNSS). The
number of participating States and
Indian tribes has varied from year to
year. The fluctuations in the scope of
surveillance could affect the
comparability of estimates.
Data for 2.11 and 2.11a–c are from
the Ross Mothers’ Survey (RMS)
conducted by Abbot Laboratories. The
RMS is an ongoing survey that is
periodically mailed to a probability
sample of new mothers selected from a
list of names that represents
approximately 80 percent of all national
births. Mothers are asked to recall the
type of milk their baby was fed in the
hospital and in each subsequent month
up to the month of the survey. Mothers
are considered to be breastfeeding if
they used either human milk exclusively
or human milk in combination with a
80 Healthy People 2000 Final Review
supplemental bottled formula or cow’s
milk.
In 1988–96, the questionnaires were
mailed to mothers at the time their
babies were 6 months old. In 1997 the
methodology changed and
questionnaires were mailed to a larger
sample of mothers with babies 1–12
months of age. Therefore, although the
overall sample grew to approximately
double the pre–1997 size, the number in
the sample for each month (including 6
months) became considerably smaller
than that of previous years. This change
affects the stability of the 6–month
figures used to monitor this objective.
Also beginning with data year 1997, the
RMS no longer collects information on
family income. Information on
education of the mother is available
from the survey to measure
socioeconomic status.
The data on inquiry about diet and
nutrition for objective 2.21 are from the
Primary Care Provider Surveys (PCPS),
which were drawn from a random
stratified sample of members of the
American College of Physicians from
four geographic regions. Provider groups
sampled included internists,
pediatricians, nurse practitioners,
obstetricians/gynecologists, and family
physicians. In 1992 response rates
varied from 50 to 80 percent across
these groups. The data on formulation of
a diet and nutrition plan represent the
proportion of providers who routinely
delivered these services to
81–100 percent of their clients who
needed a plan.
The Prevention in Primary Care
Study (PPCS) was conducted in
1997–98 to update data from the PCPS.
The design and items included in the
1997–98 study were similar to the
PCPS, but a slightly different sampling
frame was used and some items
included in the 1992 surveys were not
included in the PPCS. The professionals
were sampled from listings of all
licensed, active practitioners in the
United States whose practices were at
least 50 percent primary care. Because
of low response rates from the other
provider groups, updates are available
only for nurse practitioners.
Data for diabetes prevalence (2.24
and 2.24e) were tracked using the
National Health Interview Survey
(NHIS). Because of the NHIS redesign
implemented in 1997, data collected in
1996 and earlier are not comparable
with data from the redesigned NHIS
(see Appendix). American Indian or
Alaska Native data for 1996 for
objective 2.24a are from the Indian
Health Service (IHS) Patient
Comprehensive Care file. The file
excludes data from 25 (representing
11 percent of the population served by
IHS) of the 166 IHS service units
because data were incomplete.
Data Comparability
Overweight (2.3) was tracked with
two data sources. The primary data
source is NHANES, which provided
baselines and the 1988–94 updates for
most of the overweight objectives and
subobjectives; these data are derived
from measured height and weight. The
second data source is NHIS. This survey
provides interim estimates shown in an
earlier publication (14), updates for
Hispanic females and American
Indians/Alaska Natives, and all data for
people with disabilities. NHIS estimates
are based on self-reported heights and
weights and are not comparable with the
measured data from NHANES:
prevalence estimates of overweight from
self-reported height and weight tend to
be lower than estimates from measured
height and weight. Trends from the
NHIS self-report measures, like those
from NHANES, show a steady increase
in prevalence of overweight; this
increase, however, differs in magnitude
from that observed in the data derived
from measured height and weight.
For the use of food labels by adults
(2.13) the 1988 baseline data, and 1990,
1994, and 1995 updates are from the
Food and Drug Administration’s Health
and Diet Survey. After being given a
description of food labels, respondents
were asked if they read food labels. The
1991, 1993, 1995, and 1998 updates are
from the NHIS, which asked
respondents how often they read food
labels for calories, fat, and/or cholesterol
content. Respondents answering
‘‘always,’’ ‘‘often,’’ or ‘‘sometimes’’
were considered to be making nutritious
food selections using the food labels.
The 1985 and 1992 data for
objective 2.20 are from the National
Survey of Worksite Health Promotion
Activities, which were telephone surveys
of nongovernment worksites. Worksites
were sampled because different
worksites within the same company
could have different sets of health
promotion activities. Both active (for
example, classes) and passive methods
were included as worksite health
promotion activities. The 1995 update is
from the CDC-sponsored Worksite
Benchmark Survey, which used a
methodology very similar to the 1992
survey, but did not include passive
methods (for example, brochures) of
health promotion (15,16). The 1998–99
data are from the National Worksite
Health Promotion Survey, which
randomly sampled nongovernmental
worksites of 50 or more employees in a
particular geographic location. This
telephone survey included questions
regarding the offering of
nutrition/cholesterol education or weight
management programs for employees at
the workplace or through health plans
(17). The 1998–99 data are not
comparable to the data that were
collected earlier, although similar
questions were asked.
Proxy Data
Objective 2.7 is to increase to at
least 50 percent the proportion of
overweight people who use sound
dietary practices combined with regular
physical activity to attain appropriate
body weight. Respondents who reported
they were overweight and were
currently trying to lose weight or control
their weight by eating fewer calories
and exercising more were included in
the numerator for this objective,
although an assessment of the quality of
dietary practices has not yet been
integrated with a measure of regular
physical activity. The 1985 and 1990
questionnaires asked respondents
specifically if they were eating fewer
calories to lose weight and if they were
increasing their physical activity to lose
weight. In 1991, 1993, 1995, and 1998,
eating fewer calories and exercising
more were among a list of 10 possible
methods of losing weight in response to
the question, ‘‘Are you currently doing
any of these things to control your
weight?’’ Respondents were asked this
question if they reported they were
trying to lose weight or stay about the
same.
References
1. Frazao E. The high costs of poor eating patterns
in the United States. In: Frazao E, ed. America’s
eating habits: Changes and consequences.
Washington: U.S. Department of Agriculture,
Economic Research Service, AIB–750. 1999.
Healthy People 2000 Final Review 81
2. Murphy SL. Deaths: Final data for 1998.
National vital statistics reports 45(11). Hyattsville,
Maryland: National Center for Health Statistics.
2000.
3. Frazao E. The American diet: A costly problem.
Food Review 19:2–6. 1996.
4. National Institutes of Health. NIH Consensus
Statement: Optimal Calcium Intake. 12(4). 1994.
5. Flegal KM, Carroll MD, Kuczmarski RJ, et al.
Overweight and obesity in the United States:
Prevalence and trends, 1960–1994. International J
Obesity 22(1)39–47. 1998.
6. Kuczmarski RJ, Carroll MD, Flegal KM, et al.
Varying body mass index cutoff points to describe
overweight prevalence among U.S. adults:
NHANES III (1988–1994). Obesity Research
5(6):542–8. 1997.
7. Wolf AM and Colditz GA. Current estimates of
the economic costs of obesity in the United States.
Obesity Research 6(2):97–106. 1998.
8. U.S. Department of Agriculture and Department
of Health and Human Services. Nutrition and your
health: Dietary guidelines for Americans. 5th ed.
USDA Home and garden bulletin; no 232.
Washington: U.S. Department of Agriculture.
2000.
9. Department of Health and Human Services.
Healthy people 2010. 2d ed. With understanding
and improving health and objectives for improving
health. 2 vols. Washington: U.S. Government
Printing Office. 2000.
10. Troiano RP, Flegal KM. Overweight
prevalence among youth in the United States: Why
so many different numbers? International J
Obesity 23 (Suppl 2):S22–7. 1999.
11. National Center for Health Statistics. Healthy
people 2000 review, 1995–96. Hyattsville,
Maryland: Public Health Service. 1996.
12. Department of Health and Human Services.
Healthy people 2000 midcourse review and 1995
revisions. Washington: Public Health Service.
1995.
13. Johnson CL, et al. Declining serum total
cholesterol levels among U.S. adults. JAMA
269(23):3002–23. 1993.
14. National Center for Health Statistics. Healthy
people 2000 review, 1992. Hyattsville, Maryland:
Public Health Service. 1993.
15. Department of Health and Human Services.
Business Responds to AIDS Benchmark Survey:
Technical report. Atlanta, Georgia: Public Health
Service, Centers for Disease Control and
Prevention. 1996.
16. Centers for Disease Control and Prevention.
Cancer screening offered by worksites—United
States, 1992 and 1995. MMWR 46(19):421–4.
1997.
17. Department of Health and Human Services.
Tracking healthy people 2010. Washington: U.S.
Government Printing Office. 2000.
82 Healthy People 2000 Final Review

Table 2. Nutrition objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
2.1* Coronary heart disease deaths (age adjusted
per 100,000) ................................ 1987 135 122 118 114 114 110 108 105 100 97 - - - 100
a. Black ................................... 1987 168 158 156 151 154 147 147 140 136 133 - - - 115
2.2* Cancer deaths (age adjusted per 100,000) ......... 1987 134 135 135 133 133 132 130 128 126 124 - - - 130
a. Black ................................... 1990 182 . . . 179 178 177 174 172 168 165 161 - - - 175
2.3* Overweight prevalence (based on measured height
and weight unless otherwise indicated)
Adults 20–74 years ............................ 1976–80 26% - - - - - - - - - - - -
1,2
35% --- --- --- --- --- 20%
Male ..................................... 1976–80 24% - - - - - - - - - - - -
1,2
34% --- --- --- --- --- 20%
Female ................................... 1976–80 27% - - - - - - - - - - - -
1,2
37% --- --- --- --- --- 20%
Adolescents 12–19 years ....................... 1976–80 15% - - - - - - - - - - - -
1
24% --- --- --- --- --- 15%
a. Low-income female 20–74 years ............... 1976–80 37% - - -
3
47% --- --- --- --- --- --- --- --- 25%
b. Black female 20–74 years .................... 1976–80 44% - - - - - - - - - - - -
1,2
52% --- --- --- --- --- 30%
c. Hispanic female 20–74 years ................. ... --- --- --- --- --- --- --- --- --- --- --- 25%
Hispanic female 20 years and over
4
.......... ... --- 33% 32% 32% 33% 32% 35% ---
6
44%
6
46% --- ...
Mexican American female 20–74 years ........ 1982–84 39% - - - - - - - - - - - -
1,2
50% --- --- --- --- --- ...
Cuban female 20–74 years ................. 1982–84 34% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Puerto Rican female 20–74 years ............ 1982–84 37% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
d. American Indian/Alaska Native 20 years and over. . 1984–88 29–75% - - -
4
40%
4
36%
4
48%
4
34%
4
43% - - -
6,4
46%
6,4
45% - - - 30%
e. People with disabilities 20 years and over
4
....... 1985 36% - - - 38% 37% 38% 38% 40% - - - - - - - - - - - - 25%
f. Females with high blood pressure 20–74 years . . . 1976–80 50% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 41%
g. Males with high blood pressure 20–74 years ...... 1976–80 39% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 35%
h. Mexican American male 20–74 years ........... 1982–84 30% - - - - - - - - - - - -
1,2
37% --- --- --- --- --- 25%
2.4 Growth retardation among low-income children
5 years and under............................ 1988 11% 9% 9% 8% 8% 8% 8% 8% 8% 8% 8% 10%
a. Low-income black children under 1 year ......... 1988 15% 15% 15% 15% 16% 16% 15% 15% 15% 15% 15% 10%
b. Low-income Hispanic children under 1 year ...... 1988 13% 9% 8% 8% 7% 7% 7% 7% 7% 7% 7% 10%
c. Low-income Hispanic children 1 year ........... 1988 16% 12% 11% 9% 9% 8% 8% 8% 8% 8% 8% 10%
d. Low-income Asian/Pacific Islander children 1 year. . 1988 14% 14% 13% 12% 11% 11% 12% 11% 11% 11% 10% 10%
e. Low-income Asian/Pacific Islander children 2–4
years ................................... 1988 16% 14% 12% 11% 10% 10% 10% 9% 8% 8% 7% 10%
2.5* Dietary fat intake among people 2 years and over
5
National Health and Nutrition Examination Survey
Average percent of calories from total fat.......... 1976–80
6
36% --- --- --- ---
1
34% --- --- --- --- --- 30%
Average percent of calories from saturated fat ...... 1976–80
6
13% --- --- --- ---
1
12% --- --- --- --- --- 10%
Percent who met goal for fat ................... 1988–94
§
27% ... ... ... ... ... --- --- --- --- --- 50%
Percent who met goal for saturated fat ........... 1988–94
§
29% ... ... ... ... ... --- --- --- --- --- 50%
Continuing Survey of Food Intakes by Individuals
Average percent of calories from total fat.......... 1989–91 34% . . . . . . - - - - - - 33% 33% 33% - - - - - - - - - 30%
Average percent of calories from saturated fat ...... 1989–91 12% . . . . . . - - - - - - 11% 11% 11% - - - - - - - - - 10%
Percent who met goal for fat ................... 1989–91 22% . . . . . . - - - - - - 32% 33% 34% - - - - - - - - - 50%
Percent who met goal for saturated fat ........... 1989–91 21% . . . . . . - - - - - - 34% 35% 36% - - - - - - - - - 50%
See footnotes and key at end of table.
Healthy People 2000 Final Review 83

Table 2. Nutrition objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
2.6* Average daily intake of vegetables, fruits, and
grain products among people 2 years and over
5
Average number of servings
Vegetables and fruits ......................... 1989–91 4.1 . . . . . . - - - - - - 4.6 4.7 4.7 - - - - - - - - - 5.0
Grain products.............................. 1989–91 5.8 . . . . . . - - - - - - 6.7 6.8 6.9 - - - - - - - - - 6.0
Proportion who met Dietary Guidelines goal
Vegetables and fruits ......................... 1989–91 29% . . . . . . - - - - - - 36% 37% 35% - - - - - - - - - 50%
Grain products.............................. 1989–91 40% . . . . . . - - - - - - 50% 53% 52% - - - - - - - - - 50%
2.7* Sound weight loss practices among overweight
people 12 years and over ..................... ... --- --- --- --- --- --- --- --- --- --- --- 50%
Overweight male 18 years and over ............... 1985 25% 22% 19% - - - 17% - - - 15% - - - - - - 15% - - - 50%
Overweight female 18 years and over .............. 1985 30% 29% 22% - - - 19% - - - 19% - - - - - - 18% - - - 50%
a. Overweight Hispanic male 18 years and over ..... 1991 15% . . . . . . - - - 11% - - - 13% - - - - - - 12% - - - 24%
b. Overweight Hispanic female 18 years and over.... 1991 13% . . . . . . - - - 16% - - - 16% - - - - - - 12% - - - 22%
2.8 Foods rich in calcium (percent who consume)
5
Average of 3 or more servings daily
People 11–24 years .......................... 1989–91 20% . . . . . . - - - - - - 17% 16% 15% - - - - - - - - - 50%
Pregnant and lactating females
7
................ 1989–91 22% . . . . . . - - - - - - 20% 17% 13% - - - - - - - - - 50%
Average of 2 or more servings daily
8
Children 2–10 years ......................... 1989–91 48% . . . . . . - - - - - - 42% 41% 47% - - - - - - - - - 75%
People 25 years and over ..................... 1989–91 21% . . . . . . - - - - - - 20% 20% 21% - - - - - - - - - 50%
Proportion who met average daily goal
a. Female 11–24 years ........................ 1989–91 13% . . . . . . - - - - - - 11% 10% 8% - - - - - - - - - 50%
2.9 Salt and sodium intake (18 years and over)
Prepare foods without adding salt ................. 1989–90 43% . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - 65%
Rarely or never use salt at table .................. 1989–91 60% . . . . . . - - - - - - 56% 58% 62% - - - - - - - - - 80%
Regularly purchase foods with reduced salt
and sodium content........................... 1988 20% - - - - - - - - - - - - - - - 19% - - - - - - - - - - - - 40%
2.10 Iron deficiency prevalence
Children 1–4 years .......................... ... --- --- --- --- ---
1
6% --- --- --- --- --- 3%
Children 1–2 years .......................... 1976–80 9% - - - - - - - - - - - -
1
9% --- --- --- --- --- 3%
Children 3–4 years .......................... 1976–80 4% - - - - - - - - - - - -
1
4% --- --- --- --- --- 3%
Females of childbearing age (20–44 years) ........ 1976–80 5% - - - - - - - - - - - -
1
8% --- --- --- --- --- 3%
a. Low-income children 1–2 years ................ 1976–80 21% - - - - - - - - - - - -
1
13% --- --- --- --- --- 10%
b. Low-income children 3–4 years ................ 1976–80 10% - - - - - - - - - - - -
1
6% --- --- --- --- --- 5%
c. Low-income female 20–44 years ............... 1976–80 8% - - - - - - - - - - - -
1
12% --- --- --- --- --- 4%
Anemia prevalence
d. Alaska Native children 1–5 years .............. 1983–85 22–28%
9
38%
9
32%
9
31%
9
29%
9
27%
9
27% --- --- --- --- 10%
e. Black, low-income pregnant females
15–44 years (third trimester) ................. 1988 41% 41% 42% 43% 44% 43% 45% 46% 44% 44% - - - 20%
See footnotes and key at end of table.
84 Healthy People 2000 Final Review

Table 2. Nutrition objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
2.11* Breastfeeding
During early postpartum period
10
................. 1988 54% 52% 53% 54% 56% 57% 60% 59% 62% 64% 67% 75%
a. Low-income mothers........................ 1988
§
34% 35% 33% 35% 38% 40% 42% 42% 46% 47% 49% 75%
b. Black mothers............................. 1988 25% 23% 26% 28% 31% 33% 37% 37% 41% 45% 50% 75%
c. Hispanic mothers .......................... 1988 51% 48% 52% 52% 56% 58% 61% 61% 64% 66% 69% 75%
d. American Indian/Alaska Native mothers ......... 1988 47% 47% 46% 53% 51% 44% 52% 54% 56% 57% 62% 75%
At age 6 months .............................. 1988
§
20% 18% 18% 19% 19% 20% 22% 22% 26% 29% 31% 50%
a. Low-income mothers........................ 1988 9% 8% 8% 9% 10% 10% 11% 12% 20% 21% 20% 50%
b. Black mothers............................. 1988
§
7% 6% 7% 8% 9% 10% 11% 11% 15% 19% 20% 50%
c. Hispanic mothers .......................... 1988
§
14% 13% 15% 16% 16% 18% 20% 20% 25% 28% 29% 50%
d. American Indian/Alaska Native mothers ......... 1988 28% 27% 22% 24% 28% 24% 24% 24% 25% 26% 27% 50%
2.12* Baby bottle tooth decay
Parents and caregivers who use preventive feeding
practices ................................... 1991 55% . . . . . . - - - - - - - - - - - - - - - - - -
11
65% - - - 75%
a. Parents and caregivers with less than high school
education............................... 1991 36% . . . . . . - - - - - - - - - - - - - - - - - -
11
38% - - - 65%
b. American Indian/Alaska Native parents and
caregivers .............................. 1985–89 74% - - - - - - - - - - - - - - - - - - - - - - - -
11,12
--- --- 65%
c. Black parents and caregivers ................. 1991 48% . . . . . . - - - - - - - - - - - - - - - - - -
11
57% - - - 65%
d. Hispanic parents and caregivers ............... 1991 39% . . . . . . - - - - - - - - - - - - - - - - - -
11
46% - - - 65%
2.13 Use of food labels among people 18 years and
over ...................................... 1988 74% 76% - - - - - - - - - 74% 75% - - - - - - - - - - - - 85%
Read food labels for calories, fat, and/or cholesterol
content .................................... ... --- --- 64% --- 66% --- 66% --- --- 61% --- ...
2.14 Informative nutrition labeling
Processed foods .............................. 1988 60% - - - 66% - - - 76% - - - 96% - - - 97% - - - - - - 100%
Fresh produce ............................... 1991
§
Less
than 1%
... ... 76% --- 75% --- 73% --- --- --- 90%
Fresh seafood................................ 1991
§
0% ... ... 73% --- 75% --- 71% --- --- --- 90%
Fresh meat/poultry ............................ 1995
§
67% ... ... ... ... ... ... 58% --- --- 55% 90%
Carry-away foods ............................. ... --- --- --- --- --- --- --- --- --- --- 40%
2.15 Availability of reduced-fat processed foods ....... 1986 2,500 - - - 5,618 - - - - - - - - - - - - - - - - - - - - - - - - 5,000
2.16 Low-fat, low-calorie restaurant food choices
Proportion of large chain restaurants offering at least
one low-fat, low-calorie item .................... 1989 70% 75% - - - - - - - - - - - - - - - - - - - - - - - - - - - 90%
2.17 Nutritious school and child care food services..... ... --- --- --- --- --- --- --- --- --- --- --- 90%
Schools offering lunches with an average of:
30% or less of calories from total fat ............. 1992 1% . . . . . . . . . - - - - - - - - - - - - - - - - - - 20% . . .
Less than 10% of calories from saturated fat ....... 1992 Less
than 1%
... ... ... --- --- --- --- --- --- 15% ...
See footnotes and key at end of table.
Healthy People 2000 Final Review 85

Table 2. Nutrition objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Schools offering breakfasts in USDA program with an
average of:
30% or less of calories from total fat ............. 1992 44% . . . . . . . . . - - - - - - - - - - - - - - - - - - 78% . . .
Less than 10% of calories from saturated fat ....... 1992 4% . . . . . . . . . - - - - - - - - - - - - - - - - - - 58% . . .
Schools with initiatives to reduce fat
Drained browned meat prior to serving ........... ... --- --- --- --- --- 94% --- --- --- --- --- ...
Spooned solid fat from chilled meat .............. ... --- --- --- --- --- 79% --- --- --- --- --- ...
Did not provide butter or margarine .............. ... --- --- --- --- --- 31% --- --- --- --- --- ...
2.18 Receipt of home-delivered meals for people 65
years and over in need ....................... 1991
§
48% ... ... --- 48% --- 50% --- --- 55% --- 80%
2.19 Nutrition education in schools .................. ... --- --- --- --- --- --- --- --- --- --- --- 75%
Proportion of States requiring nutrition education...... 1990 60% . . . - - - - - - - - - 69% - - - - - - - - - - - - - - - . . .
Nutrition education in at least one class:
Middle/junior high schools ..................... ... --- --- --- --- --- 83% --- --- --- --- --- ...
Senior high schools .......................... ... --- --- --- --- --- 85% --- --- --- --- --- ...
2.20 Worksite nutrition/weight management programs
Nutrition education ............................ 1985 17% - - - - - - 31% - - - - - - - - - - - - - - - - - - - - - 50%
Weight control................................ 1985 15% - - - - - - 24% - - - - - - - - - - - - - - - - - - - - - 50%
Nutrition education and/or weight control ............ ... --- --- --- 37% --- --- --- --- --- --- --- 50%
Nutrition or cholesterol group classes, workshops, or
lectures.................................... ... --- --- --- 17% --- --- 18% --- --- ---
13
23% 50%
Weight management group classes, workshops, or
lectures.................................... ... --- --- --- 15% --- --- 14% --- --- ---
13
15% 50%
2.21 Nutrition assessment, counseling, and referral by
clinicians .................................. 1988 40–50% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 75%
Percent of clinicians routinely providing service to
81–100 percent of patients
Inquiry about diet/nutrition
Pediatricians ............................... ... --- --- --- 53% --- --- --- --- ---
14,15
--- --- 75%
Nurse practitioners .......................... ... --- --- --- 46% --- --- --- --- ---
14
43% - - - 75%
Obstetricians/gynecologists .................... ... --- --- --- 15% --- --- --- --- ---
14,15
--- --- 75%
Internists .................................. ... --- --- --- 36% --- --- --- --- ---
14,15
--- --- 75%
Family physicians ........................... ... --- --- --- 19% --- --- --- --- ---
14,15
--- --- 75%
Formulation of a diet/nutrition plan
Pediatricians ............................... ... --- --- --- 31% --- --- --- --- ---
14,15
--- --- 75%
Nurse practitioners .......................... ... --- --- --- 31% --- --- --- --- ---
14
31% - - - 75%
Obstetricians/gynecologists .................... ... --- --- --- 19% --- --- --- --- ---
14,15
--- --- 75%
Internists .................................. ... --- --- --- 33% --- --- --- --- ---
14,15
--- --- 75%
Family physicians ........................... ... --- --- --- 24% --- --- --- --- ---
14,15
--- --- 75%
2.22* Stroke deaths (age adjusted per 100,000) .......... 1987 30.4 27.7 26.8 26.2 26.5 26.5 26.7 26.4 25.9 25.1 - - - 20.0
a. Black ................................... 1987 52.5 48.4 46.8 45.0 45.0 45.4 45.0 44.2 42.5 41.4 - - - 27.0
See footnotes and key at end of table.
86 Healthy People 2000 Final Review

Table 2. Nutrition objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
2.23* Colorectal cancer deaths (age adjusted
per 100,000) ................................ 1987 14.7 13.8 13.5 13.2 13.1 13.0 12.8 12.3 12.1 12.0 - - - 13.2
a. Black ................................... 1990 18.1 . . . 17.5 17.3 17.6 17.3 17.4 16.9 16.9 16.8 - - - 16.5
2.24* Diabetes incidence and prevalence
Total population (per 1,000)
Incidence of diabetes......................... 1986–88 2.9
16
2.6
17
2.5
18
2.4
19
2.8
20
3.1
21
3.4
22
3.1 --- --- --- 2.5
Prevalence of diabetes ....................... 1986–88 28
16
26
17
27
18
28
19
30
20
30
21
31
22
31
‡
38
‡
39 - - - 25
Prevalence of diabetes (per 1,000)
a. American Indian/Alaska Native 15 years and
over in Indian Health Service areas ........... 1987 69 - - - - - - - - - - - - - - - - - -
2
90 --- --- --- 62
b. Puerto Rican 20–74 years ................... 1982–84 55 - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 49
c. Mexican American 20–74 years ............... 1982–84 54 - - - - - - - - - - - -
1
66 --- --- --- --- --- 49
d. Cuban American 20–74 years ................. 1982–84 36 - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 32
e. Black (all ages)............................ 1986–88 36
16
36
17
36
18
36
19
38
20
40
21
42
22
44
‡
55
‡
51 - - - 32
2.25* High blood cholesterol prevalence
People 20–74 years ........................... 1976–80 27% - - - - - - - - - - - -
1
19% --- --- --- --- --- 20%
Male 20–74 years ............................. 1976–80 25% - - - - - - - - - - - -
1
18% --- --- --- --- --- 20%
Female 20–74 years ........................... 1976–80 29% - - - - - - - - - - - -
1
20% --- --- --- --- --- 20%
2.26* Controlled high blood pressure
People with high blood pressure 18–74 years ........ 1976–80 11% - - - - - - - - - - - -
1
23% --- --- --- --- --- 50%
a. Males with high blood pressure 18–74 years ...... 1976–80 6% - - - - - - - - - - - -
1
17% --- --- --- --- --- 40%
b. Mexican Americans with high blood pressure
18–74 years ............................. 1988–94
§
14% ... ... ... ... ... --- --- --- --- --- 50%
c. Females with high blood pressure 70 years
and over ................................ 1988–94
§
19% ... ... ... ... ... --- --- --- --- --- 50%
2.27* Mean serum cholesterol level (mg/dL)
People 20–74 years ........................... 1976–80 213 - - - - - - - - - - - -
1
203 --- --- --- --- --- 200
Male 20–74 years ............................. 1976–80 211 - - - - - - - - - - - -
1
202 --- --- --- --- --- 200
Female 20–74 years ........................... 1976–80 215 - - - - - - - - - - - -
1
204 --- --- --- --- --- 200
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
‡
The NHIS was redesigned in 1997. Data may not be comparable with previous years; see Appendix.
1
1988–94 data.
2
Data are for people 20 years and over.
3
1988–91 data.
4
Estimate derived from self-reported height and weight.
5
Estimates are from 1-, 2-, or 3-day dietary data.
6
For people 2–74 years.
7
Estimate may be unreliable because of small cell size and/or large coefficients of variation.
8
Excluding pregnant/lactating females and breastfed children.
9
Low-income children 1–4 years.
10
Breastfed in hospital.
11
Data are for children under 2 years. Baseline data are for children 6–23 months.
12
Data are unreliable. Relative standard error is greater than 30%.
13
1998–99 data. Worksite location only.
Met
Toward
Mixed/ no change
Away
Cannot assess
Healthy People 2000 Final Review 87

14
1997-98 data.
15
Response rate for this group was too low to produce reliable estimates.
16
1988-90 data.
17
1989-91 data.
18
1990-92 data.
19
1991-93 data.
20
1992-94 data.
21
1993-95 data.
22
1994-96 data.
NOTE: Data include revisions and, therefore, may differ from data previously published.
Objective number Data source
2.1*, 2.1a National Vital Statistics System, CDC, NCHS.
2.2*, 2.2a National Vital Statistics System, CDC, NCHS.
2.3*, 2.3a, b, f, g National Health and Nutrition Examination Survey, CDC, NCHS.
2.3c, h Data for Hispanic: National Health Interview Survey, CDC, NCHS.
Baselines for Mexican American, Cuban, Puerto Rican: Hispanic Health and Nutrition Examination Survey, CDC, NCHS.
Updates for Mexican American: National Health and Nutrition Examination Survey, CDC, NCHS.
2.3d Baseline: IHS, OPEL.
Updates: National Health Interview Survey, CDC, NCHS.
2.3e National Health Interview Survey, CDC, NCHS.
2.4, 2.4a-e Pediatric Nutrition Surveillance System, CDC, NCCDPHP.
2.5* 1976–80 baselines and 1988–94 data: National Health and Nutrition Examination Survey, CDC, NCHS.
1989–91 baselines and 1994–96 updates: Continuing Survey of Food Intakes by Individuals, USDA.
2.6* Continuing Survey of Food Intakes by Individuals, USDA.
2.7*, 2.7a, b National Health Interview Survey, CDC, NCHS.
2.8, 2.8a Continuing Survey of Food Intakes by Individuals, USDA.
2.9 Preparing foods and use of salt at table: Continuing Survey of Food Intakes by Individuals, USDA.
Purchasing reduced-salt foods: Health and Diet Survey, FDA.
2.10, 2.10a-c National Health and Nutrition Examination Survey, CDC, NCHS.
2.10d Baseline: Survey of American Indians/Alaska Natives, CDC; IHS, OPEL.
Updates: Pediatric Nutrition Surveillance System, CDC, NCCDPHP.
2.10e Pregnancy Nutrition Surveillance System, CDC, NCCDPHP.
2.11*, 2.11a–c Ross Laboratories Mothers Survey.
2.11d Pediatric Nutrition Surveillance System, CDC, NCCDPHP.
2.12*, 2.12a, c, d National Health Interview Survey, CDC, NCHS.
2.12b Baseline: 1990 Baby Bottle Tooth Decay 5-year Evaluation Report, IHS.
Update: National Health Interview Survey, CDC, NCHS.
2.13 Use of food labels: Health and Diet Survey, FDA.
Read food labels: National Health Interview Survey, CDC, NCHS.
2.14 Baseline and updates for processed foods: Food Label and Package Survey, FDA.
Baselines and updates for fresh produce and seafood: Nutrition Labeling of Raw Produce and Raw Fish, FDA.
Baseline for fresh meat/poultry: Nutritional Labeling/Safe Handling Information Study: Raw Meat and Poultry, USDA.
2.15 Nielsen Company National Scantrack.
2.16 Survey of Chain Operators, National Restaurant Association.
2.17 For lunches and breakfasts: School Nutrition Dietary Assessment Study I and II, USDA.
For initiatives to reduce dietary fat: School Health Policies and Programs Study, CDC, NCCDPHP.
88 Healthy People 2000 Final Review

Objective number Data source
2.18 National Health Interview Survey, CDC, NCHS.
2.19 Baseline: National Survey of School Health Education Activities, CDC, NCCDPHP.
Update: School Health Policies and Programs Study, CDC, NCCDPHP.
2.20 National Survey of Worksite Health Promotion Activities, OPHS, ODPHP.
1995 data: Business Responds to AIDS Benchmark Survey, CDC, NCHSTP.
2.21 1988 baseline: Lewis CE. Disease prevention and health promotion practices of primary care physicians in the United States. Am J Prev Med 4:9–16.
1988.
1992 data: Primary Care Provider Surveys, OPHS, ODPHP.
1998 data: Prevention in Primary Care Study, American College of Preventive Medicine.
2.22*, 2.22a National Vital Statistics System, CDC, NCHS.
2.23*, 2.23a National Vital Statistics System, CDC, NCHS.
2.24*, 2.24e National Health Interview Survey, CDC, NCHS.
2.24a Ambulatory Utilization Data, IHS.
2.24b–d Baseline: Hispanic Health and Nutrition Examination Survey, CDC, NCHS.
Update for Mexican American: National Health and Nutrition Examination Survey, CDC, NCHS.
2.25* National Health and Nutrition Examination Survey, CDC, NCHS.
2.26*, 2.26a-c National Health and Nutrition Examination Survey, CDC, NCHS.
2.27* National Health and Nutrition Examination Survey, CDC, NCHS.
* Duplicate objective.
Healthy People 2000 Final Review 89
Nutrition Objectives
2.1*: Reduce coronary heart disease
deaths to no more than 100 per 100,000
people.
Duplicate objectives: 1.1, 3.1, and 15.1
2.1a*: Reduce coronary heart
disease deaths among blacks to no
more than 115 per 100,000 people.
Duplicate objectives: 1.1a, 3.1a, and
15.1a
2.2*: Reverse the rise in cancer deaths
to achieve a rate of no more than 130
per 100,000 people.
Duplicate objective: 16.1
2.2a*: Reverse the rise in cancer
deaths to achieve a rate of no more
than 175 per 100,000 blacks.
Duplicate objective: 16.1a
2.3*: Reduce overweight to a prevalence
of no more than 20 percent among
people aged 20 and older and no more
than 15 percent among adolescents aged
12–19.
Duplicate objectives: 1.2, 15.10, and
17.12
2.3a*: Reduce overweight to a
prevalence of no more than
25 percent among low-income
women aged 20 and older.
Duplicate objectives: 1.2a, 15.10a,
and 17.12a
2.3b*: Reduce overweight to a
prevalence of no more than
30 percent among black women
aged 20 and older.
Duplicate objectives: 1.2b, 15.10b,
and 17.12b
2.3c*: Reduce overweight to a
prevalence of no more than
25 percent among Hispanic women
aged 20 and older.
Duplicate objectives: 1.2c, 15.10c,
and 17.12c
2.3d*: Reduce overweight to a
prevalence of no more than
30 percent among American Indians
and Alaska Natives.
Duplicate objectives: 1.2d, 15.10d,
and 17.12d
2.3e*: Reduce overweight to a
prevalence of no more than
25 percent among people with
disabilities.
Duplicate objectives: 1.2e, 15.10e,
and 17.12e
2.3f*: Reduce overweight to a
prevalence of no more than
41 percent among women with high
blood pressure.
Duplicate objectives: 1.2f, 15.10f,
and 17.12f
2.3g*: Reduce overweight to a
prevalence of no more than
35 percent among men with high
blood pressure.
Duplicate objectives: 1.2g, 15.10g,
and 17.12g
2.3h*: Reduce overweight to a
prevalence of no more than
25 percent among
Mexican-American men.
Duplicate objectives: 1.2h, 15.10h,
and 17.12h
2.4: Reduce growth retardation among
low-income children aged 5 and
younger to less than 10 percent.
2.4a: Reduce growth retardation
among low-income black children
younger than age 1 to less than
10 percent.
2.4b: Reduce growth retardation
among low-income Hispanic
children younger than age 1 to less
than 10 percent.
2.4c: Reduce growth retardation
among low-income Hispanic
children aged 1 to less than
10 percent.
2.4d: Reduce growth retardation
among low-income Asian and
Pacific Islander children aged 1 to
less than 10 percent.
2.4e: Reduce growth retardation
among low-income Asian and
Pacific Islander children aged 2–4
to less than 10 percent.
2.5*: Reduce dietary fat intake to an
average of 30 percent of calories or less
and average saturated fat intake to less
than 10 percent of calories among
people aged 2 and older. In addition,
increase to at least 50 percent the
proportion of people aged 2 and older
who meet the Dietary Guidelines’
average daily goal of no more than
30 percent of calories from fat, and
increase to at least 50 percent the
proportion of people aged 2 and older
who meet the average daily goal of less
than 10 percent of calories from
saturated fat.
Duplicate objectives: 15.9 and 16.7
2.6*: Increase complex carbohydrate and
fiber-containing foods in the diets of
people aged 2 and older to an average
of five or more daily servings for
vegetables (including legumes) and
fruits, and to an average of six or more
daily servings for grain products. In
addition, increase to at least 50 percent
the proportion of people aged 2 and
older who meet the Dietary Guidelines’
average daily goal of five or more
servings of vegetables/fruits, and
increase to at least 50 percent the
proportion who meet the goal of six or
more servings of grain products.
Duplicate objective: 16.8
2.7*: Increase to at least 50 percent the
proportion of overweight people aged 12
and older who have adopted sound
dietary practices combined with regular
physical activity to attain an appropriate
body weight.
Duplicate objective: 1.7
2.7a*: Increase to at least
24 percent the proportion of
overweight Hispanic males aged 18
and older who have adopted sound
dietary practices combined with
regular physical activity to attain an
appropriate body weight.
Duplicate objective: 1.7a
2.7b*: Increase to at least
22 percent the proportion of
overweight Hispanic females aged
18 and older who have adopted
sound dietary practices combined
with regular physical activity to
attain an appropriate body weight.
Duplicate objective: 1.7b
2.8: Increase calcium intake so at least
50 percent of people aged 11–24 and
50 percent of pregnant and lactating
women consume an average of three or
more daily servings of foods rich in
calcium, and at least 75 percent of
children aged 2–10 and 50 percent of
people aged 25 and older consume an
average of two or more servings daily.
2.8a: Increase calcium intake so at
least 50 percent of females aged
11–24 consume an average of three
90 Healthy People 2000 Final Review
or more daily servings of foods rich
in calcium.
2.9: Decrease salt and sodium intake so
at least 65 percent of home meal
preparers prepare foods without adding
salt, at least 80 percent of people avoid
using salt at the table, and at least
40 percent of adults regularly purchase
foods modified or lower in sodium.
2.10: Reduce iron deficiency to less than
3 percent among children aged 1
through 4 and among women of
childbearing age.
2.10a: Reduce iron deficiency to
less than 10 percent among
low-income children aged 1–2.
2.10b: Reduce iron deficiency to
less than 5 percent among
low-income children aged 3–4.
2.10c: Reduce iron deficiency to
less than 4 percent among
low-income women of childbearing
age.
2.10d: Reduce the prevalence of
anemia to less than 10 percent
among Alaska Native children aged
1–5.
2.10e: Reduce the prevalence of
anemia to less than 20 percent
among black, low-income pregnant
women (third trimester).
2.11*: Increase to at least 75 percent the
proportion of mothers who breastfeed
their babies in the early postpartum
period and to at least 50 percent the
proportion who continue breastfeeding
until their babies are 5 to 6 months old.
Duplicate objective: 14.9
2.11a*: Increase to at least
75 percent the proportion of
low-income mothers who breastfeed
their babies in the early postpartum
period and to at least 50 percent the
proportion who continue
breastfeeding until their babies are 5
to 6 months old.
Duplicate objective: 14.9a
2.11b*: Increase to at least
75 percent the proportion of black
mothers who breastfeed their babies
in the early postpartum period and
to at least 50 percent the proportion
who continue breastfeeding until
their babies are 5 to 6 months old.
Duplicate objective: 14.9b
2.11c*: Increase to at least
75 percent the proportion of
Hispanic mothers who breastfeed
their babies in the early postpartum
period and to at least 50 percent the
proportion who continue
breastfeeding until their babies are 5
to 6 months old.
Duplicate objective: 14.9c
2.11d*: Increase to at least
75 percent the proportion of
American Indian and Alaska Native
mothers who breastfeed their babies
in the early postpartum period and
to at least 50 percent the proportion
who continue breastfeeding until
their babies are 5 to 6 months old.
Duplicate objective: 14.9d
2.12*: Increase to at least 75 percent the
proportion of parents and caregivers
who use feeding practices that prevent
baby bottle tooth decay.
Duplicate objective: 13.11
2.12a*: Increase to at least
65 percent the proportion of parents
and caregivers with less than a high
school education who use feeding
practices that prevent baby bottle
tooth decay.
Duplicate objective: 13.11a
2.12b*: Increase to at least
65 percent the proportion of
American Indian and Alaska Native
parents and caregivers who use
feeding practices that prevent baby
bottle tooth decay.
Duplicate objective: 13.11b
2.12c*: Increase to at least
65 percent the proportion of black
parents and caregivers who use
feeding practices that prevent baby
bottle tooth decay.
Duplicate objective: 13.11c
2.12d*: Increase to at least
65 percent the proportion of
Hispanic parents and caregiverswho
use feeding practices that prevent
baby bottle tooth decay.
Duplicate objective: 13.11d
2.13: Increase to at least 85 percent the
proportion of people aged 18 and older
who use food labels to make nutritious
food selections.
2.14: Achieve useful and informative
nutrition labeling for virtually all
processed foods and at least 40 percent
of ready-to-eat carry-away foods.
Achieve compliance by at least
90 percent of retailers with the voluntary
labeling of fresh meats, poultry, seafood,
fruits, and vegetables.
2.15: Increase to at least 5,000 brand
items the availability of processed food
products that are reduced in fat and
saturated fat.
2.16: Increase to at least 90 percent the
proportion of restaurants and
institutional food service operations that
offer identifiable low-fat, low-calorie
food choices, consistent with the Dietary
Guidelines for Americans.
2.17: Increase to at least 90 percent the
proportion of school lunch and breakfast
services and child care food services
with menus that are consistent with the
nutrition principles in the Dietary
Guidelines for Americans.
2.18: Increase to at least 80 percent the
receipt of home food services by people
aged 65 and older who have difficulty in
preparing their own meals or are
otherwise in need of home-delivered
meals.
2.19: Increase to at least 75 percent the
proportion of the Nation’s schools that
provide nutrition education from
preschool–12th grade, preferably as part
of comprehensive school health
education.
2.20: Increase to at least 50 percent the
proportion of worksites with 50 or more
employees that offer nutrition education
and/or weight management programs for
employees.
2.21: Increase to at least 75 percent the
proportion of primary care providers
who provide nutrition assessment and
counseling and/or referral to qualified
nutritionists or dietitians.
2.22*: Reduce stroke deaths to no more
than 20 per 100,000 people.
Duplicate objectives: 3.18 and 15.2
2.22a*: Reduce stroke deaths
among blacks to no more than 27
per 100,000.
Duplicate objectives: 3.18a and
15.2a
2.23*: Reduce colorectal cancer deaths
to no more than 13.2 per 100,000
people.
Duplicate objective: 16.5
Healthy People 2000 Final Review 91
2.23a*: Reduce colorectal cancer
deaths among blacks to no more
than 16.5 per 100,000.
Duplicate objective: 16.5a
2.24*: Reduce diabetes to an incidence
of no more than 2.5 per 1,000 people
and a prevalence of no more than 25 per
1,000 people.
Duplicate objective: 17.11
2.24a*: Reduce diabetes among
American Indians and Alaska
Natives to a prevalence of no more
than 62 per 1,000.
Duplicate objective: 17.11a
2.24b*: Reduce diabetes among
Puerto Ricans to a prevalence of no
more than 49 per 1,000.
Duplicate objective: 17.11b
2.24c*: Reduce diabetes among
Mexican-Americans to a prevalence
of no more than 49 per 1,000.
Duplicate objective: 17.11c
2.24d*: Reduce diabetes among
Cuban Americans to a prevalence of
no more than 32 per 1,000.
Duplicate objective: 17.11d
2.24e*: Reduce diabetes among
blacks to a prevalence of no more
than 32 per 1,000.
Duplicate objective: 17.11e
2.25*: Reduce the prevalence of blood
cholesterol levels of 240 mg/dL or
greater to no more than 20 percent
among adults.
Duplicate objective: 15.7
2.26*: Increase to at least 50 percent the
proportion of people with high blood
pressure whose blood pressure is under
control.
Duplicate objective: 15.4
2.26a*: Increase to at least
40 percent the proportion of men
with high blood pressure whose
blood pressure is under control.
Duplicate objective: 15.4a
2.26b*: Increase to at least
50 percent the proportion of
Mexican-Americans with high blood
pressure whose blood pressure is
under control.
Duplicate objective: 15.4b
2.26c*: Increase to at least
50 percent the proportion of women
70 years and older with high blood
pressure whose blood pressure is
under control.
Duplicate objective: 15.4c
2.27*: Reduce the mean serum
cholesterol level among adults to no
more than 200 mg/dL.
Duplicate objective: 15.6
*Duplicate objective.
92 Healthy People 2000 Final Review

Priority Area 3
Tobacco
Background
Tobacco use is the single most
preventable cause of death and disease
in the Nation (1). Each year, it causes
approximately one in five deaths (more
than 430,000) in the United States (2).
Tobacco use is associated with cancer,
heart disease, chronic obstructive
pulmonary disease, and stroke— 4 of
the 5 leading causes of death (3). If
current smoking patterns continue, an
estimated 25 million persons in the
United States who are alive today will
die prematurely from smoking-related
illnesses, including an estimated 5
million persons now under age 18 years
(4).
Smoking contributes substantially to
chronic disease and disability. It costs
the Nation an estimated $50–$73 billion
in medical expenses and $50 million in
indirect costs (5, 6). The total
smoking-attributable costs related to
complicated births among pregnant
smokers is an additional $1.4 billion (7).
Cigarette smoking during pregnancy
accounts for 17–26 percent of
low-birthweight babies (8–10). The risks
of tobacco use extend beyond the actual
users. Nearly 9 of 10 nonsmoking U.S.
residents are exposed to environmental
tobacco smoke (ETS) (1). For adult
nonsmokers, exposure to ETS increases
the risk for lung cancer and heart
disease (1, 11). Among children,
exposure to ETS may cause serious
respiratory problems (1, 11). In fact,
substantial evidence now indicates that
ETS exposure is associated with low
birthweight and sudden infant death
syndrome (11).
Other tobacco products also have
significant health consequences (1). Use
of smokeless tobacco is associated with
leukoplakia, oral cancer, and halitosis.
Periodontal degeneration and soft tissue
lesions are early indicators of these
conditions. Strong evidence also shows
causal relationships between regular
cigar use and cancers of the lungs,
larynx, oral cavity, and esophagus.
Smoking among high school
students increased significantly from
approximately 28 percent in 1991 to
35 percent in 1999 (12). Among middle
school students, 13 percent currently use
some form of tobacco (cigarettes,
smokeless tobacco, cigars, pipes, bidis,
or kreteks) (13). Differences by racial
and ethnic groups show that white teens
are taking up smoking at higher rates
than are black and Hispanic teens.
Among teens who are regular smokers,
one in three will ultimately die from
smoking. Although recent studies
indicate that current teen smoking may
have leveled or begun to decline, a great
deal of work is still needed to meet the
health promotion and disease prevention
objectives for the Nation (14).
The prevalence of smoking remains
disproportionately high among some
groups. For example, in 1998, more
than one of three American
Indians/Alaska Natives, people with low
income, and people with less than a
high school education smoked cigarettes
(14).
Primarily, because tobacco use is
addictive, of the nearly 70 percent of
smokers who want to quit smoking
completely, only 2.5 percent quit
permanently each year (15). In the past,
helping people quit smoking was the
primary focus of efforts to reduce
tobacco use, and it continues to be of
great interest with the introduction and
availability of new pharmacotherapies.
This strategy is a critical one, since
smoking cessation at all ages reduces
the risk of premature death. More
recently, this focus of tobacco control
has expanded to include strategies to
prevent individuals from ever starting to
smoke. Such efforts have centered on
young people, since the decision to use
tobacco is usually made in the teenage
years, and about one-half of young
people who take up smoking continue to
use tobacco products as adults. This
preventive strategy also includes efforts
to protect people from exposure to ETS.
Finally, efforts to reduce tobacco use
traditionally targeted individuals, but
now the focus is on both individuals and
communities.
Data Summary
Highlights
Great strides were made over the
course of the decade for many of the
national tobacco use objectives. All the
objectives targeting mortality either met
or moved toward their targets. Age-
adjusted death rates from coronary heart
disease (3.1) declined for the total
population; the rate for blacks also
declined, but at a slower rate than for
the total population. Lung cancer (3.2)
and chronic obstructive pulmonary
disease death rates (3.3) slowed to rates
below the Healthy People 2000 targets
after a rise in the previous decade.
Although the death rate for lung cancer
in women continued to rise, the rate of
increase slowed sufficiently so that the
year 2000 target was met. Oral cancer
deaths among males and females ages
45–74 years (3.17) dropped over the
course of the decade; for black males
and females in this age group the
decline in rates exceeded the decline in
rates for the total population ages 45–74
years. The mortality rate for stroke also
declined (3.18) and the subobjective for
blacks showed considerable progress,
narrowing the disparity with the total
population.
Although the proportion of high
school seniors reporting a perception of
social disapproval for smoking cigarettes
(3.21) declined, the average age of first
use of cigarettes (3.19) increased to 12.4
years and the proportion of adolescents
reporting use of cigarettes in the last
month (3.20) dropped from a baseline of
22.7 percent in 1988 to 18.2 percent in
1998. Smokeless tobacco use declined
among adolescent and young adult
males, and the target set for adolescents
ages 12–17 was met (3.9). Reports of
perception of the harm caused by using
smokeless tobacco (3.22) increased by
about a third, from 30 percent in 1987
to 41 percent in 1999.
The majority of tobacco use
objectives targeting institutions directly
affecting youth have also shown
improvement. Between 1988 and 1994,
the number of school districts providing
tobacco-free environments more than
doubled, and the number of schools
providing anti-smoking education also
increased (3.10). All 50 States and the
District of Columbia have enacted laws
prohibiting the sale and distribution of
tobacco products to youth under 18
years of age (3.13). Although these laws
are often not strictly enforced (16),
States are now required to demonstrate
that they are enforcing their tobacco
access laws in a manner that will reduce
sales violation rates (17). The number of
States with laws banning cigarette
vending machines in areas accessible to
minors increased from 12 in 1995 to 21
in 1999, but this increase was far shy of
the target of all 50 States and the
District of Columbia (3.26).
Cigarette smoking prevalence
among adults (3.4) decreased for all
Healthy People 2000 Final Review 93
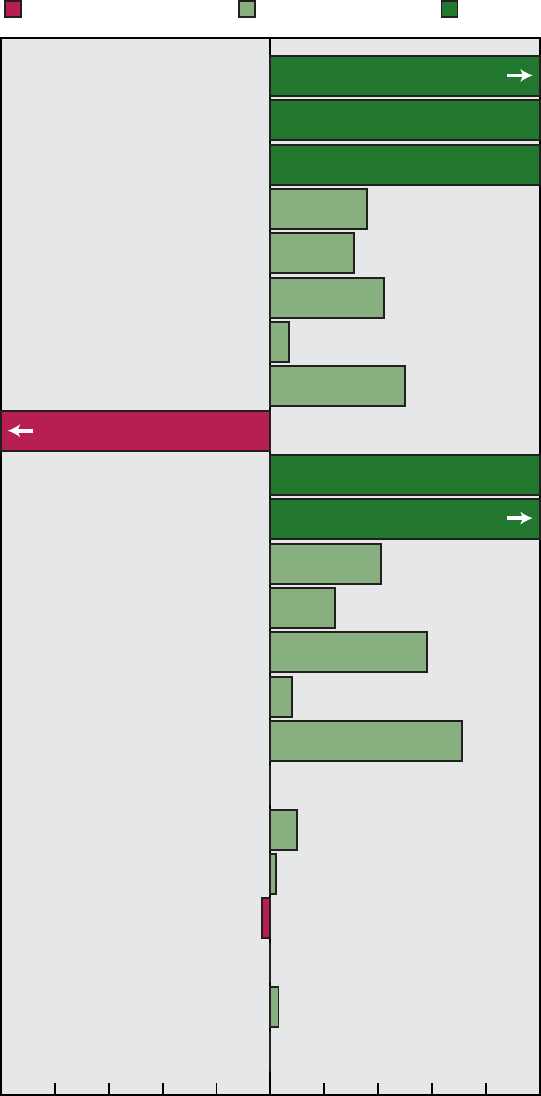
3.1 Coronary heart disease deaths
3.2 Lung cancer deaths
3.3 COPD deaths
3.4 Cigarette smoking: 18 years and over
Male
Female
3.5 Smoking initiation
3.6 Smoking cessation attempts
3.7 Smoking cessation during pregnancy
3.8 Children's exposure to smoke at home
3.9 Smokeless tobacco use: Male 12-17 years
Male 18-24 years
3.10 School districts providing: Tobacco-free environments
Anti-smoking education: High school
Middle school
3.11 Worksite smoking policies: Bans
3.12 States with clean indoor air laws: Private workplaces
Public workplaces
Restaurants
Hospitals
Public transportation
Day care centers
Grocery stores
Moved away from target Moved toward target Met target
Chart continues onto next page – see notes at the end of the chart.
36%
31%
42%
7%
50%
41%
24%
58%
8%
71%
No change
No change
No change
2%
-3%
10%
3%
Percent of target achieved
-100 -80 -60 -40 -20 0 20 40 60 80 100
109%
100%
169%
Met*
Met*
-119%
94 Healthy People 2000 Final Review
Figure 3. Final status of Tobacco objectives

Figure 3. Final status of Tobacco objectives-Con.
3.13 States with youth tobacco laws
3.14 States with plans to reduce tobacco use
3.17 Oral cancer deaths: Male 45-74 years
Female 45-74 years
3.18 Stroke deaths
3.19 Average age at first use, 12-17 years: Cigarettes
Alcohol
Marijuana
3.20 Use in the past month: Alcohol, 12-17 years
Alcohol, 18-20 years
Marijuana, 12-17 years
Marijuana, 18-25 years
Cocaine, 12-17 years
3.21 High school seniors' perception of disapproval:
Heavy alcohol use
Occasional marijuana use
Cocaine, 18-25 years
Cigarette, 12-17 years
Cocaine use once or twice
Smoking one or more packs of cigarettes a day
3.22 High school seniors' perception of harm:
Heavy alcohol use
Regular marijuana use
Cocaine use once or twice
Using smokeless tobacco regularly
3.23 Tobacco excise tax: Cigarettes
Smoking one or more packs of cigarettes a day
Smokeless tobacco
3.25 States with preemptive clean air indoor air laws
3.26 States with cigarette vending machine laws
Moved away from target Moved toward target Met target
*This objective has met its target. A progress quotient could not be calculated.
NOTE: Complete tracking data are shown in table 3. Progress quotients are not calculated for objectives 3.2, 3.3, 3.15, 3.16, and 3.24.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information. COPD is chronic
obstructive pulmonary disease.
36%
51%
80%
-68%
-3%
-14%
-4%
11%
-50%
30%
69%
4%
27%
9%
No change
No change
8%
17%
3%
23%
Percent of target achieved
-100 -80 -60 -40 -20 0 20 40 60 80 100
100%
100%
103%
200%
67%
20%
112%
-132%
-161%
Healthy People 2000 Final Review 95
groups by 1993 and then leveled off.
Smoking cessation attempts by adults
(3.6) increased; however, they peaked in
1991 and have dropped since then.
While the prevalence of smoking among
pregnant women is relatively low and
has been decreasing (18), smoking
cessation among pregnant women who
smoke (3.7) declined considerably over
the decade. Children’s exposure to
smoke at home (3.8) was reduced over
the course of the decade by about
50 percent.
The number of worksites with
smoking policies increased, as did the
number with actual bans (3.11). During
the Healthy People 2000 midcourse
review, the target for this objective was
increased from 75 percent of worksites
to 100 percent. Had it not been changed,
this objective would now be met.
All States now have tobacco control
plans (3.14), while in 1989 only 12
States had such plans. The number of
States with comprehensive laws for
clean indoor air (3.12) increased, but
only slightly. The objective to increase
the average tobacco excise tax moved
from a baseline of 31.4 percent to
22.1 percent at the end of the decade,
away from the target of 50 percent of
the average retail price of all cigarettes
and smokeless tobacco (3.23).
Summary of Progress
Data are available to assess the
progress of 24 of 26 objectives in the
tobacco priority area. Seven objectives
(3.1–3.3, 3.8, 3.13, 3.14, and 3.17) met
their targets. Ten objectives (3.4–3.6,
3.9–3.11, 3.16, 3.18, 3.19, and 3.26)
showed progress. One objective (3.25)
showed no change from the baseline
measure. Five objectives (3.12,
3.20–3.22, and 3.23) showed mixed
progress among the multiple measures
used to track the objectives. One
objective (3.7) moved away from the
target. Progress for two objectives (3.15
and 3.24) could not be assessed because
data were not available. See table 3 for
the tracking data for the objectives in
this priority area and figure 3 for a
quantitative assessment of progress.
Discussion
While significant progress was
made in reaching the year 2000 targets
for this priority area’s objectives, certain
challenges and barriers may have
contributed to preventing achievement
of all objectives. The 1990s saw a great
deal of change and progress in the area
of tobacco control. However, some
changes (for example, the Master
Settlement Agreement, the FDA ruling,
etc.) came during the latter half of the
decade and, therefore, did not have an
appreciable effect on measures of the
Healthy People 2000 objectives.
The past decade saw a number of
demographic changes among tobacco
users. The 24th Surgeon General’s
Report, Tobacco Use Among U.S.
Racial/Ethnic Minority Groups, revealed
rapid increases in smoking by teenagers
from minority groups, a trend that
threatens to reverse the progress made
during the early 1990s against lung
cancer among racial and ethnic minority
populations. Although their smoking
rates remain considerably lower than
those of whites (whose rates increased
and then began to level off during the
10 years), cigarette smoking among
African American and Hispanic
adolescents increased in the 1990s after
several years of substantial declines
(19).
The 1990s also saw changes in
tobacco products being used. For
example, there has been an increase in
the use of products other than cigarettes.
The most significant increase is in the
use of cigars. In 1999, approximately
3.7 billion cigars were smoked in the
United States (20). Cigar smoking, once
primarily an activity of older men (21),
is now an activity of teenagers, with an
estimated 6 million 14–19 year olds
reporting that they smoked a cigar
during the previous year (22). In
addition, approximately 2.7 percent of
middle school students and 6.6 percent
of high school students reported using
smokeless tobacco in 1999 (13) . Other,
more novel tobacco products are also
being used, especially by young people.
Data from 1999 indicate that use of
products such as bidis (flavored
cigarettes originally from India) and
kreteks (clove cigarettes) was
2.4 percent and 1.9 percent, respectively,
among middle school students (13).
Among high school students,
approximately 5 percent and 5.8 percent,
respectively, use bidis and kreteks (13).
Financial and other resources for
tobacco control have improved in recent
years. In 1999, the U.S. Department of
Health and Human Services (HHS) and
other organizations in the health
community developed a national tobacco
control program. Prior to that time,
activities to prevent tobacco use and
promote smoking cessation were
supported by smaller scale efforts
sponsored by, among others, the
National Cancer Institute, the Centers
for Disease Control and Prevention
(CDC), and the Robert Wood Johnson
Foundation. In 1998 settlements
between State attorneys general and the
tobacco companies to recover Medicaid
costs resulting from tobacco-related
health problems provided additional
resources for State-based tobacco
control, although the use of this money
for tobacco control purposes differs by
State. The amount of money from the
Master Settlement Agreement that is
allocated to tobacco control efforts in
each State may affect the success of the
Healthy People 2010 objectives.
In 1996 the Federal government
issued evidence-based guidelines for
smoking cessation (23) . The guidelines
were updated in 2000 and are
considered the standard of care for
tobacco use treatment (15). Five
pharmacologic treatments for nicotine
dependence are now approved by the
Food and Drug Administration (FDA).
Access to treatment has increased
because two of the products, the
nicotine patch and gum, are
over-the-counter therapies (24).
The 1990s saw the implementation
of a number of regulations, some of
which succeeded and others that failed.
In 1995 measures to restrict young
people’s access to tobacco products
were instituted in earnest through
regulations proposed by the FDA in
1995 and, in 1996, the Synar
Amendment to the Alcohol, Drug
Abuse, and Mental Health
Administration Reorganization Act.
Recently, numerous health associations,
nongovernmental organizations, and
communities have also focused
significant resources on preventing
young people from starting to use
tobacco.
In 1994 an FDA advisory
committee concluded that tobacco is
addictive and that nicotine is the drug in
tobacco that causes addiction. At that
time, an FDA regulation set 18 years as
the minimum age at which a person can
buy tobacco products and retailers are
now required to check the identification
of potential buyers under the age of 27
before selling them tobacco.
In the latter half of the 1990s, the
Courts made several rulings related to
the FDA’s authority to regulate tobacco
96 Healthy People 2000 Final Review
and its advertising. However, early in
2000 the Supreme Court ruled against
the FDA, finding it lacked authority to
regulate tobacco products.
In spite of Federal legislation in the
mid and late 1990s to prohibit smoking
in federally funded facilities and State
legislation restricting smoking in public
places, only 13 States limit smoking in
public places and in worksites and few
reported completely banning smoking or
limiting it to separately ventilated areas
in private workplaces or restaurants
(14). As of 1998, only one State
required worksites to designate
separately ventilated smoking areas and
only 20 States required worksites to
separately designate smoking areas at all
(14). Meanwhile, a growing number of
workers are demanding protection from
involuntary exposure to smoke and
increasingly more employers are
responding to their employees’ concerns
about involuntary exposure to smoke.
The Federal government took steps
in 1997 to increase the excise tax on
cigarettes. States also have authority to
increase taxes. Those States that have
increased taxes most significantly have
seen drops in smoking rates in their
States. Raising taxes is one of the most
effective interventions to decrease
tobacco use (1).
The tobacco industry continues to
promote smoking (1) . Cigarettes remain
one of the most heavily marketed
consumer products. Even though
cigarette advertisements are prohibited
on television and radio, the decade saw
an increasing trend in the amount spent
on advertising and promotion. The
Federal Trade Commission’s most recent
report shows that the tobacco industry
spent $6.73 billion on advertising and
promotion in 1998, a 19 percent increase
from 1997 (25). Several States, Federal
agencies, and other organizations
conduct media campaigns against
tobacco use. Although media campaigns
have been shown to decrease tobacco
initiation and use, resources for
campaigns have been limited. Some
changes are on the horizon since
settlements made between the State
attorneys general and the tobacco
companies in 1998–99 included several
important restrictions on tobacco
advertising and promotion to young
people.
Finally, in spite of the fact that the
U.S. Surgeon General has indicated in a
report that implementing effective
educational programs for preventing
tobacco use could postpone or prevent
smoking onset in 20 percent to
40 percent of U.S. adolescents, data
suggest that evidence-based curricula
and national guidelines have not been
widely adopted (1). Less than 5 percent
of schools nationwide are implementing
the major components of CDC’s
Guidelines for School Health Programs
to Prevent Tobacco Use and Addiction
(1).
Transition to Healthy People
2010
The year 2010 objectives are
grouped in the same categories as those
used in Healthy People 2000 (tobacco
use in population groups, cessation and
treatment, exposure to secondhand
smoke, and social and environmental
changes). Each Healthy People 2000
tobacco use objective was evaluated
based on its policy and program
importance and data availability, and
public comments were taken into
account in deciding what objectives to
include in the 2010 chapter.
To avoid duplication of objectives
across chapters, some topics previously
included in the Healthy People 2000
Tobacco Use chapter are only in the
Healthy People 2010 focus areas that
were considered their ‘‘primary homes’’
(for example, the objectives for coronary
heart disease deaths and stroke deaths
are located only in the Heart Disease
and Stroke chapter). Seven Healthy
People 2000 tobacco use objectives
moved to other focus areas in Healthy
People 2010. Prevalence measures were
expanded to include cigarettes, spit
tobacco, cigars, and other products. New
objectives for Healthy People 2010
address youth, not just adult, smoking
cessation; nonsmokers exposed to
tobacco smoke; laws on smoke-free
indoor air for territories and Tribes;
suspension or revocation of State retail
licenses for violations of laws
prohibiting the sale of tobacco to
minors; tobacco advertising and
promotions that influence adolescents
and young adults; comprehensive,
evidence-based tobacco control
programs; and the establishment of a
regulatory structure to monitor toxicity
of tobacco products.
Tobacco-related objectives also
appear in other Healthy People 2010
chapters (for example, Access to Quality
Health Services; Maternal, Infant, and
Child Health; Environmental Health;
and Oral Health). Age ranges and
subgroups were expanded in Healthy
People 2010 to better focus on
population disparities.
Tobacco use is the topic of two of
the 10 Leading Health Indicators (LHIs),
which Healthy People 2010 introduces
to serve as a barometer of the Nation’s
health. Two objective measures from the
Healthy People 2010 Tobacco Use focus
area—cigarette smoking by adolescents
and cigarette smoking by adults—are
used to measure the LHI on tobacco
use, and an additional objective
addressing nonsmokers exposed to
environmental tobacco smoke is used to
help measure the LHI for environmental
quality.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the changes between the
two decades of objectives, reflecting
new knowledge and direction for this
area.
Data Issues
Definitions
Coronary heart disease deaths (3.1)
were defined by ICD–9 codes 402,
410–414, and 429.2. These are different
from the codes used to define the
category ‘‘Diseases of heart’’ which
often appears in published tables (see
Appendix table IV).
Chronic obstructive pulmonary
disease (COPD) deaths (3.3) include
deaths due to chronic bronchitis,
emphysema, asthma, and other chronic
obstructive pulmonary diseases and
allied conditions.
Beginning in 1992, the definition of
current smoker (3.4) was modified to
specifically include persons who smoked
only some days. Prior to 1992, a current
smoker was defined by the questions:
‘‘Have you ever smoked 100 cigarettes
in your lifetime?’’ and ‘‘Do you smoke
now?’’ In 1992, cigarette smoking data
were collected for a half-sample of the
National Health Interview Survey
(NHIS) with half the respondents
(one-quarter sample) using these two
smoking questions and the other half of
respondents (one-quarter sample) using
a revised smoking question: ‘‘Do you
smoke every day, some days, or not at
all?’’ in place of the second question.
The 1992 estimate combines data
collected using both sets of questions.
Healthy People 2000 Final Review 97
Updates after 1992 are based completely
on the revised definition, which is
considered a more complete estimate of
smoking prevalence. The effect of the
new definition is a small increase in the
prevalence of smoking.
The baseline for objective 3.7
(cessation of cigarette smoking early in
pregnancy, with abstinence throughout
pregnancy) was from a 1986 telephone
interview of white women selected from
the respondents to the 1985 National
Health Interview Survey (9). Beginning
with 1991, progress toward the target
was tracked using periodic supplements
to the NHIS. The 1985 and 1991
surveys used different definitions for
smoking before pregnancy and for the
duration of quitting during pregnancy.
The 1991 measure, which focused on
women who quit during the first
trimester, is closer to the intent of the
objective but not comparable with the
1985 baseline that included women who
quit at any time during their last
pregnancy resulting in a live birth in the
previous 5 years. The data for 1998 are
defined similarly to the 1991 data. The
denominator is women ages 18–49 years
who had a live birth in the previous 5
years and smoked at any time during
their pregnancy with their last child and
the numerator is those women in the
denominator who quit smoking in the
first trimester and did not begin
smoking again during the pregnancy.
For objective 3.8 (children’s
exposure to tobacco smoke at home),
the numerator was the number of
children 6 years and under living in
households with a household resident
who smoked inside the home 4 or more
days each week. The denominator was
the number of households with children
ages 6 years and under.
Objective 3.25 sought to reduce the
number of States with preemptive clean
indoor air laws. Preemptive laws prevent
local jurisdictions from enacting more
stringent restrictions than the State law
or restrictions that vary from the State
law (26).
Data Sources
All data for objective 3.11 (worksite
smoking policies) are from telephone
surveys of nongovernment worksites of
50 or more employees. The 1985 and
1992 data for objective 3.11 were from
the Public Health Service-sponsored
National Survey of Worksite Health
Promotion Activities. Worksites were
sampled because different worksites
within the same company could have
different sets of health promotion
activities. Both active (for example,
classes) and passive (for example,
brochures) methods were counted as
worksite health promotion activities. The
1995 update was from the
CDC-sponsored Worksite Benchmark
Survey, which used a methodology very
similar to the 1992 survey, but did not
include passive methods of health
promotion (27,28). The 1998–99 data
are from the National Worksite Health
Promotion Survey. Like the 1992
survey, the designated respondent was
asked if the worksite had a formal
smoking policy that prohibits or
severely restricts smoking at the
worksite or on the job (29).
The National Household Survey on
Drug Abuse (NHSDA) was used to
measure objectives 3.9, 3.19, and 3.20
regarding substance use among
adolescents and young people.
Beginning in 1991, the survey was
expanded to include college students
living in residence halls. In 1994, an
improved questionnaire and editing
procedures were introduced, which
affect comparability with previous years.
Additionally, in 1994, data were
collected for cigarettes using a
self-administered questionnaire, unlike
previous years when questions regarding
cigarette smoking were asked by the
interviewers. This change in
questionnaire administration greatly
increased the cigarette-use estimates
among adolescents, most likely due to
the increased confidentiality of the new
methodology. The trend data for all
substances in objective 3.20 have been
recalculated to adjust for these
differences and to produce comparable
estimates from 1988 to 1997.
Data for objective 3.16, cessation
counseling and followup by clinicians,
were obtained from several different
surveys, making statements about trends
somewhat problematic. The 1986
baselines for dentists were obtained
from the Statewide Survey of Dentists
in Vermont, and the updates are from
the National Survey of Dentists,
sponsored by the University of Florida.
The 1986 baseline for internists is from
the American College of Physicians
(ACP) Membership Survey of
Prevention Practices in Adult Medicine.
The sampling frame for internists in the
1992 Primary Care Provider Surveys
(PCPS) contained a random stratified
sample of ACP members drawn from
four geographic regions with
oversampling of female members,
yielding an initial sample of 1,200
internists. Additional provider groups
sampled in the 1992 PCPS included
pediatricians, nurse practitioners,
obstetricians/gynecologists, and family
physicians. Response rates varied from
50 to 80 percent across these groups.
The PCPS data on inquiry for objective
3.16 refer to the proportion of providers
who routinely provided service to
81–100 percent of their clients. Data on
counseling about smoking cessation
represent the proportion of providers
who routinely delivered these services to
81–100 percent of their clients who
needed the intervention. Reporting of
counseling could have been independent
of the assessment made by the clinician.
The American College of
Physicians’ Prevention in Primary Care
Study (PPCS) was conducted in
1997–98 to update data from the 1992
PCPS. The design and items included in
the 1997–98 study were similar to the
PCPS, but a slightly different sampling
frame was used and some items
included in the 1992 surveys were not
included in the PPCS. The providers
were sampled from listings of all
licensed, active practitioners in the
United States whose practices were at
least 50 percent primary care. Because
of low response rates from the other
provider groups, updates are available
only for nurse practitioners.
Data Comparability
Information on objective 3.9
(smokeless tobacco use by males ages
12–24 years) was tracked by two
surveys. Males ages 12–17 years were
tracked by the NHSDA. In this survey,
smokeless tobacco use was defined as
any use of snuff or chewing tobacco in
the preceding month. For males ages
18–24 years, information was obtained
from the NHIS. The NHIS defines a
smokeless tobacco user as someone who
has used either snuff or chewing
tobacco at least 20 times and who
currently uses either of these substances
every day or some days, at the time of
the survey. Information for males ages
18–25 years was also available from the
NHSDA using the same definition as for
those ages 12–17 years. According to
the NHSDA, smokeless tobacco use
among males ages 18–24 years showed
a downward trend similar to that
observed from the NHIS. The smokeless
98 Healthy People 2000 Final Review
tobacco use prevalence estimate from
NHSDA was higher than the NHIS
estimate (11.7 percent compared with
8.2 percent, respectively, in 1992).
Differences between the NHSDA and
the NHIS may be due to differences in
the definition of smokeless tobacco use
between the two surveys and/or
methodological differences in survey
administration.
References
1. Department of Health and Human Services.
Reducing tobacco use: A report of the Surgeon
General. Atlanta, Georgia: Centers for Disease
Control and Prevention, National Center for
Chronic Disease Prevention and Health Promotion,
Office on Smoking and Health. 2000.
2. Centers for Disease Control and Prevention.
Smoking-attributable mortality and years of
potential life lost—United States, 1990. MMWR
42(33):645–9. 1993.
3. Centers for Disease Control and Prevention.
Achievements in Public Health, 1900–1999:
Tobacco use—United States, 1900–1999. MMWR
48(43):986. 1999.
4. Centers for Disease Control and Prevention.
Smoking-attributable mortality and years of
potential life lost—United States, 1984 (with
editorial note, 1997). MMWR 46(20):444–51.
1997.
5. Centers for Disease Control and Prevention.
Medical-care expenditures attributable to cigarette
smoking, United States, 1993. MMWR
43(26):469–72. 1994.
6. Herdman R, Hewitt M, Laschober M.
Smoking-related deaths and financial costs: Office
of Technology Assessment estimates for 1990
(Congressional Testimony). Washington: Office of
Technology Assessment. 1993.
7. Centers for Disease Control and Prevention.
Medical-care expenditures attributable to cigarette
smoking during pregnancy—United States, 1995.
MMWR 46(44):1048–50. 1997.
8. Department of Health and Human Services. The
health benefits of smoking cessation. Atlanta,
Georgia: Centers for Disease Control and
Prevention, National Center for Chronic Disease
Prevention and Health Promotion, Office on
Smoking and Health. 1990.
9. Fingerhut LA, Kleinman JC, Kendrick JS.
Smoking before, during, and after pregnancy. Am
J Public Health 80:541–4. 1990.
10. Chomitz VR, Cheung LW, Lieberman E. The
role of lifestyle in preventing low birth weight.
Future Child 5(1):121–38. 1995.
11. U.S. Environmental Protection Agency.
Respiratory health effects of passive smoking:
Lung cancer and other disorders. Washington: U.S.
Environmental Protection Agency, Office of
Research and Development, Office of Air and
Radiation. 1992.
12. Centers for Disease Control and Prevention.
Trends in Cigarette Smoking Among High School
Students United States, 1991–1999. MMWR
49(33):755. 2000.
13. Centers for Disease Control and Prevention.
Tobacco Use Among Middle and High School
Students United States, 1999. MMWR 48(3):
49–53. 2000.
14. Department of Health and Human Services.
Healthy people 2010. 2d ed. With understanding
and improving health and objectives for improving
health. 2 vols. Washington: U.S. Government
Printing Office. 2000.
15. Giovino GA, Shelton DM, Schooley MW.
Trends in cigarette smoking cessation in the
United States. Tobacco Control 1993; 2
(Supplement): S3–S10. 1993.
16. Department of Health and Human Services.
Preventing tobacco use among young people: A
report of the Surgeon General. Atlanta, Georgia:
Public Health Service, Centers for Disease Control
and Prevention, National Center for Chronic
Disease Prevention and Health Promotion, Office
on Smoking and Health. 1994.
17. Department of Health and Human Services.
Substance Abuse and Mental Health
Administration, Center for Substance Abuse
Prevention. Substance Abuse Prevention and
Treatment Block Grant Program, State Annual
Synar Reports. 1998–99.
18. Kvale K, Glysch RL, Gothard M, et al. Trends
in smoking during pregnancy, Wisconsin 1990 to
1996. Wisconsin Med J 99(2):63–7. 2000.
19. Department of Health and Human Services.
Tobacco use among U.S. racial/ethnic minority
groups: A report of the Surgeon General. Atlanta,
Georgia: Centers for Disease Control and
Prevention, National Center for Chronic Disease
Prevention and Health Promotion, Office on
Smoking and Health. 1998.
20. Department of Agriculture. Tobacco situation
and outlook report. TBS–246. Washington:
Economic Research Service. 2000.
21. Centers for Disease Control and Prevention.
Surveillance for selected tobacco-use
behaviors—United States, 1900–1994. MMWR 43
(SS–3). 1994.
22. Centers for Disease Control and Prevention.
Cigar smoking among teenagers—United States,
Massachusetts, and New York, 1996. MMWR
46(20):433–40. 1997.
23. Smoking Cessation Clinical Practice Guideline
Panel and Staff. Agency for Health Care Policy
and Research Smoking Cessation Clinical Practice
Guideline. JAMA 275:1270–80. 1996.
24. Shiffman S, Gitchell J, Pinney J, et al. Public
health impact of over-the-counter nicotine
medications. Tobacco Control 6:306–10. 1997.
25. Federal Trade Commission. Federal Trade
Commission report to Congress for 1998 pursuant
to the Federal Cigarette Labeling and Advertising
Act. Washington: Federal Trade Commission.
1999.
26. Centers for Disease Control and Prevention.
State laws on tobacco control: United States, 1995.
MMWR 44(SS-6):24. 1995.
27. Department of Health and Human Services.
Business Responds to AIDS Benchmark Survey:
technical report. Atlanta, Georgia: Public Health
Service, Centers for Disease Control and
Prevention. 1996.
28. Centers for Disease Control and Prevention.
Cancer screening offered by worksites—United
States, 1992 and 1995. MMWR 46(19):421–4.
1997.
29. Department of Health and Human Services.
Tracking healthy people 2010. Washington: U.S.
Government Printing Office. 2000.
Healthy People 2000 Final Review 99

Table 3. Tobacco objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
3.1* Coronary heart disease deaths (age
adjusted per 100,000) ................. 1987 135 122 118 114 114 110 108 105 100 97 - - - 100
a. Black ........................... 1987 168 158 156 151 154 147 147 140 136 133 - - - 115
3.2* Slow the rise in lung cancer deaths (age
adjusted per 100,000) ................. 1987 38.5 39.9 39.6 39.3 39.3 38.7 38.3 37.8 37.3 36.9 - - - 42
a. Female.......................... 1990 25.6 . . . 25.8 26.3 26.5 26.6 26.9 26.8 26.9 27.0 - - - 27
b. Black male ....................... 1990 86.1 . . . 83.1 81.2 80.7 77.6 75.7 73.4 70.5 68.5 - - - 91
3.3 Slow the rise in chronic obstructive
pulmonary disease deaths (age adjusted
per 100,000) ........................ 1987 18.9 19.7 20.1 19.9 21.4 21.0 20.8 21.0 21.1 21.3 - - - 25
3.4* Cigarette smoking prevalence
People 18 years and over ............... 1987 29% 25% 26%
1
27% 25% 26% 25% - - -
‡
25%
‡
24% - - - 15%
Male ............................. 1987 31% 28% 28%
1
29% 28% 28% 27% - - -
‡
28%
‡
26% - - - 15%
Female ........................... 1987 27% 23% 23%
1
25% 22% 23% 23% - - -
‡
22%
‡
22% - - - 15%
a. People with high school education or
less
20 years and over ................ 1987 34% 31% 31%
1
32% 30% 31% 30% - - -
‡
31%
‡
31% - - - 20%
b Blue-collar workers 18 years and over . . 1987 41% 36% 36%
1
36% 34% 39% 36% - - -
‡
37%
‡
36% - - - 20%
c. Military personnel .................. 1988 42% - - - - - -
1
35% --- --- 32% --- --- 30% --- 20%
d. Black 18 years and over ............. 1987 33% 26% 29%
1
28% 26% 27% 26% - - -
‡
27%
‡
25% - - - 18%
e. Hispanic 18 years and over .......... 1987 24% 23% 20%
1
21% 20% 20% 18% - - -
‡
20%
‡
19% - - - 15%
f. American Indian/Alaska Native 18 years
and over ....................... 1979-87
2
42-70% 38% 31%
1
40% 39% 40% 35% - - -
‡
32%
‡
37% - - - 20%
g. Southeast Asian male ............... 1984-88 55%
3
35%
3
36-41% - - - - - - - - - - - - - - - - - - - - - - - - 20%
h. Females of reproductive age
(18-44 years) .................... 1987 29% 26% 27%
1
28% 26% 27% 26% - - -
‡
26%
‡
25% - - - 12%
i. Pregnant females .................. 1985 25% 19% 20% - - - 20% - - - 18% 14% 13% 13% - - - 10%
j. Females who use oral contraceptives . . . 1983 36%
4
26% --- --- --- --- 24% --- --- --- --- 10%
3.5 Smoking initiation by children and
adolescents (proxy 20-24 years) ........ 1987 30% 26% 24% 28% 27% 30% 26% - - -
‡
31%
‡
29% - - - 15%
a. Lower socioeconomic status people
20-24 years
5
.................... 1987 40% 35% 33% 38% 38% 39% 31% - - -
‡
44%
‡
42% - - - 18%
3.6 Smoking cessation attempts ........... 1986 34% - - - 56% 46% 47% 46% 46% - - -
‡
44%
‡
42% - - - 50%
3.7 Smoking cessation during pregnancy
(18-49 years) ....................... 1985
6
3 9 % --- 1 6 % --- --- --- --- --- --- 1 4 % --- 6 0 %
a. Females with less than a high school
education ....................... 1985
6
28% - - -
7
--- --- --- --- --- --- ---
7
--- --- 45%
3.8* Children’s exposure to smoke at home
(6 years and under) .................. 1986 39% - - - 32% - - - 27% 27% - - - - - - - - - 20% - - - 20%
3.9* Smokeless tobacco use
Male 12-17 years ..................... 1988 6.6% - - - 5.3% 4.8% 3.9% 5.1% 4.9% 3.5% 3.7% 2.2% - - - 4%
Male 18-24 years ..................... 1987 8.9% - - - 9.9% 8.2% 7.8% 6.9% - - - - - - - - - 6.9% - - - 4%
a. American Indian/Alaska Native
18-24 years ...................... 1986-87
2
18-64% - - -
7
---
7
---
7
---
7
--- --- --- ---
7
--- --- 10%
See footnotes and key at end of table.
100 Healthy People 2000 Final Review

Table 3. Tobacco objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
3.10 Tobacco-use prevention education and
tobacco-free schools
School districts providing tobacco-free
environments .......................
1988 17% - - - - - - - - - - - -
8
36.5% - - - - - - - - - - - - - - - 100%
School districts providing anti-smoking
education
High school ........................ 1988 78% - - - - - - - - - - - - 90.7% - - - - - - - - - - - - - - - 100%
Middle school ...................... 1988 81% - - - - - - - - - - - - 82.5% - - - - - - - - - - - - - - - 100%
Elementary school................... 1988 75% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 100%
3.11 Worksites with smoking policies
Policy that bans smoking or limits it to
separately ventilated areas
50 or more employees ................. 1985 27% - - - - - - 59% - - - - - - - - - - - - - - - - - -
9
79% 100%
Any smoking policy
Medium and large companies .......... 1987 54% - - - 85% - - - - - - - - - - - - - - - - - - - - - - - - 100%
50 or more employees................ ... --- --- --- 86% --- --- 87% --- --- --- --- ...
3.12 Number of States with comprehensive
laws for clean indoor air
10
Private workplaces .................... 1995
§
1 ... ... ... ... ... ... 1 1 1 --- 51
Public workplaces ..................... 1995
§
9 ... ... ... ... ... ... 9 12 13 --- 51
Restaurants ......................... 1995 2 . . . . . . . . . . . . . . . . . . 3 3 3 - - - 51
Public transportation ................... 1995
§
17 ... ... ... ... ... ... 17 17 16 --- 51
Hospitals............................ 1995
§
8 ... ... ... ... ... ... 8 8 --- --- 51
Day care centers ..................... 1995 21 . . . . . . . . . . . . . . . . . . 21 21 22 - - - 51
Grocery stores ....................... 1995
§
4 ... ... ... ... ... ... 4 4 4 --- 51
3.13 Number of States with tobacco product
sale and distribution to youth laws
10
.... 1990 45 . . . 50 50 51 51 51 51 51 51 51 51
Number of States enforcing laws to achieve
buy rates no higher than 20 percent ...... ... --- --- --- --- --- --- --- ---
11
4 --- --- ...
3.14 Number of States with plans to reduce
tobacco use........................ 1989 12 - - - - - -
10
35 ---
10
39 ---
10
51
10
51
10
51
10
51 51
3.15 Tobacco product advertising and
promotion to youth ..................
1990
Minimal
restrictions . . . - - - - - - - - - - - - - - - - - - - - - - - - - - -
Eliminate
or severely
restrict
3.16 Cessation counseling and followup by
clinicians
Percent of clinicians routinely providing
service to at least 75% of patients
Inquiry about smoking
General dentists .................... 1986 26% - - - - - - - - - - - - 32.8% - - - - - - - - - - - - - - - 75%
Advised patients about smoking (among
patients reporting smoking)
General dentists .................... 1986 35% - - - - - - - - - - - - 64.8% - - - - - - - - - - - - - - - 75%
Internists (including sub-specialists) ...... 1986 52% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 75%
See footnotes and key at end of table.
Healthy People 2000 Final Review 101

Table 3. Tobacco objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Inquiry about smokeless tobacco use
General dentists .................... ... --- --- --- --- --- 14.4% - - - - - - - - - - - - - - - . . .
Advised patients about smokeless tobacco
use (among patients reporting smokeless
tobacco use)
General dentists .................... ... --- --- --- --- --- 75.0% - - - - - - - - - - - - - - - . . .
Percent of clinicians routinely providing
service to 81–100% of patients
Inquiry about tobacco use
Pediatricians ....................... ... --- --- --- 33% --- --- --- --- ---
12,13
--- --- 75%
Nurse practitioners .................. ... --- --- --- 51% --- --- --- --- ---
12
66% - - - 75%
Obstetricians/gynecologists ............ ... --- --- --- 49% --- --- --- --- ---
12,13
--- --- 75%
Internists .......................... ... --- --- --- 75% --- --- --- --- ---
12,13
--- --- 75%
Family physicians ................... ... --- --- --- 59% --- --- --- --- ---
12,13
--- --- 75%
Discussion of strategies to quit smoking .... ---
Pediatricians ....................... ... --- --- --- 19% --- --- --- --- ---
12,13
--- --- 75%
Nurse practitioners .................. ... --- --- --- 20% --- --- --- --- ---
12
39% - - - 75%
Obstetricians/gynecologists ............ ... --- --- --- 28% --- --- --- --- ---
12,13
--- --- 75%
Internists .......................... ... --- --- --- 50% --- --- --- --- ---
12,13
--- --- 75%
Family physicians ................... ... --- --- --- 43% --- --- --- --- ---
12,13
--- --- 75%
3.17 Oral cancer deaths (per 100,000)
Male 45–74 years ..................... 1987 13.6 13.4 12.7 12.2 12.1 11.1 11.0 10.7 10.3 10.4 - - - 10.5
Female 45–74 years ................... 1987 4.8 4.6 4.6 4.3 4.2 4.0 3.9 3.5 3.5 3.4 - - - 4.1
a. Black male 45–74 years ............. 1990 29.4 . . . 26.9 27.3 26.2 25.2 23.4 22.6 20.6 21.0 - - - 26.0
b. Black female 45–74 years ............ 1990 6.9 . . . 6.9 6.0 5.8 5.7 6.4 5.0 5.2 4.6 - - - 6.9
3.18 Stroke deaths (age adjusted per 100,000) . . 1987 30.4 27.7 26.8 26.2 26.5 26.5 26.7 26.4 25.9 25.1 - - - 20.0
a. Black ........................... 1987 52.5 48.4 46.8 45.0 45.0 45.4 45.0 44.2 42.5 41.4 - - - 27.0
3.19 Average age of first use (adolescents
12-17 years)
Cigarettes ........................... 1988 11.6 11.5 11.5 11.7 11.7 12.2 12.3 12.4 12.4 12.4 - - - 12.6
Alcohol ............................. 1988 13.1 12.8 12.6 13.0 12.9 12.8 12.6 13.1 13.1 13.1 - - - 14.1
Marijuana ........................... 1988 13.4 13.4 13.5 13.8 13.9 14.1 13.8 14.4 13.7 13.7 - - - 14.4
3.20 Use in past month by adolescents and
young adults
14
Alcohol
12-17 years ....................... 1988 33.4% 32.5% 27.0% 20.9% 23.9% 21.6% 21.1% 18.8% 20.5% 19.1% - - - 12.6%
18-20 years ....................... 1994 54.6% . . . . . . . . . . . . . . . 54.1% 50.1% 53.4% 53.5% - - - 29.0%
Hispanic 12-17 years ................ 1988 31.9% 24.2% 28.3% 20.3% 22.0% 18.3% 18.7% 19.9% 18.8% 18.9% - - - 12.0%
Marijuana
12-17 years ....................... 1988 5.4% 4.4% 3.6% 3.4% 4.0% 6.0% 8.2% 7.1% 9.4% 8.3% - - - 3.2%
18-25 years ....................... 1988 15.3% 12.7% 12.9% 10.9% 11.1% 12.1% 12.0% 13.2% 12.8% 13.8% - - - 7.8%
See footnotes and key at end of table.
102 Healthy People 2000 Final Review

Table 3. Tobacco objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Cocaine
12-17 years ....................... 1988 1.2% 0.6% 0.4% 0.3% 0.4% 0.3% 0.8% 0.6% 1.0% 0.8% - - - 0.6%
18-25 years ....................... 1988 4.8% 2.3% 2.2% 2.0% 1.6% 1.2% 1.3% 2.0% 1.2% 2.0% - - - 2.3%
Hispanic 12-17 years ................ 1988 1.4% 2.0% 1.4% 1.3% 1.1% 0.7% 0.8% 1.1% 1.0% 1.4% - - - 0.6%
Hispanic 18-25 years ................ 1994 2.2% . . . . . . . . . . . . . . . 1.1% 2.1% 1.5% 2.7% - - - 1.0%
Cigarettes
12-17 years ....................... 1988
§
22.7% 22.4% 20.9% 18.4% 18.5% 18.9% 20.2% 18.3% 19.9% 18.2% - - - 6.0%
3.21 Perception of social disapproval by high
school seniors
Heavy use of alcohol .................. 1989 56.4% 59.0% 58.1% 60.8% 58.5% 59.1% 58.0% 57.8% 56.4% 55.5% 57.6% 70%
Occasional use of marijuana ............. 1989 71.1% 76.4% 75.8% 79.2% 73.8% 69.1% 65.4% 63.1% 59.9% 60.4% 61.6% 85%
Trying cocaine once or twice............. 1989 88.9% 90.5% 91.8% 92.2% 91.1% 91.4% 91.1% 89.2% 87.3% 88.8% 88.7% 95%
Smoking one or more packs of cigarettes
perday............................ 1987 74.2% 75.3% 74.0% 76.2% 71.8% 72.4% 69.2% 69.3% 68.5% 69.0% 71.2% 95%
3.22 Perception of harm by high school
seniors
Heavy use of alcohol .................. 1989 44.0% 47.1% 48.6% 49.0% 48.3% 46.5% 45.2% 49.5% 43.0% 43.8% 43.1% 70%
Regular use of marijuana ............... 1989 77.5% 77.8% 78.6% 76.5% 72.5% 65.0% 60.8% 59.9% 58.1% 58.5% 57.4% 90%
Trying cocaine once or twice............. 1989 54.9% 59.4% 59.4% 56.8% 57.6% 57.2% 53.7% 54.2% 53.6% 54.6% 52.1% 80%
Smoking one or more packs of cigarettes
perday............................ 1987 68.6% 68.2% 69.4% 69.2% 69.5% 67.6% 65.6% 68.2% 68.7% 70.8% 70.8% 95%
Using smokeless tobacco regularly ........ 1987
§
30.0% 34.2% 37.4% 35.5% 38.9% 36.6% 33.2% 37.4% 38.6% 40.9% 41.1% 95%
3.23 Tobacco excise tax (percent of retail price)
Cigarettes ........................... 1993 31.4% . . . . . . . . . . . . 31.0% 31.6% 30.5% 31.5% 28.2% 22.1% 50%
Smokeless tobacco.................... 1993 11.8% . . . . . . . . . . . . - - - 13.8% - - -
15
13.0% - - - - - - 50%
3.24 Treatment for nicotine addiction
Health plans offering treatment ........... 1985 11% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 100%
3.25 Preemptive clean indoor air laws
States with laws .................... 1995 17 . . . . . . . . . . . . . . . . . . 17 17 17 17 0
3.26 Number of States with laws banning
cigarette vending machines in areas
accessible to minors
9
................ 1995
§
12 ... ... ... ... ... ... 14 19 20 21 51
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
‡
The NHIS was redesigned in 1997. Data may not be comparable with previous years; see Technical Notes.
1
In 1992, the definition of ‘‘current’’ changed to include ‘‘some days’’ (intermittent smoking).
2
Estimates for different tribes.
3
Vietnamese males only.
4
1988 data.
5
Among people 20-24 years with a high school education or less.
6
Baseline for white females 20-24 years.
7
Data are unreliable. Relative standard error is greater than 30 percent.
8
Middle/junior high and senior high schools only.
9
1998-99 data.
10
Includes the District of Columbia.
11
1996-97 data.
Met
Toward
Mixed/ no change
Away
Cannot assess
Healthy People 2000 Final Review 103

12
1997-98 data.
13
Response rate for this group was too low to produce reliable estimates.
14
In 1994, estimates for drug use were restricted to a core set of questions in contrast to the coding procedure in prior years. The trend data have been recalculated to adjust for these
differences and to produce comparable estimates from 1988 to 1998.
15
Method of calculation modified in 1997.
NOTE: Data include revisions and, therefore, may differ from data previously published.
Objective number Data source
3.1*, 3.1a National Vital Statistics System, CDC, NCHS.
3.2*, 3.2a, b National Vital Statistics System, CDC, NCHS.
3.3 National Vital Statistics System, CDC, NCHS.
3.4*, 3.4a, b, d, e, h National Health Interview Survey, CDC, NCHS.
3.4c Baseline, 1992, and 1995 updates: Worldwide Survey of Substance Abuse and Health Behaviors Among Military Personnel, DoD, OASD.
1998 update: DoD Survey of Health Related Behaviors, Research Triangle Institute.
3.4f Baseline: CDC.
Updates: National Health Interview Survey, CDC, NCHS.
3.4g Baseline: Local surveys.
1990 update: Jenkins CH. Cancer risks and prevention practices among Vietnamese refugees. Western J Med 153:34–9. 1990.
1991 update: Jenkins CNH, et al. Tobacco use in Vietnam: Prevalence, predictors, and the role of the transnational tobacco corporations. JAMA
227(21):1726–31. 1997; Jenkins CNH, et al. The effectiveness of a media-led intervention to reduce smoking among Vietnamese-American men. AJPH
87(6):1031–4. 1997.
3.4i Baseline and 1991 update: National Health Interview Survey, CDC, NCHS.
1993 update: National Health and Pregnancy Survey, NIH, NIDA.
1995 update: National Survey of Family Growth, CDC, NCHS.
1996–98 updates: National Vital Statistics System, CDC, NCHS.
3.4j Baseline and 1988 update: Behavioral Risk Factor Surveillance System, CDC, NCCDPHP.
1995 update: National Survey of Family Growth, CDC, NCHS.
3.5, 3.5a National Health Interview Survey, CDC, NCHS.
3.6 Baseline: Adult Use of Tobacco Survey, CDC, NCCDPHP.
Updates: National Health Interview Survey, CDC, NCHS.
3.7, 3.7a National Health Interview Survey, CDC, NCHS.
3.8* Baseline: Adult Use of Tobacco Survey, CDC, NCCDPHP.
Updates: National Health Interview Survey, CDC, NCHS.
3.9* For males 18–24 years, National Health Interview Survey, CDC, NCHS.
For males 12–17 years, National Household Survey on Drug Abuse, SAMHSA, OAS.
3.9a Baseline: National Medical Expenditure Survey of American Indians/Alaska Natives, PHS, NCHSR.
Updates: National Health Interview Survey, CDC, NCHS.
3.10 Baseline: National Survey of School Districts’ Nonsmoking Policies, NSBA, ACS, ALA, and AHA.
Updates: School Health Policies and Programs Study, CDC, NCCDPHP.
3.11* Baseline, 1991, and 1992 updates for worksites with 50 or more employees: National Survey of Worksite Health Promotion Activities, OPHS, ODPHP.
1995 data: Business Responds to AIDS Benchmark Survey, CDC, NCHSTP.
1998–99 data: National Worksite Health Promotion Survey, OPHS, ODPHP.
For medium and large companies: Nationwide Survey on Smoking in the Workplace, CDC, OSH; Bureau of National Affairs; American Society for
Personnel Administration.
3.12* Office on Smoking and Health Legislative Tracking System, CDC, NCCDPHP.
3.13 Baseline: Association of State and Territorial Health Officials Reporting System: Cancer and Cardiovascular Diseases Survey, PHF.
104 Healthy People 2000 Final Review

Objective number Data source
Updates: Office on Smoking and Health Legislative Tracking System, CDC, NCCDPHP.
1997 data for States enforcing laws: Synar Regulation Implementation: ‘‘Report to Congress on FY 1997 State Compliance,’’ SAMHSA, February 1998.
3.14 Baseline: Association of State and Territorial Health Officials Reporting System: Cancer and Cardiovascular Diseases Survey, PHF.
1992 and 1994 updates: Association of State and Territorial Health Officials Survey of State Activities on Tobacco Prevention and
1996 update: Office on Smoking and Health, CDC, NCCDPHP; Public Health Applications Branch, NIH, NCI; California Department of Health Services.
3.15 Federal Trade Commission data reported by Office on Smoking and Health, CDC, NCCDPHP.
3.16 Baseline for internists: Wells, et al. Physicians Practice Study, AJPH 76:1009–13. 1986.
Baseline for dentists: Secker-Walker, et al. Statewide Survey of Dentists’ Smoking Cessation Advice. JADA 118:37–40. 1989.
Updates for dentists: National Survey of Dentists’ and Hygienists’ Tobacco Control Activities, University of Florida.
1992 data for primary care providers: Primary Care Provider Surveys, OPHS, ODPHP.
1997–98 data for primary care providers: Prevention in Primary Care Study, American College of Preventive Medicine.
3.17*, 3.17a-b National Vital Statistics System, CDC, NCHS.
3.18*, 3.18a National Vital Statistics System, CDC, NCHS.
3.19* National Household Survey on Drug Abuse, SAMHSA, OAS.
3.20* National Household Survey on Drug Abuse, SAMHSA, OAS.
3.21* Monitoring the Future, NIH, NIDA.
3.22* Monitoring the Future, NIH, NIDA.
3.23 ‘‘The Tax Burden on Tobacco,’’ The Tobacco Institute, 1995, and the Office on Smoking and Health, CDC, NCCDPHP.
3.24 Gelb BD. Preventive Medicine and Employee Productivity. Harvard Business Review 64(2): 12. 1985.
3.25* Office on Smoking and Health Legislative Tracking System, CDC, NCCDPHP.
3.26 Office on Smoking and Health Legislative Tracking System, CDC, NCCDPHP.
* Duplicate objective.
Healthy People 2000 Final Review 105
Tobacco Objectives
3.1*: Reduce coronary heart disease
deaths to no more than 100 per 100,000
people.
Duplicate objectives: 1.1, 2.1, and 15.1
3.1a*: Reduce coronary heart
disease deaths among blacks to no
more than 115 per 100,000 people.
Duplicate objectives: 1.1a, 2.1a, and
15.1a
3.2*: Slow the rise in lung cancer deaths
to achieve a rate of no more than 42 per
100,000 people.
Duplicate objective: 16.2
3.2a*: Slow the rise in lung cancer
deaths among females to no more
than 27 per 100,000.
Duplicate objective: 16.2a
3.2b*: Slow the rise in lung cancer
deaths among black males to no
more than 91 per 100,000.
Duplicate objective: 16.2b
3.3: Slow the rise in deaths from
chronic obstructive pulmonary disease to
achieve a rate of no more than 25 per
100,000 people.
3.4*: Reduce cigarette smoking to a
prevalence of no more than 15 percent
among people aged 18 and older.
Duplicate objectives: 15.12 and 16.6
3.4a*: Reduce cigarette smoking to
a prevalence of no more than
20 percent among people with a
high school education or less aged
20 and older.
Duplicate objectives: 15.12a and
16.6a
3.4b*: Reduce cigarette smoking to
a prevalence of no more than
20 percent among blue-collar
workers aged 18 and older.
Duplicate objectives: 15.12b and
16.6b
3.4c*: Reduce cigarette smoking to
a prevalence of no more than
20 percent among military
personnel.
Duplicate objectives: 15.12c and
16.6c
3.4d*: Reduce cigarette smoking to
a prevalence of no more than
18 percent among blacks aged 18
and older.
Duplicate objectives: 15.12d and
16.6d
3.4e*: Reduce cigarette smoking to
a prevalence of no more than
15 percent among Hispanics aged
18 and older.
Duplicate objectives: 15.12e and
16.6e
3.4f*: Reduce cigarette smoking to
a prevalence of no more than
20 percent among American Indians
and Alaska Natives.
Duplicate objectives: 15.12f and
16.6f
3.4g*: Reduce cigarette smoking to
a prevalence of no more than
20 percent among Southeast Asian
men.
Duplicate objectives: 15.12g and
16.6g
3.4h*: Reduce cigarette smoking to
a prevalence of no more than
12 percent among women of
reproductive age.
Duplicate objectives: 15.12h and
16.6h
3.4i*: Reduce cigarette smoking to
a prevalence of no more than
10 percent among pregnant women.
Duplicate objectives: 15.12i and
16.6i
3.4j*: Reduce cigarette smoking to
a prevalence of no more than
10 percent among women who use
oral contraceptives.
Duplicate objectives: 15.12j and
16.6j
3.5: Reduce the initiation of cigarette
smoking by children and youth so that
no more than 15 percent have become
regular cigarette smokers by age 20.
3.5a: Reduce the initiation of
cigarette smoking by lower
socioeconomic status youth so that
no more than 18 percent have
become regular cigarette smokers
by age 20.
3.6: Increase to at least 50 percent the
proportion of cigarette smokers aged 18
and older who stopped smoking
cigarettes for at least one day during the
preceding year.
3.7: Increase smoking cessation during
pregnancy so that at least 60 percent of
women who are cigarette smokers at the
time they become pregnant quit smoking
early in pregnancy and maintain
abstinence for the remainder of their
pregnancy.
3.7a: Increase smoking cessation
during pregnancy so that at least
45 percent of women with less than
a high school education who are
cigarette smokers at the time they
become pregnant quit smoking early
in pregnancy and maintain
abstinence for the remainder of their
pregnancy.
3.8*: Reduce to no more than 20 percent
the proportion of children aged 6 and
younger who are regularly exposed to
tobacco smoke at home.
Duplicate objective: 11.17
3.9*: Reduce smokeless tobacco use by
males aged 12–24 to a prevalence of no
more than 4 percent.
Duplicate objective: 13.17
3.9a*: Reduce smokeless tobacco
use by American Indian and Alaska
Native youth to a prevalence of no
more than 10 percent.
Duplicate objective: 13.17a
3.10: Establish tobacco-free
environments and include tobacco-use
prevention in the curricula of all
elementary, middle, and secondary
schools, preferably as part of
comprehensive school health education.
3.11*: Increase to 100 percent the
proportion of worksites with a formal
smoking policy that prohibits or
severely restricts smoking at the
workplace.
Duplicate objective: 10.18
3.12*: Enact in 50 States and the
District of Columbia comprehensive
laws on clean indoor air that prohibit
smoking or limit it to separately
ventilated areas in the workplace and
enclosed public places.
Duplicate objective: 10.19
3.13: Enact in 50 States and the District
of Columbia laws prohibiting the sale
and distribution of tobacco products to
youth younger than age 18. Enforce
106 Healthy People 2000 Final Review
these laws so that the buy rate in
compliance checks conducted in all 50
States and the District Columbia is no
higher than 20 percent.
3.14: Establish in 50 States and the
District of Columbia plans to reduce
tobacco use, especially among youth.
3.15: Eliminate or severely restrict all
forms of tobacco product advertising
and promotion to which youth younger
than age 18 are likely to be exposed.
3.16: Increase to at least 75 percent the
proportion of primary care and oral
health care providers who routinely
advise cessation and provide assistance
and followup for all of their
tobacco-using patients.
3.17*: Reduce deaths due to cancer of
the oral cavity and pharynx to no more
than 10.5 per 100,000 men aged 45–74
and 4.1 per 100,000 women aged
45–74.
Duplicate objectives: 13.7 and 16.17
3.17a*: Reduce deaths due to
cancer of the oral cavity and
pharynx to no more than 26.0 per
100,000 among black males aged
45–74.
Duplicate objectives: 13.7a and
16.17a
3.17b*: Reduce deaths due to
cancer of the oral cavity and
pharynx to no more than 26.0 per
100,000 among black females aged
45–74.
Duplicate objectives: 13.7b and
16.17b
3.18*: Reduce stroke deaths to no more
than 20 per 100,000 people.
Duplicate objectives: 2.22 and 15.2
3.18a*: Reduce stroke deaths
among blacks to no more than 27
per 100,000.
Duplicate objectives: 2.22a and
15.2a
3.19*: Increase by at least 1 year the
average age of first use of cigarettes,
alcohol, and marijuana by adolescents
aged 12–17.
Duplicate objective: 4.5
3.20*: Reduce the proportion of young
people who have used alcohol,
marijuana, and cocaine, or cigarettes in
the past month as follows:
2000 target
Substance and age (percent)
Alcohol:
12–17 years 12.6
18–20 years 29.0
Marijuana:
12–17 years 3.2
18–25 years 7.8
Cocaine:
12–17 years 0.6
18–25 years 2.3
Use in past month 2000 target
(percent)
Alcohol:
Hispanic 12–17 years 12.0
Cocaine:
Hispanic 12–17 years 0.6
Hispanic 18–25 years 1.0
Cigarettes:
12–17 years 6.0
Duplicate objective: 4.6
3.21*: Increase the proportion of high
school seniors who perceive social
disapproval of heavy use of alcohol,
occasional use of marijuana, and
experimentation with cocaine, or regular
use of tobacco, as follows:
2000 target
(percent)
Heavy use of alcohol 70
Occasional use of marijuana 85
Trying cocaine once or twice 95
Smoking one or more packs of
cigarettes per day 95
Duplicate objective: 4.9
3.22*: Increase the proportion of high
school seniors who associate physical or
psychological harm with the heavy use
of alcohol, occasional use of marijuana,
experimentation with cocaine, or regular
use of tobacco, as follows:
2000 target
(percent)
Heavy use of alcohol 70
Regular use of marijuana 90
Trying cocaine once or twice 80
Smoking one or more packs of
cigarettes per day 95
Using smokeless tobacco
regularly 95
Duplicate objective: 4.10
3.23: Increase the average (State and
Federal combined) tobacco excise tax to
at least 50 percent of the average retail
price of all cigarettes and smokeless
tobacco.
3.24: Increase to 100 percent the
proportion of health plans that offer
treatment of nicotine addiction (e.g.,
tobacco use cessation counseling by
health care providers, tobacco use
cessation classes, prescriptions for
nicotine replacement therapies, and/or
other cessation services).
3.25*: Reduce to zero the number of
States that have clean indoor air laws
preempting stronger clean indoor air
laws on the local level.
Duplicate objective: 10.20
3.26: Enact in 50 States and the District
of Columbia laws banning cigarette
vending machines except in places
inaccessible to minors.
* Duplicate objective.
Healthy People 2000 Final Review 107

Priority Area 4
Substance Abuse:
Alcohol and Other
Drugs
Background
Substance abuse and its related
problems are among society’s most
pervasive health and social concerns.
Each year, about 100,000 deaths in the
United States are related to alcohol
consumption (1). Illicit drug abuse and
related acquired immunodeficiency
syndrome (AIDS) deaths account for at
least another 12,000 deaths. In 1995, the
economic cost of alcohol and drug
abuse was $276 billion (2). This
represents more than $1,000 for every
man, woman, and child in the United
States to cover the costs of health care,
motor vehicle crashes, crime, lost
productivity, and other adverse outcomes
of alcohol and drug abuse.
Data Summary
Highlights
Three of the 20 Healthy People
2000 objectives have been met or
surpassed. By 1996, all 50 States had
established and were monitoring
comprehensive plans to ensure that
underserved populations had access to
alcohol and drug treatment programs
(4.12). In 1995, more than 90 percent of
the worksites with 50 or more
employees had adopted policies on
alcohol and drugs (4.14), which exceeds
the target of 60 percent.
Alcohol-related motor vehicle crash
deaths (4.1) have declined dramatically
from 9.8 deaths per 100,000 persons in
1987, when the baseline was
established, to 5.8 per 100,000 in 1999.
However, the target, which was revised
downward from 8.5 to 5.5 during the
1995 Healthy People Midcourse Review
(3), was not met. This reduction has
been attributed in part to the passage of
State laws mandating administrative
license revocation (4.15) and setting
maximum blood alcohol concentration
(BAC) levels of 0.08 percent for drivers
21 years and older and establishing zero
tolerance for alcohol in the blood of
drivers under the age of 21 years (4.18).
The cirrhosis death rate (4.2) has
declined to an age-adjusted rate of 7.2
deaths per 100,000 persons, although the
age-adjusted rate for American
Indians/Alaska Natives remains
significantly higher than that of other
racial and ethnic groups. Progress has
also been made on increasing the age of
first use of cigarettes (4.5).
Past month use of illegal substances
showed mixed results. Alcohol use by
adolescents 12–17 years has declined
substantially from 33.4 percent in 1988,
when the baseline was established, to
19.1 percent in 1998 (4.6). Marijuana
use for youths 12–17 years is more
prevalent now than it was a decade ago,
although use by this age group has been
decreasing since 1997 (4.6). The
problem of heavy drinking (4.7) persists
among high school students in the
30–percent range and in the 40–percent
range for college students.
Trends in the perceived harmfulness
of different substances are also mixed
(4.10). In recent years, high school
seniors’ perception of harm from regular
marijuana use has declined substantially,
moving away from the target despite
initial improvements. While high school
seniors’ perception of harm from heavy
alcohol use (4.10) increased somewhat
in the early 1990s, it has subsequently
declined, ending the decade largely
unchanged from the baseline.
High school seniors’ perception of
social disapproval for heavy alcohol use
has increased, while perceptions of
social disapproval for regular use of
marijuana, trying cocaine once or twice,
smoking one or more packs of cigarettes
per day, and using smokeless tobacco
are generally moving away from their
respective targets (4.9), resulting in a
mixed assessment for this objective.
For the total population, rates of
drug-related deaths (4.3) and drug
abuse-related emergency department
visits have increased (4.4).
Summary of Progress
Data to assess trends toward the
year 2000 targets are available for 16 of
the 20 objectives in the Substance
Abuse priority area. The target for one
objective (4.12) was met and the targets
for two objectives (4.11 and 4.14) have
been surpassed. Progress toward targets
is shown for seven objectives (4.1, 4.2,
4.5, 4.7, 4.8, 4.15, and 4.18). Trends are
generally moving away from targets for
two objectives (4.3 and 4.4). Mixed
results are shown for four objectives
(4.6, 4.9, 4.10, and 4.19). No updates
are available for four objectives (4.13,
4.16, 4.17, and 4.20). See table 4 for the
tracking data for the objectives in this
priority area and figure 4 for a
quantitative assessment of progress.
Discussion
Twice in the 20th century, in the
1970s and again in the 1990s, drug use
rose. In the later part of the 1980s, drug
use fell, although illegal drug use never
disappeared entirely. Beginning around
1990, teens and preteens began to adopt
more permissive attitudes toward drugs.
Soon thereafter, actions followed
perceptions and the use of illegal drugs
increased among young people 12–17
years. Supplemental data from 1999
indicate that 6.7 percent or 14.8 million
Americans 12 years and over were
current users of illicit drugs. This is
down from 1979, when 14.1 percent of
the U.S. population 12 years and over
were current drug users (4) . By
historical standards, present drug rates
are relatively low. The fluctuation in
trends reinforces the need for education
as a continuous process for parents,
communities, and policymakers, as well
as for children.
The direct and indirect public health
impact of substance abuse is
widespread. Alcohol abuse and drug
abuse among youth are strongly
associated with risk-taking behavior,
including sexual risk-taking behavior
(5). Intravenous drug use is a major
vector for the transmission of infectious
diseases, including HIV/AIDS, hepatitis
B and hepatitis C (6).
Alcohol consumption has significant
adverse consequences for public health.
Alcohol use has been linked with a
substantial portion of injuries and deaths
from motor vehicle crashes, falls, fires,
and drownings (7). It is also a factor in
homicide, suicide, marital violence, and
child abuse (8). Excessive drinking has
consequences for virtually every part of
the body. Heavy alcohol use increases
risk for cirrhosis and other liver
disorders. Drinking also may increase
the risk for developing cancer of the
colon and rectum (9). Women’s risk of
developing breast cancer increases
slightly if they drink two or more drinks
per day (10).
The problem of heavy drinking
persists among high school students and
for college students. Recently enacted
Federal law requires the withholding of
108 Healthy People 2000 Final Review

4.1 Alcohol-related motor vehicle deaths
4.2 Cirrhosis deaths
4.3 Drug-related deaths
4.4 Drug abuse-related emergency room visits
4.5 Average age of first use, 12-17 years: Cigarettes
Alcohol
Marijuana
4.6 Use in past month: Alcohol, 12-17 years
Alcohol, 18-20 years
Marijuana, 12-17 years
Marijuana, 18-25 years
Cocaine, 12-17 years
Cocaine, 18-25 years
College students
Occasional marijuana use
4.9 High school seniors' perception of disapproval:
Heavy alcohol use
4.8 Alcohol consumption (gallons per capita)
Cocaine use once or twice
Cigarette, 12-17 years
4.7 Heavy drinking in past 2 weeks: High school seniors
Smoking one or more packs of cigarettes a day
4.10 High school seniors' perception of harm:
Heavy alcohol use
Cocaine use once or twice
Regular marijuana use
Smoking one or more packs of cigarettes a day
4.12 Number of States with access to treatment programs
4.11 Anabolic steroid use: Male high school seniors
4.14 Alcohol policies at worksites with 50 or more employees
Using smokeless tobacco regularly
4.15 States with administrative license suspension/revocation laws
Other drug policies
4.18 States with blood alcohol tolerance levels:
.02% or less, under 21 years
0.8%, 21 years and over
Moved away from target Moved toward target Met target
NOTE: Complete tracking data are shown in table 4. Progress quotients are not calculated for objectives 4.13, 4.16, 4.17, 4.19, and 4.20.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
22%
80%
No change
-68%
-14%
-3%
-4%
-11%
30%
69%
4%
20%
67%
44%
18%
67%
9%
8%
17%
55%
21%
Percent of target achieved
-100 -80 -60 -40 -20
0
20 40 60 80 100
93%
112%
27%
112%
100%
100%
100%
100%
-132%
-132%
-132%
-132%
Healthy People 2000 Final Review 109
Figure 4. Final status of Substance Abuse: Alcohol and Other Drug objectives
Federal highway funds from States that
do not adopt a BAC of 0.08 percent as
the standard for driving while
intoxicated. Beginning in 2004, a
gradually increasing percent of these
funds (up to a maximum of 8 percent)
will be withheld from States that fail to
comply (11). In 1998, 16 States and the
District of Columbia had 0.08 as their
threshold BAC tolerance level for
driving while alcohol-impaired.
Transition to Healthy People
2010
Healthy People 2010 increases the
number of objectives related to
substance abuse from 20 in Healthy
People 2000 to 25. Many of the
objectives in Healthy People 2000 are
retained in Healthy People 2010,
including objectives on alcohol-related
motor vehicle crashes (4.1), cirrhosis
deaths (4.2), drug-related (drug-induced)
deaths (4.3), drug-related hospital
emergency department visits (4.4),
average age of first use (4.5), past
month use of illicit substances by
adolescents (4.6), binge drinking (4.7),
alcohol consumption (4.8), peer
disapproval of substance abuse (4.9),
perception of risk (4.10), steroid use
(4.11), administrative license revocation
laws (4.15), and blood alcohol
concentration levels (4.18).
Objectives related to the prevention
of tobacco use included in the Healthy
People 2000 priority area have been
reorganized into the Tobacco Use focus
area in Healthy People 2010. Such a
move, however, is not intended to lessen
the understanding that the prevention
and treatment of substance abuse
requires that all abused substances be
addressed—from tobacco and alcohol to
marijuana and other illicit drugs.
In addition, several objectives have
been added to Healthy People 2010.
They include objectives dealing with
drug-related motor vehicle crash deaths,
injuries caused by alcohol-related motor
vehicle crashes, injuries caused by
drug-related motor vehicle crashes,
alcohol-related emergency department
visits, adolescents riding in a car with a
driver who has been drinking, alcohol-
and drug-related violence, lost
productivity, adults exceeding guidelines
for low-risk drinking, and inhalant use
by adolescents. Also added are
objectives related to the treatment gap
for illicit drugs, treatment in correctional
institutions, treatment for injection drug
use, treatment gap for problem alcohol
use, hospital emergency department
referrals, and communities using
partnerships or coalition models to
conduct substance abuse prevention
efforts.
Several objectives included in other
focus areas of Healthy People 2010 are
directly related to the objectives
included in the Substance Abuse focus
area. For example, school health
education and community health
promotion programs (Educational and
Community-Based Programs), AIDS
among persons who inject drugs (HIV),
deaths from unintentional injuries
(Injury and Violence Prevention), fetal
alcohol syndrome (Maternal, Infant, and
Child Health), and primary care
screening and assessment (Mental
Health and Mental Disorders). All 21
objectives in the focus area on Tobacco
Use are related to the Substance Abuse
chapter, especially the objectives on
adolescent tobacco use, initiation of
tobacco use, age at first tobacco use,
smoking cessation by adolescents, and
adolescent disapproval of smoking.
Substance Abuse is one of the 10
Leading Health Indicators (LHIs), which
Healthy People 2010 introduces to serve
as a barometer of the Nation’s health.
Three objectives from the Substance
Abuse focus area—adolescent use of
alcohol or any illicit drug, adult use of
any illicit drug, and binge drinking by
adults—are used to measure this LHI.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the differences between the
two decades of objectives, reflecting
new knowledge and directions in this
area.
Data Issues
Definitions
All deaths attributed to chronic liver
disease and cirrhosis (whether or not
they are specified as alcohol related) are
tracked in objective 4.2 as an indicator
of abusive alcohol consumption. The
entries on death certificates are often not
specific enough to identify all
alcohol-related liver disease deaths.
Estimates of the proportion of the total
chronic liver disease and cirrhosis
deaths that are alcohol-related range
from 41 to 95 percent (12).
Data from the National Vital
Statistics System are used to track
drug-related deaths (4.3). Although the
objective discusses drug-related deaths,
it is tracked by a category of deaths that
is more accurately termed
‘‘drug-induced deaths.’’ The category
includes deaths whose underlying cause
was drug dependence, nondependent use
of drugs, and poisoning from drugs, all
of which may include medically
prescribed drugs. It excludes
unintentional injuries, homicides, and
other causes indirectly related to drug
use. See Appendix table IV for a list of
specific ICD–9 codes. This objective
will continue to be tracked in Healthy
People 2010 with the more accurate title
‘‘drug-induced deaths.’’
Objectives 4.7, 4.9, and 4.10 refer
to recent heavy drinking and heavy
alcohol use. Heavy alcohol use is
defined as having five or more drinks
on one occasion. Recent heavy drinking
is having five or more drinks on one
occasion in the past 2 weeks.
Data Sources
Alcohol-related motor vehicle
crashes (4.1) are tracked using data from
the Department of Transportation’s
Fatality Analysis Reporting System
(formerly the Fatal Accident Reporting
System) (FARS). The FARS
supplements death certificate data with
information on the circumstances of the
death to determine whether the death
was alcohol related. The National Vital
Statistics System does not specify
alcohol-related motor vehicle crashes.
The 1992 baselines for objective
4.14 are from the National Survey of
Worksite Health Promotion Activities,
which was a telephone survey of
nongovernment worksites. Worksites
were sampled, because different
worksites within the same company
could have different sets of health
promotion activities. Both active (for
example, classes) and passive (for
example, brochures) methods were
counted as worksite health promotion
activities. The 1995 update is from the
Centers for Disease Control and
Prevention (CDC) sponsored Worksite
Benchmark Survey, which used a
methodology very similar to the 1992
survey, but did not include passive
methods of health promotion (13,14).
The 1992 data on inquiry about
alcohol consumption and other drug
abuse for objective 4.19 are from
Primary Care Provider Surveys (PCPS).
110 Healthy People 2000 Final Review
The sample was drawn from the
membership rolls of professional
organizations for pediatricians, nurse
practitioners, family physicians,
obstetricians/gynecologists, and
internists. Response rates varied from 50
to 80 percent across these groups. The
data on inquiry (from PCPS) about
work-related risks represent the
proportion of providers who routinely
queried 81–100 percent of their patients
about these risks. The data on
counseling refer to the proportion of
providers who routinely provided these
services to patients who needed the
services. The Prevention in Primary
Care Study (PPCS) was conducted in
1997–98. The design and items included
in the 1997–98 study were similar to the
PCPS, but the sampling frame was
slightly different and some items
included in the 1992 surveys were not
included in the PPCS. The professionals
were sampled from listings of all
licensed, active practitioners in the
United States whose practices were at
least 50 percent primary care. Because
of low response rates from the other
provider groups, updates are available
only for nurse practitioners.
Data Comparability
The National Household Survey on
Drug Abuse (NHSDA) is used to
measure objectives 4.5 and 4.6
regarding substance use among
adolescents and young people. The
targets for objective 4.6 are consistent
with the goals established by the Office
of the National Drug Control Policy,
Executive Office of the President.
Beginning in 1991, the NHSDA was
expanded to include college students
living in residence halls. Thus, results
for people 18–25 years for marijuana
and cocaine use and people 18–20 years
for alcohol use are not directly
comparable with measures from
previous years. Additionally, improved
questionnaire and editing procedures
were introduced with the 1994 survey,
which restricted determination of drug
use to a core set of questions in contrast
to a broader coding procedure in prior
years. The trend data for all substances
in objective 4.6 have been recalculated
to adjust for these differences and to
produce comparable estimates from
1988 to 1998.
References
1. McGinnis JM, Foege WH. Actual causes of
death in the United States. JAMA 270:2207–12.
1993.
2. Harwood H, Fountain D, Livermore G. The
economic costs of alcohol and drug abuse in the
United States, 1992. Rockville, Maryland:
National Institutes of Health. 1998.
3. Department of Health and Human Services.
Healthy people 2000 midcourse review and 1995
revisions. Washington, DC: Public Health Service.
1996.
4. Office of National Drug Control Policy.
National drug control strategy 2001 annual report.
Washington, DC: Government Printing Office.
2001.
5. Rahdert E, Czechowicz D. (eds). Adolescent
drug abuse: Clinical assessment and therapeutic
interventions. NIDA Research Monograph No.
156. NIH Publication No. 95–3908. Rockville,
Maryland: National Institutes of Health. 1995.
6. Garfein R.S, Vlahov D, Galai N, et al. Viral
infections in short-term injection drug users; the
prevalence of the hepatitis B, hepatitis C, human
immunodeficiency, human T-lymphotropic virus.
Am J Public Health 88:655–61. 1996.
7. National Institute on Alcohol Abuse and
Alcoholism. Ninth special report to the U.S.
Congress on alcohol and health from the Secretary
of Health and Human Services. Rockville,
Maryland: National Institutes of Health. 1997.
8. Roizen J. Issues in the epidemiology of alcohol
and violence. In: Martin S, ed. Alcohol and
interpersonal violence: Fostering multidisciplinary
perspectives. Rockville, Maryland: National
Institutes of Health. 1993.
9. National Institute on Alcohol Abuse and
Alcoholism. Alcohol and cancer. Alcohol Alert. No
21. Rockville, Maryland: National Institutes of
Health. 1993.
10. Reichmann ME. Alcohol and breast cancer.
Alcohol Health and Research World. 18(3):182–4.
1994.
11. Center for Science in the Public Interest.
Action Alerts, Oct. 4, 2000.
http://www.cspinet.org/booze/08_update2.htm.
12. National Institute on Alcohol Abuse and
Alcoholism. County alcohol problem indicators
1979–85. U.S. Alcohol epidemiologic data
reference manual; vol 3, 3d ed. Washington:
Department of Health and Human Services. 1991.
13. Centers for Disease Control and Prevention
Cancer screening offered by worksites—United
States, 1992 and 1995. MMWR 46(19):421–4.
1997.
14. Survey: Technical report. Atlanta: Public
Health Service, Centers for Disease Control and
Prevention. 1996.
Healthy People 2000 Final Review 111

Table 4. Substance Abuse: Alcohol and Other Drugs objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
4.1* Alcohol-related motor vehicle deaths (per 100,000). . 1987 9.8 8.9 7.9 7.0 6.8 6.4 6.6 6.5 6.1 5.9 5.8 5.5
a. American Indian/Alaska Native male ............ 1987 40.4 34.3 32.2 31.4 26.8 28.0 - - - - - - - - - - - - - - - 35.0
b. People 15-24 years ........................ 1987
§
20.9 18.6 17.2 14.2 13.2 13.0 12.8 12.9 11.7 11.7 11.5 12.5
4.2 Cirrhosis deaths (age adjusted per 100,000) ........ 1987 9.2 8.6 8.3 8.0 7.9 7.9 7.6 7.5 7.4 7.2 - - - 6
a. Black male ............................... 1987 22.6 20.0 17.4 17.2 16.1 15.9 14.7 13.8 12.9 12.3 - - - 12
b. American Indian/Alaska Native ................ 1987 20.5 19.8 20.4 21.6 21.0 21.4 24.3 20.7 20.6 22.0 - - - 10
c. Hispanic
1
................................ 1990
§
14.2 . . . 13.8 13.5 13.4 13.7 12.9 12.6 12.0 11.7 - - - 10
4.3 Drug-related deaths (age adjusted per 100,000) ..... 1987 3.8 3.6 3.8 4.3 4.8 5.0 5.1 5.2 5.6 5.9 - - - 3
a. Black ................................... 1990 5.7 . . . 6.6 6.8 8.3 8.6 8.5 8.0 8.2 8.1 - - - 3
b. Hispanic
1
................................ 1990 4.3 . . . 3.9 5.6 6.4 6.0 6.0 5.9 5.7 5.9 - - - 3
4.4 Drug abuse-related emergency room visits
(per 100,000) ............................... 1991 175.8 . . . . . . 191.4 203.9 225.2 222.5 207.2 221.5 225.4 228.2 140.6
4.5* Average age of first use (adolescents 12-17 years) . .
Cigarettes ................................... 1988 11.6 11.5 11.5 11.7 11.7 12.2 12.3 12.4 12.4 12.4 - - - 12.6
Alcohol ..................................... 1988 13.1 12.8 12.6 13.0 12.9 12.8 12.6 13.1 13.1 13.1 - - - 14.1
Marijuana ................................... 1988 13.4 13.4 13.5 13.8 13.9 14.1 13.8 14.4 13.7 13.7 - - - 14.4
4.6* Use in past month by adolescents and young
adults
2
Alcohol
12-17 years ............................... 1988 33.4% 32.5% 27.0% 20.9% 23.9% 21.6% 21.1% 18.8% 20.5% 19.1% - - - 12.6%
18-20 years ............................... 1994 54.6% . . . . . . . . . . . . . . . 54.1% 50.1% 53.4% 53.5% - - - 29.0%
Hispanic 12-17 years ........................ 1988 31.9% 24.2% 28.3% 20.3% 22.0% 18.3% 18.7% 19.9% 18.8% 18.9% - - - 12.0%
Marijuana
12-17 years ............................... 1988 5.4% 4.4% 3.6% 3.4% 4.0% 6.0% 8.2% 7.1% 9.4% 8.3% - - - 3.2%
18-25 years ............................... 1988 15.3% 12.7% 12.9% 10.9% 11.1% 12.1% 12.0% 13.2% 12.8% 13.8% - - - 7.8%
Cocaine
12-17 years ............................... 1988 1.2% 0.6% 0.4% 0.3% 0.4% 0.3% 0.8% 0.6% 1.0% 0.8% - - - 0.6%
18-25 years ............................... 1988 4.8% 2.3% 2.2% 2.0% 1.6% 1.2% 1.3% 2.0% 1.2% 2.0% - - - 2.3%
Hispanic 12-17 years ........................ 1988 1.4% 2.0% 1.4% 1.3% 1.1% 0.7% 0.8% 1.1% 1.0% 1.4% - - - 0.6%
Hispanic 18-25 years ........................ 1994 2.2% . . . . . . . . . . . . . . . 1.1% 2.1% 1.5% 2.7% - - - 1.0%
Cigarettes
12-17 years ............................... 1988 22.7% 22.4% 20.9% 18.4% 18.5% 18.9% 20.2% 18.3% 19.9% 18.2% - - - 6.0%
4.7 Heavy drinking in past 2 weeks
3
High school seniors ........................... 1989 33.0% 32.2% 29.8% 27.9% 27.5% 28.2% 29.8% 30.2% 31.3% 31.5% 30.8% 28.0%
College students .............................. 1989 41.7% 41.0% 42.8% 41.4% 40.2% 40.0% 40.0% 38.3% 40.7% 38.9% 40.0% 32.0%
4.8 Alcohol consumption (gallons per capita, persons
14 years and over) ........................... 1987 2.54 2.45 2.31 2.31 2.25 2.21 2.17 2.19 2.18 - - - - - - 2.0
4.9* Perception of social disapproval by high school
seniors
Heavy use of alcohol .......................... 1989 56.4% 59.0% 58.1% 60.8% 58.5% 59.1% 58.0% 57.8% 56.4% 55.5% 57.6% 70%
Occasional use of marijuana ..................... 1989 71.1% 76.4% 75.8% 79.2% 73.8% 69.1% 65.4% 63.1% 59.9% 60.4% 61.6% 85%
Trying cocaine once or twice..................... 1989 88.9% 90.5% 91.8% 92.2% 91.1% 91.4% 91.1% 89.2% 87.3% 88.8% 88.7% 95%
Smoking one or more packs of cigarettes per day..... 1987 74.2% 75.3% 74.0% 76.2% 71.8% 72.4% 69.2% 69.3% 68.5% 69.0% 71.2% 95%
See footnotes and key at end of table.
112 Healthy People 2000 Final Review

Table 4. Substance Abuse: Alcohol and Other Drugs objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
4.10* Perception of harm by high school seniors
Heavy use of alcohol .......................... 1989 44.0% 47.1% 48.6% 49.0% 48.3% 46.5% 45.2% 49.5% 43.0% 43.8% 43.1% 70%
Regular use of marijuana ....................... 1989 77.5% 77.8% 78.6% 76.5% 72.5% 65.0% 60.8% 59.9% 58.1% 58.5% 57.4% 90%
Trying cocaine once or twice..................... 1989 54.9% 59.4% 59.4% 56.8% 57.6% 57.2% 53.7% 54.2% 53.6% 54.6% 52.1% 80%
Smoking one or more packs of cigarettes per day..... 1987 68.6% 68.2% 69.4% 69.2% 69.5% 67.6% 65.6% 68.2% 68.7% 70.8% 70.8% 95%
Using smokeless tobacco regularly ................ 1987
§
30.0% 34.2% 37.4% 35.5% 38.9% 36.6% 33.2% 37.4% 38.6% 40.9% 41.1% 95%
4.11 Anabolic steroid use
Male high school seniors........................ 1989 4.7% 5.0% 3.6% 3.5% 3.5% 3.8% 3.8% 3.2% 4.1% 2.8% - - - 3.0%
4.12 Number of States with access to treatment
programs.................................. 1996 50 . . . . . . . . . . . . . . . . . . . . . 50 50 50 50
4.13 Alcohol and drug education in schools........... 1996 86% . . . . . . . . . . . . . . . . . . . . . - - - - - - - - - 100%
Provided students with some instruction ............ 1987 63% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 100%
Provided students with counseling................. 1987 39% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 100%
Referred students for clinical assessments .......... 1987 23% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 100%
Provided students with instruction in at least one
course
Middle/junior and senior high schools ............ ... --- --- --- --- --- 90.4% - - - - - - - - - - - - - - - 100%
4.14 Worksite alcohol and drug policies 50 or more
employees ................................. ... --- --- --- --- --- --- --- --- --- --- --- 60%
Alcohol ..................................... 1992 88% . . . . . . . . . - - - - - - 92% - - - - - - - - - - - - 60%
Other drugs ................................. 1992 89% . . . . . . . . . - - - - - - 96% - - - - - - - - - - - - 60%
4.15 Number of States with administrative license
suspension/revocation laws
4
.................. 1990 29 . . . 30 - - - 35 38 - - - - - - 40 41 - - - 51
4.16 Number of States with policies to reduce minors’
access to alcohol ........................... 1996 46 . . . . . . . . . . . . . . . . . . . . . - - - - - - - - - 50
4.17 Number of States with restrictions on promotion
of alcohol to children and adolescents .......... 1996 13 . . . . . . . . . . . . . . . . . . . . . - - - - - - - - - 20
4.18 Number of States with blood alcohol concentration
tolerance levels
Zero tolerance (0.02% or less) for people under
21 years ................................... 1993 9 . . . . . . . . . . . .
4
21 ---
4
38
4
46
4
50 - - - 50
0.08% for people 21 years and over ............... 1993 7 . . . . . . . . . . . . 11 - - - 14 15 16 - - - 50
4.19 Screening, counseling, and referral by clinicians ... ... --- --- --- --- --- --- --- --- --- --- --- 75%
Percent of clinicians routinely providing service
to 81–100% of patients
Inquiry about alcohol consumption (12 years and over)
Pediatricians ............................... 1992 29% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
Nurse practitioners .......................... 1992 45% . . . . . . . . . - - - - - - - - - - - - - - -
5
52% - - - 75%
Obstetricians/gynecologists .................... 1992 34% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
Internists .................................. 1992 63% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
Family physicians ........................... 1992 39% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
See footnotes and key at end of table.
Healthy People 2000 Final Review 113

Table 4. Substance Abuse: Alcohol and Other Drugs objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Inquiry about other drug use (12 years and over)
Pediatricians ............................... 1992 28% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
Nurse practitioners .......................... 1992 43% . . . . . . . . . - - - - - - - - - - - - - - -
5
36% - - - 75%
Obstetricians/gynecologists .................... 1992 32% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
Internists .................................. 1992 34% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
Family physicians ........................... 1992 23% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
Referral to alcohol treatment
Pediatricians ............................... 1992 26% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
Nurse practitioners .......................... 1992 19% . . . . . . . . . - - - - - - - - - - - - - - -
5
22% - - - 75%
Obstetricians/gynecologists .................... 1992 24% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
Internists .................................. 1992 33% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
Family physicians ........................... 1992 28% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
Referral to drug abuse treatment
Pediatricians ............................... 1992 32% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
Nurse practitioners .......................... 1992 19% . . . . . . . . . - - - - - - - - - - - - - - -
5
25% - - - 75%
Obstetricians/gynecologists .................... 1992 28% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
Internists .................................. 1992 35% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
Family physicians ........................... 1992 28% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 75%
4.20 Number of States with Hospitality Resource
Panels .................................... 1994 8 . . . . . . . . . . . . . . . - - - - - - - - - - - - - - - 30
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
1
Excludes data from States lacking an Hispanic-origin item on their death certificate or for which Hispanic-origin data were not of sufficient quality. See Appendix.
2
In 1994, estimates for drug use were restricted to a core set of questions in contrast to the coding procedure in prior years. The trend data have been recalculated to adjust for these
differences and to produce comparable estimates from 1988 to 1997.
3
Recent heavy drinking is defined as having 5 or more drinks on 1 occasion in the previous 2-week period as monitored by self-reports.
4
Includes the District of Columbia.
5
1997–98 data.
6
Response rate for this group was too low to produce reliable estimates.
NOTE: Data include revisions and, therefore, may differ from data previously published in these reports and other publications.
Met
Toward
Mixed/ no change
Away
Cannot assess
114 Healthy People 2000 Final Review

Objective number Data source
4.1*, 4.1b Fatality Analysis Reporting System, DOT, NHTSA.
4.1a Fatality Analysis Reporting System, DOT, NHTSA and National Vital Statistics System, CDC, NCHS.
4.2, 4.2a–c National Vital Statistics System, CDC, NCHS.
4.3, 4.3a–b National Vital Statistics System, CDC, NCHS.
4.4 Drug Abuse Warning Network, SAMHSA, OAS.
4.5* National Household Survey on Drug Abuse, SAMHSA, OAS.
4.6* National Household Survey on Drug Abuse, SAMHSA, OAS.
4.7 Monitoring the Future, NIH, NIDA.
4.8 Alcohol Epidemiology Data System, NIH, NIAAA.
4.9* Monitoring the Future, NIH, NIDA.
4.10* Monitoring the Future, NIH, NIDA.
4.11 Monitoring the Future, NIH, NIDA.
4.12 Substance Abuse Prevention and Treatment Block Grant Applications, SAMHSA, CSAT.
4.13 1987 baseline: Report to Congress and the White House on the Nature and Effectiveness of Federal, State, and Local Drug Prevention Education
Programs. DOE. 1987.
1996 baseline: Substance Abuse Prevention and Treatment Block Grant Applications, SAMHSA, CSAT.
1994 data: School Health Policies and Programs Study, CDC, NCCDPHP.
4.14 Baseline: National Survey of Worksite Health Promotion Activities, OPHS, ODPHP.
Update: Business Responds to AIDS Benchmark Survey, CDC, NCHSTP.
4.15 Baseline: Office of Alcohol and State Programs, DOT, NHTSA.
Updates: Office of Safety Recommendations, DOT, NHTSA.
4.16 Substance Abuse Prevention and Treatment Block Grant Applications, SAMHSA, CSAT.
4.17 Substance Abuse Prevention and Treatment Block Grant Applications, SAMHSA, CSAT.
4.18 Baseline: Office of Alcohol and State Programs, DOT, NHTSA.
Updates: Office of Safety Recommendations, DOT, NHTSA.
4.19 Baseline: Primary Care Provider Surveys, OPHS, ODPHP.
Update: Prevention in Primary Care Study, American College of Preventive Medicine.
4.20 California Coordinating Council on Responsible Beverage Service, National Survey Report.
* Duplicate objective.
Healthy People 2000 Final Review 115
Substance Abuse:
Alcohol and Other
Drugs Objectives
4.1*: Reduce deaths caused by
alcohol-related motor vehicle crashes to
no more than 5.5 per 100,000 people.
Duplicate objective: 9.23
4.1a*: Reduce deaths among
American Indian and Alaska Native
men caused by alcohol-related
motor vehicle crashes to no more
than 35.0 per 100,000.
Duplicate objective: 9.23a
4.1b*: Reduce deaths among people
aged 15–24 caused by
alcohol-related motor vehicle
crashes to no more than 12.5 per
100,000.
Duplicate objective: 9.23b
4.2: Reduce cirrhosis deaths to no more
than 6 per 100,000 people.
4.2a: Reduce cirrhosis deaths
among black men to no more than
12 per 100,000.
4.2b: Reduce cirrhosis deaths
among American Indians and
Alaska Natives to no more than 10
per 100,000.
4.2c: Reduce cirrhosis deaths
among Hispanics to no more than
10 per 100,000.
4.3: Reduce drug-related deaths to no
more than 3 per 100,000 people.
4.3a: Reduce drug-related deaths
among blacks to no more than 3 per
100,000.
4.3b: Reduce drug-related deaths
among Hispanics to no more than 3
per 100,000.
4.4: Reduce drug abuse-related hospital
emergency department visits by at least
20 percent.
4.5*: Increase by at least 1 year the
average age of first use of cigarettes,
alcohol, and marijuana by adolescents
aged 12–17.
Duplicate objective: 3.19
4.6*: Reduce the proportion of young
people who have used alcohol,
marijuana, and cocaine, or cigarettes in
the past month as follows:
2000 target
Substance and age (percent)
Alcohol:
12–17 years 12.6
18–20 years 29.0
Marijuana:
12–17 years 3.2
18–25 years 7.8
Cocaine:
12–17 years 0.6
18–25 years 2.3
Use in past month 2000 target
(percent)
Alcohol:
Hispanic 12–17 years 12.0
Cocaine:
Hispanic 12–17 years 0.6
Hispanic 18–25 years 1.0
Cigarettes:
12–17 years 6.0
Duplicate objective: 3.20
4.7: Reduce the proportion of high
school seniors and college students
engaging in recent occasions of heavy
drinking of alcoholic beverages to no
more than 28 percent of high school
seniors and 32 percent of college
students.
4.8: Reduce alcohol consumption by
people aged 14 and older to an annual
average of no more than 2 gallons of
ethanol per person.
4.9*: Increase the proportion of high
school seniors who perceive social
disapproval of heavy use of alcohol,
occasional use of marijuana, and
experimentation with cocaine, or regular
use of tobacco, as follows:
2000 target
(percent)
Heavy use of alcohol 70
Occasional use of marijuana 85
Trying cocaine once or twice 95
Smoking one or more packs of
cigarettes per day 95
Duplicate objective: 3.21
4.10*: Increase the proportion of high
school seniors who associate physical or
psychological harm with the heavy use
of alcohol, occasional use of marijuana,
experimentation with cocaine, or regular
use of tobacco, as follows:
Heavy use of alcohol 70
Regular use of marijuana 90
Trying cocaine once or twice 80
Smoking one or more packs of
cigarettes per day 95
Using smokeless tobacco
regularly 95
Duplicate objective: 3.22
4.11: Reduce to no more than 3 percent
the proportion of male high school
seniors who use anabolic steroids.
4.12: Establish and monitor in 50 States
comprehensive plans to ensure access to
alcohol and drug treatment programs for
traditionally underserved people.
4.13: Provide to children in all school
districts and private schools primary and
secondary school educational programs
on alcohol and other drugs, preferably
as part of comprehensive school health
education.
4.14: Extend adoption of alcohol and
drug policies for the work environment
to at least 60 percent of worksites with
50 or more employees.
4.15: Extend to 50 States administrative
driver’s license suspension/revocation
laws or programs of equal effectiveness
for people determined to have been
driving under the influence of
intoxicants.
4.16: Increase to 50 the number of
States that have enacted and enforce
policies, beyond those in existence in
1989, to reduce access to alcoholic
beverages by minors.
4.17: Increase to at least 20 the number
of States that have enacted statutes to
restrict promotion of alcoholic beverages
that are focused principally on young
audiences.
4.18: Extend to 50 States legal blood
alcohol concentration tolerance levels of
.08 percent for motor vehicle drivers
aged 21 and older and zero tolerance
(.02 percent and lower) for those
younger than age 21.
4.19: Increase to at least 75 percent the
proportion of primary care providers
who screen for alcohol and other drug
use problems and provide counseling
and referral as needed.
4.20: Increase to 30 the number of
States with Hospitality Resource Panels
(including representatives from State
regulatory, public health, and highway
safety agencies, law enforcement,
insurance associations, alcohol retail and
licensed beverage associations) to ensure
a process of management and server
training and define standards of
responsible hospitality.
*Duplicate objective.
116 Healthy People 2000 Final Review

Priority Area 5
Family Planning
Background
Family planning provides
individuals with the information and
means to exercise personal choice in
determining whether and when to
become parents (1). Family planning
entails both the prevention of
unintended pregnancies and the
achievement of planned, wanted
pregnancies—important for improving
birth outcomes, women’s health, and the
health of families. Family planning has
had a significant impact on the health
and well-being of persons in the United
States through the promotion of
reproductive health education and
services (2).
While adolescent females receive
considerable attention in family planning
initiatives, all women of childbearing
age require assistance with family
planning. The goal of reducing
unintended pregnancy is a significant
public health concern in the United
States. Each year, publicly subsidized
contraceptive services help women
avoid an estimated 1.3 million
unintended pregnancies (3). Access to
family planning services is an important
determinant of prenatal outcomes
generally, and unintended pregnancies
affect healthy starts for children. Women
who did not want to become pregnant
are less likely to have prenatal care in
the first trimester, compared with
women who wanted a child at the time
they became pregnant (4).
To the extent that effective family
planning widens the intervals between
births and ensures that pregnancies are
intended, babies will be born healthier.
Problems attendant to poor family
planning exact serious health and social
costs. Low birthweight (5), high rates of
infant mortality (6), and inadequate
monetary and family support (7) are
some of the consequences of poor
family planning. Research suggests that
educating young potential parents about
the financial, welfare, and social costs
of pregnancy may improve
decisionmaking, which, in turn, may
reduce the likelihood of an unintended
pregnancy (8).
Despite advances in contraceptive
technology and demonstrated
effectiveness of prevention initiatives,
nearly one-half of all pregnancies in the
United States are unintended (9). While
data show a decline in the rates of
unintended pregnancy (which may be
attributable to higher rates of
contraceptive use and improved quality
of contraceptives) (10), more needs to
be done to insure that all pregnancies
are intended (7). Promoting the use of
effective contraceptive methods and
improving the effectiveness with which
all methods are used could help
significantly in further reducing the
levels of unintended pregnancy (10, 11).
Data Summary
Highlights
Teenage pregnancy rates (5.1) have
declined considerably in the 1990s. The
teenage pregnancy rate was 98.7 per
1,000 women ages 15–19 years in 1996,
down 15 percent from its high point of
116.5 in 1991. The pregnancy rate was
67.8 for women ages 15–17 years in
1996, down from its high point of 80.3
in 1990. Data for objective 5.2 show
that in 1995, 49 percent of pregnancies
were unintended, a decrease of
13 percent since 1988, but still short of
the year 2000 target of 30 percent.
The proportions of both males and
females 15 years of age who engaged in
sexual intercourse during the past 3
months (5.5) have declined, although
both were short of the year 2000 targets.
The proportion of females 15–19 years
using contraception at first intercourse
(5.6) have increased between 1988 and
1995 from 65 to 76 percent. However,
use of contraception during most recent
intercourse decreased just as
dramatically from 78 to 71 percent over
the same time period for females of the
same age group.
The proportion of teens that have
discussed sexuality with their parents
(5.8) increased significantly, from
66 percent in 1986 to 80 percent in
1995, although it did not meet the year
2000 target of 85 percent. It should be
noted that more recent 1998 data from
the National Health Interview Survey
(NHIS) for adolescents 10–17 years
show a loss of some of the progress
made earlier, as reported from 1994
NHIS data.
Summary of Progress
Of the 12 family planning Healthy
People 2000 objectives, progress was
made in eight objectives (5.1–5.3, 5.5,
5.7, 5.8, 5.11, and 5.12), although none
reached their targets. Progress was
mixed for three objectives (5.4, 5.6, and
5.10). Progress for objective 5.10 is
based on limited updates showing only
nurse practitioner data. Data were not
available to update one objective (5.9).
See table 5 for the tracking data for the
objectives in this priority area and
figure 5 for a quantitative assessment of
progress.
Discussion
Although short of the year 2000
targets, decreases in adolescent
pregnancy (5.1) were promising. Among
the factors accounting for the overall
falling teenage pregnancy rates are
decreases in sexual activity, increases in
condom use, and the adoption of
injectable and implant contraceptives
(12). Objective 5.4 sought to reduce the
proportion of adolescents who have
engaged in sexual intercourse to no
more than 15 percent by age 15 and no
more than 40 percent by age 17. The
progress was mixed with declines in
proportions of all females, males, and
black males age 15. There was a slight
increase for all females age 17 and a
decrease for all black females age 17.
The pregnancy rates for non-Hispanic
black and Hispanic teenagers were about
twice as high as the rates for
non-Hispanic white teenagers. The lower
pregnancy rates for non-Hispanic white
teenagers is due to both the lower
proportion sexually active and the lower
pregnancy rate among those who are
sexually active. In 1995 about one out
of three sexually active black and
Hispanic teenagers became pregnant
compared with about one out of six
sexually active non-Hispanic white
teenagers (12).
Substantial decreases occurred in
unintended pregnancy rates (5.2) and,
correspondingly, the use of
contraceptives among females 15–44
years of age at risk for unintended
pregnancy increased (5.12). For black
females 15–44 years of age, the rate of
unintended pregnancies declined from
78 percent in 1988 to 72 percent in
1995, and the use of contraceptives for
this population increased from
78.9 percent in 1982 to 89.9 percent in
1995. Also, the pregnancy rate for
females using a contraceptive method
declined (5.7); this may have
contributed to the decline in unintended
Healthy People 2000 Final Review 117

5.1 Adolescent pregnancy
5.2 Unintended pregnancy
5.3 Infertility
5.4 Adolescents who ever had sexual intercourse:
Female 15 years
Male 15 years
Female 17 years
Male 17 years
5.5 Adolescent abstinence from sexual intercourse in
past 3 months: Female 15-17 years
Male 15-17 years
5.6 Contraception use: First intercourse female 15-19 years
First intercourse male 15-19 years
Recent intercourse female 15-19 years
Pill and condom use, recent intercourse female 15-19 years
5.7 Failure of contraceptive: Female 15-44 years
5.12 Contraception use: Female 15-44 years
5.8 Discussed sexuality with parents 13-18 years
Pill and condom use, recent intercourse in-school
male 15-17 years
Pill and condom use, last intercourse male 17-19 years
Moved away from target Moved toward target Met target
NOTE: Complete tracking data are shown in table 5. Progress quotients are not calculated for objectives 5.9, 5.10, and 5.11.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
41%
27%
57%
-58%
-10%
42%
33%
50%
57%
8%
44%
2%
7%
1%
29%
63%
Percent of target achieved
-100 -80 -60 -40 -20
0
20 40 60 80 100
21%
74%
118 Healthy People 2000 Final Review
Figure 5. Final status of Family Planning objectives
pregnancies. Overall, the rate of
unintended pregnancies in the United
States is still higher than other
developed countries. Canada’s rate is
39 percent, and the Netherlands’ rate is
only 6 percent (10).
Early sexual intercourse among
American adolescents represents a
significant public health problem.
Reduction of sexual activity at early
ages is an important public health
objective because sexual activity at early
ages is associated with more partners
and more frequent intercourse and the
concomitant risks of sexually
transmitted diseases, including HIV, and
unintended pregnancy (13). Although
other developed countries have similar
rates of early sexual intercourse, the
United States has one of the highest
teenage pregnancy rates in the world
(14). Abstinence from sexual intercourse
by adolescents increased (5.5), as did
their use of contraceptives (5.6).
Adolescents are now more likely to use
contraceptives, especially condoms, at
first intercourse than they were at the
beginning of the decade; black
adolescent females are particularly likely
to use injectable and implant
contraceptives (15).
Transition to Healthy People
2010
Many of the topics covered in the
Healthy People 2000 priority area on
Family Planning have been retained in
Healthy People 2010, with an emphasis
on increasing the proportion of
pregnancies that are intended, as well as
reducing adolescent pregnancy. In light
of the serious consequences of
unintended pregnancies, in 1995, the
Institute of Medicine issued a
comprehensive report supporting actions
to establish a new national norm in
which all pregnancies should be
intended, that is consciously and clearly
desired at the time of conception (5).
The importance of developing this norm
is supported in Healthy People 2010.In
a departure from Healthy People 2000,
which focused primarily on adolescent
pregnancy, Healthy People 2010 adopts
the broader perspective that every
pregnancy should be intended and
highlights improvement opportunities to
achieve this goal.
In addition, some new topics have
been included to address key issues such
as adequate spacing between
pregnancies, male involvement in
pregnancy prevention and reproductive
health, access to emergency
contraception and insurance coverage
for contraception. A new objective has
been added that addresses encouraging
females of all ages to space their
pregnancies adequately in order to lower
their risk of adverse perinatal outcomes.
A new developmental objective (an
objective without current baseline data)
has been included to increase the
proportion of health care providers who
provide emergency contraception. The
U.S. Guide to Clinical Preventive
Services (16) identifies postcoital
administration of emergency
contraceptive pills (ECP) after
unprotected intercourse as an effective
means of reducing subsequent
pregnancy. Increased public awareness,
including culturally and linguistically
competent education about ECP as well
as direct access to and insurance
reimbursement for ECP, would
contribute significantly toward
attainment of this objective and
reductions in the rates of unintended
pregnancies.
The Healthy People 2010 Family
Planning chapter also includes a new
developmental objective addressing male
involvement in pregnancy prevention
and family planning efforts. The next
cycle of the National Survey of Family
Growth (NSFG), which is expected to
be conducted in 2002, is being expanded
to include questions directed to males,
providing an avenue for
institutionalizing data collection about
male fertility that will be reflected in the
Healthy People 2010 objectives.
Another developmental objective
seeks to increase the proportion of
health insurance policies that cover
contraceptive supplies and services. In a
1995 report, the Institute of Medicine
concluded that among the reasons for
high rates of unintended pregnancy in
the United States was lack of
contraceptive coverage by private health
insurance (7). Both newer managed care
insurance plans and traditional
fee-for-service insurance plans are more
likely to pay for general gynecological
services than they are to cover
contraceptive services or supplies (17).
A 1993 survey conducted by the Alan
Guttmacher Institute found that half of
indemnity plans and 7 percent of health
maintenance organizations (HMOs) do
not cover nonpermanent contraception.
The survey also found that plans that do
cover contraceptive services and/or
supplies are often inconsistent in which
methods they cover and have a
pronounced bias toward covering
permanent surgical methods (18).
The Healthy People 2010 objective
to increase the proportion of adolescents
who abstain from sexual intercourse or
use condoms if currently sexually active
included in the Sexually Transmitted
Diseases focus area was adapted from
several family planning objectives in
Healthy People 2000. This objective has
been designated as a measure of one of
the Leading Health Indicators (LHI).
Abstinence is the only method of
complete protection from unintended
pregnancy and sexually transmitted
diseases. Condoms, if used correctly and
consistently, can help prevent both as
well. The LHIs are discussed in further
detail in the Introduction.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010, summarizes the
differences between the two decades of
objectives, reflecting new knowledge
and direction in this area.
Data Issues
Definitions
Data for objective 5.3 (infertility) is
from the NSFG. Infertility is defined as
the failure of a couple to conceive after
12 months of sexual intercourse without
contraception.
Data Sources
Data for objective 5.1 (adolescent
pregnancy) are based on three outcomes
of pregnancy: live births, fetal losses,
and abortions. Data on live births are
collected annually through the National
Vital Statistics System. For Hispanic
births, it should be noted that the
number of States reporting Hispanic
origin data in their vital statistics has
increased during the monitoring period
(see Appendix). Data for estimates on
fetal losses come from the NSFG, which
is conducted at multiyear intervals; the
most recent data available are from
1995. The 1995 data showed higher
rates than reported by this source for the
1988 NSFG. Fetal loss rates are affected
by the degree to which losses are
detected at very early gestations; it is
believed that these estimates reflect
more complete reporting, rather than a
‘‘real’’ increase in the fetal loss rate.
More information is provided in Vital
Healthy People 2000 Final Review 119
and Health Statistics Report series 21
no. 56 (12).
Estimates of the number of
abortions comes from the Abortion
Provider Survey, conducted by the Alan
Guttmacher Institute (AGI), a
nongovernment organization. The
estimation of the number of abortions is
complex. Nationally, valid data on
abortion are available from only two
sources and the methods of data
collection differ. The Centers for
Disease Control and Prevention (CDC)
collects annual information primarily
through reports from State health
agencies, which vary in completeness,
and the AGI collects data from a direct
survey of all known abortion providers,
which is conducted on a periodic basis.
As a result, the number of abortions
reported by CDC tend to be lower than
the numbers published by AGI. The data
from the Abortion Provider Survey are
adjusted using demographic
characteristics of women obtaining
abortions (in States that report abortions
to CDC) to produce national estimates.
More details are provided in Vital and
Health Statistics series 21 no. 56 (12).
The diversity of sources and the
variability of reporting intervals
complicate tracking of this objective.
The baseline data on inquiry about
family planning for objective 5.10
(counseling by clinicians) are from the
Primary Care Provider Surveys (PCPS)
and refer to the proportion of providers
who routinely provided counseling to
81–100 percent of their female clients of
childbearing age. The sample for the
study was drawn from the membership
rolls of provider organizations for
pediatricians, family physicians,
obstetricians/gynecologists, nurse
practitioners, and internists. Response
rates varied from 50–80 percent across
provider groups. The Prevention in
Primary Care Study (PPCS) was
conducted in 1997–98 to update data
from the PCPS. The design and items
included in the 1997–98 study were
similar to the PCPS, but a slightly
different sampling frame was used and
some items included in the 1992 surveys
were not included in the PPCS. The
professionals were sampled from listings
of all licensed, active practitioners in the
United States whose practices were at
least 50 percent primary care. Because
of low response rates from the other
provider groups, updates are available
only for nurse practitioners.
Data Comparability
Baseline and the 1995 data for ‘‘all
females’’ for objectives 5.4 (adolescent
postponement of sexual intercourse), 5.5
(adolescent abstinence), and 5.6
(contraception use) are from the NSFG.
Baseline and the 1995 data for ‘‘all
males’’ for objectives 5.4, 5.5, and 5.6
are from the National Survey of
Adolescent Males (NSAM). Additional
tracking data for 1990–99 from the
Youth Risk Behavior Survey (YRBS)
are also displayed for these objectives,
but are not directly comparable to the
baselines or targets. The YRBS surveys
adolescents in schools and reports data
by grade rather than age. The NSFG and
the NSAM surveys include all
adolescents regardless of their school
enrollment status. Data from the 1992
National Health Interview Survey
(NHIS) suggest that sexual intercourse
is more common and condom use is less
common among out-of-school youth
14–19 years of age, than among
in-school youth in the same age group.
However, estimates for in-school youth
were very close to those for the total
youth population (19).
The baseline for objective 5.8
(human sexuality discussion) came from
a one-time study by the Planned
Parenthood Foundation that provided
data on persons 13–18 years of age who
had discussed sexuality with their
parents. The 1994 and 1998 data came
from the NHIS, a population-based
survey that provided data on persons
10–17 years of age; 1995 data came
from the NSFG, which provided data on
females 18–19 years old.
References
1. Department of Health and Human Services.
Healthy people 2000: National health promotion
and disease prevention objectives. Washington:
Public Health Service. 1991.
2. Center for Disease Control and Prevention.
Achievements in public health, 1990–99: Family
planning. MMWR 48(47);1073–80.
3. Forrest JD, Samara R. Impact of publicly
funded contraceptive services on unintended
pregnancies and implications for medicaid
expenditures. Fam Plann Perspect 28:188–95.
1996.
4. Kost K, Landry D, Darroch J. Predicting
maternal behaviors during pregnancy: Does
intention status matter? Fam Plann Perspect
30(20):79–88. 1998.
5. Institute of Medicine, NAS. Preventing low
birthweight. Washington. 1985.
6. Centers for Disease Control and Prevention.
Infant mortality by marital status of mother.
United States, 1983. MMWR 39(30):521–2. 1990.
7. Brown SS, Eisenberg L, eds. The best
intentions, unintended pregnancy and the
well-being of children and families. Washington,
DC: National Academy Press.1995.
8. Jarman BJ. Enhancing social and life skills:
Preventing pregnancy among middle school
students. Presentation at what works conference.
University of Indiana. Nov. 1994.
9. Data are from the National Survey of Family
Growth, Centers for Disease Control and
Prevention, National Center for Health Statistics.
1995.
10. Henshaw SK. Unintended pregnancy in the
U.S. Family Planning Perspectives 30(1) 24–69.
1998.
11. Fu J, Darroch JE, Hass, T, et.al. Contraceptive
failure rates: new estimates from the 1995 national
survey of family growth. Fam Plann Perspect
31(2):56–63. 1999.
12. Ventura SJ, Mosher WD, Curtin SC, et.al.
Trends in pregnancies and pregnancy rates by
outcome: Estimates from the United States,
1976–96. National Center for Health Statistics.
Vital Health Stat 21(56):2, 56–57. 2000.
13. Department of Health and Human Services.
Trends in the well-being of America’s children
and youth:1997. Washington: Office of the
Assistant Secretary for Planning and Evaluation.
1997.
14. Singh S, Darroch JE. Adolescent pregnancy
and childbearing: levels and trends in developed
countries. Fam Plann Perspect 32:14–23. 2000.
15. Ventura S, Curtin SC, Matthews TJ. Teenage
births in the United States: National and state
trends 1990–96. National Vital Statistics System.
Hyattsville, Maryland: National Center for Health
Statistics. 1998.
16. Department of Health and Human Services.
Guide to clinical prevention services. 2nd ed.
Washington: Department of Health and Human
Services. 1995.
17. Landry DJ, Forrest JD. Private physicians’
provision of contraceptive services. Fam Plann
Perspect 28(5):203–9. 1996.
18. The Alan Guttmacher Institute. Uneven and
unequal: Insurance coverage and reproductive
health services. New York: the Institute. 1994.
19. Centers for Disease Control and Prevention.
Health risk behaviors among adolescents who do
and do not attend school: United States, 1992.
MMWR 43:129–32. 1994.
120 Healthy People 2000 Final Review

Table 5. Family Planning objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
5.1 Adolescent pregnancy
Pregnancies (per 1,000)
1
Female 10-14 years ......................... 1990
§
3.5 . . . 3.4 3.4 3.3 3.3 3.0 2.8 - - - - - - - - - . . .
Female 15-17 years ......................... 1990
§
80.3 . . . 79.8 77.3 76.8 75.5 71.7 67.8 - - - - - - - - - 50
Live births (per 1,000)
Female 10-14 years ...................... 1990
§
1.4 . . . 1.4 1.4 1.4 1.4 1.3 1.2 1.1 1.0 - - - . . .
Female 15-17 years ...................... 1990
§
37.5 . . . 38.7 37.8 37.8 37.6 36.0 33.8 32.1 30.4 - - - . . .
Abortions (per 1,000)
Female 10-14 years ...................... 1990
§
1.5 . . . 1.4 1.5 1.4 1.3 1.2 1.1 - - - - - - - - - . . .
Female 15-17 years ...................... 1990
§
26.5 . . . 24.3 23.1 22.5 21.4 19.9 19.0 - - - - - - - - - . . .
Fetal losses (per 1,000)
Female 10-14 years ...................... 1990
§
0.5 . . . 0.5 0.5 0.5 0.6 0.5 0.5 - - - - - - - - - . . .
Female 15-17 years ...................... 1990
§
16.2 . . . 16.8 16.4 16.5 16.5 15.9 15.0 - - - - - - - - - . . .
a. Pregnancies, black adolescents (per 1,000)
1
Female 15-19 years ...................... 1990
§
215 . . . 215 211 205 195 178 178 - - - - - - - - - 120
Female 15-17 years ...................... 1990
§
161 . . . 160 156 153 146 133 128 - - - - - - - - - . . .
Live births (per 1,000)
Female 15-19 years ...................... 1990
§
113 . . . 116 112 109 105 96 94 88 85 - - - . . .
Female 15-17 years ...................... 1990
§
82 . . . 84 81 80 76 70 67 61 57 - - - . . .
Abortions (per 1,000)
Female 15–19 years
2
..................... 1990
§
80 ... 77 76 75 70 63 66 --- --- --- ...
Female 15-17 years ...................... 1990
§
5 5 ... 5 3 5 2 5 1 4 8 4 4 4 4 --- --- --- ...
Fetal losses (per 1,000)
Female 15-19 years
2
..................... 1990
§
2 2 ... 2 2 2 2 2 1 2 1 1 9 1 8 --- --- --- ...
Female 15-17 years ...................... 1990
§
2 3 ... 2 3 2 3 2 2 2 1 1 9 1 8 --- --- --- ...
b. Pregnancies, Hispanic adolescents (per 1,000)
1
Female 15-19 years ...................... 1990
§
156 . . . 165 168 166 167 163 157 - - - - - - - - - 105
Live births (per 1,000)
Female 15-19 years
3
..................... 1990
§
100 . . . 107 107 107 108 107 102 97 94 - - - . . .
Abortions (per 1,000)
Female 15-19 years ...................... 1990
§
3 9 ... 4 0 4 3 4 2 4 2 3 9 3 7 --- --- --- ...
Fetal losses (per 1,000)
Female 15-19 years ...................... 1990
§
1 7 ... 1 8 1 8 1 8 1 8 1 8 1 7 --- --- --- ...
5.2 Unintended pregnancy (female 15-44 years) ....... 1988 56% - - - - - - - - - - - - - - - 49% - - - - - - - - - - - - 30%
a. Black female (15-44 years) .................. 1988 78% - - - - - - - - - - - - - - - 72% - - - - - - - - - - - - 40%
b. Hispanic female (15-44 years) ................ 1988 55% - - - - - - - - - - - - - - - 48% - - - - - - - - - - - - 30%
5.3 Infertility
Married couples with wives 15-44 years ............ 1988 7.9% - - - - - - - - - - - - - - - 7.1% - - - - - - - - - - - - 6.5%
a. Black couples with wives 15-44 years .......... 1988 12.1% - - - - - - - - - - - - - - -
4
10.5% - - - - - - - - - - - - 9%
b. Hispanic couples with wives 15-44 years ........ 1988 12.4% - - - - - - - - - - - - - - - 7.0% - - - - - - - - - - - - 9%
See footnotes and key at end of table.
Healthy People 2000 Final Review 121

Table 5. Family Planning objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
5.4* Adolescents who ever engaged in sexual
intercourse
Adolescents 15 years
All females ................................ 1988 27% - - - - - - - - - - - - - - - 22% - - - - - - - - - - - - 15%
In-school females.......................... ... --- 35% 36% --- 37% --- 38% --- 44% --- 43% ...
All males .................................. 1988 33% - - - - - - - - - - - - - - - 27% - - - - - - - - - - - - 15%
In-school males ........................... ... --- 48% 44% --- 45% --- 42% --- 42% --- 34% ...
a. All black males ............................ 1988 69% - - - - - - - - - - - - - - - 58% - - - - - - - - - - - - 15%
In-school non-Hispanic black males .......... ... --- --- 79% --- 82% --- 77% --- 75% --- 68% ...
Adolescents 17 years
All females ................................ 1988 50% - - - - - - - - - - - - - - - 51% - - - - - - - - - - - - 40%
In-school females.......................... ... --- 62% 66% --- 66% --- 67% --- 62% --- 40% ...
All males .................................. 1988 66% - - - - - - - - - - - - - - - 53% - - - - - - - - - - - - 40%
In-school males ........................... ... --- 73% 68% --- 68% --- 65% --- 60% --- 44% ...
b. All black males ............................ 1988 90% - - - - - - - - - - - - - - - 79% - - - - - - - - - - - - 40%
In-school non-Hispanic black males .......... ... --- --- 90% --- 92% --- 88% --- 85% --- 82% ...
c. All black females 15-17 years ................. 1988 66% - - - - - - - - - - - - - - -
4
4 8 % --- --- --- --- 4 0 %
In-school non-Hispanic black females ......... ... --- --- 8 4 % --- 8 0 % --- 7 5 % --- 7 3 % --- 7 3 % ...
5.5* Adolescent abstinence from sexual intercourse for
previous 3 months
All sexually active females 15-17 years ............ 1988 23.6% - - - - - - - - - - - - - - - 27% - - - - - - - - - - - - 40%
In-school sexually active females 15-17 years ...... ... --- 2 4 % 2 5 % --- 2 5 % --- 2 3 % --- 2 3 % --- 2 5 % ...
All sexually active males 15-17 years .............. 1988 33% - - - - - - - - - - - - - - - 37% - - - - - - - - - - - - 40%
In-school sexually active males 15-17 years ....... ... --- 3 0 % 3 6 % --- 3 3 % --- 3 4 % --- 3 2 % --- 3 2 % ...
5.6 Contraception use by sexually active adolescents
Female
First intercourse (15-19 years) ................... 1988 65% - - - - - - - - - - - - - - - 76% - - - - - - - - - - - - 90%
Recent intercourse (15-19 years) ................. 1988 78% - - - - - - - - - - - - - - - 71% - - - - - - - - - - - - 90%
Recent intercourse (In school, 15-17 years) ......... ... --- 7 8 % 8 1 % --- 8 3 % --- 8 3 % --- 8 5 % --- 8 3 % ...
Oral contraceptive and the condom at most
recent intercourse (15-19 years) ................. 1988 2% - - - - - - - - - - - - - - - 8% - - - - - - - - - - - - 90%
Male
Contraception use at most recent intercourse
(15-19 years) ............................... 1990 78% . . . - - - - - - - - - - - - 79% - - - - - - - - - - - - 90%
Contraception use at most recent intercourse
(In school, 15-17 years) ....................... ... --- --- 8 3 % --- 8 4 % --- 8 5 % --- 8 1 % --- 8 6 % ...
Birth control pills and condoms at most recent
intercourse (In school, 15-17 years) .............. 1990
§
2.0% . . . 3.3% - - - 2.9% - - - 3.6% - - - 4.8% - - - 4.2% 90%
Condom and pill use at last intercourse
(17-19 years) ............................... 1988 15% - - - - - - - - - - - - - - - 16% - - - - - - - - - - - - 90%
Condom and pill use at last intercourse
(In school, 17 years and over)................... ... --- 2% 3% --- 4% --- 7% --- 5% --- 6% ...
See footnotes and key at end of table.
122 Healthy People 2000 Final Review

Table 5. Family Planning objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
5.7 Failure of contraceptive method for females
15-44 years ................................ 1988 14% - - - - - - - - - - - - - - - 12% - - - - - - - - - - - - 7%
a. Black female 15-44 years .................... 1988 18% - - - - - - - - - - - - - - - 19% - - - - - - - - - - - - 8%
b. Hispanic female 15-44 years ................. 1988 16% - - - - - - - - - - - - - - - 15% - - - - - - - - - - - - 8%
5.8 Persons ages 13–18 years who have discussed
sexuality with parents........................ 1986 66% - - - - - - - - - - - -
5
73%
6
8 0 % --- --- --- --- 8 5 %
Persons ages 10-17 who have discussed human
sexuality with parents, church, or school ........... ... --- --- --- --- --- 8 9 %
7
9 8 % --- --- 9 2 % --- ...
5.9 Family planning counseling .................... 1984 60% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 90%
5.10* Age-appropriate preconception counseling by
clinicians .................................. ... --- --- --- --- --- --- --- --- --- --- --- 6 0 %
Percent of clinicians routinely providing service
to 81-100% of patients
Inquiry about family planning (female, childbearing age)
Pediatricians ............................... 1992 18% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 60%
Nurse practitioners .......................... 1992 53% . . . . . . . . . - - - - - - - - - - - - - - -
8
42% - - - 60%
Obstetricians/gynecologists .................... 1992 48% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 60%
Internists .................................. 1992 24% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 60%
Family physicians ........................... 1992 28% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 60%
Counseling about family planning
Pediatricians ............................... 1992 36% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 60%
Nurse practitioners .......................... 1992 53% . . . . . . . . . - - - - - - - - - - - - - - -
8
40% - - - 60%
Obstetricians/gynecologists .................... 1992 65% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 60%
Internists .................................. 1992 26% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 60%
Family physicians ........................... 1992 36% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 60%
5.11* Clinic services for HIV and other sexually
transmitted diseases
Family planning clinics ......................... 1989 40% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 50%
Title X funded family planning clinics
STD testing (excluding HIV).................... ... --- --- --- --- --- 95% --- --- --- --- --- ...
STD counseling (excluding HIV) ................ ... --- --- --- --- --- 98% --- --- --- --- --- ...
STD treatment (excluding HIV) ................. ... --- --- --- --- --- 93% --- --- --- --- --- ...
Gonorrhea
Client testing
10
............................. ... --- 97% --- --- --- --- --- --- --- --- --- ...
Client treatment............................. ... --- 82% --- --- --- --- --- --- --- --- --- ...
Partner notification
11
......................... ... --- 23% --- --- --- --- --- --- --- --- --- ...
Partner testing.............................. ... --- 60% --- --- --- --- --- --- --- --- --- ...
Partner treatment ........................... ... --- 62% --- --- --- --- --- --- --- --- --- ...
Syphilis
Client testing
10
............................. ... --- 86% --- --- --- --- --- --- --- --- --- ...
Client treatment............................. ... --- 48% --- --- --- --- --- --- --- --- --- ...
Partner notification
11
......................... ... --- 29% --- --- --- --- --- --- --- --- --- ...
Partner testing.............................. ... --- 57% --- --- --- --- --- --- --- --- --- ...
Partner treatment ........................... ... --- 40% --- --- --- --- --- --- --- --- --- ...
See footnotes and key at end of table.
Healthy People 2000 Final Review 123

Table 5. Family Planning objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Chlamydia
Client testing
10
............................. ... --- 66% --- --- --- --- --- --- --- --- --- ...
Client treatment............................. ... --- 73% --- --- --- --- --- --- --- --- --- ...
Partner notification
11
......................... ... --- 15% --- --- --- --- --- --- --- --- --- ...
Partner testing.............................. ... --- 29% --- --- --- --- --- --- --- --- --- ...
Partner treatment ........................... ... --- 50% --- --- --- --- --- --- --- --- --- ...
HIV
Client pretest counseling ...................... ... --- 66% --- --- --- 82% --- --- --- --- --- ...
Client testing ............................... ... --- 60% --- --- --- 74% --- --- --- --- --- ...
5.12 Contraception use
Female 15-44 years ........................... 1982 88.2%
12
90.1% - - - - - - - - - - - - 92.5% - - - - - - - - - - - - 95%
a. Black female 15-44 years .................... 1982 78.9%
12
84.7% - - - - - - - - - - - - 89.9% - - - - - - - - - - - - 95%
b. Female 15-44 years under 100% poverty ........ 1982 79.6%
12
80.2% - - - - - - - - - - - - 92.1% - - - - - - - - - - - - 95%
c. Female 15-19 years under 200% poverty ........ 1982 67.4%
12
74.9% - - - - - - - - - - - - 84.8% - - - - - - - - - - - - 95%
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
1
Pregnancy rates are calculated from the number of births, fetal losses, and abortions.
2
Nonwhite adolescents.
3
Excludes data from States lacking an Hispanic-origin item on their birth or death certificate.
4
Data for non-Hispanic black females.
5
Data represent the proportion of people 10-17 years who had discussed human sexuality with parents. Proportions for school and church were 76 percent and 32 percent, respectively.
6
1995 data are for females 18–19 years who have ever discussed birth control methods, how pregnancy occurs, or STDs with a parent, or have had a sex educat ion class on birth control
methods, STDs, safe sex, or abstinence.
7
1995 data are for females 18–19 years who have ever discussed birth control methods, how pregnancy occurs, or STDs with a parent.
8
1997–98 data.
9
Response rate for this group was too low to produce reliable estimates.
10
Includes testing at initial visit, at annual visit, or if symptomatic.
11
By family planning clinic staff via telephone or mail.
12
1988 data.
NOTES: Data include revisions and, therefore, may differ from data previously published in these reports and other publications. STD is sexually transmitted disease. HIV is human
immunodeficiency virus.
Met
Toward
Mixed/ no change
Away
Cannot assess
124 Healthy People 2000 Final Review

Objective number Data source
5.1, 5.1a, b Abortion Provider Survey, Alan Guttmacher Institute; National Vital Statistics System, CDC, NCHS; National Survey of Family Growth, CDC, NCHS.
National Vital Statistics System, CDC, NCHS.
National Survey of Family Growth, CDC, NCHS.
5.2, 5.2a, b National Survey of Family Growth, CDC, NCHS.
5.3, 5.3a, b National Survey of Family Growth, CDC, NCHS.
5.4* Baseline and updates for all females and all black females: National Survey of Family Growth, CDC, NCHS.
Baseline and updates for all males and all black males: National Survey of Adolescent Males, NIH, NICHD.
1990–99 data for in-school females and males: Youth Risk Behavior Survey, CDC, NCCDPHP.
5.5* Baseline and update for all females: National Survey of Family Growth, CDC, NCHS.
Baseline and update for all males: National Survey of Adolescent Males, NIH, NICHD.
1990–99 data for in-school females and males: Youth Risk Behavior Survey, CDC, NCCDPHP.
5.6 All females: National Survey of Family Growth, CDC, NCHS.
All males: National Survey of Adolescent Males, NIH, NICHD.
1990–99 data for in-school females and males: Youth Risk Behavior Survey, CDC, NCCDPHP.
5.7, 5.7a, b National Survey of Family Growth, CDC, NCHS.
5.8 Baseline: Planned Parenthood Federation of America, Inc., 1986.
1994 and 1998 updates: National Health Interview Survey, CDC, NCHS.
1995 update: National Survey of Family Growth, CDC, NCHS.
5.9 Baseline: Mech EB. Unpublished. Orientation of Pregnancy Counselors toward Adoption. 1984.
5.10* Baseline: Primary Care Provider Surveys, OPHS, ODPHP.
Update: Prevention in Primary Care Study, American College of Preventive Medicine.
5.11* Baseline: State Family Planning Directors.
1990 data: National Questionnaire on Provision of STD and HIV Services by Family Planning Clinics, OPA.
1994 data: The Urban Institute. Family Planning Clinics: Current status and recent changes in services, clients, staffing, and income sources. March 1994.
5.12, 5.12a–c Forrest JD and Singh S. The Sexual and Reproduction Behavior of American Women, 1982–88. Family Planning Perspectives 22(5):206–14. 1 990.
1995 updates: National Survey of Family Growth, CDC, NCHS.
* Duplicate objective. See full text of objective following this table.
Healthy People 2000 Final Review 125
Family Planning
Objectives
5.1: Reduce pregnancies among females
aged 15–17 to no more than 50 per
1,000 adolescents.
5.1a: Reduce pregnancies among
black adolescent females aged
15–19 to no more than 120 per
1,000.
5.1b: Reduce pregnancies among
Hispanic adolescent females aged
15–19 to no more than 105 per
1,000.
5.2: Reduce to no more than 30 percent
the proportion of all pregnancies that are
unintended.
5.2a: Reduce to no more than
40 percent the proportion of all
pregnancies among black females
that are unintended.
5.2b: Reduce to no more than
30 percent the proportion of all
pregnancies among Hispanic
females that are unintended.
5.3: Reduce the prevalence of infertility
to no more than 6.5 percent.
5.3a: Reduce the prevalence of
infertility among black couples to
no more than 9 percent.
5.3b: Reduce the prevalence of
infertility among Hispanic couples
to no more than 9 percent.
5.4*: Reduce the proportion of
adolescents who have engaged in sexual
intercourse to no more than 15 percent
by age 15 and no more than 40 percent
by age 17.
Duplicate objectives: 18.3 and 19.9
5.4a*: Reduce the proportion of
black males aged 15 years who
have engaged in sexual intercourse
to no more than 15 percent.
Duplicate objectives: 18.3a and
19.9a
5.4b*: Reduce the proportion of
black males aged 17 years who
have engaged in sexual intercourse
to no more than 40 percent.
Duplicate objectives: 18.3b and
19.9b
5.4c*: Reduce the proportion of
black females aged 17 years who
have engaged in sexual intercourse
to no more than 40 percent.
Duplicate objectives: 18.3c and
19.9c
5.5*: Increase to at least 40 percent the
proportion of ever sexually active
adolescents aged 17 and younger who
have not had sexual intercourse during
the previous 3 months.
Duplicate objectives: 18.15 and 19.16
5.6: Increase to at least 90 percent the
proportion of sexually active, unmarried
people aged 15–24 who use
contraception, especially combined
method contraception that both
effectively prevents pregnancy and
provides barrier protection against
disease.
5.7: Increase the effectiveness with
which family planning methods are
used, as measured by a decrease to no
more than 7 percent in the proportion of
women experiencing pregnancy despite
use of a contraceptive method.
5.7a: Increase the effectiveness with
which family planning methods are
used, as measured by a decrease to
no more than 8 percent in the
proportion of black females
experiencing pregnancy in the last
year despite use of a contraceptive
method.
5.7b: Increase the effectiveness with
which family planning methods are
used, as measured by a decrease to
no more than 8 percent in the
proportion of Hispanic females
experiencing pregnancy in the last
year despite use of a contraceptive
method.
5.8: Increase to at least 85 percent the
proportion of people aged 10–18 who
have discussed human sexuality,
including correct anatomical names,
sexual abuse, and values surrounding
sexuality, with their parents and/or have
received information through another
parentally endorsed source, such as
youth, school, or religious programs.
5.9: Increase to at least 90 percent the
proportion of family planning counselors
who offer accurate information about all
options, including prenatal care and
delivery, infant care, foster care, or
adoption and pregnancy termination to
their patients with unintended
pregnancies.
5.10*: Increase to at least 60 percent the
proportion of primary care providers
who provide age-appropriate
preconception care and counseling.
Duplicate objective: 14.12
5.11*: Increase to at least 50 percent the
proportion of family planning clinics,
maternal and child health clinics,
sexually transmitted disease clinics,
tuberculosis clinics, drug treatment
centers, and primary care clinics that
provide on site primary prevention and
provide or refer for secondary
prevention services for HIV infection
and bacterial sexually transmitted
diseases (gonorrhea, syphilis, and
Chlamydia) to high-risk individuals and
their sex or needle-sharing partners.
Duplicate objectives: 18.13 and 19.11
5.12: Increase to at least 95 percent the
proportion of all females aged 15–44 at
risk of unintended pregnancy who use
contraception.
5.12a: Increase to at least
95 percent the proportion of black
females aged 15–44 at risk of
unintended pregnancy who use
contraception.
5.12b: Increase to at least
95 percent the proportion of females
aged 15–44 with income less than
100 percent of poverty at risk of
unintended pregnancy who use
contraception.
5.12c: Increase to at least 95 percent
the proportion of females aged
15–19 years under 200 percent of
poverty at risk of unintended
pregnancy who use contraception.
*Duplicate objective.
126 Healthy People 2000 Final Review

Priority Area 6
Mental Health and
Mental Disorders
Background
Major mental disorders continue to
affect large numbers of persons in the
United States. Suicide is one of the most
serious consequences of these disorders,
but other physical, emotional, social,
and economic costs occur as well—not
only for individuals but also for
families, communities, and government.
The most common disorders include
forms of anxiety, panic, agoraphobia,
social phobia, simple phobia,
post-traumatic stress and generalized
anxiety, mood disorders, major
depressive episodes, and manic episodes
or dysthymia. Other major conditions
that significantly affect quality of life
include schizophrenia and other
nonaffective psychoses, conduct
disorders, and antisocial personality
disorders.
In 1992, nearly 40 million persons
in the United States between the ages of
15 and 54 years experienced a major
mental disorder, approximately
24 percent of the population, slightly
more than the number experiencing
cardiovascular disorders. The lifetime
estimate of the prevalence of mental
illness is even higher (49 percent) (1,2).
Individuals affected by major mental
disorders are at unusually high risk for
suicide, as well as for social disabilities,
risk-laden life trajectories, and the
development of other disorders, mental
and physical. The costs precipitated by
these major disorders in the United
States approximated $150 billion in
1996. Treatment costs accounted for $69
billion, including approximately $37
billion from public funds; lost
productivity or premature death
accounted for another $75 billion; and
criminal justice involvement with the
mentally ill and property destruction
cost some $6 billion (3).
In 1990, more than 5 million people
were admitted to mental health facilities
for treatment; about 62 percent were
treated on an outpatient basis in
hospitals, mental health clinics, and
other facilities, with self-help and
human service organizations providing
most supplementary and alternative care
(4). Public insurance pays for treatment
of roughly 48 percent of those admitted
for inpatient treatment and 35 percent of
those admitted as outpatients (5).
Despite the volume of persons who
received treatment, only 40 percent of
those who experience mental illness
receive treatment (6), and only
one-quarter of these receive care from
the mental health sector (1).
Among people with early onset
disorders, the probability of no lifetime
treatment contact or delay in contact is
unusually high. This is particularly
likely for those with childhood-onset
mood or anxiety disorders, which are
also more severe and disabling than
later onset disorders. However, the
majority of persons who experience
other major disorders eventually do seek
treatment. On average, this takes place
between 6 and 14 years later, depending
on the disorder. Comorbidity appears to
be part of this treatment-seeking
process, increasing the likelihood of
both 12-month (34 percent) and lifetime
(42 percent) treatment contact (1).
Altogether, when severity and
persistence of the disorder or level of
associated role impairment is taken into
account (among persons 18–54 years of
age), the likelihood of comorbid
substance use, service use, frequent
service use, and service use in the
mental health specialty sector and in
self-help groups increases markedly.
Given these facts, avoiding
disabling outcomes of the major mental
disorders, intervening early in their
course, and preventing their onset all
become important strategies for
improving the health of the Nation.
Advances in research, such as those
reviewed in a recent Institute of
Medicine report (7), have provided
additional impetus for this complex
undertaking. Numerous conditions,
biologic and genetic vulnerabilities,
acute or chronic physical dysfunction,
insecure attachment styles, personality
characteristics, and a range of
environments, have been found to affect
both mental disorders and mental health.
Addressing this array of contingencies
through preventive interventions requires
a mix of minimizing risk factors and
maximizing protective factors in a
combination of preventive and treatment
interventions.
Data Summary
Highlights
By the end of the decade, progress
in achieving objectives was most
remarkable for those concerned with
enhancing protective factors and those
concerned with reducing
mortality/physical morbidity linked to
disorder.
The enhancements involved both
system development and individual
action. Mutual help clearinghouses
became available to all 50 States (6.12).
The number of adults seeking help with
their emotional and personal problems
(6.8) increased as anticipated and moved
toward the target for those with
disabilities (6.8a). The use of
community support among people with
severe mental disorders (6.6) also
increased past the targeted 30 percent.
The reductions in mortality and
morbidity linked to disorder involved
both suicide and physical illness.
Suicide rates overall, and in particular
among males ages 65 years and over,
and among adolescents 15–19 years of
age, declined below the target (6.1, 6.1c,
and 6.1a); suicide rates among males
20–34 years of age moved toward the
target (6.1b). Adverse health effects
from stress declined as anticipated
among adults in general (6.5) and
moved toward the target for those with
disabilities (6.5a).
On the other hand, significant
reversals were seen specific to
objectives involving interventions in
high-risk environments and suicidality
among adolescents. Objectives regarding
the number of States meeting nationally
recognized prison suicide prevention
standards (6.8) and adults not taking
steps to control stress (6.9) lost ground.
Objectives concerning suicide attempts
among adolescents (14–17 years of age)
and among female adolescents (6.2,
6.2a) also lost ground.
Summary of Progress
Year 2000 targets were met for five
objectives (6.1, 6.5, 6.6, 6.8, and 6.12).
One objective moved toward its target
(6.4), although progress was based on
estimated data from the prior decade.
Seven objectives moved away from their
targets (6.2, 6.7, 6.9, 6.10, 6.11, 6.14,
and 6.15).Two of these objectives (6.7
and 6.15) had very slight changes and
have not been updated since a 1990–92
Healthy People 2000 Final Review 127
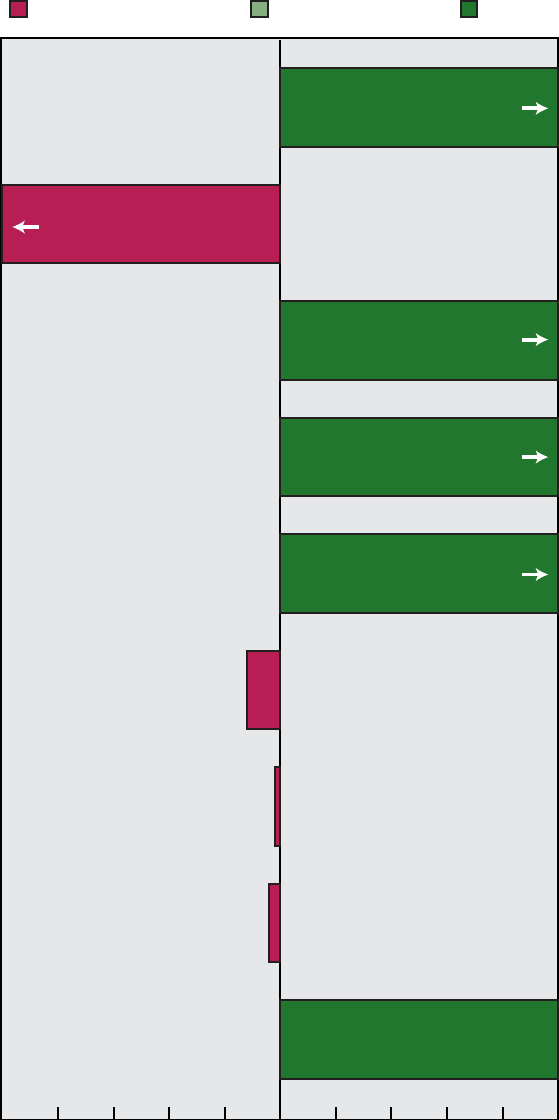
6.1 Suicide
6.2 Suicide attempts among adolescents
6.5 People with adverse health effects from stress
6.6 Use of community support among people with
severe mental disorders
6.8 People seeking help with emotional problems
6.9 People not taking steps to control stress
6.11 Worksite stress management programs
6.12 States with mutual help clearinghouses
6.10 States meeting prison suicide prevention standards
Moved away from target Moved toward target Met target
NOTE: Complete tracking data are shown in table 6. Progress quotients are not calculated for objectives 6.3, 6.4, 6.7, 6.13, 6.14, and 6.15.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
-12%
-2%
-4%
Percent of target achieved
-100 -80 -60 -40 -20 0 20 40 60 80 100
114%
108%
131%
125%
100%
-167%
128 Healthy People 2000 Final Review
Figure 6. Final status of Mental Health and Mental Disorders objectives

estimate. Another two objectives (6.10
and 6.11) that appeared to move away
from targets did show progress based on
supplemental data. Objective (6.13)
showed mixed results. Progress for 6.13
and 6.14 was based on data limited to
nurse practitioners only. No national
data were available for one objective
(6.3). Details regarding measurement
issues are available in the following
section on data issues. See table 6 for
the tracking data for the objectives in
this priority area and figure 6 for a
quantitative assessment of progress.
Discussion
The 1990s saw a tremendous
increase in awareness of mental health
issues with the recognition that mental
health in part reflects a healthy
community. This is reflected in several
of the objectives that obtained the
targets set for the year 2000; a rise in
people seeking help with emotional
problems (6.8), a rise in the use of
community support among people with
severe mental disorders (6.6), a decline
in people with adverse health effects
from stress (6.5), a decline in suicide
(6.1), and all States being covered by
mutual help clearinghouses (6.12).
In 1999, the end of the ‘‘Decade of
the Brain’’ saw the first Surgeon
General’s Report on Mental Health (3),
which called attention to the body of
research testing effective treatments and
service delivery models and their
implications for advancing the state of
mental health in the Nation. Factors
influencing trends in mental health
include: increased third-party financing
and the provision of services through
managed care, an international
recognition of the ‘‘burden of disease’’
that documents depression as the leading
cause of disability worldwide among
persons 5 years of age and older, and a
rising concern with the translation of
resilience research into services research
and practice.
Other national events that have
provided guidance for mental health
concerns at the end of the decade (1999)
include:
The White House Conference on
Mental Health
National antistigma campaign
The Surgeon General’s Call to
Action on Suicide Prevention
The Surgeon General’s conference
on Children’s Mental Health
Inclusion of the treatment of
recognized depression as a National
Leading Health Indicator for the
decade 2000–2010
Transition to Healthy People
2010
Data development, increasing access
to services, eliminating disparities, and
providing for mental health needs across
the life span continue as themes into the
new decade for Healthy People 2010.
There are 14 objectives included in the
Mental Health and Mental Disorders
chapter for Healthy People 2010,
presented in terms of mental health
status improvement, treatment
expansion, and State activities. Only two
objectives are taken directly from
Healthy People 2000, suicide (6.1) and
suicide attempts among adolescents (6.2)
(with suicide age-adjusted to the 2000
population). Treatment of depression
among adults (6.7) appears in Healthy
People 2010 as well as Healthy People
2000, although the objective has been
expanded to monitor other serious
mental illnesses such as anxiety
disorders and schizophrenia as well. The
objective to track the percent of
worksites that have stress management
programs (6.11) has also been included
in Healthy People 2010; it has been
relocated in the Occupational Health and
Safety chapter.
The Healthy People 2010 objective
for adults with recognized depression
who receive treatment, which was
retained from Healthy People 2000
(with a different data source and age
group), has been designated as a
measure of one of the Leading Health
Indicators (LHI). The LHIs are
discussed in further detail in the
Introduction.
Four new objectives were added to
Healthy People 2010, they include:
reduce the proportion of homeless adults
who have serious mental illness,
increase the proportion of persons with
serious mental illness who are
employed, increase the number of States
that track satisfaction with the mental
health services received, and increase
the number of States with an operational
mental health plan that addresses mental
health crisis interventions, ongoing
screening, and treatment services for the
elderly.
In addition, seven new
developmental objectives (objectives
without current baseline data) appear for
mental health in Healthy People 2010.
They include: reduce the relapse rates
for persons with eating disorders,
increase the number of persons seen in
primary health care who receive mental
health screening and assessment,
increase the proportion of children with
mental health problems who receive
treatment, increase the proportion of
juvenile justice facilities that screen new
admissions for mental health problems,
increase the proportion of persons with
co-occurring substance abuse and mental
disorders who receive treatment for both
disorders, increase the proportion of
local governments with
community-based jail diversion
programs for adults with serious mental
illness, and increase the number of
States with an operational mental health
plan that addresses cultural competence.
Baseline data are expected to be
developed for these objectives by 2004.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the changes between the
two decades of objectives, reflecting
new knowledge and direction for this
area.
Data Issues
Definitions
Operational definitions and data
collection specifications for all Healthy
People 2000 objectives in Priority Area
6 have been published in the National
Center for Health Statistics’ Healthy
People Statistical Notes series (8). Data
issues are discussed and references are
cited for expanded discussions of the
data systems that provide data for the
national objectives. Where appropriate,
the text of the questionnaire items used
to measure the objectives is also
provided. See Appendix table VII for
further information.
Objective 6.1 (suicide) is monitored
using data from the National Vital
Statistics System (NVSS). The data are
compiled from death certificates
submitted by the States. Differentiating
suicide deaths from accidental deaths
relies heavily on judgment by the
medical legal officer (for example,
coroner or medical examiner). A key
element of this determination is the
establishment of intent by the deceased.
This determination may be based on
information about prior suicide attempts,
a statement or note by the deceased
Healthy People 2000 Final Review 129
indicating their intent to commit suicide,
or other clinical information (for
example, serious mental illness) (9).
Objective 6.2 (adolescent suicide
attempts) is monitored with data from
the Youth Risk Behavior Survey
(YRBS), a school-based survey. Suicide
attempts are self-reported and are
limited to those that required medical
attention in the last 12 months. The
exclusion of adolescents not in school
from the data used to monitor objective
6.2 may underestimate the actual
number of suicide attempts (10). Data
from the 1992 National Health Interview
Survey (NHIS) youth supplement
suggest that other types of violent
behavior (weapons-carrying and
fighting) are higher among youth (14–19
years of age) not in school than among
those in school; the estimates for
fighting and weapons-carrying for
in-school youth were very close to
estimates for the total population. (The
NHIS youth supplement did not include
questions on suicide attempts.) Reliance
on self-report of suicide attempts that
resulted in hospitalization, without
validation from medical sources, may
also affect the accuracy of estimates.
However, a recent study by the Centers
for Disease Control and Prevention
(CDC) indicates that estimates among
in-school youth are highly reliable (11).
The wording and baseline data for
objective 6.10 (suicide prevention in
jails) were established using States as
the organizational level for monitoring
and implementing suicide prevention
protocols in jails. However, jails are
usually under the jurisdiction of counties
or municipalities. State-level data on
jails are limited; the alternative data
track the objective using jails as the unit
of analysis. Data from the National
Census of Jails, conducted by the
Bureau of Justice Statistics, are only
available for 1993 but later updates are
expected. Additional data are from the
American Correctional Association’s
(ACA) list of jails, which are ACA
accredited; their accreditation requires
that suicide prevention policies and
training be implemented in the jail.
However, not all jails seek ACA
accreditation; this selection bias suggests
that these data may not be nationally
representative.
Data Sources
Data for objective 6.9 (people not
taking steps to control stress) are from
the Prevention Index, Rodale Press. Data
are collected from a random sample
telephone survey weighted to census
data for sex, race, education, and region
of the country.
Baseline data for objectives 6.13
(clinical review of childhood mental
functions) and 6.14 (clinical review of
adult mental functions) are from the
Primary Care Providers Surveys (PCPS).
The samples for the surveys were drawn
from the membership rolls of provider
organizations for family physicians,
nurse practitioners, internists,
obstetricians/gynecologists, and
pediatricians (6.14). Response rates
ranged from 50 to 80 percent. The data
on assessment and screening represent
the proportion of providers who
routinely queried 81–100 percent of their
patients about a particular type of
mental function. Data on treatment and
referral refer to the proportion of
providers who provided or referred
patients who needed the services. The
Prevention in Primary Care Study
(PPCS) was conducted in 1997–98 to
update data from the PCPS. The design
and items included in the 1997–98 study
were similar to the PCPS, but a slightly
different sampling frame was used and
some items included in the 1992 surveys
were not included in the PPCS. The
professionals were sampled from listings
of all licensed, active practitioners in the
United States whose practices were at
least 50 percent primary care. Because
of low response rates from the other
provider groups, updates are available
only for nurse practitioners. These
updates are based on a partial
characterization of medical screening
activity and may not fully represent the
primary care provider community.
Data Comparability
Baselines for objectives 6.4 (adult
mental disorders), 6.7 (treatment for
depression), and 6.15 (prevalence of
depression) came from the National
Institute for Mental Health (NIMH)
Epidemiologic Catchment Area (ECA)
studies conducted in five metropolitan
areas during the early 1980s. This
household survey used the Diagnostic
Interview Schedule (DIS) and the
Diagnostic and Statistical Manual of
Mental Disorders (DSM-III) criteria to
estimate 1-month prevalences, which
were used to set the baseline and target.
The updates for these objectives come
from the National Comorbidity Survey
(NCS), which is a national survey that
collects prevalence data using the
Composite International Diagnostic
Interview (CIDI) and DSM-IIIR and IV
criteria. Planned monitoring of the
objectives originally involved reanalysis
of ECA data to produce 1-year
prevalence estimates and recoding of
NCS data to reflect DSM-III categories.
Subsequent conduct of an
NCS-replication in the year 2000 will
provide comparative data more
appropriate to the decade, the currently
used diagnostic scheme, and
generalization to the national population.
These prevalence and treatment findings
will be available in 2001.
Data for objective 6.11 (worksite
stress management programs) are from
the National Survey of Worksite Health
Promotion Activities, which were
telephone surveys of nongovernment
worksites. Some of the businesses
surveyed had multiple worksites with
different health promotion activities.
Additionally, both active (for example,
classes) and passive (for example,
brochures) methods were counted as
worksite health promotion activities.
References
1. Kessler R, et al. Lifetime and 12-month
prevalence of DSM-IIIR psychiatric disorders in
the U.S. Arch Gen Psychiatry 51:8–19. 1994.
2. National Advisory Mental Health Council.
Health care reform for Americans with severe
mental illness: Report of the National Advisory on
Mental Health Council. Am J Psychiatry 150:
1447–64. 1993.
3. Department of Health and Human Services.
Mental Health: A report of the Surgeon General.
Rockville, Maryland: Department of Health and
Human Services, Substance Abuse and Mental
Health Services Administration, Center for Mental
Health Services, National Institutes of Health,
National Institute of Mental Health. 1999.
4. Rouse BA (ed). Substance abuse and mental
health statistical sourcebook. Rockville, Maryland:
Substance Abuse and Mental Health Services
Administration, 180–4. 1997.
5. Frank RG, et al. Paying for mental health and
substance abuse care. Health Aff 13: 237–42.
1994.
6. Robins LN, et al. An overview of psychiatric
disorders in America. In: Robins LN, Regier DA,
eds. Psychiatric disorders in America: The
Epidemiologic Catchment Area Study. New York:
Free Press. 1991.
7. Mrazek PJ, Haggery RJ, eds. Reducing risks for
mental disorders. Washington: Institutes of
Medicine, National Academy Press. 1994.
8. Seitz F, Jonas B. Operational definitions for
year 2000 objectives: Priority area 6, Mental
health and mental disorders. Healthy people
130 Healthy People 2000 Final Review
statistical notes; no 16. Hyattsville, Maryland:
National Center for Health Statistics. 1998.
9. Rosenberg M, et al. Operational criteria for the
determination of suicide. J Forensic Sci
33:1445–6. 1988.
10. Centers for Disease Control and Prevention.
Health risk behaviors among adolescents who do
and do not attend school: United States, 1992.
MMWR 43:129–32. 1994.
11. Brener N, et al. Reliability of the Youth Risk
Behavior Survey questionnaire. Paper presented at
the annual meeting of the American Public Health
Association, Oct 1994.
Healthy People 2000 Final Review 131

Table 6. Mental Health and Mental Disorders objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
6.1* Suicide (age adjusted per 100,000) ............... 1987 11.7 11.5 11.4 11.1 11.3 11.2 11.2 10.8 10.6 10.4 - - - 10.5
a. Adolescents 15-19 years (per 100,000) ......... 1987 10.2 11.1 11.0 10.8 10.9 11.1 10.5 9.7 9.5 8.9 - - - 8.2
b. Male 20-34 years (per 100,000) ............... 1987 25.2 25.1 25.1 24.5 25.5 26.5 26.3 24.2 23.4 22.9 - - - 21.4
c. White male 65 years and over (per 100,000)...... 1987 46.7 44.4 42.7 41.0 40.9 38.9 38.7 37.8 36.1 38.2 - - - 39.2
d. American Indian/Alaska Native male
(age adjusted per 100,000) ................. 1987 20.1 21.0 19.2 17.9 18.7 23.8 20.1 20.0 21.3 21.4 - - - 17.0
6.2* Suicide attempts among adolescents 14–17 years . . 1990 2.1% . . . 1.7% - - - 2.7% - - - 2.8% - - - 2.6% - - - 2.6% 1.8%
a. Female 14-17 years ........................ 1991 2.5% . . . . . . - - - 3.8% - - - 3.4% - - - 3.3% - - - 3.1% 2.0%
6.3 Mental disorders
Children and adolescents 18 years and under ........ 1988 20% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 17%
6.4 Mental disorders among adults 18–54 years
(1-month prevalence) ......................... 1981–85 12.6% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 10.7%
Mental disorders among adults 18-54 years
(1-year prevalence) ........................... ... ---
1
20.4% - - -
2
16.0% - - - - - - - - - - - - - - - - - - - - - . . .
6.5 Adverse health effects from stress for people
18 years and over ........................... 1985 44.2% 40.6% - - - - - - 39.2% - - - 33.9% - - - - - - 33.7% - - - 35%
a. People 18 years and over with disabilities........ 1985 53.5% 54.2% - - - - - - 54.9% - - - 49.1% - - - - - - - - - - - - 40%
6.6 Use of community support among people 18 years
and over with severe mental disorders .......... 1986 15% - - - - - - - - - - - -
3
34.6% - - - - - - - - - - - - - - - 30%
6.7 Treatment for depression among people
18–54 years (6-month services) ................. 1981-85 31% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 54%
Treatment for depression among people 18–54 years
(1-year services) ............................ ... ---
1
34.7% - - -
2
34.2% - - - - - - - - - - - - - - - - - - - - - . . .
6.8 People 18 years and over seeking help with
emotional/personal problems .................. 1985 11.1% 12.5% - - - - - - 14.3% - - -
4
18.9% - - - - - - 22.2% - - - 20%
a. People 18 years and over with disabilities........ 1985 14.7% 17.0% - - - - - - 19.8% - - -
4
26.6% - - - - - - - - - - - - 30%
6.9 People 18 years and over not taking steps to
control stress .............................. 1985
§
31% --- 33% --- 36% 34% 34% --- --- --- --- 5%
6.10* Suicide prevention in jails
Number of States meeting nationally recognized suicide
prevention standards.......................... 1992
§
2 ... ... ... --- --- 2 1 --- --- --- 50
Proportion of jails with suicide policies.............. ... --- --- --- --- 79.5% - - - - - - - - - - - - - - - - - - . . .
Proportion of jails with ACA accreditation............ ... --- 1% 1% 1% 2% 2% 2% --- --- --- 3% ...
6.11 Worksite stress management programs .......... 1985 26.6% - - - - - - 37.0% - - - - - - - - - - - - - - - - - -
5
26.0% 40%
6.12 Mutual self-help network
Number of States with mutual help clearinghouses .... 1995 8 . . . . . . . . . . . . . . . . . . - - - - - - - - - - - - 50
Number of Federal clearinghouses ................ 1995
6
2 ... ... ... ... ... ... --- --- --- --- ...
6.13 Clinician review of patients’ mental functioning .... ... --- --- --- --- --- --- --- --- --- --- --- 60%
Percent of clinicians routinely providing service to
81-100% of patients
Inquiry about cognitive functioning
Nurse practitioners .......................... 1992 35% . . . . . . . . . - - - - - - - - - - - - - - -
7
19% - - - 60%
Obstetricians/gynecologists .................... 1992 9% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 60%
See footnotes and key at end of table.
132 Healthy People 2000 Final Review

Table 6. Mental Health and Mental Disorders objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Internists .................................. 1992 18% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 60%
Family physicians ........................... 1992 7% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 60%
Inquiry about emotional/behavioral functioning
Nurse practitioners .......................... 1992 40% . . . . . . . . . - - - - - - - - - - - - - - -
7
26% - - - 60%
Obstetricians/gynecologists .................... 1992 12% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 60%
Internists .................................. 1992 25% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 60%
Family physicians ........................... 1992 13% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 60%
Treatment/referral for cognitive problems
Nurse practitioners .......................... 1992 20% . . . . . . . . . - - - - - - - - - - - - - - -
7
24% - - - 60%
Obstetricians/gynecologists .................... 1992 20% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 60%
Internists .................................. 1992 27% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 60%
Family physicians ........................... 1992 21% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 60%
Treatment/referral for emotional/behavioral problems
Nurse practitioners .......................... 1992 23% . . . . . . . . . - - - - - - - - - - - - - - -
7
33% - - - 60%
Obstetricians/gynecologists .................... 1992 23% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 60%
Internists .................................. 1992 35% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 60%
Family physicians ........................... 1992 27% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 60%
6.14 Clinician review of children’s mental functioning ... ... --- --- --- --- --- --- --- --- --- --- --- 75%
Percent of clinicians routinely providing service to
81–100% of patients
Inquiry about cognitive functioning
Pediatricians ................................ 1992 62% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 75%
Inquiry about emotional/behavioral functioning
Pediatricians ............................... 1992 47% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 75%
Treatment/referral for cognitive problems
Pediatricians ............................... 1992 51% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 75%
Treatment/referral for emotional/behavioral problems
Pediatricians ............................... 1992 45% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 75%
Inquiry about parent-child relationship
Pediatricians ............................... 1992 55% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 75%
Nurse practitioners .......................... 1992 55% . . . . . . . . . - - - - - - - - - - - - - - -
7
51% - - - 75%
Family physicians ........................... 1992 36% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 75%
Treatment/referral for parent-child interaction problems
Pediatricians ............................... 1992 34% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 75%
Nurse practitioners .......................... 1992 24% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 75%
Family physicians ........................... 1992 29% . . . . . . . . . - - - - - - - - - - - - - - -
7,8
--- --- 75%
See footnotes and key at end of table.
Healthy People 2000 Final Review 133

Table 6. Mental Health and Mental Disorders objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
6.15 Prevalence of depression in people 18–54 years
(1-month prevalence) ......................... 1981-85 5.1% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 4.3%
Prevalence of depression in people 18–54 years
(1-year prevalence)........................... ... ---
1
10.9% - - -
2
11.1% - - - - - - - - - - - - - - - - - - - - - . . .
a. Female 18-54 years (1-month prevalence) ....... 1981-85 6.6% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 5.5%
Female 18-54 years (1-year prevalence) ........ ... ---
1
14.2% - - -
2
13.1% - - - - - - - - - - - - - - - - - - - - - . . .
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
1
1981-85 data. Data are for noninstitutionalized, nonrural, white, black, or Hispanic people 18–54 years.
2
1990-92 data. Data are for noninstitutionalized, nonrural, white, black, or Hispanic people 18–54 years.
3
Estimate represents the proportion of people with mental disorders (excluding substance disorders only) in the past year which interfered with their ability to work or find work and who sought
help from community mental health services.
4
Estimate represents the proportion of people who sought help from a family member, therapist, or minister/rabbi. Individual values were: family members, 15.6 percent (21.0 percent for people
with disabilities), therapist, 6.8 percent (11.8 percent for people with disabilities), and minister/rabbi, 4.4 percent (6.9 percent for people with disabilities).
5
1998-99 data. Only 26 percent of worksites had onsite stress management programs, but 48 percent offered stress management either onsite or through t he health plan.
6
Clearinghouses provide coverage for all 50 states, establishing the required network.
7
1997-98 data.
8
Response rate for this group was too low to produce reliable estimates.
NOTE: Data include revisions and, therefore, may differ from data previously published in these reports and other publications.
Met
Toward
Mixed/ no change
Away
Cannot assess
134 Healthy People 2000 Final Review

Objective number Data source
6.1*, 6.1a-d National Vital Statistics System, CDC, NCHS.
6.2, 6.2a* Youth Risk Behavior Survey, CDC, NCCDPHP.
6.3 Bird HR. Estimates of the prevalence of childhood maladjustment in a community survey in Puerto Rico. Archives of Gen Psychiatry 45:1120–26. 1988 .
Costello EJ, et al. Psychiatric disorders in pediatric primary care: Prevalence risk factors.
Archives of Gen Psychiatry 45:1107–16. 1988.
6.4 Baseline: Epidemiologic Catchment Area Study, NIH, NIMH.
1990 and 1992 data: National Comorbidity Survey, University of Michigan.
6.5, 6.5a National Health Interview Survey, CDC, NCHS.
6.6 Baseline: National Institute of Mental Health Community Support Program Client Follow-up Study, SAMHSA.
1994 data: National Health Interview Survey, CDC, NCHS.
6.7 Baseline: Epidemiologic Catchment Area Study, NIH, NIMH.
Updates: National Comorbidity Survey, University of Michigan.
6.8, 6.8a National Health Interview Survey, CDC, NCHS.
6.9 Prevention Index, Rodale Press, Inc.
6.10* Baseline and updates (States): National Study of Jails, National Center on Institutions and Alternatives.
Baseline (suicide policies): National Census of Jails, DOJ, BJS.
Baseline and update (ACA accreditation): American Correctional Association.
6.11 National Survey of Worksite Health Promotion Activities, OPHS, ODPHP; 1998–99 update: National Worksite Health Promotion Survey, OPHS, ODPHP .
6.12 SAMHSA.
6.13 Baseline: Primary Care Provider Surveys, OPHS, ODPHP.
Update: Prevention in Primary Care Study, American College of Preventive Medicine.
6.14 Baseline: Primary Care Provider Surveys, OPHS, ODPHP.
Update: Prevention in Primary Care Study, American College of Preventive Medicine.
6.15 Baseline: Epidemiologic Catchment Area Study, NIH, NIMH.
1990 and 1992 data: National Comorbidity Survey, University of Michigan.
* Duplicate objective. See full text of objective following this table.
Healthy People 2000 Final Review 135
Mental Health and
Mental Disorders
Objectives
6.1*: Reduce suicides to no more than
10.5 per 100,000 people.
Duplicate objective: 7.2
6.1a*: Reduce suicides among
youth aged 15–19 to no more than
8.2 per 100,000.
Duplicate objective: 7.2a
6.1b*: Reduce suicides among men
aged 20–34 to no more than 21.4
per 100,000.
Duplicate objective: 7.2b
6.1c*: Reduce suicides among white
men aged 65 and older to no more
than 39.2 per 100,000.
Duplicate objective: 7.2c
6.1d*: Reduce suicides among
American Indian and Alaska Native
men to no more than 17.0 per
100,000.
Duplicate objective: 7.2d
6.2*: Reduce to 1.8 percent the
incidence of injurious suicide attempts
among adolescents aged 14–17.
Duplicate objective: 7.8
6.2a*: Reduce to 2.0 percent the
incidence of injurious suicide
attempts among female adolescents
aged 14–17.
Duplicate objective: 7.8a
6.3: Reduce to less than 17 percent the
prevalence of mental disorders among
children and adolescents.
6.4: Reduce the prevalence of mental
disorders (exclusive of substance abuse)
among adults living in the community to
less than 10.7 percent.
6.5: Reduce to less than 35 percent the
proportion of people aged 18 and older
who report adverse health effects from
stress within the past year.
6.5a: Reduce to less than 40 percent
the proportion of people with
disabilities who report adverse
health effects from stress within the
past year.
6.6: Increase to at least 30 percent the
proportion of people aged 18 and older
with severe, persistent mental disorders
who use community support programs.
6.7: Increase to at least 54 percent the
proportion of people with major
depressive disorders who obtain
treatment.
6.8: Increase to at least 20 percent the
proportion of people aged 18 and older
who seek help in coping with personal
and emotional problems.
6.8a: Increase to at least 30 percent
the proportion of people with
disabilities who seek help in coping
with personal and emotional
problems.
6.9: Decrease to no more than 5 percent
the proportion of people aged 18 and
older who report experiencing
significant levels of stress who do not
take steps to reduce or control their
stress.
6.10*: Increase to 50 the number of
States with officially established
protocols that engage mental health,
alcohol and drug, and public health
authorities with corrections authorities to
facilitate identification and appropriate
intervention to prevent suicide by jail
inmates.
Duplicate objective: 7.18
6.11: Increase to at least 40 percent the
proportion of worksites employing 50 or
more people that provide programs to
reduce employee stress.
6.12: Establish a network to facilitate
access to mutual self-help activities,
resources, and information by people
and their family members who are
experiencing emotional distress resulting
from mental or physical illness.
6.13: Increase to at least 60 percent the
proportion of primary care providers
who routinely review with patients their
patients’ cognitive, emotional, and
behavioral functioning and the resources
available to deal with any problems that
are identified.
6.14: Increase to at least 75 percent the
proportion of providers of primary care
for children who include assessment of
cognitive, emotional, and parent-child
functioning with appropriate counseling,
referral, and followup, in their clinical
practices.
6.15: Reduce the prevalence of
depressive (affective) disorders among
adults living in the community to less
than 4.3 percent.
6.15a: Reduce the prevalence of
depressive (affective) disorders
among women living in the
community to less than 5.5 percent.
*Duplicate objective.
136 Healthy People 2000 Final Review

Priority Area 7
Violent and Abusive
Behavior
Background
Injuries and deaths due to violence
and abusive behavior continue to be
pervasive in the United States and cost
the Nation over $200 billion annually.
Violence claims the lives of many of the
Nation’s youth and threatens the health
and well-being of persons of all ages.
Each year approximately 50,000 persons
in the United States die from
violence-related injuries. On an average
day, 53 persons die from homicide, and
a minimum of 18,000 persons survive
interpersonal assaults, 84 persons
complete suicide, and as many as 3,000
persons attempt suicide (1).
The United States has the highest
rates of lethal childhood violence when
compared to other industrialized
countries (2). In 1997, nearly 19,000
children 19 years of age and under died
of injuries, one-third from violence and
two-thirds from unintentional injury (3).
Of all homicide victims in 1997,
37 percent were 24 years of age and
under (4). Homicide remains the second
leading cause of death for young
persons 15–24 years of age and the
leading cause of death for African
Americans in this age group (5).
Data from 1979 through 1993
indicate that the total homicide rate
increased due to increases in
firearm-related homicides (6,7).
Furthermore, for each of the 32,436
persons killed by a gunshot wound in
the United States in 1997,
approximately 2 more people were
treated for nonfatal wounds in hospital
emergency departments (8).
Suicide is the eighth leading cause
of death in the United States. In 1997
suicide was the third leading cause of
death for children 10–14 years of age
and young persons 15–24 years of age
(5). The economic costs of suicide
among young persons 15–24 years of
age has been estimated at over $2.26
billion per year and, when attempted
suicide is considered, this cost increases
to over $3.19 billion (9).
Unfortunately, youth continue to be
involved as both perpetrators and
victims of violence. In 1998, 5,506
young people 15–24 years of age were
victims of homicide, an average of 15
youth homicide victims per day.
Homicide is second only to motor
vehicle crashes as a cause of injury
deaths for persons 15–24 years of age
(10). For each death there are at least
100 nonfatal violence-related injuries.
For adolescents 12 years of age and
older, physical assault victimization
occurred twice as often as in the general
population. Additionally, according to a
report by the U.S. Department of
Justice, assaults were significantly
higher among males. The report also
states that the number of assaults were
higher for those with lower household
incomes (less than $7,500) when
compared to the rates of assault
victimization of households with greater
annual incomes (more than $35,000)
(11).
Intimate partner violence and sexual
assault threaten people of all walks of
life. Even though both females and
males experience these types of
violence, male victimization of females
is more common in intimate partner
violence and sexual assault. Although
most assault victims survive, they suffer
physically and emotionally. In 1995,
almost 5,000 females in the United
States were murdered. In those cases for
which the Federal Bureau of
Investigation had data on the
relationship between the offender and
the victim, 85 percent were killed by
someone they knew. In 1994, more than
500,000 females were seen in hospital
emergency departments for violence-
related injuries, and 37 percent of those
females were there for injuries inflicted
by spouses, ex-spouses, or nonmarital
partners (12).
Child maltreatment continues to be
a major concern. In 1997, an estimated
1,196 fatalities were due to child
maltreatment in the 50 States and the
District of Columbia. In the same year,
the rate of child victims was 13.9 per
1,000 children in the general population.
Based on information from 39 States,
75.4 percent of the perpetrators were the
victim’s parents, 10.2 percent were
relatives, and 1.9 percent were
individuals in other caretaking
relationships (13).
Data Summary
Highlights
While violent and abusive behaviors
continue to be major causes of death,
injury, and stress in the United States,
during the last decade there were many
indications of progress. All three violent
mortality objectives met their year 2000
targets. The age-adjusted homicide rate
for the total population (7.1) decreased
to 7.1 per 100,000 persons in 1998,
surpassing the year 2000 target of 7.2
per 100,000. Additionally, homicide
rates decreased for young adult black
and Hispanic males and all American
Indian/Alaska Natives, and to below the
year 2000 target for black females.
Suicide rates (7.2) in the total
population (age adjusted) fell to 10.4 per
100,000 in 1998, below the year 2000
target of 10.5. Suicide rates also
declined for adolescents 15–19 years of
age, males 20–34 years of age, and
white males 65 years of age and over.
On the other hand, suicide rates for
American Indian/Alaska Native males
increased from 20.1 per 100,000 persons
in 1987 to 21.4 per 100,000 in 1998.
Firearm-related deaths (7.3) declined for
the total population (age adjusted). The
rate in 1998 was 11.3 per 100,000
persons, which was below the year 2000
target of 11.6.
The incidence of rape and attempted
rape for females 12 years of age and
over (7.7) declined substantially, from
332 per 100,000 in 1992 to 160 in 1998,
even though it fell short of its year 2000
target of 108.
There was progress for two
objectives related to adolescent
behaviors, physical fighting (7.9) and
weapon-carrying (7.10) among
adolescents 14–17 years of age. The
incidence of physical fighting and
weapon-carrying among youth is
measured by the number of incidents
per 100 students per month. The number
of incidents of physical fighting
decreased from 137 in 1991 to 106 in
1999, surpassing the year 2000 target of
110. The incidence of weapon-carrying
decreased from 107 incidents in 1991 to
68 in 1999, which surpassed the year
2000 target of 86.
Another objective that surpassed the
year 2000 target was the implementation
in States of child death review systems
for unexplained child deaths (7.13). The
number of States (including the District
of Columbia) increased from 33 in 1991
to 48 in 1996, above the year 2000
target of 45.
Two objectives moved dramatically
away from the year 2000 targets. The
objectives were (7.6) reduce assault
injuries among people 12 years of age
Healthy People 2000 Final Review 137

7.1 Homicide
7.2 Suicide
7.3 Firearm-related deaths
7.8 Suicide attempts among adolescents
7.7 Rape and attempted rape
7.6 Assault injuries
7.9 Physical fighting among adolescents
7.10 Weapon-carrying by adolescents
7.11 Firearms in the home stored loaded and unlocked
7.16 Conflict resolution in a required course
7.18 States meeting prison suicide prevention standards
7.19 States with firearm storage laws
7.15 Battered women turned away from shelters
7.13 States with child death review systems
Moved away from target Moved toward target Met target
NOTE: Complete tracking data are shown in table 7. Progress quotients are not calculated for objectives 7.4, 7.5, 7.12, 7.14, and 7.17.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
-50%
-2%
Percent of target achieved
-100 -80 -60 -40 -20 0 20 40 60 80 100
108%
110%
108%
115%
186%
125%
100%
35%
25%
77%
-167%
-87%
138 Healthy People 2000 Final Review
Figure 7. Final status of Violent and Abusive Behavior objectives
and older and (7.8) reduce injurious
suicide attempts by adolescents.
Disturbingly, injurious suicide attempts
among adolescents 14–17 years of age
increased from 2.1 percent in 1990 to
2.6 percent in 1997.
Summary of Progress
The priority area of Violent and
Abusive Behavior contained a total of
19 objectives. Seven objectives, (7.1,
7.2, 7.3, 7.9, 7.10, 7.13, and 7.16) met
their respective year 2000 targets. Four
(7.5, 7.7, 7.11, and 7.19) progressed
toward the year 2000 targets. Five
objectives (7.4, 7.6, 7.8, 7.15, and 7.18)
moved away from the year 2000 target,
although three of these (7.4, 7.15, and
7.18) had definitional issues making
monitoring difficult (see Data Issues).
Three objectives (7.12, 7.14, and 7.17)
remained without baselines. See table 7
for the tracking data for the objectives
in this priority area and figure 7 for a
quantitative assessment of progress.
Discussion
As the 21st century begins, the
public, private, and government sectors
must work together to prevent violence.
Studies have demonstrated
interrelationships between risk factors
for different types of violence but much
more research is needed. Intervention
evaluations have demonstrated a limited
number of effective programs for
violence prevention. Further, some
interventions appear to be effective but
their ability to reduce morbidity and
mortality needs to be evaluated (14).
Given the diversity of communities and
circumstances, many more types of
prevention programs and interventions
need to be developed and evaluated.
The reasons for decline in many of
the measures of violence-related
objectives during the last decade are not
well understood. The decline
demonstrated in the latter part of the
decade may be due to multiple risk and
protective factors. Factors that may have
influenced the decline are changes in
prevention activities, demographics, the
economy, and the prevalence of
substance abuse.
Measurement of progress within the
area of family and intimate violence
prevention had three objectives (7.5, 7.6,
and 7.7) for which measurements were
affected by the redesign of the National
Crime Victimization Survey in 1992.
The revised questions elicited higher
rates for rape and other sex offenses, as
well as crimes committed by relatives
and acquaintances. The sensitive nature
of this issue makes it difficult to study.
Therefore, knowledge is limited about
the factors that affect the likelihood that
males will behave violently toward
females, the factors that endanger or
protect females from violence, and the
physical and emotional consequences of
such violence for families and their
children.
In the area of child maltreatment,
data systems are needed to identify new
cases and to characterize associated
causes of maltreatment. While some
long-term studies on home-visitation
programs for young mothers have
shown potential for preventing child
abuse and neglect, other existing
interventions and their effectiveness
need to be evaluated.
Developing data to measure
progress is critical for many of the areas
in injury and violence prevention. While
many reliable data sources have
remained constant over time, major
information gaps still exist, especially
for race, ethnicity, and special
populations. Addressing these gaps
poses a major challenge for the next
decade.
Transition to Healthy People
2010
The structure of Healthy People
changed significantly for Priority Area
7—Violent and Abusive Behavior and
Priority Area 9—Unintentional Injuries.
For several reasons, these two areas
were combined into one Healthy People
2010 Focus Area titled ‘‘Injury and
Violence Prevention.’’ One consideration
for merging the two subjects was having
all of the unintentional injury and
violence-related objectives in one place
to facilitate a better focus on the entire
injury area. Another consideration was
that many injuries produce the same
outcome but result from different causal
factors. For example, a nonfatal spinal
cord injury can be caused by an
unintentional event, such as a motor
vehicle crash, or by a violence-related
event, such as a domestic dispute or
attempted robbery. Finally, the same
interventions can reduce injuries for
both unintentional and violence-related
injuries. For instance, efforts to promote
proper storage of firearms in homes can
help reduce the risk of unintentional and
intentionally self-inflicted, or assaultive
firearm-related injuries in the home.
Overall, there are fewer objectives
for the two subject areas. The Healthy
People 2000 had 45 objectives in the
Unintentional Injuries and Violent and
Abusive Behavior chapters compared
with 39 objectives in Healthy People
2010 Injury and Violence Prevention
chapter. For unintentional injuries
Healthy People 2000 had 29 objectives
compared with 19 objectives in the
Healthy People 2010 edition. Violent
and Abusive Behaviors had 19
objectives in Healthy People 2000
compared with 8 objectives in Healthy
People 2010. The Healthy People 2010
chapter has a new section titled ‘‘Injury
Prevention’’ containing 12 objectives
that relate to both subject areas (for
example, nonfatal head injuries and
nonfatal spinal cord injuries, child
fatality review, emergency department
and hospital discharge surveillance
systems).
Additionally, some Healthy People
2000 objectives were eliminated because
they either lacked baseline data, a
national data source, progress, standard
definitions, or were too multifaceted and
could not be tracked. Some objectives
were revised for Healthy People 2010.
For example, objective 7.13
(unexplained child death review
systems) exceeded the year 2000 target
but was revised as a developmental
objective (an objective without current
baseline data) for Healthy People 2010.
The revised objective is more specific
and will measure State-level child
fatality reviews of deaths for children 14
years of age and under. The year 2000
objective was more general and did not
state a specific age group. In general for
Healthy People 2010, there is greater
specificity and also a better sense of the
potential for baseline data and
continuous tracking for the next decade.
Also, for Healthy People 2010, all age-
adjusted rates use a year 2000 standard
population.
Many injuries and injury-related
deaths occur in specific population
groups (such as infants, children, and
the elderly) where the intentionality of
the injury is unknown and requires more
detailed investigation. Examination of
these cases, documentation of the
events, and surveillance efforts can help
communities to better understand the
causes and to develop interventions to
Healthy People 2000 Final Review 139
prevent injuries which are a growing
public health concern for the Nation.
The Healthy People 2010 objective
for homicide, which was retained from
Healthy People 2000 (with a minor
variation), has been designated as a
measure of one of the Leading Health
Indicators (LHI). Homicide data are the
most accurate violent crime data
collected and as such are a reliable
indicator of violent crime in general.
The LHIs are discussed in further detail
in the Introduction.
Two objectives were transferred to
Focus Area 18—Mental Health and
Mental Disorders: Suicides, and
adolescent suicide attempts.
Healthy People 2010 contains 19
other related objectives within 6 other
focus areas. Those related areas are:
Focus Area 1, Access to Quality Health
Services, with three related objectives;
Focus Area 7, Educational and
Community-Based Programs, with one
objective; Focus Area 8, Environmental
Health, with three objectives; Focus
Area 18, Mental Health and Mental
Disorders, with two related objectives;
Focus Area 20, Occupational Safety and
Health, with four objectives; and Focus
Area 26, Substance Abuse, with six
related objectives.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the changes between the
two decades of objectives, reflecting
new knowledge and direction for this
area.
Data Issues
Definitions
Objective 7.1 (homicide) is
monitored using data from the National
Vital Statistics System (NVSS) and
excludes homicides attributed to legal
intervention. It should be noted that the
number of States reporting Hispanic
origin data in their vital statistics has
varied from year to year (see Appendix).
The data on spousal homicide (7.1b) are
from the Federal Bureau of
Investigation; the 1993 and 1994 data
are for spouses 15 years of age and over
and are not directly comparable to
previous data.
Objective 7.2 (suicide) is monitored
using data from the NVSS. The data are
compiled from death certificates
submitted by the States. Differentiating
suicide deaths from accidental deaths
relies heavily on judgment by the
medical legal officer (for example,
coroner or medical examiner). A key
element of this determination is the
establishment of intent by the deceased.
This determination may be based on
information about prior suicide attempts,
a statement or note by the deceased
indicating their intent to commit suicide,
or other clinical information (for
example, serious mental illness) (15).
Update data for objective 7.4 were
collected using a different
definition—the endangerment
standard—than the baseline, so the data
points are not directly comparable.
The baseline and target for
objective 7.5 (partner abuse) were
established using the National Institute
of Mental Health’s survey of family
violence, which measured incidents of
violence among couples. This survey
will not be repeated, so the objective is
monitored using data from the Bureau
of Justice Statistics’ National Crime
Victimization Survey that is tracking
violence between intimates (for
example, spouses, ex-spouses, or
boyfriends). The data used to track the
objective report incidents per 1,000
women, which reflects the intent of the
objective.
Data for objectives 7.6 (assault
injuries) and 7.7 (rape and attempted
rape) come from the National Crime
Victimization Survey, which provides
self-reported victimizations. The
numbers of offenses reported in this
survey generally exceed those reported
to police and other law enforcement
agencies. However, because of their
personal nature, some offenses such as
rape are underreported in the crime
survey (16). The data for objective 7.6
include injuries from completed rapes,
attempted and completed robberies with
injury, and completed aggravated and
simple assaults with injury. In 1992, the
Survey was redesigned; the revised
questions elicit higher rates for rape,
other sex offenses, and crimes
committed by relatives and
acquaintances. The baseline for
objective 7.7 has been revised using the
1992 data to reflect this measurement
change.
Data for objectives 7.8 (adolescent
suicide attempts), 7.9 (physical fighting
among adolescents), and 7.10 (weapon
carrying) are measured using the
school-based Youth Risk Behavior
Survey (YRBS) and rely on student
self-report. Self-reported suicide
attempts are limited to those that
occurred in the last 12 months and
required medical attention. Data from
the 1992 National Health Interview
Survey (NHIS) youth supplement
indicate higher levels of
weapon-carrying and fighting among
youth (14–19 years of age) not in school
than among youth the same age in
school, although the estimates for
in-school youth were very close to the
estimates for the total population (17).
The NHIS supplement did not include
questions on suicide attempts. Because
YRBS data are used for ongoing
monitoring of objective 7.8, the
exclusion of adolescents not in school
may underestimate youth suicide
attempts. The reliance on self-report
without external validation of
weapon-carrying, suicide attempts, and
fighting may affect the validity of these
estimates, although a study by the
Centers for Disease Control and
Prevention indicated that the results are
highly reliable (18).
Objective 7.11 (inappropriate
firearm storage) is measured using data
from the NHIS. The numerator is the
number of people who have a firearm in
or around the house that is stored loaded
and unlocked. The denominator is the
number of people who report having a
firearm in or around the house. Data on
the proportion of the total population
having a firearm in or around the house
are also footnoted in the summary table.
Objective 7.19 (handgun storage
laws) relates to State laws that vary
across States in populations targeted,
penalties, and liability.
Data Comparability
The update for objective 7.15
(shelter availability for battered women)
comes from the same source (National
Coalition Against Domestic Violence) as
the baseline, but was collected
differently and is not directly
comparable.
A reliable source of data for
objective 7.18 (suicide prevention in
jails) was not developed. The wording
and baseline data for the objective were
established with States as the
organizational level for monitoring and
implementing suicide prevention
protocols in jails. Jails are usually under
the jurisdiction of counties or
municipalities. State-level data on jails
are limited; the alternative data track the
objective using jails as the unit of
analysis. Data from the National Census
of Jails, conducted by the Bureau of
Justice Statistics, were only available for
140 Healthy People 2000 Final Review
1993. Additional data are from the
American Correctional Association’s
(ACA) list of jails that are
ACA-accredited; their accreditation
requires that suicide prevention policies
and training be implemented in the jail.
However, not all jails seek ACA
accreditation; this selection bias suggests
that these data may not be nationally
representative.
Data Availability
Data are not currently available for
objectives 7.12 (emergency room
protocols), 7.14 (followup on abused
children), and 7.17 (comprehensive
violence prevention programs). No
update was obtained for objective 7.16
(conflict resolution), which met its target
at baseline.
References
1. Moscicki EK, O’Carroll PW, Rae DS, et al.
Suicide ideation and attempts: The Epidemiologic
Catchment Area Study. In: Report of the
Secretary’s Task Force on Youth Suicide. Vol 4.
Washington: Department of Health and Human
Services. 1989.
2. Centers for Disease Control and Prevention.
Rates of homicide, suicide, and firearm-related
death among children—26 industrialized countries,
1950–93. MMWR 46(5):101. 1995.
3. National Center for Injury Prevention and
Control. U.S. Injury Mortality Statistics. Centers
for Disease Control and Prevention. 1997.
http:\\www.cdc.gov/ncipc/osp/usmort.htm.
4. National Center for Health Statistics. National
vital statistics system, general mortality table,
numbers of deaths. 1997. Hyattsville, Maryland:
Public Health Service. 2000.
5. Hoyert DL, Kochanek KD, Murphy SL. Deaths:
Final Data for 1997. National vital statistics
reports; vol 47 no 19. Hyattsville, Maryland:
National Center for Health Statistics. 1999.
6. Fingerhut LA, Ingram DD, Feldman JJ. Firearm
and nonfirearm homicide among persons 15–19
years of age: Differences by level of urbanization.
United States 1979–89. JAMA 267(22): 3048–53.
1992.
7. Fingerhut LA, Ingram DD, Feldman JJ.
Homicide rates among U.S. teenagers and young
adults: Differences by mechanism, level of
urbanization, race, and sex, 1987 through 1995.
JAMA 280(5): 423–7. 1998.
8. Centers for Disease Control and Prevention.
Nonfatal and fatal firearm-related injuries—United
States, 1993–1997. MMWR 48(45): 1029–34.
1999.
9. Weinstein MC, Saturno, PJ. Economic impact
of youth suicides and suicide attempts. In: Report
of the Secretary’s task force on youth suicide. Vol
4. Washington: Department of Health and Human
Services. 1989.
10. National Center for Health Statistics. Health,
United States, 2000 with Adolescent Health
Chartbook. Hyattsville, Maryland: 2000.
11. Bureau of Justice Statistics. Criminal
Victimization 1998: Changes 1997–98 with trends,
1993–98. Washington: Department of Justice.
1999.
12. Bureau of Justice Statistics. Violence-related
injuries treated in hospital emergency departments.
Washington: Department of Justice. 1997.
13. Department of Health and Human Services,
Administration on Children, Youth, and Families.
Child maltreatment 1997: Reports from the states
to the national child abuse and neglect data
system. Washington: U.S. Government Printing
Office. 1999.
14. National Center for Injury Prevention and
Control. Best practices for preventing violence by
children and adolescents: A source book. Atlanta,
Georgia: National Center for Injury Prevention and
Control. 1999.
15. Rosenberg M, et al. Operational criteria for the
determination of suicide. J Forensic Sci
33:1445–6. 1988.
16. Department of Justice, Bureau of Justice
Statistics. The crime of rape. Washington. 1985.
17. Centers for Disease Control and Prevention.
Health risk behaviors among adolescents who do
and do not attend school: United States, 1992.
MMWR 43:129–32. 1994.
18. Brener N, et al. Reliability of the Youth Risk
Behavior Survey questionnaire. Paper presented at
the annual meeting of the American Public Health
Association. Oct 1994.
Healthy People 2000 Final Review 141

Table 7. Violent and Abusive Behavior objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
7.1 Homicide (age adjusted per 100,000).............. 1987 8.5 10.1 10.8 10.3 10.6 10.1 9.2 8.3 7.8 7.1 - - - 7.2
a. Children 3 years and under (per 100,000)........ 1987 3.9 4.4 4.9 4.5 4.9 4.6 4.5 4.5 4.3 4.4 - - - 3.1
b. Spouses 15-34 years (per 100,000) ............ 1987 1.7
1
1.5
2
1.1
2
1.1
2
1.1
2
1.0 --- --- --- --- --- 1.4
c. Black male 15-34 years (per 100,000) .......... 1987 91.1 130.5 140.8 134.2 140.5 133.8 114.6 105.7 97.6 84.9 - - - 72.4
d. Hispanic male 15-34 years (per 100,000)
3
....... 1987 41.3 53.2 55.7 56.8 52.4 52.2 49.7 39.2 34.9 33.5 - - - 33.0
e. Black female 15-34 years (per 100,000) ......... 1987 20.2 22.1 24.1 22.7 23.7 21.0 18.5 16.1 14.4 13.3 - - - 16.0
f. American Indian/Alaska Native (age adjusted
per 100,000) ............................. 1987 11.2 10.7 12.2 10.3 10.7 11.8 11.5 9.9 10.8 9.5 - - - 9.0
7.2* Suicide (age adjusted per 100,000) ............... 1987 11.7 11.5 11.4 11.1 11.3 11.2 11.2 10.8 10.6 10.4 - - - 10.5
a. Adolescents 15-19 years (per 100,000) ......... 1987 10.2 11.1 11.0 10.8 10.9 11.1 10.5 9.7 9.5 8.9 - - - 8.2
b. Male 20-34 years (per 100,000) ............... 1987 25.2 25.1 25.1 24.5 25.5 26.5 26.3 24.2 23.4 22.9 - - - 21.4
c. White male 65 years and over (per 100,000)...... 1987 46.7 44.4 42.7 41.0 40.9 38.9 38.7 37.8 36.1 38.2 - - - 39.2
d. American Indian/Alaska Native male (age
adjusted per 100,000) ...................... 1987 20.1 21.0 19.2 17.9 18.7 23.8 20.1 20.0 21.3 21.4 - - - 17.0
7.3 Firearm-related deaths (age adjusted per 100,000) . . . 1990 14.6 . . . 15.2 14.9 15.6 15.1 13.9 12.9 12.2 11.3 - - - 11.6
a. Black ................................... 1990 33.4 . . . 35.4 34.4 37.6 35.5 30.3 28.5 25.7 22.7 - - - 30.0
7.4 Child abuse and neglect (per 1,000).............. 1986 22.6 - - - - - - - - - 41.9 - - - - - - - - - - - - - - - - - - Less
than 2
Incidence of types of maltreatment
a. Physical abuse ............................ 1986 4.9 - - - - - - - - -
4
9.1 - - - - - - - - - - - - - - - - - - Less
than 4
b. Sexual abuse ............................. 1986 2.1 - - - - - - - - -
4
4.5 - - - - - - - - - - - - - - - - - - Less
than 2
c. Emotional abuse........................... 1986 3.0 - - - - - - - - -
4
7.9 - - - - - - - - - - - - - - - - - - Less
than 3
d. Neglect.................................. 1986 14.6 - - - - - - - - -
4,5
29.2 - - - - - - - - - - - - - - - - - - Less
than 1
7.5 Partner abuse (per 1,000 couples)................ 1985 30.0 - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 27.0
Assaults by intimates (per 1,000 females 12 years
and over)
6
.................................. ... --- --- --- 8.8 9.8 9.1 8.6 7.5 --- 7.2 --- ...
7.6 Assault injuries (per 1,000 people 12 years
and over)
6
.................................. 1986 9.7 10.3 11.0 9.3 12.3 12.7 11.7 10.5 - - - 10.2 - - - 8.7
7.7 Rape and attempted rape (per 100,000)
6
Female 12 years and over ...................... 1992
§
332 . . . . . . . . . 267 273 193 144 156 160 - - - 108
a. Female 12-34 years ........................ 1992
§
607 . . . . . . . . . 477 527 374 249 349 - - - - - - 225
Sexual assaults (per 100,000 females 12 years
and over) .................................. 1992 184 . . . . . . . . . 138 96 84 84 102 110 - - - . . .
7.8* Suicide attempts among adolescents 14–17 years . . 1990 2.1% . . . 1.7% - - - 2.7% - - - 2.8% - - - 2.6% - - - 2.6% 1.8%
a. Female 14-17 years ........................ 1991 2.5% . . . . . . - - - 3.8% - - - 3.4% - - - 3.3% - - - 3.1% 2.0%
7.9 Physical fighting among adolescents 14–17 years
(incidents per 100 students per month) ............ 1991 137 . . . . . . - - - 137 - - - 128 - - - 115 - - - 106 110
a. Non-Hispanic black male 14-17 years .......... 1991 207 . . . . . . - - - 203 - - - 181 - - - 175 - - - 159 160
See footnotes and key at end of table.
142 Healthy People 2000 Final Review

Table 7. Violent and Abusive Behavior objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
7.10 Weapon-carrying by adolescents 14–17 years
(incidents per 100 students per month) ............ 1991 107 . . . . . . - - - 92 - - - 81 - - - 74 - - - 68 86
a. Non-Hispanic black adolescents 14–17 .......... 1991 134 . . . . . . - - - 117 - - - 85 - - - 84 - - - 71 105
7.11 Proportion of people 18 years and over with
firearms in the home that are stored loaded and
unlocked .................................. 1994
7
20% ... ... ... ... ... --- --- ---
7
19% - - - 16%
7.12 Emergency room protocols for victims of
violence ................................... ... --- --- --- --- --- --- --- --- --- --- --- 90%
7.13 Number of States with child death review
systems ................................... 1991 33 . . . . . . 32 - - - 40 - - -
8
48 --- --- --- 45
7.14 Number of States that followup abused children ... ... --- --- --- --- --- --- --- --- --- --- --- 30
7.15 Battered women turned away from shelters ....... 1987 40% - - - - - - - - - - - - - - - - - - 66% - - - - - - - - - 10%
7.16 Conflict resolution in a required course
Proportion of middle/junior and senior high schools .... 1994 58.3% . . . . . . . . . . . . . . . - - - - - - - - - - - - - - - 50%
7.17 Local comprehensive violence prevention
programs.................................. ... --- --- --- --- --- --- --- --- --- --- --- 80%
7.18* Suicide prevention in jails
Number of States meeting nationally recognized suicide
prevention standards.......................... 1992
§
2 ... ... ... --- --- 2 1 --- --- --- 50
Proportion of jails with suicide policies.............. ... --- --- --- --- 79.5% - - - - - - - - - - - - - - - - - - . . .
Proportion of jails with ACA accreditation............ ... --- 1% 1% 1% 2% 2% 2% --- --- --- 3% ...
7.19* Number of States with firearm storage laws ....... 1989
§
1 1 5 8 11 13 15 15 15 15 18 50
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
1
1989 data.
2
Includes married men and women aged 15 and older. Data include deaths from legal intervention (E970–E978) in addition to E960–E969 and are not compara ble to other data for this
objective.
3
Excludes data from States lacking Hispanic-origin item on their death certificates or for which Hispanic-origin data were not of sufficient quality. See appendix.
4
Rates were computed using the Endangerment Standard.
5
Rate includes both physically and emotionally neglected children.
6
The victimization survey was redesigned in 1992. The revised questions elicit higher rates for rape, other sex offenses, and crimes committed by relatives and acquaintances.
7
In 1994, 37.4 percent reported having a firearm in or around the home; in 1998, this number decreased to 35 percent.
8
Number includes State teams in 38 States and the District of Columbia, and 9 additional States where county/local teams serve the majority of the population at risk.
NOTE: Data include revisions and, therefore, may differ from data previously published in these reports and other publications.
Met
Toward
Mixed/ no change
Away
Cannot assess
Healthy People 2000 Final Review 143

Objective number Data source
7.1, 7.1a-f National Vital Statistics System, CDC, NCHS.
7.1b Numerator obtained from DOJ, FBI.
7.2*, 7.2a-d National Vital Statistics System, CDC, NCHS.
7.3 National Vital Statistics System, CDC, NCHS.
7.4, 7.4a-d National Incidence of Child Abuse and Neglect Survey, Administration for Children and Families, NCCAN.
7.5 Baseline: National Family Violence Survey, NIH, NIMH.
1992–96 and 1998 data: National Crime Victimization Survey, DOJ, BJS.
7.6 National Crime Victimization Survey, DOJ, BJS.
7.7, 7.7a National Crime Victimization Survey, DOJ, BJS.
7.8,7.8a* Youth Risk Behavior Survey, CDC, NCCDPHP.
7.9 Youth Risk Behavior Survey, CDC, NCCDPHP.
7.10 Youth Risk Behavior Survey, CDC, NCCDPHP.
7.11 National Health Interview Survey, CDC, NCHS.
7.13 Baseline: Annual 50-State Survey, National Committee for Prevention of Child Abuse.
Updates: National Incidence of Child Abuse and Neglect Survey, Administration for Children and Families, NCCAN.
7.15 Domestic Violence Statistical Survey, National Coalition Against Domestic Violence.
7.16 School Health Policies and Programs Study, CDC, NCCDPHP.
7.18* Baseline and updates (States): National Study of Jails, National Center on Institutions and Alternatives.
Baseline (suicide policies): National Census of Jails, DOJ, BJS.
Baseline and update (ACA accreditation): American Correctional Association.
7.19* Office of Planning, Evaluation and Legislation, CDC, NCIPC; National Conference of State Legislatures.
* Duplicate objective. See full text of objective following this table.
144 Healthy People 2000 Final Review
Violent and Abusive
Behavior Objectives
7.1: Reduce homicides to no more than
7.2 per 100,000 people.
7.1a: Reduce homicides among
children aged 3 and younger to no
more than 3.1 per 100,000 children.
7.1b: Reduce homicides among
spouses aged 15–34 to no more
than 1.4 per 100,000.
7.1c: Reduce homicides among
black men aged 15–34 to no more
than 72.4 per 100,000.
7.1d: Reduce homicides among
Hispanic men aged 15–34 to no
more than 33.0 per 100,000.
7.1e: Reduce homicides among
black women aged 15–34 to no
more than 16.0 per 100,000.
7.1f: Reduce homicides among
American Indians and Alaska
Natives to no more than 9.0 per
100,000.
7.2*: Reduce suicides to no more than
10.5 per 100,000 people.
Duplicate objective: 6.1
7.2a*: Reduce suicides among
youth aged 15–19 to no more than
8.2 per 100,000.
Duplicate objective: 6.1a
7.2b*: Reduce suicides among men
aged 20–34 to no more than 21.4
per 100,000.
Duplicate objective: 6.1b
7.2c*: Reduce suicides among white
men aged 65 and older to no more
than 39.2 per 100,000.
Duplicate objective: 6.1c
7.2d*: Reduce suicides among
American Indian and Alaska Native
men to no more than 17.0 per
100,000.
Duplicate objective: 6.1d
7.3: Reduce firearm-related deaths to no
more than 11.6 per 100,000 people from
major causes.
7.3a: Reduce firearm-related deaths
among blacks to no more than 30.0
per 100,000 people from major
causes.
7.4: Reverse to less than 22.6 per 1,000
children the rising incidence of
maltreatment of children younger than
age 18.
7.4a: Reverse to less than 4.9 per
1,000 children the rising incidence
of maltreatment of children younger
than age 18.
7.4b: Reverse to less than 2.1 per
1,000 children the rising incidence
of sexual abuse of children younger
than age 18.
7.4c: Reverse to less than 3.0 per
1,000 children the rising incidence
of emotional abuse of children
younger than age 18.
7.4d: Reverse to less than 14.6 per
1,000 children the rising incidence
of neglect of children younger than
age 18.
7.5: Reduce physical abuse directed at
women by male partners to no more
than 27 per 1,000 couples.
7.6: Reduce assault injuries among
people aged 12 and older to no more
than 8.7 per 1,000.
7.7: Reduce rape and attempted rape of
women aged 12 and older to no more
than 108 per 100,000 women.
7.7a: Reduce rape and attempted
rape of women aged 12–34 to no
more than 225 per 100,000.
7.8*: Reduce to 1.8 percent the
incidence of injurious suicide attempts
among adolescents aged 14–17.
Duplicate objective: 6.2
7.8a*: Reduce to 2.0 percent the
incidence of injurious suicide
attempts among female adolescents
aged 14–17.
Duplicate objective: 6.2a
7.9: Reduce to 110 per 100 the incidents
of physical fighting among adolescents
aged 14–17.
7.9a: Reduce to 160 per 100 the
incidents of physical fighting among
black males aged 14–17.
7.10: Reduce to 86 per 100 the incidents
of weapon-carrying by adolescents aged
14–17.
7.10a: Reduce to 105 per 100 the
incidents of weapon-carrying by
blacks aged 14–17.
7.11: Reduce by 20 percent the
proportion of people who possess
weapons that are inappropriately stored
and therefore dangerously available.
7.12: Extend protocols for routinely
identifying, treating, and properly
referring suicide attempters, victims of
sexual assault, and victims of spouse,
elder, and child abuse to at least
90 percent of hospital emergency
departments.
7.13: Extend to at least 45 States
implementation of unexplained child
death review systems.
7.14: Increase to at least 30 the number
of States in which at least 50 percent of
children identified as neglected or
physically or sexually abused receive
physical and mental evaluation with
appropriate followup as a means of
breaking the intergenerational cycle of
abuse.
7.15: Reduce to less than 10 percent the
proportion of battered women and their
children turned away from emergency
housing due to lack of space.
7.16: Increase to at least 50 percent the
proportion of elementary and secondary
schools that teach nonviolent conflict
resolution skills, preferably as a part of
comprehensive school health education.
7.17: Extend coordinated,
comprehensive violence prevention
programs to at least 80 percent of local
jurisdictions with populations over
100,000.
7.18*: Increase to 50 the number of
States with officially established
protocols that engage mental health,
alcohol and drug, and public health
authorities with corrections authorities to
facilitate identification and appropriate
intervention to prevent suicide by jail
inmates.
Duplicate objective: 6.10
7.19*: Enact in 50 States and the
District of Columbia laws requiring that
firearms be properly stored to minimize
access and the likelihood of discharge
by minors.
Duplicate objective: 9.25
*Duplicate objective.
Healthy People 2000 Final Review 145

Priority Area 8
Educational and
Community-Based
Programs
Background
Educational and community-based
programs are instrumental in creating an
environment that is conducive to leading
a healthy lifestyle. These programs,
designed to reach people outside of the
traditional health care setting, are
fundamental to promoting health and
increasing the quality of life by
producing a supportive social and
physical environment capable of
reinforcing positive behaviors and
changing negative behaviors that
contribute to many of today’s leading
health threats.
Health and quality of life rely on
much more than a well-functioning
health and medical care system; they
depend on the community to encourage
and provide leadership, collaboration,
and initiatives that are fundamental to
progress. To successfully address health
and quality of life issues, communities
must draw on many components
including public health, health care,
civic, voluntary health, and faith
organizations; local governments;
schools; park and recreation
departments; and other interested groups
as well as private citizens. Working
within existing systems or networks,
such as schools or park departments,
community efforts can yield results that
can improve the health and lives of a
large segment of the population. While
some community-based programs
address a single risk factor or prominent
health problem, others are taking a more
comprehensive, holistic approach to
health and healthy communities.
Data Summary
Highlights
The average number of years of
healthy life for the total population (8.1)
increased from a baseline of 64 years in
1990 to 65.2 years in 1998, exceeding
the Healthy People 2000 target. Among
special populations targeted by the
objective, Hispanics met the year 2000
target, while progress was made for
blacks and older adults.
High school completion rates (8.2)
remained relatively stable compared
with their 1992 baselines. Worksites
with 50 or more employees that
provided employee health promotion
programs (8.6) saw significant growth
and exceeded the year 2000 target,
while the participation of blue-collar
employees 18 years of age and over in
these programs (8.7) declined from the
1994 baseline, yet still met their target.
The proportion of hospitals offering
community health promotion programs
(8.12) also met its target. Family
discussion of health issues with children
10 years of age and over (8.9) moved
away from its target.
Summary of Progress
Of the 14 Educational and
Community-Based Programs objectives,
4 met (8.1, 8.6, 8.7, and 8.13) and 2
progressed (8.12 and 8.14) toward the
year 2000 targets. One objective (8.8)
showed no change from baseline while
two objectives (8.3 and 8.9) showed
mixed progress. Two objectives (8.2 and
8.3) moved away from the year 2000
targets. Five objectives could not be
assessed because they either lacked data
beyond the baseline data (8.4, 8.5, 8.8,
and 8.11) or because they had proxy
data but lacked baseline data (8.10). See
table 8 for the tracking data for the
objectives in this priority area and
figure 8 for a quantitative assessment of
progress.
Discussion
In the past decade, the Nation has
seen progress in a number of areas—but
greater achievements are needed in the
areas of educational and
community-based programs. To make
further headway in this area, health
promotion programs need to be sensitive
to the diverse cultural norms and beliefs
of the people for whom the programs
are intended. Achieving such sensitivity
is a continuing challenge as the Nation’s
population becomes increasingly diverse.
To ensure that interventions are
culturally appropriate, linguistically
competent, and appropriate for the needs
of all groups (racial, ethnic, gender,
sexual orientation, disability status, and
age) within the community, members of
the populations served, and gatekeepers
must be involved in the community
assessment and planning process (1).
Community assessment helps to
identify the cultural traditions and
beliefs of the community and the
education, literacy level, and language
preferences necessary for the
development of appropriate materials
and programs. In addition, a community
assessment can help identify levels of
social capital and community capacity.
Such assessments help identify the
skills, resources, and abilities needed to
manage health improvement programs in
communities (2,3).
Educational and community-based
programs must be supported by
accurate, appropriate, and accessible
information derived from a science base.
Increasing evidence supports the
efficacy and effectiveness of health
education and health promotion in
schools, worksites, health care facilities,
and community-based programming (4).
Gaps in research include the
dissemination and diffusion of effective
programs, new technologies, policies,
relationships between settings, and
approaches to disadvantaged and special
populations (5).
Communities need to be involved as
partners in conducting research ensuring
that the content of the prevention efforts
developed is tailored to meet the needs
of the communities and populations
being served. Communities also need to
be involved as equal partners in research
to enhance the appropriateness and
sustainability of science-based
interventions and prevention programs
and ensure that the lessons of research
are transferred back to the community.
Sustainability is necessary for successful
research to be translated into programs
of lasting benefit to communities.
The impact of social ecology on
behavior and the successes of
environmental and policy approaches to
health promotion and disease prevention
need further documentation. Techniques
to evaluate community processes and
community health improvement methods
and models need to be refined and
disseminated so that other communities
can learn from and duplicate successful
strategies. Issues of partnering and the
role of collaborative efforts to increase
the capacity of individuals and
communities to achieve long-term
outcomes and improvements in health
status are not fully understood (2) and
should be evaluated. Mechanisms need
to be developed to share what is learned
in an appropriate and timely manner
with communities.
146 Healthy People 2000 Final Review

8.1 Years of healthy life
8.2 Completion of high school
8.3 Preschool child development programs:
Eligible children offered Head Start
8.8 Health promotion programs for older adults
8.7 Participation in worksite health promotion
activities, hourly workers 18 years and over
8.6 Worksite health promotion activities:
50 or more employees
8.9 Family discussion of health issues
Discussion on HIV/AIDS (grades 9-12)
8.12 Hospital-based patient education: Registered hospitals
Immunizations
Prenatal care
Primary care
8.14 Effective public health systems, health assurance:
Health education
Hospital-based community health promotion:
Community hospitals
Moved away from target Moved toward target Met target
NOTE: Complete tracking data are shown in table 8. Progress quotients are not calculated for objectives 8.4, 8.5, 8.10, 8.11, and 8.13.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
-25%
Percent of target achieved
-100 -80 -60 -40 -20 0 20 40 60 80 100
125%
120%
175%
100%
100%
100%
12%
16%
2%
No change
43%
55%
63%
Healthy People 2000 Final Review 147
Figure 8. Final status of Educational and Community-Based Programs objectives

Transition to Healthy People
2010
The Healthy People 2010 focus area
on Educational and Community-Based
Programs has been reorganized to
emphasize four major settings—schools
(including colleges and universities),
worksites, health care sites, and the
community; and three major intervention
strategies, educational, policy, and
environmental. The four settings serve
as channels to reach desired audiences
as well as apply strategies in as broad a
population as possible. Among the more
effective community health promotion
programs are those that implement
comprehensive intervention plans with
multiple intervention strategies, such as
educational, policy, and environmental
within these various settings. These
settings also provide major social
structures for intervening at the policy
level to facilitate healthful choices (3).
Thus in Healthy People 2010, the
objectives are organized by setting, with
objectives addressing special populations
included in the community setting. The
objectives also reflect a mix of
educational, policy, and environmental
strategies. In the school setting, the
objective addressing high school
completion was retained as was the
objective addressing school health
education, but because of measurement
difficulties, health behaviors that
account for the majority of morbidity
and mortality were emphasized rather
than a comprehensive school health
education. Health risk behavior
information for colleges and universities
was also retained. A new objective
regarding the school nurse to student
ratio was added to address the rapidly
increasing number of children who need
health care during the school day.
Under the worksite setting,
objectives addressing the provision of
worksite health promotion and employee
participation in worksite health
promotion activities were both retained.
Under the health care setting, the
objective on hospital-based patient
education and community health
promotion was expanded to three
separate objectives, with some
modifications. The three objectives
address the provision of patient and
family education, satisfaction with
patient and family education received,
and finally, on health care organizations
sponsorship of community health
promotion activities. The focus on
health care organizations expands the
previous focus on hospitals. Health care
organizations are defined to include
hospitals, managed care organizations,
home health organizations, long-term
care facilities, and community-based
health care organizations.
Under the community setting, the
objective measuring community health
promotion programs addressing Healthy
People focus areas was retained
targeting multiple healthy focus areas
instead of a specific number. The
objective addressing culturally
appropriate and linguistically competent
community health promotion was
modified to reflect the new Healthy
People focus areas. The objective
addressing older adult participation in
health promotion activities was also
retained.
Objectives addressing years of
healthy life, preschool child
development programs, family
discussion of health issues, television
partnerships with community
organizations for health promotion, and
effective public health systems were
dropped. A modified version of the
latter objective was retained in the new
Healthy People focus area called Public
Health Infrastructure. Appendix table III,
a crosswalk between Healthy People
2000 and Healthy People 2010
objectives, summarizes the differences
between the two decades of objectives,
reflecting new knowledge and directions
in this area.
Data Issues
Years of Healthy Life
Years of healthy life is a summary
measure of health that combines
mortality (quantity of life) and
morbidity and disability (quality of life)
into a single measure. The concept of
increasing the span of healthy life is one
of the three Healthy People 2000 goals
and a specific measure has been
developed to track this objective in three
priority areas (8.1, 17.1, and 21.1). The
data used to track the objective come
from the National Vital Statistics System
(mortality) and the National Health
Interview Survey (NHIS) (morbidity).
The NHIS was redesigned in 1997 and,
therefore, data for 1997–98 may not be
comparable with those from previous
years. See the Appendix for a discussion
of the changes to the NHIS. The
methodology for the Healthy People
2000 years of healthy life measure,
developed by NCHS and outside
consultants, is published in Healthy
People Statistical Notes series (6).
Definitions
Objective 8.4 does not include a
definition of comprehensive school
health education. However, the Centers
for Disease Control and Prevention
(CDC) uses an operational definition
that includes eight elements (2). Data
for the variables from the 1994 School
Health Policies and Programs Study
(SHPPS) used to measure these
elements are shown in table 8. Schools
must have addressed all elements of the
operational definition to meet the
criteria for comprehensive school health
education. In 1994, only 2.3 percent of
schools included all eight elements.
Objective 8.7 asks for the
proportion of hourly workers who
participated regularly in
employer-sponsored health promotion
activities. The 1994 baseline indicates
the number of people who participated
in employer-sponsored health promotion
programs in the past year in the
following occupational categories:
Precision production, craft, and
repair occupations
Operators, fabricators, and laborers
Transportation and material moving
occupations
Handlers, equipment cleaners,
helpers, and laborers
Family discussions of health issues
(8.9) are defined as discussions in the
past month among family members 10
years of age and over about the
following topics: nutrition, exercise,
safety, tobacco use, sexual
behavior/sexually transmitted diseases,
or illegal drugs. In 1994, 83 percent of
people had discussed at least one of
these topics with family members in the
month prior to interview. This exceeds
the year 2000 target of 75 percent.
Data Sources
The data used to track objectives
8.2 (completion of high school) and 8.3
(preschool child development programs)
come from the National Center for
Education Statistics (NCES) (7). Both
objectives 8.2 and 8.3 are consistent
with the National Education Goals. The
data for objective 8.2 include those who
received high school diplomas as well
as those who received alternative
credentials, such as a General Education
148 Healthy People 2000 Final Review
Development (GED) certificate. Data for
1992 and 1993 are for persons 19–20
years of age. Beginning with data for
1994, figures for high school completion
are available only for people 18–24
years of age.
Data for objective 8.6 (worksite
health promotion programs) are from the
National Survey of Worksite Health
Promotion Activities, which were
telephone surveys of nongovernment
worksites. Some of the businesses
surveyed had multiple worksites with
different health promotion activities.
Additionally, both active (for example,
classes) and passive (for example,
brochures) methods were counted as
worksite health promotion activities.
Proxy Data and Data Availability
Proxy data for 1992–93 from the
National Association of City and County
Health Officials are shown for objective
8.10. These data show the percent of the
43 reporting States in which at least
90 percent of local health departments
reported providing services that
addressed three or more Healthy People
2000 priority areas. The data represent
the local health departments’ report of
whether a program or service existed.
The survey did not determine whether
the program or service was a health
promotion effort that involved citizen
participation, included community
assessment, or had measurable
objectives. Information on the
proportion of the State population
reached by the services or programs was
not available.
Objective 8.11 is measured using
proxy data from the local health
departments obtained through the
National Profile of Local Health
Departments, NACCHO. The objective
was developed to track the proportion of
counties that have established culturally
and linguistically appropriate community
health promotion programs for racial
and ethnic minority populations.
Local health department refers to
any local component of the public
health system, defined as an
administrative and service unit of local
or State government concerned with
health and carrying some responsibility
for the health of a jurisdiction smaller
than a State. Baseline data for objective
8.11 represent the proportion of local
health departments who indicated they
provided programs or interventions in
the past year that were adapted to meet
the special language and cultural needs
of any racial/ethnic minority population
served. Local health departments
included are those in which a racial or
ethnic minority group constitutes more
than 10 percent of the population. Data
shown are the proportion of local health
departments providing culturally and
linguistically appropriate programs by
Healthy People 2000 priority area.
The Media Health Partnerships
Survey was developed by CDC to
measure partnerships between network
television affiliates and community
health organizations (objective 8.13).
The survey, conducted September
1995–January 1996, determined that all
television network affiliates in the top
20 media markets devote a substantial
effort to health promotion and disease
prevention through partnerships with
community groups, organizations, and/or
agencies. Based on these findings,
objective 8.13 has been achieved and
the survey will not be repeated.
Objective 8.14, which focuses on
the proportion of people served by local
health departments, is being monitored
by the proportion of health departments
carrying out the core functions of public
health. The core functions of public
health have been defined as assessment,
policy development, and assurance.
Local health department refers to any
local component of the public health
system, defined as an administrative and
service unit of local or State government
concerned with health and carrying
some responsibility for the health of a
jurisdiction smaller than a State.
References
1. Department of Health and Human Services.
Healthy people 2010. 2d ed. Washington: U.S.
Government Printing Office. 2000.
2. Steckler A, Allegrante JP, Altman D, et al.
Health education intervention strategies:
Recommendations for future research. Health Educ
Q 22:307–28. 1995.
3. Kreuter MW, Lezin NA, Young LA.
Community-based collaborative mechanisms:
Implications for practitioners. Health Promotion
Practice 1(1):2000.
4. King AC, Jeffrey RW, Fridinger F, et al.
Environmental and policy approaches to
cardiovascular disease prevention through physical
activity: Issues and opportunities. Health Educ Q
22:499–511. 1995.
5. Mullen PD, Evans D, Forster J, et al. Settings
as an important dimension in health
education/promotion policy, programs, and
research. Health Educ Q 22:329–45. 1995.
6. Erickson P, Wilson R, Shannon I. Years of
Healthy Life. Healthy people statistical notes no 7.
Hyattsville, Maryland: National Center for Health
Statistics. 1995.
7. National Education Goals Panel. The national
education goals report: Building a nation of
learners. Washington: U.S. Government Printing
Office. 1996.
Healthy People 2000 Final Review 149

Table 8. Educational and Community-Based Programs objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
8.1* Years of healthy life .......................... 1990 64.0 . . . 63.9 63.7 63.5 63.8 63.9 64.2
‡
64.8
‡
65.2 - - - 65
a. Black ................................... 1990 56.0 . . . 56.0 55.6 55.2 55.6 56.0 56.5
‡
57.4
‡
57.8 - - - 60
b. Hispanic
1
................................ 1990 64.8 . . . 63.6
2
64.0 63.2 64.2 64.0 64.7
‡
65.8
‡
66.3 - - - 65
c. People 65 years and over
3
................... 1990 11.9 . . . 11.8 11.9 11.9 12.1 12.0 12.2
‡
12.0
‡
12.2 - - - 14
8.2 Completion of high school (18-24 years) .......... 1992
§
86% . . . . . . . . . 86% 86% 85% 86% 86% 85% - - - 90%
a. Hispanic ................................. 1992
§
62% . . . . . . . . . 64% 62% 63% 67% 67% 63% - - - 90%
b. Black ................................... 1992
§
82% . . . . . . . . . 82% 83% 84% 82% 82% 81% - - - 90%
8.3 Preschool child development programs
Eligible children 4 years afforded opportunity to enroll in
Head Start ................................. 1990 47% . . . 55% - - - - - - - - - - - - - - - 40% 48% - - - 100%
Low-income children receiving 1 year of Head Start
services prior to entering kindergarten or first grade . . . . . - - - - - - - - - 58% 57% 58% 54% 49% - - - - - - - - - . . .
Disabled children 3-5 years enrolled in preschool ..... ... --- --- 5 6 % --- 5 6 % --- 6 3 % 6 2 % --- --- 7 3 % ...
8.4 Schools with comprehensive school health
education
All eight criteria met ........................... 1994 2.3% . . . . . . . . . . . . . . . - - - - - - - - - - - - - - - 75%
A documented, sequential program .............. 1994
§
48% ... ... ... ... ... --- --- --- --- --- ...
At least one health education course ............. 1994
§
77% ... ... ... ... ... --- --- --- --- --- ...
Instruction in six key behavioral areas ............ 1994
§
47% ... ... ... ... ... --- --- --- --- --- ...
Focus on skill development .................... 1994
§
39% ... ... ... ... ... --- --- --- --- --- ...
Health education teachers adequately trained ...... 1994 53% . . . . . . . . . . . . . . . - - - - - - - - - - - - - - - . . .
Designated coordinator for health education ....... 1994 38% . . . . . . . . . . . . . . . - - - - - - - - - - - - - - - . . .
Involvement of parents, health professionals, and
other concerned community members ........... 1994
§
31% ... ... ... ... ... --- --- --- --- --- ...
Evaluation of health education program during the
past 2 years .............................. 1994
§
67% ... ... ... ... ... --- --- --- --- --- ...
8.5 Health promotion in post-secondary institutions
Higher education institutions offering health promotion
activities ................................... 1989–90 20% . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - 50%
College students 18-24 years who report receiving
information from their college or university on:
Tobacco use prevention....................... ... --- --- --- --- --- --- 32% --- --- --- --- ...
Alcohol and other drug use prevention............ ... --- --- --- --- --- --- 59% --- --- --- --- ...
Violence prevention .......................... ... --- --- --- --- --- --- 38% --- --- --- --- ...
Injury prevention and safety.................... ... --- --- --- --- --- --- 26% --- --- --- --- ...
Suicide prevention........................... ... --- --- --- --- --- --- 21% --- --- --- --- ...
Pregnancy prevention ........................ ... --- --- --- --- --- --- 34% --- --- --- --- ...
Sexually transmitted disease prevention .......... ... --- --- --- --- --- --- 53% --- --- --- --- ...
AIDS or HIV infection prevention ................ ... --- --- --- --- --- --- 58% --- --- --- --- ...
Dietary behaviors and nutrition ................. ... --- --- --- --- --- --- 34% --- --- --- --- ...
Physical activity and fitness .................... ... --- --- --- --- --- --- 40% --- --- --- --- ...
See footnotes and key at end of table.
150 Healthy People 2000 Final Review

Table 8. Educational and Community-Based Programs objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
8.6 Worksite health promotion activities
Worksites with 50 or more employees (using 1985
analysis) ................................... 1985 65% - - - - - - 81% - - - - - - - - - - - - - - - - - - 90% 85%
Worksites with 50 or more employees (using 1992
analysis) ................................... ... --- --- --- 92% --- --- --- --- --- --- --- ...
Medium and large companies having a wellness
program ................................... 1987 63% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 85%
8.7 Hourly workers 18 years and over participating in
health promotion activities .................... 1994
§
23% ... ... ... ... ... --- --- --- 20% --- 20%
8.8 Health promotion programs for older adults
People 65 years and over participating in at least one
health promotion program through a facility serving
older adults................................. 1995 12% . . . . . . . . . . . . . . . . . . - - - - - - 12% - - - 90%
8.9 Family discussion of health issues—persons
10 years and over ........................... 1994 83% . . . . . . . . . . . . . . . - - - - - - - - - 69% - - - 75%
Nutrition .................................. ... --- --- --- --- --- 67% --- --- --- 54% --- ...
Physical activity............................. ... --- --- --- --- --- 66% --- --- --- 48% --- ...
Sexual behavior............................. ... --- --- --- --- --- 39% --- --- --- 22% --- ...
Tobacco .................................. ... --- --- --- --- --- 47% --- --- --- 33% --- ...
Alcohol ................................... ... --- --- --- --- --- 38% --- --- --- 26% --- ...
Illegal drugs................................ ... --- --- --- --- --- 33% --- --- --- 22% --- ...
Safety .................................... ... --- --- --- --- --- 50% --- --- --- 41% --- ...
Among 9th–12th grade students engaging in family
discussion of HIV/AIDS ........................ 1989 54% 53% 61% - - - 66% - - - 63% - - - 63% - - - - - - 75%
8.10 Number of States with community health programs
addressing at least three Healthy People 2000
objectives that reach 40% of State population .... ... --- --- --- --- --- --- --- --- --- --- --- 50
Proportion of States with 90 percent of local health
departments providing services that address three or
more Healthy People 2000 priority areas........... ... --- --- --- ---
4
81% --- --- --- --- --- --- ...
8.11 Counties with programs for racial/ethnic minority
groups .................................... ... --- --- --- --- --- --- --- --- --- --- --- 50%
Percent of local health departments providing culturally
and linguistically appropriate services
Health promotion
Physical activity and fitness .................. 1996-97 21% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Nutrition ................................. 1996-97 44% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Tobacco ................................. 1996-97 24% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Alcohol and other drugs ..................... 1996-97 26% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Family planning ........................... 1996-97 42% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Mental health and mental disorders ............ 1996-97 18% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Violent and abusive behavior ................. 1996-97 25% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Education and community-based programs ...... 1996-97 33% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
See footnotes and key at end of table.
Healthy People 2000 Final Review 151

Table 8. Educational and Community-Based Programs objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Health protection
Unintentional injuries ....................... 1996-97 19% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Occupational safety and health ............... 1996-97 13% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Environmental health ....................... 1996-97 22% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Food and drug safety ....................... 1996-97 18% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Oral health ................................ 1996-97 25% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Preventive services
Maternal and infant health ................... 1996-97 47% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Heart disease and stroke .................... 1996-97 28% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Cancer .................................. 1996-97 30% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Diabetes and other chronic disabling conditions . . . 1996-97 26% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
HIV infection ............................. 1996-97 45% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Sexually transmitted diseases ................ 1996-97 41% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Immunization and infectious diseases........... 1996-97 48% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Clinical preventive services .................. 1996-97 35% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
Surveillance and data systems ................. 1996-97 14% . . . . . . . . . . . . . . . . . . . . . . . . - - - - - - 50%
8.12 Hospital-based patient education and community
health promotion
Patient education programs
Registered hospitals ......................... 1987 68% 86% - - - - - - 90% - - - - - - - - - - - - - - - 80% 90%
Health maintenance organizations ............... ... --- --- --- --- 90% --- --- --- --- --- --- 90%
Health education classes .................... ... ---
5
75% --- 84% ... --- --- --- --- --- --- ...
Nutrition counseling ........................ ... ---
5
85% --- 87% ... --- --- --- --- --- --- ...
Smoking cessation classes................... ... --- --- --- 67% ... --- --- --- --- --- --- ...
Community health promotion programs
Community hospitals ....................... 1987 60% 77% - - - - - - 90% - - - - - - - - - - - - - - - - - - 90%
8.13 Television partnerships with community
organizations for health promotion ............. 1995-96 100% . . . . . . . . . . . . . . . . . . . . . - - - - - - - - - 75%
8.14 Effective public health systems
Local health departments reporting:
Health assessment
Behavioral risk assessment .................... 1990 33% . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - 90%
Morbidity data .............................. 1990 49% . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - 90%
Reportable disease .......................... 1990 87% . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - 90%
Vital records and statistics ..................... 1990 64% . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - 90%
Surveillance — chronic disease ................. 1990 55% . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - 90%
Surveillance — communicable disease ........... 1990 92% . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - 90%
Policy development functions and services
Health code development and enforcement ........ 1990 59% . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - 90%
Health planning ............................. 1990 57% . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - 90%
See footnotes and key at end of table.
152 Healthy People 2000 Final Review
Table 8. Educational and Community-Based Programs objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Health assurance
Health education ............................ 1990 74% . . . - - - - - -
4
84% --- --- --- --- --- --- 90%
Child health................................ 1990 84% . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - 90%
Immunizations .............................. 1990 92% . . . - - - - - -
4
96% --- --- --- --- --- --- 90%
Prenatal care............................... 1990 59% . . . - - - - - -
4
64% --- --- --- --- --- --- 90%
Primary care ................................. 1990 22% . . . - - - - - -
4
30% --- --- --- --- --- --- 90%
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
‡
The NHIS was redesigned in 1997. Data may not be comparable with previous years; see Appendix.
1
Estimate based on preliminary data. Excludes mortality data from States lacking a Hispanic-origin item on their death certificate or for which Hispanic-origin data were not of sufficient quality.
See Appendix.
2
Estimate derived from 1991-93 health status data and 1992 mortality data.
3
Years of healthy life remaining for those surviving to age 65.
4
1992–1993 data. Data are from 43 States and represent local health department’s report of whether a program or service existed.
5
1988 data.
NOTES: Data include revisions and, therefore, may differ from data previously published in these reports and other publications. HIV is human immunodeficiency virus. AIDS is acquired
immunodeficiency syndrome.
Objective number Data source
8.1*, 8.1a-c National Vital Statistics System, CDC, NCHS.
National Health Interview Survey, CDC, NCHS.
8.2, 8.2a-b National Center for Education Statistics, National Education Goals Panel.
8.3 Head Start data: Head Start Bureau: Administration on Children, Youth, and Families.
Data on disabled children: National Center for Education Statistics, National Education Goals Panel.
8.4 School Health Policies and Programs Study, CDC, NCCDPHP.
8.5 Baseline: Health Promotion on Campus Survey and Directory, American College Health Association.
Data for college students: National College Health Risk Behavior Survey, CDC, NCCDPHP.
8.6 Baseline and updates for worksites with 50 or more employees : National Survey of Worksite Health Promotion Activities OPHS, ODPHP.
Baseline for medium and large companies: Health Research Institute Biennial Survey, Health Research Institute.
8.7 National Health Interview Survey, CDC, NCHS.
8.8 National Health Interview Survey, CDC, NCHS.
8.9 1989 baseline: Secondary School Student Health Risk Survey, CDC, NCCDPHP.
1990–97 data: Youth Risk Behavior Survey, CDC, NCCDPHP.
1994 and 1998 data: National Health Interview Survey, CDC, NCHS.
8.10 National Profile of Local Health Departments, National Association of City and County Health Officials.
8.11 National Profile of Local Health Departments, National Association of City and County Health Officials.
8.12 Annual Survey of Hospitals, AHA.
HMO Industry Profile, Group Health Association of America, Inc.
8.13 Media Health Partnerships Survey, CDC, NCCDPHP.
8.14 National Profile of Local Health Departments, National Association of City and County Health Officials.
* Duplicate objective. See full text of objective following this table.
Met
Toward
Mixed/ no change
Away
Cannot assess
Healthy People 2000 Final Review 153
Educational and
Community-Based
Programs Objectives
8.1*: Increase years of healthy life to at
least 65 years.
Duplicate objectives: 17.1 and 21.1
8.1a*: Increase years of healthy life
among black persons to at least 60
years.
Duplicate objectives: 17.1a and
21.1a
8.1b*: Increase years of healthy life
among Hispanics to at least 65
years.
Duplicate objectives: 17.1b and
21.1b
8.1c*: Increase years of healthy life
among people aged 65 and older to
at least 14 years remaining at age
65.
Duplicate objectives: 17.1c and
21.1c
8.2: Increase the high school graduation
rate to at least 90 percent, thereby
reducing risks for multiple problem
behaviors and poor mental and physical
health.
8.2a: Increase the high school
graduation rate among Hispanics to
at least 90 percent, thereby reducing
risks for multiple problem behaviors
and poor mental and physical
health.
8.2b: Increase the high school
graduation rate among blacks to at
least 90 percent, thereby reducing
risks for multiple problem behaviors
and poor mental and physical
health.
8.3: Achieve for all disadvantaged
children and children with disabilities
access to high quality and
developmentally appropriate preschool
programs that help prepare children for
school, thereby improving their
prospects with regard to school
performance, problem behaviors, and
mental and physical health.
8.4: Increase to at least 75 percent the
proportion of the Nation’s elementary
and secondary schools that provide
planned and sequential kindergarten–
12th grade comprehensive school health
education.
8.5: Increase to at least 50 percent the
proportion of postsecondary institutions
with institution-wide health promotion
programs for students, faculty, and staff.
8.6: Increase to at least 85 percent the
proportion of workplaces with 50 or
more employees that offer health
promotion activities for their employees,
preferably as part of a comprehensive
employee health promotion program.
8.7: Increase to at least 20 percent the
proportion of hourly workers who
participate regularly in
employer-sponsored health promotion
activities.
8.8: Increase to at least 90 percent the
proportion of people aged 65 and older
who had the opportunity to participate
during the preceding year in at least one
organized health promotion program
through a senior center, lifecare facility,
or other community-based setting that
serves older adults.
8.9: Increase to at least 75 percent the
proportion of people aged 10 and older
who have discussed issues related to
nutrition, physical activity, sexual
behavior, tobacco, alcohol, other drugs,
or safety with family members on at
least one occasion during the preceding
month.
8.10: Establish community health
promotion programs that separately or
together address at least three of the
Healthy People 2000 priorities and reach
at least 40 percent of each State’s
population.
8.11: Increase to at least 50 percent the
proportion of counties that have
established culturally and linguistically
appropriate community health promotion
programs for racial and ethnic minority
populations.
8.12: Increase to at least 90 percent the
proportion of hospitals, health
maintenance organizations, and large
group practices that provide patient
education programs, and to at least
90 percent the proportion of community
hospitals that offer community health
promotion programs addressing the
priority health needs of their
communities.
8.13: Increase to at least 75 percent the
proportion of local television network
affiliates in the top 20 television markets
that have become partners with one or
more community organizations around
one of the health problems addressed by
the Healthy People 2000 objectives.
8.14: Increase to at least 90 percent the
proportion of people who are served by
a local health department that is
effectively carrying out the core
functions of public health.
*Duplicate objective.
154 Healthy People 2000 Final Review

Priority Area 9
Unintentional
Injuries
Background
Unintentional injuries are the fifth
leading cause of death in the United
States, accounting for more than 97,000
deaths in 1998 (1). They are a major
cause of disabilities and hospitalization;
in 1998 there were nearly 29 million
visits to emergency rooms because of
unintentional injuries (2). Unintentional
injuries have a significant impact on
health care costs; in 1998 alone, medical
expenses attributable to unintentional
injuries were estimated at nearly $80
billion; when costs for losses of income
and productivity, insurance and legal
administration, motor-vehicle damage,
fire loss and losses by employers are
included, the estimate climbs to $480.5
billion (3).
Motor vehicle injuries remain the
most costly and fatal of unintentional
injuries. The National Safety Council
estimates motor vehicle crashes cost the
United States $169 billion in lost wages,
medical expenses, and administrative
costs. However, efforts to reduce motor
vehicle-related injuries show promise.
The National Highway Traffic Safety
Administration (NHTSA) estimates that
if all occupants in passenger vehicles
had used their safety belts, nearly
one-half of the 41,000 motor vehicle
deaths in 1996 could have been
prevented (4). Teens are overrepresented
among motor vehicle deaths. While
constituting 10 percent of the total
population, teenagers account for
15 percent of motor vehicle deaths (5).
Implementation of graduated licensing
programs may help reduce the number
of teenage motor vehicle deaths (6).
Motorcyclists are also
disproportionally represented among
motor vehicle deaths; per mile traveled,
16 motorcyclists are killed for each
automobile occupant (5). Motorcycle
helmet use is associated with less severe
injuries and lower health care costs (7).
In 1995 alone, 791 lives would have
been saved if all motorcyclists wore
helmets (8). While States requiring
helmet use increased during the early
1990’s, the number began declining in
1997.
Alcohol use has long been
recognized as contributing to motor
vehicle deaths and injuries. The rate of
alcohol-related motor vehicle deaths has
declined between 1987 and 1999. In
part, this may be attributed to
improvements in the engineering and
safety of motor vehicles. However,
NHTSA has also highlighted the impact
of the passage and enforcement of laws
limiting drinking to 21 years of age and
over (9).
Although less visible, fall-related
injury and death rates among older
persons in the United States are
increasing. Falls and fall-related injuries
are the second leading cause of injury
deaths among people 65–84 years of age
and the leading cause for people 85
years of age and over (1).
Data Summary
Highlights
Significant progress has been seen
in the past decade regarding deaths due
to unintentional injuries. The overall
age-adjusted rate of deaths from
unintentional injuries (9.1) decreased,
from 34.7 per 100,000 persons in 1987
to 30.1 in 1998, as did the rates for
special populations. The targeted
components of unintentional injuries
including motor-vehicle crashes (9.3),
drowning (9.5), and fire (9.6) also
decreased.
Particularly noteworthy were
objectives that targeted motor vehicle
fatalities and use of vehicle occupant
restraints. The rate of deaths caused by
motor vehicle crashes (9.3) decreased
from 2.4 per 100 million vehicle miles
traveled in 1987 to 1.6 in 1998. The rate
of deaths from motor vehicle crashes
per 100,000 persons also declined, from
19.2 in 1987 to 15.3 in 1998. The death
rate for motorcyclists involved in
collisions decreased from 42.5 per 100
million vehicle miles traveled in 1987 to
22.3 in 1998, surpassing the Healthy
People 2000 target of 25.6. The rate of
motorcyclists’ deaths per 100,000
persons declined from 1.7 in 1987 to 0.8
in 1998, also surpassing the target of
0.9. For pedestrians struck by motor
vehicles, the death rate decreased from
2.8 per 100,000 persons in 1987 to 1.8
in 1999, exceeding the objective target
of 2.0.
Corresponding to the decrease in
motor vehicle deaths, the use of safety
belts by motor vehicle occupants (9.12)
increased from 42 percent in 1988 to 67
in 1999. Use of helmets by
motorcyclists (9.13) increased from
60 percent in 1988 to 67 in 1998. The
number of States with laws requiring the
use of safety belts for all ages (9.14)
increased from 33 in 1989 to 49 in
1999. The number of States having a
graduated driver licensing system for
novice drivers and riders under the age
of 18 (9.26) increased from zero in 1994
to 23 in 1999.
The rate of nonfatal head injuries
(9.9) decreased from 118 per 100,000
persons in 1988 to 61 in 1998, far
surpassing the year 2000 target of 106.
The rate of nonfatal spinal cord injuries
(9.10) decreased from 5.3 per 100,000
persons in 1988 to 4.5 in 1998, also
exceeding the year 2000 target of 5.0.
The rate of nonfatal poisonings
requiring emergency department
treatment (9.8) decreased from 104
visits per 100,000 persons in 1986 to 33
in 1999, well below the year 2000 target
of 88. The rate for children 4 years of
age and under decreased from 664 in
1986 to 418 in 1999 also surpassing the
target of 520.
The rate of hospitalizations for
nonfatal injuries (9.2) decreased from
832 hospitalizations per 100,000 persons
in 1988 to 564 in 1998, surpassing by
34 percent the year 2000 target of 754.
For black males, the rate declined from
1,007 in 1991 to 628 in 1998,
surpassing the target of 856 by
36 percent.
Moving in the other direction, the
hospitalization rate for hip fractures
among people 65 years of age and over
(9.7) increased from 714 hospitalizations
per 100,000 persons in 1988 to 863 in
1998. However, the rate for hip fractures
for white women 85 years of age and
over, the group at highest risk,
decreased marginally from 2,721 in
1988 to 2,690 in 1998.
Summary of Progress
By the end of the decade, targets
had been met for seven objectives: 9.2,
9.6, 9.8–9.10 , 9.18, and 9.22. Eleven
objectives made substantial progress:
9.1, 9.3, 9.5, 9.12, 9.13, 9.16, 9.19,
9.23, and 9.24–9.26 . The two measures
used to track one objective (9.14)
showed mixed progress. Objective 9.21
also showed mixed progress, although
this was based on data only for nurse
practitioners (see Data Issues). Two
objectives (9.7 and 9.17) moved away
from the Healthy People 2000 target.
However, more recent supplemental data
Healthy People 2000 Final Review 155

9.1 Unintentional injury deaths
9.2 Unintentional injury hospitalizations
9.3 Motor vehicle deaths: Per 100 million miles traveled
Per 100,000 population
9.4 Fall-related deaths
9.5 Drowning deaths
9.6 Residential fire deaths
9.7 Hip fractures among older adults
9.8 Nonfatal poisoning
9.9 Nonfatal head injuries
9.10 Nonfatal spinal cord injuries
9.12 Motor vehicle occupant protection systems
9.13 Helmet use: Motorcyclists
9.15 States with handguns designed to protect children
Bicyclists
Motorcycle helmet laws
9.14 States with: Safety belt laws
9.17 Residences with smoke detectors
9.18 Injury prevention instruction in required course
9.19 Use of protective equipment: Baseball - headgear
Baseball - mouth guard
Football - headgear
Soccer - headgear
Soccer - mouth guard
9.22 States with linked EMS and trauma systems
Football - mouth guard
9.23 Alcohol-related motor vehicle deaths
9.24 States with bicycle helmet laws
9.25 States with firearm storage laws
9.26 States with graduated licensing systems
Moved away from target Moved toward target Met target
NOTE: Complete tracking data are shown in table 9. Progress quotients are not calculated for objectives 9.11, 9.16, 9.20, and 9.21.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
78%
63%
-4%
-5%
58%
19%
5%
18%
14%
5%
No change
No change
8%
15%
35%
66%
Percent of target achieved
-100 -80 -60 -40 -20 0 20 40 60 80 100
344%
140%
444%
475%
267%
100%
108%
24%
35%
-139%
94%
89%
85%
93%
156 Healthy People 2000 Final Review
Figure 9. Final status of Unintentional Injuries objectives
for objective 9.17 (smoke detectors)
does show progress in the proportion of
people with a working smoke detector
on each floor of their residence,
88 percent in 1998 (see Data Issues).
Two objectives showed no change (9.4
and 9.15). Data sources were never
developed for two objectives (9.11 and
9.20). See table 9 for the tracking data
for the objectives in this priority area
and figure 9 for a quantitative
assessment of progress.
Discussion
The decline in the rate of deaths
from motor vehicle crashes can be
attributed to a combination of factors;
raising the legal drinking age to 21,
increased use of safety belts and child
restraint seats, zero tolerance for
drinking and driving by youth, stricter
law enforcement, administrative license
revocation, graduated driver licensing
systems, and stiffer penalties upon
conviction for drunk driving (10).
Alcohol continues to be a major
risk factor for all unintentional injuries,
contributing to an estimated 38 percent
of motor vehicle-related deaths (11).
Raising the minimum legal drinking age
to 21 years was accompanied by
reduced alcohol consumption, traffic
crashes, and related fatalities among
young persons under age 21 years.
Reductions in alcohol-related traffic
crashes are associated with many policy
and program measures, among them,
administrative revocation of licenses for
drinking and driving and lower legal
blood alcohol limits for youth and
adults. Community programs involving
multiple city departments and private
citizens have reduced driving after
drinking and traffic deaths and injuries.
In addition, a combination of
community mobilization, media
advocacy, and enhanced law
enforcement has been shown to reduce
alcohol-related traffic crashes and sales
of alcohol to minors. Alcohol
consumption has also been linked with a
substantial proportion of injuries and
deaths from falls, fires, and drowning.
Smoke alarms reduce by half the
risk of dying from a fire, while home
sprinkler systems decrease the risk of
death by 65 percent and property loss by
48 percent (10). As a result, operable
residential smoke alarms can be highly
effective in preventing fire-related
deaths. It is important to understand that
any smoke alarm will offer adequate
warning for escape, provided that the
alarm is listed by an independent testing
laboratory and is properly installed and
maintained. Fire safety initiatives
targeted at the home and at increased
smoke alarm use remain the key to any
reductions in residential fire deaths. Five
major strategies are: first, continued
widespread public fire safety education
is needed discussing fire prevention and
how to avoid serious injury or death if
fire occurs. Second, more people must
use and maintain smoke alarms and
develop and practice escape plans.
Third, wider use of residential sprinklers
must be aggressively pursued. Fourth,
additional ways must be developed to
make home products fire safe.
Regulations requiring child-resistant
lighters are a good example. The wider
use of upholstered furniture and
mattresses that are more resistant to
cigarette ignitions are examples of
changes that have already accomplished
much. Fifth, the special fire safety needs
of population groups at high risk, such
as the young, older adults, and the
disadvantaged need to be addressed
(12,13).
Child-resistant packaging, product
reformulation and interventions by
poison control centers and health
professionals all helped reduce the
childhood poison-related death rate by
38 percent from 1987 to 1998 (14).
Also, prescription drug interactions
contribute to frailty and loss of visual
acuity in the elderly and, consequently,
to their high incidence of falls and
resultant fractures. The death rate from
falls among those 85 years and over is
almost eight times the rate for those
65–84 years of age and has increased by
almost 21 percent over the past decade.
Wearing a bicycle helmet reduces
the risk of head injury by as much as
85 percent (15). Increases in usage may
be possible with the introduction of
multisport helmets and the help of the
media to institutionalize the wearing of
these helmets.
Transition to Healthy People
2010
Most of the topics covered in the
Healthy People 2000 priority areas on
Unintentional Injuries (Priority Area 9)
and Violent and Abusive Behavior
(Priority Area 7) were reorganized into
one Focus Area: Injury and Violence
Prevention in Healthy People 2010. The
rationale for combining these two focus
areas was that many injuries produce the
same outcome although they may result
from different factors. For example, a
nonfatal spinal cord injury produces the
same outcome whether it is caused by
an unintentional motor vehicle crash or
an attempted suicide. Understanding
injuries allows for development and
implementation of effective prevention
interventions. Some interventions can
reduce injuries from both unintentional
and violence-related episodes. For
instance, efforts to promote proper
storage of firearms in homes can help
reduce the risk of assault with a firearm
and intentional self-inflicted and
unintentional firearm-related injuries in
the home (16). Higher taxes on
alcoholic beverages are associated with
lower death rates from motor vehicle
crashes and lower rates for some
categories of violent crime, including
rape (17,18).
Many injuries and injury-related
deaths occur in some population groups
(such as younger children from birth to
age 4 years) where the intentionality of
the injury is unknown and requires more
detailed investigation. As these cases are
examined, interventions can be
developed to address ways injuries
occur. An example of this, which is
emerging as a growing public health
concern, is poisonings in children.
Overall there are fewer objectives
for the two subject areas. Healthy
People 2000 had 45 objectives in the
Unintentional Injuries and Violence and
Abusive Behaviors chapters compared
with 39 objectives in the Healthy People
2010 Injury and Violence Prevention
chapter. For unintentional injuries
Healthy People 2000 had 29 objectives
compared with 19 objectives in the
Healthy People 2010 edition. Violence
had 19 objectives in Healthy People
2000 compared with 8 objectives in
Healthy People 2010. The Healthy
People 2010 chapter has a new section
titled ‘‘Injury Prevention’’ containing 12
objectives that relate to both subject
areas (for example, nonfatal head
injuries and nonfatal spinal cord injuries,
child fatality review, emergency
department and hospital discharge
surveillance systems).
The Healthy People 2010 objective
for deaths due to motor vehicle crashes,
which was retained from Healthy People
2000 (with a minor variation), has been
designated as a measure of one of the
Leading Health Indicators (LHI). Motor
Healthy People 2000 Final Review 157
vehicle crashes are the most common
cause of serious injury and so provide
an indication of overall serious injury
rates. The LHIs are discussed in further
detail in the Introduction.
Some of the objectives contained in
the unintended injury priority area of
Healthy People 2000 have been
transferred to more specific focus areas
in Healthy People 2010. These are listed
as related objectives in Healthy People
2010.
Appendix
table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the changes between the
two decades of objectives, reflecting
new knowledge and direction for this
area.
Data Issues
Definitions
Objective 9.2 (nonfatal
unintentional injuries) is tracked with
data from the National Hospital
Discharge Survey (NHDS) maintained
by the National Center for Health
Statistics (NCHS), Centers for Disease
Control and Prevention (CDC). The
ICD–9 codes designated for this
objective include both unintentional and
intentional injuries (see Appendix
table V). The two types of injuries
cannot be distinguished at the national
level because currently only 22 States
mandate the use of E-codes (external
causes) on hospital discharge forms.
NCHS is working with States to
increase the use of E-codes.
Data for objective 9.6 (residential
fire deaths) include all fire-related
deaths. While 90 percent occur at home,
a small proportion occur elsewhere. The
1990 baseline data for fire-related deaths
for Puerto Ricans (9.6g) have been
revised. The original baseline published
in the Midcourse Review and 1995
Revisions (19) included data for 45
States and the District of Columbia. It
did not include data for New York City
where about 40 percent of the U.S.
Puerto Rican population resides. The
revised baseline, which includes data for
47 States (including New York) and the
District of Columbia, is considerably
lower than originally published and, in
fact, has met the year 2000 target for
this subobjective. The number of States
reporting Hispanic origin data on birth
and death certificates has varied from
year to year; see the Appendix for more
information.
Objective 9.7 (hip fractures among
older adults) is also monitored with data
from the NHDS. These rates are based
on extremely small numbers and must
be interpreted cautiously. Data on race
are not reported by many hospitals
because of omission of a race field on
hospital discharge reporting forms. More
hospitals have automated their discharge
systems in recent years and are using
these forms (UB–82 and UB–92). A
comparison of NHDS data with the
National Health Interview Survey
(NHIS) data for people who reported
being hospitalized (NHIS data were
adjusted to exclude hospitalizations of 1
day or less) indicated that
underreporting for whites was roughly
22 percent in 1991; the difference for
blacks was negligible (20).
Objective 9.11 refers to secondary
conditions, which occur as a result of a
spinal cord injury and include a
pathology, an impairment, a functional
limitation, or a disability.
Objectives 9.14 (safety belt and
motorcycle helmet laws), 9.15 (handgun
design laws), 9.22 (trauma linking
systems), 9.24 (bicycle helmet laws),
9.25 (handgun storage laws), and 9.26
(graduated driver licensing) all relate to
State laws or programs that vary across
States in populations targeted, penalties,
and liability.
The baseline and target for
objective 9.17 (smoke detectors) are
based on estimates of the proportion of
homes with working smoke detectors;
this is somewhat different from the
intent of the objective, which focuses on
working smoke detectors on each
habitable floor. Findings from a 1993
survey conducted by the Consumer
Product Safety Commission (CPSC)
indicate that 52 percent of households
had at least one functional smoke
detector on each floor (21). Data from
the 1993 NHIS did not specify a
working smoke detector. The 1994 and
1998 supplemental data on the
proportion of homes with working
smoke detectors are from the NHIS, a
different source than that used for the
baseline.
Data Sources
Data for objective 9.3 (motor
vehicle crash deaths) and the
subobjectives (except d and g) are crude
rates from the Department of
Transportation’s Fatality Analysis
Reporting System (formerly the Fatal
Accident Reporting System) (FARS).
See the Appendix for a discussion of
crude and age-adjusted rates and the
chapter on Priority Area 4 for a
description of FARS. The rates for 9.3d
(American Indian/Alaska Native) and
9.3g (Mexican American) are
age-adjusted data from the National
Vital Statistics System.
Baseline data for objective 9.21
(injury prevention counseling) are from
the Primary Care Provider Surveys
(PCPS). The sample for the survey was
drawn from the membership rolls of
professional associations of internists,
family physicians, nurse practitioners,
pediatricians, and obstetricians/
gynecologists. Response rates from these
groups varied from 50–80 percent. The
data on inquiry about seat-belt use and
falls in the home represent the
proportion of practitioners who routinely
queried 81–100 percent of their patients
about these risks. The data on
counseling about these issues represent
the proportion of providers who
routinely provided these services to their
patients who needed the services. The
basis for counseling may be independent
of the inquiry made by the clinicians.
The Prevention in Primary Care
Study (PPCS) was conducted in
1997–98 to update data from the PCPS.
The design and items included in the
1997–98 study were similar to the
PCPS, but a slightly different sampling
frame was used and some items
included in the 1992 surveys were not
included in the PPCS. The professionals
were sampled from listings of all
licensed, active practitioners in the
United States whose practices were at
least 50 percent primary care. Because
of low response rates from the other
provider groups, updates are available
only for nurse practitioners.
Alcohol-related motor vehicle
crashes (9.23) are tracked using data
from FARS. The FARS supplements
death certificate data with information
on the circumstances of the death to
determine whether the death was alcohol
related. The National Vital Statistics
System does not specify alcohol-related
motor vehicle crashes.
Data Comparability
Data for 9.8 (nonfatal poisonings)
are from the National Electronic Injury
Surveillance System (NEISS), which is
maintained by the CPSC. This system
158 Healthy People 2000 Final Review
does not utilize ICD–9 or other
conventional injury coding mechanisms.
Injuries reported in the system are
limited to those related to products
regulated by CPSC in a given year.
Therefore, variation in the numbers and
types of products affect the number of
injuries reported in the system. The
baseline and estimates were again
revised in 1997 to accommodate a new
sampling design.
In 1992, data collection for
objectives 9.12 (motor vehicle occupant
protection systems) and 9.13 (helmet use
by motorcyclists and bicyclists) was
expanded from 19 metropolitan areas to
all 50 States. The data collection
methods (direct observation) were
unchanged.
Proxy Data
Tracking data for 9.16 (fire
suppression systems) are from the U.S.
Fire Administration’s National Fire
Incident Reporting System (NFIRS) and
indicate the proportion of fires in
residential properties that have
automatic suppression systems. Data on
localities for this objective are not
available.
Regarding objective 9.19 (protective
sports equipment), use of protective
equipment in college sports is required
by the National Collegiate Athletic
Association. The additional data are
from the NHIS and represent the
proportions of children playing baseball,
softball, football, or soccer who use
headgear or mouthguards.
References
1. National Vital Statistics System. Centers for
Disease Control and Prevention, National Center
for Health Statistics. 1999.
2. McCaig LF. National Hospital Ambulatory
Medical Care Survey: 1998 Emergency department
summary. Advance data from vital and health
statistics; no.313. Hyattsville, Maryland: National
Center for Health Statistics. 2000.
3. National Safety Council. Injury facts.
Washington. July 2000.
4. National Highway Traffic Safety
Administration. Occupant protection facts.
Washington: Department of Transportation. 1996.
5. Fatality Analysis Reporting System. Department
of Transportation, National Highway Traffic Safety
Administration. 1997.
6. National Highway Traffic Safety
Administration. Traffic safety facts 1996: Young
drivers. Washington: Department of
Transportation. 1997.
7. Hertz ES. The effect of helmet law repeal on
motorcycle fatalities: A four-year update. Research
Notes. Washington: National Center for Statistics
and Analysis, National Highway Traffic Safety
Administration. 1990.
8. Rowland J, et al. Motorcycle helmet use and
injury outcome and hospitalization costs from
crashes in Washington State. Am J Public Health
86(1):41–5. 1996.
9. National Highway Traffic Safety
Administration. Fatal Accident Reporting System,
1989. A review of information on fatal traffic
crashes in the United States, 1989. Washington:
Department of Transportation. 1991.
10. Healthy People 2000 Progress Review:
Unintentional injuries. Aug. 5, 1999.
11. National Highway Traffic Safety
Administration. Traffic safety facts 1998: Alcohol.
Washington: National Highway Traffic Safety
Administration. 1999.
12. Hall JR. Patterns of civilian fire casualties in
home fires by age and sex, 1993–97. Quincy:
National Fire Protection Association. Apr. 2000.
13. Fahy RF, Miller AL. How Being Poor Affects
Fire Risk. Fire Journal 83(1):28. 1989.
14. National Safe Kids Campaign. Poison.
Washington. 1999.
15. National Safe Kids Campaign. Preventing a
fall, protecting a child. Washington. 1999.
16. Cummings P, Grossman DC, Rivara FP, et al.
State gun storage laws and child mortality due to
firearms. JAMA 278 (13). 1997.
17. Chaloupka FJ, Saffer H, Grossman M. Alcohol
control policies and motor vehicle fatalities.
Journal of Legal Studies 22:161–86. 1993.
18. Cook PJ, Moor MJ. Economic perspectives on
reducing alcohol-related violence. In: Martin SE,
ed. Alcohol and interpersonal violence: Fostering
multidisciplinary perspectives. Based on a
workshop on alcohol-related violence sponsored
by National Institute on Alcohol Abuse and
Alcoholism, May 14–15, 1992. Rockville,
Maryland: National Institutes of Health. 1993.
19. Department of Health and Human Services.
Healthy people 2000 midcourse review and 1995
revisions. Washington: Public Health Service.
1995.
20. Kozak LJ. Underreporting of race in the
National Hospital Discharge Survey. Hyattsville,
Maryland: National Center for Health Statistics.
1995.
21. Smith CL. Smoke detector operability survey
report on findings. Directorate for Economic
Analysis. Consumer Product Safety Commission.
1994.
Healthy People 2000 Final Review 159

Table 9. Unintentional Injuries objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
9.1 Unintentional injury deaths (age adjusted
per 100,000) ........................ 1987 34.7 32.5 31.0 29.4 30.3 30.3 30.5 30.4 30.1 30.1 - - - 29.3
a. American Indian/Alaska Native ......... 1987 66.0 59.0 58.3 57.3 58.1 58.3 56.7 57.6 58.5 55.6 - - - 53.0
b. Black male ........................ 1987
§
68.0 62.4 61.0 56.7 59.8 58.5 57.6 55.7 54.2 54.4 - - - 51.9
c. White male ........................ 1987
§
49.8 46.4 43.9 41.9 42.7 42.7 43.0 42.4 42.0 42.2 - - - 42.9
d. Mexican American male
1
............. 1990
§
53.1 . . . 47.2 46.5 48.6 46.1 44.6 45.4 43.0 44.2 - - - 43.0
9.2 Unintentional injury hospitalizations
(per 100,000)
2
........................ 1988
§
832 780 764 714 699 654 635 612 582 564 - - - 754
a. Black male ........................ 1991 1,007 . . . . . . 969 893 847 911 730 637 628 - - - 856
9.3 Motor vehicle crash deaths
Per 100 million vehicle miles traveled (VMT) . . 1987 2.4 2.1 1.9 1.7 1.7 1.7 1.7 1.7 1.6 1.6 1.6 1.5
Per 100,000 population .................. 1987 19.2 17.9 16.5 15.4 15.6 15.6 15.9 15.9 15.7 15.4 15.3 14.2
a. Children 14 years and under (per
100,000) ......................... 1987 6.3 5.3 5.1 4.8 4.9 5.1 4.9 4.8 4.6 4.4 4.2 4.4
b. People 15–24 years (per 100,000) ...... 1987 36.3 33.4 31.4 28.0 28.5 28.9 29.9 28.9 27.4 26.7 27.2 26.8
c. People 70 years and over (per 100,000) . . 1987 22.9 22.9 22.3 21.9 22.9 23.4 23.3 23.3 23.9 23.3 22.4 20
d. American Indian/Alaska Native (age
adjusted per 100,000) ............... 1987 37.7 33.2 33.4 32.0 32.3 31.4 33.1 34.0 32.3 31.8 - - - 32.0
e. Motorcyclists (per 100 million VMT) ..... 1987
§
42.5 33.8 30.6 25.1 24.7 22.7 22.7 21.8 20.9 22.3 - - - 25.6
(per 100,000) ........... 1987 1.7 1.3 1.1 0.9 0.9 0.9 0.9 0.8 0.8 0.8 0.8 0.9
f. Pedestrians (per 100,000) ............. 1987 2.8 2.6 2.3 2.2 2.2 2.1 2.1 2.0 2.0 1.9 1.8 2.0
g. Mexican American (age adjusted per
100,000)
1
........................ 1990 20.9 . . . 18.9 17.5 18.1 18.7 17.7 18.0 17.0 16.7 - - - 18.0
9.4 Fall-related deaths (age adjusted per
100,000) ......................... 1987 2.7 2.7 2.6 2.5 2.5 2.5 2.6 2.7 2.7 2.7 - - - 2.3
a. People 65–84 years (per 100,000) ...... 1987 18.1 17.8 18.0 17.6 17.8 18.3 18.5 19.9 20.7 21.5 - - - 14.4
b. People 85 years and over (per 100,000) . . 1987 133.0 143.1 147.5 147.3 149.5 147.0 152.0 159.6 160.3 162.7 - - - 105.0
c. Black male 30–69 years (per 100,000) . . . 1987 8.1 6.8 6.2 5.3 5.5 5.4 4.8 5.3 4.5 4.9 - - - 5.6
d. American Indian/Alaska Native (age
adjusted per 100,000) ............... 1990 3.2 . . . 3.1 3.1 4.3 3.2 3.8 2.9 4.0 3.5 - - - 2.8
9.5 Drowning deaths (age adjusted per
100,000) ............................ 1987 2.1 1.9 1.9 1.6 1.7 1.5 1.7 1.5 1.5 1.6 - - - 1.3
a. Children 4 years and under (per
100,000) ......................... 1987
§
4.3 3.4 3.6 3.2 3.2 2.8 3.7 2.8 2.7 2.9 - - - 2.3
b. Male 15–34 years (per 100,000) ........ 1987 4.5 4.0 4.1 3.4 3.6 3.1 4.6 3.0 3.2 3.4 - - - 2.5
c. Black male (age adjusted per 100,000) . . . 1987 6.6 5.0 5.8 4.1 4.3 4.0 4.1 3.9 3.5 4.4 - - - 3.6
d. American Indian/Alaska Native (age
adjusted per 100,000) ............... 1990 4.3 . . . 3.8 4.0 4.3 4.3 3.5 3.3 3.8 3.0 - - - 2.0
9.6 Residential fire deaths
Fire-related deaths (age adjusted per
100,000)
3
........................... 1987
§
1.7 1.5 1.5 1.4 1.3 1.4 1.2 1.2 1.1 1.0 - - - 1.2
a. Children 4 years and under (per
100,000) ......................... 1987
§
4.5 3.5 3.8 3.4 3.6 3.5 2.6 2.4 2.1 1.6 - - - 3.3
See footnotes and key at end of table.
160 Healthy People 2000 Final Review

Table 9. Unintentional Injuries objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
b. People 65 years and over (per 100,000) . . 1987
§
4.9 4.1 3.9 3.7 3.7 3.5 3.6 3.8 3.5 3.2 - - - 3.3
c. Black male (age adjusted per 100,000) . . . 1987
§
6.4 5.2 5.1 4.9 4.6 4.5 4.2 3.9 3.4 3.1 - - - 4.3
d. Black female (age adjusted per 100,000). . 1987
§
3.3 2.7 2.6 2.3 2.6 2.4 2.4 2.1 2.0 1.8 - - - 2.6
e. Residential fire deaths caused by
smoking.......................... 1987
§
20% 19% 16% 18% 16% 16% 17% 17% - - - - - - - - - 8%
f. American Indian/Alaska Native (age
adjusted per 100,000) ............... 1990 2.1 . . . 2.3 2.5 2.5 3.1 3.1 1.9 2.0 1.8 - - - 1.4
g. Puerto Rican (age adjusted per 100,000). . 1990
§
1.8 . . . 1.4 1.4 1.0 1.1 1.3 1.6 1.2 1.0 - - - 2.0
9.7 Hip fractures among adults 65 years and
over (per 100,000) .................... 1988 714 776 814 757 841 815 818 934 879 863 - - - 607
a. White female 85 years and over ........ 1988 2,721 3,075 3,091 2,368 3,035 2,815 2,604 2,804 2,879 2,690 - - - 2,177
9.8 Nonfatal poisoning (per 100,000) ......... 1986
§
104 68 64 61 52 43 43 41 41 38 33 88
a. Children 4 years and under (per 100,000) . . 1986
§
664 705 638 626 597 518 499 470 460 450 418 520
9.9 Nonfatal head injuries (per 100,000) ....... 1988 118 110 104 92 90 84 87 79 75 61 - - - 106
9.10 Nonfatal spinal cord injuries (per 100,000). . 1988 5.3 4.4 6.4 3.6 4.7 3.9 4.6 4.8 4.8 4.5 - - - 5.0
a. Male............................. 1988 9.6 6.9 9.8 4.8 6.7 7.1 6.9 6.5 6.1 7.4 - - - 7.1
9.11 Incidence of secondary conditions
associated with traumatic spinal cord
injuries............................. ... --- --- --- --- --- --- --- --- --- --- ---
20%
reduction
9.12 Motor vehicle occupant protection
systems ............................ 1988 42% 49% 59% 62% 66% 67% 68% 68% 69% 69% 67% 85%
a. Children 4 years and under............ 1988 48% 50% 55% - - - 60% 60% - - - 61% - - - 92% - - - 70%
9.13 Helmet use by motorcyclists and bicyclists
Motorcyclists.......................... 1988 60% 60% 62% - - - - - - 63% - - - 64% 67% 67% - - - 80%
Bicyclists............................. 1988 8% - - - 18% - - - - - - - - - - - - - - - - - - - - - - - - 50%
9.14 Safety belt and helmet use laws
Number of States with safety belt laws
4
..... 1989 33 36 41 44 45 48 49 49 49 49 - - - 50
Number of States with motorcycle helmet use
laws
5
.............................. 1989 22 23 24 24 25 25 25 25 22 22 21 50
9.15 Number of States with handgun design to
protect children...................... 1989 0 0 - - - - - - - - - - - - - - - - - - - - - - - - - - - 50
9.16 Fire suppression sprinkler installation
codes (number of localities) ............. 1989 700 - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 2,000
Proportion of residential fires with suppression
equipment........................... ... ---
6
2.4% 2.5% 2.7% 2.6% - - - - - - - - - - - - - - - - - - . . .
9.17 Smoke detectors
In inhabited residential dwellings ........... 1985 81% 82% - - - - - - 80% - - - - - - - - - - - - - - - - - - 100%
At least one on each habitable floor....... ... --- --- --- --- 52% --- --- --- --- --- --- 100%
Proportion of people with at least one
detector ............................ ... ---
7
68.5% - - - - - - 87.6% 92.7% - - - - - - - - - - - - - - - . . .
At least one on each floor .............. ... --- --- --- --- 66% 87% --- --- --- 88% --- ...
See footnotes and key at end of table.
Healthy People 2000 Final Review 161

Table 9. Unintentional Injuries objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
9.18 Injury prevention instruction in required
course
Proportion of middle/junior and senior high
schools ............................. 1994 66.2% . . . . . . . . . . . . . . . - - - - - - - - - - - - - - - 50%
9.19* Protective equipment in sporting and
recreation events .................... ... --- --- --- --- --- --- --- --- --- --- --- 100%
National Collegiate Athletic Association
Football ............................ 1988 Required - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Hockey ............................ 1988 Required - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Lacrosse ........................... 1988 Required - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
High school football .................... 1988 Required - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Amateur boxing........................ 1988 Required - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Amateur ice hockey .................... 1988 Required - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Use of protective headgear and mouth guards
among children who play sports
Baseball/softball
Headgear......................... ... --- --- 35% --- --- --- --- --- --- 47% --- ...
Mouth guard ...................... ... --- --- 7% --- --- --- --- --- --- 12% --- ...
Football
Headgear......................... ... --- --- 72% --- --- --- --- --- --- 77% --- ...
Mouth guard ...................... ... --- --- 72% --- --- --- --- --- --- 76% --- ...
Soccer
Headgear......................... ... --- --- 4% --- --- --- --- --- --- 9% --- ...
Mouth guard ...................... ... --- --- 7% --- --- --- --- --- --- 14% --- ...
9.20 Number of States with design standards for
roadway safety ...................... ... --- --- --- --- --- --- --- --- --- --- --- 50
9.21 Injury prevention counseling by primary
care providers ....................... ... --- --- --- --- --- --- --- --- --- --- --- 50%
Percent of clinicians routinely providing service
to 81–100% of patients
Inquiry about seat belt/child seat use
Pediatricians ........................ 1992 45% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 50%
Nurse practitioners ................... 1992 29% . . . . . . . . . - - - - - - - - - - - - - - -
8,10
39% - - - 50%
Obstetricians/gynecologists ............. 1992 6% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 50%
Internists ........................... 1992 11% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 50%
Family physicians .................... 1992 16% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 50%
Inquiry about hazards for falls in the home
(65 years and over)
Nurse practitioners ................... 1992 15% . . . . . . . . . - - - - - - - - - - - - - - -
8
14% - - - 50%
Internists ........................... 1992 10% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 50%
Family physicians .................... 1992 7% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 50%
See footnotes and key at end of table.
162 Healthy People 2000 Final Review

Table 9. Unintentional Injuries objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Advice about seat belt/child seat use
Pediatricians ........................ 1992 58% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 50%
Nurse practitioners ................... 1992 32% . . . . . . . . . - - - - - - - - - - - - - - -
8,11
30% - - - 50%
Obstetricians/gynecologists ............. 1992 18% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 50%
Internists ........................... 1992 15% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 50%
Family physicians .................... 1992 29% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 50%
Advice about prevention of falls in the home
(65 years and over)
Nurse practitioners ................... 1992 17% . . . . . . . . . - - - - - - - - - - - - - - -
8
18% - - - 50%
Internists ........................... 1992 17% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 50%
Family physicians .................... 1992 15% . . . . . . . . . - - - - - - - - - - - - - - -
8,9
--- --- 50%
9.22 Number of States with linked emergency
medical services and trauma systems .... 1993 7 . . . . . . . . . . . . - - - - - - - - - - - - 21 - - - 20
9.23* Alcohol-related motor vehicle deaths
(per 100,000) ........................ 1987 9.8 8.9 7.9 7.0 6.8 6.4 6.6 6.5 6.1 5.9 5.8 5.5
a. American Indian/Alaska Native male ..... 1987 40.4 34.3 32.2 31.4 26.8 28.0 - - - - - - - - - - - - - - - 35.0
b. People 15–24 years ................. 1987
§
20.9 18.6 17.2 14.2 13.7 13.0 12.8 12.9 11.7 11.7 11.5 12.5
9.24 Number of States with bicycle helmet
laws ............................... 1994 9 . . . . . . . . . . . . . . . 13 14 15 15 - - - 50
9.25* Number of States with firearm storage
laws ............................... 1989
§
1 1 5 8 11 13 15 15 15 15 18 50
9.26 Number of States with graduated licensing
systems ............................ 1993
§
0 ... ... ... ... --- --- --- 11 --- 23 35
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
1
Excludes data from States lacking Hispanic-origin item on their death certificates or for which Hispanic-origin data were not of sufficient quality. See appendix.
2
Data include intentional and unintentional injuries and injuries where the intent was not known.
3
Includes all deaths due to fires and flames regardless of location.
4
The District of Columbia, Puerto Rico, and other U.S. possessions also have a safety belt law.
5
The District of Columbia and Puerto Rico also have a motorcycle helmet law.
6
1989 data.
7
1985 data.
8
1997–98 data.
9
Response rate for this group was too low to produce reliable estimates.
10
Data represent the proportion of nurse practitioners who inquired about child safety seat use only. Seventeen percent of the nurse practitioners inquired about seat belt use.
11
Data represent the proportion of nurse practitioners who provided advice about child safety seat use only. Data on advice on seat-belt use were not available.
NOTE: Data include revisions and, therefore, may differ from data previously published in these reports and other publications.
Objective number Data source
9.1, 9.1a–d National Vital Statistics System, CDC, NCHS.
9.2 National Hospital Discharge Survey, CDC, NCHS.
9.3, 9.3a–c, e, f Fatality Analysis Reporting System, DOT, NHTSA.
9.3d, g National Vital Statistics System, CDC, NCHS.
Met
Toward
Mixed/ no change
Away
Cannot assess
Healthy People 2000 Final Review 163

Objective number Data source
9.4, 9.4a–d National Vital Statistics System, CDC, NCHS.
9.5, 9.5a–d National Vital Statistics System, CDC, NCHS.
9.6, 9.6a–d, f, g National Vital Statistics System, CDC, NCHS.
9.6e National Fire Incident Reporting System, FEMA, U.S. Fire Administration.
9.7, 9.7a National Hospital Discharge Survey, CDC, NCHS.
9.8, 9.8a National Electronic Injury Surveillance System, CPSC, Directorate for Epidemiology.
9.9 National Hospital Discharge Survey, CDC, NCHS.
9.10, 9.10a National Hospital Discharge Survey, CDC, NCHS.
9.12 Baseline and 1989–91 updates: 19 Cities Survey, DOT, NHTSA.
1992–97 updates: Population Weighted State Surveys, DOT, NHTSA.
1998–99 updates: National Occupant Protection Use Survey, DOT, NHTSA.
9.12a Baseline: 19 Cities Survey, DOT, NHTSA.
1992–93 updates: Population Weighted State Surveys, DOT, NHTSA.
1994–98 updates: National Occupant Protection Use Survey, DOT, NHTSA.
9.13 Baseline: 19 Cities Survey, DOT, NHTSA.
Updates: National Occupant Protection Use Survey , DOT, NHTSA.
9.14 DOT, NHTSA.
9.15 Telephone Survey on Handgun Laws, CDC, NCIPC.
9.16 Baseline (localities): Fire Suppression Sprinkler Codes, FEMA, U.S. Fire Administration.
1990–93 data: National Fire Incident Reporting System, FEMA, U.S. Fire Administration.
9.17 Baseline and 1990–93 updates: Prevention Index, Rodale Press for Inhabited residential dwellings.
1993 data: Smoke Detector Operability Survey, Consumer Product Safety Commission for inhabited residential dwellings on each habitable floor.
Proportion of people with smoke detectors: National Health Interview Survey, CDC, NCHS.
Proportion of people with one on each floor: National Health Interview Survey, CDC, NCHS.
9.18 School Health Policies and Programs Study, CDC, NCCDPHP.
9.19* 1988 baseline: CDC, NCPS; NIH, NIDR.
1991 data: National Health Interview Survey, CDC, NCHS.
9.21 Baseline: Primary Care Provider Surveys, OPHS, ODPHP.
Update: Prevention in Primary Care Study, American College of Preventive Medicine.
9.22 CDC, NCIPC.
9.23*, 9.23b Fatality Analysis Reporting System, DOT, NHTSA.
9.23a Fatality Analysis Reporting System, DOT, NHTSA; National Vital Statistics System, CDC, NCHS.
9.24 National SAFEKIDS Campaign.
9.25* Office of Planning, Evaluation and Legislation, CDC, NCIPC; National Conference of State Legislatures.
9.26 Insurance Institute for Highway Safety.
*Duplicate objective. See full text of objective following this table.
164 Healthy People 2000 Final Review
Unintentional
Injuries Objectives
9.1: Reduce deaths caused by
unintentional injuries to no more than
29.3 per 100,000 people.
9.1a: Reduce deaths among
American Indians and Alaska
Natives caused by unintentional
injuries to no more than 53.0 per
100,000 people.
9.1b: Reduce deaths among black
males caused by unintentional
injuries to no more than 51.9 per
100,000 people.
9.1c: Reduce deaths among white
males caused by unintentional
injuries to no more than 42.9 per
100,000.
9.1d: Reduce deaths among
Mexican–American males caused by
unintentional injuries to no more
than 43.0 per 100,000.
9.2: Reduce nonfatal unintentional
injuries so that hospitalizations for this
condition are no more than 754 per
100,000 people.
9.2a: Reduce nonfatal unintentional
injuries among black males so that
hospitalizations for this condition
are no more than 856 per 100,000
people.
9.3: Reduce deaths caused by motor
vehicle crashes to no more than 1.5 per
100 million vehicle miles traveled and
14.2 per 100,000 people.
9.3a: Reduce deaths among children
aged 14 and younger caused by
motor vehicle crashes to no more
than 4.4 per 100,000.
9.3b: Reduce deaths among youth
aged 15–24 caused by motor
vehicle crashes to no more than
26.8 per 100,000.
9.3c: Reduce deaths among people
aged 70 and older caused by motor
vehicle crashes to no more than 20
per 100,000.
9.3d: Reduce deaths among
American Indians and Alaska
Natives caused by motor vehicle
crashes to no more than 32 per
100,000.
9.3e: Reduce deaths among
motorcyclists caused by motor
vehicle crashes to no more than
25.6 per 100 million vehicle miles
traveled and 0.9 per 100,000.
9.3f: Reduce deaths among
pedestrians caused by motor vehicle
crashes to no more than 2.0 per
100,000.
9.3g: Reduce deaths among
Mexican-Americans caused by
motor vehicle crashes to no more
than 18 per 100,000.
9.4: Reduce deaths from falls and
fall-related injuries to no more than 2.3
per 100,000 people.
9.4a: Reduce deaths among people
aged 65–84 from falls and
fall-related injuries to no more than
14.4 per 100,000.
9.4b: Reduce deaths among people
aged 85 and older from falls and
fall-related injuries to no more than
105 per 100,000.
9.4c: Reduce deaths among black
men aged 30–69 from falls and
fall-related injuries to no more than
5.6 per 100,000.
9.4d: Reduce deaths among
American Indians and Alaska
Natives from falls and fall-related
injuries to no more than 2.8 per
100,000.
9.5: Reduce drowning deaths to no more
than 1.3 per 100,000 people.
9.5a: Reduce drowning deaths
among children aged 4 and younger
to no more than 2.3 per 100,000.
9.5b: Reduce drowning deaths
among men aged 15–34 to no more
than 2.5 per 100,000.
9.5c: Reduce drowning deaths
among black males to no more than
3.6 per 100,000.
9.5d: Reduce drowning deaths
among American Indians and
Alaska Natives to no more than 2.0
per 100,000.
9.6: Reduce residential fire deaths to no
more than 1.2 per 100,000 people.
9.6a: Reduce residential fire deaths
among children aged 4 and younger
to no more than 3.3 per 100,000.
9.6b: Reduce residential fire deaths
among people aged 65 and older to
no more than 3.3 per 100,000.
9.6c: Reduce residential fire deaths
among black males to no more than
4.3 per 100,000.
9.6d: Reduce residential fire deaths
among black females to no more
than 2.6 per 100,000.
9.6e: Reduce residential fire deaths
from residential fires caused by
smoking to no more than 8 percent.
9.6f: Reduce residential fire deaths
among American Indians and
Alaska Natives to no more than 1.4
per 100,000.
9.6g: Reduce residential fire deaths
among Puerto Ricans to no more
than 2.0 per 100,000.
9.7: Reduce hip fractures among people
aged 65 and older so that
hospitalizations for this condition are no
more than 607 per 100,000 people.
9.7a: Reduce hip fractures among
white women aged 85 and older so
that hospitalizations for this
condition are no more than 2,177
per 100,000.
9.8: Reduce nonfatal poisoning to no
more than 88 emergency department
treatments per 100,000 people.
9.8a: Reduce nonfatal poisoning
among children aged 4 and younger
to no more than 520 emergency
department treatments per 100,000.
9.9: Reduce nonfatal head injuries so
that hospitalizations for this condition
are no more than 106 per 100,000
people.
9.10: Reduce nonfatal spinal cord
injuries so that hospitalizations for this
condition are no more than 5 per
100,000 people.
9.10a: Reduce nonfatal spinal cord
injuries among males so that
hospitalizations for this condition
are no more than 7.1 per 100,000.
9.11: Reduce by 20 percent the
incidence of secondary conditions (i.e.,
pressure sores) associated with traumatic
spinal cord injuries.
9.12: Increase use of safety belts and
child safety seats to at least 85 percent
of motor vehicle occupants.
Healthy People 2000 Final Review 165
9.12a: Increase use of child restraint
systems among children aged 4 and
younger involved in potentially fatal
crashes to 70 percent.
9.13: Increase use of helmets to at least
80 percent of motorcyclists and at least
50 percent of bicyclists.
9.14: Extend to 50 States laws requiring
safety belt and motorcycle helmet use
for all ages.
9.15: Enact in 50 States laws requiring
that new handguns be designed to
minimize the likelihood of discharge by
children.
9.16: Extend to 2,000 local jurisdictions
the number whose codes address the
installation of fire suppression sprinkler
systems in those residences at highest
risk for fires.
9.17: Increase the presence of functional
smoke detectors to at least one on each
habitable floor of all inhabited
residential dwellings.
9.18: Provide academic instruction on
injury prevention and control, preferably
as part of comprehensive school health
education, in at least 50 percent of
public school systems (grades K–12).
9.19*: Extend requirement of the use of
effective head, face, eye, and mouth
protection to all organizations, agencies,
and institutions sponsoring sporting and
recreation events that pose risks of
injury.
Duplicate objective: 13.16
9.20: Increase to at least 50 the number
of States that have design standards for
markings, signing, and other
characteristics of the roadway
environment to improve the visual
stimuli and protect the safety of older
drivers and pedestrians.
9.21: Increase to at least 50 percent the
proportion of primary care providers
who routinely provide age–appropriate
counseling on safety precautions to
prevent unintentional injury.
9.22: Extend to 20 States the capability
to link emergency medical services,
trauma systems, and hospital data.
9.23*: Reduce deaths caused by
alcohol-related motor vehicle crashes to
no more than 5.5 per 100,000 people.
Duplicate objective: 4.1
9.23a*: Reduce deaths among
American Indian and Alaska Native
men caused by alcohol-related
motor vehicle crashes to no more
than 35.0 per 100,000.
Duplicate objective: 4.1a
9.23b*: Reduce deaths among
people aged 15–24 caused by
alcohol-related motor vehicle
crashes to no more than 12.5 per
100,000.
Duplicate objective: 4.1b
9.24: Extend to 50 States laws requiring
helmets for bicycle riders.
9.25*: Enact in 50 States and the
District of Columbia laws requiring that
firearms be properly stored to minimize
access and the likelihood of discharge
by minors.
Duplicate objective: 7.19
9.26: Increase to 35 the number of
States having a graduated driver
licensing system for novice drivers and
riders under the age of 18.
*Duplicate objective.
166 Healthy People 2000 Final Review

Priority Area 10
Occupational Safety
and Health
Background
Workplace injuries and illness
continue to place an enormous burden
on the Nation’s workers and economy.
Each day, an average of 137 persons die
from work-related illness (1,2) and an
additional 17 die from work-related
injuries (3). Each year, about 70
adolescent workers 17 years of age and
under die from work-related injuries (4).
In 1992, the estimated economic burden
for occupational illnesses and injuries
was $171 billion (5), while in 1998, the
estimated societal cost of occupational
injuries alone was $125.1 billion (6).
In 1999, highway crashes continued
to be the leading cause of occupational
death, accounting for 25 percent of
work-related fatalities (3). During the
same year and for the first time in the
decade, an increase in falls and a
simultaneous decline in homicides
resulted in falls becoming the second
and homicides the third leading cause of
death in the workplace (3). The highest
occupational fatality rates are observed
in mining, agriculture, forestry and
fishing, and construction (3).
Data Summary
Highlights
Since the development of Healthy
People 2000, the Nation has experienced
marked success in meeting the targets in
several areas of occupational safety and
health. The rate of work-related
homicides per 100,000 workers (10.16)
met its target level of 0.5 in 1998. The
number of workers who became infected
with hepatitis B virus through
occupational exposure (10.5) was
reduced to 243 cases in 1999, well
beyond the target level of 623 cases.
Both the percent of worksites with
safety and health programs (10.12) and
the percent of worksites with back
injury prevention and rehabilitation
programs (10.13) exceeded their target
by 1999. In addition, the age-adjusted
fatality rate for occupational lung
disease per million workers 15 years of
age and older (10.17) was reduced to
6.6 in 1998, beyond the target rate of
7.7.
Occupational lung disease exposure
standards (10.11) are applicable in all 50
States and the District of Columbia, the
target level, up from 15 in 1970. The
number of States with programs for
small business safety and health (10.14)
also increased to the target level of 50
States and the District of Columbia.
In other areas, progress toward the
Healthy People 2000 targets was made.
The rate of work-related injury deaths
per 100,000 workers (10.1) decreased to
4.5 in 1999 from the 1983–87 baseline
average rate of 6, the rate of nonfatal
work-related injuries per 100,000
workers (10.2) was 6.2 in 1998, down
from 7.7 in 1983–87, and the percent of
businesses that ban smoking (10.18)
increased from 27 percent in 1985 to
79 percent in 1999. The target for this
objective (10.18) was raised from
75 percent to 100 percent at the
midcourse review.
Moving substantially in the other
direction, that is, away from their
targets, were cumulative trauma
disorders (10.3), worksite occupant
protection systems (10.6), occupational
noise exposure (10.7), and occupational
lead exposure (10.8). For objective 10.8
the number of States reporting has
increased over the last decade, which
makes the raw number of cases reported
understate the progress made; the rate of
cases per million population has steadily
decreased since 1995.
Summary of progress
Of the 20 occupational safety and
health objectives, 7 (10.5, 10.11–10.14,
10.16, and 10.17) have achieved their
targets. Seven objectives (10.1, 10.2,
10.4, 10.9, 10.10, 10.15, and 10.18)
moved toward the year 2000 targets.
Progress for objective 10.15 was toward
the year 2000 target, but is based on
limited data from nurse practitioners
only (see Data Issues). Four objectives
(10.3, and 10.6–10.8) moved away from
the targets. Progress for the compound
objective (10.19) was mixed. One
objective (10.20) showed no change
from the baseline level. See table 10 for
the tracking data for the objectives in
this priority area and figure 10 for a
quantitative assessment of progress.
Discussion
The barriers to achieving the
Healthy People 2000 objectives vary;
however, there are a number of common
themes. Numerous
conditions—including hepatitis B,
occupational lead exposure, and
occupational injuries—are preventable,
but the lack of public awareness of
prevention measures or the view that
these conditions are an ‘‘acceptable
risk’’ of employment interfere with basic
prevention. Gaps in scientific knowledge
also present a barrier to achieving the
objectives. For instance, in the case of
cumulative trauma and other
musculoskeletal disorders, limitations in
the amount of technological and
scientific information inhibit precise
assessment of risks in some jobs with
complex and multiple exposures to
several risk factors. Difficulties in
systematically evaluating the
effectiveness of interventions also
impeded progress toward the Healthy
People 2000 targets.
In addition, there is a lack of
cost-benefit studies in occupational
safety and health that would help
motivate companies to institute and
maintain occupational safety and health
programs, such as back injury and
hearing loss prevention programs.
Moreover, difficulties in diagnosing and
establishing the work relatedness of
some conditions, such as occupational
skin disorders/diseases and low back
injuries, also serve as a barrier to
progress.
The national occupational safety
and health community is pursuing a
number of strategies to address these
and other barriers. Such efforts include
health education and communication
campaigns, academic training, research
on intervention effectiveness and health
services, and surveillance strategic
planning. In addition, the National
Institute for Occupational Safety and
Health (NIOSH) is working with
partners to track occupational injury and
illness disparities among population
subgroups. In all of these areas,
partnerships among industry, labor,
government, academia, and professional
organizations are enhancing the Nation’s
ability to target resources and expertise.
Also addressing the barriers to
achieving the Healthy People 2000
objectives is the National Occupational
Research Agenda (NORA), a framework
to guide occupational safety and health
research into the 21st century.
Approximately 500 organizations and
individuals provided input into the
development of the agenda. The NORA
process resulted in a consensus about a
Healthy People 2000 Final Review 167
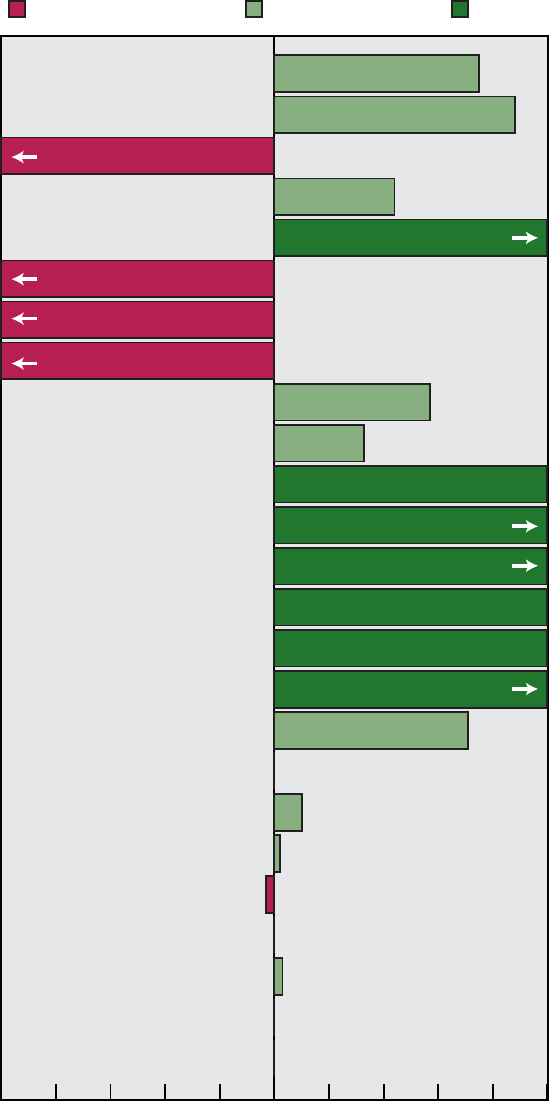
10.1 Work-related injury deaths
10.2 Nonfatal work-related injuries
10.3 Cumulative trauma disorders
10.4 Occupational skin disorders
10.5 Hepatitis B infection among exposed workers
10.6 Worksite occupant protection systems
10.7 Occupational noise exposure
10.8 Occupational lead exposure
10.9 Hepatitis B immunization among exposed workers
10.10 States with occupational health and safety plans
10.11 States with occupational lung disease standards
10.14 States with small business safety/health programs
10.12 Worksite health and safety programs
10.13 Worksite back injury prevention programs
10.16 Work-related homicides
10.17 Occupational lung disease deaths
10.18 Worksite smoking policies: Bans
10.19 States with clean indoor air laws: Private workplaces
Restaurants
Public transportation
Public workplaces
Hospitals
Day care centers
Grocery stores
10.20 States with preemptive clean indoor air laws
Moved away from target Moved toward target Met target
NOTE: Complete tracking data are shown in table 10. Progress quotients are not calculated for objective 10.15.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
44%
75%
-3%
33%
57%
71%
10%
2%
No change
No change
No change
No change
3%
Percent of target achieved
-100 -80 -60 -40 -20
0
20 40 60 80 100
115%
342%
114%
100%
100%
138%
100%
-390%
-281%
-463%
-104%
88%
168 Healthy People 2000 Final Review
Figure 10. Final status of Occupational Safety and Health objectives
set of top 21 research priorities. The
priority areas are organized under the
more general categories of Disease and
Injury, Work Environment and
Workforce, and Research Tools and
Approaches. Because of the significant
overlap between the NORA research
priority areas and the Healthy People
2000 objectives, NORA was
instrumental in moving the Nation
toward the year 2000 targets and will
likewise serve as a catalyst for
advancing the objectives established in
Healthy People 2010 (7) over the next
decade.
Transition to Healthy People
2010
Many of the Healthy People 2000
occupational safety and health objectives
are included in the new set of objectives
for Healthy People 2010, with baselines
and targets adjusted to reflect gains
made, as well as changes in data
systems. Several new objectives have
been added to address work organization
issues, health care worker hazards, and
engineering interventions in agriculture.
In addition, several Healthy People 2000
objectives that cannot be tracked
reliably or that have low relative value
for monitoring improved outcomes in
worker safety and health have been
revised, replaced, or dropped from the
Healthy People 2010 objectives.
Objectives on work-related injury
deaths and work-related injuries are
continued in Healthy People 2010 and
are addressed by the NORA priority
area of Traumatic Injuries. Also, the
objective on overexertion or repetitive
motion which is addressed by NORA’s
Low Back Disorders and
Musculoskeletal Disorders of the Upper
Extremities and the objective on
occupational skin diseases or disorders,
addressed by NORA’s Allergic and
Irritant Dermatitis, are included in the
next decade. A number of other health
outcomes are continued in Healthy
People 2010 including: work-related
noise-induced hearing loss, addressed by
NORA’s Hearing Loss; elevated blood
lead levels from work exposure,
addressed by NORA’s Fertility and
Pregnancy Abnormalities; work-related
homicides and work-related assaults,
addressed by NORA’s Traumatic
Injuries; and, pneumoconiosis deaths,
addressed by NORA’s Asthma and
Chronic Obstructive Pulmonary
Diseases. Finally, two Healthy People
2010 objectives are new: worksite stress
reduction programs, addressed by
NORA’s Organization of Work; and
needlestick injuries, addressed by
NORA’s Infectious Diseases.
Seven of the Healthy People 2000
objectives were not included in Healthy
People 2010: worksite occupant
protection system mandates; State
occupational safety and health plans;
occupational lung disease exposure
standards; worksite safety and health
programs; worksite injury prevention
and rehabilitation programs; State
programs for small business safety and
health; and clinician assessment of
occupational health exposures.
Healthy People 2000 objectives on
hepatitis B infections and hepatitis B
immunizations are addressed by Healthy
People 2010 objectives in the
Immunization and Infectious Diseases
chapter. In addition, Healthy People
2000 objectives on worksites with
smoking policies, States with
comprehensive laws for clean indoor air,
and preemptive clean indoor air laws are
addressed by Healthy People 2010
objectives in the Tobacco Use chapter.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the changes between the
two decades of objectives, reflecting
new knowledge and direction for this
area.
Data Issues
Definitions
Objective 10.20 seeks to reduce the
number of States with preemptive clean
indoor air laws. Preemptive laws prevent
local jurisdictions from enacting more
stringent restrictions than the State law
or restrictions that vary from the State
law (8).
Data Sources
Since 1992, the data for objective
10.1 (work-related injury deaths) have
come from the Census for Fatal
Occupational Injuries (CFOI), Bureau of
Labor Statistics (BLS). Prior to 1992,
the data came from the Annual Survey
on Occupational Injuries and Illnesses
(ASOII). ASOII relied on a single data
source to capture occupational fatalities:
a survey of employer logs of
occupational deaths in approximately
50,000 workplaces. The survey
undercounted occupational fatalities by
as much as 60 percent (9). The CFOI
uses a minimum of two data sources to
identify occupational deaths. The
primary sources are death certificates;
State workers’ compensation reports;
coroner, medical examiner, or autopsy
reports; and Occupational Safety and
Health Administration (OSHA) reports.
The rates for 1993 were rounded to
whole numbers by BLS. National
Traumatic Occupational Fatalities
Surveillance System (NTOF) data
(reported by NIOSH) can also be used
to monitor this objective, but NTOF
uses only death certificates and may
underestimate some categories of
work-related injury deaths. CFOI and
NTOF are also used to track objective
10.16 on workplace homicides.
The subobjective on adolescent
work injuries (10.2f) is tracked with
data from the National Electronic Injury
Surveillance System (NEISS) under an
interagency agreement between NIOSH
and the Consumer Product Safety
Commission (CPSC) and does not
utilize ICD–9 codes or other
conventional injury reporting
mechanisms. The data are collected in
hospital emergency rooms and are
limited to injuries attributable to a
specific list of regulated products and
devices. Hence, the data collected are
subject to annual variations in what is
specified in product safety or regulatory
codes. The baseline for adolescent
worker injuries is an extrapolation of
data from the last 6 months of calendar
year 1992 and is limited to workers ages
15–17 years. The updates for 1996 and
1997 are for fiscal years; the 1998 data
are for the calendar year. The main
objective and the other subobjectives for
10.2 are tracked using data from ASOII.
The data used to report on the
status of objective 10.7 (occupational
noise exposure) come from the U.S. Air
Force Hearing Conservation Database.
The data report exposures for civilian
and military employees in a wide range
of industrial and service occupations.
NIOSH is currently developing the
Sentinel Event Notification System for
Occupational Risk (SENSOR) and the
Occupational Hearing and Conservation
database and has issued guidelines and
sponsored workshops designed to
address this important occupational
health issue.
The data for objective 10.8
(occupational lead exposure) are from
Healthy People 2000 Final Review 169
State registries that report adult blood
lead levels. These State data are
compiled by NIOSH in the Adult Blood
Lead Epidemiology Survey (ABLES).
The number of reporting States has
increased since the baseline was
established; this increase has affected
the number of cases reported; in fiscal
year 1999, 25 States were reporting.
Objective 10.11 (State exposure
standards for occupational lung disease)
was achieved because Federal standards
applicable in all 50 States and the
District of Columbia were established
for airborne asbestos fibers, cotton dust,
coal mine dust, and silica dust. The
parallel objective 10.17 (pneumoconiosis
deaths) is tracked with the number of
deaths as reported in the National Vital
Statistics System (NVSS).
The 1985, 1987, and 1992 data for
objectives 10.12 (worksite safety and
health programs), 10.13 (worksite back
injury prevention), and 10.18 (worksite
smoking policies) are from the National
Surveys of Worksite Health Promotion
Activities, which were telephone surveys
of nongovernment worksites of 50 or
more employees. Worksites were
sampled instead of companies, because
different worksites within the same
company could have different sets of
health promotion activities. Both active
(for example, classes) and passive (for
example, brochures) methods were
counted as worksite health promotion
activities. The 1995 updates were
measured by the Centers for Disease
Control and Prevention
(CDC)-sponsored Worksite Benchmark
Survey, which used a methodology very
similar to the 1992 survey, but did not
include passive methods of health
promotion (10,11). The 1998–99 data
for 10.13 and 10.18 also came from the
National Worksite Health Promotion
Survey. Like the 1992 survey, the
designated respondent was asked if the
worksite had specific policies or
prevention activities (12).
Baseline data for objective 10.15
(screening for occupational health
exposure) are from the Primary Care
Provider Surveys (PCPS). The PCPS
sample was drawn from the membership
rolls of professional organizations for
pediatricians, nurse practitioners, family
physicians, obstetricians/gynecologists,
and internists. Response rates varied
from 50 to 80 percent across these
groups. The data on inquiry about
work-related risks represent the
proportion of providers who routinely
queried 81–100 percent of their patients
about these risks. The data on
counseling refer to the proportion of
providers who routinely provided these
services to patients who needed the
services. The Prevention in Primary
Care Study (PPCS) was conducted in
1997–98 to update data from the PCPS.
The design and items included in the
1997–98 study were similar to the
PCPS, but a slightly different sampling
frame was used and some items
included in the 1992 surveys were not
included in the PPCS. The professionals
were sampled from listings of all
licensed, active practitioners in the
United States whose practices were at
least 50 percent primary care. Because
of low response rates from the other
provider groups, updates are available
only for nurse practitioners.
Data Comparability
The baseline data for objective 10.9
(hepatitis immunizations) were collected
by OSHA’s Regulatory Impact Analysis;
the updates are from CDC’s National
Center for Infectious Diseases. The
baseline for objective 10.10 (State
occupational health and safety plans)
came from the Public Health
Foundation’s unintentional injuries
survey; the updates are from OSHA. For
both objectives, the data may not be
comparable and statements about trends
must be made with caution. It should
also be noted that all updates for
objective 10.16 are from the Bureau of
Labor Statistics’ CFOI and the baseline
data were from NIOSH.
References
1. Centers for Disease Control and Prevention.
National occupational research agenda. MMWR
45:445–6. 1996.
2. Centers for Disease Control and Prevention.
Clarification. MMWR 45:495. 1996.
3. Bureau of Labor Statistics. Census of fatal
occupational injuries summary.
http://stats.bls.gov/news.release/cfoi.nr0.htm. Aug.
17, 2000.
4. National Institute for Occupational Safety and
Health. National Traumatic Occupational Fatalities
Surveillance System. Morgantown, West Virginia:
National Institute for Occupational Safety and
Health. 1999.
5. Leigh JP, Markowitz SB, Fahs M, et al.
Occupational injury and illness in the United
States: Estimates of costs, morbidity, and
mortality. Arch Int Med 157(14):1557–68. 1997.
6. National Safety Council. Accident Facts, 1999.
Itasca, Illinois: National Safety Council. 2000.
7. Department of Health and Human Services.
Healthy People 2010. 2d ed. With Understanding
and Improving Health. 2 vols. Washington: U.S.
Government Printing Office. 2000.
8. Centers for Disease Control and Prevention.
State laws on tobacco control: United States, 1995.
MMWR 44(SS–6):24. 1995.
9. Toscano G, Windau J. Fatal work injuries:
Census for 31 States. Monthly Labor Review:3–8.
1992.
10. Department of Health and Human Services.
Business Responds to AIDS Benchmark Survey:
Technical report. Atlanta, Georgia: Public Health
Service, Centers for Disease Control and
Prevention. 1996.
11. Centers for Disease Control and Prevention.
Cancer screening offered by worksites—United
States, 1992 and 1995. MMWR 46(19):421–4.
1997.
12. Department of Health and Human Services.
Tracking Healthy People 2010. Washington, DC:
U.S. Government Printing Office. 2000.
170 Healthy People 2000 Final Review

Table 10. Occupational Safety and Health objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
10.1 Work-related injury deaths (per 100,000
workers) ........................ 1983-87 6.0 4.3 4.3 5.0 5.0 5.0 4.9 4.8 4.8 4.5 4.5 4.0
a. Mine workers .................. 1983-87 30.3
1
17.3 15.6 27.0 26.0 27.0 25.0 26.8 25 23.6 21.5 21
b. Construction workers ............ 1983-87 25.0 20.6 16.6 14.0 14.0 15.0 14.6 13.9 14.1 14.6 14.0 17
c. Transportation workers ........... 1983-87 15.2 10.0 8.1 13.0 13.0 14 13.1 13.1 13.2 11.8 12.6 10
d. Farm workers .................. 1983-87 14.0 23.8 - - - 24.0 26.0 24 22.5 22.8 23.9 24.1 23.6 9.5
10.2 Nonfatal work-related injuries (per 100
full-time workers
2
................. 1983-87 7.7 8.3 7.9 8.3 7.9 7.7 7.5 6.9 6.6 6.2 - - - 6
a. Construction workers
2
............ 1983-87 14.9 14.1 12.8 12.9 12.0 11.5 10.4 9.7 9.3 8.7 - - - 10
b. Nursing and personal care workers
2
. 1983-87 12.7 15.4 15.0 18.2 16.9 16.5 17.8 16.2 15.9 13.8 - - - 9
c. Farm workers
2
................. 1983-87 12.4 12.3 11.1 11.5 10.9 9.7 9.9 8.9 8.7 8.0 - - - 8
d. Transportation workers
2
.......... 1983-87 8.3 9.4 9.1 8.8 9.1 9.0 8.7 8.4 7.9 7.0 - - - 6
e. Mine workers
2
................. 1983-87 8.3 8.1 7.1 7.0 6.5 6.0 6.0 5.3 5.7 4.7 - - - 6
f. Adolescent workers (15–17 years)
3
. . 1992 5.8 . . . . . . . . . - - - - - - - - - 4.8 4.8
4
4.9 - - - 3.8
10.3 Cumulative trauma disorders (per
100,000 full-time workers) ........... 1987 100 241 297 368 383 411 378 335 320 285 - - - 60
a. Manufacturing industry workers..... 1987 355 867 1,046 1,241 1,267 1,362 1,258 1,104 1,061 960 - - - 150
b. Meat product workers ............ 1987 3,920 8,245 8,802 8,475 8,532 8,750 7,720 6,116 6,860 5,979 - - - 2,000
10.4 Occupational skin disorders (per
100,000 full-time workers) ........... 1983-87 64 79 77 82 76 81 79 69 67 60 - - - 55
10.5* Hepatitis B infections among
occupationally exposed workers
(number of cases)................. 1987 3,090 1,258 2,576 1,923 727 506 407 391 383 377 243 623
10.6 Worksite occupant protection system
mandates....................... 1992 82.4% . . . . . . . . . - - - - - - 85% - - - - - - - - -
5
47% 95%
10.7 Occupational noise exposure
6
(average
noise levels exceeding 85 db) ........ 1989 16% 20.5% 23.8% 21.5% 19.9% - - - - - - - - - - - - - - - - - - 15%
10.8 Occupational lead exposure (blood
concentration greater than 25 µg/dL). . . 1988
7
4,804
8
4,531 - - -
9
8,886
10
9,571
11
11,068
11
11,660
12
12,706
13
12,688
14
10,501
14
9,790 0
10.9* Hepatitis B immunizations among
occupationally exposed workers
15
. . . 1989 37% - - - - - - 50% - - - 67% - - - - - - - - - - - - - - - 90%
10.10 Number of States with occupational
health and safety plans
16
.......... 1989 10 - - - - - - 23 - - - - - - - - - - - - - - - - - - 23 51
10.11 Number of States with occupational
lung disease exposure standards
16
. . 1970
17
51 51 51 51 51 51 51 51 51 51 51 51
10.12 Worksite health and safety programs . 1992 63.8% . . . . . . . . . - - - - - - 85% - - - - - - - - - - - - 70%
10.13 Worksite back injury prevention and
rehabilitation programs ........... 1985 28.6% - - - - - - 32.5% - - - - - - - - - - - - - - - - - -
5
53% 50%
Back injury classes, workshops or
lectures......................... ... --- ... ... 24% --- --- 26% --- --- --- --- ...
10.14 Number of States with programs for
small business safety and health
16
. . 1991 26 . . . . . . - - - 51 51 51 51 51 51 - - - 51
10.15 Clinician assessment of occupational
health exposures ................ ... --- --- --- --- --- --- --- --- --- --- --- 75%
See footnotes and key at end of table.
Healthy People 2000 Final Review 171

Table 10. Occupational Safety and Health objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Percent of clinicians routinely providing
service to 81–100% of patients
Inquiry about work-related health risks
(16 years and over)
Pediatricians .................... 1992 7% . . . . . . . . . - - - - - - - - - - - - - - -
18,19
--- --- 75%
Nurse practitioners ............... 1992 14% . . . . . . . . . - - - - - - - - - - - - - - -
18
14% - - - 75%
Obstetricians/gynecologists ......... 1992 6% . . . . . . . . . - - - - - - - - - - - - - - -
18,19
--- --- 75%
Internists ....................... 1992 14% . . . . . . . . . - - - - - - - - - - - - - - -
18,19
--- --- 75%
Family physicians ................ 1992 7% . . . . . . . . . - - - - - - - - - - - - - - -
18,19
--- --- 75%
Counseling about work-related health
risks
Pediatricians .................... 1992 8% . . . . . . . . . - - - - - - - - - - - - - - -
18,19
--- --- 75%
Nurse practitioners ............... 1992 10% . . . . . . . . . - - - - - - - - - - - - - - -
18
12% - - - 75%
Obstetricians/gynecologists ......... 1992 10% . . . . . . . . . - - - - - - - - - - - - - - -
18,19
--- --- 75%
Internists ....................... 1992 9% . . . . . . . . . - - - - - - - - - - - - - - -
18,19
--- --- 75%
Family physicians ................ 1992 8% . . . . . . . . . - - - - - - - - - - - - - - -
18,19
--- --- 75%
10.16 Work-related homicides (per 100,000
workers) ........................ 1980-89 0.7 - - - - - - 0.9 0.8 0.9 0.8 0.7 0.6 0.5 0.5 0.5
10.17 Occupational lung disease
(pneumoconiosis) deaths among
people 15 years and over (age
adjusted per million) ............... 1990
§
10.6 . . . 9.8 8.8 8.7 8.1 8.2 7.8 7.0 6.6 - - - 7.7
Number of pneumoconiosis deaths among
people 15 years and over ........... ... --- 3,644 3,486 3,230 3,238 3,126 3,151 3,114 2,928 2,790 - - - 3,000
10.18* Worksites with smoking policies
Policy that bans smoking or limits it to
separately ventilated areas 50 or more
employees ...................... 1985 27% - - - - - - 59% - - - - - - - - - - - - - - - - - -
5
79% 100%
Any smoking policy
Medium and large companies ......... 1987 54% - - - 85% - - - - - - - - - - - - - - - - - - - - - - - - 100%
50 or more employees .............. ... --- --- --- 86% --- --- 87% --- --- --- --- ...
10.19* Number of States with comprehensive
laws for clean indoor air
16
Private workplaces ............... 1995
§
1 ... ... ... ... ... ... 1 1 1 --- 51
Public workplaces ................ 1995
§
9 ... ... ... ... ... ... 9 12 13 --- 51
Restaurants..................... 1995 2 . . . . . . . . . . . . . . . . . . 3 3 3 - - - 51
Public transportation .............. 1995
§
17 ... ... ... ... ... ... 17 17 16 --- 51
Hospitals ....................... 1995
§
8 ... ... ... ... ... ... 8 8 --- --- 51
Day care centers ................. 1995 21 . . . . . . . . . . . . . . . . . . 21 21 22 - - - 51
Grocery stores................... 1995
§
4 ... ... ... ... ... ... 4 4 4 --- 51
10.20* Preemptive clean indoor air laws
States with laws ................. 1995 17 . . . . . . . . . . . . . . . . . . 17 17 17 17 0
- - - Data not available. Final objective status:
. . . Category not applicable.
Met
Toward
Mixed/ no change
Away
Cannot assess
172 Healthy People 2000 Final Review

§
Baseline has been revised.
1
1989 data.
2
Data include work-related injuries and illnesses.
3
Data are for adolescents age 15–17 who sought medical treatment in an emergency room and are reported by fiscal year.
4
Data are for the calendar year 1998.
5
1998–99 data.
6
Data represent a cross-section of civilian and military employees.
7
Data are from 7 States.
8
Data are from 13 States.
9
Data are from 18 States.
10
Data are from 17 States.
11
Data are from 22 States.
12
Data are from 24 States.
13
Data are from 27 States.
14
Data are from 25 States.
15
Health care workers only.
16
Includes the District of Columbia.
17
Pursuant to the enactment of the Federal Coal Mine Health and Safety Act of 1969 (PL91–173, amended by PL95–164) and the Occupational Safety and Health Act of 1970 (PL91–596),
Federal Standards have been established for occupational exposure to airborne asbestos fibers, cotton dust, coal mine dust, and silica dust. These exposure limits apply in all 50 States and
U.S. Territories.
18
1997–98 data.
19
Response rate for this group was too low to produce reliable estimates.
NOTE: Data include revisions and, therefore, may differ from data previously published in these reports and other publications.
Objective number Data source
10.1, 10.1a-d Baseline and 1990–91 updates: Annual Survey of Occupational Injuries and Illnesses, DOL, BLS.
1992–99 updates: Census of Fatal Occupational Injuries, DOL, BLS.
10.2, 10.2a-e Annual Survey of Occupational Injuries and Illnesses, DOL, BLS.
10.2f National Electronic Injury Surveillance System, CPSC.
10.3, 10.3a-b Annual Survey of Occupational Injuries and Illnesses, DOL, BLS.
10.4 Annual Survey of Occupational Injuries and Illnesses, DOL, BLS.
10.5* National Notifiable Disease Surveillance System, CDC, EPO.
Sentinel Counties Study of Acute Viral Hepatitis, Viral Hepatitis Surveillance Program, CDC, NCID.
10.6 Baseline: National Survey of Worksite Health Promotion Activities, OPHS, ODPHP. 1995 Update: Business Responds to HIV/AIDS Benchmark Survey,
CDC, NCHSTP; 1998–99 Update: National Worksite Health Promotion Survey, OPHS, ODPHP.
10.7 U.S. Air Force Hearing Conservation Database, DoD.
10.8 Adult Elevated Blood Lead Level Registries, CDC, NIOSH.
10.9* Baseline: Regulatory Impact Analysis of OSHA Final Rule on Occupational Exposure to Bloodborne Pathogens, DOL, OSHA, ORA.
Updates: CDC, NCID.
10.10 Baseline: Association of State and Territorial Health Officials Reporting System: Unintentional Injuries Survey, PHF.
Updates: DOL, OSHA.
10.11 CDC, NIOSH.
10.12 Baseline: National Survey of Worksite Health Promotion Activities, OPHS, ODPHP.
1995 Update: Business Responds to HIV/AIDS Benchmark Survey, CDC, NCHSTP.
10.13 Baseline and 1992 updates: National Survey of Worksite Health Promotion Activities, OPHS, ODPHP.
1995 update: Business Responds to AIDS Benchmark Survey, CDC, NCHSTP; 1999 Update: National Worksite Health Promotion Survey, OPHS, ODPHP.
10.14 CDC, NIOSH.
10.15 Baseline: Primary Care Provider Surveys, OPHS, ODPHP.
Update: Prevention in Primary Care Study, American College of Preventive Medicine.
Healthy People 2000 Final Review 173

Objective number Data source
10.16 Baseline: National Traumatic Occupational Fatalities, CDC, NIOSH.
Updates: Census of Fatal Occupational Injuries, DOL, BLS.
10.17 National Vital Statistics System, CDC, NCHS.
10.18* Baseline, 1991 and 1992 updates for worksites with 50 or more employees: National Survey of Worksite Health Promotion Activities, OPHS, ODPHP.
1995 data: Business Responds to AIDS Benchmark Survey, CDC, NCHSTP.
1998–99 update: National Worksite Health Promotion Survey, OPHS, ODPHP.
For medium and large companies: Nationwide Survey on Smoking in the Workplace, CDC, OSH; Bureau of National Affairs; American Society for
Personnel Administration.
10.19* Office of Smoking and Health Legislative Tracking System, CDC, NCCDPHP.
10.20* Office of Smoking and Health Legislative Tracking System, CDC, NCCDPHP.
*Duplicate objective. See full text of objective following this table.
174 Healthy People 2000 Final Review
Occupational Safety
and Health
Objectives
10.1: Reduce deaths from work-related
injuries to no more than 4 per 100,000
full-time workers.
10.1a: Reduce deaths among mine
workers from work-related injuries
to no more than 21 per 100,000
full-time workers.
10.1b: Reduce deaths among
construction workers from
work-related injuries to no more
than 17 per 100,000 full-time
workers.
10.1c: Reduce deaths among
transportation workers from
work-related injuries to no more
than 10 per 100,000 full-time
workers.
10.1d: Reduce deaths among farm
workers from work-related injuries
to no more than 9.5 per 100,000
full-time workers.
10.2: Reduce work-related injuries
resulting in medical treatment, lost time
from work, or restricted-work activity to
no more than 6 cases per 100 full-time
workers.
10.2a: Reduce work-related injuries
among construction workers
resulting in medical treatment, lost
time from work, or restricted-work
activity to no more than 10 cases
per 100 full-time workers.
10.2b: Reduce work-related injuries
among nursing and personal care
workers resulting in medical
treatment, lost time from work, or
restricted-work activity to no more
than 9 cases per 100 full-time
workers.
10.2c: Reduce work-related injuries
among farm workers resulting in
medical treatment, lost time from
work, or restricted-work activity to
no more than 8 cases per 100
full-time workers.
10.2d: Reduce work-related injuries
among transportation workers
resulting in medical treatment, lost
time from work, or restricted-work
activity to no more than 6 cases per
100 full-time workers.
10.2e: Reduce work-related injuries
among mine workers resulting in
medical treatment, lost time from
work, or restricted-work activity to
no more than 6 cases per 100
full-time workers.
10.2f: Reduce work-related injuries
among adolescent workers resulting
in medical treatment, lost time from
work, or restricted-work activity to
no more than 3.8 cases per 100
full-time workers.
10.3: Reduce cumulative trauma
disorders to an incidence of no more
than 60 cases per 100,000 full-time
workers.
10.3a: Reduce cumulative trauma
disorders among manufacturing
industry workers to an incidence of
no more than 150 cases per 100,000
full-time workers.
10.3b: Reduce cumulative trauma
disorders among meat product
workers to an incidence of no more
than 2,000 cases per 100,000
full-time workers.
10.4: Reduce occupational skin disorders
or diseases to an incidence of no more
than 55 per 100,000 full-time workers.
10.5*: Reduce hepatitis B infections
among occupationally exposed workers
to an incidence of no more than 623
cases.
Duplicate objective: 20.3e
10.6: Increase to at least 95 percent the
proportion of worksites with 50 or more
employees that mandate employee use
of occupant protection systems, such as
seat belts, during all work-related motor
vehicle travel.
10.7: Reduce to no more than 15 percent
the proportion of workers exposed to
average daily noise levels that exceed
85 dBA.
10.8: Eliminate exposures which result
in workers having blood lead
concentrations greater than 25 ug/dL of
whole blood.
10.9*: Increase hepatitis B
immunization levels to 90 percent
among occupationally exposed workers.
Duplicate objective: 20.11
10.10: Implement occupational safety
and health plans in 50 States for the
identification, management, and
prevention of leading work-related
diseases and injuries within the State.
10.11: Establish in 50 States exposure
standards adequate to prevent the major
occupational lung diseases to which
their worker populations are exposed
(byssinosis, asbestosis, coal workers’
pneumoconiosis, and silicosis).
10.12: Increase to at least 70 percent the
proportion of worksites with 50 or more
employees that have implemented
programs on worker health and safety.
10.13: Increase to at least 50 percent the
proportion of worksites with 50 or more
employees that offer back injury
prevention and rehabilitation programs.
10.14: Establish in 50 States either
public health or labor department
programs that provide consultation and
assistance to small businesses to
implement safety and health programs
for their employees.
10.15: Increase to at least 75 percent the
proportion of primary care providers
who routinely elicit occupational health
exposures as a part of patient history
and provide relevant counseling.
10.16: Reduce deaths from work-related
homicides to no more than 0.5 per
100,000 full-time workers.
10.17: Reduce the overall age-adjusted
mortality rate for four major preventable
occupational lung diseases (byssinosis,
asbestosis, coal workers’
pneumoconiosis, and silicosis) to 7.7 per
100,000.
10.18*: Increase to 100 percent the
proportion of worksites with a formal
smoking policy that prohibits or
severely restricts smoking at the
workplace.
Duplicate objective: 3.11
10.19*: Enact in 50 States and the
District of Columbia comprehensive
laws on clean indoor air that prohibit
smoking or limit it to separately
ventilated areas in the workplace and
enclosed public places.
Duplicate objective: 3.12
10.20*: Reduce to zero the number of
States that have clean indoor air laws
preempting stronger clean indoor air
laws on the local level.
Duplicate objective: 3.25
*Duplicate objective.
Healthy People 2000 Final Review 175

Priority Area 11
Environmental
Health
Background
Environmental factors play a central
role in human development, health, and
disease. A mainstay of public health
practice since the late 1800s (1),
environmental protection addresses both
the physical and social environments.
The physical environment includes the
air, water, and soil through which
exposure to chemical, biological, and
physical agents can occur. The social
environment encompasses housing,
transportation, urban development, land
use, industry, and agriculture and results
in exposures to conditions such as
work-related stress, injury, and violence.
An estimated 25 percent of
preventable illnesses worldwide can be
attributed to poor environmental quality,
with diarrheal diseases and respiratory
infections heading the list (2).
Poor air quality contributes to
respiratory illness, cardiovascular
disease, and cancer. In the United States,
air pollution alone is associated with
about $40– $50 billion in health-related
costs annually (3). Two indicators of air
quality, both outdoor and indoor, are
ozone and environmental tobacco smoke
(ETS), respectively. In 1997,
approximately 43 percent of the U.S.
population lived in areas that failed to
meet federally established health-based
standards for ozone (3). From 1988 to
1994, 65 percent of nonsmokers were
exposed to ETS. An estimated 125
million children were exposed to ETS in
their homes in 1996 (3).
In 1995, 85 percent of persons
served by community water systems
received water that met Federal
standards (3). Between 1987 and 1996,
an average of 15.5 waterborne disease
outbreaks per year occurred, of which 6
originated from community water
systems that supplied drinking water (3).
Toxic substances and wastes pose a
significant public health threat. Exposure
to lead is evidenced by elevated blood
lead levels in children. In the area of
waste management, more than one-half
of the U.S. population was served by
curbside recycling by the end of the
decade.
Increasingly, environmental quality
is becoming a global concern. The
potential for health risks to spread is
heightened as ever-increasing numbers
of people and products cross national
borders. For example, pesticides that are
not registered or that are restricted in
the United States potentially could enter
this country through imported fruits,
vegetables, and seafood produced
abroad, adding an additional burden to
the U.S. public health system.
Data Summary
Highlights
Asthma hospitalizations (11.1) for
the population as a whole declined over
the decade, exceeding the year 2000
target in 1998. Although the rates for
the nonwhite population, children 14
years of age and under, and women over
25 years of age also declined, they
remained significantly higher than their
year 2000 targets. Reducing the number
of children with elevated blood lead
levels (11.4) from 3 million to 393,000
was one of the most significant
achievements in environmental health in
the latter part of the 20th century. The
final number of children, however, was
short of the target of 300,000 set for the
year 2000.
The number of waterborne
outbreaks (11.3) declined through the
decade and was less than the year 2000
target of 11 for each year between 1996
and 1998. The targets for increasing the
number of curbside and household
hazardous waste recycling programs
(11.5) and the average amount of solid
waste produced ‘‘after recovery’’ (11.8)
were also met by the close of the
decade, although the average amount of
solid waste before recovery (11.8)
increased above the set target limit. The
proportion of children exposed to
tobacco smoke at home (11.7) was
reduced to below the target as well.
Progress in the proportion of rivers,
lakes, and estuaries that met designated
uses (11.10) was split with recreational
activities moving toward or meeting
year 2000 targets and consumable fish
moving away from the targets in all
cases.
The proportion of people who knew
what radon was and had tested their
homes for radon (11.6) increased in
1998 compared with previous years and
was three times that reported in 1990.
Only a fraction of these homes exceed
the level identified as dangerous (four
picocuries) by the U.S. Environmental
Protection Agency. The proportion of
homes in which smokers or former
smokers lived that had been tested for
radon increased in 1998. As of 1997, 28
States required disclosure of radon test
results at the time of home sales (11.13).
While this number represents a major
increase over the 1989 baseline of one
State, it falls short of the year 2000
target of 30 States.
The proportion of people living in
counties that did not exceed ambient air
standards for any of the six air
pollutants (ozone, carbon monoxide,
nitrogen dioxide, sulfur dioxide,
particulates, and lead) (11.5) increased
substantially and, measured individually,
only fell short of the year 2000 target
for ozone.
Summary of Progress
Of the 17 environmental health
objectives, 4 (11.1, 11.3, 11.15, and
11.17) met the year 2000 target and 9
showed progress toward the year 2000
targets (11.4–11.7, 11.9, 11.11–11.13,
and 11.16). Progress for objective 11.14
is difficult to evaluate (see Data Issues).
One objective (11.2) moved away from
its target. Progress for two objectives
(11.8 and 11.10) was mixed. See
table 11 for the tracking data for the
objectives in this priority area and
figure 11 for a quantitative assessment
of progress.
Discussion
The decline in childhood lead
poisoning in the United States represents
a public health success. The dramatic
reduction was a result of research to
identify persons at risk, professional and
public education campaigns to ‘‘spread
the word,’’ broad-based screening
measures to find those at risk, and
effective community efforts to clean up
problem areas, namely, substandard
housing units. However, despite the
success achieved, more remains to be
done before childhood lead poisoning
becomes a disease of the past. Although
childhood lead poisoning occurred in all
population groups, the risk was higher
for persons having low income, living in
older housing, and belonging to certain
racial and ethnic groups.
Urban sprawl has become an
increasingly important concern in the
United States for several reasons:
increased outdoor air pollution in major
urban areas, reduced quality of life due
176 Healthy People 2000 Final Review

11.1 Asthma hospitalizations
11.2 Serious mental retardation: Children 10 years, IQ<50
11.3 Waterborne diseases
11.4 Children with: Blood lead levels >15 µg/dL
Blood lead levels >25 µg/dL
11.5 People in counties not exceeding air pollutant standards
11.6 Homes with radon testing
11.7 Toxic agent releases: DHHS carcinogens
ATSDR most toxic substances
11.8 Solid waste, per person per day
Solid waste after recovery, per person per day
Rivers supporting recreational activities
11.10 Rivers supporting consumable fish
11.9 Community water systems meeting safety standards
Lakes supporting consumable fish
Lakes supporting recreational activities
Estuaries supporting consumable fish
Estuaries supporting recreational activities
11.11 Homes tested for lead paint
11.13 States requiring radon disclosure
11.15 Population served by: Curbside recycling
Household hazardous waste recycling
11.12 States with radon minimizing construction standards
11.17 Children's exposure to smoke at home
Moved away from target Moved toward target Met target
*This objective has moved away from its target. A progress quotient could not be calculated.
NOTE: Complete tracking data are shown in table 11. Progress quotients are not calculated for objectives 11.14 and 11.16.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
75%
76%
36%
-20%
-45%
-28%
78%
7%
27%
22%
Away*
15%
Percent of target achieved
-100 -80 -60 -40 -20
0
20 40 60 80 100
118%
120%
106%
100%
302%
104%
100%
100%
-967%
33%
97%
93%
Healthy People 2000 Final Review 177
Figure 11. Final status of Environmental Health objectives

to the loss of free time and the stress of
increased commuting time, and less
green space in major metropolitan areas.
For example, between 1983 and 1995,
the average annual vehicle miles
traveled increased 80 percent (4). These
conditions may lead to increases in
asthma and other respiratory conditions,
which often are triggered or worsened
by substances found in the air. Although
the public health community has
focused several activities on asthma, it
continues to be a significant burden. The
direct economic and health care costs of
asthma and other respiratory conditions
were estimated to be $6.2 billion in
1990, projected to have risen to $14.5
billion by the year 2000. Indirect costs
include an estimated 10 million schools
days missed each year by children with
asthma and $1 billion of lost
productivity of their parents caring for
them (5).
Although improvements in
environmental public health are possible,
complete success is often elusive. For
instance, the waterborne disease
objective (11.3) exceeded the target;
however, infectious agents still
contaminate drinking water. Animals
continue to carry diseases to human
populations, and outbreaks of
once-common intestinal diseases (for
example, typhoid fever), although less
frequent, still occur. These outbreaks
underscore the need to maintain and
improve programs developed in the first
half of the 20th century to ensure the
safety of food and water. The challenge
is to retain these basic capacities in the
21st century, with the added
responsibilities for dealing with
emerging hazards. The control of
well-known hazards must coexist with
ongoing research and the development
of strategies and methods to understand
and control new hazards.
An increase in public awareness of
environmental health issues is key to
public health advances. Education—at
all levels—is a cornerstone of broad
prevention efforts. Improving the
availability of environmental health data
will impact on continued improvement
for the environmental health objectives.
Surveillance systems play a crucial role
to track and understand exposures.
Substantial improvement has occurred in
the establishment of State-based
surveillance systems, especially for
childhood, and to a lesser extent adult,
lead poisoning.
Transition to Healthy People
2010
The 1990s saw extensive expansion
and increased visibility of environmental
health-related issues in both the
scientific and public arenas. The
heightened concern over environmental
health issues had the effect of
broadening the field. As a result, the
number and breadth of the Healthy
People 2010 Environmental Health
objectives increased.
Because of the expansion of the
field, an effort was made to convene a
more comprehensive working group to
develop the plan for the focus area. In
addition to the agencies serving as co-
leads for the focus area, members were
recruited from other Federal agencies,
national organizations, and professional
associations with areas of expertise
relevant to environmental health. The
expansion of the field was further
supported by the extensive input
received during the public comment
period regarding the subject matter of
environmental health.
Healthy People 2010 builds on the
experience gained from Healthy People
2000 by presenting objectives that
address developments and shortcomings
encountered during the decade. For
example, for clean air standards,
Healthy People 2010 contains an
objective that takes into consideration
Federal standards and additional
programs as well as a series of
objectives that call for the elimination of
unclean air in cities. Also, because a
system to accurately and effectively
measure the cleanup of hazardous waste
sites was not developed, the Healthy
People 2010 objective has been
substantially changed, incorporating
other types of contaminated sites.
Although substantially expanded,
the Healthy People 2010 objectives
retained three Healthy People 2000
objectives and retained the substance of
six others. Because of the significant
expansion decided on by the
environmental health work group and to
facilitate management, the new chapter
is divided into six sections:
Outdoor Air Quality
Water Quality
Toxics and Waste
Healthy Homes and Healthy
Communities
Infrastructure and Surveillance
Global Environmental Health
Some proposed objectives and
topics were eliminated from the set of
final objectives because of the expanse
of the environmental health field. In
addition, a number of objectives and
topics such as food, noise, and
respiratory-related environmental factors
were subsumed into other focus areas
(Food Safety, Occupational Safety and
Health, and Respiratory Diseases) to
avoid duplication of objectives.
The objective addressing exposure
to air that does not meet EPA’s standard
for ozone was retained from Healthy
People 2000 (with some variation) and
has been designated as a measure of one
of the Healthy People 2010 Leading
Health Indicators (LHIs). Under the
objective, a high standard of
improvement has been set for ozone
because ozone levels require the most
improvement of the outdoor air
pollutants. The LHIs are discussed in
further detail in the Introduction.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the changes between the
two decades of objectives, reflecting
new knowledge and direction for this
area.
Data Issues
Definitions
Data for objective 11.1 (asthma
hospitalizations) come from the National
Hospital Discharge Survey (NHDS)
maintained by the National Center for
Health Statistics (NCHS), Centers for
Disease Control and Prevention (CDC).
Data for the survey are obtained from
approximately 480 hospitals throughout
the United States. Data on race (required
for objective 11.1a asthma
hospitalizations for blacks and
nonwhites) are not reported by many
hospitals due to omission of a race field
on hospital discharge reporting forms.
More hospitals have automated their
discharge systems in recent years and
are using these forms (UB–82 and
UB–92). A comparison of NHDS data
with National Health Interview Survey
(NHIS) data for people who reported
being hospitalized indicated that
underreporting for whites was roughly
22 percent in 1991; the difference in
reporting for blacks was negligible (6).
(NHIS data were adjusted to exclude
hospitalizations of 1 day or less.)
178 Healthy People 2000 Final Review
The baseline data for 11.2 (mental
retardation) were revised to be
comparable with data from the
Metropolitan Atlanta Developmental
Disabilities Surveillance Program, which
uses school counts of children classified
as mentally retarded.
Data for 11.3 are from the CDC’s
Waterborne Surveillance System, which
compiles data from States; reporting is
voluntary. Only water intended for
drinking is included. Variations in the
level of reporting can produce large
fluctuations in the number of outbreaks
reported from year to year. An outbreak
can be defined when as few as two
people are affected by waterborne
disease or poisoning; however, the
numbers of people affected by the one
outbreak in 1994 (the year the target
was met) were as high as 400,000
people. Epidemiological evidence is
used to link the cause of the outbreak to
water.
The updates for 11.4 are from the
National Health and Nutrition
Examination Survey (NHANES) III
(1988–94). The children tested in
NHANES III were 1–5 years of age
compared with 6 months–5 years of age
for the 1984 baseline projected from
NHANES II (1976–80) data.
Additionally, the special population was
identified using the Bureau of Census
Poverty Income Ratio rather than a
discrete family income level. It should
be noted that the 1988–94 update data
surpassed the original year 2000 target
for this objective. The current, more
ambitious, target set at the midcourse
review was not attained.
Data for 11.5 (air quality) are
affected by a range of meteorological
factors (for example, temperature and
wind) and may vary considerably on an
annual basis. The data are also limited
by the fact that not all counties have
monitoring stations. Individual exposure
within counties varies greatly and health
effects from poor air quality are
mitigated by a wide range of individual
factors (for example, personal sensitivity
to pollutants, other health conditions,
and use of health services). Additionally,
health effects from some pollutants may
occur at levels lower than those
specified in the National Ambient Air
Quality Standards (NAAQS). Data
issues for this objective are discussed in
more detail in Healthy People Statistical
Notes No. 9 (7).
Data for 11.7 (toxic agent release)
are from the Toxic Chemical Release
Inventory maintained by EPA. The
inventory estimates of prior year
releases are provided to EPA by
industry, which periodically revises
these estimates. These revisions are
permitted under the Community Right to
Know Act of 1986; however, they
complicate monitoring of this objective.
Data for 11.8 are estimates of per
capita waste production and per capita
recycling. While pounds of waste
produced have increased beyond the
target set for the year 2000, pounds
recycled have increased at a faster rate
so that final levels of waste after
recovery have decreased and met the
year 2000 target.
Although drinking water quality has
improved, data for 11.9 (safe drinking
water) have remained relatively
unchanged for the past 5 years because
of an increase in the number of
maximum contaminant level (MCL)
standards used to define safe drinking
water. For the past several years,
compliance has also been based on
reporting and treatment standards, as
well as contaminants. Additionally, the
proportions reported for this objective
reflect the proportion of community
water systems, rather than the proportion
of the population (which is stated in the
objective). The proportion of the
population served by community water
systems has increased over the years;
currently they serve nearly 98 percent of
the population.
Data for objective 11.15 (hazardous
waste recycling) include both permanent
(year-round) and temporary (1 day)
recycling programs. The data for
temporary programs in 1995 are
estimated numbers.
For objective 11.17 (children’s
exposure to tobacco smoke at home),
the definition of regular exposure is
defined as the occurrence of tobacco
smoking anywhere in the home on 4 or
more days each week.
Proxy Data
Updates for 11.6 (radon testing)
come from the NHIS and represent the
proportion of people living in homes
where a survey respondent reported that
they knew what radon was and had
tested their home for radon; the
objective calls for an increase in the
proportion of homes that had been
tested. The update data for children
represent the proportion of homes with
children 6 years of age and under where
the respondent reported testing for
radon. The update data on smokers were
limited to those who reported smoking
at home 4 or more days a week.
Data for 11.11 (lead paint testing)
are also provided by the NHIS and
represent the proportion of people who
reported testing their homes (if built
before 1950) for lead paint, rather than
the proportion of homes built before
1950 tested for lead-based paint as
called for in the objective.
Data Availability
There were no further updates for
tracking disclosure of lead paint (11.13)
beyond 1991. Federal regulations
promulgated in 1996 require disclosure
of the presence of lead paint in all
pre-1978 houses in all 50 States during
sales or leasing.
The Agency for Toxic Substances
and Disease Registry (ATSDR) reported
that, in 1995, EPA and States had
followed 90 percent of their
recommendations at National Priorities
List (NPL) sites with health concerns or
hazards where ATSDR had made
recommendations (11.14). This level of
concurrence, however, will vary from
year to year because the number of NPL
sites continues to vary and there may be
a lag between the time that sites are
listed; ATSDR makes recommendations
and EPA and States act on those
recommendations.
The data for objective 11.16
(sentinel environmental diseases) include
data reported by States in surveys
conducted by the Council of State and
Territorial Epidemiologists (CSTE) and
Public Health Foundation (PHF).
Additionally, CDC is working with other
government and nongovernment
organizations to develop guidelines to
improve State capacity to conduct
environmental surveillance.
References
1. Commissioned Corps of the U.S. Public Health
Service, HHS.
http://www.os.dhhs.gov/phs/corps/direct1.html#history.
June 14, 2000.
2. World Health Organization. Fact sheet 170.
Geneva, Switzerland: World Health Organization.
1997.
3. Department of Health and Human Services.
Healthy People 2010. 2d ed. With understanding
and improving health and objectives for improving
health. 2 vols. Washington: U.S. Government
Printing Office. 2000.
Healthy People 2000 Final Review 179
4. Hu PS. Summary of travel trends 1995
nationwide personal transportation survey.
Washington: Department of Transportation. 1999.
5. Department of Health and Human Services.
Office of Science Policy. Action Against Asthma.
A strategic plan for the Department of Health and
Human Services. Washington: Department of
Health and Human Services. 2000.
6. Kozak LJ. Underreporting of race in the
National Hospital Discharge Survey. Advance data
from vital and health statistics; no 265.
Hyattsville, Maryland: National Center for Health
Statistics. 1995.
7. Seitz F, Plepys C. Monitoring air quality in
healthy people 2000. Healthy people statistical
notes; no 9. Hyattsville, Maryland: National
Center for Health Statistics. 1995.
180 Healthy People 2000 Final Review

Table 11. Environmental Health objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
11.1 Asthma hospitalizations (per 100,000) ....... 1987 188 192 196 183 183 174 194 179 179 155 - - - 160
a. Black and other nonwhite ............... 1987 334 340 349 380 290 353 368 360 342 292 - - - 265
b. Children 14 years and under............. 1987 284 308 339 344 280 295 369 338 358 277 - - - 225
c. Female 25 years and over .............. 1988 229 235 235 216 198 202 219 201 197 191 - - - 183
11.2* Serious mental retardation (per 1,000)
Children 10 years with IQ less than 50 ........ 1985-87
§
3 . 1 --- ---
1
3.6 --- --- --- --- --- --- --- 2.0
11.3 Waterborne diseases (number of outbreaks) . . . 1988 16 14 15 19 17 11 16 6 7 10 - - - 11
a. People served by community water systems . . 1988 4 3 2 5 9 5822 6--- 2
11.4 Blood lead levels among children (6 months to
5 years)
Levels exceeding 15 µg/dL ................. 1984 3 million - - - - - - - - - - - -
2
393,000 - - - - - - - - - - - - - - - 300,000
Levels exceeding 25 µg/dL ................. 1984 234,000 - - - - - - - - - - - -
2
59,000 - - - - - - - - - - - - - - - 0
a. Inner-city low-income black children
Levels exceeding 15 µg/dL ................. 1984 234,900 - - - - - - - - - - - -
2
93,000 - - - - - - - - - - - - - - - 75,000
Levels exceeding 25 µg/dL ................. 1984 36,700 - - - - - - - - - - - -
2
18,000 - - - - - - - - - - - - - - - 0
11.5 Proportion of people in counties that have
not exceeded standards for air pollutants
3
Total population (any of the following
pollutants)............................. 1988 49.7% 69.4% 65.3% 78.4% 76.5% 75.1% 67.1% 81.3% 78.9% 76.5% - - - 85%
Ozone ................................ 1988 53.6% 74.2% 72.0% 82.1% 79.5% 79.9% 71.5% 83.3% 80.7% 79.5% - - - 85%
Carbon monoxide ........................ 1988 87.8% 91.1% 92.0% 94.3% 95.4% 93.9% 95.2% 94.9% 96.4% 95.9% - - - 85%
Nitrogen dioxide ......................... 1988 96.6% 96.5% 96.5% 100% 100% 100% 100% 100% 100% 100% - - - 85%
Sulfur dioxide ........................... 1988 99.3% 99.4% 98.0% 100% 99.4% 100% 100% 99.9% 99.9% 100% - - - 85%
Particulates............................. 1988 89.4% 92.3% 91.4% 89.6% 97.5% 94.8% 90.2% 97.1% 96.8% 97.3% - - - 85%
Lead.................................. 1988 99.3% 97.8% 94.1% 98.1% 97.8% 98.3% 98.1% 98.3% 99.0% 98.3% - - - 85%
11.6 Proportion of homes with radon testing ..... 1989
Less
than
5%
4
5.6%
4
8.7% - - -
4
11.4%
4
11.0% - - - - - - - - -
4
17.5% - - - 40%
a. Homes with smokers and former smokers
5
. . 1990 5.2% . . . 6.9% - - - 10.3% 8.8% - - - - - - - - - 16.0% - - - 50%
b. Homes with children ................... 1990
6
6.8% . . .
7
10.7% - - -
7
13.8%
7
13.1% - - - - - - - - -
7
20.0% - - - 50%
11.7 Toxic agent releases
DHHS list of carcinogens (billion pounds) ...... 1988
§
0.35 0.28 0.23 0.21 0.19 0.18 0.17 - - - - - - - - - - - - 0.12
ATSDR list of the most toxic chemicals (billion
pounds) 275 substances .................. 1988
§
2.15 1.75 2.04 1.39 1.64 1.04 1.02 - - - - - - - - - - - - 1.08
11.8 Solid waste (average pounds per person
per day) .............................. 1988 4.0 4.3 - - - 4.5 4.4 4.5 4.4 4.3 4.4 4.5 - - - 4.3
After recovery (recycling and composing) ...... 1988 3.5 3.6 - - - 3.6 3.4 3.4 3.3 3.2 3.2 3.2 - - - 3.2
11.9 People receiving safe drinking water........ ... --- --- --- --- --- --- 98% --- --- --- --- ---
Proportion of community water systems meeting
standards ............................. 1988
§
73% 73% 72% 72% 68% 66% 73% 73% 74% 76% 77% 85%
Number of Maximum Contaminant Level
Standards in force....................... ... --- 36 57 81 81 81 81 81 81 81 --- ...
See footnotes and key at end of table.
Healthy People 2000 Final Review 181

Table 11. Environmental Health objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
11.10 Waters supporting beneficial uses
Rivers supporting:
Consumable fish ..................... 1992 89% . . . . . . . . . - - - 95% - - - 85% - - - 88% - - - 94%
Recreational activities ................. 1992 71% . . . . . . . . . - - - 77% - - - 79% - - - 72% - - - 85%
Lakes supporting:
Consumable fish ..................... 1992 64% . . . . . . . . . - - - 82% - - - 63% - - - 59% - - - 82%
Recreational activities ................. 1992 77% . . . . . . . . . - - - 81% - - - 75% - - - 80% - - - 88%
Estuaries supporting:
Consumable fish ..................... 1992 94% . . . . . . . . . - - - 92% - - - 76% - - - 65% - - - 97%
Recreational activities ................. 1992 83% . . . . . . . . . - - - 85% - - - 84% - - - 91% - - - 91%
11.11 Homes built before 1950 tested for lead-based
paint ................................
1990
Less
than
5% ... --- --- 9% --- --- --- --- 15% --- 50%
11.12 Number of States with construction standards
to minimize radon concentrations ......... 1989 1 3 - - - - - -
8
3 --- --- --- 6 --- --- 35
11.13 Number of States requiring disclosure of lead
and radon concentrations
Lead-based paint and radon ................ 1989 1 1 3 - - - - - - - - - - - - - - - 28 - - - - - - 30
Lead-based paint ........................ 1989 2 2 5 - - - - - - - - - - - -
9
50
9
50
9
50
9
50 30
Radon ................................ 1989 1 3 5 - - - 13 - - -
10
26 --- 28 --- --- 30
11.14 Significant health risks from hazardous
waste sites
Number of sites on National Priority List ..... 1990
§
1,079 . . . - - - 1,199 - - - - - - 1,232 1,210 - - - 1,200 1,400 . . .
Percent of sites identified with public health
hazards/concerns where ATSDR
recommendations were implemented ....... ... --- --- --- --- --- ---
11
90% - - - - - - - - - - - - 100%
11.15 Recyclable materials and household
hazardous waste programs
Population served by curbside recycling ..... 1991 26% . . . . . . 30% 39% 42% 46% 51% 51% 51% - - - 50%
Permanent and temporary household
hazardous waste recycling programs
12
.... 1991 802 . . . . . . 867 1,223 - - -
13
2,184 - - -
14
3,000 - - - - - - 1,529
Permanent programs .................... 1991 96 . . . . . . - - - - - - - - - 284 - - - - - - - - - - - - 215
Temporary programs .................... 1991 706 . . . . . . - - - - - - - - -
13
1,900 - - - - - - - - - - - - 1,314
States with at least one program ........... ... ---
15
28 50 50 50 50 50 50 50 50 50 . . .
11.16 Number of States that track sentinel
environmental diseases
Plans established and monitored ........... 1990 0 . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - 35
Federal funds for surveillance ............. ... --- --- --- 8 10 19 27 31 36 --- --- ...
State data collection for specific diseases
Childhood lead poisoning ............... ... --- --- --- --- --- --- --- --- 51 --- 51 ...
Adult nonoccupational lead poisoning...... ... --- --- --- --- --- --- --- --- 28 --- 33 ...
Mercury poisoning .................... ... --- --- --- --- 9 --- --- --- 15 --- 17 ...
Arsenic poisoning..................... ... --- --- --- --- 8 --- --- --- 11 --- 14 ...
See footnotes and key at end of table.
182 Healthy People 2000 Final Review
Table 11. Environmental Health objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Cadmium poisoning ................... ... --- --- --- --- 7 --- --- --- 11 --- 13 ...
Methemglobinemia .................... ... --- --- --- --- --- --- --- --- 9 --- 10 ...
Acute chemical poisoning............... ... --- --- --- --- 6 --- --- --- 8 --- 10 ...
Carbon monoxide poisoning ............. ... --- --- --- --- 4 --- --- --- 7 --- 9 ...
Heatstroke .......................... ... --- --- --- --- --- --- --- --- 4 --- 5 ...
Hypothermia ........................ ... --- --- --- --- --- --- --- --- 4 --- 5 ...
11.17* Children’s exposure to smoke at home
(6 years and under) ..................... 1986 39% - - - 32% - - - 27% 27% - - - - - - - - - 20% - - - 20%
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
1
1991–92 data.
2
1988–94 data for children 1–5 years.
3
Data are based on 1990 county population estimates.
4
The measure includes people in homes where an adult reported they knew what radon was.
5
The measure includes people in homes where an adult reported that they smoked in their homes 4 or more days a week and knew what radon was.
6
The measure includes people in homes where an adult reported that they had children 16 years or under and knew what radon was.
7
The measure includes people in homes where an adult reported that they had children 6 years or under and knew what radon was.
8
EPA developed model standards for control of radon in buildings in 1993.
9
In 1996, EPA promulgated regulation requiring disclosure of lead-based paint in all pre-1987 housing during sales or leasing.
10
Includes one territory in addition to the States.
11
90 percent represents 253 sites.
12
Permanent programs are fixed locations where hazardous waste can be recycled year round; temporary programs are short-term sites where hazardous waste can be recycled. There has
been a steady increase in permanent programs (from 27 in 1988 to 96 in 1991), whereas the number of temporary programs has declined.
13
Estimated.
14
In 1997, there were more than 3,000 household hazardous waste permanent collection programs and collection events throughout the United States.
15
1987 data.
NOTE: Data include revisions and, therefore, may differ from data previously published in these reports and other publications.
Met
Toward
Mixed/ no change
Away
Cannot assess
Healthy People 2000 Final Review 183

Objective number Data source
11.1, 11.1a-c National Hospital Discharge Survey, CDC, NCHS.
11.2* Baseline: Metropolitan Atlanta Developmental Disabilities Study, CDC, NCEH.
Update: Metropolitan Atlanta Developmental Disabilities Surveillance Program, CDC, NCEH.
11.3, 11.3a Waterborne Surveillance System, CDC, NCEH.
11.4, 11.4a National Health and Nutrition Examination Survey, CDC, NCHS.
11.5 National Air Quality and Emissions Trends Report, EPA, AIRS, OAR.
11.6 Baseline: EPA, OAR, Office of Radiation Programs.
11.6a,b National Health Interview Survey, CDC, NCHS.
11.7 Toxic Chemical Release Inventory, EPA, OPPTS.
11.8 Characterization of Municipal Solid Waste in the United States, EPA, OSWER.
11.9 EPA Federal Reporting Data Base; EPA, Office of Water, Office of Ground Water and Drinking Water.
11.10 National Water Quality Inventory, EPA, Office of Water.
11.11 National Health Interview Survey, CDC, NCHS.
11.12 Environmental Law Institute.
11.13 Alliance to End Childhood Lead Poisoning, Environmental Law Institute.
11.14 National Priorities List, EPA, OSWER; Hazardous Substance Release and Health Effects Database, CDC, ATSDR.
11.15 Biocycle Journal of Waste Recycling; Wastewatch Center.
11.16 For funding data: CDC, NCEH.
For State data collection: PHF; Council for State and Territorial Epidemiologists.
11.17* Baseline: Adult Use of Tobacco Survey, CDC, NCCDPHP.
Update: National Health Interview Survey, CDC, NCHS.
*Duplicate objective. See full text of objective following this table.
184 Healthy People 2000 Final Review
Environmental
Health Objectives
11.1: Reduce asthma morbidity, as
measured by a reduction in asthma
hospitalizations to no more than 160 per
100,000 people.
11.1a: Reduce asthma morbidity
among blacks and other nonwhites,
as measured by a reduction in
asthma hospitalizations to no more
than 265 per 100,000 people.
11.1b: Reduce asthma morbidity
among children, as measured by a
reduction in asthma hospitalizations
to no more than 225 per 100,000
people.
11.1c: Reduce asthma morbidity
among women, as measured by a
reduction in asthma hospitalizations
to no more than 183 per 100,000
people.
11.2*: Reduce the prevalence of serious
mental retardation among school-aged
children to no more than 2 per 1,000
children.
Duplicate objective: 17.8
11.3: Reduce outbreaks of waterborne
disease from infectious agents and
chemical poisoning to no more than 11
per year.
11.3a: Reduce outbreaks of
waterborne disease from infectious
agents and chemical poisoning
among people served by community
water systems to no more than 2
per year.
11.4: Reduce the prevalence of blood
lead levels exceeding 15 ug/dL and 25
ug/dL among children aged 6 months–5
years to no more than 300,000 and zero,
respectively.
11.4a: Reduce the prevalence of
blood lead levels exceeding 15
ug/dL and 25 ug/dL among
inner-city low-income black
children (annual family income less
than $6,000 in 1984 dollars) to no
more than 75,000 and zero,
respectively.
11.5: Reduce human exposure to criteria
air pollutants, as measured by an
increase to at least 85 percent in the
proportion of people who live in
counties that have not exceeded any
Environmental Protection Agency
standard for air quality in the previous
12 months.
11.6: Increase to at least 40 percent the
proportion of homes in which
homeowners/occupants have tested for
radon concentrations and that have
either been found to pose minimal risk
or have been modified to reduce risk to
health.
11.6a: Increase to at least 50 percent
the proportion of homes with
smokers and former smokers in
which homeowners/occupants have
tested for radon concentrations and
that have either been found to pose
minimal risk or have been modified
to reduce risk to health.
11.6b: Increase to at least
50 percent the proportion of homes
with children in which home-
owners/occupants have tested for
radon concentrations and that have
either been found to pose minimal
risk or have been modified to
reduce risk to health.
11.7: Reduce human exposure to toxic
agents by decreasing the release of
hazardous substances from industrial
facilities:
65 percent decrease in the
substances on the Department of
Health and Human Services list of
carcinogens.
50 percent reduction in the
substances on the Agency for Toxic
Substances and Disease Registry
(ATSDR) priority list of the most
toxic chemicals.
11.8: Reduce human exposure to solid
waste-related water, air, and soil
contamination, as measured by a
reduction in average pounds of
municipal solid waste produced per
person each day to no more than 4.3
pounds before recovery and 3.2 pounds
after recovery.
11.9: Increase to at least 85 percent the
proportion of people who receive a
supply of drinking water that meets the
safe drinking water standards established
by the Environmental Protection
Agency.
11.10: Reduce potential risks to human
health from surface water, as measured
by an increase in the proportion of
assessed rivers, lakes, and estuaries that
support beneficial uses, such as
consumable fishing and recreational
activities.
2000
Waters supporting target
beneficial use (percent)
Rivers supporting:
Consumable fish 94
Recreational activities 85
Lakes supporting:
Consumable fish 82
Recreational activities 88
Estuaries supporting:
Consumable fish 97
Recreational activities 91
11.11: Perform testing for lead-based
paint in at least 50 percent of homes
built before 1950.
11.12: Expand to at least 35 the number
of States in which at least 75 percent of
local jurisdictions have adopted
construction standards and techniques
that minimize elevated indoor radon
levels in those new building areas
locally determined to have elevated
radon levels.
11.13: Increase to at least 30 the number
of States requiring that prospective
buyers be informed of the presence of
lead-based paint and radon
concentrations in all buildings offered
for sale.
11.14: Eliminate significant health risks
from National Priority List hazardous
waste sites, as measured by performance
of clean-up at these sites sufficient to
eliminate immediate and significant
health threats as specified in health
assessments completed at all sites.
11.15: Establish curbside recycling
programs that serve at least 50 percent
of the U.S. population and continue to
increase household hazardous waste
collection programs.
Recyclable materials
and household 2000
hazardous waste target
programs (percent)
Percentage of population
served by curbside
recycling programs 50
2000
Permanent and temporary target
household hazardous (number of
waste collection events events)
Permanent 215
Temporary 1,314
Total 1,529
Healthy People 2000 Final Review 185
11.16: Establish and monitor in at least
35 States plans to define and track
sentinel environmental diseases.
11.17*: Reduce to no more than
20 percent the proportion of children
aged 6 and younger who are regularly
exposed to tobacco smoke at home.
Duplicate objective: 3.8
*Duplicate objective.
186 Healthy People 2000 Final Review

Priority Area 12
Food and Drug
Safety
Background
Food Safety
Reducing the number of foodborne
illnesses improves the public health
more than most people might realize. An
estimated 76 million illnesses, 325,000
hospitalizations, and 5,000 deaths each
year may be associated with
microorganisms in food (1). Economic
costs resulting from foodborne illnesses
are estimated at over $3 billion each
year for hospitalizations and between
$20 billion and $40 billion each year for
the cost of lost productivity (2).
Foodborne disease cases and outbreaks
sometimes result from failures in
protective systems, but are often the
result of improper food handling.
Children, the very old, and people with
immunologic deficiencies are at
increased risk of infection and death
resulting from food contamination.
Reductions in foodborne illness will
save lives, improve the quality of life,
and save medical and other costs.
Drug Safety
While drugs provide great public
benefits, their use carries a certain
degree of risk with the possibility of
injury and harm. Adverse drug reactions,
defined as injuries resulting from
medical intervention related to a drug,
are estimated to play a role in more than
100,000 deaths nationwide each year
(3). New drugs, which are being
introduced at an increasing rate, are
becoming more powerful and more
complex. Older adults, who use more
prescription and nonprescription
medicines than younger people, are at
increased risk of suffering adverse drug
reactions (4,5). The physiological
changes associated with increasing age
and particular diseases and conditions
may alter the effects of drugs. In
addition, use of multiple medications
increases the risk of an adverse outcome
(4,5).
With the increasing number and
complexity of prescription drugs, with
more drugs being taken by increasingly
large numbers of elderly patients, and
with patients seeing multiple
practitioners, it becomes more vital that
each participant in the health care
system—the patient, the health care
provider, pharmaceutical manufacturers,
and the regulators—be well informed
with up-to-date information.
Data Summary
Highlights
Food Safety
One of the successes for food safety
in the last decade of the 20th century
was the drop in the incidence of
listeriosis (12.1). The decrease was
enough to reach the year 2000 target of
0.5 cases per 100,000 persons by 1992.
Except for 1998, when large-scale
outbreaks raised the incidence to 0.6 per
100,000, the incidence of listeriosis has
remained at or below the target level
ever since. The incidence of infection
from other pathogens (12.1) fell as well,
with the year 2000 targets being met for
Salmonella species (13.6 per 100,000);
Campylobacter jejuni (17.5 per
100,000); Escherichia coli O157:H7 (2.8
per 100,000); and Listeria
monocytogenes (0.5 per 100,000) (6).
Significant progress was made in
reducing the number of outbreaks
caused by Salmonella serotype
Enteritidis (12.2). Outbreaks dropped by
43 percent, from 77 outbreaks in 1987
to 44 outbreaks in 1999. Progress was
also made toward the adoption by States
and U.S. Territories of the Food Code
(12.4)—first published in 1993 and
updated and revised biennially since
then. By 1999, a total of 82 percent of
States and Territories either had adopted
(46 percent) or were in the process of
adopting (36 percent) the code (7).
Federal, State, local, Territorial, and
Tribal agencies adopt the code through
their regular legal processes.
Drug Safety
By 1995, 98 percent of pharmacies
were using computers, thus surpassing
the year 2000 target of 75 percent for
linked pharmacy systems (12.5).
Early in the 1990’s, the target of
75 percent set for primary care providers
routinely reviewing with their older
patients all prescribed and
over-the-counter medications each time
a new medication is prescribed, and
maintaining a list of current medications
used by their patients (12.6) was
surpassed by internists, with 77 percent
conducting such reviews and 84 percent
maintaining medication lists. In 1998,
the proportion of nurse practitioners
who routinely maintained a medication
list for 81–100 percent of patients 65
years of age and over and who reviewed
medication when prescribing for the
same proportion of patients had
increased to 71 percent and 68 percent,
respectively, compared with 63 percent
and 55 percent in 1992.
As of mid–2000, reporting of
serious adverse drug reactions (12.7)
had dropped to 65 percent as a
proportion of all events reported to the
Food and Drug Administration’s (FDA)
MedWatch, missing the target of
75 percent and falling below the 1993
baseline of 69 percent.
In 1998, about three-quarters of
people who obtained new prescriptions
received written information about these
prescriptions at the pharmacy (12.8), up
from 32 percent in 1992. Information
includes directions for use (how much
to take and how often to take it) and
possible risks (precautions and side
effects). Written information from the
doctor’s office increased only slightly,
from 14 percent in 1992 to 16 percent in
1998. About 14 percent of consumers
reported receiving verbal counseling on
new prescriptions from pharmacies,
while 24 percent received verbal
information from prescribers. These
proportions are the same or somewhat
lower, respectively, than 2 years earlier,
in 1996.
Summary of Progress
Two of the eight food and drug
safety objectives met their targets (12.1
and 12.5). Five objectives (12.2–12.4,
12.6, and 12.8) show progress toward
their respective targets, and one
objective (12.7) is moving away from
the target. See table 12 for the tracking
data for the objectives in this priority
area and figure 12 for a quantitative
assessment of progress.
Discussion
Food Safety
The significant progress made in the
United States in food safety can be
attributed, in part, to increased consumer
awareness, knowledge, and concern
about foodborne illness. The proportion
of people who thought that foodborne
Healthy People 2000 Final Review 187
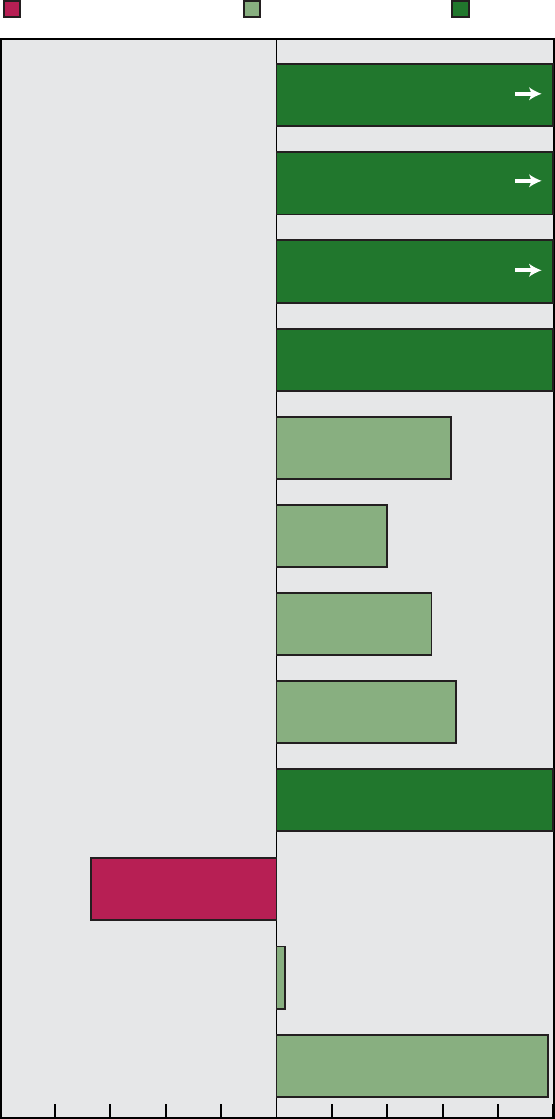
12.1 Foodborne infections: Salmonella species
Campylobacter jejuni
Escherichia coli O157:H7
12.2 Salmonella enteritidis outbreaks
Listeria monocytogenes
12.3 Refrigeration and cutting board practices:
Refrigeration of perishable foods
Washing cutting boards with soap
12.4 States adopting FDA Food Code protection
standards
12.7 Serious adverse event drug reports
12.8 Written information: Prescribers
Dispensers
12.5 Computer utilization by pharmacies
Moved away from target Moved toward target Met target
NOTES: Complete tracking data are shown in table 12. Progress quotients are not calculated for objective 12.6.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
FDA is Food and Drug Administration.
-67%
Percent of target achieved
-100 -80 -60 -40 -20
0
20 40 60 80 100
128%
150%
100%
200%
100%
98%
3%
63%
40%
56%
65%
188 Healthy People 2000 Final Review
Figure 12. Final status of Food and Drug Safety objectives
illness had increased during the previous
5 years rose from 26 percent in 1993 to
40 percent in 1998 (8). The proportion
of people who thought that
contamination of food by
microorganisms was a serious food
safety problem increased from
36 percent in 1993 to 55 percent in 1998
(8). Between 1993 and 1998, several
large foodborne outbreaks occurred.
Large multi-State outbreaks of illness
from E. coli O157:H7, from hamburgers
in 1993 and from fresh apple juice in
1996, caused serious illness and deaths.
These outbreaks galvanized action by
industry, agencies with responsibility for
food safety, and consumers. After the E.
coli O157:H7 outbreaks from ground
beef, produce, and apple juice, more and
more consumers have become familiar
with the risk and even the names of
some foodborne pathogens, particularly
Salmonella species and E. coli O157:H7
(8).
Means to further improve food
safety and reduce foodborne illness and
outbreaks include regulatory
implementation of safe food handling
labels; development and regulatory
implementation of the Food Safety
Initiative (with cooperation and
increased emphasis on surveillance,
enforcement, research, traceback from
illness outbreaks to determine causes
and correct them, and education);
regulatory adoption of the Food Code;
development of key safe food handling
practices and messages (for example, the
Partnership for Food Safety Education’s
FightBac!™ messages of ‘‘clean,
separate, chill, and cook to proper
temperature’’ and the U.S. Department
of Agriculture (USDA) Food Safety and
Inspection Service’s Thermy™, the
thermometer use campaign); mandatory
use of Hazard Analysis and Critical
Control Point (HACCP) systems for
seafood (1995), meat and poultry
(1996), and most recently, for juice (9);
development of and training in Good
Agricultural Practices and Good
Manufacturing Practices for produce and
for eggs; and using quantitative
microbial risk assessments to aid in
developing action plans for foodborne
pathogens (10,11)
Substantial advances were made in
developing infrastructure and data
gathering systems in the 1990s. During
1996, CDC, FDA, USDA, and
participating agencies in several States
and local metropolitan areas piloted an
active surveillance program, the
Foodborne Illness Active Surveillance
Network (FoodNet). Early success of
FoodNet in obtaining better foodborne
illness estimates resulted in increased
funding and cooperation for more sites
and other illness-causing organisms to
be added, especially following the
announcement and funding of the Food
Safety Initiative in 1997 (2).
Another data gathering system,
PulseNet, allows comparison of pulsed
field gel electrophoresis results against a
database to determine if cases may be
caused by the same strain of a
microorganism, such as L.
monocytogenes. The importance of this
network, used by cooperating scientists
in CDC, FDA, FSIS, and States, is that
widespread outbreaks can now be
detected.
Drug Safety
One possible explanation for the
decrease in the proportion of adverse
event drug reports voluntarily sent to
FDA that are regarded as serious is the
lack of health care organizations that are
linked in an integrated system that
monitors and reports adverse events. At
the end of the 20th century, little
collaboration exists between Federal
authorities and researchers with
pharmacoepidemiological databases to
monitor suspected associations between
specific drug exposure and specific
adverse events and in estimating such
risk.
Transition to Healthy People
2010
Healthy People 2010 includes a
focus area on Food Safety and another
on Medical Product Safety, which
replace the Healthy People 2000 priority
area on Food and Drug Safety. The
Healthy People 2010 food safety
objectives were chosen, expanded, or
changed using a scientific basis for
assessing public health improvements.
The science-based Food Safety Initiative
(FSI) guided development of the
objectives and the chapter (2).
Food Safety
Healthy People 2010 has seven
food safety objectives, several of which
have multiple subobjectives. In addition
to the four microorganisms tracked for
foodborne infections in Healthy People
2000, Healthy People 2010 added three
developmental subobjectives to track the
incidence of Cyclospora cayetanensis,
congenital Toxoplasmosis gondii (both
parasites), and hemolytic uremic
syndrome. A subobjective was added to
the objective on foodborne outbreaks for
tracking outbreaks of E. coli O157:H7.
Other new objectives address
antimicrobial resistance of Salmonella
species, anaphylactic deaths from food
allergens, consumer food safety
practices, retail food safety practices,
and exposure to organophosphate
pesticide residues.
The Healthy People 2010 Food
Safety focus area also indicates related
objectives in other focus areas—
Environmental Health, Immunization
and Infectious Diseases, Occupational
Safety and Health, and Public Health
Infrastructure.
Medical Product Safety
The Medical Product Safety focus
area in Healthy People 2010 moves
beyond the issues of drug safety to
encompass biological products and
medical devices as well. It addresses the
issues covered in Healthy People 2000
with a recognition that such topics
pertain to medical products overall. The
focus area includes objectives on
monitoring adverse medical events,
linked automated information systems,
receipt of useful information about
prescriptions from pharmacies, and
receipt of oral counseling about
medications from prescribers and
dispensers. An objective on the supply
of blood from donors is also included.
Objectives related to medical product
safety are found in other Healthy People
2010 focus areas—Access to Quality
Health Care, Cancer, Chronic Kidney
Disease, Diabetes, Educational and
Community-Based Programs, Family
Planning, Health Communication, Heart
Disease and Stroke, Injury and Violence
Prevention, Public Health Infrastructure,
and Respiratory Diseases.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the differences between the
two decades of objectives, reflecting on
changes and new knowledge in the area
of food and drug safety.
Data Issues
Definitions
For objective 12.4, States and
Territories with at least one agency
Healthy People 2000 Final Review 189
adopting the Food Code are considered
to have adopted it. In the earlier part of
the decade, there was a high proportion
of States and Territories reviewing the
Code; in the later years, as more States
and Territories adopted it, the proportion
reviewing it declined. In 2000, the
proportions reviewing and adopting the
Code did not add to 100 percent, as
some States and Territories were neither
reviewing nor had adopted the Code.
The definition of a serious adverse
event (12.7) includes events that are life
threatening and require intervention to
prevent permanent damage as well as
death, hospitalization, disability, and
congenital anomaly (12).
For objective 12.8, receipt of useful
information for new prescriptions, a
prescriber is anyone who is authorized
to prescribe, including physicians, nurse
practitioners, and physician assistants,
depending on the State law. Dispensers
are persons authorized to dispense
prescription medications and include
physicians and pharmacists (12).
Data Sources
Starting in 1996, foodborne illness
incidence data for objective 12.1 have
been obtained from FoodNet. Although
the data are collected in geographically
distinct areas, surveillance is active; the
estimates provided are thought to be
better estimates than those obtained in
the past through national passive
reporting of data. Before FoodNet,
national data and national estimates
were not available for Campylobacter
species and E. coli O157:H7. FoodNet
is the principal foodborne disease
component of CDC’s Emerging
Infections Program (EIP). It is a
collaborative project of the CDC, nine
EIP sites (California, Colorado,
Connecticut, Georgia, New York,
Maryland, Minnesota, Oregon, and
Tennessee), the USDA and FDA. The
project consists of active surveillance
for foodborne diseases and related
epidemiologic studies designed to help
public health officials better understand
the epidemiology of foodborne diseases
in the United States. Foodborne diseases
include infections caused by bacteria
such as Salmonella, Shigella,
Campylobacter, and Vibrio species,
Escherichia coli O157, Listeria
monocytogenes, Yersinia enterocolitica,
and parasites such as Cryptosporidium
and Cyclospora species. In 1995,
FoodNet surveillance began in five
locations: California, Connecticut,
Georgia, Minnesota, and Oregon. Each
year the surveillance area, or catchment,
has expanded, with the inclusion of
additional counties or additional sites
(New York and Maryland in 1998,
Tennessee in 2000, and Colorado in
2001). The total population of the
current catchment is 25.4 million
persons, or 10 percent of the United
States population. Additional
information about FoodNet, including
annual reports, is available on the
Internet at http://www.cdc.gov/foodnet.
MedWatch, which is used to track
objective 12.7, is the FDA Medical
Products Reporting and Safety
Information Program. It provides
important and timely clinical
information about safety issues
involving medical products, including
prescription and over-the-counter drugs,
biologics, dietary supplements, and
medical devices. MedWatch allows
healthcare professionals and consumers
to report serious problems that they
suspect are associated with the drugs
and medical devices they prescribe,
dispense, or use. Further information is
available on the Internet at
http://www.fda.gov/medwatch/index.html.
The baseline data on maintenance
of a current medication list and
medication review for older patients by
primary care providers for objective
12.6 are from the Primary Care Provider
Surveys (PCPS), drawn from a random
stratified sample of members of the
American College of Physicians from
four geographic regions. Provider groups
sampled included internists,
pediatricians, nurse practitioners,
obstetricians/gynecologists, and family
physicians. In 1992, response rates
varied between 50–80 percent across
these groups. The data represent the
proportion of providers who routinely
delivered these services to
81–100 percent of their clients 65 years
of age and over.
The Prevention in Primary Care
Study (PPCS) was conducted in
1997–98 to update data from the PCPS.
The design and items included in the
1997–98 study were similar to the
PCPS, but a slightly different sampling
frame was used and some items
included in the 1992 surveys were not
included in the PPCS. The professionals
were sampled from listings of all
licensed, active practitioners in the
United States whose practices were at
least 50 percent primary care. Because
of low response rates from the other
provider groups, updates are available
only for nurse practitioners. Progress for
this objective was based on the reports
of internists, who had met the target at
baseline, and nurse practitioners.
Data Comparability
Starting in 1996, FoodNet active
surveillance estimates have been used to
track objectives 12.1 and 12.2 in place
of passively collected national data.
Although FoodNet sites have been
added since 1996 to include a much
larger portion of the U.S. population, the
estimates reported for Healthy People
2000 are based only on the five original
(1996) FoodNet sites to keep the data as
comparable as possible. Annual
incidence rates are calculated using
reported cases as the numerator and
census estimates for individual
catchment areas as the denominator (6).
Various surveillance systems of
CDC, including the Salmonella
Surveillance System, the Campylobacter
Surveillance System, and the Bacterial
Meningitis Surveillance System, were
used to monitor progress for objectives
12.1 and 12.2 for data through 1994.
The Salmonella Surveillance System is a
passive laboratory-based system that
uses reports from 49 States, FDA, and
USDA. This system measures the
incidence of infection from Salmonella
species (12.1) and the number of
outbreaks caused by Salmonella
Enteritidis (12.2). Many factors,
including the intensity of surveillance,
the severity of the illness, access to
medical care, and association with a
recognized outbreak, affect whether the
infection will be reported. When
reporting is incomplete, the incidence of
salmonellosis is substantially
underreported.
The incidence of foodborne Listeria
monocytogenes-induced infections (12.1)
was measured until 1994 using the
Bacterial Meningitis Surveillance
System. This is an active
laboratory-based surveillance system
conducted in six States; it counts all
cases of bacterial meningitis and other
invasive bacterial diseases caused by the
five most common pathogens causing
bacterial meningitis, including Listeria
monocytogenes. The participating
surveillance areas represent several
regions throughout the country and a
population of 33.5 million, 14 percent of
the U.S. population.
The Campylobacter Surveillance
System is a passive system that receives
190 Healthy People 2000 Final Review
weekly reports of laboratory isolates of
Campylobacter. The number of
participating States has increased each
year. Surveillance mechanisms,
including laboratory isolation
procedures, vary from State to State.
Baseline data for refrigeration and
cutting-board practices (12.3) were
obtained from the 1988 Health and Diet
Survey, FDA/USDA (HDS). Updates are
from the Food Safety Survey, FDA, and
USDA (FSS). The FSS is based on 20-
to 30-minute telephone interviews with
consumers to determine food safety
knowledge, concern level, food handling
practices, perception of risk, and
consumption of potentially hazardous
foods. In 1992–93, 1,620 people were
surveyed and in 1997–98, 2,001 people
were surveyed.
Proxy Data
Objective 12.5, to increase the
proportion of pharmacies and other
dispensers of prescription medications
that use linked systems to provide alerts
to potential adverse drug reactions
among medications dispensed by
different sources to individual patients,
is being tracked with proxy data. The
data show the proportion of pharmacies
with individual computer capability or
the proportion of pharmacies that are
part of a larger more integrated system
such as a chain store computer system.
References
1. Mead PS, Slutsker L, Dietz V, et al.
Food-related illness and death in the United States.
Emerging Infectious Diseases 5(5):607–25. 1999.
2. Food and Drug Administration, U.S.
Department of Agriculture, and U.S.
Environmental Protection Agency. Food safety
from farm to table: A national food safety
initiative. Report to the President, May 1997.
Washington: FDA, USDA, EPA. 1997.
3. Lazarou J, Pomeranz, PH, Cory PN. Incidence
of adverse drug reactions in hospitalized patients.
A meta-analysis of prospective studies. JAMA 279
(15):1200–05. 1998.
4. Institute of Medicine. To err is human: Building
a safer health system. Washington: National
Academy Press. 2000.
5. National Center for Health Statistics. Healthy
people 2000 review 1998–99. Hyattsville,
Maryland: Public Health Service. 1999.
6. Centers for Disease Control and Prevention.
FoodNet 1999 surveillance results: Final report,
January 1999. Atlanta, Georgia: Centers for
Disease Control and Prevention. 1999.
http://www.cdc.gov/foodnet.
7. Food and Drug Administration, Center for Food
Safety and Applied Nutrition. Status of food code
adoptions. 2000.
http://vm.cfsan.fda.gov/∼ear/fcadopt.html.
8. Fein SB, Levy A. 2000. Safety of Consumer
Food Preparation and Consumption Behaviors.
Paper presented at the Healthy People 2010
Conference, January 25, Washington. 2000.
9. Food and Drug Administration. 21 CFR Part
120; Hazard Analysis and Critical Control Point
(HAACP). Procedures for the safe and sanitary
processing and importing of juice; Final Rule. FR
66(13):6137–6202. Jan. 19, 2001.
10. Food and Drug Administration and Food
Safety and Inspection Service, U.S. Department of
Agriculture. Relative risk to public health from
foodborne Listeria monocytogenes among selected
categories of ready-to-eat foods. Draft risk
assessment document and risk management action
plan. FR 66(13):5515–5517. Jan. 19, 2001.
11. Food and Drug Administration. Public health
impact of Vibrio parahaemolyticus in raw
molluscan shellfish. Draft risk assessment
document. FR 66(13):5517–5518. Jan. 19, 2001.
12. Department of Health and Human Services.
Healthy People 2000 midcourse review and 1995
revisions. Washington: Public Health Service.
1995.
Healthy People 2000 Final Review 191

Table 12. Food and Drug Safety objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
12.1 Foodborne infections (cases per 100,000)
Salmonella species .......................... 1987 18 16 16 14 15 15 - - - 15 14 12 14 16
Campylobacter jejuni ......................... 1987 50 - - - - - - - - - - - - - - - - - - 24 25 21 18 25
Escherichia coli O157:H7...................... 1987 8 - - - - - - - - - - - - - - - - - - 3 2 3 3 4
Listeria monocytogenes ....................... 1987 0.7 0.77 0.61 0.45 0.44 0.42 - - - 0.5 0.5 0.6 0.5 0.5
12.2 Salmonella serotype Enteritidis outbreaks .......... 1989 77 68 68 59 63 44 56 50 46 49 44 25
12.3 Refrigeration and cutting board practices
For refrigeration of perishable foods.............. 1988 70% - - - - - - - - -
1
72% --- --- --- --- --- --- 75%
For washing cutting boards with soap ............ 1988 66% - - - - - - - - -
1
65% --- --- --- ---
2
71% - - - 75%
For washing utensils with soap ................. 1988 55% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 75%
12.4 Use of FDA Food Code by at least one agency
within each State and U.S. Territory
(proportion of States and Territories)
Adopting food protection standards .............. 1994
§
2% ... ... ... ... ... --- 8% 16% ---
3
46% 70%
Reviewing food protection standards ............. ... --- --- --- --- --- 80% --- 82% --- ---
4
36% . . .
12.5 Linked pharmacy systems
5
Computer utilization by pharmacies .............. 1993 95% . . . . . . . . . . . . - - - 98% - - - - - - - - - - - - 75%
12.6 Providers reviewing medication for older patients
Percent of clinicians routinely providing service to
81–100% of patients 65 years and over ........... ... --- --- --- --- --- --- --- --- --- --- --- 75%
Maintenance of current medication list
Nurse practitioners .......................... 1992 63% . . . . . . . . . - - - - - - - - - - - - - - -
2
71% - - - 75%
Obstetricians/gynecologists .................... 1992 64% . . . . . . . . . - - - - - - - - - - - - - - -
2,6
--- --- 75%
Internists .................................. 1992 84% . . . . . . . . . - - - - - - - - - - - - - - -
2,6
--- --- 75%
Family physicians ........................... 1992 70% . . . . . . . . . - - - - - - - - - - - - - - -
2,6
--- --- 75%
Review of medications when prescribing
Nurse practitioners .......................... 1992 55% . . . . . . . . . - - - - - - - - - - - - - - -
2
68% - - - 75%
Obstetricians/gynecologists .................... 1992 60% . . . . . . . . . - - - - - - - - - - - - - - -
2,6
--- --- 75%
Internists .................................. 1992 77% . . . . . . . . . - - - - - - - - - - - - - - -
2,6
--- --- 75%
Family physicians ........................... 1992 63% . . . . . . . . . - - - - - - - - - - - - - - -
2,6
--- --- 75%
12.7 Adverse event drug reports
Proportion voluntarily sent to FDA regarded as
serious .................................... 1993 69% . . . . . . . . . . . . 70% 69% 63% 66% 62%
7
65% 75%
12.8 Receipt of verbal and written information for new
prescriptions ............................... ... --- --- --- --- --- --- --- --- --- --- --- 75%
Written information
From prescribers ............................ 1992 14% . . . . . . . . . - - - 15% - - - 16% - - - 16% - - - 75%
From dispensers ............................ 1992
§
32% ... ... ... --- 59% --- 71% --- 74% --- 75%
Verbal counseling
From prescribers ............................ ... --- ... ... ... ... ... ... 26% --- 24% --- ...
From pharmacists ........................... ... --- ... ... ... ... ... ... 14% --- 14% --- ...
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
Met
Toward
Mixed/ no change
Away
Cannot assess
192 Healthy People 2000 Final Review

1
1992-93 data.
2
1997-98 data.
3
2000 data (at least one agency in each of 25 States + the District of Columbia).
4
2000 data (at least one agency in 18 of 50 States and 1 of 6 Territories).
5
A linked system is one with individual computer capability or one that is part of a larger more integrated system such as a chain store computer system.
6
Response rate for this group was too low to produce reliable estimates.
7
Cumulative total as of July 20, 2000. In 1999, the estimate was 63%.
NOTE: Data include revisions and, therefore, may differ from data previously published in these reports and other publications.
Objective number Data source
12.1 Salmonella Surveillance System, CDC, NCID.
Campylobacter Surveillance System, CDC, NCID.
Bacterial Meningitis Surveillance System, CDC, NCID.
1996–99 updates: Foodborne Disease Active Surveillance Network (FoodNet), CDC, USDA, FDA.
12.2 Salmonella Surveillance System, CDC, NCID.
1996–99 updates: Foodborne Disease Active Surveillance Network (FoodNet), CDC, USDA, FDA.
12.3 Baselines: Health and Diet Survey, USDA/FDA.
Updates: Food Safety Survey, FDA.
12.4 Listing of Reported Adoptions by Local, State, and National Jurisdictions, FDA, CFSAN.
12.5 Baseline: National Association of Retail Druggists.
Update: American Society for Automated Pharmacies.
12.6 Baseline: Primary Care Provider Surveys, OPHS, ODPHP.
Update: Prevention in Primary Care Study, American College of Preventive Medicine.
12.7 MedWatch, FDA.
12.8 FDA.
Healthy People 2000 Final Review 193
Food and Drug
Safety Objectives
12.1: Reduce infections caused by key
foodborne pathogens to incidences of no
more than:
2000 target
Disease (per 100,000)
Salmonella species 16
Campylobacter 25
Escherichia coli O157:H7 4
Listeria monocytogenes 0.5
12.2: Reduce outbreaks of infections due
to Salmonella enteritidis to fewer than
25 outbreaks yearly.
12.3: Increase to at least 75 percent the
proportion of households in which
principal food preparers routinely refrain
from leaving perishable food out of the
refrigerator for over 2 hours and wash
cutting boards and utensils with soap
after contact with raw meat and poultry.
12.4: Extend to at least 70 percent the
proportion of States and territories that
have implemented Food Code 1993 for
institutional food operations and to at
least 70 percent the proportion that have
adopted the new uniform food
protection code that sets recommended
standards for regulation of all food
operations.
12.5: Increase to at least 75 percent the
proportion of pharmacies and other
dispensers of prescription medications
that use linked systems to provide alerts
to potential adverse drug reactions
among medications dispensed by
different sources to individual patients.
12.6: Increase to at least 75 percent the
proportion of primary care providers
who routinely review with their patients
aged 65 and older all prescribed and
over-the-counter medicines taken by
their patients each time a new
medication is prescribed.
12.7: Increase to at least 75 percent the
proportion of the total number of
adverse event reports voluntarily sent
directly to FDA that are regarded as
serious.
12.8: Increase to at least 75 percent the
proportion of people who receive useful
information verbally and in writing for
new prescriptions from prescribers or
dispensers.
194 Healthy People 2000 Final Review

Priority Area 13
Oral Health
Background
Oral health is an essential and
integral component of health throughout
life (1). Poor oral health and untreated
oral diseases and conditions can have a
significant impact on quality of life.
Oral and craniofacial diseases result in
needless pain and suffering; difficulty in
speaking, chewing, and swallowing;
increased costs of care; loss of
self-esteem; decreased economic
productivity through lost work and
school days; and, in extreme cases,
death (2). Conditions significantly
affecting quality of life include dental
caries, periodontal diseases, and oral
cancer. In addition, cleft lip and palate,
oral and facial pain, and reduced
salivary flow affect a substantial
proportion of the general population.
Approximately 70 million persons
in the United States have untreated
dental decay (3), despite dramatic
declines in this infectious disease among
children and adults in recent decades.
Untreated tooth decay is a problem
affecting 16 percent of children 2–4
years of age, 29 percent of those 6–8
years of age, 20 percent of adolescents
15 years of age, and approximately
30 percent of all adults (4). Disparities
of at least 50 percent, with some
approaching 100 percent, exist among
children and adults, by race and
ethnicity, as well as income and level of
education.
Almost 25 percent of U.S. adults
have severe periodontal disease that
results in loss of connective tissue and
bone that support teeth. This is a leading
cause of bleeding, pain, infection, tooth
mobility, and tooth loss. Tobacco use,
especially cigarette smoking, is a
significant risk factor for periodontal
disease, accounting for up to one-half of
all cases of periodontitis (5). Recent
studies pointing to associations between
periodontal diseases and low birth
weight and premature births (6–8), as
well as between periodontitis and heart
disease and stroke (9–11) have
heightened research interest in these
diseases.
Some 31,000 new cases of oral and
pharyngeal cancers were expected to be
diagnosed in 1999, and approximately
8,100 persons were expected to die from
the diseases (12). Oral and pharyngeal
cancers are the 7th most common
cancers found among white males (4th
most common among black males) and
the 14th most common among U.S.
females. The 5-year survival rate for
oral and pharyngeal cancers is only
53 percent (13), and most of these
cancers are diagnosed at late stages (14).
Only 13 percent of U.S. adults 40 years
and over in 1998 reported having had an
oral cancer examination in the past year
(15), which is the recommended interval
(16).
Many persons in the United States
do not receive essential preventive and
treatment services, which results in
disparities in oral health status (17).
Through increased access to appropriate
and timely care, individuals can enjoy
improved oral health. Barriers to care
include cost; lack of dental insurance,
public programs, or providers from
underserved racial and ethnic groups;
and fear of dental visits. Additionally,
some people may have limited oral
health literacy and may not understand
the value of oral health or may not be
able to find or understand information
about how to obtain services.
Data Summary
Highlights
Although the percent of
6–8-year-old children with untreated
dental decay (13.2) increased very
slightly from 28 percent at baseline
(1986–87) to the final data point of
29 percent (1988–94), the prevalence of
dental sealants (13.8) doubled for 8 year
olds—from 11 percent at baseline to
23 percent, still far from its 50 percent
goal. By 1988–94, fewer adolescents 15
years of age (20 percent) had untreated
dental decay (13.2) than in 1986–87
(24 percent); the prevalence of dental
sealants tripled for 14 year olds (13.8),
from 8 percent at baseline to 24 percent.
Black and Hispanic children, whose
baseline levels were substantially higher
for dental caries and lower for sealant
prevalence, showed little improvement
in either untreated decay or dental
sealants. The percent of adolescents 15
years of age who had ever had dental
decay (13.1), improved substantially,
almost to the target of 60 percent,
dropping from 78 percent in 1986–87 to
61 percent in 1988–94. Among children
6–8 years of age, however, little
improvement was noted during that
period—54 percent versus 52 percent.
Oral cancer mortality (13.7) among
both men and women 45–74 years of
age, showed a steady improvement over
the 1990s, surpassing their targets. Male
deaths per 100,000 were reduced from
13.6 in 1987 to 10.4 in 1998; for
females the reduction over the same
time period was from 4.8 to 3.4. Black
males, however, with a rate of 21.0
continue to have extremely high
mortality rates due to oral cancer.
Smokeless tobacco use (13.17) among
12–17-year-old males also reached its
target, going from 6.6 percent in 1988 to
2.2 percent in 1998; improvement
among young adults 18–24 years of age
was less, from 8.9 percent to 6.9 percent
in 1998. Among adults 35–44 years of
age, the percentage who had lost no
teeth (13.3), remained unchanged
(31 percent) during the decade of the
1990s. Fewer adults had complete tooth
loss (13.4) in 1998 compared with
1986—29 percent versus 36 percent. For
adults with incomes of less than
$15,000, the reduction in complete tooth
loss was from 46 percent in 1986 to
41 percent in 1998.
Substantial improvement over the
decade was noted in the percent of
people who self-reported a past year
dental visit. Children 5 years of age
(13.12) and adults over 35 years of age
(13.14) both reached over half of their
goal from baseline. The percentage of
children with a past year dental visit
increased from 66 percent in 1986 to
75 percent in 1997 (with gains for black
children from 51 percent to 72 percent;
for Hispanic children from 51 percent to
63 percent); for adults, the data were
54 percent at baseline and 65 percent in
1998.
Smaller gains were made in the
percent of people served by community
water fluoridation (13.9), oral health
care for infants with cleft lips and/or
palates (13.15), the percent of adults
35–44 years of age with destructive
periodontal disease (13.6), and use of
protective equipment in sporting and
recreation events (13.16).
Summary of Progress
More than three-quarters of the 17
objectives that address oral health
showed progress over the decade, with
one objective (13.7) meeting its target.
Twelve objectives (13.1, 13.4, 13.6,
13.8, 13.9, 13.11–13.17) moved toward
their targets. Smokeless tobacco (13.17)
met the target for adolescents 12–17
Healthy People 2000 Final Review 195

13.1 Dental caries: 6-8 years
15 years
13.2 Untreated dental caries: 6-8 years
15 years
13.3 No tooth loss: 35-44 years
13.4 Complete tooth loss: 65 years and over
13.5 Gingivitis: 35-44 years
13.6 Periodontal diseases: 35-44 years
13.7 Oral cancer deaths: Male 45-74 years
Female 45-74 years
13.8 Protective sealants: 8 years
13.11 Baby bottle tooth decay preventive practices
13.9 Water fluoridation: People served by
fluoridated community water systems
14 years
13.12 Oral health screening, referral, and followup:
Children age 5 years who visited dentist in past year
13.14 Regular dental visits: 35 years and over
13.15 State recording and referring infants
with cleft lip and/or palates
13.16 Use of protective equipment: Baseball - headgear
Baseball - mouth guard
Soccer - headgear
Soccer - mouth guard
13.17 Smokeless tobacco use: Male 12-17 years
Football - mouth guard
Football - headgear
Male 18-24 years
Moved away from target Moved toward target Met target
NOTE: Complete tracking data are shown in table 13. Progress quotients are not calculated for objectives 13.10 and 13.13.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
44%
-64%
-13%
30%
17%
19%
41%
18%
No change
14%
Percent of target achieved
-100 -80 -60 -40 -20 0 20 40 60 80 100
103%
200%
69%
169%
5%
41%
7%
38%
31%
50%
44%
94%
11%
8%
5%
196 Healthy People 2000 Final Review
Figure 13. Final status of Oral Health objectives
years of age (but not for young adults
18–24 years of age, which made
progress). Two objectives (13.5 and
13.10) moved away from their targets.
One objective (13.3) had no change, and
one objective (13.2), showed mixed
progress. See table 13 for the tracking
data for the objectives in this priority
area and figure 13 for a quantitative
assessment of progress.
Discussion
Although progress was made for the
majority of oral health objectives,
generally progress was relatively small.
In addition, some of the data used to
assess progress (for example, the
1988–94 National Health and Nutrition
Examination Survey (NHANES) and the
1992 Fluoridation Census) were
somewhat dated. It is possible that the
availability of more timely data might
have demonstrated more progress.
The level of available resources for
oral health at the local, State, and
Federal levels, including capacity and
infrastructure, declined during the
1990s. Thus development and
implementation of strategies to achieve
the Healthy People 2000 objectives
were compromised. An increased focus
on oral health in the late 1990s, lead by
the Surgeon General’s Report on Oral
Health (1), should help achieve
improvements in oral health and quality
of life for individuals and communities.
If initiatives, partnerships, and
collaborations flourish, then oral health
literacy will increase, access to
preventive and restorative services for
all persons in need will improve,
surveillance of oral diseases or
conditions will be enhanced, and
appropriate research will explore new
ways to improve oral health for
everyone in the United States.
Transition to Healthy People
2010
The Healthy People 2010 oral
health chapter objectives were
developed by a 26-member workgroup,
composed equally of representatives of
Healthy People Consortium member
organizations and of Public Health
Service representatives. An 11-member
Review Panel also participated in the
process that began in late 1997 and was
completed in 1999.
Of the 17 Healthy People 2000
objectives related to oral health, 15
objectives were carried over to Healthy
People 2010 in some form. Three
objectives—oral cancer mortality,
smokeless tobacco, and orofacial
injuries—went to other chapters; in two
instances, two objectives for Healthy
People 2000 were merged into one
objective for Healthy People 2010. Thus
10 of the 17 objectives in the new
Healthy People 2010 chapter were a
part of the Healthy People 2000
objectives. New objectives relate to
stage of detection of oral cancer lesions,
receipt of annual examination for oral
cancer, receipt of dental services for
low-income children, school-based
health centers with oral health
components, health departments or
community centers with oral health
components, oral health surveillance,
and increasing the number of public
health trained professionals in oral
health programs.
Oral health-related objectives are
found throughout the Healthy People
2010 document. Three objectives in
other chapters are of particular note:
oral cancer mortality in the Cancer
chapter, minority oral health
professionals in the Access to Care
chapter, and diabetics who have had a
past year dental visit in the Diabetes
chapter.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the differences between the
two decades of objectives, reflecting
new knowledge and direction for this
area.
Data Issues
Definitions
Operational definitions and data
collection specifications for all Healthy
People 2000 objectives in Priority Area
13 have been published in the National
Center for Health Statistics’ Healthy
People Statistical Notes series (18). Data
issues are discussed and references are
cited for expanded discussions of the
data systems that provide data for the
national objectives. When appropriate,
the text of questionnaire items used to
measure the objectives is also provided.
For objective 13.3, no tooth loss is
defined as having 28 natural teeth
exclusive of third molars. Destructive
periodontal disease (13.6) is defined as
one or more sites with 4 millimeters or
greater loss of tooth attachment.
Objective 13.9 targets optimal
levels of fluoride in community water
systems. Optimal levels of fluoride are
determined by the mean maximum daily
air temperature over a 5–year period and
range between 0.7 and 1.2 parts of
fluoride per 1 million parts of water
(ppm).
Objective 13.11 addresses feeding
practices that prevent baby bottle tooth
decay. The measure used to establish a
baseline for this objective for the total
population, caregivers with less than a
high school education (13.11a), blacks
(13.11c), and Hispanics (13.11d)isfor
children 6–23 months. The preventive
feeding practices are either that the child
no longer uses a bottle, never used a
bottle, or if the child still uses a bottle,
that no bottle was given at bedtime
(excluding bottles with plain water)
during the past 2 weeks.
Data Comparability
Baseline data for 1986–87 from the
National Survey of Dental Caries in
U.S. school children for objectives 13.1,
13.2, and 13.8 and 1988–94 updates
from the NHANES III are not strictly
comparable because of different
sampling designs. The 1986–87 survey
sampled only children attending schools
while the NHANES III sampled all
noninstitutionalized children.
Changes in the National Health
Interview Survey (NHIS) questions on
oral health between 1989 and 1991
affect comparability of information on
the proportion of 5-year old children
and adults 35 years and over who
visited a dentist in the past 12 months
(13.12 and 13.14, respectively). In 1986
and 1989, the question on dental visits
in the past 12 months followed an
introductory statement and questions
about dental visits and problems in the
past 2 weeks (19,20). The introduction
and question on visits in the past 2
weeks were not included in the 1991
and more recent surveys. These may
have differentially affected recall about
visits in the past 12 months. A second
difference is that the proportion of
people who had visited a dentist in the
past 12 months was based on a question
about the interval since the last dental
visit in the 1986 and 1989 surveys.
Since 1991, this measure was obtained
from a question about the number of
visits to a dentist in the past year.
Finally, in 1986 and 1989 oral health
Healthy People 2000 Final Review 197
data for adults were obtained from a
knowledgeable respondent who provided
information for all people in the
household. Beginning in 1991, an adult
sampled from each family provided
information only for himself or herself
and not others in the household. A
knowledgeable adult provided
information for children in all survey
years.
The National Household Survey on
Drug Abuse is used to measure
objective 13.17 regarding smokeless
tobacco use among adolescents. An
improved questionnaire and editing
procedures were introduced with the
1994 survey and affect comparability
with previous years, especially for
tobacco use among adolescents. For
males 12–17 years of age, a smokeless
tobacco user is someone who has used
snuff or chewing tobacco in the
preceding month. For males 18–24 years
of age, a smokeless tobacco user is
someone who has used either snuff or
chewing tobacco at least 20 times and
who currently uses snuff or chewing
tobacco.
Proxy Data
Nationally representative data on
topical or systemic fluoride use among
people not receiving optimally
fluoridated public water are not readily
obtainable (13.10). It is difficult to
identify a national sample of people
who are not served by a fluoridated
water system. Survey interview methods
are limited because many people cannot
accurately state the fluoridation status of
their water supply. For this reason, a
proxy measure—the proportion of all
U.S. residents who use fluoride—is used
as the revised baseline and has been
used to monitor progress toward
achieving this objective. The original
baseline showing use of fluoride
products among people without
fluoridated water was approximated
from the 1989 NHIS data and
information on water fluoridation
patterns in the United States.
The additional data for objective
13.16 (protective sports equipment) are
from the NHIS and represent the
proportions of children playing baseball,
softball, football, or soccer who use
headgear or mouthguards.
References
1. Department of Health and Human Services.
Oral health in America: A report of the Surgeon
General. Rockville, Maryland: National Institutes
of Health, National Institute of Dental and
Craniofacial Research. 2000.
2. Reisine S, Locker D. Social, psychological, and
economic impacts of oral conditions and
treatments. In: Cohen LK and Gift HC, eds.
Disease prevention and oral health promotion:
Socio-dental sciences in action. Copenhagen:
Munksgaard and la Fédération Dentaire
Internationale, 33–71. 1995.
3. National Center for Health Statistics. National
Health and Nutrition Examination Survey III,
1988–1994. Hyattsville, Maryland: Centers for
Disease Control and Prevention, unpublished data.
4. Department of Health and Human Services.
Healthy people 2010. 2d ed. With understanding
and improving health and objectives for improving
health. 2 vols. Washington: U.S. Government
Printing Office. 2000.
5. Tomar SL, Asthma S. Smoking-attributable
periodontitis in the United States: Findings from
NHANES III. J Periodontol 71:743–51. 2000.
6. Dasanayake AP. Poor periodontal health of the
pregnant woman as a risk factor for low birth
weight. Ann Periodont 3:206–11. 1998.
7. Offenbacher S, Katz V, Fertik G, et al.
Periodontal infection as a possible risk factor for
preterm low birth weight. Ann Periodont
67(Suppl.10):1103–13. 1995.
8. Davenport ES, Williams CE, Sterne JA, et al.
The East London study of maternal chronic
periodontal disease and preterm low birth weight
infants: Study design and prevalence data. Ann
Periodont 3:213–21. 1998.
9. Beck JD, Offenbacher S, Williams R, et al.
Periodontitis: A risk factor for coronary heart
disease? Ann Periodont 3:127–41. 1998.
10. Genco RJ. Periodontal disease and risk for
myocardial infarction and cardiovascular disease.
Cardiovascular Reviews and Reports 19:34–40.
1998.
11. Slavkin HC. Does the mouth put the heart at
risk? J Amer Dental Assoc 130:109–13. 1999.
12. Landis SH, Murray T, Bolden S, et al. Cancer
statistics, 1999. Cancer Journal for Clinicians
49:8–31. 1999.
13. Greenlee RT, Murray T, Bolden S, et al.
Cancer statistics, 2000. Cancer Journal for
Clinicians 50:7–33. 2000.
14. National Institutes of Health. SEER cancer
statistics review 1973–1996. Bethesda, Maryland:
National Cancer Institute. 1999.
http://www.seer.ims.nci.nih.gov/Publications/
CSR1973_1996 June 15, 1999.
15. Centers for Disease Control and Prevention,
National Center for Health Statistics. National
Health Interview Survey, unpublished data. 1998.
16. Horowitz AM, Nourjah PA. Patterns of
screening for oral cancer among U.S. adults. J
Public Health Dent 56:331–5. 1996.
17. U.S. General Accounting Office. Report of
Congressional Requestors. Oral health in low-
income populations. Washington: U.S. General
Accounting Office. 2000.
18. Vargas C, Schober S, Gift H. Operational
definitions for year 2000 objectives: Priority area
13, oral health. Healthy people statistical notes; no
12. Hyattsville, Maryland: National Center for
Health Statistics. 1997.
19. Jack SS, Bloom B. Use of dental services and
dental health, United States, 1986. National Center
for Health Statistics. Vital Health Stat 10(165).
1988.
20. Bloom B, Gift HC, Jack SS. Dental services
and oral health, United States, 1989. National
Center for Health Statistics. Vital Health Stat
10(183). 1992.
198 Healthy People 2000 Final Review

Table 13. Oral Health objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
13.1 Dental caries
Children 6-8 years ......................... 1986–87 54% - - - - - - - - - - - -
1
5 2 % --- --- --- --- --- 3 5 %
Adolescents 15 years ....................... 1986–87 78% - - - - - - - - - - - -
1
61% --- --- --- --- --- 60%
a. Children 6-8 years whose parents have
less than high school education ........... 1986–87 70% - - - - - - - - - - - -
1
66% --- --- --- --- --- 45%
b. American Indian/Alaska Native children
6-8 years
Primary or permanent teeth ............. ... --- --- 88% --- --- --- --- --- --- --- 89% 45%
Primary teeth ........................ 1983–84 92% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Permanent teeth ...................... 1983–84 52% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
c. Black children 6-8 years .................. 1986–87 56% - - - - - - - - - - - -
1
5 0 % --- --- --- --- --- 4 0 %
d. American Indian/Alaska Native adolescents
15 years............................. 1983–84 93% - - - 90% - - - - - - - - - - - - - - - - - - - - - 88% 70%
13.2 Untreated dental caries
Children 6-8 years ......................... 1986–87 28% - - - - - - - - - - - -
1
2 9 % --- --- --- --- --- 2 0 %
a. Children whose parents have less than a
high school education................... 1986–87 43% - - - - - - - - - - - -
1
44% --- --- --- --- --- 30%
b. American Indian/Alaska Native children....... 1983–84 64% - - - 72% - - - - - - - - - - - - - - - - - - - - - 69% 35%
c. Black children .......................... 1986–87
§
38% --- --- --- ---
1
36% --- --- --- --- --- 25%
d. Hispanic children ....................... 1982–84
§,2
45% --- --- --- ---
1,2
48% --- --- --- --- --- 25%
Adolescents 15 years ....................... 1986–87 24% - - - - - - - - - - - -
1
20% --- --- --- --- --- 15%
e. Adolescents whose parents have less than a
high school education................... 1986–87 41% - - - - - - - - - - - -
1
29% --- --- --- --- --- 25%
f. American Indian/Alaska Native adolescents .... 1983–84 84% - - - 61% - - - - - - - - - - - - - - - - - - - - - 67% 40%
g. Black adolescents....................... 1986–87 38% - - - - - - - - - - - -
1
29% --- --- --- --- --- 20%
h. Hispanic adolescents .................... 1982–84
§,2
45% --- --- --- ---
1,2
36% --- --- --- --- --- 25%
13.3 No tooth loss
People 35-44 years ........................ 1985–86 31% - - - - - - - - - - - -
1
3 1 % --- --- --- --- --- 4 5 %
13.4 Complete tooth loss
People 65 years and over .................... 1986 36% 32% 32% - - - 30% - - - - - - - - -
‡
30%
‡
29% - - - 20%
a. Low-income people
Annual family income less than $15,000...... 1986 46% 45% 45% - - - 42% - - - - - - - - -
‡
44%
‡
41% - - - 25%
Annual family income below poverty level..... ... --- --- --- --- 48% --- --- ---
‡
46%
‡
44% --- ...
b. American Indian/Alaska Native ............. 1991 42% - - - - - - - - -
3
--- --- --- ---
3
---
3
- - - 29% 20%
13.5 Gingivitis
People 35-44 years ........................ 1985–86 41% - - - - - - - - - - - -
1
4 8 % --- --- --- --- --- 3 0 %
a. Low-income people (annual family income
less than $12,500) ..................... 1985–86 50% - - - - - - - - - - - -
1
66% --- --- --- --- --- 35%
b. American Indian/Alaska Native ............. 1983–84 95% - - - 96% - - - - - - - - - - - - - - - - - - - - - 96% 50%
c. Hispanic .............................. ... --- --- --- --- --- --- --- --- --- --- --- 50%
Mexican American ...................... 1982–84 74% - - - - - - - - - - - -
1
64% --- --- --- --- --- ...
Cuban ............................... 1982–84 79% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Puerto Rican .......................... 1982–84 82% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
See footnotes and key at end of table.
Healthy People 2000 Final Review 199

Table 13. Oral Health objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
13.6 Periodontal diseases
People 35-44 years ........................ 1985-86 25% - - - - - - - - - - - -
1
22% --- --- --- --- --- 15%
13.7* Oral cancer deaths (per 100,000)
Male 45-74 years .......................... 1987 13.6 13.4 12.7 12.2 12.1 11.1 11.0 10.7 10.3 10.4 - - - 10.5
Female 45-74 years ........................ 1987 4.8 4.6 4.6 4.3 4.2 4.0 3.9 3.5 3.5 3.4 - - - 4.1
a. Black male 45-74 years .................. 1990 29.4 . . . 26.9 27.3 26.2 25.2 23.4 22.6 20.6 21.0 - - - 26.0
b. Black female 45-74 years ................. 1990 6.9 . . . 6.9 6.0 5.8 5.7 6.4 5.0 5.2 4.6 - - - 6.9
13.8 Protective sealants
Children 8 years ........................... 1986-87 11% - - - - - - - - - - - -
1
23% --- --- --- --- --- 50%
Adolescents 14 years ....................... 1986-87 8% - - - - - - - - - - - -
1
24% --- --- --- --- --- 50%
a. Black 8 years .......................... 1986-87
§
4 % --- --- --- ---
1
1 1 % --- --- --- --- --- 5 0 %
b. Black 14 years ......................... 1986-87
§
3 % --- --- --- ---
1
5% --- --- --- --- --- 50%
c. Hispanic 8 years ........................ 1986-87
§
9 % --- --- --- ---
1,2
7 % --- --- --- --- --- 5 0 %
d. Hispanic 14 years ....................... 1986-87
§
6 % --- --- --- ---
1,2
7 % --- --- --- --- --- 5 0 %
13.9 Water fluoridation
People served by community water systems ...... 1989 61% - - - - - - 62% - - - - - - - - - - - - - - - - - - - - - 75%
13.10 Topical and systemic fluorides
People in nonfluoridated areas who use fluoride . . . 1989 50% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 85%
Proportion of people (national) using:
Toothpaste containing fluoride ............... ... ---
4
94% --- --- --- --- --- --- --- --- --- ...
Fluoride mouthrinse
Children and adolescents 6–17 years ........ ... ---
5
22.0% - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
People 18 years and over ................ ... ---
5
7.7% - - - - - - - - - - - - - - - - - - - - - 7.1% - - - . . .
Fluoride supplements
Children and adolescents 2–16 years ........ ... ---
5
10.3% - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
13.11* Baby bottle tooth decay
Parents and caregivers who use preventive feeding
practices ................................ 1991 55% . . . . . . - - - - - - - - - - - - - - - - - -
6
65% - - - 75%
a. Parents and caregivers with less than high
school education ...................... 1991 36% . . . . . . - - - - - - - - - - - - - - - - - -
6
38% - - - 65%
b. American Indian/Alaska Native parents and
caregivers ........................... 1985-89 74% - - - - - - - - - - - - - - - - - - - - - - - -
3,6
--- --- 65%
c. Black parents and caregivers .............. 1991 48% . . . . . . - - - - - - - - - - - - - - - - - -
6
57% - - - 65%
d. Hispanic parents and caregivers ............ 1991 39% . . . . . . - - - - - - - - - - - - - - - - - -
6
46% - - - 65%
13.12 Oral health screening, referral, and followup
Children 5 years who visited the dentist in the past
year ................................... 1986 66%
5
60% 63% --- --- --- --- --- 75% 70% --- 90%
a. Black 5 years .......................... 1991 51% . . . . . . - - - - - - - - - - - - - - - 72% 67% - - - 90%
b. Hispanic 5 years........................ 1991 51% . . . . . . - - - - - - - - - - - - - - - 63% 64% - - - 90%
13.13 Oral health care at institutional facilities ....... . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 100%
Nursing facilities ........................... 1990 Required - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Federal prisons............................ ... --- --- --- --- --- --- --- --- --- --- --- ...
Nonfederal prisons ......................... ... --- --- --- --- --- --- --- --- --- --- --- ...
200 Healthy People 2000 Final Review

Table 13. Oral Health objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Juvenile homes............................ ... --- --- --- --- --- --- --- --- --- --- --- ...
Detention facilities.......................... ... --- --- --- --- --- --- --- --- --- --- --- ...
13.14 Regular dental visits
People 35 years and over .................... 1986 54%
5
55% 58% --- 61% --- --- ---
‡
63%
‡
65% - - - 70%
a. Edentulous people 35 years and over ........ 1986 11%
5
13% 13% --- 16% --- --- ---
‡
20%
‡
21% - - - 50%
b. People 65 years and over................. 1986 42%
5
43% 47% --- 51% --- --- ---
‡
55%
‡
56% - - - 60%
c. Black 35 years and over .................. 1991 43% . . . . . . - - - 46% - - - - - - - - -
‡
53%
‡
53% - - - 60%
d. Mexican American 35 years and over ........ 1991 38% . . . . . . - - - 45% - - - - - - - - -
‡
47%
‡
49% - - - 60%
e. Puerto Rican 35 years and over ............ 1991 51% . . . . . . - - - 37% - - - - - - - - -
‡
54%
‡
56% - - - 60%
13.15 Oral health care for infants with cleft lip and/or
palate
Number of States with systems for recording and
referring infants with cleft lip and/or palates
Systems to identify and refer ................ 1989 11 - - - - - - - - - 23 - - - - - - - - - - - - - - - - - - 40
Systems to identify infants .................. 1989 25 - - - - - - - - - 34 - - - - - - - - - - - - - - - - - - 40
Systems to refer for care ................... 1989 20 - - - - - - - - - 31 - - - - - - - - - - - - - - - - - - 40
Systems to identify, refer, and follow up for
care..................................
1993
§
16 ... ... ... ... --- --- --- --- --- --- 40
13.16* Protective equipment in sporting and recreation
events ................................. . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 100%
National Collegiate Athletic Association
Football ................................ 1988 Required - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Hockey ................................ 1988 Required - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Lacrosse ............................... 1988 Required - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
High school football ........................ 1988 Required - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Amateur boxing............................ 1988 Required - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Amateur ice hockey ........................ 1988 Required - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Use of protective headgear and mouth guards
among children who play sports
Baseball/softball
Headgear ........................... ... --- --- 35% --- --- --- --- --- --- 47% --- ...
Mouth guard ......................... ... --- --- 7% --- --- --- --- --- --- 12% --- ...
Football
Headgear ........................... ... --- --- 72% --- --- --- --- --- --- 77% --- ...
Mouth guard ......................... ... --- --- 72% --- --- --- --- --- --- 76% --- ...
Soccer
Headgear ........................... ... --- --- 4% --- --- --- --- --- --- 9% --- ...
Mouth guard ......................... ... --- --- 7% --- --- --- --- --- --- 14% --- ...
13.17* Smokeless tobacco use
Male 12-17 years .......................... 1988 6.6% - - - 5.3% 4.8% 3.9% 5.1% 4.9% 3.5% 3.7% 2.2% - - - 4%
Male 18-24 years .......................... 1987 8.9% - - - 9.9% 8.2% 7.8% 6.9% - - - - - - - - - 6.9% - - - 4%
a. American Indian/Alaska Native
18-24 years ............................. 1986-87
7
18-64 - - -
3
---
3
---
3
---
3
--- --- --- ---
3
--- --- 10%
- - - Data not available. Final objective status:
Met
Toward
Mixed/ no change
Away
Cannot assess
Healthy People 2000 Final Review 201

. . . Category not applicable.
§
Baseline has been revised.
‡
The NHIS was redesigned in 1997. Data may not be comparable with previous years; see Appendix.
1
1988–94 data.
2
Data are for Mexican Americans.
3
Data are unreliable. Relative standard error is greater than 30%.
4
1986 data.
5
1989 data.
6
Data are for children under 2 years. Baseline data are for children 6–23 months.
7
Estimates for different tribes.
NOTE: Data include revisions and, therefore, may differ from those previously published in these reports and other publications.
Objective number Data source
13.1, 13.1c Baseline: National Survey of Dental Caries in U.S. School Children, 1986–87, NIH, NIDR.
Update: National Health and Nutrition Examination Survey, CDC, NCHS.
13.1a Baseline: North Carolina Oral Health School Survey, North Carolina Division of Dental Health, University of North Carolina School of Public Health.
Update: National Health and Nutrition Examination Survey, CDC, NCHS.
13.1b, d Baseline: Survey of Oral Health, IHS.
Update: Oral Health Status and Treatment Needs Survey of American Indians/Alaska Natives, IHS.
13.2, 13.2c, g Baseline: National Survey of Dental Caries in U.S. School Children, 1986–87, NIH, NIDR.
Update: National Health and Nutrition Examination Survey, CDC, NCHS.
13.2a, e Baseline: North Carolina Oral Health School Survey, North Carolina Division of Dental Health, University of North Carolina School of Public Health.
Update: National Health and Nutrition Examination Survey, CDC, NCHS.
13.2b, f Baseline: Survey of Oral Health, IHS.
Update: Oral Health Status and Treatment Needs Survey of American Indians/Alaska Natives, IHS.
13.2d, h Baseline: Hispanic Health and Nutrition Examination Survey, CDC, NCHS.
Update: National Health and Nutrition Examination Survey, CDC, NCHS.
13.3 Baseline: National Survey of Oral Health in U.S. Employed Adults and Seniors, 1985–86, NIH, NIDR.
Update: National Health and Nutrition Examination Survey, CDC, NCHS.
13.4, 13.4a National Health Interview Survey, CDC, NCHS.
13.4b Baseline: Oral Health Status and Treatment Needs Survey of American Indians/Alaska Natives, IHS.
Update: National Health Interview Survey, CDC, NCHS.
13.5, 13.5a Baseline: National Survey of Oral Health in U.S. Employed Adults and Seniors, 1985–86, NIH, NIDR.
Update: National Health and Nutrition Examination Survey, CDC, NCHS.
13.5b Baseline: Survey of Oral Health, IHS.
Update: Oral Health Status and Treatment Needs Survey of American Indians/Alaska Natives, IHS.
13.5c Baseline: Hispanic Health and Nutrition Examination Survey, CDC, NCHS.
Update: National Health and Nutrition Examination Survey, CDC, NCHS.
13.6 Baseline: National Survey of Oral Health in U.S. Employed Adults and Seniors, 1985–86, NIH, NIDR.
Update: National Health and Nutrition Examination Survey, CDC, NCHS.
13.7*, 13.7a, b National Vital Statistics System, CDC, NCHS.
13.8 Baseline: National Survey of Dental Caries in U.S. School Children, 1986–1987, NIH, NIDR.
Update: National Health and Nutrition Examination Survey, CDC, NCHS.
13.9 Fluoridation Census, CDC, NCPS.
13.10 National Health Interview Survey, CDC, NCHS.
13.11*, 13.11a, c, d National Health Interview Survey, CDC, NCHS.
13.11b 1990 Baby Bottle Tooth Decay 5-Year Evaluation Report, IHS.
13.12 National Health Interview Survey, CDC, NCHS.
202 Healthy People 2000 Final Review

Objective number Data source
13.13 Baseline: HCFA.
13.14 National Health Interview Survey, CDC, NCHS.
13.15 State Public Health Dentists Survey, Illinois State Health Department.
13.16* 1988 baseline: CDC, NCPS; NIH, NIDR.
1991 data: National Health Interview Survey, CDC, NCHS.
13.17* For males 18–24 years, National Health Interview Survey, CDC, NCHS.
For males 12–17 years, National Household Survey on Drug Abuse, SAMHSA, OAS.
13.17a Baseline: National Medical Expenditure Survey of American Indians/Alaska Natives, PHS, NCHSR.
Updates: National Health Interview Survey, CDC, NCHS.
*Duplicate objective. See full text of objective following this table.
Healthy People 2000 Final Review 203
Oral Health
Objectives
13.1: Reduce dental caries (cavities) so
that the proportion of children with one
or more caries (in permanent or primary
teeth) is no more than 35 percent among
children aged 6–8 and no more than
60 percent among adolescents aged 15.
13.1a: Reduce dental caries
(cavities) so that the proportion of
children with one or more caries (in
permanent or primary teeth) is no
more than 45 percent among
children aged 6–8 whose parents
have less than a high school
education.
13.1b: Reduce dental caries
(cavities) so that the proportion of
children with one or more caries (in
permanent or primary teeth) is no
more than 45 percent among
American Indian and Alaska Native
children aged 6–8.
13.1c: Reduce dental caries
(cavities) so that the proportion of
children with one or more caries (in
permanent or primary teeth) is no
more than 40 percent among black
children aged 6–8.
13.1d: Reduce dental caries
(cavities) so that the proportion of
adolescents with one or more caries
(in permanent teeth) is no more
than 70 percent among American
Indian and Alaska Native
adolescents aged 15.
13.2: Reduce untreated dental caries so
that the proportion of children with
untreated caries (in permanent or
primary teeth) is no more than
20 percent among children aged 6–8 and
no more than 15 percent among
adolescents aged 15.
13.2a: Reduce untreated dental
caries so that the proportion of
lower socioeconomic status children
aged 6–8 (those whose parents have
less than a high school education)
with untreated dental caries (in
permanent or primary teeth) is no
more than 30 percent.
13.2b: Reduce untreated dental
caries so that the proportion of
American Indian and Alaska Native
children aged 6–8 with untreated
caries (in permanent or primary
teeth) is no more than 35 percent.
13.2c: Reduce untreated dental
caries so that the proportion of
black children aged 6–8 with
untreated caries (in permanent or
primary teeth) is no more than
25 percent.
13.2d: Reduce untreated dental
caries so that the proportion of
Hispanic children aged 6–8 with
untreated caries (in permanent or
primary teeth) is no more than
25 percent.
13.2e: Reduce untreated dental
caries so that the proportion of
lower socioeconomic status
adolescents aged 15 (those whose
parents have less than a high school
education) with untreated dental
caries (in permanent or primary
teeth) is no more than 25 percent.
13.2f: Reduce untreated dental
caries so that the proportion of
American Indian and Alaska Native
adolescents aged 15 with untreated
caries (in permanent or primary
teeth) is no more than 40 percent.
13.2g: Reduce untreated dental
caries so that the proportion of
black adolescents aged 15 with
untreated caries (in permanent or
primary teeth) is no more than
20 percent.
13.2h: Reduce untreated dental
caries so that the proportion of
Hispanic adolescents aged 15 with
untreated caries (in permanent or
primary teeth) is no more than
25 percent.
13.3: Increase to at least 45 percent the
proportion of people aged 35–44 who
have never lost a permanent tooth due
to dental caries or periodontal diseases.
13.4: Reduce to no more than 20 percent
the proportion of people aged 65 and
older who have lost all of their natural
teeth.
13.4a: Reduce to no more than
25 percent the proportion of
low-income people (annual family
income less than $15,000) aged 65
and older who have lost all of their
natural teeth.
13.4b: Reduce to no more than
20 percent the proportion of
American Indians and Alaska
Natives aged 65 and older who
have lost all of their natural teeth.
13.5: Reduce the prevalence of
gingivitis among people aged 35–44 to
no more than 30 percent.
13.5a: Reduce the prevalence of
gingivitis among low-income people
(annual family income less than
$12,500) aged 35–44 to no more
than 35 percent.
13.5b: Reduce the prevalence of
gingivitis among American Indians
and Alaska Natives aged 35–44 to
no more than 50 percent.
13.5c: Reduce the prevalence of
gingivitis among Hispanics aged
35–44 to no more than 50 percent.
13.6: Reduce destructive periodontal
diseases to a prevalence of no more than
15 percent among people aged 35–44.
13.7*: Reduce deaths due to cancer of
the oral cavity and pharynx to no more
than 10.5 per 100,000 men aged 45–74
and 4.1 per 100,000 women aged
45–74.
Duplicate objectives: 3.17 and 16.17
13.7a*: Reduce deaths due to
cancer of the oral cavity and
pharynx to no more than 26.0 per
100,000 among black males aged
45–74.
Duplicate objectives: 3.17a and
16.17a
13.7b*: Reduce deaths due to
cancer of the oral cavity and
pharynx to no more than 6.9 per
100,000 among black females aged
45–74.
Duplicate objectives: 3.17b and
16.17b
13.8: Increase to at least 50 percent the
proportion of children who have
received protective sealants on the
occlusal (chewing) surfaces of
permanent molar teeth.
13.8a: Increase to at least
50 percent the proportion ofblack
children aged 8 who have received
protective sealants on the occlusal
(chewing) surfaces of permanent
molar teeth.
13.8b: Increase to at least
50 percent the proportion of black
children aged 14 who have received
protective sealants on the occlusal
(chewing) surfaces of permanent
molar teeth.
204 Healthy People 2000 Final Review
13.8c: Increase to at least 50 percent
the proportion of Hispanic children
aged 8 who have received
protective sealants on the occlusal
(chewing) surfaces of permanent
molar teeth.
13.8d: Increase to at least
50 percent the proportion of
Hispanic children aged 14 who have
received protective sealants on the
occlusal (chewing) surfaces of
permanent molar teeth.
13.9: Increase to at least 75 percent the
proportion of people served by
community water systems providing
optimal levels of fluoride.
13.10: Increase use of professionally or
self-administered topical or systemic
(dietary) fluorides to at least 85 percent
of people not receiving optimally
fluoridated public water.
13.11*: Increase to at least 75 percent
the proportion of parents and caregivers
who use feeding practices that prevent
baby bottle tooth decay.
Duplicate objective: 2.12
13.11a*: Increase to at least
65 percent the proportion of parents
and caregivers with less than a high
school education who use feeding
practices that prevent baby bottle
tooth decay.
Duplicate objective: 2.12a
13.11b*: Increase to at least
65 percent the proportion of
American Indian and Alaska Native
parents and caregivers who use
feeding practices that prevent baby
bottle tooth decay.
Duplicate objective: 2.12b
13.11c*: Increase to at least
65 percent the proportion of black
parents and caregivers who use
feeding practices that prevent baby
bottle tooth decay.
Duplicate objective: 2.12c
13.11d*: Increase to at least
65 percent the proportion of
Hispanic parents and caregivers
who use feeding practices that
prevent baby bottle tooth decay.
Duplicate objective: 2.12d
13.12: Increase to at least 90 percent the
proportion of all children entering
school programs for the first time who
have received an oral health screening,
referral, and followup for necessary
diagnostic, preventive, and treatment
services.
13.12a: Increase to at least
90 percent the proportion of all
black children aged 5 who have
received an oral health screening,
referral, and followup for necessary
diagnostic, preventive, and
treatment services.
13.12b: Increase to at least
90 percent the proportion of
Hispanic children aged 5 who have
received an oral health screening,
referral, and followup for necessary
diagnostic, preventive, and
treatment services.
13.13: Extend to all long-term
institutional facilities the requirement
that oral examinations and services be
provided no later than 90 days after
entry into these facilities.
13.14: Increase to at least 70 percent the
proportion of people aged 35 and older
using the oral health care system during
each year.
13.14a: Increase to at least
50 percent the proportion of
edentulous people using the oral
health care system during each year.
13.14b: Increase to at least
60 percent the proportion of people
aged 65 and older using the oral
health care system during each year.
13.14c: Increase to at least
60 percent the proportion of blacks
aged 35 and older using the oral
health care system during each year.
13.14d: Increase to at least
60 percent the proportion of
Mexican-Americans aged 35 and
older using the oral health care
system during each year.
13.14e: Increase to at least
60 percent the proportion of Puerto
Ricans aged 35 and older using the
oral health care system during each
year.
13.15: Increase to at least 40 the number
of States that have an effective system
for recording and referring infants with
cleft lips and/or palates to craniofacial
anomaly teams.
2000 target
Identification and referral (number of
of infants with clefts States)
States with system to identify
clefts 40
States with system to refer
for care 40
States with system to follow-up 40
States with system to identify
and refer 40
13.16*: Extend requirement of the use
of effective head, face, eye, and mouth
protection to all organizations, agencies,
and institutions sponsoring sporting and
recreation events that pose risk of injury.
Duplicate objective: 9.19
13.17*: Reduce smokeless tobacco use
by males aged 12–24 to a prevalence of
no more than 4 percent.
Duplicate objective: 3.9
13.17a*: Reduce smokeless tobacco
use by American Indian and Alaska
Native youth to a prevalence of no
more than 10 percent.
Duplicate objective: 3.9a
*Duplicate objective.
Healthy People 2000 Final Review 205

Priority Area 14
Maternal and Infant
Health
Background
Improving the health of mothers
and infants remains a national priority.
The health of mothers and infants is
both a reflection of the current health
status of the population and a predictor
of the health of the next generation.
Infant mortality is a particularly
critical measure of a Nation’s health and
a worldwide indicator of health status
and social well-being. In 1998, 28,371
infants died before their first birthday
(1). This represents an infant mortality
rate of 7.2 deaths per 1,000 live births,
the lowest rate ever recorded for the
United States. However, this
achievement masks the persistent
disparities among racial and ethnic
groups; the mortality rate among black
infants remained more than double that
of white infants.
In addition, as infant mortality rates
have declined, the rates of two
important risk factors for infant
mortality, preterm birth and low birth
weight, are increasing (2). Therefore,
concern for maternal and infant health
encompasses not just the immediate
indicators of maternal and infant
survival but the risk and protective
factors that contribute to the health of
this population, including preconception
counseling, early prenatal care,
breastfeeding, newborn screening, and
primary care in infancy.
Data Summary
Highlights
A major achievement in the area of
maternal and infant health is the
reduction in infant mortality (14.1) from
the baseline rate of 10.1 deaths per
1,000 live births in 1987 to 7.2 in 1998.
This overall decline incorporates
decreases in both neonatal mortality
(14.1d) and postneonatal mortality
(14.1g). In fact, the target for
postneonatal mortality of 2.5 deaths per
1,000 live births was exceeded in 1998,
when a rate of 2.4 was recorded.
Significant improvements were seen
within each of the population groups
measured for this objective, including
blacks, American Indians/Alaska
Natives, and particularly Puerto Ricans.
Puerto Ricans have met or exceeded
each of the objectives for infant,
neonatal, and postneonatal mortality.
Progress toward other objectives
was more modest. An increasing
proportion of new mothers of all racial
and ethnic groups were breastfeeding in
the early postpartum period (14.9) and
at 6 months postpartum, with 67 percent
of women breastfeeding in the hospital
and 31 percent still doing so 6 months
later. Increases were noted among low-
income women, black women, Hispanic
women, and American Indians/Alaska
Natives. In addition, a greater
percentage of pregnant women received
prenatal care in the first trimester
(14.11); this proportion increased from
76 percent in 1987 to 83 in 1999. The
rate of cesarean delivery (14.8) has
declined as well, with the majority of
the decrease attributable to the
widespread promotion of vaginal birth
after cesarean (VBAC) delivery for
women who have had a previous
cesarean. The cesarean delivery rate
among women with a previous cesarean
declined from 91.2 percent in 1987 to
69.3 in 1998, approaching the target of
65.
Unfortunately, ground was lost on
several important indicators. The rate of
low birth weight (14.5) has risen
steadily since the baseline year, from
6.9 percent in 1987 to 7.6 in 1998. The
rate of very low birth weight has also
increased, from 1.2 percent to 1.4, over
the same period. (It is notable, however,
that the low birth weight rate among
black infants, while it did not improve
over this period, was essentially stable
at 13 percent.) Low and very low birth
weight infants are at significantly
increased risk of infant death, and those
who survive are more likely to
experience long-term developmental
disabilities. Another disturbing trend is
the increase in the rate of maternal
mortality (14.3), particularly among
black women. This rate has risen from
6.6 maternal deaths per 100,000 live
births to 7.1 for the population as a
whole, and the rate for blacks has
increased from 14.9 to 17.1 in 1998.
It is important to note that the wide
racial and ethnic disparities seen in
many of these objectives did not narrow
substantially. In 1998, there was still a
twofold differential between black and
white rates of infant mortality, and the
maternal mortality rate for blacks
remained three times greater than that of
whites. Black women also had lower
rates of early prenatal care and
breastfeeding than whites or Hispanics.
Summary of Progress
Of the 17 maternal and infant health
objectives included in Healthy People
2000, data are available to assess the
progress for 15 objectives. One
objective (14.7) and five subobjectives
(14.1c, f, and j, infant, neonatal, and
postneonatal mortality among Puerto
Ricians; 14.1g, overall postneonatal
mortality; and 14.15, newborn screening
for galactosemia) have met their targets.
Overall, eight objectives (14.1, 14.2,
14.6, 14.8, 14.9, 14.11, 14.13, 14.15)
have moved toward their targets. For
three objectives (14.3–14.5), movement
has been away from their targets. No
change was recorded for one objective
(14.17).
For two of the remaining objectives
(14.10 and 14.12), progress among the
multiple measures showed mixed trends.
For the remaining two (14.14 and
14.16), no data are available to assess
progress. See table 14 for the tracking
data for the objectives in this priority
area and figure 14 for a quantitative
assessment of progress.
Discussion
The decline in infant mortality over
the past 10 years is likely due to the
widespread application of advances in
medical knowledge. Much of the
decrease in neonatal mortality is
probably attributable to the introduction
in the early 1990s of synthetic
surfactant, which allows small infants
with undeveloped lungs to breathe,
decreasing the incidence of
intraventricular hemorrhage and the
severity of respiratory disease in
preterm, very small infants (3,4). The
dramatic decline in postneonatal
mortality is probably the result of
widespread public education campaigns
about the importance of infant sleep
position. Much research has shown that
nonprone sleeping position (that is,
sleeping on the side or back rather than
the stomach) greatly decreases the risk
of SIDS among infants (5,6). The
American Academy of Pediatrics has
therefore recommended that healthy
full-term infants be put down to sleep
on their backs (7), and the National
Institute for Child Health and Human
206 Healthy People 2000 Final Review

14.1 Infant mortality
14.2 Fetal deaths
14.3 Maternal mortality
14.4 Fetal alcohol syndrome*
14.5 Low birthweight
Very low birthweight
14.6 Recommended weight gain during pregnancy
14.7 Severe complications of pregnancy
14.8 Cesarean delivery
14.9 Breastfeeding: Early postpartum
At 6 months
14.10 Abstinence during pregnancy: Tobacco
Alcohol
Cocaine
Marijuana*
14.15 Newborn screening: Sickle cell anemia
14.17 Spina bifida and other neural tube defects
Galactosemia
14.11 Prenatal care in the first trimester
14.13 Screening for fetal abnormalities
Moved away from target Moved toward target Met target
* For explanation, see Data Comparability in chapter text.
NOTE: Complete tracking data are shown in table 14. Progress quotients are not calculated for objectives 14.12, 14.14, and 14.16.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
31%
-50%
-37%
-15%
41%
62%
80%
37%
49%
13%
36%
No change
No change
Percent of target achieved
-100 -80 -60 -40 -20
0
20 40 60 80 100
20%
108%
129%
94%
-450%
-100%
98%
Healthy People 2000 Final Review 207
Figure 14. Final status of Maternal and Infant Health objective
Development, National Institutes of
Health, and the Maternal and Child
Health Bureau, Health Resources and
Services Administration, instituted the
‘‘Back to Sleep’’ campaign in 1994 to
educate parents and physicians about
this recommendation.
While the achievement of the target
regarding the rate of severe pregnancy
complications is commendable, the
reasons for this progress bear some
explanation. This objective monitors
complications by measuring the number
of hospitalizations for diagnoses related
to pregnancy complications. The 1990s
saw a dramatic increase in the use of
managed care plans for the delivery of
medical services in both the public and
private sectors. One of the major cost-
saving strategies used by these plans is
to substitute ambulatory for inpatient
care whenever possible; therefore, the
statistic reported here may not be a true
reflection of the incidence of severe
pregnancy complications.
The increasing rate of low birth
weight bears further scrutiny as well.
Much of the increase in this rate since
1990 is attributable to increases in the
rate of multiple births, which are much
more likely than singleton births to be
of low birth weight. Because of the
increased use of assisted reproductive
technologies, the number of twin births
has risen 62 percent, and the rate of
triplets or higher-order births rose
470 percent since 1980. These babies
have a low birth weight rate of more
than 50 percent, while the low birth
weight rate among singletons is
approximately 6 percent. In fact, the low
birth weight rate among singleton births
rose only 1 percent between 1989 and
1998 (2).
Transition to Healthy People
2010
The Maternal and Infant Health
chapter of Healthy People 2010 was
developed by an interagency,
multidisciplinary work group co-led by
the Maternal and Child Health Bureau,
Health Resources and Services
Administration, and the Centers for
Disease Control and Prevention. The
group includes researchers,
representatives of Federal agencies, and
national membership organizations. The
workgroup began meeting in October
1997 to review the maternal and infant
health objectives in Healthy People
2000, to propose new objectives, to
consider additions suggested by the
public, and to set targets for the
objectives after they had been selected.
The resulting chapter has become
both broader, expanding to encompass
objectives related to child health, birth
defects, and developmental disabilities,
and deeper, including new indicators of
maternal and infant health that are based
on the considerable scientific progress
of the last 10 years. The chapter has
been retitled the Maternal, Infant, and
Child Health chapter in recognition of
its broader scope, and contains 23
objectives in 8 categories. Objectives
focusing on mortality include, in
addition to infant, fetal and maternal
deaths, perinatal deaths, deaths of
children 5–14 years of age, as well as
those of adolescents and young adults.
Objectives addressing risk factors
include new objectives on preterm births
and infant sleep position. A new, more
detailed focus on developmental
disabilities has been added, with new
objectives on the incidence of several
specific conditions and folic acid intake
among women of childbearing age.
Finally, a set of objectives has been
added that addresses specific issues for
children with special health care needs,
including the provision of medical
homes and Statewide service systems
for these children.
Nearly every chapter of Healthy
People 2010 contains objectives that
relate directly to the goals of the
Maternal, Infant, and Child Health
chapter. These include the objectives
under Access to Quality Health Services
on the accessibility of health care and
health insurance, those under Family
Planning on adolescent pregnancy and
sexual activity, those under
Immunization and Infectious Diseases
on immunization coverage for children,
those under Injury and Violence
Prevention on a range of child injury
risks, and those under Oral Health on
the prevalence of dental disorders and
access to dental care.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the changes between the
two decades of objectives, reflecting
new knowledge and direction for this
area.
Data Issues
Definitions
Operational definitions and data
collection specifications for all Healthy
People 2000 objectives in Priority Area
14 have been published in the National
Center for Health Statistics’ (NCHS)
Healthy People Statistical Notes series
(8). Data issues are discussed and
references are cited for expanded
discussions of the systems that provide
data for the national objectives. When
appropriate, the text of questionnaire
items used to measure the objectives is
also provided.
In 1989, NCHS changed the method
for tabulating race for live births,
assigning the race of mother to the
infant rather than using the previous,
more complicated algorithm for race of
child. This change affects the natality
data by race in this chapter. In addition,
because live births comprise the
denominator of infant mortality
(including neonatal and postneonatal),
maternal mortality, and fetal death rates,
these rates are also affected. These
changes are described in greater detail
in other NCHS publications (9,10).
Quantitatively, the change in the basis
for tabulating live births by race results
in more births to the white population
and fewer births to the black population
and other races. Because of changes in
the denominators, infant mortality rates
(14.1), fetal death rates (14.2), and
maternal mortality rates (14.3) under the
new classification tend to be lower for
white infants and higher for infants of
other races than they would be when
computed by the previous method. For
characteristics of birth such as percent
low birthweight (14.5) and percent
receiving early care (14.11), the racial
disparities tend to be larger when data
are tabulated by race of mother rather
than race of child.
The special target populations for
racial subgroups in this priority area
were monitored with the ‘‘new’’ data by
race of mother. Data prior to 1989 were
recomputed by race of mother to allow
comparable trend comparisons.
Studies indicate that, in the past,
infant mortality for minorities other than
blacks from the mortality files had been
seriously underestimated (11). Therefore,
infant mortality (14.1) for American
Indians/Alaska Natives (AI/AN) and for
Puerto Ricans were monitored with data
208 Healthy People 2000 Final Review
from the Linked Infant Birth and Infant
Death Files, which categorizes deaths by
the race of mother as reported on the
birth certificate. Beginning in 1995, data
are based on a period file using
weighted data and are not strictly
comparable with the unweighted cohort
linked file data used for previous years
(8). The 1995 weighted infant mortality
rates are between less than 1 percent and
5 percent higher than unweighted rates
for 1995 (12).
For objective 14.6, recommended
weight gain is the amount of weight
gained during pregnancy as
recommended in the 1990 National
Academy of Sciences’ report, Nutrition
During Pregnancy.
Data Sources
Data for objective 14.7 come from
the National Hospital Discharge Survey
(NHDS) maintained by NCHS. Data for
the survey are obtained from
approximately 480 hospitals throughout
the United States. Data on race are not
reported by many hospitals due to
omission of a race field on hospital
discharge reporting forms. More
hospitals have automated their discharge
systems in recent years and are using
these forms (UB–82 and UB–92). A
comparison of NHDS data with National
Health Interview Survey (NHIS) data
for people who reported being
hospitalized (NHIS data were adjusted
to exclude hospitalizations of 1 day or
less) indicated that underreporting for
whites was roughly 22 percent in 1991;
the difference in reporting for blacks
was negligible (13).
Data for 14.9 and 14.9a–c are from
the Ross Mothers’ Survey (RMS)
conducted by Abbot Laboratories. The
RMS is an ongoing survey that is
periodically mailed to a probability
sample of new mothers selected from a
list of names that represents
approximately 80 percent of all national
births. In the RMS, mothers are asked to
recall the type of milk their baby was
fed in the hospital and in each
subsequent month up to the month of
the survey. Mothers are considered to be
breastfeeding if they used either human
milk exclusively or human milk in
combination with a supplemental bottled
formula or cow’s milk. In 1988–96, the
questionnaires were mailed to mothers
when their babies were 6 months old. In
1997, the methodology changed and
questionnaires were mailed to a larger
sample of mothers with babies 1–12
months of age. Therefore, although the
overall sample is now approximately
double the pre–1997 size, the number in
the sample for each month (including 6
months) is considerably smaller than
that of previous years. This change
affects the stability of the 6-month
figures now used to monitor this
objective. Also beginning with 1997
data, the RMS no longer collects
information on family income.
Information on education of mother
from the survey is available to measure
socioeconomic status.
Breastfeeding among AI/AN
mothers (14.9d) is tracked by the
Pediatric Nutrition Surveillance System
(PedNSS). The number of participating
States and Indian tribes has varied from
year to year. The fluctuations in State
and tribal participation could affect the
comparability of these data.
Data for objective 14.12 are from
the Primary Care Provider Surveys
(PCPS). The sample was drawn from
the membership rolls of professional
organizations for pediatricians, nurse
practitioners, family physicians,
obstetricians/gynecologists, and
internists. Response rates varied from 50
to 80 percent across these groups. The
data on inquiry about preconception
counseling represent the proportion of
providers who routinely asked
81–100 percent of their patients about
family planning. The data on counseling
refer to the proportion of providers who
routinely counseled 81–100 percent of
their patients who needed the services.
The Prevention in Primary Care
Study (PPCS) was conducted in
1997–98 to update data from the PCPS.
The design and items included in the
1997–98 study were similar to the
PCPS, but a slightly different sampling
frame was used and some items
included in the 1992 surveys were not
included in the PPCS. The professionals
were sampled from listings of all
licensed, active practitioners in the
United States whose practices were at
least 50 percent primary care. Because
of low response rates from the other
provider groups, updates are available
only for nurse practitioners.
Data Comparability
Data on fetal alcohol syndrome
(FAS) (14.4) and spina bifida and other
neural tube defects (14.17) are by year
of birth. Cases received after the end of
the data year are assigned to the year of
the birth. Therefore, data for previous
years include revisions and may differ
from those previously published.
The trends for FAS and spina bifida
should be interpreted with caution.
These data from baseline (1987 and
1990, respectively) through 1993 were
obtained from the Birth Defects
Monitoring Program (BDMP) from
hospitals participating in the
Commission on Professional and
Hospital Activities (CPHA). The number
of participating hospitals has declined
substantially in recent years, resulting in
a decrease in the proportion of U.S.
births covered by the BDMP. In 1981,
24 percent of all births (19 percent of
black births) were covered compared
with only 5 percent (and only 2 percent
of black births) in 1993. As a result, the
relatively small number of births in the
BDMP may not be representative of all
U.S. births. The increasing trend in FAS
may also be a function of improved
identification and reporting, rather than
an actual increase in incidence of the
condition. There was not a sufficient
number of CPHA hospitals in 1994 to
compute a reliable rate for either
objective 14.4 or 14.17. At present, the
BDMP no longer exists.
The decreased number of births in
the BDMP also made tracking FAS for
AI/AN problematic (14.4a). In 1993 the
BDMP contained only about 500 births
(or 1 percent of AI/AN births) to AI/AN
mothers compared with 13 percent in
1981. As a result, FAS data beyond
1990 for AI/AN are considered
unreliable and are not shown in the
table.
In 1996, data from the National
Birth Defects Prevention Network
(NBDPN) became available and is being
used to track objective 14.17. The
NBDPN is a collaborative effort
between the Centers for Disease Control
and Prevention and more than 30 States
to collect comparable population-based
birth defect data.
The data on substance use during
pregnancy (14.10) come from multiple
sources. The 1985 baseline data on
smoking are from the NHIS and the
1988 baseline data on alcohol, cocaine,
and marijuana come from the National
Maternal and Infant Health Survey. The
1992 and 1994–98 updates on tobacco
are from the information listed on the
certificate of live birth and the 1993
updates on all substances are from the
National Pregnancy and Health Survey.
Although the estimates from these
sources are relatively consistent,
Healthy People 2000 Final Review 209
differences in methodology among the
data systems suggest that changes over
time should be interpreted with caution.
Proxy Data
Objective 14.13 addresses the
percent of women enrolled in prenatal
care who are offered screening and
counseling on prenatal detection of fetal
abnormalities. The data used to track the
objective are the number of pregnant
women (per 100 live births) who were
screened for alpha-fetoprotein levels for
the purpose of detecting babies with
fetal Down’s syndrome (8).
References
1. Murphy SL. Deaths: Final data for 1998.
National vital statistics reports; vol 48 no11.
Hyattsville, Maryland: Public Health Service,
Centers for Disease Control and Prevention. 2000.
2. Ventura SJ, Martin JA, Curtin SC, et al. Births:
Final data for 1998. National vital statistics
reports; vol 48 no 3. Hyattsville, Maryland: Public
Health Service, Centers for Disease Control and
Prevention. 2000.
3. Palta M, Weinstein MR, McGuinness G, et al.
A population study. Mortality and morbidity after
availability of surfactant therapy. Newborn Lung
Project. Archives of Pediatric and Adolescent
Medicine 148(12):1295–1301. 1994.
4. Schoendorf KC, Kiely JL. Birth weight and
age–specific analysis of the 1990 U.S. infant
mortality drop. Was it surfactant? Arch Pediatr
Adolesc Med 151(2):129–34. 1997.
5. Øyen N, Markestad T, Skjærven R, et al.
Combined effects of sleeping position and prenatal
risk factors in sudden infant death syndrome: The
Nordic epidemiological SIDS study. Pediatrics
100(4):613–21. 1997.
6. Willinger M, Hoffman HJ, Hartford RB. Infant
sleep position and risk for sudden infant death
syndrome: Report of meeting held January 13–14,
1994. National Institutes of Health, Bethesda,
Maryland. Pediatrics 93(5):814–819. 1994.
7. American Academy of Pediatrics, AAP Task
Force on Infant Sleep Position and SIDS.
Changing concepts of sudden infant death
syndrome: Implications for infant sleeping
environment and sleep position. Elk Grove
Village, Illinois: American Academy of Pediatrics.
2000.
8. Heck KE, Klein RJ. Operational definitions for
year 2000 objectives: Priority area 14, maternal
and infant health. Healthy people statistical notes
no 14 (revised). Hyattsville, Maryland: National
Center for Health Statistics. 1998.
9. National Center for Health Statistics. Advance
report of final natality statistics, 1989. Monthly
vital statistics report; vol 40 no 8 supp.
Hyattsville, Maryland. 1991.
10. National Center for Health Statistics. Health,
United States, 1992. Hyattsville, Maryland: Public
Health Service. 1993.
11. Hahn RA, Mulinare J, Teutsh SM.
Inconsistencies in coding of race and ethnicity
between birth and death in U.S. infants: A new
look at infant mortality, 1983 through 1985.
JAMA 267:259–63. 1992.
12. Ventura SJ, Martin JA, Curtin SC, et al.
Advance report of final natality statistics, 1996.
Monthly vital statistics report; vol 46 no 11 supp.
Hyattsville, Maryland: National Center for Health
Statistics. 1998.
13. Kozak LJ. Underreporting of race in the
National Hospital Discharge Survey. Advance data
from vital and health statistics; no 265.
Hyattsville, Maryland: Centers for Disease Control
and Prevention, National Center for Health
Statistics. 1995.
210 Healthy People 2000 Final Review

Table 14. Maternal and Infant Health objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
14.1 Infant mortality (under 1 year per 1,000 live births) . . . 1987 10.1 9.2 8.9 8.5 8.4 8.0 7.6 7.3 7.2 7.2 - - - 7
a. Black ................................... 1987 18.8 18.0 17.6 16.8 16.5 15.8 15.1 14.7 14.2 14.3 - - - 11
b. American Indian/Alaska Native ................ 1984 13.4 13.1 11.3 - - - - - - - - - 9.0 10.0 8.7 9.3 - - - 8.5
c. Puerto Rican
1
............................. 1984 12.9 9.9 9.7 - - - - - - - - - 8.9 8.6 7.9 7.8 - - - 8
d. Neonatal mortality (under 28 days per 1,000 live
births) ................................. 1987 6.5 5.7 5.6 5.4 5.3 5.1 4.9 4.8 4.8 4.8 - - - 4.5
e. Neonatal mortality among black ............... 1987 12.3 11.6 11.2 10.8 10.7 10.2 9.8 9.6 9.4 9.5 - - - 7
f. Neonatal mortality among Puerto Rican
1
......... 1984 8.6 6.9 6.1 - - - - - - - - - 6.1 5.6 5.4 5.2 - - - 5.2
g. Postneonatal mortality (28 days–11 months per
1,000 live births).......................... 1987 3.6 3.4 3.4 3.1 3.1 2.9 2.7 2.5 2.5 2.4 - - - 2.5
h. Postneonatal mortality among black infants....... 1987 6.4 6.4 6.3 6.0 5.8 5.6 5.3 5.1 4.8 4.8 - - - 4
i. Postneonatal mortality among American
Indian/Alaska Native infants .................. 1984 7.0 7.0 5.8 - - - - - - - - - 5.1 5.3 4.2 4.3 - - - 4
j. Postneonatal mortality among Puerto Rican
infants
1
................................. 1984 4.3 3.0 3.5 - - - - - - - - - 2.8 3.0 2.5 2.6 - - - 2.8
14.2 Fetal deaths ................................ 1987 7.6 7.5 7.3 7.4 7.1 7.0 7.0 6.9 6.8 6.8 - - - 5
(20 weeks or more gestation per 1,000 live births plus
fetal deaths)
a. Black ................................... 1987 13.1 13.3 12.8 13.3 12.8 12.5 12.7 12.5 12.5 12.5 - - - 7.5
14.3 Maternal mortality (per 100,000 live births) ......... 1987 6.6 8.2 7.9 7.8 7.5 8.3 7.1 7.6 8.4 7.1 - - - 3.3
a. Black ................................... 1987 14.9 22.4 18.3 20.8 20.5 18.5 22.1 20.3 20.8 17.1 - - - 5
14.4 Fetal alcohol syndrome (per 1,000 live births)....... 1987 0.22 0.40 0.37 0.52 0.67 - - - - - - - - - - - - - - - - - - 0.12
a. American Indian/Alaska Native ................ 1987 4.0 5.2 - - - - - - - - - - - - - - - - - - - - - - - - - - - 2.0
b. Black ................................... 1987 0.8 1.4 1.9 2.3 5.4 - - - - - - - - - - - - - - - - - - 0.4
14.5 Low birth weight (less than 2,500 grams) 1987 6.9% 7.0% 7.1% 7.1% 7.2% 7.3% 7.3% 7.4% 7.5% 7.6%
p
7.6% 5%
Very low birth weight (less than 1,500 grams) ........ 1987 1.2% 1.3% 1.3% 1.3% 1.3% 1.3% 1.4% 1.4% 1.4% 1.4%
p
1.5% 1%
a. Low birth weight among black infants ........... 1987 13.0% 13.3% 13.6% 13.3% 13.3% 13.2% 13.1% 13.0% 13.0% 13.0%
p
13.1% 9%
b. Very low birth weight among black infants........ 1987 2.8% 2.9% 3.0% 3.0% 3.0% 3.0% 3.0% 3.0% 3.0% 3.1%
p
3.1% 2%
c. Low birth weight among Puerto Rican infants
1
.... 1990 9.0% . . . 9.4% 9.2% 9.2% 9.1% 9.4% 9.2% 9.4% 9.7% - - - 6%
d. Very low birth weight among Puerto Rican
infants
1
................................. 1990 1.6% . . . 1.7% 1.7% 1.7% 1.6% 1.8% 1.7% 1.8% 1.9% - - - 1%
14.6 Recommended weight gain during pregnancy ..... 1980
2,§
68%
3
75% --- --- --- --- --- --- --- --- --- 85%
14.7 Severe complications of pregnancy
(per 100 deliveries) ........................... 1987 22 18 18 17 15 15 14 14 13 13 - - - 15
a. Black ................................... 1991 28 . . . . . . 26 24 25 24 23 21 19 - - - 16
14.8 Cesarean delivery (per 100 deliveries)............. 1987 24.4 23.5 23.5 23.6 22.8 22.0 20.8 21.8 21.5 22.5 - - - 15
a. Primary (first time) cesarean delivery ........... 1987 17.4 16.8 17.1 16.8 16.3 15.8 15.5 15.7 15.6 15.6 - - - 12
b. Repeat cesarean deliveries (females with previous
cesarean delivery) ......................... 1987 91.2 79.6 75.8 74.9 74.6 70.3 64.5 66.4 65.6 69.3 - - - 65
14.9* Breastfeeding
During early postpartum period
4
.................. 1988 54% 52% 53% 54% 56% 57% 60% 59% 62% 64% 67% 75%
a. Low-income mothers........................ 1988
§
34% 35% 33% 35% 38% 40% 42% 42% 46% 47% 49% 75%
b. Black mothers............................. 1988 25% 23% 26% 28% 31% 33% 37% 37% 41% 45% 50% 75%
See footnotes and key at end of table.
Healthy People 2000 Final Review 211

Table 14. Maternal and Infant Health objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
c. Hispanic mothers .......................... 1988 51% 48% 52% 52% 56% 58% 61% 61% 64% 66% 69% 75%
d. American Indian/Alaska Native mothers ......... 1988 47% 47% 46% 53% 51% 44% 52% 54% 56% 57% 62% 75%
At age 6 months .............................. 1988
§
20% 18% 18% 19% 19% 20% 22% 22% 26% 29% 31% 50%
a. Low-income mothers........................ 1988 9% 8% 8% 9% 10% 10% 11% 12% 20% 21% 20% 50%
b. Black mothers............................. 1988
§
7% 6% 7% 8% 9% 10% 11% 11% 15% 19% 20% 50%
c. Hispanic mothers .......................... 1988
§
14% 13% 15% 16% 16% 18% 20% 20% 25% 28% 29% 50%
d. American Indian/Alaska Native mothers ......... 1988 28% 27% 22% 24% 28% 24% 24% 24% 25% 26% 27% 50%
14.10 Abstinence from alcohol, tobacco, and drug use
during pregnancy
Tobacco .................................... 1985 75% 79% 80% 83% 80% 85% 86% 86% 87% 87% - - - 90%
Alcohol ..................................... 1988 79% - - - - - - - - - 81% - - - - - - - - - - - - - - - - - - 95%
Cocaine .................................... 1988 99% - - - - - - - - - 99% - - - - - - - - - - - - - - - - - - 100%
Marijuana ................................... 1988 98% - - - - - - - - - 97% - - - - - - - - - - - - - - - - - - 100%
14.11 Prenatal care in the first trimester (percent
of live births) ................................ 1987 76.0% 75.8% 76.2% 77.7% 78.9% 80.2% 81.3% 81.9% 82.5% 82.8%
p
83.2% 90%
a. Black ................................... 1987 60.8% 60.6% 61.9% 63.9% 66.0% 68.3% 70.4% 71.4% 72.3% 73.3%
p
74.0% 90%
b. American Indian/Alaska Native ................ 1987 57.6% 57.9% 59.9% 62.1% 63.4% 65.2% 66.7% 67.7% 68.1% 68.8% - - - 90%
c. Hispanic
1
................................ 1987 61.0% 60.2% 61.0% 64.2% 66.6% 68.9% 70.8% 72.2% 73.7% 74.3%
p
74.5% 90%
14.12* Age-appropriate preconception counseling by
clinicians .................................. ... --- --- --- --- --- --- --- --- --- --- --- 60%
Percent of clinicians routinely providing service
to 81–100% of patients
Inquiry about family planning (female, childbearing age)
Pediatricians ............................... 1992 18% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 60%
Nurse practitioners .......................... 1992 53% . . . . . . . . . - - - - - - - - - - - - - - -
5
42% - - - 60%
Obstetricians/gynecologists .................... 1992 48% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 60%
Internists .................................. 1992 24% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 60%
Family physicians ........................... 1992 28% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 60%
Counseling about family planning
Pediatricians ............................... 1992 36% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 60%
Nurse practitioners .......................... 1992 53% . . . . . . . . . - - - - - - - - - - - - - - -
5
40% - - - 60%
Obstetricians/gynecologists .................... 1992 65% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 60%
Internists .................................. 1992 26% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 60%
Family physicians ........................... 1992 36% . . . . . . . . . - - - - - - - - - - - - - - -
5,6
--- --- 60%
14.13 Screening for fetal abnormalities (percent of live
births) .................................... 1988 29% - - - - - - 51% - - - - - - - - - - - - - - - - - - - - - 90%
14.14 Pregnant women and infants receiving
risk-appropriate care......................... ... --- --- --- --- --- --- --- --- --- --- --- 90%
14.15 Newborn screening and treatment
Screened by State-sponsored programs for genetic
disorders and other conditions................... ... --- --- --- --- --- --- --- --- --- --- --- 95%
Testing positive for disease and receiving appropriate
treatment .................................. ... --- --- --- --- --- --- --- --- --- --- --- 90%
See footnotes and key at end of table.
212 Healthy People 2000 Final Review

Table 14. Maternal and Infant Health objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Sickle cell anemia screening ..................... 1987
7
33%
8
89% --- --- --- --- --- --- --- --- --- 90%
Black infants ............................... 1987
7
57%
9
77% --- --- --- --- --- --- --- --- --- 95%
Galactosemia screening (38 States) ............... 1987 70% 97% - - - - - - - - - - - - - - - - - - - - - - - - - - - 95%
Newborns diagnosed positive for sickle cell anemia
receiving treatment ........................... ... --- 95% --- --- --- --- --- --- --- --- --- 90%
Newborns diagnosed positive for galactosemia
receiving treatment ........................... ... --- 100% - - - - - - - - - - - - - - - - - - - - - - - - - - - 95%
14.16 Babies receiving primary care .................. ... --- --- --- --- --- --- --- --- --- --- --- 90%
14.17 Spina bifida and other neural tube defects
(per 10,000 live births) ........................ 1990 6 . . . 7 6 7 - - - - - - 6 - - - - - - - - - 3
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
p
Preliminary data.
1
Excludes data from States lacking an Hispanic-origin item on their birth or death certificate or for which Hispanic-origin data were not of sufficient quality.
2
1980 data for married females who had a full-term live birth and prenatal care.
3
1988 data.
4
Breastfed in hospital.
5
1997-98 data.
6
Response rate for this group was too low to produce reliable estimates.
7
Based on 20 States reporting.
8
Based on 43 States reporting.
9
Based on 9 States reporting.
NOTE: Data may include revisions and, therefore, may differ from data previously published in these reports and other publications.
Objective number Data source
14.1, 14.1a-j National Vital Statistics System, CDC, NCHS.
14.2, 14.2a National Vital Statistics System, CDC, NCHS.
14.3, 14.3a National Vital Statistics System, CDC, NCHS.
14.4, 14.4a, b Birth Defects Monitoring Program, CDC, NCEH.
14.5, 14.5a–d National Vital Statistics System, CDC, NCHS.
14.6 Baseline: National Natality Survey, CDC, NCHS.
Update: National Maternal and Infant Health Survey, CDC, NCHS.
14.7 National Hospital Discharge Survey, CDC, NCHS.
14.8, 14.8a, b National Hospital Discharge Survey, CDC, NCHS.
14.9*, 14.9a-c Ross Laboratories Mother Survey.
14.9d Pediatric Nutrition Surveillance System, CDC, NCCDPHP.
14.10 Baseline: National Maternal and Infant Health Survey, CDC, NCHS.
1992 and 1994–98 updates: National Vital Statistics System, CDC, NCHS.
1993 updates: National Pregnancy and Health Survey, NIH, NIDA.
14.11, 14.11a-c National Vital Statistics System, CDC, NCHS.
14.12* Baseline: Primary Care Provider Surveys, OPHS, ODPHP.
Update: Prevention in Primary Care Study, American College of Preventive Medicine.
Met
Toward
Mixed/ no change
Away
Cannot assess
Healthy People 2000 Final Review 213

Objective number Data source
14.13 College of American Pathologists, Foundation for Blood Research.
14.15 Council of Regional Networks for Genetic Services.
14.17 Birth Defects Monitoring Program, CDC, NCEH.
1996 update: National Birth Defects Prevention Network, CDC, NCEH.
* Duplicate objective. See full text of objective following this table.
214 Healthy People 2000 Final Review
Maternal and Infant
Health Objectives
14.1: Reduce the infant mortality rate to
no more than 7 per 1,000 live births.
14.1a: Reduce the infant mortality
rate among blacks to no more than
11 per 1,000 live births.
14.1b: Reduce the infant mortality
rate among American Indians and
Alaska Natives to no more than 8.5
per 1,000 live births.
14.1c: Reduce the infant mortality
rate among Puerto Ricans to no
more than 8 per 1,000 live births.
14.1d: Reduce the neonatal
mortality rate to no more than 4.5
per 1,000 live births.
14.1e: Reduce the neonatal
mortality rate among blacks to no
more than 7 per 1,000 live births.
14.1f: Reduce the neonatal mortality
rate among Puerto Ricans to no
more than 5.2 per 1,000 live births.
14.1g: Reduce the postneonatal
mortality rate to no more than 2.5
per 1,000 live births.
14.1h: Reduce the postneonatal
mortality rate among blacks to no
more than 4 per 1,000 live births.
14.1i: Reduce the postneonatal
mortality rate among American
Indians and Alaska Natives to no
more than 4 per 1,000 live births.
14.1j: Reduce the postneonatal
mortality rate among Puerto Ricans
to no more than 2.8 per 1,000 live
births.
14.2: Reduce the fetal death rate (20 or
more weeks of gestation) to no more
than 5 per 1,000 live births plus fetal
deaths.
14.2a: Reduce the fetal death rate
(20 or more weeks of gestation)
among blacks to no more than 7.5
per 1,000 live births plus fetal
deaths.
14.3: Reduce the maternal mortality rate
to no more than 3.3 per 100,000 live
births.
14.3a: Reduce the maternal
mortality rate among black women
to no more than 5 per 100,000 live
births.
14.4: Reduce the incidence of fetal
alcohol syndrome to no more than 0.12
per 1,000 live births.
14.4a: Reduce the incidence of fetal
alcohol syndrome among American
Indians and Alaska Natives to no
more than 2 per 1,000 live births.
14.4b: Reduce the incidence of fetal
alcohol syndrome among blacks to
no more than 0.4 per 1,000 live
births.
14.5: Reduce low birthweight to an
incidence of no more than 5 percent of
live births and very low birthweight to
no more 1 percent of live births.
14.5a: Reduce low birthweight
among blacks to an incidence of no
more than 9 percent of live births.
14.5b: Reduce very low birthweight
among blacks to no more 2 percent
of live births.
14.5c: Reduce low birthweight
among Puerto Ricans to an
incidence of no more than 6 percent
of live births.
14.5d: Reduce very low birthweight
among Puerto Ricans to no more
1 percent of live births.
14.6: Increase to at least 85 percent the
proportion of mothers who achieve the
minimum recommended weight gain
during their pregnancies.
14.7: Reduce severe complications of
pregnancy to no more than 15 per 100
deliveries.
14.7a: Reduce severe complications
of pregnancy among blacks to no
more than 16 per 100 deliveries.
14.8: Reduce the cesarean delivery rate
to no more than 15 per 100 deliveries.
14.8a: Reduce the primary (first
time) cesarean delivery rate to no
more than 12 per 100 deliveries.
14.8b: Reduce the repeat cesarean
delivery rate to no more than 65 per
100 deliveries among women who
had a previous cesarean delivery.
14.9*: Increase to at least 75 percent the
proportion of mothers who breastfeed
their babies in the early postpartum
period and to at least 50 percent the
proportion who continue breastfeeding
until their babies are 5 to 6 months old.
Duplicate objective: 2.11
14.9a*: Increase to at least
75 percent the proportion of
low-income mothers who breastfeed
their babies in the early postpartum
period and to at least 50 percent the
proportion who continue
breastfeeding until their babies are 5
to 6 months old.
Duplicate objective: 2.11a
14.9b*: Increase to at least
75 percent the proportion of black
mothers who breastfeed their babies
in the early postpartum period and
to at least 50 percent the proportion
who continue breastfeeding until
their babies are 5 to 6 months old.
Duplicate objective: 2.11b
14.9c*: Increase to at least
75 percent the proportion of
Hispanic mothers who breastfeed
their babies in the early postpartum
period and to at least 50 percent the
proportion who continue
breastfeeding until their babies are 5
to 6 months old.
Duplicate objective: 2.11c
14.9d*: Increase to at least
75 percent the proportion of
American Indian and Alaska Native
mothers who breastfeed their babies
in the early postpartum period and
to at least 50 percent the proportion
who continue breastfeeding until
their babies are 5 to 6 months old.
Duplicate objective: 2.11d
14.10: Increase abstinence from tobacco
use by pregnant women to at least
90 percent, increase abstinence from
alcohol by pregnant women to at least
90 percent and increase abstinence from
cocaine and marijuana to 100 percent.
14.11: Increase to at least 90 percent the
proportion of all pregnant women who
receive prenatal care in the first
trimester of pregnancy.
14.11a: Increase to at least
90 percent the proportion of
pregnant black women who receive
prenatal care in the first trimester of
pregnancy.
14.11b: Increase to at least
90 percent the proportion of
pregnant American Indian and
Alaska Native women who receive
prenatal care in the first trimester of
pregnancy.
Healthy People 2000 Final Review 215
14.11c: Increase to at least
90 percent the proportion of
pregnant Hispanic women who
receive prenatal care in the first
trimester of pregnancy.
14.12*: Increase to at least 60 percent
the proportion of primary care providers
who provide age-appropriate
preconception care and counseling.
Duplicate objective: 5.10
14.13: Increase to at least 90 percent the
proportion of women enrolled in
prenatal care who are offered screening
and counseling on prenatal detection of
fetal abnormalities.
14.14: Increase to at least 90 percent the
proportion of pregnant women and
infants who receive risk-appropriate
care.
14.15: Increase to at least 95 percent the
proportion of newborns screened by
State-sponsored programs for genetic
disorders and other disabling conditions
and to 90 percent the proportion of
newborns testing positive for disease
who receive appropriate treatment.
14.16: Increase to at least 90 percent the
proportion of babies aged 18 months
and younger who receive recommended
primary care services at the appropriate
intervals.
14.17: Reduce the incidence of spina
bifida and other neural tube defects to 3
per 10,000 live births.
*Duplicate objective.
216 Healthy People 2000 Final Review

Priority Area 15
Heart Disease and
Stroke
Background
One of the most important public
health achievements of the 20th century
has been the decline in age-adjusted
mortality rates from cardiovascular
disease (CVD). Between 1950 and 1999,
the age-adjusted death rate from CVD
declined 60 percent (1). Despite this
dramatic improvement, an estimated 60
million Americans have CVD, with
more than 950,000 Americans dying
from the illness each year. On the
whole, CVD accounts for more than
40 percent of all U.S. deaths (2). Heart
disease and stroke, the two main
components of CVD, are the first and
third leading causes of death,
respectively (3). Heart disease and
stroke continue to be major causes of
premature, permanent disability among
working adults (4); stroke alone
accounts for disability among more than
1 million people nationwide. The
economic cost and lost productivity
attributed to CVD was $327 billion in
2000 (5). Coronary heart disease (CHD)
accounts for the largest proportion of
heart disease, with about 12 million
people affected (5). The age-adjusted
death rates, which peaked in the
mid-1960s, declined 60 percent for CHD
and 66 percent for stroke between 1963
and 1998. However, recent age-adjusted
rates suggest a slowing in the rate of
decline for CHD mortality and a
leveling for stroke mortality (6).
Historically, the age-adjusted death rates
for both CHD and stroke have been
higher for African Americans than for
the total U.S. population. Recent data
show that the overall decline in CHD
death rates between 1987 and 1998 was
not as large in the African American
population; however, there was a
narrowing of the disparity observed in
the stroke death rates for African
Americans over this period.
Data Summary
Highlights
Progress has been made in reducing
mortality from CVD. Between 1987 and
1998, the age-adjusted death rate for
CHD (15.1) declined 28 percent,
surpassing the year 2000 target in 1998
with a rate of 97 deaths per 100,000
persons. The age-adjusted mortality rate
for stroke (15.2) declined 17 percent to
25.1 deaths per 100,000 persons in
1998.
Detection and treatment of CVD
risk factors, especially high blood
pressure and high blood cholesterol,
have contributed significantly to the
decline in CHD and stroke death rates
(7,8). The prevalence of high blood
pressure has declined from 39.7 percent
of adults 20–74 years of age (1976–80),
to 23.1 percent (1988–94) (9). The
percent of persons with hypertension
whose blood pressure was controlled
(15.4) increased from 11 percent to
23 percent during the same time period.
Furthermore, the mean serum cholesterol
level (15.6) fell from 213 mg/dL in
1976–80 to 203 mg/dL in 1988–94 and
the percent of persons with high blood
cholesterol (15.7) fell from 27 to 19,
below the year 2000 target set at
20 percent.
Prevention research studies have
shown benefits from engaging in
preventive health and health promotion
behaviors such as quitting smoking and
being physically active (10,11). The
percent of adults 18 years of age and
over who were current smokers (15.12)
fell from 29 percent in 1987 to
24 percent in 1998. Participation in
moderate physical activity (15.11) five
or more times per week increased over
the decade, meeting the year 2000 target
of 30 percent, whereas the proportion of
adults participating in physical activity
seven or more times per week moved
toward, but did not meet the 30 percent
target. Even so, overweight prevalence
(15.10) remains a major public health
challenge, with the percent of
overweight American adults increasing
from 26 percent in 1976–80 to
35 percent in 1988–94. A similar trend
has been observed among all gender and
racial groups and across regions (12).
Another concern is end-stage renal
disease (ESRD). The death rate for
ESRD (15.3) increased from 14.4 deaths
per 100,000 persons at baseline (1987)
to 32 per 100,000 in 1998, moving
away from the target of 13 per 100,000.
Summary of Progress
Of the 17 objectives in the Heart
Disease and Stroke Priority Area, 3
objectives (15.1, 15.7, and 15.8) either
met or exceeded their year 2000 targets.
Data for 12 objectives (15.2, 15.4–15.6,
15.9, and 15.11–15.17) show
improvements toward their respective
year 2000 targets, while the remaining
two objectives (15.3 and 15.10) have
moved away from their targets. See
table 15 for the tracking data for the
objectives in this priority area and
figure 15 for a quantitative assessment
of progress.
Discussion
Reasons for the declines in heart
disease and stroke may vary by region
of the country, socioeconomic status,
and population demographics (that is,
age, sex, and racial/ethnic group).
Prevention efforts and improvements in
early detection, treatment, and care have
resulted in a number of beneficial
trends, which may have contributed to
declines in heart disease and stroke.
These trends include a decline in
cigarette smoking, decrease in mean
blood pressure levels, increase in
controlled hypertension, decrease in
mean blood cholesterol levels, and
improved medical care (1).
In addition to clinical interventions,
public health outreach and education
programs and media campaigns (13–15),
may have contributed significantly to
increasing treatment and control of high
blood pressure, increasing awareness of
high blood cholesterol, and decreasing
prevalence of high blood cholesterol.
Efforts to promote early
identification and treatment of patients
with a heart attack and implementation
interventions to prevent recurrence of
cardiovascular events have also likely
had a significant impact on improving
CVD-related outcomes. Nevertheless,
there are still major challenges that lie
ahead.
The alarming trend in the
prevalence of overweight may be
associated with environmental and
lifestyle changes that occurred in the
United States during this period,
including the availability of more food
and food with higher energy content,
unhealthy eating habits, and diminished
opportunities in daily life to burn energy
(16). In addition, the increase in
overweight may also be associated with
the finding that only about 10 percent of
office visits included counseling directed
toward weight reduction, 19 percent of
visits included counseling for physical
activity, and 23 percent included dietary
counseling (17).
Healthy People 2000 Final Review 217

15.1 Coronary heart disease deaths
15.2 Stroke deaths
15.3 End-stage renal disease
15.4 Controlled high blood pressure
15.5 Taking action to control blood pressure
15.6 Mean serum cholesterol level: 20-74 years
Male
Female
15.7 High blood cholesterol prevalence: 20-74 years
Male
Female
15.8 Awareness of high blood cholesterol condition
15.9 Total dietary fat: Average percent of calories
Met goal for saturated fat
15.10 Overweight prevalence: 20-74 years
Met goal for fat
Saturated fat: Average percent of calories
Male
Female
Adolescents 12-19 years
15.11 Light to moderate physical activity:
5 or more times per week
15.12 Cigarette smoking: 18 years and over
Male
7 or more times per week
Female
15.13 People given blood pressure values
15.17 Laboratory accuracy in cholesterol measurement
Moved away from target Moved toward target Met target
*This objective has moved away from its target. A progress quotient could not be calculated.
NOTE: Complete tracking data are shown in table 15. Progress quotients are not calculated for objectives 15.14, 15.15 and 15.16.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
51%
31%
77%
82%
73%
50%
52%
Away*
29%
36%
31%
42%
52%
Percent of target achieved
-100 -80 -60 -40 -20 0 20 40 60 80 100
109%
140%
100%
100%
100%
114%
25%
43%
-150%
-250%
-143%
-1,257%
91%
84%
218 Healthy People 2000 Final Review
Figure 15. Final status of Heart Disease and Stroke objectives
The higher ESRD death rate may be
associated with complications from
prolonged periods of undiagnosed
hypertension or type 2 diabetes (18).
Screening for, and aggressive treatment
of, hypertension and hyperglycemia are
likely interventions to reduce the
incidence of ESRD and subsequent
death.
Transition to Healthy People
2010
Like the preceding Healthy People
2000 Heart Disease and Stroke chapter,
the Healthy People 2010 focus area
addresses broad issues related to CVD
and stroke, and the associated
modifiable risk factors of high blood
pressure and high blood cholesterol. The
Healthy People 2010 goal of improving
cardiovascular health and quality of life
through the prevention of the
development of risk factors; detection,
and treatment of risk factors; early
identification and treatment of heart
attacks and strokes; and prevention of
recurrent cardiovascular events, builds
on the detection, evaluation, and
management focus of Healthy People
2000. There is also an expanded focus
on reducing cardiovascular- and
stroke-related health disparities among
racial, ethnic, and geographic
populations, as well as an emphasis on
the critical need for new, innovative
science-based prevention methods that
can be widely disseminated and utilized
by professionals, patients, and the public
in general.
As in Healthy People 2000, Healthy
People 2010 also highlights related
objectives focusing on key components
(for example, risk factors,
health/medical services, community
outreach, effective communication) to
the prevention, detection, and treatment
of heart disease and stroke. These
objectives can be found in the chapters
on Access to Quality Health Services,
Chronic Kidney Disease, Educational
and Community-Based Programs, Health
Communication, Nutrition and
Overweight, Physical Activity and
Fitness, Public Health Infrastructure, and
Tobacco Use.
However, there are differences
between the two chapters. Several of the
17 objectives that were in Healthy
People 2000 were not carried over to
Healthy People 2010. A total of nine
objectives were either moved to other
chapters (15.9, 15.10, 15.11, and 15.16)
or dropped from the Healthy People
2010 initiative (15.3, 15.8, 15.12, 15.15,
and 15.17). The eight remaining
objectives from Healthy People 2000
were combined with eight new
objectives in Healthy People 2010,
created to address the four components
of the overall goal for heart disease and
stroke. The new objectives include
knowledge of symptoms of heart attack
and importance of calling 9-1-1,
artery-opening therapy, bystander
response to cardiac arrest,
out-of-hospital emergency cardiac care,
heart failure hospitalizations, knowledge
of early warning signs of stroke, high
blood pressure prevalence, and
LDL-cholesterol level in CHD patients.
With the exception of two objectives
(heart failure hospitalizations and high
blood pressure prevalence), all are
developmental objectives (currently lack
baseline data).
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the differences between the
two decades of objectives, reflecting
new knowledge and direction for this
area.
Data Issues
Definitions
Coronary heart disease deaths (15.1)
are defined by ICD–9 codes 402,
410–414, and 429.2. These are different
from the codes used to define the
category ‘‘Diseases of heart,’’ which
often appears in published tables (see
Appendix table IV).
Objective 15.4 addresses the
proportion of people with hypertension
whose blood pressure is under control.
High blood pressure is defined as blood
pressure greater than or equal to 140
mm Hg systolic and/or 90 mm Hg
diastolic and/or taking antihypertensive
medication. The estimates used to track
this objective define control as
maintaining a blood pressure less than
140 mm HG systolic and 90 mm Hg
diastolic through the use of
antihypertensive medication only and do
not include other nonpharmacologic
treatments such as weight loss, low
sodium diets, and restriction of alcohol.
Objective 15.5, to increase the
proportion of people with hypertension
who are taking action to control their
blood pressure, is measured by
self-reported data from the National
Health Interview Survey (NHIS). In this
survey, people with high blood pressure
are defined as those who report that
they have been told they have high
blood pressure on two or more
occasions by a doctor or health
professional. People with high blood
pressure were considered to be taking
action if they report that they were told
either to take blood pressure medication,
diet to lose weight, cut down on salt, or
exercise. High blood cholesterol (15.7)
is defined as serum cholesterol levels of
240 mg/dL or higher (19).
Overweight (15.10) for adults is
defined as a body mass index (BMI) of
27.8 kilograms per meter squared for
males, and 27.3 kilograms per meter
squared for females. For adolescents,
overweight is the sex- and age-specific
85th percentile from NHANES II.
Current international research indicates
that a lower BMI of 25 kilograms per
meter squared may be more clinically
relevant to increased risk of
cardiovascular disease (20,21). See the
Nutrition chapter (Priority Area 2) for
additional information.
Beginning in 1992, the definition of
current smoker (15.12) was modified to
specifically include persons who smoked
only ‘‘some days.’’ Prior to 1992, a
current smoker was defined by the
questions ‘‘Have you ever smoked 100
cigarettes in your lifetime?’’ and ‘‘Do
you smoke now?’’ In 1992, cigarette
smoking data were collected for a
half-sample of the National Health
Interview Survey with half the
respondents (one-quarter sample) using
these two smoking questions and the
other half of respondents (one-quarter
sample) using a revised smoking
question: ‘‘Do you smoke everyday,
some days, or not at all?’’ in place of
the second question. The 1992 estimate
combines data collected using both sets
of questions. Updates after 1992 are
based completely on the revised
definition, which is considered a more
complete estimate of smoking
prevalence. The effect of the new
definition is a small increase in the
number of smokers.
Objective 15.15 seeks to increase
the proportion of primary care providers
who provide appropriate therapy for
high blood cholesterol. This objective
was tracked by the median blood
cholesterol level at initiation of diet and
drug therapy. In 1990, 54 percent of
physicians reported that they initiate diet
Healthy People 2000 Final Review 219
therapy and 60 percent initiate drug
therapy at these median levels.
Data Comparability
Overweight (15.10) was tracked
with two data sources. The primary data
source was NHANES, which provided
baseline data for most of the overweight
objectives and the 1988–94 updates;
these data are derived from measured
height and weight. The second data
source was NHIS. This survey provides
interim estimates shown in an earlier
publication (22), updates for Hispanic
females and American Indians/Alaska
Natives, and all data for people with
disabilities. NHIS estimates are based on
self-reported heights and weights and
are not comparable with the actual
measured data from NHANES;
prevalence estimates of overweight from
self-reported height and weight are
lower. Trends from the NHIS self-report
measures, like those from NHANES,
show a steady increase in prevalence of
overweight; this increase is, however,
different in magnitude from that
observed in the data derived from
measured height and weight.
Objective 15.11 (light-to-moderate
physical activity) was tracked with the
NHIS. The list of activities asked for on
the NHIS was not identical from year to
year. The 1985 and 1990 surveys did
not ask about some activities for people
65 years and over; thus, the data shown
are for people 18–64 years of age. The
1991, 1995, and 1998 surveys asked
about some different activities than the
previous surveys, but people of all age
groups were asked the same questions.
Because of these differences, 1985 and
1990 data are not comparable to later
data. (See Priority Area 1 for more
information.)
Objective 15.13 addresses blood
pressure screening and whether people
know if their blood pressure is normal
or high. Baseline data and 1990 updates
show the proportion of people 18 years
of age and over who had their blood
pressure measured within the preceding
2 years by a health professional or other
trained observer and who were given the
diastolic and systolic values of the
measure. The 1991, 93, 94, and 98
updates are the proportion of people 18
years of age and over who had their
blood pressure checked and can state
whether their blood pressure was high,
low, borderline, or normal.
The 1985 and 1992 data for
objective 15.16 are from the National
Survey of Worksite Health Promotion
Activities, which were telephone surveys
of nongovernment worksites of 50 or
more employees. Worksites were
sampled, because different worksites
within the same company could have
different sets of health promotion
activities. Both active (for example,
classes) and passive (for example,
brochures) methods were counted as
worksite health promotion activities. The
1995 update comes from the Centers for
Disease Control and Prevention-
sponsored Worksite Benchmark Survey,
which used a methodology very similar
to the 1992 survey but did not include
passive methods of health promotion
(23,24).
References
1. Centers for Disease Control and Prevention.
Declines in death from heart disease and
stroke—United States, 1990–99. MMWR
48(30):649–55. 1999.
2. National Heart Lung and Blood Institute.
Morbidity and mortality: 2000 chartbook on
cardiovascular, lung, and blood disease. Bethesda,
Maryland: National Institutes of Health, Public
Health Service. 2000.
3. Murphy SL. Deaths: Final data for 1998.
National vital statistics reports; vol 48 no 11.
Hyattsville, Maryland: National Center for Health
Statistics. 2000.
4. Department of Health and Human Services.
Healthy people 2010. 2nd ed. With understanding
and improving health and objectives for improving
health. 2 vols. Washington: US Government
Printing Office. 2000.
5. National Heart, Lung, and Blood Institute. Fact
book fiscal year 1999. Bethesda, Maryland:
National Institutes of Health, Public Health
Service. 2000.
6. National Heart, Lung, and Blood Institute. Fact
book fiscal year 1996. Bethesda, Maryland:
National Institutes of Health, Public Health
Service. 1997.
7. Roccella EJ, Horan MJ. The National High
Blood Pressure Education Program: Measuring
progress and assessing its impact. Health Psychol
(Suppl.7): 237–303. 1998.
8. Long-Term Intervention with Pravastatin in
Ischaemic Disease (LIPID) Study Group.
Prevention of cardiovascular events and death with
pravastatin in patients with coronary heart disease
and a broad range of initial cholesterol levels.
New Engl J Med 339(19): 1349–57. 1998.
9. National Center for Health Statistics. Health,
United States, 2000. Hyattsville, Maryland: Public
Health Service. 2000.
10. The Health Benefits of Smoking Cessation. A
report of the Surgeon General. Atlanta, Georgia:
Department of Health and Human Services, Public
Health Service, Centers for Disease Control and
Prevention, National Center for Disease Prevention
and Health Promotion, Office on Smoking and
Health. 1990.
11. He J, Whelton PK, Appel LJ, et al. Long-term
effects of weight loss and dietary sodium
reduction on incidence of hypertension.
Hypertension 35(2): 544–9. 2000.
12. Mokdad AH, Serdula MK, Dietz WH, et al.
The spread of the obesity epidemic in the United
States, 1991–1998. JAMA 282 (16): 1519–22.
1999.
13. National Heart Attack Alert Program
coordination committee: Working group report on
educational strategies to prevent prehospital delay
in patients at high risk for acute myocardial
infarction. Bethesda, Maryland: National Institutes
of Health, National Heart, Lung and Blood
Institute. 1997.
14. National High Blood Pressure Education
Program: National High Blood Pressure Education
Program Working Group Report on Primary
Prevention of Hypertension. Bethesda, Maryland:
National Institutes of Health, National Heart, Lung
and Blood Institute. 1993.
15. Cleeman JI and Lenfant C. The National
Cholesterol Education Program: Progress and
prospects. JAMA 280 (24): 2099–2104. 1998.
16. Koplan JP, Dietz WH. Caloric imbalance and
public health policy. JAMA 282 (16):1579–81.
1999.
17. Centers for Disease Control and Prevention.
Missed opportunities in preventive counseling for
cardiovascular disease—United States 1995.
MMWR 47(5): 91–5. 1998.
18. Reikes ST. Trends in end-stage renal disease.
Epidemiology, morbidity, and mortality. Postgrad
Med 108(1):124–6, 129–31, 135–6. 2000.
19. Johnson CL, et al. Declining serum total
cholesterol levels among U.S. adults. JAMA
269(23):3002–23. 1993.
20. Flegal KM, Carroll MD, Kuczmarski RJ, et al.
Overweight and obesity in the United States:
Prevalence and trends, 1960–94. Int J Obes
22(1)39–47. 1998.
21. Kuczmarski RJ, Carroll MD, Flegal KM, et al.
Varying body mass index cutoff points to describe
overweight prevalence among U.S. adults:
NHANES III (1988–94). Obesity Research
5(6):542–8. 1997.
22. National Center for Health Statistics. Healthy
people 2000 review, 1992. Hyattsville, Maryland:
Public Health Service. 1993.
23. Department of Health and Human Services.
Business Responds to AIDS Benchmark Survey:
Technical report. Atlanta, Georgia: Public Health
Service, Centers for Disease Control and
Prevention. 1996.
24. Centers for Disease Control and Prevention.
Cancer screening offered by worksites—United
States, 1992 and 1995. MMWR 46(19):421–4.
1997.
220 Healthy People 2000 Final Review

Table 15. Heart Disease and Stroke objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
15.1* Coronary heart disease deaths (age adjusted
per 100,000) ........................... 1987 135 22 118 114 114 110 108 105 100 97 - - - 100
a. Black .............................. 1987 168 158 156 151 154 147 147 140 136 133 - - - 115
15.2* Stroke deaths (age adjusted per 100,000) ..... 1987 30.4 27.7 26.8 26.2 26.5 26.5 26.7 26.4 25.9 25.1 - - - 20.0
a. Black .............................. 1987 52.5 48.4 46.8 45.0 45.0 45.4 45.0 44.2 42.5 41.4 - - - 27.0
15.3 End-stage renal disease (per 100,000) ....... 1987 14.4 19.2 21.4 23.4 24.1 28.8 27.8 28.7 30.8 32.0 - - - 13.0
a. Black .............................. 1987 34.0 47.8 52.7 57.2 59.5 69.6 69.1 71.5 72.3 75.4 - - - 30.0
15.4* Controlled high blood pressure
People with high blood pressure 18–74 years . . . 1976–80 11% - - - - - - - - - - - -
1
23% --- --- --- --- --- 50%
a. Males with high blood pressure 18–74 years. 1976–80 6% - - - - - - - - - - - -
1
17% --- --- --- --- --- 40%
b. Mexican Americans with high blood pressure
18–74 years........................ 1988–94
§
14% ... ... ... ... ... --- --- --- --- --- 50%
c. Females with high blood pressure 70 years
and over .......................... 1988–94
§
19% ... ... ... ... ... --- --- --- --- --- 50%
15.5 Taking action to control blood pressure
People with high blood pressure 18 years
and over .............................. 1985 79% 80% - - - - - - - - - 87% - - - - - - - - - 89% - - - 90%
a. White hypertensive male 18–34 years...... 1985 51% 54% - - - - - - - - - 64% - - - - - - - - - 57% - - - 80%
b. Black hypertensive male 18–34 years ...... 1985 63% 56% - - - - - - - - - 70% - - - - - - - - - 67% - - - 80%
15.6* Mean serum cholesterol level (mg/dL)
People 20–74 years ...................... 1976–80 213 - - - - - - - - - - - -
1
203 --- --- --- --- --- 200
Male 20–74 years ........................ 1976–80 211 - - - - - - - - - - - -
1
202 --- --- --- --- --- 200
Female 20–74 years ...................... 1976–80 215 - - - - - - - - - - - -
1
204 --- --- --- --- --- 200
15.7* High blood cholesterol prevalence
People 20–74 years ...................... 1976–80 27% - - - - - - - - - - - -
1
19% --- --- --- --- --- 20%
Male 20–74 years ........................ 1976–80 25% - - - - - - - - - - - -
1
18% --- --- --- --- --- 20%
Female 20–74 years ...................... 1976–80 29% - - - - - - - - - - - -
1
20% --- --- --- --- --- 20%
15.8 Awareness of high blood cholesterol
condition
Adults 18 years and over with high blood
cholesterol ............................ 1988 30% 44% - - - - - - - - - - - - 60% - - - - - - - - - - - - 60%
15.9* Dietary fat intake among people 2 years and
over
2
National Health and Nutrition Examination Survey
Average percent of calories from total fat..... 1976–80
3
36% --- --- --- ---
1
34% --- --- --- --- --- 30%
Average percent of calories from saturated fat . 1976–80
3
13% --- --- --- ---
1
12% --- --- --- --- --- 10%
Percent who met goal for total fat .......... 1988–94
§
27% ... ... ... ... ... --- --- --- --- --- 50%
Percent who met goal for saturated fat ...... 1988–94
§
29% ... ... ... ... ... --- --- --- --- --- 50%
Continuing Survey of Food Intakes by Individuals
Average percent of calories from total fat .... 1989–91 34% . . . . . . - - - - - - 33% 33% 33% - - - - - - - - - 30%
Average percent of calories from saturated fat . 1989–91 12% . . . . . . - - - - - - 11% 11% 11% - - - - - - - - - 10%
Percent who met goal for total fat .......... 1989–91 22% . . . . . . - - - - - - 32% 33% 34% - - - - - - - - - 50%
Percent who met goal for saturated fat ...... 1989–91 21% . . . . . . - - - - - - 34% 35% 36% - - - - - - - - - 50%
See footnotes and key at end of table.
Healthy People 2000 Final Review 221

Table 15. Heart Disease and Stroke objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
15.10* Overweight prevalence (based on measured
height and weight unless otherwise indicated)
Adults 20–74 years ....................... 1976–80 26% - - - - - - - - - - - -
1,4
35% --- --- --- --- --- 20%
Male ................................ 1976–80 24% - - - - - - - - - - - -
1,4
34% --- --- --- --- --- 20%
Female .............................. 1976–80 27% - - - - - - - - - - - -
1,4
37% --- --- --- --- --- 20%
Adolescents 12–19 years .................. 1976–80 15% - - - - - - - - - - - -
1
24% --- --- --- --- --- 15%
a. Low-income female 20–74 years.......... 1976–80 37% - - -
5
47% --- --- --- --- --- --- --- --- 25%
b. Black female 20–74 years............... 1976–80 44% - - - - - - - - - - - -
1,4
52% --- --- --- --- --- 30%
c. Hispanic female 20–74 years ............ ... --- --- --- --- --- --- --- --- --- --- --- 25%
Hispanic female 20 years and over
(self-reported) ..................... ... --- 33% 32% 32% 33% 32% 35% ---
‡
44%
‡
46% --- ...
Mexican American female 20–74 years . . . 1982–84 39% - - - - - - - - - - - -
1,4
50% --- --- --- --- --- ...
Cuban female 20–74 years............ 1982–84 34% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Puerto Rican female 20–74 years ....... 1982–84 37% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
d. American Indian/Alaska Native 20 years and
over .............................. 1984–88 29–75% - - -
6
40%
6
36%
6
48%
6
34%
6
43% - - -
‡,6
46%
‡,6
45% - - - 30%
e. People with disabilities 20 years and over
(self-reported) ....................... 1985 36% - - - 38% 37% 38% 38% 40% - - - - - - - - - - - - 25%
f. Females with high blood pressure 20–74
years .............................. 1976–80 50% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 41%
g. Males with high blood pressure 20–74 years. 1976–80 39% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 35%
h. Mexican American male 20–74 years ...... 1982–84 30% - - - - - - - - - - - -
1,4
37% --- --- --- --- --- 25%
15.11* Light-to-moderate physical activity
People 6 years and over................... ... --- --- --- --- --- --- --- --- --- --- --- 30%
People 18 years and over
5 or more times per week ................ 1985
7
22%
7
23%
8
24% --- --- --- 23% --- --- 30% --- 30%
7 or more times per week ................ 1985
7
16%
7
16%
8
17% --- --- --- 16% --- --- 20% --- 30%
a. Hispanic 18 years and over
5 or more times per week.............. 1991 20% . . . . . . - - - - - - - - - 22% - - - - - - 29% - - - 25%
15.12* Cigarette smoking prevalence
People 18 years and over .................. 1987 29% 25% 26%
9
27% 25% 26% 25% - - -
‡
25%
‡
24% - - - 15%
Male .................................. 1987 31% 28% 28%
9
29% 28% 28% 27% - - -
‡
28%
‡
26% - - - 15%
Female ................................ 1987 27% 23% 23%
9
25% 22% 23% 23% - - -
‡
22%
‡
22% - - - 15%
a. People with high school education or less
20 years and over ................... 1987 34% 31% 31%
9
32% 30% 31% 30% - - -
‡
31%
‡
31% - - - 20%
b. Blue-collar workers 18 years and over ..... 1987 41% 36% 36%
9
36% 34% 39% 36% - - -
‡
37%
‡
36% - - - 20%
c. Military personnel ..................... 1988 42% - - - - - -
9
35% --- --- 32% --- --- 30% --- 20%
d. Black 18 years and over 1987 33% 26% 29%
9
28% 26% 27% 26% - - -
‡
27%
‡
25% - - - 18%
e. Hispanic 18 years and over ............. 1987 24% 23% 20%
9
21% 20% 20% 18% - - -
‡
20%
‡
19% - - - 15%
f. American Indian/Alaska Native 18 years and
over ............................... 1979–87
10
42–70% 38% 31%
9
40% 39% 40% 35% - - -
‡
32%
‡
37% - - - 20%
g. Southeast Asian male .................. 1984–88 55%
11
35%
11
36–41 - - - - - - - - - - - - - - - - - - - - - - - - 20%
h. Females of reproductive age (18–44 years) . 1987 29% 26% 27%
9
28% 26% 27% 26% - - -
‡
26%
‡
25% - - - 12%
i. Pregnant females...................... 1985 25% 19% 20% - - - 20% - - - 18% 14% 13% 13% - - - 10%
j. Females who use oral contraceptives....... 1983 36%
12
26% --- --- --- --- 24% --- --- --- --- 10%
222 Healthy People 2000 Final Review

Table 15. Heart Disease and Stroke objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
15.13 Knowledge of blood pressure values
People given blood pressure values .......... 1985 61% 76% - - - - - - - - - - - - - - - - - - - - - - - - - - - 90%
People who can state blood pressure is high,
low, or normal .......................... ... --- --- 84% --- 85% 84% --- --- --- 85% --- ...
a. Mexican American male 18 years and over . . 1991 69% . . . . . . - - - 68% 68% - - - - - - - - - 62% - - - 90%
15.14 Blood cholesterol checked in past 5 years
People 18 years and over .................. 1993 66% . . . . . . . . . . . . - - - - - - - - - - - - 67% - - - 75%
Ever checked ......................... 1988 59% 65% 63% - - - 71% - - - 75% - - - - - - 72% - - - . . .
Within past 2 years ..................... 1988 52% - - - 50% - - - 54% - - - - - - - - - - - - 57% - - - . . .
Ever checked
a. Black 18 years and over ................ 1991 56% . . . . . . - - - 68% - - - - - - - - - - - - 67% - - - 75%
b. Mexican American 18 years and over ...... 1991 42% . . . . . . - - - 55% - - - - - - - - - - - - 48% - - - 75%
c. American Indian/Alaska Native 18 years and
over .............................. 1991 46% . . . . . . - - - 60% - - - - - - - - - - - - 53% - - - 75%
Within past two years
d. Mexican American 18 years and over 1991 33% . . . . . . - - - 38% - - - - - - - - - - - - 38% - - - 75%
e. American Indian/Alaska Native 18 years and
over .............................. 1991 38% . . . . . . - - - 50% - - - - - - - - - - - - 42% - - - 75%
f. Asians/Pacific Islander 18 years and over . . . 1991 45% . . . . . . - - - 44% - - - - - - - - - - - - 54% - - - 75%
15.15
Primary care providers who provide
appropriate therapy for high blood
cholesterol ........................... ... --- --- --- --- --- --- --- --- --- --- --- 75%
Median cholesterol level when diet therapy is
initiated (mg/dL) ........................ 1986 240–259 200–219 - - - - - - - - - - - - 200–219 - - - - - - - - - - - - . . .
Median cholesterol level when drug therapy is
initiated (mg/dL) ........................ 1986 300–319 240–259 - - - - - - - - - - - - 240–259 - - - - - - - - - - - - . . .
15.16
Worksite blood pressure/cholesterol
education programs
High blood pressure and/or cholesterol activity . . 1992 35.0% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 50%
High blood pressure activity ................ 1985 16.5% - - - - - -
13
29% --- --- --- --- --- --- --- ...
Nutrition education activity ................. 1985 16.8% - - - - - - 31% - - - - - - - - - - - - - - - - - - - - - . . .
Blood pressure screening .................. ... --- --- --- 32% --- --- 16% --- --- --- --- ...
15.17 Laboratory accuracy in cholesterol
measurement.......................... 1985 53%
14
84% --- --- --- --- --- --- --- --- --- 90%
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
‡
The NHIS was redesigned in 1997. Data may not be comparable with previous years; see Appendix.
1
1988–94 data.
2
Estimates are from 1-, 2-, or 3-day dietary data.
3
For people 2–74 years.
4
People 20 years and over.
5
1988–91 data.
6
Estimate derived from self-reported height and weight.
7
Data are for people 18–64 years of age.
8
Operational definition changed from previous tracking data.
Met
Toward
Mixed/ no change
Away
Cannot assess
Healthy People 2000 Final Review 223

9
In 1992, the definition of ‘‘current’’ changed to include ‘‘some days’’ (intermittent smoking).
10
Estimates for different tribes.
11
Vietnamese males only.
12
1988 data.
13
Includes classes, individual counseling, and resource materials.
14
1987 data.
NOTE: Data may include revisions and, therefore, may differ from data previously published in these reports and other publications.
Objective number Data source
15.1*, 15.1a National Vital Statistics System, CDC, NCHS.
15.2*, 15.2a National Vital Statistics System, CDC, NCHS.
15.3, 15.3a United States Renal Data System, NIH.
15.4*, 15.4a–c National Health and Nutrition Examination Survey, CDC, NCHS.
15.5, 15.5a, b National Health Interview Survey, CDC, NCHS.
15.6* National Health and Nutrition Examination Survey, CDC, NCHS.
15.7* National Health and Nutrition Examination Survey, CDC, NCHS.
15.8 Baseline: Health and Diet Survey, FDA.
Update: Cholesterol Awareness Survey, NIH, NHLBI.
15.9* 1976–80 baselines and 1988–94 data: National Health and Nutrition Examination Survey, CDC, NCHS.
1989–91 baselines and 1994–96 updates: Continuing Survey of Food Intakes by Individuals, USDA.
15.10*, 15.10a, b, f, g National Health and Nutrition Examination Survey, CDC, NCHS.
15.10c, h Data for Hispanic: National Health Interview Survey, CDC, NCHS.
Baseline for Mexican American, Cuban, Puerto Rican: Hispanic Health and Nutrition Examination Survey, CDC, NCHS.
Updates for Mexican American: National Health and Nutrition Examination Survey, CDC, NCHS.
15.10d Baseline: IHS, OPEL.
Updates: National Health Interview Survey, CDC, NCHS.
15.10e National Health Interview Survey, CDC, NCHS.
15.11*, 15.11a National Health Interview Survey, CDC, NCHS.
15.12*, 15.12a, b, d, e, h National Health Interview Survey, CDC, NCHS.
15.12c Baseline, 1992, and 1995 updates: Worldwide Survey of Substance Abuse and Health Behaviors Among Military Personnel, DoD, OASD.
1998 update: DoD Survey of Health Related Behaviors, Research Triangle Institute.
15.12f Baseline: CDC, 1987.
Updates: National Health Interview Survey, CDC, NCHS.
15.12g Baseline: Local surveys.
1990 update: Jenkins CH. Cancer risks and prevention practices among Vietnamese refugees. Western J of Med 153:34–9. 1990.
1991 update: Jenkins CNH, et al. Tobacco use in Vietnam: Prevalence, predictors, and the role of the transnational tobacco corporations. JAMA
227(21):1726-31. 1997; Jenkins CNH, et al. The effectiveness of a media-led intervention to reduce smoking among Vietnamese-American men. AJPH
87(6):1031–4. 1997.
15.12i Baseline and 1991 update: National Health Interview Survey, CDC, NCHS.
1993 update: National Health and Pregnancy Survey, NIH, NIDA.
1995 update: National Survey of Family Growth, CDC, NCHS.
1996–98 updates: National Vital Statistics System, CDC, NCHS.
15.12j 1983 and 1988 data: Behavioral Risk Factor Surveillance System, CDC, NCCDPHP.
1995 update: National Survey of Family Growth, CDC, NCHS.
15.13 National Health Interview Survey, CDC, NCHS.
224 Healthy People 2000 Final Review

Objective number Data source
15.14 Baseline: Health and Diet Survey, FDA.
1991 and 1993 updates: National Health Interview Survey, CDC, NCHS.
1995 update: Cholesterol Awareness Survey, NIH, NHLBI.
15.15 Cholesterol Awareness Physicians Survey, NIH, NHLBI.
15.16 1985 and 1992 data: National Survey of Worksite Health Promotion Activities, OPHS, ODPHP.
1995 data: Business Responds to AIDS Benchmark Survey, CDC, NCHSTP.
15.17 Comprehensive Chemistry Survey of Laboratories Using Enzymatic Methods, College of American Pathologists.
* Duplicate objective. See full text of objective following this table.
Healthy People 2000 Final Review 225
Heart Disease and
Stroke Objectives
15.1*: Reduce coronary heart disease
deaths to no more than 100 per 100,000
people.
Duplicate objectives: 1.1, 2.1, and 3.1
15.1a*: Reduce coronary heart
disease deaths among blacks to no
more than 115 per 100,000 people.
Duplicate objectives: 1.1a, 2.1a, and
3.1a
15.2*: Reduce stroke deaths to no more
than 20 per 100,000 people.
Duplicate objectives: 2.22 and 3.18
15.2a*: Reduce stroke deaths
among blacks to no more than 27
per 100,000.
Duplicate objectives: 2.22a and
3.18a
15.3: Reverse the increase in end-stage
renal disease (requiring maintenance
dialysis or transplantation) to attain an
incidence of no more than 13 per
100,000.
15.3a: Reverse the increase in
end-stage renal disease (requiring
maintenance dialysis or
transplantation) among black
persons to attain an incidence of no
more than 30 per 100,000.
15.4*: Increase to at least 50 percent the
proportion of people with high blood
pressure whose blood pressure is under
control.
Duplicate objective: 2.26
15.4a*: Increase to at least
40 percent the proportion of men
with high blood pressure whose
blood pressure is under control.
Duplicate objective: 2.26a
15.4b*: Increase to at least
50 percent the proportion of
Mexican-Americans with high blood
pressure whose blood pressure is
under control.
Duplicate objective: 2.26b
15.4c*: Increase to at least
50 percent the proportion of women
70 years and older with high blood
pressure whose blood pressure is
under control.
Duplicate objective: 2.26c
15.5: Increase to at least 90 percent the
proportion of people with high blood
pressure who are taking action to help
control their blood pressure.
15.5a: Increase to at least
80 percent the proportion of white
hypertensive men aged 18–34 who
are taking action to help control
their blood pressure.
15.5b: Increase to at least
80 percent the proportion of black
hypertensive men aged 18–34 who
are taking action to help control
their blood pressure.
15.6*: Reduce the mean serum
cholesterol level among adults to no
more than 200 mg/dL.
Duplicate objective: 2.27
15.7*: Reduce the prevalence of blood
cholesterol levels of 240 mg/dL or
greater to no more than 20 percent
among adults.
Duplicate objective: 2.25
15.8: Increase to at least 60 percent the
proportion of adults with high blood
cholesterol who are aware of their
condition and are taking action to
reduce their blood cholesterol to
recommended levels.
15.9*: Reduce dietary fat intake to an
average of 30 percent of calories or less
and average saturated fat intake to less
than 10 percent of calories among
people aged 2 and older. In addition,
increase to at least 50 percent the
proportion of people aged 2 and older
who meet the Dietary Guidelines’
average daily goal of no more than
30 percent of calories from fat, and
increase to at least 50 percent the
proportion of people aged 2 and older
who meet the average daily goal of less
than 10 percent of calories from
saturated fat.
Duplicate objectives: 2.5 and 16.7
15.10*: Reduce overweight to a
prevalence of no more than 20 percent
among people aged 20 and older and no
more than 15 percent among adolescents
aged 12–19.
Duplicate objectives: 1.2, 2.3, and 17.12
15.10a*: Reduce overweight to a
prevalence of no more than
25 percent among low-income
women aged 20 and older.
Duplicate objectives: 1.2a, 2.3a, and
17.12a
15.10b*: Reduce overweight to a
prevalence of no more than
30 percent among black women
aged 20 and older.
Duplicate objectives: 1.2b, 2.3b, and
17.12b
15.10c*: Reduce overweight to a
prevalence of no more than
25 percent among Hispanic women
aged 20 and older.
Duplicate objectives: 1.2c, 2.3c, and
17.12c
15.10d*: Reduce overweight to a
prevalence of no more than
30 percent among American Indians
and Alaska Natives.
Duplicate objectives: 1.2d, 2.3d, and
17.12d
15.10e*: Reduce overweight to a
prevalence of no more than
25 percent among people with
disabilities.
Duplicate objectives: 1.2e, 2.3e, and
17.12e
15.10f*: Reduce overweight to a
prevalence of no more than
41 percent among women with high
blood pressure.
Duplicate objectives: 1.2f, 2.3f, and
17.12f
15.10g*: Reduce overweight to a
prevalence of no more than
35 percent among men with high
blood pressure.
Duplicate objectives: 1.2g, 2.3g, and
17.12g
15.10h*: Reduce overweight to a
prevalence of no more than
25 percent among
Mexican-American men.
Duplicate objectives: 1.2h, 2.3h, and
17.12h
15.11*: Increase to at least 30 percent
the proportion of people aged 6 and
older who engage regularly, preferably
daily, in light-to-moderate physical
activity for at least 30 minutes per day.
Duplicate objectives: 1.3 and 17.13
15.11a*: Increase to at least
25 percent the proportion of
Hispanics aged 18 and older who
engage regularly, preferably daily,
226 Healthy People 2000 Final Review
in light to moderate physical
activity for at least 30 minutes per
day 5 or more times per week.
Duplicate objectives: 1.3a and
17.13a
15.12*: Reduce cigarette smoking to a
prevalence of no more than 15 percent
among people aged 18 and older.
Duplicate objectives: 3.4 and 16.6
15.12a*: Reduce cigarette smoking
to a prevalence of no more than
20 percent among people aged 20
and older with a high school
education or less.
Duplicate objectives: 3.4a and 16.6a
15.12b*: Reduce cigarette smoking
to a prevalence of no more than
20 percent among blue-collar
workers aged 18 and older.
Duplicate objectives: 3.4b and 16.6b
15.12c*: Reduce cigarette smoking
to a prevalence of no more than
20 percent among military
personnel.
Duplicate objectives: 3.4c and 16.6c
15.12d*: Reduce cigarette smoking
to a prevalence of no more than
18 percent among blacks aged 18
and older.
Duplicate objectives: 3.4d and 16.6d
15.12e*: Reduce cigarette smoking
to a prevalence of no more than
15 percent among Hispanics aged
18 and older.
Duplicate objectives: 3.4e and 16.6e
15.12f*: Reduce cigarette smoking
to a prevalence of no more than
20 percent among American Indians
and Alaska Natives.
Duplicate objectives: 3.4f and 16.6f
15.12g*: Reduce cigarette smoking
to a prevalence of no more than
20 percent among Southeast Asian
men.
Duplicate objectives: 3.4g and 16.6g
15.12h*: Reduce cigarette smoking
to a prevalence of no more than
12 percent among women of
reproductive age.
Duplicate objectives: 3.4h and 16.6h
15.12i*: Reduce cigarette smoking
to a prevalence of no more than
10 percent among pregnant women.
Duplicate objectives: 3.4i and 16.6i
15.12j*: Reduce cigarette smoking
to a prevalence of no more than
10 percent among women who use
oral contraceptives.
Duplicate objectives: 3.4j and 16.6j
15.13: Increase to at least 90 percent the
proportion of adults who have had their
blood pressure measured within the
preceding 2 years and can state whether
their blood pressure was normal or high.
15.13a: Increase to at least
90 percent the proportion of
Mexican-American men who have
had their blood pressure measured
within the preceding 2 years and
can state whether their blood
pressure was normal or high.
15.14: Increase to at least 75 percent the
proportion of adults who have had their
blood cholesterol checked within the
preceding 5 years.
15.14a: Increase to at least
75 percent the proportion of blacks
who have ever had their blood
cholesterol checked.
15.14b: Increase to at least
75 percent the proportion of
Mexican-Americans who have ever
had their blood cholesterol checked.
15.14c: Increase to at least
75 percent the proportion of
American Indians/Alaska Natives
who have ever had their blood
cholesterol checked.
15.14d: Increase to at least
75 percent the proportion of
Mexican-Americans who have had
their blood cholesterol checked
within the preceding 2 years.
15.14e: Increase to at least
75 percent the proportion of
American Indians/Alaska Natives
who have had their blood
cholesterol checked within the
preceding 2 years.
15.14f: Increase to at least
75 percent the proportion of
Asian/Pacific Islanders who have
had their blood cholesterol checked
within the preceding 2 years.
15.15: Increase to at least 75 percent the
proportion of primary care providers
who initiate diet and, if necessary, drug
therapy at levels of blood cholesterol
consistent with current management
guidelines for patients with high blood
cholesterol.
15.16: Increase to at least 50 percent the
proportion of worksites with 50 or more
employees that offer high blood pressure
and/or cholesterol education and control
activities to their employees.
15.17: Increase to at least 90 percent the
proportion of clinical laboratories that
meet the recommended accuracy
standard for cholesterol measurement.
*Duplicate objective.
Healthy People 2000 Final Review 227

Priority Area 16
Cancer
Background
Cancer is the second leading cause
of death in the United States, accounting
for nearly one out of every four deaths
(1). An estimated 1,220,000 Americans
are expected to be diagnosed with
cancer in 2000 and approximately
552,000 are expected to die of cancer.
These estimates are based on an
increase in the number of older
Americans who are at higher risk for
developing the disease (2); one-half of
the new cases occur in persons 65 years
and over (3). Although cancer remains a
major health problem in the United
States, there is evidence that the
prospects of preventing and surviving
cancer continue to improve. Specifically,
perhaps as much as 50 percent or more
of cancer incidence can be prevented
through smoking cessation and changed
dietary habits (4). The scientific
evidence for smoking as a cause of
cancer has been recognized for over 40
years. The evidence for diet has
emerged over the past decade and has
progressed to the extent that
recommendations for prudent dietary
changes, such as less fat and more fruits
and vegetables, can now be made.
Data Summary
Highlights
Trends for most objectives related
to cancer mortality (16.1–16.5, and
16.17) improved for the total population
in 1998. For all cancers (16.1) the
mortality rate in 1998 surpassed the year
2000 target. For lung cancer mortality,
objective 16.2 met the target that was
set to slow the rise in the rate; until
1990 the trend for lung cancer mortality
had been rising at a rate that would
have exceeded the target. However, the
rate actually declined in 1991 for the
first time in at least 50 years and again
in 1992. Lung cancer mortality remained
level in 1993, then dropped again in
1994–98. In 1998 the age-adjusted death
rate for colorectal cancer (16.5)
continued to decline and surpassed the
year 2000 target. However, the rate of
improvement in cancer mortality for the
total population was not observed for all
population subgroups. Improvement was
also observed in cancer risk factors such
as smoking (16.6) and dietary fat intake
(16.7). Data for 1998 indicate that
substantial progress was made in
increasing the numbers of women
receiving breast exams and
mammograms in the past two years
(16.11). The proportions of people who
have received other cancer screening
tests such as Pap tests (16.12), fecal
occult blood tests and
proctosigmoidoscopy exams (16.13), and
oral exams (16.14) also increased over
the last decade.
Summary of Progress
Seven objectives (16.1–16.3, 16.5,
16.11, 16.15 and 16.17) met the year
2000 targets. Progress toward the year
2000 targets was made for 8 of the 17
objectives (16.4, 16.6–16.8, 16.10,
16.12, 16.13 and 16.16). It should be
noted that in many cases the actual
improvement is small. Progress for two
objectives (16.9 and 16.14) was mixed.
See table 16 for the tracking data for the
objectives in this priority area and
figure 16 for a quantitative assessment
of progress.
Discussion
Cancer death rates vary by gender,
race, and ethnicity. Age-adjusted male
and female cancer death rates peaked in
1990 at 164.7 and 111.7 per 100,000,
respectively. After the peak year,
through 1998, male cancer deaths for all
sites decreased on average by
1.5 percent per year, and female deaths
decreased on average by 0.8 percent per
year.
While there has been significant
progress in reaching the year 2000
targets, all population groups have not
benefitted equally. African Americans
are about 34 percent more likely to die
of cancer than are whites and more than
two times as likely to die of cancer as
Asian/Pacific Islanders, American
Indians, or Hispanics. However, some
specific forms of cancer affect other
ethnic groups at rates higher than the
national average (for example, stomach
and liver cancers among Asian
American populations and colon and
rectum cancers among Alaska Natives).
Also, certain racial and ethnic groups
have lower survival rates than whites for
most cancers. The challenge to
understanding these differences between
the races provides an opportunity to
reduce illness and death and to improve
survival rates.
The growing knowledge in
behavioral and population research and
cancer control science is allowing the
Nation to better understand and address
the burden of cancer. By studying
patterns of cancer, searching for its
causes, more fully comprehending the
behaviors of individuals and
communities, and understanding the
impact of the public health and medical
systems—the tools to effectively reduce
that burden are being created. In
addition, the increased understanding of
cancer’s disproportionate effect on some
population groups is paving the way for
effective interventions.
Transition to Healthy People
2010
Most of the Healthy People 2000
objectives were retained in Healthy
People 2010, some with new targets that
will further challenge the United States
to achieve even better health. Reducing
the number of new cases and deaths
from cancer remains a challenge for
Healthy People 2010. Further
improvement in the cancer risk
behaviors and cancer screening
utilization are important objectives to
achieve reduced cancer.
Several new objectives were
developed in Healthy People 2010.
Prostate and melanoma cancer deaths
have risen rapidly, so new objectives
were introduced. Also, the importance of
following life-long healthy behaviors is
emphasized by adding a focus on
adolescent cancer risk behaviors to
complement the adult risk behavior
objectives. Improving long-term survival
rates is included as a new objective in
Healthy People 2010. To evaluate this,
improved data will be required from
State cancer registries. Therefore,
increasing the number of these registries
is also a new objective.
Some of the topics covered in the
Healthy People 2000 priority area on
Cancer were reorganized into other
focus area chapters in Healthy People
2010. The rationale for this change was
to eliminate duplication of similar
objectives in separate focus areas.
Several objectives in the Cancer focus
area for Healthy People 2000 were
moved to other chapters, including
228 Healthy People 2000 Final Review

-100 -80 -60 -40 -20
0
20 40 60 80 100
16.1 Cancer deaths
16.2 Lung cancer deaths
16.3 Female breast cancer deaths
16.4 Cervical cancer deaths
16.5 Colorectal cancer deaths
16.6 Cigarette smoking prevalence: 18 years and over
Male
Female
16.7 Total dietary fat: Average percent of calories
Met goal for fat
Saturated fat: Average percent of calories
Met goal for saturated fat
16.8 Average servings: Vegetables/fruits
Grain products
Met guidelines: Vegetables/fruits
Use sun screen
16.9 Protection from sun, 18 years and older:
Limit sun exposure
Grain products
Wear protective clothes
16.11 Breast exam/mammogram: 50 years and over
16.12 Pap test, 18 years and over: Ever
Past 3 years
16.13 Colon screening, 50 years and older:
Fecal occult blood test (past 2 years)
Sigmoidoscopy (ever)
16.14 Screening in past year, 50 years and over: Oral exam
Skin exam
16.15 Pap test quality: Monitoring labs
Digital rectal exam
16.16 FDA certified mammography facilities
16.17 Oral cancer deaths: Male 45-74 years
Female 45-74 years
Moved away from target Moved toward target Met target
*This objective has met its target. A progress quotient could not be calculated.
NOTES: Complete tracking data are shown in table 16. Progress quotients are not calculated for objective 16.10.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
FDA is Food and Drug Administration.
250%
550%
120%
100%
111%
103%
200%
180%
175%
Met*
-13%
-18%
80%
35%
40%
42%
71%
13%
54%
55%
43%
25%
36%
33%
50%
52%
67%
29%
31%
7%
-4%
Percent of target achieved
Healthy People 2000 Final Review 229
Figure 16. Final status of Cancer objectives
Nutrition and Overweight, Oral Health
and Tobacco Use.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the differences between the
two decades of objectives, reflecting
new knowledge and direction for this
area.
Data Issues
The death rates shown in objectives
16.1–16.5 are age adjusted to the 1940
U.S. population. (See Appendix for
more information on age adjustment.)
The National Cancer Institute age
adjusts cancer deaths to the 1970 U.S.
population. When the 1970 standard
population is used, the equivalent
baseline, interim, and target rates are all
somewhat higher than those generated
using the 1940 population. However, the
trends are very similar.
Definitions
Beginning in 1992, the definition of
current smoker (16.6) was modified to
specifically include persons who smoked
only ‘‘some days.’’ Prior to 1992, a
current smoker was defined by the
questions ‘‘Have you ever smoked 100
cigarettes in your lifetime?’’ and ‘‘Do
you smoke now?’’ In 1992, cigarette
smoking data were collected for a
half-sample of the National Health
Interview Survey with half the
respondents (one-quarter sample) using
these two smoking questions and the
other half of respondents (one-quarter
sample) using a revised smoking
question: ‘‘Do you smoke everyday,
some days, or not at all?’’ in place of
the second question. The 1992 estimate
combines data collected using both sets
of questions. Updates after 1992 are
based completely on the revised
definition, which is considered a more
complete estimate of smoking
prevalence. The effect of the new
definition is a small increase in the
number of smokers.
Estimates for objective 16.8 (fruit,
vegetable, and grain intakes) exclude
fruits and vegetables eaten as part of
potato chips, condiments, fruit-flavored
candies, jellies, and jams.
Two subobjectives in this chapter,
16.11b (mammograms) and 16.12d (Pap
tests), target women with low income.
Prior to 1993, these subobjectives were
tracked with data for women with
annual family incomes of less than
$10,000. Because of changes in the
poverty level over time, beginning with
data for 1993, these subobjectives were
tracked with data for women with
annual family incomes below the census
poverty threshold (see Appendix for
more information).
Data from the Health Care
Financing Administration indicate that
virtually all laboratories meet the
standards set by the Clinical Laboratory
Improvements Act (16.15). In 1993–96,
only 8 of 3,200 laboratories were cited
with deficiencies and terminated from
medical payments until the deficiencies
were corrected. Therefore, the year 2000
target of 100 for this objective is
considered met.
Data Sources
Data for 1992 for objective 16.10
(tobacco, diet, and cancer screening and
counseling) are from the Primary Care
Provider Surveys (PCPS). The data on
formulation of a diet/nutrition plan
represent the proportion of providers
who routinely queried 81–100 percent of
their patients about these risks. The data
on strategies to quit smoking refer to the
proportion of providers who routinely
provided these services to patients who
needed the services. The sample was
drawn from the membership rolls of
professional organizations for
pediatricians, nurse practitioners, family
physicians, obstetricians/gynecologists,
and internists. Response rates varied
from 50 to 80 percent across these
groups. The data on counseling refer to
the proportion of providers who
routinely provided these services to
patients who needed the services.
The Prevention in Primary Care
Study (PPCS) was conducted in
1997–98 to update data from the PCPS.
The design and items included in the
1997–98 study were similar to the
PCPS, but a slightly different sampling
frame was used and some items
included in the 1992 surveys were not
included in the PPCS. The professionals
were sampled from listings of all
licensed, active practitioners in the
United States whose practices were at
least 50 percent primary care. Because
of low response rates from the other
provider groups, updates are available
only for nurse practitioners.
Data Comparability
For objective 16.16, 1990 baseline
and 1992 update data represent the
proportion of mammography facilities
that were certified by the American
College of Radiology. The 1995–97
updates measure how well the 4,200
facilities performing mammograms met
the Mammogram Quality Standards Act
(MQSA) quality standards. ‘‘No
noncompliances’’ means the facility was
in full compliance with MQSA. Level 1
findings are the most serious and
facilities with level 1 findings receive a
warning letter from the Food and Drug
Administration (FDA) and must respond
to it. Although level 2 and level 3
findings are considered less serious, they
also must be corrected. Information on
the types of violations included in these
levels has been published by FDA (5).
References
1. American Cancer Society. Cancer facts and
figures, 2000. Atlanta, Georgia: American Cancer
Society, Inc. 2000.
2. American Cancer Society. Cancer facts and
figures, 1994. Atlanta, Georgia: American Cancer
Society, Inc. 1994.
3. Reis LAG, Kosary CL, Hankey BF, et al. SEER
Cancer Statistics Review, 1973–1996. Bethesda,
Maryland: National Cancer Institute. 1999.
4. National Cancer Institute, Division of Cancer
Prevention and Control. Fiscal Year 1994 Annual
Report. Rockville, Maryland: National Institutes of
Health. 1994.
5. Center for Devices and Radiological Health.
Mammography matters; vol 2 issue 3. Columbia,
Maryland: Food and Drug Administration. 1995.
230 Healthy People 2000 Final Review

Table 16. Cancer objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
16.1* Cancer deaths (age adjusted per
100,000) ....................... 1987 134 135 135 133 133 132 130 128 126 124 - - - 130
a. Black ....................... 1990 182 . . . 179 178 177 174 172 168 165 161 - - - 175
16.2* Slow the rise in lung cancer deaths
(age adjusted per 100,000) ......... 1987 38.5 39.9 39.6 39.3 39.3 38.7 38.3 37.8 37.3 36.9 - - - 42
a. Female...................... 1990 25.6 . . . 25.8 26.3 26.5 26.6 26.9 26.8 26.9 27.0 - - - 27
b. Black male ................... 1990 86.1 . . . 83.1 81.2 80.7 77.6 75.7 73.4 70.5 68.5 - - - 91
16.3 Female breast cancer deaths (age
adjusted per 100,000) ............. 1987 23.0 23.1 22.7 21.9 21.5 21.3 21.0 20.2 19.4 18.8 - - - 20.6
a. Black female.................. 1990 27.5 . . . 27.7 27.0 27.1 26.9 27.5 26.5 26.7 25.3 - - - 25
16.4 Cervical cancer deaths (age adjusted
per 100,000) .................... 1987 2.8 2.8 2.7 2.7 2.6 2.7 2.5 2.5 2.5 2.3 - - - 1.3
a. Black female.................. 1990 5.9 . . . 5.7 6.1 5.7 5.0 5.2 4.7 4.8 4.4 - - - 3
b. Hispanic female
1
.............. 1977-83 3.6 3.3 3.0 3.4 3.1 3.5 3.1 3.1 3.0 2.5 - - - 2
16.5* Colorectal cancer deaths (age
adjusted per 100,000) ............. 1987 14.7 13.8 13.5 13.2 13.1 13.0 12.8 12.3 12.1 12.0 - - - 13.2
a. Black ....................... 1990 18.1 . . . 17.5 17.3 17.6 17.3 17.4 16.9 16.9 16.8 - - - 16.5
16.6* Cigarette smoking prevalence
People 18 years and over ........... 1987 29% 25% 26%
2
27% 25% 26% 25% - - -
‡
25%
‡
24% - - - 15%
Male ......................... 1987 31% 28% 28%
2
29% 28% 28% 27% - - -
‡
28%
‡
26% - - - 15%
Female ....................... 1987 27% 23% 23%
2
25% 22% 23% 23% - - -
‡
22%
‡
22% - - - 15%
a. People with high school education
or less 20 years and over ....... 1987 34% 31% 31%
2
32% 30% 31% 30% - - -
‡
31%
‡
31% - - - 20%
b. Blue-collar workers 18 years and
over........................ 1987 41% 36% 36%
2
36% 34% 39% 36% - - -
‡
37%
‡
36% - - - 20%
c. Military personnel .............. 1988 42% - - - - - -
2
35% --- --- 32% --- --- 30% --- 20%
d. Black 18 years and over ......... 1987 33% 26% 29%
2
28% 26% 27% 26% - - -
‡
27%
‡
25% - - - 18%
e. Hispanic 18 years and over ...... 1987 24% 23% 20%
2
21% 20% 20% 18% - - -
‡
20%
‡
19% - - - 15%
f. American Indian/Alaska Native
18 years and over ............. 1979-87
3
42–70% 38% 31%
2
40% 39% 40% 35% - - -
‡
32%
‡
37% - - - 20%
g. Southeast Asian male ........... 1984-88 55%
4
35%
4
36–41% - - - - - - - - - - - - - - - - - - - - - - - - 20%
h. Females of reproductive age
(18–44 years) ................ 1987 29% 26% 27%
2
28% 26% 27% 26% - - -
‡
26%
‡
25% - - - 12%
i. Pregnant females............... 1985 25% 19% 20% - - - 20% - - - 18% 14% 13% 13% - - - 10%
j. Females who use oral
contraceptives ................ 1983 36%
5
26% --- --- --- --- 24% --- --- --- --- 10%
16.7* Dietary fat intake among people
2 years and over
6
National Health and Nutrition
Examination Survey
Average percent of calories from
total fat ...................... 1976-80
7
3 6 % --- --- --- ---
8
34% --- --- --- --- --- 30%
See footnotes and key at end of table.
Healthy People 2000 Final Review 231

Table 16. Cancer objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Average percent of calories from
saturated fat .................. 1976-80
7
13% --- --- --- ---
8
12% --- --- --- --- --- 10%
Percent who met goal for total fat . . . 1988-94
§
27% ... ... ... ... ... --- --- --- --- --- 50%
Percent who met goal for saturated
f a t.......................... 1988-94
§
29% ... ... ... ... ... --- --- --- --- --- 50%
Continuing Survey of Food Intakes by
Individuals
Average percent of calories from
total fat ...................... 1989-91 34% . . . . . . - - - - - - 33% 33% 33% - - - - - - - - - 30%
Average percent of calories from
saturated fat .................. 1989-91 12% . . . . . . - - - - - - 11% 11% 11% - - - - - - - - - 10%
Percent who met goal for fat ....... 1989-91 22% . . . . . . - - - - - - 32% 33% 34% - - - - - - - - - 50%
Percent who met goal for saturated
f a t.......................... 1989-91 21% . . . . . . - - - - - - 34% 35% 36% - - - - - - - - - 50%
16.8* Average daily intake of vegetables,
fruits, and grain products among
people 2 years and over
6
Average number of servings
Vegetables and fruits ............. 1989-91 4.1 . . . . . . - - - - - - 4.6 4.7 4.7 - - - - - - - - - 5.0
Grain products.................. 1989-91 5.8 . . . . . . - - - - - - 6.7 6.8 6.9 - - - - - - - - - 6.0
Proportion who met Dietary Guidelines
goal
Vegetables and fruits ............. 1989-91 29% . . . . . . - - - - - - 36% 37% 35% - - - - - - - - - 50%
Grain products.................. 1989-91 40% . . . . . . - - - - - - 50% 53% 52% - - - - - - - - - 50%
16.9 Actions to limit sun exposure
Among people 18 years and over those
very likely to:
Limit sun exposure .............. 1992
§
32% ... ... ... --- --- --- --- --- 27% --- 60%
Use sun screen ................. 1992
§
29% ... ... ... --- --- --- --- --- 31% --- 60%
Wear protective clothing .......... 1992 28% . . . . . . . . . - - - - - - - - - - - - - - - 24% - - - 60%
Avoid artificial ultraviolet light....... ... --- --- --- --- --- --- --- --- --- --- --- 60%
16.10 Tobacco, diet, and cancer screening
and counseling by clinicians
Smoking patients ................. 1986
9
52%
9,10
96% --- --- --- --- --- --- --- --- --- 75%
Digital rectal ..................... ... ---
10
49% --- --- --- --- --- --- --- --- --- 75%
Blood stool ...................... ... ---
10
56% --- --- --- --- --- --- --- --- --- 75%
Proctoscopic exam ................ ... ---
10
23% --- --- --- --- --- --- --- --- --- 75%
Breast physical ................... ... ---
10
78% --- --- --- --- --- --- --- --- --- 75%
Mammogram .................... ... ---
10
37% --- --- --- --- --- --- --- --- --- 75%
Pap test ........................ ... ---
10
55% --- --- --- --- --- --- --- --- --- 75%
Percent of clinicians routinely providing
service to 81–100% of patients
See footnotes and key at end of table.
232 Healthy People 2000 Final Review

Table 16. Cancer objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Formulation of diet/nutrition plan
Pediatricians ................... ... --- --- --- 31% --- --- --- --- ---
11,12
--- --- 75%
Nurse practitioners .............. ... --- --- --- 31% --- --- --- --- ---
11
31% - - - 75%
Obstetricians/gynecologists ........ ... --- --- --- 19% --- --- --- --- ---
11,12
--- --- 75%
Internists ...................... ... --- --- --- 33% --- --- --- --- ---
11,12
--- --- 75%
Family physicians ............... ... --- --- --- 24% --- --- --- --- ---
11,12
--- --- 75%
Discussion of strategies to quit smoking
Pediatricians ................... ... --- --- --- 19% --- --- --- --- ---
11,12
--- --- 75%
Nurse practitioners .............. ... --- --- --- 20% --- --- --- --- ---
11
39% - - - 75%
Obstetricians/gynecologists ........ ... --- --- --- 28% --- --- --- --- ---
11,12
--- --- 75%
Internists ...................... ... --- --- --- 50% --- --- --- --- ---
11,12
--- --- 75%
Family physicians ............... ... --- --- --- 43% --- --- --- --- ---
11,12
--- --- 75%
16.11 Breast examination and mammogram
Female 50 years and over (preceding
1–2 years) ..................... 1987 25% 49% - - - 51% 55% 56% - - - - - - - - - 64% - - - 60%
a. Hispanic female 50 years and over . 1987 18% 42% - - - 47% 47% 50% - - - - - - - - - 60% - - - 60%
b. Low-income female 50 years and
over (annual family income less
than $10,000)
13
............... 1987 15% 32% - - - 32% 39% 38% - - - - - - - - - 48% - - - 60%
c. Female 50 years and over with less
than high school education ...... 1987 16% 35% - - - 35% 42% 42% - - - - - - - - - 51% - - - 60%
d. Female 70 years and over ....... 1987 18% 39% - - - 39% 44% 45% - - - - - - - - - 56% - - - 60%
e. Black female 50 years and over . . . 1987 19% 43% - - - 48% 54% 56% - - - - - - - - - 62% - - - 60%
16.12 Pap test
14
Ever received .................... 1987 88% - - - - - - 93% 95% 94% - - - - - - - - - 93% - - - 95%
a. Hispanic female 18 years and over . 1987 75% - - - - - - 88% 88% 91% - - - - - - - - - 85% - - - 95%
b. Female 70 years and over ....... 1987 76% - - - - - - 86% 91% 90% - - - - - - - - - 88% - - - 95%
c. Female 18 years and over with less
than high school education ...... 1987 79% - - - - - - 87% 91% 91% - - - - - - - - - 86% - - - 95%
d. Low-income female 18 years and
over (annual family income less
than $10,000)
13
............... 1987 80% - - - - - - 89% 89% 91% - - - - - - - - - 86% - - - 95%
Received within preceding 3 years .... 1987 75% - - - - - - 74% 78% 77% - - - - - - - - - 79% - - - 85%
a. Hispanic female 18 years and over . 1987 66% - - - - - - 74% 77% 74% - - - - - - - - - 75% - - - 80%
b. Female 70 years and over ....... 1987 44% - - - - - - 46% 54% 53% - - - - - - - - - 56% - - - 70%
c. Female 18 years and over with less
than high school education ...... 1987 58% - - - - - - 58% 64% 62% - - - - - - - - - 66% - - - 75%
d. Low-income female 18 years and
over (annual family income less
than $10,000)
13
............... 1987 64% - - - - - - 65% 71% 72% - - - - - - - - - 70% - - - 80%
See footnotes and key at end of table.
Healthy People 2000 Final Review 233

Table 16. Cancer objective status—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
16.13 Fecal occult blood test and
proctosigmoidoscopy (50 years and
over)
Received fecal occult blood testing
within preceding 2 years ...........
1987 27% - - - - - - 30% - - - - - - - - - - - - - - - 35% - - - 50%
Ever received proctosigmoidoscopy . . . 1987 25% - - - - - - 33% - - - - - - - - - - - - - - - 37% - - - 40%
People 65 years and over with routine
checkup in past 2 years who had a
fecal occult blood test .............
... --- --- 36% --- --- --- --- --- --- --- --- ...
16.14 Oral, skin, and digital rectal
examinations
People 50 years and over (during past
year)..........................
... --- --- --- --- --- --- --- --- --- --- --- 40%
Oral.......................... ... --- --- --- 9% --- --- --- --- --- 13% --- ...
Skin ......................... ... --- --- --- 17% --- --- --- --- --- 16% --- ...
Digital rectal ................... 1987 27% - - - - - - 38% - - - - - - - - - - - - - - - 34% - - - . . .
16.15 Pap test quality
Monitoring cytology laboratory ........ 1988-92
15
100% . . . . . . . . . - - - - - - - - -
16,17
100% - - - - - - - - - 100%
16.16 Monitoring and certifying
mammography facilities
Certified by FDA .................. 1990 18–21% . . . - - - 64% - - - - - - - - - - - - - - - - - - - - - 100%
Mammogram Quality Standards Act
compliance
No non-compliances . . . - - - - - - - - - - - - - - - - - - 31.9% 48.2% 56.4% - - - - - - . . .
Level 3 findings ................. ... --- --- --- --- --- --- 47.1% 38.6% 28.6% - - - - - - . . .
Level 2 findings ................. ... --- --- --- --- --- --- 18.8% 11.9% 13.9% - - - - - - . . .
Level 1 findings ................. ... --- --- --- --- --- --- 2.2% 1.4% 1.1% - - - - - - . . .
16.17* Oral cancer deaths (per 100,000)
Male 45-74 years ................. 1987 13.6 13.4 12.7 12.2 12.1 11.1 11.0 10.7 10.3 10.4 - - - 10.5
Female 45-74 years ............... 1987 4.8 4.6 4.6 4.3 4.2 4.0 3.9 3.5 3.5 3.4 - - - 4.1
a. Black male 45-74 years ......... 1990 29.4 . . . 26.9 27.3 26.2 25.2 23.4 22.6 20.6 21.0 - - - 26.0
b. Black female 45-74 years ........ 1990 6.9 . . . 6.9 6.0 5.8 5.7 6.4 5.0 5.2 4.6 - - - 6.9
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
‡
The NHIS was redesigned in 1997. Data may not be comparable with previous years; see Appendix.
1
Updates for 1990-98 exclude data from States lacking an Hispanic-origin item on their death certificate or for which Hispanic-origin data were not of sufficient quality.
2
In 1992, the definition of ‘‘current’’ changed to include ‘‘some days’’ (intermittent smoking).
3
Estimates for different tribes.
4
Vietnamese males only.
5
1988 data.
6
Estimates are from 1-, 2-, or 3-day dietary data.
7
For people 2-74 years.
8
1988-94 data.
9
Data reflect tobacco screening and counseling only.
10
1989 data.
Met
Toward
Mixed/ no change
Away
Cannot assess
234 Healthy People 2000 Final Review

11
1997-98 data.
12
Response rate for this group was too low to produce reliable estimates.
13
Beginning with 1993, data are for women with family incomes below the Census poverty threshold.
14
Includes women without a uterine cervix.
15
15 of 3,200 laboratories closed or limited in cytology testing.
16
1993-96 data.
17
8 of 3,200 laboratories terminated from medical payments until differences corrected.
NOTE: Data include revisions and, therefore, may differ from data previously published in these reports and other publications.
Objective number Data source
16.1*, 16.1a National Vital Statistics System, CDC, NCHS.
16.2*, 16.2a, b National Vital Statistics System, CDC, NCHS.
16.3, 16.3a National Vital Statistics System, CDC, NCHS.
16.4, 16.4a National Vital Statistics System, CDC, NCHS.
16.4b Baseline: Surveillance, Epidemiology, and End Results, NIH, NCI.
Updates: National Vital Statistics System, CDC, NCHS.
16.5*, 16.5a National Vital Statistics System, CDC, NCHS.
16.6*,16.6a, b, d, e, h National Health Interview Survey, CDC, NCHS.
16.6c Baseline, 1992, and 1995 updates: Worldwide Survey of Substance Abuse and Health Behaviors Among Military Personnel, DoD, OASD.
1998 update: DoD Survey of Health Related Behaviors, Research Triangle Institute.
16.6f Baseline: CDC.
Updates: National Health Interview Survey, CDC, NCHS.
16.6g Baseline: Local surveys.
1990 update: Jenkins CH. Cancer risks and prevention practices among Vietnamese refugees. Western J of Med 153:34–9. 1990.
1991 update: Jenkins CNH, et al. Tobacco use in Vietnam: Prevalence, predictors, and the role of the transnational tobacco corporations.
JAMA 227(21):1726–31. 1997; Jenkins CNH, et al. The effectiveness of a media-led intervention to reduce smoking among Vietnamese-American men.
AJPH 87(6):1031–4. 1997.
16.6i Baseline and 1991 update: National Health Interview Survey, CDC, NCHS.
1993 update: National Health and Pregnancy Survey, NIH, NIDA.
1995 update: National Survey of Family Growth, CDC, NCHS.
1996–98 updates: National Vital Statistics System, CDC, NCHS.
16.6j 1983 baseline and 1988 update: Behavioral Risk Factor Surveillance System, CDC, NCCDPHP.
1995 update: National Survey of Family Growth, CDC, NCHS.
16.7* 1976–80 and 1988–94 data: National Health and Nutrition Examination Survey, CDC, NCHS.
1989–91 baselines and 1994–96 updates: Continuing Survey of Food Intakes by Individuals, USDA.
16.8* Continuing Survey of Food Intakes by Individuals, USDA.
16.9 National Health Interview Survey, CDC, NCHS.
16.10 1986 baseline: Physician Practice Study, University of Chicago.
1989 updates: Survey of Physician’s Attitudes and Practices in Early Cancer Detection, NIH, NCI.
1992 data: Primary Care Provider Surveys, OPHS, ODPHP.
1997–98 data: Prevention in Primary Care Study, American College of Preventive Medicine.
16.11, 16.11a-d National Health Interview Survey, CDC, NCHS.
16.12, 16.12a-d National Health Interview Survey, CDC, NCHS.
16.13 National Health Interview Survey, CDC, NCHS.
16.14 National Health Interview Survey, CDC, NCHS.
16.15 Clinical Laboratory Improvements Act, HCFA.
16.16 Baseline and 1992 update: American College of Radiology. 1995–97 data: Mammography Quality Assurance Program, FDA.
16.17*, 16.17a,b National Vital Statistics System, CDC, NCHS.
* Duplicate objective.
Healthy People 2000 Final Review 235
Cancer Objectives
16.1*: Reverse the rise in cancer deaths
to achieve a rate of no more than 130
per 100,000 people.
Duplicate objective: 2.2
16.1a*: Reverse the rise in cancer
deaths among blacks to achieve a
rate of no more than 175 per
100,000 people.
Duplicate objective: 2.2a
16.2*: Slow the rise in lung cancer
deaths to achieve a rate of no more than
42 per 100,000 people.
Duplicate objective: 3.2
16.2a*: Slow the rise in lung cancer
deaths among females to no more
than 27 per 100,000.
Duplicate objective: 3.2a
16.2b*: Slow the rise in lung cancer
deaths among black males to no
more than 91 per 100,000.
Duplicate objective: 3.2b
16.3: Reduce breast cancer deaths to no
more than 20.6 per 100,000 women.
16.3a: Reduce breast cancer deaths
among black females to no more
than 25 per 100,000 women.
16.4: Reduce deaths from cancer of the
uterine cervix to no more than 1.3 per
100,000 women.
16.4a: Reduce deaths from cancer
of the uterine cervix among black
females to no more than 3 per
100,000 women.
16.4b: Reduce deaths from cancer
of the uterine cervix among
Hispanic females to no more than 2
per 100,000 women.
16.5*: Reduce colorectal cancer deaths
to no more than 13.2 per 100,000
people.
Duplicate objective: 2.23
16.5a*: Reduce colorectal cancer
deaths among blacks to no more
than 16.5 per 100,000 people.
Duplicate objective: 2.23a
16.6*: Reduce cigarette smoking to a
prevalence of no more than 15 percent
among people aged 18 and older.
Duplicate objectives: 3.4 and 15.12
16.6a*: Reduce cigarette smoking
to a prevalence of no more than
20 percent among people aged 20
and older with a high school
education or less.
Duplicate objectives: 3.4a and
15.12a
16.6b*: Reduce cigarette smoking
to a prevalence of no more than
20 percent among blue-collar
workers aged 18 and older.
Duplicate objectives: 3.4b and
15.12b
16.6c*: Reduce cigarette smoking to
a prevalence of no more than
20 percent among military
personnel.
Duplicate objectives: 3.4c and
15.12c
16.6d*: Reduce cigarette smoking
to a prevalence of no more than
18 percent among blacks aged 18
and older.
Duplicate objectives: 3.4d and
15.12d
16.6e*: Reduce cigarette smoking to
a prevalence of no more than
15 percent among Hispanics aged
18 and older.
Duplicate objectives: 3.4e and
15.12e
16.6f*: Reduce cigarette smoking to
a prevalence of no more than
20 percent among American Indians
and Alaska Natives.
Duplicate objectives: 3.4f and
15.12f
16.6g*: Reduce cigarette smoking
to a prevalence of no more than
20 percent among Southeast Asian
men.
Duplicate objectives: 3.4g and
15.12g
16.6h*: Reduce cigarette smoking
to a prevalence of no more than
12 percent among women of
reproductive age.
Duplicate objectives: 3.4h and
15.12h
16.6i*: Reduce cigarette smoking to
a prevalence of no more than
10 percent among pregnant women.
Duplicate objectives: 3.4i and
15.12i
16.6j*: Reduce cigarette smoking to
a prevalence of no more than
10 percent among women who use
oral contraceptives.
Duplicate objectives: 3.4j and
15.12j
16.7*: Reduce dietary fat intake to an
average of 30 percent of calories or less
and average saturated fat intake to less
than 10 percent of calories among
people aged 2 and older. In addition,
increase to at least 50 percent the
proportion of people aged 2 and older
who meet the Dietary Guidelines’
average daily goal of no more than
30 percent of calories from fat, and
increase to at least 50 percent the
proportion of people aged 2 and older
who meet the average daily goal of less
than 10 percent of calories from
saturated fat.
Duplicate objectives: 2.5 and 15.9
16.8*: Increase complex carbohydrate
and fiber-containing foods in the diets of
adults to five or more daily servings for
vegetables (including legumes) and
fruits, and to six or more daily servings
for grain products. In addition, increase
to at least 50 percent the proportion of
people aged 2 and older who meet the
Dietary Guidelines’ average daily goal
of 5 or more servings of
vegetables/fruits, and increase to at least
50 percent the proportion who meet the
goal of 6 or more servings of grain
products.
Duplicate objective: 2.6
16.9: Increase to at least 60 percent the
proportion of people of all ages who
limit sun exposure, use sunscreens and
protective clothing when exposed to
sunlight, and avoid artificial sources of
ultraviolet light (e.g., sun lamps, tanning
booths).
16.10: Increase to at least 75 percent the
proportion of primary care providers
who routinely counsel patients about
tobacco-use cessation, diet modification,
and cancer screening
recommendations,which includes
providing information on the potential
benefit or harm attributed to the various
screening modalities and discussion of
risk factors associated with breast,
prostate, cervical, colorectal, and lung
cancers.
16.11: Increase to at least 60 percent
those women aged 50 and older who
have received a clinical breast
236 Healthy People 2000 Final Review
examination and a mammogram within
the preceding 1 to 2 years.
16.11a: Increase to at least
60 percent Hispanic women aged 50
and older who have received a
clinical breast examination and a
mammogram within the preceding 2
years.
16.11b: Increase to at least
60 percent low-income women aged
50 and older who have received a
clinical breast examination and a
mammogram within the preceding 2
years.
16.11c: Increase to at least
60 percent women aged 50 and
older with less than high school
education who have received a
clinical breast examination and a
mammogram within the preceding 2
years.
16.11d: Increase to at least
60 percent women aged 70 and
older who have received a clinical
breast examination and a
mammogram within the preceding 2
years.
16.11e: Increase to at least
60 percent black women aged 50
and older who have received a
clinical breast examination and a
mammogram within the preceding 2
years.
16.12: Increase to at least 95 percent the
proportion of women aged 18 and older
who have ever received a Pap test, and
to at least 85 percent those who received
a Pap test within the preceding 1 to 3
years.
16.12a: Increase to at least
95 percent the proportion of
Hispanic women aged 18 and older
who have ever received a Pap test,
and to at least 80 percent those who
received a Pap test within the
preceding 3 years.
16.12b: Increase to at least
95 percent the proportion of women
aged 70 and older who have ever
received a Pap test, and to at least
70 percent those who received a
Pap test within the preceding 3
years.
16.12c: Increase to at least
95 percent the proportion of women
aged 18 and older with less than a
high school education who have
ever received a Pap test, and to at
least 75 percent those who received
a Pap test within the preceding 3
years.
16.12d: Increase to at least
95 percent the proportion of
low-income women (annual family
income less than $10,000) aged 18
and older who have ever received a
Pap test, and to at least 80 percent
those who received a Pap test
within the preceding 3 years.
16.13: Increase to at least 50 percent the
proportion of people aged 50 and older
who have received fecal occult blood
testing within the preceding 1 to 2
years, and to at least 40 percent those
who have ever received
proctosigmoidoscopy.
16.14: Increase to at least 40 percent the
proportion of people aged 50 and older
visiting a primary care provider in the
preceding year who have received oral,
skin, and digital rectal examinations
during one such visit.
16.15: Ensure that Pap tests meet quality
standards by monitoring and certifying
all cytology laboratories.
16.16: Ensure that mammograms meet
quality standards by inspecting and
certifying 100 percent of mammography
facilities according to the requirements
of the Mammography Quality Standards
Act.
16.17*: Reduce deaths due to cancer of
the oral cavity and pharynx to no more
than 10.5 per 100,000 men aged 45–74
and 4.1 per 100,000 women aged
45–74.
Duplicate objectives: 3.17 and 13.7
16.17a*: Reduce deaths due to
cancer of the oral cavity and
pharynx to no more than 26.0 per
100,000 among black males aged
45–74.
Duplicate objectives: 3.17a and
13.7a
16.17b*: Reduce deaths due to
cancer of the oral cavity and
pharynx to no more than 6.9 per
100,000 among black females aged
45–74.
Duplicate objectives: 3.17b and
13.7b
*Duplicate objective.
Healthy People 2000 Final Review 237

Priority Area 17
Diabetes and
Chronic Disabling
Conditions
Background
Diabetes and chronic disabling
conditions continue to pose a significant
challenge to the public health of the
Nation. As the population of the United
States ages, more people are expected to
be affected by these conditions. These
conditions can impinge on quality of life
and lead to physical, emotional, social,
and economic costs, not only for
individuals, but also for their families
and the Nation. Chronic disabling
conditions that significantly affect the
quality of life encompass diabetes,
chronic kidney disease, arthritis,
deformities or orthopedic impairments,
and mental retardation. Other conditions
include asthma, hearing and visual
impairments, overweight, and peptic
ulcer disease. (1–4).
Some 800,000 new cases of
diabetes are diagnosed each year, or
2,200 each day (5,6). The prevalence of
diabetes, as well as diabetes-related
complications, is increasing in the
United States (5–7). End stage renal
disease (ESRD) among persons with
diabetes has almost tripled in the past
decade (8). The number of persons with
diabetes increased steadily during the
1990’s; 1988–94 data showed that 10.5
million persons had been diagnosed with
diabetes, while 5.5 million persons
estimated to have the disease were
undiagnosed (1). Furthermore, because
diabetes is most common among
persons over 60 years of age (9), the
number of persons with diabetes is
expected to rise as the population ages.
Diabetes remains the seventh leading
cause of death (10).
An estimated 54 million persons in
the United States, or nearly 20 percent
of the population, are living with some
level of disability (11). The direct
medical and indirect annual costs
associated with disability are more than
$300 million, or 4 percent of the gross
national product (12).
Asthma is a serious and growing
health problem. An estimated 14.9
million persons in the United States
have asthma (13). The number of people
with asthma increased by 102 percent
between the years 1979–80 and 1993–94
(14). Asthma is responsible for about
500,000 hospitalizations (13), 5,000
deaths (13), and 134 million days of
restricted activity a year (14). Yet most
of the problems caused by asthma could
be averted if persons with asthma and
their health care providers managed the
disease according to established
guidelines. Effective management of
asthma comprises four major
components: controlling exposure to
factors that trigger asthma episodes,
adequately managing asthma with
medicine, monitoring the disease by
using objective measures of lung
function, and educating asthma patients
to become partners in their own care
(15,16). Such prevention efforts are
essential to interrupt the progression
from disease to functional limitation and
disability and to improve the quality of
life for persons with asthma.
The various forms of arthritis—the
leading cause of disability in the United
States (17)—affect more than 15 percent
of the total U.S. population (43 million
persons) and more than 20 percent of
the adult population (18–21). Other
musculoskeletal conditions can also lead
to disability. About 13–18 percent of
women 50 years of age and over and
3–6 percent of men 50 years of age and
over have osteoporosis (22), a reduction
in bone mass or density that leads to
deterioration or fragile bones, whose
major health consequence is an
increased risk of fractures. Chronic back
conditions are also common and can be
debilitating. In the United States, back
pain is the most frequent cause of
activity limitation in people under 45
years of age (23,24), the second most
frequent reason for physician visits, the
fifth ranking reason for hospitalization,
and the third most common reason for
surgical procedures (25).
Data Summary
Highlights
By the end of the 20th century,
notable strides had been made in
improving the lives of people with
diabetes and chronic disabling
conditions. The average number of years
of healthy life for the total population
(17.1) increased from a baseline of 64
years in 1990 to 65.2 years in 1998,
exceeding the Healthy People 2000
target. Among special populations
targeted by the objective, Hispanics
have met the year 2000 target, while
progress was made for blacks and older
adults. Assuming full compliance with
the Americans with Disabilities Act of
1990, that prohibits employers from
discriminating against disabled
individuals, all worksites had policies or
programs for hiring people with
disabilities (17.19).
Several other measures of chronic
disability improved over the decade. The
rates of both significant hearing
impairments (17.6) and significant visual
impairments (17.7) decreased.
Participation in moderate (17.13)
physical activity five or more times per
week increased over the decade, meeting
the year 2000 target of 30 percent,
whereas the proportion of adults
participating in physical activity seven
or more times per week moved toward,
but did not meet the 30–percent target.
The proportion of perimenopausal
women receiving counseling about
estrogen replacement therapy (17.18)
increased from 80 percent in 1994 to
89 percent in 1998. A greater proportion
of people with diabetes had dilated eye
exams (17.23) in 1998 than in 1989, an
8-percent increase.
In other areas, however, measures
moved away from the year 2000 targets.
The rate of activity limitation due to
chronic back conditions (17.5) increased
from 21.9 per 1,000 persons in 1986–88
to 27.9 per 1,000 persons in 1994–96.
Both the incidence and prevalence of
diabetes (17.11) increased as did the rate
of diabetes-related deaths (17.9). The
prevalence of peptic ulcer (17.21)
increased from the 1991 baseline of
20 percent to 28 percent in 1998.
Summary of Progress
Data are available to assess the
progress of 22 of the 23 objectives that
address diabetes and chronic disabling
conditions. Two objectives (17.1 and
17.19) met their year 2000 targets. Six
objectives (17.6, 17.7, 17.13, 17.18,
17.22, and 17.23) moved toward the
year 2000 targets. Eleven objectives
(17.2–17.5, 17.8, 17.9, 17.11, 17.12,
17.15, 17.16, and 17.21) moved away
from their targets. See table 17 for the
tracking data for the objectives in this
priority area and figure 17 for a
quantitative assessment of progress.
For three objectives (17.10, 17.14,
and 17.17), progress among the multiple
measures used for tracking was mixed.
Progress could not be assessed for one
238 Healthy People 2000 Final Review

-100 -80 -60 -40 -20
0
20 40 60 80 100
17.1 Years of healthy life
17.2 Limitation in major activity
17.3 Difficulty with self-care: 70 years and over
17.4 People with asthma with activity limitation
17.5 Activity limitation due to chronic back conditions
17.6 Significant hearing impairment
17.7 Significant visual impairment
17.8 Serious mental retardation: Children 10 years, IQ <50
17.9 Diabetes-related deaths
17.10 Diabetes related complications: End-stage
renal disease
Blindness
Lower extremity amputation
17.11 Diabetes: Incidence
Male
17.12 Overweight prevalence: 20-74 years
Prevalence
Female
Adolescents 12-19 years
17.13 Light-to-moderate physical activity:
5 or more times per week
7 or more times per week
17.14 Patient education: People with diabetes
People with asthma
17.16 Early detection hearing impairment in children
17.18 Perimenopausal females counseled about estrogen
replacement therapy: Female 40-60 years
Female 50-60 years
Female 40-49 years
17.19 Worksites with policies for hiring people
with disabilities
17.21 Prevalence of peptic ulcer
17.23 People with diabetes who had a dilated eye
exam in the past year
Moved away from target Moved toward target Met target
* This objective has moved away from its target. A progress quotient could not be calculated.
NOTE: Complete tracking data are shown in table 17. Progress quotients are not calculated for objective 17.15, 17.17, 17.20, and 17.22.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
120%
100%
100%
166%
-50%
-75%
-45%
-43%
-2%
-43%
23%
29%
79%
90%
19%
9%
55%
3%
No change
Away*
-13%
-2%
-411%
-143%
-250%
-150%
-367%
-207%
-1,800%
Percent of target achieved
Healthy People 2000 Final Review 239
Figure 17. Final status of Diabetes and Chronic Disabling Conditions objectives

objective (17.20) because it lacked
baseline data.
Discussion
Several factors account for what is
considered to be a diabetes epidemic,
including the following: behavioral
elements (improper nutrition, for
example, increased fat consumption;
decreased physical activity; obesity);
demographic changes (aging, increased
growth of at-risk populations); improved
ascertainment and surveillance systems
that more completely capture the actual
burden of diabetes; and the relative
weakness of interventions to change
individual, community, or organizational
behaviors (5,7,26,27). Several other
interrelated factors influence the impact
of diabetes, including genetics, cultural
and community traditions, and
socioeconomic status. In addition,
unanticipated scientific breakthroughs,
the characteristics of the health care
system, and the level of patient
knowledge and empowerment all have a
great impact on the disease burden
associated with diabetes.
Increased insulin resistance and
gradual deterioration in the function of
insulin-producing cells may explain the
high prevalence of diabetes among older
people. The rise in diabetes among
teenagers may be due to obesity,
improper nutrition, and lack of physical
activity, the latter of which has been
linked to increased television viewing
(1).
Particularly within certain racial and
ethnic groups, four potential individual
reasons account for a greater burden of
diabetes:
Increased number of cases of
diabetes, which is expected to lead to
more amputations, death, and other
complications.
Increased seriousness of diabetes,
for example, the presence of
hyperglycemia or other serious
comorbid conditions, such as high blood
pressure or elevated blood lipids.
Inadequate access to proper diabetes
prevention and control programs.
Unfortunately, many diabetes at-risk
groups reside in medically underserved
areas or are without adequate insurance
and thus do not receive these types of
preventive services.
Improper quality of care, which can
result in ineffective diabetes prevention
programs.
Identifying the reasons for
disparities in diabetes health outcomes is
important in tailoring programs to those
specific areas where deficiencies exist.
Collection of racial and ethnic health
services data for all health activities is
critical to designate the reason for the
greater disease burden.
Scientific research has led to greater
asthma control than was available in the
early 1980s (6). Effective management
of asthma includes four components:
avoiding or controlling the factors that
may make asthma worse (for example,
environmental and occupational
allergens and irritants), taking
appropriate medications tailored to the
severity of the disease, objective
monitoring of the disease by the patient
and the health care professional, and
actively involving the patient in
managing the disease (16). Effective
asthma management reduces the need
for hospitalizations and urgent care
visits (in either an emergency
department or physician’s office) and
enables patients to enjoy normal
activities (28,29).
Advances in human genetics related
to asthma are expected to provide better
information about the contribution of
genetic variation to the development of
disease when people are exposed to
certain environmental factors and
variation in individual response to
therapy. The use of this genetic
information will improve targeted
disease prevention and health
management strategies for respiratory
diseases.
Genetic research may soon identify
persons at high risk for certain types of
arthritis and thereby offer a better target
for interventions. Current medical care
offers considerable relief from pain and
other symptoms for all types of arthritis.
Available interventions often are not
used, however, because of the popular
belief that arthritis is part of normal
aging, which a person can do nothing
about, and that arthritis affects only
older persons. However, early diagnosis
and aggressive treatment of rheumatoid
arthritis with disease-modifying drugs,
for example, appear to reduce its
symptoms and related disability (30–35).
Educational and behavioral
interventions also can relieve symptoms
and reduce disability. Telephone contacts
with clinicians and several land-based
and water-based exercise programs have
had beneficial outcomes (36–39). The
Arthritis Self-Help Course, a 6-week,
2-hour per week educational
intervention, has been shown to reduce
pain up to 20 percent beyond outcomes
achieved through conventional medical
care (40). The course has the additional
benefit of reducing medical care costs
by reducing the number of physician
visits for arthritis (40,41). These and
other effective interventions currently
are underused, with some interventions
reaching less than 1 percent of target
populations (42). Countering myths
about arthritis and applying available
interventions can help reduce the impact
of this health problem (1).
A wide range of interventions
prevent or reduce lower back problems.
These interventions may include
activities designed to reduce the
physical demands of work activities by
redesigning the task or to address the
individual’s specific needs, such as
strength or endurance training or
counseling for nutrition and lifestyle
changes. Ergonomic interventions that
are directed at changing the job or work
environment have proved effective in
reducing risk of occupational low back
pain (1). Thus, it is reasonable to
assume that ergonomic approaches
would be effective in preventing chronic
lower back pain (LBP) as well. Even in
a nonwork environment, using
ergonomic principles can reduce the
physical demands of an activity.
Interventions involving training in
proper lifting techniques, physical
conditioning, and weight loss have been
investigated in programmatically
oriented studies. These have shown that
workplace interventions may have an
effect on low back disorders (43). The
overall benefits of exercise, nutrition,
and lifestyle changes on an individual’s
health and well-being would certainly
justify efforts in this area. Also,
interventions directed at improving
strength and endurance may have an
important impact on reducing activity
limitations due to chronic LBP.
Transition to Healthy People
2010
Several topics covered in the
Healthy People 2000 priority area on
Diabetes and Chronic Disabling
Conditions were reorganized into
separate focus area chapters in Healthy
People 2010. Providing for greater
emphasis on individual conditions,
Healthy People 2010 contains several
240 Healthy People 2000 Final Review
new chapters—including Arthritis,
Osteoporosis, and Chronic Back
Conditions; Chronic Kidney Disease;
Diabetes; Respiratory Diseases;
Disability and Secondary Conditions;
and Vision and Hearing.
In Healthy People 2010, Chronic
Kidney Disease has a revised measure
to track end-stage renal disease (ESRD)
incidence. The Healthy People 2010
focus area has also added measures that
address cardiovascular disease deaths
among persons with ESRD, counseling
for persons with chronic kidney failure,
kidney transplantation waiting lists,
kidney failure due to diabetes, and
medical therapy for persons with
diabetes and proteinuria.
Diabetes has expanded its area to
track the proportion of persons with
diabetes that have been diagnosed with
the disease, diabetes death rates among
persons with diabetes, cardiovascular
disease deaths among persons with
diabetes, gestational diabetes, and foot
ulcers. In addition to the receipt of
annual dilated eye exams among persons
with diabetes, there are several measures
in Healthy People 2010 that address
secondary and tertiary prevention of
complications associated with diabetes
including annual urinary microalbumin
measurements, annual glycosylated
hemoglobin measurements, annual foot
examinations, annual dental
examinations, aspirin therapy, as well as
self-blood-glucose-monitoring.
As a section in the Respiratory
Diseases focus area, asthma patient
education continues to be measured in
Healthy People 2010, with other
measures continued from Healthy
People 2000, such as asthma
hospitalizations and activity limitation
due to asthma. Asthma has several new
measures in Healthy People 2010, which
include asthma deaths, emergency
department visits for asthma, school or
work days missed by persons with
asthma due to asthma, appropriate
asthma care, and surveillance systems
for the collection of asthma-related data.
In Healthy People 2010, both the
Vision and Hearing focus area and
Disability and Secondary Conditions
focus area expand on the objectives
included in Healthy People 2000. The
disability focus area deals with
promoting health and preventing disease
among persons with disabilities, rather
than preventing disabling conditions.
The Vision and Hearing focus area
covers screening, visual impairments
and hearing loss and the appropriate
treatments, as well as protective devices
to keep hearing and vision intact.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the differences between the
two decades of objectives, reflecting
new knowledge and directions in these
areas.
Data Issues
Years of Healthy Life
Years of healthy life is a summary
measure of health that combines
mortality (quantity of life) and
morbidity and disability (quality of life)
into a single measure. The concept of
increasing the span of healthy life is one
of the three Healthy People 2000 goals
and a specific measure has been
developed to track this objective in three
priority areas (8.1, 17.1, and 21.1). The
data used to track the objective come
from the National Vital Statistics System
(mortality) and the National Health
Interview Survey (NHIS) (morbidity).
The NHIS was redesigned in 1997 and,
therefore, data for 1997–98 may not be
comparable with those from previous
years. See the Appendix for a discussion
of the changes to the NHIS. The
methodology for the Healthy People
2000 years of healthy life measure,
developed by NCHS and outside
consultants, is published in Healthy
People Statistical Notes series (44).
Definitions
Subobjective 17.2a (limitation in
major activity due to chronic conditions)
targets people with low income.
Originally, this subobjective was tracked
with data for people with annual family
incomes of less than $10,000. Because
of changes in the poverty level over
time, data are also shown for people
with family incomes below Census
poverty threshold (see Appendix).
The 1984–85 baseline figures for
objective 17.3 were derived by
combining estimates for the
noninstitutionalized population from the
1984 NHIS Supplement on Aging
(SOA) with data for those in nursing
homes from the 1985 National Nursing
Home Survey (NNHS). The 1984 SOA
asked about seven specific personal care
activities, also referred to as activities of
daily living (ADLs) for persons 65 years
of age and over. Because of the way the
questions were asked on the NNHS,
only five ADLs (bathing, dressing, using
the toilet, getting in and out of bed or
chair, and eating) were used for tracking
this objective. The numerator included
respondents to the SOA who said they
had ‘‘any difficulty’’ performing at least
two ADLs combined with patients for
whom administrators reported to the
NNHS as ‘‘receiving assistance’’ with at
least two ADLs. The denominator for
the baseline was the civilian,
noninstitutionalized population 65 years
of age and over plus the nursing home
population 65 years of age and over.
The update for this objective is derived
from combined data from the 1994
NHIS Second Supplement on Aging
(SOA II) and data from the 1995 NNHS
using the same questions as were used
for the baseline. However, because of
the way data were collected in the SOA
II, the 1994–95 update is for persons 70
years of age and over. The 1984–85 data
were also computed for ages 70 years of
age and over to provide a comparison
with the 1994–95 update. For objective
17.4, activity limitation refers to any
self-reported limitation in activity
attributed to asthma.
The 1990 baseline data for
diabetes-related deaths for Puerto Ricans
(17.9d) have been revised. The original
baseline published in the Midcourse
Review and 1995 Revisions (45)
included data for 45 States and the
District of Columbia. It did not include
data for New York where more than
one-half of the U.S. Puerto Rican
population resides. The revised baseline,
which includes data for 47 States and
the District of Columbia (including New
York), is considerably lower than
originally published and, in fact, has
met the year 2000 target for this
subobjective. The number of States
reporting Hispanic origin data on their
birth and death certificates has varied
from year to year; see Appendix for
more information.
Overweight (17.12) for adults is
defined as a body mass index (BMI) of
27.8 kilograms per meter squared for
males, and 27.3 kilograms per meter
squared for females. For adolescents,
overweight is the sex- and age-specific
85th percentile from NHANES II
(46,47). See the Nutrition chapter
(Priority Area 2) for additional
information.
Objective 17.22 addresses the
development and implementation of a
Healthy People 2000 Final Review 241
national process to identify significant
gaps in the Nation’s disease prevention
and health promotion data. Progress was
made, although it is difficult to quantify.
See objective 22.4 in the Data and
Surveillance chapter (Priority Area 22)
for additional information.
Data Sources
Data for objective 17.6 (significant
hearing impairment) are from the NHIS,
and are based on self-reported hearing
impairment (that is, deafness in one or
both ears or any trouble hearing in one
or both ears). These data are a proxy
measure for significant hearing
impairment. Hearing impairment covers
the range of hearing deficits from mild
loss in one ear to profound loss in both
ears. Generally, inability to hear sounds
at levels softer (less intense) than 20
decibels (dB) constitutes abnormal
hearing. Significant hearing impairment
is defined as having hearing thresholds
for speech poorer than 25 dB.
Significant visual impairment (17.7)
is also measured using the NHIS.
Significant visual impairment is
generally defined as a permanent
reduction in visual acuity and/or field of
vision that is not correctable with
eyeglasses or contact lenses. Severe
visual impairment is defined as inability
to read ordinary newsprint even with
corrective lenses. For this objective,
self-reported blindness in one or both
eyes and other self-reported visual
impairments (that is, any trouble seeing
with one or both eyes even when
wearing glasses or color blindness) from
the NHIS is used as a proxy measure
for significant visual impairment.
Diabetes-related mortality data
(17.9) are derived from the
multiple-cause-of-death files. Data
include all mentions of diabetes on the
death certificate, whether as an
underlying or contributing cause of
death. Diabetes is approximately three
times as likely to be listed as a
contributing cause of death than as the
underlying cause.
Data on lower extremity amputation
for objective 17.10–17.10c come from
the National Hospital Discharge Survey
(NHDS) maintained by the National
Center for Health Statistics (NCHS),
Centers for Disease Control and
Prevention (CDC). Data for the survey
are obtained from approximately 480
hospitals throughout the United States.
Data on race are not reported by many
hospitals due to omission of a race field
on hospital discharge reporting forms.
Automation of the hospital discharge
systems has led to an increase in the use
of these forms (UB–82 and UB–92) in
recent years. A comparison of NHDS
data with those who reported being
hospitalized in the NHIS (NHIS data
were adjusted to exclude hospitalizations
of 1 day or less) indicated that
underreporting for whites is roughly
22 percent in 1991; the difference in
reporting for blacks was negligible (48).
Data for diabetes prevalence (17.11
and 17.11e) were tracked using the
NHIS. Due to the NHIS redesign that
was implemented in 1997, data collected
in 1996 and earlier are not comparable
with data from the redesigned NHIS,
starting with 1997 (see Appendix).
American Indian/Alaska Native data for
1996 for objective 17.11a are from the
Indian Health Service (IHS) Patient
Comprehensive Care file. The file
excludes data from 25 of the 166 IHS
service units (representing 11 percent of
the population served by IHS) because
data were incomplete.
Baseline data for objectives 17.15
and 17.17 (clinical assessment of
childhood development and cognitive
assessment of older adults) are from the
Primary Care Provider Surveys (PCPS).
The data on testing/evaluation-inquiry
represent the proportion of providers
who routinely queried 81–100 percent of
their patients about specific functioning.
The data on treatment/referral refer to
the proportion of providers who
routinely provided these services to
patients who needed the services. The
sample was drawn from the membership
rolls of professional organizations for
pediatricians, nurse practitioners, family
physicians, obstetricians/gynecologists,
and internists. Response rates varied
from 50 to 80 percent across these
groups. The Prevention in Primary Care
Study (PPCS) was conducted in
1997–98 to update data from the PCPS.
The design and items included in the
1997–98 study were similar to the
PCPS, but a slightly different sampling
frame was used and some items
included in the 1992 surveys were not
included in the PPCS. The professionals
were sampled from listings of all
licensed, active practitioners in the
United States whose practices were at
least 50 percent primary care. Because
of low response rates from the other
provider groups, updates are available
only for nurse practitioners.
Data Comparability
The baseline data for 17.8 (mental
retardation) were revised to be
comparable with data from the
Metropolitan Atlanta Developmental
Disabilities Surveillance Program, which
uses school counts of children classified
as mentally retarded.
Overweight (17.12) was tracked
with two data sources. The primary data
source is the NHANES, which provided
baseline data for most of the overweight
objectives and the 1988–94 updates;
these data are derived from measured
height and weight. The second data
source is the NHIS. This survey
provides interim estimates shown in an
earlier publication (49), updates for
Hispanic females and American
Indians/Alaska Natives, and all data for
people with disabilities. NHIS estimates
are based on self-reported heights and
weights and are not comparable with the
actual measured data from NHANES;
prevalence estimates of overweight from
self-reported height and weight are
lower. Trends from the NHIS self-report
measures, like those from NHANES,
show a steady increase in prevalence of
overweight; this increase however, is
different in magnitude from that
observed in the data derived from
measured height and weight.
Objective 17.13 (light-to-moderate
physical activity) is being tracked with
the NHIS. The list of activities asked by
the NHIS has not been identical from
year to year. The 1985 and 1990 surveys
did not ask about some activities for
people 65 years of age and over; thus,
the data shown are for people 18–64
years of age. The 1991, 1995, and 1998
surveys asked about some activities
different from the previous surveys, but
people of all age groups were asked the
same questions. Because of these
differences, 1985 and 1990 data are not
comparable to later data. (See Priority
Area 1 for more information.)
References
1. Department of Health and Human Services.
Healthy people 2010. 2d ed. Washington: U.S.
Government Printing Office. 2000.
2. Department of Health and Human Services.
Healthy people 2000 midcourse review and 1995
revisions. Washington: Public Health Service.
1995.
3. Department of Health and Human Services.
Healthy people 2000: National health promotion
242 Healthy People 2000 Final Review
and disease prevention objectives. Washington:
Public Health Service. 1990.
4. National Center for Health Statistics. Healthy
people 2000 review, 1998–99. Hyattsville,
Maryland: Public Health Service. 1999.
5. Clark C. How should we respond to the
worldwide diabetes epidemic? Diabetes Care
21:475–6. 1998.
6. Burke J, Williams K, Gaskill S, et al. Rapid rise
in the incidence of type 2 diabetes from 1987 to
1996: Results from the San Antonio Heart Study.
Arch of Internal Med 159:1450–7. 1999.
7. King H, Aubert R, Herman H. Global burden of
diabetes, 1995–2025: Prevalence, numerical
estimates and projections. Diabetes Care
21:1414–31. 1997.
8. U.S. Renal Data System. 1999 Annual Data
Report. Appendix, Table A–6. Bethesda, Maryland:
National Institutes of Health, National Institute of
Diabetes and Digestive and Kidney Diseases.
1999.
9. Vita A, Terry R, Hubert H, et al. Aging, health
risk and cumulative disability. N Engl J Med
338:1035–41. 1998.
10. Murphy SL. Deaths: Final data for 1998.
National vital statistics reports; vol 48 no 11.
Hyattsville, Maryland: National Center for Health
Statistics. 2000.
11. McNeil JM. Americans with disabilities
1994–95. Current Population Reports P7061:3–6.
1997.
12. Institute of Medicine. Enabling America:
Assessing the role of rehabilitation science and
engineering. Washington: National Academy Press.
1997.
13. National Heart, Lung, and Blood Institute.
Data fact sheet. Asthma statistics. Bethesda,
Maryland: National Institutes of Health, Public
Health Service. 1999.
14. National Center for Health Statistics. Current
estimates from the National Health Interview
Survey, 1990. Vital Health Stat 10(194). 1997.
15. Benson V, Marano MA. Current estimates
from the National Health Interview Survey, 1995.
Vital Health Stat 10(199):1–428. 1998.
16. National Asthma Education and Prevention
Program. Expert Panel Report 2: Guidelines for
the diagnosis and management of asthma.
Bethesda, Maryland: National Institutes of Health.
1997.
17. Centers for Disease Control and Prevention.
Current trends: Prevalence of disabilities and
associated health conditions—United States,
1991–1992. MMWR 43(40):730–1, 737–9. 1994.
18. Centers for Disease Control and Prevention.
Arthritis prevalence and activity
limitations—United States, 1990. MMWR
43(24):433–8. 1994.
19. Lawrence RC, Helmick CG, Arnett FC, et al.
Estimates of the prevalence of arthritis and
selected musculoskeletal disorders in the United
States. Arthritis & Rheumatism 41(5):778–99.
1998.
20. Helmick CG, Lawrence RC, Pollard RA, et al.
Arthritis and other rheumatic conditions: Who is
affected now, who will be affected later? National
Arthritis Data Work Group. Arthritis Care and
Research 8:203–11. 1995.
21. Centers for Disease Control and Prevention.
Impact of arthritis and other rheumatic conditions
on the health-care system. MMWR 48(17):349–53.
1999.
22. Looker AC, Orwoll ES, Johnston CC, et al.
Prevalence of low femoral bone density in older
U.S. adults from NHANES III. J Bone Mineral
Res 12(11):1761–8. 1997.
23. Anderson GB. Epidemiological aspects on low
back pain in industry. Spine 6(1):53–60. 1981.
24. Kelsey JL, White AA, Pastides H, et al. The
impact of musculoskeletal disorders on the
population of the United States. J Bone Joint Surg
61(7):959–64. 1979.
25. Praemer A, Furner S, Rice DP.
Musculoskeletal conditions in the United States.
Park Ridge, Illinios: American Academy of
Orthoscopic Surgery. 1992.
26. Centers for Disease Control and Prevention.
Diabetes Surveillance, 1997. Atlanta, Georgia:
Department of Health and Human Services. 1997.
27. Centers for Disease Control and Prevention.
Trends in the prevalence and incidence of
self-reported diabetes mellitus—United States,
1980–1994. MMWR 46:1014–18. 1997.
28. Evans D, Mellins R, Lobach K, et al.
Improving care for minority children with asthma:
Professional education in public health clinics.
Pediatrics 99:157–64. 1997.
29. Institute of Medicine. Cleaning the air: Asthma
and indoor air exposures. Washington: National
Academy Press. 2000.
30. Weinblatt ME. Rheumatoid arthritis: Treat
now, not later! (Editorial). Ann Intern Med
124:773–4. 1996.
31. Van der Heide A, Jacobs JW, Bijlsma JW, et
al. The effectiveness of early treatment with
‘‘second-line’’ antirheumatic drugs. A randomized,
controlled trial. Ann Intern Med 124(8):699–707.
1996.
32. Fries JF, Williams CA, Morrfeld D, et al.
Reduction in long-term disability in patients with
rheumatoid arthritis by disease-modifying
antirheumatic drug-based treatment strategies.
Arthritis Rheum 39:616–22. 1996.
33. Egsmose C, Lund B, Borg G, et al. Patients
with rheumatoid arthritis benefit from early 2nd
line therapy: 5-year follow up of a prospective
double blind placebo controlled study. J
Rheumatol 22:2208–13. 1995.
34. Kirwan JR. The Arthritis and Rheumatism
Council, Low-Dose Glucocorticoid Study Group.
The effect of glucocorticoids on joint destruction
in rheumatoid arthritis. N Engl J Med
333(3):142–6. 1995.
35. Emery P, Salmon M. Early rheumatoid
arthritis: Time to aim for remission? Ann Rheum
Dis 54:944–7. 1995.
36. Maisiak R, Austin J, Heck L. Health outcomes
of two telephone interventions for patients with
rheumatoid arthritis or osteoarthritis. Arthritis
Rheum 39:1391–9. 1996.
37. Minor MA. Arthritis and exercise: The times
they are a-changin’. (Editorial). Arthritis Care and
Research 9:9–81. 1996.
38. Minor MA, Kay DR. Arthritis. In: Exercise
management for persons with chronic diseases and
disabilities. Champaign, Illinois: Human Kinetics.
1997.
39. Puett DW, Griffin MR. Published trials of
nonmedicinal and noninvasive therapies for hip
and knee osteoarthritis. Ann Intern Med
121(2):133–140. 1994.
40. Lorig KR, Mazonson PD, Holman HR.
Evidence suggesting that health education for
self-management in patients with chronic arthritis
has sustained health benefits while reducing health
care costs. Arthritis Rheum 36(4):439–446. 1993.
41. Kruger JMS, Helmick CG, Callahan LF, et al.
Cost-effectiveness of the Arthritis Self- Help
Course. Arch Intern Med. 1998.
42. Boutaugh M, Group Vice President, Health
Promotion, Arthritis Foundation. Personal
communication. Aug. 1999.
43. Violinn E. Do workplace interventions prevent
low-back disorders? If so: Why?: A
methodological commentary. Ergonomics
42(1):258–72. 1999.
44. Erickson P, Wilson R, Shannon I. Years of
Healthy Life. Healthy people statistical notes no 7.
Hyattsville, Maryland: National Center for Health
Statistics. 1995.
45. Department of Health and Human Services.
Healthy People 2000 midcourse review and 1995
revisions. Washington: Public Health Service.
1995.
46. Flegal KM, Carroll MD, Kuczmarski RJ, et al.
Overweight and obesity in the United States:
Prevalence and trends, 1960–94. Intern J Obes
22(1)39–47. 1998.
47. Kuczmarski RJ, Carroll MD, Flegal KM, et al.
Varying body mass index cutoff points to describe
overweight prevalence among U.S. adults:
NHANES III (1988–94). Obesity Research
5(6):542–8. 1997.
48. Kozak LJ. Underreporting of race in the
National Hospital Discharge Survey. Advance data
from vital and health statistics; no 265.
Hyattsville, Maryland: National Center for Health
Statistics. 1995.
49. National Center for Health Statistics. Healthy
people 2000 review, 1992. Hyattsville, Maryland:
Public Health Service. 1993.
Healthy People 2000 Final Review 243

Table 17. Diabetes and Chronic Disabling Conditions objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
17.1* Years of healthy life .................... 1990 64.0 . . . 63.9 63.7 63.5 63.8 63.9 64.2
‡
64.8
‡
65.2 - - - 65
a. Black ............................. 1990 56.0 . . . 56.0 55.6 55.2 55.6 56.0 56.5
‡
57.4
‡
57.8 - - - 60
b. Hispanic
1
.......................... 1990 64.8 . . . 63.6
2
64.0 63.2 64.2 64.0 64.7
‡
65.8
‡
66.3 - - - 65
c. People 65 years and over
3
............. 1990 11.9 . . . 11.8 11.9 11.9 12.1 12.0 12.2
‡
12.0
‡
12.2 - - - 14
17.2 Limitation in major activity due to chronic
conditions ........................... 1988 9.4% 9.3% 9.6% 10.3% 10.6% 10.3% 10.1% 10.0% - - - - - - - - - 8%
a. Low-income people
Annual family income less than $10,000. . 1988 18.9% 19.2% 19.6% 20.2% 20.9% 21.1% 21.4% 22.7% - - - - - - - - - 15%
Below poverty level ................. ... --- 14.6% 15.5% 16.2% 16.5% 16.8% 17.1% 17.5% - - - - - - - - - . . .
b. American Indian/Alaska Native .......... 1983–85 13.4%
4
12.3%
5
12.0%
6
12.6%
7
12.4%
8
13.3%
9
13.5%
10
14.9% - - - - - - - - - 11%
c. Black ............................. 1988 11.2% 10.7% 11.0% 12.2% 12.6% 12.5% 12.2% 12.5% - - - - - - - - - 9%
d. Puerto Rican ....................... 1989–91 11.7% . . . . . .
6
12.0%
7
12.7%
8
13.4%
9
13.4%
10
13.0% - - - - - - - - - 10%
17.3* People with difficulty performing self-care
activities (per 1,000)
People 65 years and over ................. 1984–85 111 - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 90
People 70 years and over ................. 1984–85
§
141 --- --- --- --- ---
11
163 --- --- --- --- 90
a. People 85 years and over.............. 1984–85 371 - - - - - - - - - - - - - - -
11
471 --- --- --- --- 325
b. Black 65 years and over ............... 1984–85
§
132 --- --- --- --- --- --- --- --- --- --- 98
Black 70 years and over ............... 1984–85
§
166 --- --- --- --- ---
11
218 --- --- --- --- 98
17.4 People with asthma with activity limitation
due to asthma ........................ 1986–88 19.4%
4
20.4%
5
21.8%
6
21.8%
7
22.5%
8
22.0%
9
20.7%
10
19.6 - - - - - - - - - 10%
a. Black ............................. 1989–91 30.5% . . . . . .
6
30.3%
7
32.1%
8
31.5%
9
29.1%
10
27.0 - - - - - - - - - 19%
b. Puerto Ricans
12
..................... ... --- --- --- --- --- --- --- --- --- --- --- 22%
17.5 Activity limitation due to chronic back
conditions (per 1,000) .................. 1986–88 21.9
4
23.7
5
25.1
6
25.3
7
27.3
8
28.1
9
28.8
10
27.9 - - - - - - - - - 19
17.6 Significant hearing impairment (per 1,000) . . 1986–88 88.9
4
89.5
5
89.7
6
93.5
7
93.6
8
91.9
9
89.0
10
85.1 - - - - - - - - - 82
a. People 45 years and over.............. 1986–88 203
4
206.2
5
205.2
6
215.7
7
213.2
8
207.4
9
200.4
10
195.9 - - - - - - - - - 180
17.7 Significant visual impairment (per 1,000) .... 1986–88 34.5
4
32.5
5
31.7
6
32.8
7
34.8
8
35.1
9
34.0
10
31.3 - - - - - - - - - 30
a. People 65 years and over.............. 1986–88 87.7
4
81.8
5
78.0
6
79.8
7
87.4
8
88.3
9
84.6
10
84.2 - - - - - - - - - 70
17.8* Serious mental retardation (per 1,000)
Children 10 years with IQ less than 50 ....... 1985–87
§
3.1 --- ---
13
3.6 --- --- --- --- --- --- --- 2
17.9 Diabetes-related deaths (age adjusted per
100,000) ............................. 1986 38 38 38 38 40 40 40 41 41 41 - - - 34
a. Black ............................. 1986 67 71 71 71 74 73 76 76 76 76 - - - 58
b. American Indian/Alaska Native .......... 1986 46 53 51 57 60 58 63 63 66 66 - - - 41
c. Mexican American
14
.................. 1990
§
55.7 . . . 50.3 51.1 56.6 55.6 56.7 60.1 62.6 60.7 - - - 50
d. Puerto Rican
14
...................... 1990
§
40.7 . . . 47.2 48.7 48.5 57.8 63.2 58.5 51.7 49.1 - - - 42
17.10 Diabetes-related complications
People with diabetes
End-stage renal disease (ESRD) (per 1,000) . 1987 1.5 2.5 2.5 2.7 2.4 3.3 3.4 4.1
‡
3.2
‡
3.3 - - - 1.4
Blindness (age adjusted per 1,000) ........ 1987 2.2 2.5 2.4 2.3 2.1 2.2 - - - - - - - - - - - - - - - 1.4
Lower extremity amputation (per 1,000) ..... 1987 8.2 8.6 6.2 7.8 7.3 8.6 9.4 11.1
‡
8.1
‡
8.1 - - - 4.9
Perinatal mortality (among infants of females
with established diabetes) .............. 1988 5% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 2%
See footnotes and key at end of table.
244 Healthy People 2000 Final Review

Table 17. Diabetes and Chronic Disabling Conditions objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Major congenital malformations............... 1988 8% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 4%
ESRD due to diabetes (per 1,000)
a. Black with diabetes..................... 1983–86 2.2
15
3.1 --- ---
16
5.7
17
5.0
18
5.2
19
5.5 - - -
‡,20
5.7 - - - 2.0
b. American Indian/Alaska Native in Indian
Health Service areas with diabetes
21
....... 1983–86 2.1 4.2 4.4 5.4 - - - - - - - - - - - - - - - - - - - - - 1.9
Lower extremity amputations due to diabetes
(per 1,000)
c. Black with diabetes ..................... 1987
§
9.0 8.0 11.1 8.6 8.6 9.1 10.2 10.1
‡
9.1
‡
11.7 - - - 6.1
17.11* Diabetes incidence and prevalence
Total population (per 1,000)
Incidence of diabetes..................... 1986–88 2.9
4
2.6
5
2.5
6
2.4
7
2.8
8
3.1
9
3.4
10
3.1 --- --- --- 2.5
Prevalence of diabetes ................... 1986–88 28
4
26
5
27
6
28
7
30
8
30
9
31
10
31
‡
---
‡
--- --- 25
Prevalence of diabetes (per 1,000)
a. American Indian/Alaska Native 15 years and
over in Indian Health Service areas ........ 1987 69 - - - - - - - - - - - - - - - - - -
21
90 --- --- --- 62
b. Puerto Rican (ages 20–74 years) .......... 1982–84 55 - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 49
c. Mexican American (ages 20–74 years) ...... 1982–84 54 - - - - - - - - - - - -
22
66 --- --- --- --- --- 49
d. Cuban American (ages 20–74 years) ....... 1982–84 36 - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 32
e. Black (all ages)........................ 1986–88 36
4
36
5
36
6
36
7
38
8
40
9
42
10
44
‡
---
‡
--- --- 32
17.12* Overweight prevalence (Based on measured
height and weight unless otherwise indicated)
Adults 20–74 years ........................ 1976–80 26% - - - - - - - - - - - -
21,23
3 --- --- --- --- --- 20%
Male ................................. 1976–80 24% - - - - - - - - - - - -
21,23
3 --- --- --- --- --- 20%
Female ............................... 1976–80 27% - - - - - - - - - - - -
21,23
3 --- --- --- --- --- 20%
Adolescents 12–19 years ................... 1976–80 15% - - - - - - - - - - - -
23
24% --- --- --- --- --- 15%
a. Low-income female 20–74 years........... 1976–80 37% - - -
24
47% --- --- --- --- --- --- --- --- 25%
b. Black female 20–74 years................ 1976–80 44% - - - - - - - - - - - -
21,23
5 --- --- --- --- --- 30%
c. Hispanic female 20–74 years ............. ... --- --- --- --- --- --- --- --- --- --- --- 25%
Hispanic female 20 years and over
(self-reported)
25
...................... ... --- 33% 32% 32% 33% 32% 35% ---
‡
44%
‡
46% --- ...
Mexican American female 20–74 years..... 1982–84 39% - - - - - - - - - - - -
21,23
50% --- --- --- --- --- ...
Cuban female 20–74 years ............. 1982–84 34% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Puerto Rican female 20–74 years......... 1982–84 37% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
d. American Indian/Alaska Native 20 years and
over ............................... 1984–88 29–75% - - -
25
40%
25
36%
25
48%
25
34%
25
43% - - -
‡,25
46
‡,25
45 - - - 30%
e. People with disabilities 20 years and over
(self-reported)
25
....................... 1985 36% - - - 38% 37% 38% 38% 40% - - - - - - - - - - - - 25%
f. Females with high blood pressure 20–74
years ............................... 1976-80 50% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 41%
g. Males with high blood pressure 20–74 years. . 1976–80 39% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 35%
h. Mexican American male 20–74 years ....... 1982–84 30% - - - - - - - - - - - -
21,23
3 --- --- --- --- --- 25%
17.13* Light to moderate physical activity
People 6 years and over.................... ... --- --- --- --- --- --- --- --- --- --- --- 30%
See footnotes and key at end of table.
Healthy People 2000 Final Review 245

Table 17. Diabetes and Chronic Disabling Conditions objectives—Con
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
People 18 years and over
5 or more times per week ................ 1985
26
22%
26
23%
27
24% --- --- --- 23% --- --- 30% --- 30%
7 or more times per week ................ 1985
26
16%
26
16%
27
17% --- --- --- 16% --- --- 20% --- 30%
a. Hispanic 18 years and over
5 or more times per week ................ 1991 20% . . . . . . - - - - - - - - - 22% - - - - - - 29% - - - 25%
17.14 Patient education for people with chronic and
disabling conditions ....................
... --- --- --- --- --- --- --- --- --- --- --- 40%
a. People with diabetes (classes) ........... 1983-84 32%
28
3 3 % 3 9 % --- 4 3 % --- --- --- --- 4 2 % --- 7 5 %
People with diabetes (counseling) ......... 1983-84 68% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
b. People with asthma (classes) ............ 1991 9% . . . . . . - - - 10% - - - - - - - - - - - - 8% - - - 50%
c. Black with diabetes (classes) ............ 1991 34% . . . . . . - - - 50% - - - - - - - - - - - - 45% - - - 75%
d. Hispanic with diabetes (classes) .......... 1991 27% . . . . . . - - - 26% - - - - - - - - - - - - 31% - - - 75%
17.15 Clinician assessment of childhood
development ..........................
... --- --- --- --- --- --- --- --- --- --- --- 80%
Percent of clinicians routinely providing service to
81–100% of patients (children)
Visual acuity testing (3 years and over)
Pediatricians .......................... 1992 55% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Nurse practitioners ..................... 1992 49% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Family physicians ...................... 1992 30% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Hearing testing (3 years and over)
Pediatricians .......................... 1992 47% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Nurse practitioners ..................... 1992 46% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Family physicians ...................... 1992 19% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Evaluation of speech
Pediatricians .......................... 1992 65% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 80%
Nurse practitioners ..................... 1992 51% . . . . . . . . . - - - - - - - - - - - - - - -
29
49% - - - 80%
Family physicians ...................... 1992 39% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 80%
Evaluation of motor development
Pediatricians .......................... 1992 72% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 80%
Nurse practitioners ..................... 1992 56% . . . . . . . . . - - - - - - - - - - - - - - -
29
53% - - - 80%
Family physicians ...................... 1992 45% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 80%
Treatment/referral for vision problems
Pediatricians .......................... 1992 67% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Nurse practitioners ..................... 1992 35% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Family physicians ...................... 1992 56% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Treatment/referral for hearing problems
Pediatricians .......................... 1992 66% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Nurse practitioners ..................... 1992 35% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Family physicians ...................... 1992 55% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Treatment/referral for speech problems
Pediatricians .......................... 1992 62% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Nurse practitioners ..................... 1992 34% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Family physicians ...................... 1992 48% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
See footnotes and key at end of table.
246 Healthy People 2000 Final Review

Table 17. Diabetes and Chronic Disabling Conditions objectives—Con
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Treatment/referral for motor problems
Pediatricians .......................... 1992 55% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Nurse practitioners ..................... 1992 33% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
Family physicians ...................... 1992 49% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 80%
17.16 Early detection of significant hearing
impairment in children (average age in
months)
31
............................. 1988 24–30 - - - 27 - - - - - - - - - - - - - - - - - - 29 - - - 12
a. Black
12
............................. ... --- --- --- --- --- --- --- --- --- --- --- 12
17.17 Clinician assessment of cognitive and other
functioning in older adults
Percent of clinicians routinely providing service to
81–100% of patients (adults aged 65 years and
over)
Visual acuity testing
Nurse practitioners ..................... 1992 24% . . . . . . . . . - - - - - - - - - - - - - - -
29
19% - - - 60%
Obstetricians/gynecologists ............... 1992 3% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Internists ............................. 1992 15% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Family physicians ...................... 1992 12% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Hearing acuity testing
Nurse practitioners ..................... 1992 16% . . . . . . . . . - - - - - - - - - - - - - - -
29
13% - - - 60%
Obstetricians/gynecologists ............... 1992 2% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Internists ............................. 1992 9% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Family physicians ...................... 1992 7% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Evaluation of physical mobility
Nurse practitioners ..................... 1992 41% . . . . . . . . . - - - - - - - - - - - - - - -
29
35% - - - 60%
Obstetricians/gynecologists ............... 1992 18% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Internists ............................. 1992 42% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Family physicians ...................... 1992 26% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Evaluation for dementia
Nurse practitioners ..................... 1992 28% . . . . . . . . . - - - - - - - - - - - - - - -
29
22% - - - 60%
Obstetricians/gynecologists ............... 1992 9% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Internists ............................. 1992 23% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Family physicians ...................... 1992 13% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Inquiry about urinary incontinence
Nurse practitioners ..................... 1992 33% . . . . . . . . . - - - - - - - - - - - - - - -
29
24% - - - 60%
Obstetricians/gynecologists ............... ... --- --- --- --- --- --- --- --- ---
29,30
--- --- 60%
Internists ............................. 1992 30% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Family physicians ...................... 1992 15% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Treatment/referral for vision problems
Nurse practitioners ..................... 1992 33% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 60%
Obstetricians/gynecologists ............... 1992 35% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 60%
Internists ............................. 1992 63% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 60%
Family physicians ...................... 1992 54% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 60%
See footnotes and key at end of table.
Healthy People 2000 Final Review 247

Table 17. Diabetes and Chronic Disabling Conditions objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Treatment/referral for hearing problems
Nurse practitioners ..................... 1992 30% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 60%
Obstetricians/gynecologists ............... 1992 34% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 60%
Internists ............................. 1992 52% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 60%
Family physicians ...................... 1992 46% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 60%
Prescription of mobility aids/modification of living
environment to improve mobility
Nurse practitioners ..................... 1992 18% . . . . . . . . . - - - - - - - - - - - - - - -
29
25% - - - 60%
Obstetricians/gynecologists ............... 1992 15% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Internists ............................. 1992 31% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Family physicians ...................... 1992 25% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Investigation of/referral for treatable causes of
dementia
Nurse practitioners ..................... 1992 31% . . . . . . . . . - - - - - - - - - - - - - - -
29
28% - - - 60%
Obstetricians/gynecologists ............... 1992 27% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Internists ............................. 1992 54% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Family physicians ...................... 1992 40% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Treatment/referral for urinary incontinence
Nurse practitioners ..................... 1992 31% . . . . . . . . . - - - - - - - - - - - - - - -
29
28% - - - 60%
Obstetricians/gynecologists ............... 1992 56% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Internists ............................. 1992 37% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
Family physicians ...................... 1992 31% . . . . . . . . . - - - - - - - - - - - - - - -
29,30
--- --- 60%
17.18 Perimenopausal females counseled about
estrogen replacement therapy
Female 40–60 years .................... 1994 80% . . . . . . . . . . . . . . . - - - - - - - - - 89% - - - 90%
Female 40–49 years .................. 1994 76% . . . . . . . . . . . . . . . - - - - - - - - - 87% - - - 90%
Female 50–60 years .................. 1994 83% . . . . . . . . . . . . . . . - - - - - - - - - 90% - - - 90%
17.19 Worksites with policies for hiring people with
disabilities
32
Worksites with a voluntary policy............. 1986 37% 100% 100% 100% 100% 100% 100% 100% 100% 100% 100% 75%
17.20 Service systems for children with or at risk of
chronic and disabling conditions (number of
States) ............................... ... --- --- --- --- --- --- --- --- --- --- --- 50
17.21 Prevalence of peptic ulcer (per 1,000)
33
People 18 years and over .................. 1991 19.9 . . . . . . 23.7 24.4 23.0 22.0 21.2
‡
30.4
‡
27.7 - - - 18
17.22* Identify gaps in health data ............... 1990 None
identified
--- --- --- ---
34
---
35
---
36
---
36
---
37
---
37
--- ...
Establish mechanisms to meet needs ......... 1990 None
established
--- --- --- ---
34
---
35
---
36
---
36
---
37
---
37
--- ...
17.23 People with diabetes who had a dilated eye
exam in the past year
People 18 years and over .................. 1989 49% - - -
24
52% --- --- --- --- --- --- 53% --- 70%
- - - Data not available. Final objective status:
. . . Category not applicable.
Met
Toward
Mixed/ no change
Away
Cannot assess
248 Healthy People 2000 Final Review

§
Baseline has been revised.
‡
The NHIS was redesigned in 1997. Data may not be comparable with previous years; see Appendix.
1
Estimate based on preliminary data. Excludes mortality data from States lacking an Hispanic-origin item on their death certificate or for which Hispanic-origin data were not of sufficient quality.
See Appendix.
2
Estimate derived from 1991–93 health status data and 1992 mortality data.
3
Years of healthy life remaining for those surviving to age 65.
4
1988–90 data.
5
1989–91 data.
6
1990–92 data.
7
1991–93 data.
8
1992–94 data.
9
1993–95 data.
10
1994–96 data.
11
1994–95 data.
12
Data are unreliable. Relative standard error is greater than 30%.
13
1991–92 data.
14
Excludes data from States lacking an Hispanic-origin item on their death certificates or for which Hispanic-origin data were not of sufficient quality. See appendix.
15
1986–89 data.
16
1990–93 data.
17
1991–94 data.
18
1992–95 data.
19
1993–96 data.
20
1995–98 data.
21
Data are for people 20 years and over.
22
Crude data from 1988–94.
23
1988–94 data.
24
1988–91 data.
25
Estimate derived from self-reported height and weight.
26
Data are for people 18–64 years.
27
Operational definition changed from previous tracking data.
28
1989 data.
29
1997–98 data.
30
Response rate for this group was too low to produce reliable estimates.
31
Among hearing-impaired children 4–6 years.
32
Assuming full compliance, achieved through passage of the Americans with Disabilities Act of 1990.
33
Includes stomach, duodenal, and peptic ulcers.
34
The National Committee on Vital and Health Statistics established a Subcommittee on State and Community Health Statistics. The Subcommittee’s charge (in part) is to work with Federal
and State agencies and appropriate private agencies to review and identify gaps in current health statistics.
35
The Healthy People 2000 Midcourse Review added 111 additional subobjectives for major population groups at highest risk for disease, injury, and disability.
36
As part of the planning process for 2010, data gaps are being identified and mechanisms to address these gaps are being considered.
37
Healthy People 2010 is addressing data gaps by requiring data for race, ethnicity, socioeconomic status, and disability measures for population-based objectives and by listing developmental
objectives.
NOTE: Data may include revisions and, therefore, may differ from data previously published.
Objective number Data source
17.1*, 17.1a–c National Vital Statistics System, CDC, NCHS; National Health Interview Survey, CDC, NCHS.
17.2, 17.2a–c National Health Interview Survey, CDC, NCHS.
17.3*, 17.3a, b National Health Interview Survey, CDC, NCHS; National Nursing Home Survey, CDC, NCHS.
17.4 National Health Interview Survey, CDC, NCHS.
17.5 National Health Interview Survey, CDC, NCHS.
17.6, 17.6a National Health Interview Survey, CDC, NCHS.
17.7, 17.7a National Health Interview Survey, CDC, NCHS.
17.8* Baseline: Metropolitan Atlanta Developmental Disabilities Study, CDC, NCEH.
Update: Metropolitan Atlanta Developmental Disabilities Surveillance Program, CDC, NCEH.
17.9, 17.9a, b National Vital Statistics System, CDC, NCHS.
Healthy People 2000 Final Review 249

Objective number Data source
17.10 For blindness: Massachusetts Blind Registry, Massachusetts Commission on the Blind.
For perinatal mortality and congenital malfunctions: Clinical series and selected State data.
For ESRD:
Denominator: National Health Interview Survey, CDC, NCHS;
Numerator: United States Renal Data System, NIH.
For amputation:
Denominator: National Health Interview Survey, CDC, NCHS;
Numerator: National Hospital Discharge Survey, CDC, NCHS.
17.10a Denominator: National Health Interview Survey, CDC, NCHS;
Numerator: United States Renal Data System, NIH.
17.10b IHS, OPEL.
17.10c Denominator: National Health Interview Survey, CDC, NCHS;
Numerator: National Hospital Discharge Survey, CDC, NCHS.
17.11*, 17.11e National Health Interview Survey, CDC, NCHS.
17.11a Ambulatory Utilization Data, IHS.
17.11b–d Baseline: Hispanic Health and Nutrition Examination Survey, CDC, NCHS.
Update for Mexican American: National Health and Nutrition Examination Survey, CDC, NCHS.
17.12*, 17.12a,
b, f, g
National Health and Nutrition Examination Survey, CDC, NCHS.
17.12c, h Data for Hispanic: National Health Interview Survey, CDC, NCHS.
Baselines for Mexican American, Cuban, Puerto Rican: Hispanic Health and Nutrition Examination Survey, CDC, NCHS.
Updates for Mexican American: National Health and Nutrition Examination Survey, CDC, NCHS.
17.12d Baseline: IHS, OPEL.
Updates: National Health Interview Survey, CDC, NCHS.
17.12e National Health Interview Survey, CDC, NCHS.
17.13*, 17.13a National Health Interview Survey, CDC, NCHS.
17.14a 1983–84 baseline: Halpern M. The impact of diabetes education in Michigan. Diabetes 38(2):151A, 1989.
1991 baseline and updates: National Health Interview Survey, CDC, NCHS.
17.14b National Health Interview Survey, CDC, NCHS.
17.15 Baseline: Primary Care Provider Surveys, OPHS, ODPHP.
Update: Prevention in Primary Care Study, American College of Preventive Medicine.
17.16 1988 baseline: Annual Survey of Hearing Impaired Children and Youth, Commission on Education of the Deaf.
1991 baseline and updates: National Health Interview Survey, CDC, NCHS.
17.17 Baseline: Primary Care Provider Surveys, OPHS, ODPHP.
Update: Prevention in Primary Care Study, American College of Preventive Medicine.
17.18 National Health Interview Survey, CDC, NCHS.
17.19 Baseline: Survey of Persons with Disability, International Center for the Disabled.
Updates: Americans with Disabilities Act of 1990.
17.21 National Health Interview Survey, CDC, NCHS.
17.22* Subcommittee on State and Community Health Statistics, NCVHS; CDC, NCHS; OPHS, ODPHP.
17.23 Baseline: National Health Interview Survey, CDC, NCHS.
Update: National Health and Nutrition Examination Survey, CDC, NCHS.
* Duplicate objective. See full text of objective following this table.
250 Healthy People 2000 Final Review

Diabetes and
Chronic Disabling
Conditions
Objectives
17.1*: Increase years of healthy life to
at least 65 years.
Duplicate objectives: 8.1 and 21.1
17.1a*: Increase years of healthy
life among blacks to at least 60
years.
Duplicate objectives: 8.1a and 21.1a
17.1b*: Increase years of healthy
life among Hispanics to at least 65
years.
Duplicate objectives: 8.1b and 21.1b
17.1c*: Increase years of healthy
life among people aged 65 and
older to at least 14 more years of
healthy life.
Duplicate objectives: 8.1c and 21.1c
17.2: Reduce to no more than 8 percent
the proportion of people who experience
a limitation in major activity due to
chronic conditions.
17.2a: Reduce to no more than
15 percent the proportion of
low-income people (annual family
income of less than $10,000 in
1988) who experience a limitation
in major activity due to chronic
conditions.
17.2b: Reduce to no more than
11 percent the proportion of
American Indians and Alaska
Natives who experience a limitation
in major activity due to chronic
conditions.
17.2c: Reduce to no more than
9 percent the proportion of blacks
who experience a limitation in
major activity due to chronic
conditions.
17.2d: Reduce to no more than
10 percent the proportion of Puerto
Ricans who experience a limitation
in major activity due to chronic
conditions.
17.3*: Reduce to no more than 90 per
1,000 people the proportion of all
people aged 65 and older who have
difficulty in performing two or more
personal care activities, thereby
preserving independence.
Duplicate objective: 1.13 and age-related
objective for people aged 65 and older
17.3a*: Reduce to no more than
325 per 1,000 people the proportion
of all people aged 85 and older who
have difficulty in performing two or
more personal care activities,
thereby preserving independence.
Duplicate objective: 1.13a
17.3b*: Reduce to no more than 98
per 1,000 people the proportion of
blacks aged 65 and older who have
difficulty in performing two or more
personal care activities, thereby
preserving independence.
Duplicate objective: 1.13b
17.4: Reduce to no more than 10 percent
the proportion of people with asthma
who experience activity limitation.
17.4a: Reduce to no more than
19 percent the proportion of blacks
with asthma who experience activity
limitation.
17.4b: Reduce to no more than
22 percent the proportion of Puerto
Ricans with asthma who experience
activity limitation.
17.5: Reduce activity limitation due to
chronic back conditions to a prevalence
of no more than 19 per 1,000 people.
17.6: Reduce significant hearing
impairment to a prevalence of no more
than 82 per 1,000 people.
17.6a: Reduce significant hearing
impairment among people aged 45
and older to a prevalence of no
more than 180 per 1,000.
17.7: Reduce significant visual
impairment to a prevalence of no more
than 30 per 1,000 people.
17.7a: Reduce significant visual
impairment among people aged 65
and older to a prevalence of no
more than 70 per 1,000.
17.8*: Reduce the prevalence of serious
mental retardation among school-aged
children to no more than 2 per 1,000
children.
Duplicate objective: 11.2
17.9: Reduce diabetes-related deaths to
no more than 34 per 100,000.
17.9a: Reduce diabetes-related
deaths among blacks to no more
than 58 per 100,000.
17.9b: Reduce diabetes-related
deaths among American Indians and
Alaska Natives to no more than 48
per 100,000.
17.9c: Reduce diabetes-related
deaths among Mexican-Americans
to no more than 50 per 100,000.
17.9d: Reduce diabetes-related
deaths among Puerto Ricans to no
more than 42 per 100,000.
17.10: Reduce the most severe
complications of diabetes as follows:
Complications among
people with diabetes: 2000 target
End-stage renal disease 1.4 per 1,000
Blindness 1.4 per 1,000
Lower extremity amputation 4.9 per
1,000
Perinatal mortality
1
2 percent
Major congenital malformation 4 percent
1
Among infants of women with established
diabetes.
17.10a: Reduce end-stage renal
disease due to diabetes among black
persons with diabetes to no more
than 2 per 1,000.
17.10b: Reduce end-stage renal
disease due to diabetes among
American Indians and Alaska
Natives with diabetes to no more
than 1.9 per 1,000.
17.10c: Reduce lower extremity
amputations due to diabetes among
blacks with diabetes to no more
than 6.1 per 1,000.
17.11*: Reduce diabetes to an incidence
of no more than 2.5 per 1,000 people
and a prevalence of no more than 25 per
1,000 people.
Duplicate objective: 2.24
17.11a*: Reduce diabetes among
American Indians and Alaska
Natives to a prevalence of no more
than 62 per 1,000.
Duplicate objective: 2.24a
17.11b*: Reduce diabetes among
Puerto Ricans to a prevalence of no
more than 49 per 1,000.
Duplicate objective: 2.24b
17.11c*: Reduce diabetes among
Mexican-Americans to a prevalence
of no more than 49 per 1,000.
Healthy People 2000 Final Review 251
Duplicate objective: 2.24c
17.11d*: Reduce diabetes among
Cuban Americans to a prevalence of
no more than 32 per 1,000.
Duplicate objective: 2.24d
17.11e*: Reduce diabetes among
blacks to a prevalence of no more
than 32 per 1,000.
Duplicate objective: 2.24e
17.12*: Reduce overweight to a
prevalence of no more than 20 percent
among people aged 20 and older and no
more than 15 percent among adolescents
aged 12–19.
Duplicate objectives: 1.2, 2.3, and 15.10
17.12a*: Reduce overweight to a
prevalence of no more than
25 percent among low-income
women aged 20 and older.
Duplicate objectives: 1.2a, 2.3a, and
15.10a
17.12b*: Reduce overweight to a
prevalence of no more than
30 percent among black women
aged 20 and older.
Duplicate objectives: 1.2b, 2.3b, and
15.10b
17.12c*: Reduce overweight to a
prevalence of no more than
25 percent among Hispanic women
aged 20 and older.
Duplicate objectives: 1.2c, 2.3c, and
15.10c
17.12d*: Reduce overweight to a
prevalence of no more than
30 percent among American Indians
and Alaska Natives.
Duplicate objectives: 1.2d, 2.3d, and
15.10d
17.12e*: Reduce overweight to a
prevalence of no more than
25 percent among people with
disabilities.
Duplicate objectives: 1.2e, 2.3e, and
15.10e
17.12f*: Reduce overweight to a
prevalence of no more than
41 percent among women with high
blood pressure.
Duplicate objectives: 1.2f, 2.3f, and
15.10f
17.12g*: Reduce overweight to a
prevalence of no more than
35 percent among men with high
blood pressure.
Duplicate objectives: 1.2g, 2.3g, and
15.10g
17.12h*: Reduce overweight to a
prevalence of no more than
35 percent among
Mexican-American men.
Duplicate objectives: 1.2h, 2.3h, and
15.10h
17.13*: Increase to at least 30 percent
the proportion of people aged 6 and
older who engage regularly, preferably
daily, in light-to-moderate physical
activity for at least 30 minutes per day.
Duplicate objectives: 1.3 and 15.11
17.13a*: Increase to at least
25 percent the proportion of
Hispanics aged 18 and older who
engage regularly, preferably daily,
in light to moderate physical
activity for at least 30 minutes per
day 5 or more times per week.
Duplicate objectives: 1.3a and
15.11a
17.14: Increase to at least 40 percent the
proportion of people with chronic and
disabling conditions who receive formal
patient education including information
about community and self-help
resources as an integral part of the
management of their condition.
17.14a: Increase to at least
75 percent the proportion of people
with diabetes who receive formal
patient education including
information about community and
self-help resources as an integral
part of the management of their
condition.
17.14b: Increase to at least
50 percent the proportion of people
with asthma who receive formal
patient education including
information about community and
self-help resources as an integral
part of the management of their
condition.
17.14c: Increase to at least
75 percent the proportion of blacks
with diabetes who receive formal
patient education including
information about community and
self-help resources as an integral
part of the management of their
condition.
17.14d: Increase to at least
75 percent the proportion of
Hispanics with diabetes who receive
formal patient education including
information about community and
self-help resources as an integral
part of the management of their
condition.
17.15: Increase to at least 80 percent the
proportion of providers of primary care
for children who routinely refer or
screen infants and children for
impairments of vision, hearing, speech
and language, and assess other
developmental milestones as part of
well-child care.
17.16: Reduce the average age at which
children with significant hearing
impairment are identified to no more
than 12 months.
17.16a: Reduce the average age at
which black children with
significant hearing impairment are
identified to no more than 12
months.
17.17: Increase to at least 60 percent the
proportion of providers of primary care
for older adults who routinely evaluate
people aged 65 and older for urinary
incontinence and impairments of vision,
hearing, cognition, and functional status.
17.18: Increase to at least 90 percent the
proportion of perimenopausal women
who have been counseled about the
benefits and risks of estrogen
replacement therapy (combined with
progestin, when appropriate) for
prevention of osteoporosis.
17.19: Increase to at least 75 percent the
proportion of worksites with 50 or more
employees that have a policy or
program for the hiring of people with
disabilities.
17.20: Increase to 50 the number of
States that have service systems for
children with or at risk of chronic and
disabling conditions, as required by
Public Law 101–239.
17.21: Reduce the prevalence of peptic
ulcer disease to no more than 18 per
1,000 people aged 18 and older by
preventing its recurrence.
17.22*: Develop and implement a
national process to identify significant
gaps in the Nation’s disease prevention
and health promotion data, including
data for racial and ethnic minorities,
people with low incomes, and people
252 Healthy People 2000 Final Review
with disabilities, and establish
mechanisms to meet these needs.
Duplicate objective: 22.4
17.23: Increase to 70 percent the
proportion of people with diabetes who
have an annual dilated eye exam.
*Duplicate objective.
Healthy People 2000 Final Review 253

Priority Area 18
HIV Infection
Background
The epidemic of human
immunodeficiency virus (HIV) and
acquired immunodeficiency syndrome
(AIDS) presents social, economic, and
public health challenges to individuals
in the United States and globally.
Although significant progress has been
made in understanding the disease and
developing both prevention strategies
and treatments since the first AIDS case
was discovered in the United States in
1981, HIV remains a deadly infection
for which there are only limited
treatments; no vaccine or cure exists.
HIV/AIDS has been reported in virtually
every racial and ethnic group, every age
group, and every socioeconomic group
in every State and most large cities in
the United States. Since the early 1980s
four distinct populations have been
identified to be at greatest risk: men
who have sex with men (MSM);
injection drug users (IDUs);
heterosexual persons in certain racial
and ethnic populations (African
Americans and Hispanics); and infants
who are susceptible to perinatal
transmission caused by undetected or
untreated HIV infection in pregnant
women (1–3). The proportion of
different population groups affected by
HIV/AIDS has changed over time.
Comparing the 1980s to the 1990s, the
proportion of AIDS cases among white
MSM declined while the proportion of
AIDS cases among women and men
increased in select racial and ethnic
groups, especially among African
Americans and Hispanics. Moreover,
increases among all women have
occurred. Reported AIDS cases among
women have increased steadily since the
1980’s and accounted for nearly
18 percent of cases reported in 1999 (4).
By the end of 1999, more than 733,000
cases of AIDS had been reported, and
over 430,000 persons had died of AIDS.
Estimates of the number of people
infected with HIV in the United States
range from 800,000 to 900,000. (4–6).
Transmission of HIV infection can be
prevented through changes in high-risk
behaviors. Prevention is an important
cost-effective component of the control
of HIV infection. Two biomedical
interventions have demonstrated
possibilities in reducing the spread of
this deadly disease. First, antiretroviral
combination therapy lowers viral load,
which may translate to lower infectivity
and, second, evidence suggests that
treatment of other sexually transmitted
diseases (STDs) can reduce the spread
of heterosexually transmitted HIV
infection. Despite advances in
prevention and medical treatment to
improve survival among infected
persons, HIV infection remains a major
public health threat in the United States.
Data Summary
Highlights
By the end of the 20th century,
successes had been achieved in the HIV
Infection priority area. The publication,
dissemination, and implementation of
specific guidelines to test donated blood
for HIV has resulted in a reduction in
transfusion-related AIDS (18.7). The
estimated risk of transfusion-transmitted
HIV dropped from 1 case per
40,000–150,000 units of donated blood
in 1989 to 1 per 625,000 units in 1997.
Assuming full compliance with the
Occupational Safety and Health
Administration bloodborne pathogen
standard promulgated in 1991,
100 percent of worksites had regulations
to protect workers from occupational
exposure to bloodborne infections,
including HIV (18.14) (5,7). Several
other measures related to HIV infection
status improved over the decade. For
example, AIDS incidence continues to
decrease and has met its year 2000
target. The estimated number of AIDS
cases per 100,000 population by year of
diagnosis decreased between 1997 and
1998 (18.1) (see Data Issues). The
number of cases diagnosed in 1998
among MSM (18.1a) and IDUs (18.1c)
also decreased compared with the
number of cases in 1997. Since 1992,
HIV incidence in the United States has
been relatively stable, with an estimated
40,000 new HIV infections expected to
occur each year. The latest estimates of
HIV infection (18.2) indicated that
800,000 to 900,000 persons are infected
with HIV (4–6).
In 1999, among in-school male and
female adolescents 15 years of age, 34
and 43 percent, respectively, engaged in
sexual intercourse (18.3), whereas
68 percent among black adolescent
males in this age group engaged in
sexual intercourse. Among 17 year olds,
the proportion of adolescents engaging
in sexual intercourse decreased in all
groups (44 percent, 40 percent, and
82 percent, respectively). In addition, the
proportion of condoms used at last
sexual intercourse by the partners of
sexually active unmarried females 15–44
years of age (18.4) increased from
19 percent to 25 percent between 1988
and 1995. Between 1997 and 1999, the
proportion of in-school sexually active
females (15–17 years of age) abstaining
from sexual intercourse for the previous
3 months (18.15) improved from
23 percent to 25 percent. About a quarter
of a million people in the United States
are unaware that they are infected with
AIDS (8); knowledge of serostatus is
key to HIV-positive people benefiting
from prevention and treatment. The
proportion of HIV-positive persons who
knew their serostatus (18.8) decreased
over the decade. Between 1995 and
1998, the proportion of individuals who
tested positive for HIV infection and
returned for counseling decreased from
83 percent to 63 percent. Some
78 percent of persons who were tested at
a publicly funded site completed posttest
counseling sessions in 1998 (8).
Knowledge of HIV status is a critical
prevention strategy and essential for
entry into care services. Once rapid HIV
tests are available, test results can be
provided in the same day, eliminating
the need for a person to return for test
results.
Summary of Progress
Data to assess progress are available
for 14 of the 17 objectives in this
priority area. Five objectives have met
(18.10, and 18.14) or exceeded (18.1,
18.2, and 18.7) the year 2000 targets.
Objective 18.10 (HIV and other STD
education curricula) was met at baseline,
however, supplemental data for the
objective indicate a trend that could be
moving in the wrong direction. Data
show progress toward the year 2000
targets for an additional five objectives
(18.4, 18.5, 18.6, 18.13, and 18.15).
Objectives 18.8, 18.9, and 18.12 moved
away from the year 2000 targets. For
one objective (18.3), progress among the
multiple measures used for tracking was
mixed. Data beyond baseline are not
available for assessing the status of two
objectives (18.11 and 18.16). Baseline
data are not available for one objective
(18.17). See table 18 for the tracking
data for the objectives in this priority
254 Healthy People 2000 Final Review

18.1 AIDS incidence
§
18.2 HIV prevalence
18.3 Adolescents who ever had sexual intercourse:
Female 15 years
18.4 Condom use at last intercourse:
Female 15-44 years
Female 17 years
Male 17 years
Male 15 years
18.5 Injecting drug users enrolled in treatment
18.6 Injecting drug users not in treatment who did
not share needles
18.7 Risk of transfusion-transmitted HIV infection
18.14 Regulations to protect workers from occupational
exposure to bloodborne infections, including HIV
18.15 Adolescent abstinence from sexual intercourse in
past 3 months: Female 15-17 years
Male 15-17 years
18.12 Outreach HIV programs for drug users
18.8 HIV-infected people who know their serostatus
Moved away from target Moved toward target Met target
* This objective has met its target. A progress quotient could not be calculated.
NOTES: Complete tracking data are shown in table 18. Progress quotients are not calculated for objectives 18.9, 18.10, 18.11, 18.13, 18.16,
and 18.17. See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
AIDS is aquired immunodeficiency syndrome. HIV is human immunodeficiency virus.
-10%
-6%
Percent of target achieved
-100 -80 -60 -40 -20
0
20 40 60 80 100
Met*
Met*
131%
100%
57%
21%
66%
59%
42%
33%
50%
19%
-127%
§
For explanation, see Data Comparability in chapter text.
Healthy People 2000 Final Review 255
Figure 18. Final status of HIV Infection objectives
area and figure 18 for a quantitative
assessment of progress.
Discussion
Historically, AIDS incidence data
have served as the basis for assessing
needs for prevention and treatment
programs. However, because of the
effect of potent antiretroviral therapies,
AIDS incidence no longer can provide
unbiased information on HIV incidence
patterns. Rather, persons reported with
AIDS will increasingly represent
persons who were diagnosed too late for
them to benefit from treatments, persons
who either did not seek or had no
access to care, or persons who failed
treatment (4,5). A nationwide system of
HIV reporting is needed to better track
where HIV infections are occurring. By
the end of 1999, 34 States had adopted
some type of HIV reporting for adults
and adolescents. AIDS incidence rates
for minority populations, particularly
African Americans and Hispanics,
continue to increase, revealing the
continuing trend of racial minority
groups being disproportionately
impacted by the epidemic. In 1998, an
estimated 17,847 AIDS cases were
diagnosed among MSM (18.1a). This
was a decrease from 1997 and part of a
continuing trend. The decline is a result
of prevention activities and the impact
of and access to potent antiretroviral
therapies that are delaying progression
to AIDS in many HIV-infected
individuals. During the next decade,
increased emphasis will be placed on
increasing the number of people who
know their HIV serostatus. For
HIV-infected persons and those not
infected but at increased risk for HIV,
linkages with appropriate medical,
prevention, and other supportive services
increases the likelihood of maintaining
health, enhancing longevity and quality
of life, and reducing the risk of
transmitting or acquiring HIV. Thus,
learning one’s status is an important
element of prevention.
In the 21st century, strategies for
reducing HIV transmission will continue
to evolve. Future strategies will continue
to focus on the disproportionate impact
of HIV/AIDS among certain
racial/ethnic groups; prevention
strategies for populations that are
particularly high risk (for example,
IDUs and incarcerated persons);
increasing the number of persons who
learn their HIV status; and providing
access to highly active antiretroviral
therapy (HAART), thereby reducing
deaths and HIV-associated illnesses and
infections of others. The effect of
treatment on AIDS incidence and the
shift in emphasis from AIDS to HIV
require new ways of thinking about
surveillance data. With respect to AIDS
data, year-to-year changes reflect
differences in the historic pattern of
HIV infection and differences in access
to and utilization of care. Until the
estimated proportion of new HIV
diagnoses is available, the number and
characteristics of persons living with
diagnosed HIV infection and those
living with AIDS will be helpful in
planning for prevention, treatment, and
other services needed to further reduce
transmission and to improve survival
and quality of life for infected persons.
The next few years will represent a time
of transition. AIDS incidence will
continue to be affected by therapy; at
the same time, HIV reporting will
gradually be implemented by an
increasing number of States (4,5,9).
Transition to Healthy People
2010
The HIV Infection focus area was
changed for Healthy People 2010 to
reflect improvements in HIV/AIDS
treatment, surveillance, and medical
knowledge. In order to better track the
rise in incidence of AIDS cases in
particular populations in the United
States, the AIDS incidence objective in
Healthy People 2000 has been expanded
into five separate objectives in Healthy
People 2010: AIDS incidence for
adolescents and adults (13 years of age
and older), AIDS cases among MSM,
AIDS cases among persons who inject
drugs (IDUs), and AIDS cases among
men who have sex with men and who
also inject drugs. A developmental
objective, adapted from Healthy People
2000, is included in Healthy People
2010 that will monitor new cases of
HIV infection among the adolescent and
adult population. This objective is
expected to be tracked once HIV case
surveillance is implemented nationwide.
There is an objective modified from
Healthy People 2000 to monitor the
proportion of substance abuse treatment
facilities that offer HIV/AIDS education,
counseling, and support. Two new
objectives were developed to track
HIV/AIDS activities among the
incarcerated population, including
comprehensive HIV/AIDS, STD, and
TB education and voluntary HIV
counseling and testing. An additional
two objectives to track tuberculosis
among HIV-positive persons and another
to track perinatally acquired HIV
infection were added.
As new therapies continue to be
developed and as more people with
HIV/AIDS are living longer lives,
HIV-infected persons need access to
life-enhancing treatments. Therefore, an
objective regarding treatment guidelines
was added to Healthy People 2010 to
reflect the need for equal access to
appropriate care and treatment services
to maintain a healthy life. New
objectives on AIDS-related deaths, the
interval between HIV infection and
AIDS diagnosis, and the interval
between AIDS diagnosis and death from
AIDS were developed to identify
populations who are not benefitting
from current treatment therapies and to
guide where to direct resources.
There is a modification of the
Healthy People 2000 objective that
tracks the proportion of positive HIV
tests for which persons return for
counseling. This modified measure in
Healthy People 2010 will track the
number of HIV-positive persons who
know their serostatus.
The Healthy People 2000 objective
on sexual activity and abstinence in the
past 3 months among adolescents is split
into two objectives in Healthy People
2010: Focus Area 25, Sexually
Transmitted Diseases has an objective to
increase the proportion of adolescents,
grades 9–12, who abstain from sexual
intercourse or use condoms if currently
sexually active and Focus Area 13, HIV
Infection has an objective to increase
the proportion of sexually active
persons, 18–44 years of age, who use
condoms. These objectives were
included in 1 of the 10 Leading Health
Indicators for Healthy People 2010,
Responsible Sexual Behavior.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the differences between the
two decades of objectives, reflecting
new knowledge and directions in this
area.
Data Issues
In January 1993, an expanded AIDS
case (18.1) definition was implemented
256 Healthy People 2000 Final Review
for the HIV/AIDS Surveillance System
(6). The expanded definition added
pulmonary tuberculosis, recurrent
pneumonia, and invasive cervical cancer
to the list of diseases that indicate that
AIDS has fully developed among
HIV-infected people (6). In addition, the
new definition included HIV-infected
people with a CD4 cell count below 200
cells per microliter of blood, regardless
of whether those persons have
opportunistic infections, neoplasms, or
any other symptoms of HIV infection.
These changes resulted in cases being
diagnosed earlier in the course of the
disease and effected a temporary
increase in the number of cases reported
after January 1, 1993. The expanded
definition increased the number of cases
diagnosed in 1992 and 1993 because it
applied to cases diagnosed in earlier
years if they were reported after the
expanded definition was implemented in
1993. The decline in 1994 and 1995
represented the continued but waning
effect of the change in AIDS reporting
criteria. In 1995, the Centers for Disease
Control and Prevention (CDC) published
estimates of the incidence of cases of
AIDS opportunistic illnesses (including
HIV dementia and wasting syndrome)
by year of diagnosis (10). This
improved comparability for trend
purposes. In 1999, CDC published
recommendations for a nationwide
system for reporting cases of HIV
infection (5). The expansion of the
surveillance system to include all
persons who have been diagnosed with
HIV will enhance knowledge of the
scope and impact of the epidemic.
The National Household Survey on
Drug Abuse (NHSDA) provides updates
to monitor objective 18.5 on the
proportion of injecting drug users in the
past year who were enrolled in any drug
abuse treatment program in the past year
(11). For 1991 through 1993, persons
defined as IDUs in the past year were
those who used any drug with a needle
for nonmedical reasons. For 1994,
persons defined as IDUs in the past year
were those who used a needle to inject
cocaine, heroin, a stimulant, or an
anabolic steroid in the past year. For
1995, persons defined as IDUs in the
past year were those who used a needle
to inject cocaine, heroin, or a stimulant
in the past year. Enumeration of IDUs is
difficult because of the illegality of the
behavior. Therefore, the number of
IDUs may be underestimated using this
data source. In addition, the NHSDA
will miss an unknown proportion of
injecting drug users who are homeless,
institutionalized, or difficult to locate.
The NHSDA data are not comparable to
the baseline measure, which was
estimated from various sources.
Recent data on the proportion of
IDUs not in treatment who use
uncontaminated injecting equipment
(18.6) are available from the
Cooperative Agreement for AIDS
Community-Based Outreach and
Intervention Research Program from the
National Institute on Drug Abuse
(NIDA). Baseline data were from a
similar research project, the National
AIDS Demonstration Research Program,
also from NIDA. Data from both data
sources are from selected cities and are
not nationally representative. The
measure to monitor this objective is the
proportion of current injecting drug
users who did not share needles during
the last 30 days. Injecting drug users are
newly recruited study participants who
report injecting drugs during the past 30
days and whose drug-using behavior is
confirmed by observation of track marks
or positive urine tests.
Data Sources
Data for objective 18.1 on the
number of AIDS cases by year of
diagnosis are available from the CDC’s
HIV/AIDS Surveillance System and are
adjusted for both delayed and
incomplete reporting (12). Data on
AIDS cases are more often published by
year of report than by year of diagnosis.
Approximately 20 percent of AIDS cases
are reported more than a year after
diagnosis. The estimated number of
AIDS cases by year of diagnosis
changes as new data become available
because AIDS cases diagnosed in
previous years continue to be reported
and because the adjustment factor for
delays in reporting changes as new data
become available. The adjustment factor
for underreporting is based on the
assumption that 90 percent of all AIDS
cases are eventually reported (13).
Healthy People 2000 data from this
surveillance system cover the 50 States
and the District of Columbia only. The
data usually published by the AIDS
Surveillance System also include United
States dependencies, possessions, and
nations in free association with the
United States.
The baseline data on counseling to
prevent HIV and other STD’s for
objective 18.9 are from the Primary
Care Provider Surveys (PCPS). The
sample was drawn from the membership
rolls of professional organizations for
pediatricians, nurse practitioners, family
physicians, obstetricians/gynecologists,
and internists. Response rates varied
from 50 to 80 percent across these
groups. The data on counseling refer to
the proportion of providers who
routinely delivered these services to
81–100 percent of their patients who
needed the services.
The Prevention in Primary Care
Study (PPCS) was conducted in
1997–98 to update data from the PCPS.
The design and items included in the
1997–98 study were similar to the
PCPS, but a slightly different sampling
frame was used and some items
included in the 1992 surveys were not
included in the PPCS. The professionals
were sampled from listings of all
licensed, active practitioners in the
United States whose practices were at
least 50 percent primary care. Because
of low response rates from the other
provider groups, updates are available
only for nurse practitioners.
Data for objective 18.16 are from
the CDC-sponsored Worksite
Benchmark Survey, which was a
telephone survey of nongovernment
worksites. Worksites were sampled
because different worksites within the
same company could have different sets
of health promotion activities. Active
methods (for example, classes) were
counted as worksite health promotion
activities; passive methods (for example,
brochures) were not included (14,15).
Data Comparability
Beginning with data for 1996, the
incidence of AIDS cases (18.1)is
computed for the age group 13 years
and over. For previous years, the age
group 18 years and over was used. In
addition, the methodology for
determining AIDS incidence has
changed. Because of the impact of new
combination therapies, researchers can
no longer reliably predict the number of
people who will be diagnosed with
AIDS opportunistic infections each year.
There is no longer a way to determine
the length of time it takes an infected
individual on treatment to develop an
opportunistic infection because treatment
has slowed the progression of disease
for many individuals and the duration of
the effects of these drugs are not certain.
Moreover, after 1996, AIDS incidence
will no longer provide an indication of
Healthy People 2000 Final Review 257
trends in HIV transmission. Therefore,
estimates of AIDS incidence from 1986
to 1996 are presented to assess the
direction of the epidemic before the
impact of new combination therapies.
Also beginning with 1996 data, the
number of AIDS cases for MSM’s
excludes those who were also IDU’s
and the number of cases of IDU’s
excludes those who also were MSM’s.
In 1996, the number of AIDS cases
among those who were both IDU’s and
MSM’s was 2,680.
Baseline and 1995 data for ‘‘all
females’’ for objective 18.3 (adolescent
postponement of sexual intercourse), for
females 15–44 years of age and 15–19
years of age for objective 18.4 (condom
use at last sexual intercourse), and for
‘‘all females’’ for objective 18.15
(adolescent abstinence) are from the
NSFG. Baseline and 1995 data for ‘‘all
males’’ for these objectives are from the
National Survey of Adolescent Males
(NSAM). Biennial tracking data from
the Youth Risk Behavior Survey
(YRBS) are also displayed for these
objectives, but are not directly
comparable to the baselines or the
targets. The YRBS is a school-based
survey and thus does not include
teenagers who are not in school and
who are potentially at higher risk of
these behaviors (16). YRBS data, shown
by age in this report, are published by
grade only in other publications. (See
text for Priority Area 5 for more
information.)
The update for objective 18.7
comes from Lackritz et al., who
obtained data from the American Red
Cross on donations collected between
January 1992 through December 1993
(17). The more sensitive screening
enzyme immunosorbent assay antibody
test introduced in 1992 is one possible
reason for the estimated risk decreasing
remarkably between 1990 and 1992–93.
Proxy Data
Objective 18.8 targets an increase in
the proportion of HIV-infected people
who know their serostatus. A reported
posttest counseling session from a
publicly funded site is used as a
measure for knowledge of serostatus.
This objective is being measured by the
percent of positive HIV tests for which
tested people returned for counseling.
Some people who were tested and
returned for counseling may have had
more than one test during the year. CDC
believes that because of numerous
operational difficulties, posttest
counseling is under-reported by publicly
funded sites. These numbers, therefore,
are probably an underestimate, and
represent the lowest possible percent.
Data Availability
No national data are routinely
available that directly measure HIV
seroprevalence among the general
population (18.2). Estimates of the
prevalence of HIV infection in the U.S.
population as a whole are based on
mathematical models using back
calculation, a statistical method that
estimates the number of prior HIV
infections that would account for the
number of AIDS cases that have
subsequently occurred (12) as well as
serostatus data from the Survey on
Childbearing Women and from the
National Health and Nutrition
Examination Survey III (18,19).
Nationally representative estimates of
HIV seroprevalence among high-risk
groups are not available. Information on
the proportion infected among men who
have sex with men and injecting drug
users has been obtained from
seroprevalence studies conducted in
clinical settings as part of a sentinel
surveillance system conducted by CDC
in collaboration with State and local
health departments (20). The
surveillance system covers various
clinical settings in selected metropolitan
areas. Seroprevalence estimates for men
who have sex with men are based on
anonymous surveys conducted in STD
clinics. For injecting drug users,
estimates are based on surveys among
drug users entering treatment programs.
Clients attending STD clinics and drug
treatment programs are not
representative of all persons with these
high-risk behaviors.
References
1. Centers for Disease Control and Prevention.
Update: Trends in AIDS incidence, deaths, and
prevalence—United States, 1996. MMWR
46:165–93. 1997.
2. Centers for Disease Control and Prevention.
HIV/AIDS Surveillance Report, 1998; vol 10 no 2.
1998.
3. Lindegren ML, Byers RH, Thomas P, et al.
Trends in perinatal transmission of HIV/AIDS in
the United States. JAMA 282:531–8. 1999.
4. Centers for Disease Control and Prevention.
HIV/AIDS Surveillance Report, 1999; vol 11 no 2.
1999.
5. Centers for Disease Control and Prevention.
Guidelines for national human immunodeficiency
virus case surveillance, including monitoring for
human immunodeficiency virus infection and
acquired immunodeficiency syndrome. MMWR
48(RR–13). 1999.
6. Centers for Disease Control and Prevention.
1993 revised classification system for HIV
infection and expanded surveillance case definition
for AIDS among adolescents and adults. MMWR
41(RR 17):1–19. 1992.
7. Occupational exposure to bloodborne
pathogens; final rules (29 CFR 1910.1030).
Federal Register: 56:64004–182. Dec. 6, 1991.
8. Department of Health and Human Services.
Healthy people 2010. 2d ed. With understanding
and improving health and objectives for improving
health. 2 vols. Washington: U.S. Government
Printing Office. 2000.
9. Fleming PL, Whortley PM, Karon JM, et al.
Tracking the HIV epidemic: Current issues, future
challenges. AJPH 90:1037–41. 2000.
10. Centers for Disease Control and Prevention.
Surveillance for AIDS-defining opportunistic
illnesses, 1992–1997. In CDC Surveillance
Summaries, April 16, 1999. MMWR 48(SS–2).
1999.
11. Unpublished estimates from the National
Household Survey on Drug Abuse. Prevalence
Branch, Substance Abuse and Mental Health
Services Administration. 1997.
12. Centers for Disease Control and Prevention.
HIV/AIDS Surveillance Report 9(2):1–39. 1998.
13. Rosenblum L, Buehler JW, Morgan MW, et al.
The completeness of AIDS case reporting, 1988: A
multisite collaborative surveillance project. AJPH
82:1495–9. 1992.
14. Department of Health and Human Services.
Business Responds to AIDS Benchmark Survey:
Technical report. Atlanta, Georgia: Public Health
Service. Centers for Disease Control and
Prevention. 1996.
15. Centers for Disease Control and Prevention.
Cancer screening offered by worksites—United
States, 1992 and 1995. MMWR 46(19): 421–4.
1997.
16. Centers for Disease Control and Prevention.
Health risk behaviors among adolescents who do
and do not attend schools: United States, 1992.
MMWR 43:129–32. 1994.
17. Lackritz EM, Satten GA, Aberle-Grasse J, et
al. Estimated risk of transmission of the human
immunodeficiency virus by screened blood in the
United States. N Engl J Med 333(26):1721–5.
1995.
18. Karon JM, Rosenberg PS, McQuillan G, et al.
Prevalence of HIV infection in the United States,
1984–92. JAMA 276:126–31. 1996.
19. Karon JM. Methods for estimating HIV
prevalence in the United States. Washington:
Department of Health and Human Services,
National AIDS Clearinghouse. 1996.
20. Centers for Disease Control and Prevention.
National HIV serosurveillance summary, vol 3.
Results through 1992. HIV/NCID/11–93/036.
Atlanta, Georgia: Department of Health and
Human Services. 1994.
258 Healthy People 2000 Final Review

Table 18. HIV Infection objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
18.1 Slow the rise in incidence of AIDS
cases (per 100,000 population
18 years and over) ............... 1989 17.0 20.9 25.2 33.1 32.8 29.8 28.6
1
27.8
1
23.1
1
19.5 - - - 43
a. Men who have sex with men
(number of cases) ............. 1989 27,000 28,574 34,005 42,706 39,326 34,146 30,696
1,2
23,420
1,2
21,341
1,2
17,847 - - - 48,000
b. Black (non-Hispanic)............ 1989 44.4 59.0 73.0 100.8 107.8 102.9 100.5
1
110.9
1
98.1
1
82.9 - - - 136
c. Hispanic ..................... 1989 34.9 33.1 39.9 51.5 53.7 49.4 47.1
1
48.4
1
40.5
1
33.0 - - - 76
d. Female...................... 1989 3.5 5.3 6.9 9.9 11.1 10.9 11.2
1
11.6
1
10.4
1
8.8 - - - 13
e. Injecting drug users (number of
cases) ...................... 1989 10,300 12,466 15,696 21,899 23,399 20,734 19,100
1,3
15,583
1,3
15,697
1,3
12,099 - - - 25,000
18.2 Slow the rise in prevalence of HIV
infection (per 100,000 population
13 years and over) ............... 1989 400 400 - - - 310–420 - - - - - - - - - - - - - - - - - - - - - 400
a. Men who have sex with men
(15 years and over)
4
........... 1989
§
15,000–
61,800
17,400–
60,900 - - -
5
3,900–
47,400 - - - - - - - - - - - - - - - - - - - - - 20,000
b. Injecting drug users (15 years and
over)
6
...................... 1989
§
0–48,200 0–49,300 - - -
5
600–
52,900 - - - - - - - - - - - - - - - - - - - - - 40,000
c. Females giving birth to live-born
infants (15–44 years) ........... 1989 160 160 170 170 160 160 - - - - - - - - - - - - - - - 100
18.3* Adolescents who ever engaged in
sexual intercourse
Adolescents 15 years
All females .................... 1988 27% - - - - - - - - - - - - - - - 22% - - - - - - - - - - - - 15%
In-school females.............. ... --- 35% 36% --- 37% --- 38% --- 44% --- 43% ...
All males ...................... 1988 33% - - - - - - - - - - - - - - - 27% - - - - - - - - - - - - 15%
In-school males ............... ... --- 48% 44% --- 45% --- 42% --- 42% --- 34% ...
a. All black males ................ 1988 69% - - - - - - - - - - - - - - - 58% - - - - - - - - - - - - 15%
In-school non-Hispanic black
males...................... ... --- --- 79% --- 82% --- 77% --- 75% --- 68% ...
Adolescents 17 years
All females .................... 1988 50% - - - - - - - - - - - - - - - 51% - - - - - - - - - - - - 40%
In-school females.............. ... --- 62% 66% --- 66% --- 67% --- 62% --- 40% ...
All males ...................... 1988 66% - - - - - - - - - - - - - - - 53% - - - - - - - - - - - - 40%
In-school males ............... ... --- 73% 68% --- 68% --- 65% --- 60% --- 44% ...
b. All black males ................ 1988 90% - - - - - - - - - - - - - - - 79% - - - - - - - - - - - - 40%
In-school non-Hispanic black
males...................... ... --- --- 90% --- 92% --- 88% --- 85% --- 82% ...
c. All black females 15–17 years..... 1988 66% - - - - - - - - - - - - - - -
7
48% --- --- --- --- 40%
In-school non-Hispanic black
females .................... ... --- --- 84% --- 80% --- 75% --- 73% --- 73% ...
18.4* Condom use at last sexual
intercourse
Sexually active unmarried females
15–44 years (by their partners) ...... 1988 19% - - - - - - - - - - - - - - - 25% - - - - - - - - - - - - 50%
See footnotes and key at end of table.
Healthy People 2000 Final Review 259

Table 18. HIV Infection objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
a. Sexually active females
15–19 years (by their partners) . . . 1988 26% - - - - - - - - - - - - - - - 37% - - - - - - - - - - - - 60%
Sexually active females 15–19
years in grades 9–12 (by their
partners) .................. ... --- 40% 38% --- 46% --- 49% --- 51% --- 51% ...
b. Sexually active males 15–19 years . 1988 57% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 75%
Sexually active males 15–19
years in grades 9–12 ........ ... --- 49% 54% --- 59% --- 61% --- 63% --- 66% ...
c. Injecting drug users ............ 1992
8
34% ... ... ... --- --- --- --- --- --- --- 75%
d. Black female 15–44 years (by their
partners) .................... 1988 12.4% - - - - - - - - - - - - - - -
7
25% --- --- --- --- 75%
18.5 Injecting drug users enrolled in
treatment ...................... 1989 11% - - - 28.7% 29.8% 45.9% 47.8% 34.1% - - - - - - - - - - - - 50%
18.6 Injecting drug users not in treatment
who did not share needles (in
previous 30 days) ................ 1991 30.8% . . . . . . - - -
9
57.7%
10
60.6%
11
60.0% - - - - - - - - - - - - 75%
18.7 Risk of transfusion-transmitted
HIV infection (per units of
blood) ......................... 1989
1 per
40,000–
150,000
1 per
221,000 - - - - - -
12
1 per
450,000–
660,000 - - - - - - - - -
1 per
625,000 - - - - - -
1 per
250,000
18.8 HIV-infected people who know their
serostatus
Percent of positive HIV tests for which
people returned for counseling ...... 1990 72.5% . . . - - - - - - - - - - - - 83% 74% - - - 63% - - - 80%
18.9* Clinician counseling to prevent HIV
and other sexually transmitted
diseases ...................... 1987
13
10% --- --- --- --- --- --- --- --- --- --- 75%
Percent of clinicians routinely providing
service to 81–100% of patients
a. Providers practicing in high
incidence areas ............... ... --- --- --- --- --- --- --- --- --- --- --- 90%
b. Family physicians .............. 1992 27% . . . . . . . . . - - - - - - - - - - - - - - -
14,15
--- --- 75%
c. Internists..................... 1992 30% . . . . . . . . . - - - - - - - - - - - - - - -
14,15
--- --- 75%
d. Nurse practitioners ............. 1992 50% . . . . . . . . . - - - - - - - - - - - - - - -
14
45% - - - 75%
e. Obstetricians/gynecologists....... 1992 46% . . . . . . . . . - - - - - - - - - - - - - - -
14,15
--- --- 75%
f. Pediatricians .................. 1992 46% . . . . . . . . . - - - - - - - - - - - - - - -
14,15
--- --- 75%
g. Mental health care providers...... ... --- --- --- --- --- --- --- --- --- --- --- 75%
18.10* HIV and other STD education
curricula
Schools offering at least one STD
class.......................... 1988 95% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 95%
Proportion of middle and senior high
schools:
With HIV prevention in required
courses ...................... ... --- --- --- --- --- 86% --- --- --- --- --- ...
See footnotes and key at end of table.
260 Healthy People 2000 Final Review

Table 18. HIV Infection objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
With STD prevention in required
courses ...................... ... --- --- --- --- --- 84% --- --- --- --- --- ...
18.11* HIV and STD education for students
at colleges and universities
Students 18 years and over given:
AIDS or HIV infection prevention
information ................... 1995 49.1% . . . . . . . . . . . . . . . . . . - - - - - - - - - - - - 90%
STD prevention information ........ 1995 43.4% . . . . . . . . . . . . . . . . . . - - - - - - - - - - - - 90%
Students 18 years and over taught
about AIDS or HIV in a college class. . 1995 41.4% . . . . . . . . . . . . . . . . . . - - - - - - - - - - - - 90%
18.12 Outreach HIV programs for drug
users (cities with populations greater
than 100,000) ................... 1991 35% . . . . . . 32% - - - - - - - - - - - - - - - - - - - - - 90%
18.13 Clinic services for HIV and other
sexually transmitted diseases ..... ... --- --- --- --- --- --- --- --- --- --- --- 50%
Family planning clinics ............. 1989 40% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Title X funded family planning clinics
STD testing (excluding HIV)........ ... --- --- --- --- --- 95% --- --- --- --- --- ...
STD counseling (excluding HIV) .... ... --- --- --- --- --- 98% --- --- --- --- --- ...
STD treatment (excluding HIV) ..... ... --- --- --- --- --- 93% --- --- --- --- --- ...
Gonorrhea
Client testing
16
................. ... --- 97% --- --- --- --- --- --- --- --- --- ...
Client treatment................. ... --- 82% --- --- --- --- --- --- --- --- --- ...
Partner notification
17
............. ... --- 23% --- --- --- --- --- --- --- --- --- ...
Partner testing.................. ... --- 60% --- --- --- --- --- --- --- --- --- ...
Partner treatment ............... ... --- 62% --- --- --- --- --- --- --- --- --- ...
Syphilis
Client testing
16
................. ... --- 86% --- --- --- --- --- --- --- --- --- ...
Client treatment................. ... --- 48% --- --- --- --- --- --- --- --- --- ...
Partner notification
17
............. ... --- 29% --- --- --- --- --- --- --- --- --- ...
Partner testing.................. ... --- 57% --- --- --- --- --- --- --- --- --- ...
Partner treatment ............... ... --- 40% --- --- --- --- --- --- --- --- --- ...
Chlamydia
Client testing
16
................. ... --- 66% --- --- --- --- --- --- --- --- --- ...
Client treatment................. ... --- 73% --- --- --- --- --- --- --- --- --- ...
Partner notification
17
............. ... --- 15% --- --- --- --- --- --- --- --- --- ...
Partner testing.................. ... --- 29% --- --- --- --- --- --- --- --- --- ...
Partner treatment ............... ... --- 50% --- --- --- --- --- --- --- --- --- ...
HIV
Client pretest counseling .......... ... --- 66% --- --- --- 82% --- --- --- --- --- ...
Client testing ................... ... --- 60% --- --- --- 74% --- --- --- --- --- ...
See footnotes and key at end of table.
Healthy People 2000 Final Review 261

Table 18. HIV Infection objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
18.14 Regulations to protect workers from
occupational exposure to
bloodborne infections, including
HIV
Proportion of work places ........... 1992 100% . . . . . . . . . 100% 100% 100% 100% 100% 100% 100% 100%
18.15* Adolescent abstinence from sexual
intercourse for previous 3 months
All sexually active females
15–17 years .................... 1988 23.6% - - - - - - - - - - - - - - - 27% - - - - - - - - - - - - 40%
In-school sexually active females
15–17 years .................... ... --- 24% 25% --- 25% --- 23% --- 23% --- 25% ...
All sexually active males 15–17 years . . 1988 33% - - - - - - - - - - - - - - - 37% - - - - - - - - - - - - 40%
In-school sexually active males 15–17
years ......................... ... --- 30% 36% --- 33% --- 34% --- 32% --- 32% ...
18.16 Comprehensive HIV/AIDS workplace
programs
Proportion of businesses with policies,
management training, and employee
education:
Small businesses (15–49
employees) ................... 1995 2% . . . . . . . . . . . . . . . . . . - - - - - - - - - - - - 10%
Medium businesses (50–749
employees) ................... ... --- --- --- --- --- --- 7% --- --- --- --- ...
Large businesses (750 or more
employees) ................... 1995 25% . . . . . . . . . . . . . . . . . . - - - - - - - - - - - - 50%
Proportion of businesses with policies:
Small businesses (15–49
employees) ................... ... --- --- --- --- --- --- 18% --- --- --- --- ...
Medium businesses (50–749
employees) ................... ... --- --- --- --- --- --- 42% --- --- --- --- ...
Large businesses (750 or more
employees) ................... ... --- --- --- --- --- --- 79% --- --- --- --- ...
Proportion of businesses with
management training:
Small businesses (15–49
employees) ................... ... --- --- --- --- --- --- 18% --- --- --- --- ...
Medium businesses (50–749
employees) ................... ... --- --- --- --- --- --- 41% --- --- --- --- ...
Large businesses (750 or more
employees) ................... ... --- --- --- --- --- --- 77% --- --- --- --- ...
Proportion of businesses with employee
education:
Small businesses (15–49
employees) ................... ... --- --- --- --- --- --- 6% --- --- --- --- ...
See footnotes and key at end of table.
262 Healthy People 2000 Final Review

Table 18. HIV Infection objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Medium businesses (50–749
employees) ................... ... --- --- --- --- --- --- 16% --- --- --- --- ...
Large businesses (750 or more
employees) ................... ... --- --- --- --- --- --- 32% --- --- --- --- ...
Federal government departments and
agencies ....................... 1995 80% . . . . . . . . . . . . . . . . . . - - - - - - - - - - - - 100%
18.17 Linkages between substance abuse
treatment programs and primary
care clinics
Federally funded primary care clinics . . . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 40%
Federally funded substance abuse
treatment programs............... ... --- --- --- --- --- --- --- --- --- --- --- 40%
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
1
Beginning with 1996, data are for people 13 years and over and methodology is changed. See text for Priority Area 18.
2
Beginning with 1996 data, excludes men who are also injecting drug users. See text for Priority Area 18.
3
Beginning with 1996 data, excludes men who also had sex with men. See text for Priority Area 18.
4
Range of clinic-specific HIV prevalence rates among men who have sex with men attending sexually transmitted disease clinics participating in CDC’s National Serosurveillance Program.
5
1991–92 data.
6
Range of clinic-specific HIV prevalence rates among injecting drug users attending sexually transmitted disease clinics participating in CDC’s National Serosurveillance Program.
7
Data are for non-Hispanic black females.
8
Data are for male and female injecting drug users (married and unmarried) who report having vaginal sex within the last 6 months and reported using a condom ‘‘always’’ or ‘‘sometimes.’’
9
Data are for January 1992 through April 1993.
10
Data are for May 1993 through December 1995.
11
Data are for January 1992 through July 1996.
12
1992–93 data.
13
Data are for new patients only.
14
1997–98 data.
15
Response rate for this group was too low to produce reliable estimates.
16
Includes testing at initial visit, at annual visit, or if symptomatic.
17
By family planning clinic staff via telephone or mail.
NOTES: Data include revisions and, therefore, may differ from data previously published in these reports and other publications. AIDS is acquired immunodeficiency syndrome. HIV is human
immunodeficiency virus. STD is sexually transmitted disease.
Objective number Data source
18.1,18.1a–e HIV/AIDS Surveillance System, CDC, NCHSTP.
18.2, 18.2a–c Baseline and 1990 update: CDC, NCHSTP.
1992 update for total population: Karon JM, et. al. Prevalence of HIV infection in the United States, 1984 to 1992. JAMA 276(2):126–31. 1996.
18.3* Baseline and updates for all females and all black females: National Survey of Family Growth, CDC, NCHS.
Baseline and updates for all males and all black males: National Survey of Adolescent Males, NIH, NICHD.
1990–99 data for in-school females and males: Youth Risk Behavior Survey, CDC, NCCDPHP.
18.4*, 18.4d National Survey of Family Growth, CDC, NCHS.
18.4a Baseline: National Survey of Family Growth, CDC, NCHS.
1990–99 data for in-school females and males: Youth Risk Behavior Survey, CDC, NCCDPHP.
18.4b Baseline: National Survey of Adolescent Males, NIH, NICHD.
Met
Toward
Mixed/ no change
Away
Cannot assess
Healthy People 2000 Final Review 263

Objective number Data source
1990–99 data for in-school females and males: Youth Risk Behavior Survey, CDC, NCCDPHP.
18.4c National AIDS Demonstration Research Program, NIH, NIDA.
18.5 Baseline: NIH, NIDA.
Updates: National Household Survey on Drug Abuse, SAMHSA, OAS.
18.6 Baseline: National AIDS Demonstration Research Program, NIH, NIDA.
Updates: Cooperative Agreement for AIDS Community-based Outreach/Intervention Research Program, NIH, NIDA.
18.7 Baseline: American Association of Blood Banks.
1990 update: Comprehensive Blood Donations Data Set, CDC, NCHSTP.
1992–93 update: Lackritz EM, et al. Estimated risk of transmission of the human immunodeficiency virus by screened blood in the United States. NEJM
333(26):1721–5. 1995.
1997 update: Busch MP and Kleinman SH. Nucleic acid amplification testing of blood donors for transfusion-transmitted infectious diseases. Transfusion
40:143–59. 2000.
18.8 HIV Counseling and Testing Data Sites System, CDC, NCHSTP.
18.9* 1987 baseline: Sexual history-taking and counseling practices of primary care physicians, Lewis CE and Freeman HE. Western Journal of Medicine 147:
165–7. 1987.
1992 baseline: Primary Care Provider Surveys, OPHS, ODPHP.
Update: Prevention in Primary Care Study, American College of Preventive Medicine.
18.10* Baseline: AIDS education: Public school programs require more student information and teacher training. GAO, 1990.
1994 data: School Health Policies and Programs Study, CDC, NCCDPHP.
18.11 National College Health Risk Behavior Survey, CDC, NCCDPHP.
18.12 CDC, NCHSTP.
18.13* Baseline: State Family Planning Directors.
1990 data: National Questionnaire on Provision of STD and HIV Services by Family Planning Clinics, OPA.
1994 data: The Urban Institute. Family planning clinics: Current status and recent changes in services, clients, staffing and income sources. March 1994.
18.14 Occupational exposure to bloodborne pathogens; final rule (29 CFR 1910, 1030). Federal Register 56:64004–182. December 6, 1991.
18.15* Baseline and update for all females: National Survey of Family Growth, CDC, NCHS.
Baseline and update for all males: National Survey of Adolescent Males, NIH, NICHD.
1990–99 data for in-school females and males: Youth Risk Behavior Survey, CDC, NCCDPHP.
18.16 Businesses: Business Responds to AIDS Benchmark Survey, CDC, NCHSTP.
Federal government: CDC.
* Duplicate objective. See full text of objective following this table.
264 Healthy People 2000 Final Review
HIV Infection
Objectives
18.1: Confine annual incidence of
diagnosed AIDS cases to no more than
43 per 100,000 population.
18.1a: Confine annual incidence of
diagnosed AIDS cases among men
who have sex with men to no more
than 48,000 cases.
18.1b: Confine annual incidence of
diagnosed AIDS cases among
blacks to no more than 136 per
100,000 population.
18.1c: Confine annual incidence of
diagnosed AIDS cases among
Hispanics to no more than 76 per
100,000 population.
18.1d: Confine annual incidence of
diagnosed AIDS cases among
women to no more than 13 per
100,000 population.
18.1e: Confine annual incidence of
diagnosed AIDS cases among
injecting drug users to no more than
25,000.
18.2: Confine the prevalence of HIV
infection to no more than 400 per
100,000 people.
18.2a: Confine the prevalence of
HIV infection among men who
have sex with men to no more than
20,000 per 100,000.
18.2b: Confine the prevalence of
HIV infection among injecting drug
users to no more than 40,000 per
100,000.
18.2c: Confine the prevalence of
HIV infection among women giving
birth to live-born infants to no more
than 100 per 100,000.
18.3*: Reduce the proportion of
adolescents who have engaged in sexual
intercourse to no more than 15 percent
by age 15 and no more than 40 percent
by age 17.
Duplicate objectives: 5.4 and 19.9
18.3a*: Reduce the proportion of
black males aged 15 years who
have engaged in sexual intercourse
to no more than 15 percent.
Duplicate objectives: 5.4a and 19.9a
18.3b*: Reduce the proportion of
black males aged 17 years who
have engaged in sexual intercourse
to no more than 40 percent.
Duplicate objectives: 5.4b and 19.9b
18.3c*: Reduce the proportion of
black females aged 17 years who
have engaged in sexual intercourse
to no more than 40 percent.
Duplicate objectives: 5.4c and 19.9c
18.4*: Increase to at least 50 percent the
proportion of sexually active, unmarried
people who used a condom at last
sexual intercourse.
Duplicate objective: 19.10
18.4a*: Increase to at least
60 percent the proportion of
sexually active, unmarried young
women aged 15–19 whose partners
used a condom at last sexual
intercourse.
Duplicate objective: 19.10a
18.4b*: Increase to at least
75 percent the proportion of
sexually active, unmarried young
men aged 15–19 who used a
condom at last sexual intercourse.
Duplicate objective: 19.10b
18.4c*: Increase to at least
75 percent the proportion of
injecting drug users who used a
condom at last sexual intercourse.
Duplicate objective: 19.10c
18.4d*: Increase to at least
75 percent the proportion of black
women aged 15–44 whose partners
used a condom at last sexual
intercourse.
Duplicate objective: 19.10d
18.5: Increase to at least 50 percent the
estimated proportion of all injecting
drug users who are in drug abuse
treatment programs.
18.6: Increase to at least 75 percent the
estimated proportion of active injecting
drug users who use only new or
properly decontaminated syringes,
needles, and other drug paraphernalia
(‘‘works’’).
18.7: Reduce to no more than 1 per
250,000 units of blood and blood
components the risk of transfusion-
transmitted HIV infection.
18.8: Increase to at least 80 percent the
proportion of HIV-infected people who
know their serostatus.
18.9*: Increase to at least 75 percent the
proportion of primary care and mental
health care providers who provide
appropriate counseling on the prevention
of HIV and other sexually transmitted
diseases.
Duplicate objective: 19.14
18.9a*: Increase to at least
90 percent the proportion of primary
care and mental health care
providers who practice in areas of
high AIDS and sexually transmitted
disease incidence, who provide
appropriate counseling on the
prevention of HIV and other
sexually transmitted diseases.
Duplicate objective: 19.14a
18.9b*: Increase to at least
75 percent the proportion of family
physicians who provide appropriate
counseling on the prevention of
HIV and other sexually transmitted
diseases.
Duplicate objective: 19.14b
18.9c*: Increase to at least
75 percent the proportion of
internists who provide appropriate
counseling on the prevention of
HIV and other sexually transmitted
diseases.
Duplicate objective: 19.14c
18.9d*: Increase to at least
75 percent the proportion of nurse
practitioners who provide
appropriate counseling on the
prevention of HIV and other
sexually transmitted diseases.
Duplicate objective: 19.14d
18.9e*: Increase to at least
75 percent the proportion of
obstetricians/gynecologists who
provide appropriate counseling on
the prevention of HIV and other
sexually transmitted diseases.
Duplicate objective: 19.14e
18.9f*: Increase to at least
75 percent the proportion of
pediatricians who provide
appropriate counseling on the
prevention of HIV and other
sexually transmitted diseases.
Duplicate objective: 19.14f
Healthy People 2000 Final Review 265
18.9g*: Increase to at least
75 percent the proportion of mental
health care providers who provide
appropriate counseling on the
prevention of HIV and other
sexually transmitted diseases.
Duplicate objective: 19.14g
18.10*: Increase to at least 95 percent
the proportion of schools that have
appropriate HIV and other STD
education curricula for students in
4th–12th grade, preferably as part of
comprehensive school health education,
based upon scientific information that
includes the way HIV and other STDs
are prevented and transmitted.
Duplicate objective: 19.12
18.11*: Increase to at least 90 percent
the proportion of students who received
HIV and other STD information,
education, or counseling on their college
or university campus.
Duplicate objective: 19.17
18.12: Increase to at least 90 percent the
proportion of cities with populations
over 100,000 that have outreach
programs to contact drug users
(particularly injecting drug users) to
deliver HIV-risk-reduction messages.
18.13*: Increase to at least 50 percent
the proportion of family planning
clinics, maternal and child health clinics,
sexually transmitted disease clinics,
tuberculosis clinics, drug treatment
centers, and primary care clinics that
provide on site primary prevention and
provide or refer for secondary
prevention services for HIV infection
and bacterial sexually transmitted
diseases (gonorrhea, syphilis, and
chlamydia) to high-risk individuals and
their sex or needle-sharing partners.
Duplicate objectives: 5.11 and 19.11
18.14: Extend to all facilities where
workers are at risk for occupational
transmission of HIV regulations to
protect workers from exposure to blood
borne infections, including HIV
infection.
18.15*: Increase to at least 40 percent
the proportion of ever sexually active
adolescents aged 17 and younger who
have not had sexual intercourse for the
previous 3 months.
Duplicate objectives: 5.5 and 19.16
18.16: Increase to at least 50 percent the
proportion of large businesses and to
10 percent the proportion of small
businesses that implemented a
comprehensive HIV/AIDS workplace
program. Increase to 100 percent the
proportion of Federal Government
departments and agencies that
implemented a comprehensive
HIV/AIDS workplace program.
18.17: Increase to at least 40 percent the
number of federally funded primary care
clinics that have formal established
linkages with substance abuse treatment
programs and increase to at least
40 percent the number of federally
funded substance abuse treatment
programs that have formal established
linkages with primary care clinics.
*Duplicate objective.
266 Healthy People 2000 Final Review

Priority Area 19
Sexually Transmitted
Diseases
Background
Sexually transmitted diseases
(STDs) refer to the more than 25
infectious organisms transmitted
primarily through sexual activity. STD
rates have declined substantially in the
United States since 1936 when programs
for their prevention and control were
established through collaborative efforts
of Federal, State, and local health
authorities. Throughout the 1990s,
considerable progress was realized as
programs continued and were expanded
(1). Nevertheless, STDs remain at
epidemic levels in this country, and the
United States continues to record the
highest STD rates in the industrialized
world (2). STDs disproportionately
affect women, infants, adolescents, and
communities of color (1). Syphilis
remains one of the most glaring
examples of racial disparities in health;
1999 rates among African Americans
were 30 times those among white
Americans (3). Gonorrhea and
chlamydia rates are highest among
adolescents and young adults. More than
65 million Americans are currently
living with an incurable viral STD such
as genital herpes or human
papillomavirus infection (HPV) (4).
Overall, an estimated 15 million new
cases of non-HIV STDs, such as
syphilis, chlamydia, gonorrhea, herpes,
hepatitis B, and HPV infection, occur at
an annual direct and indirect cost of at
least $10 billion. Approximately
25 percent of new STD cases occur
among adolescents (2). These infections
frequently cause severe, life-long
consequences, especially among women,
adolescents, and infants. The
consequences of STDs include
involuntary infertility, potentially fatal
tubal pregnancy, other adverse
pregnancy outcomes such as stillbirths
and newborn (congenital) infections,
increased risk of HIV transmission, and
cervical and liver cancers (1).
STD transmission is sustained by
the complex interaction of biological,
social, and behavioral factors (1). For
example, STDs are frequently
asymptomatic or can be so mild as to be
unrecognized by infected persons. This
factor helps sustain transmission to
others. Frequently there is a very long
lag time between infection and
complications such as cervical cancer
(from HPV infection) or infertility (from
chlamydia or gonorrhea). Often people
do not recognize the connection between
an earlier STD and a resulting health
problem. Women are at higher risk than
men for most STDs and often suffer
more severe consequences (2). STDs
disproportionately affect disenfranchised
persons, especially those who are in
social networks where high-risk sexual
behaviors are common or access to
high-quality health care is limited (5).
Access to health care is essential for
early detection, treatment, and
behavior-change counseling for STDs.
Groups with the highest rates of STDs
are often the same groups in which
access to health services is most limited.
Complicating matters is the continued
stigma associated with STDs and
Americans’ general discomfort with
openly discussing intimate aspects of
life, especially those related to sex. The
latter two factors greatly impede efforts
to change sexual behaviors on the part
of parents, educators, and health care
providers.
Because of the challenging
dynamics of STD transmission, STD
prevention efforts must use diverse,
multifactorial interventions (1).
Behavioral interventions are essential.
They can help people abstain from
sexual intercourse, delay initiation of
intercourse, reduce the number of sex
partners, and increase the use of
effective physical barriers such as
condoms. Biological interventions are
equally important. They include
screening, diagnosis, treatment, and
early antimicrobial prophylaxis of sex
partners.
Data Summary
Highlights
During the 1990s, notable progress
was made in reducing the incidence and
complications associated with common
bacterial STDs. From 1985 to 1996,
rates of gonorrhea (19.1) decreased
nearly 10 percent annually. Rates
stabilized between 1996 and 1997, and,
between 1997 and 1999, gonorrhea rates
increased by 9 percent. The 1999
gonorrhea rate of 133 per 100,000
population approached, but did not
meet, the Healthy People 2000 target of
100. Gonorrhea rates remain high
among adolescents, young adults, and
African Americans.
In the 1990s, chlamydia (19.2)
emerged as the most commonly reported
notifiable infectious disease in the
United States (6). An estimated 3
million people contract chlamydia each
year, a decline from more than 4 million
cases per year in the early 1980s (4).
The Healthy People 2000 objective of
less than 5 percent chlamydia positivity
in women tested in family planning
clinics was achieved for women 20–24
years of age (4.5 percent positivity in
1999). Positivity declined in women
15–19 years of age, from 12.2 percent in
1988 to 6.6 percent in 1999.
Untreated gonorrhea and chlamydia
infection in women can lead to pelvic
inflammatory disease (PID). As
gonorrhea and chlamydia morbidity
declined through the 1990s, so did the
incidence of PID. There were steady
declines in PID hospitalizations and PID
visits to physicians (19.6). PID
hospitalizations decreased from a
baseline of 311 per 100,000 females
15–44 years of age to a rate of 155 in
1998. Visits to physicians for PID
decreased from 431,000 visits per year
in 1988 to 251,000 in 1999. This
exceeded the Healthy People 2000
target of 290,000.
The 1990s began with the United
States experiencing one of the highest
rates of primary and secondary (P&S)
syphilis (19.3) (20.3 per 100,000
population) since the 1940s. Remarkable
progress occurred during the past
decade. The 1999 P&S syphilis rate was
the lowest since reporting began in
1941. The P&S syphilis rate declined by
88 percent since 1990 to a 1999 rate of
2.5 cases per 100,000 for the general
population. The Healthy People 2000
target of less than 4 per 100,000 was
achieved. The Healthy People 2000
target for African Americans of less than
30 per 100,000 was also achieved with a
reported rate of 15 per 100,000 in 1999.
Congenital syphilis (19.4) occurs when
infants acquire syphilis from their
mothers during pregnancy. The
congenital syphilis rate in the United
States peaked in 1991 at 107 cases per
100,000 live births and declined by
87 percent to 14 per 100,000 in 1999.
The Healthy People 2000 objectives for
congenital syphilis were achieved for
the general population (14.3 per 100,000
live births in 1999 versus Healthy
People 2000 target of 40), African
Healthy People 2000 Final Review 267

19.1 Gonorrhea
19.2 Chlamydia: Female 15-19 years
Female 20-24 years
19.3 Primary and secondary syphilis
19.4 Congenital syphilis
19.5 Annual number: Genital herpes
Genital warts
19.6 Pelvic inflammatory disease, female 15-44 years:
Hospitalizations
Initial visits to physicians
19.7 Sexually transmitted Hepatitis B (number of cases)
19.8 Repeat gonorrhea infection
19.9 Adolescents who ever had sexual intercourse:
Female 15 years
Male 15 years
19.10 Condom use at last intercourse:
Female 15-44 years
Male 15-17 years
19.16 Adolescent abstinence from sexual intercourse
in past 3 months: Female 15-17 years
Female 17 years
Male 17 years
Moved away from target Moved toward target Met target
NOTE: Complete tracking data are shown in table 19. Progress quotients are not calculated for objectives 19.11, 19.12, 19.13, 19.14, 19.15,
and 19.17. See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
78%
-10%
50%
33%
42%
56%
19%
57%
Percent of target achieved
-100 -80 -60 -40 -20 0 20 40 60 80 100
75%
21%
150%
111%
114%
115%
128%
135%
-249%
84%
268 Healthy People 2000 Final Review
Figure 19. Final status on Sexually Transmitted Diseases objectives
Americans (57.9 in 1999 versus 175),
and Hispanics (20.4 in 1999 versus 50).
Fewer first-time health care
consultations for genital warts (19.5)
(240,000 consultations) were seen in
1999 compared with 1988 (290,000).
The Healthy People 2000 target of
fewer than 246,500 genital warts
consultations was achieved. The
opposite trend occurred for genital
herpes (19.5), with 163,000
consultations recorded in 1988 and
224,000 recorded in 1999. The number
of cases of sexually transmitted hepatitis
B infection (19.7) declined, with
reported cases decreasing from 48,000
in 1987 to 25,000 in 1999, thus
surpassing the year 2000 target of fewer
than 30,500 cases.
Some progress was made in
reducing the proportion of adolescents
engaging in sexual intercourse and for
increasing condom use at last
intercourse (19.10b). All groups of
in-school male and female teenagers
(except 15-year-old females) reported a
lower proportion of having engaged in
sexual intercourse in 1999 compared
with 1990. Condom use at last
intercourse by partners of sexually
active in-school females 15–19 years of
age increased from 40 percent in 1990
to 51 percent in 1999. Sexually active
in-school males 15–19 years of age
increased condom use at last intercourse
from 49 percent in 1990 to 66 percent in
1999.
Summary of Progress
Data to assess trends are available
for 14 of 17 objectives in this priority
area. Three objectives (19.3, 19.4, and
19.7) have met or exceeded the year
2000 targets, including all of the special
population targets. Seven objectives
(19.1, 19.2, 19.6, 19.8, 19.10, 19.11, and
19.16) moved toward their targets. One
objective (19.12) was met at baseline;
however, supplemental data for the
objective indicate a trend that could be
moving in the wrong direction. Progress
was mixed for objectives 19.5 and 19.9.
Update data were obtained for objective
19.14 for nurse practitioners only and
indicated that the objective is moving
away from its target. The status for
three objectives (19.13, 19.15, and
19.17) could not be assessed because
data subsequent to the baseline measures
are unavailable. See table 19 for the
tracking data for the objectives in this
priority area and figure 19 for a
quantitative assessment of progress.
Discussion
The STD achievements of the 1990s
demonstrate that STD prevention
programs are highly effective. When
programs are expanded to meet demand,
new technologies can be quickly
adopted, and program efforts are
consistently combined with policies to
expand clinical and community
partnerships for prevention.
Chlamydia declined throughout
much of the 1990s, mainly because of
increased efforts to screen and treat
women. However, from 1997 to 1999,
chlamydia test positivity in family
planning clinics actually increased in 8
out of 10 regions. These reported
increases are most likely due to changes
in newly available and better laboratory
tests and expanded screening programs
to populations with higher levels of
disease. Efforts to create a dedicated
infertility prevention program focusing
on chlamydia and gonorrhea began in
1994 after the success of pilot programs
involving family planning clinics and
public health laboratories and wide
availability of new, cost-effective
diagnostic technologies. Since then,
chlamydia screening has become
increasingly available in publicly funded
family planning and STD clinics. From
1988 to 1999, the Pacific Northwest
experienced a 62 percent decline in
infection among women tested for
chlamydia in family planning clinics (8).
The percentage of women testing
positive for chlamydia—chlamydia
positivity—in family planning clinics by
State provides a good indication of
where the disease remains most
widespread. The highest level of
infection tends to be in areas where
screening and treatment have not been
widely implemented. The greatest
declines generally have been in areas
with the most effective and prolonged
screening programs. Future challenges
will include expanded screening in
women to meet regional demand,
expanded screening programs for men,
and expanded venues in which high-risk
persons can be screened.
In 1999, 79 percent of U.S. counties
reported no P&S syphilis; 25 counties
were responsible for 50 percent of new
cases (3). The unprecedented low rate of
syphilis overall, combined with the
well-documented geographic
concentration of new cases, has created
a unique opportunity to eliminate
syphilis in the United States. In October
1999, the Public Health Service
launched the National Plan to Eliminate
Syphilis from the United States (8). The
elimination effort will expand and
enhance strategies that worked in the
1990s to reduce syphilis to record low
levels: strengthening community
involvement and partnerships, enhancing
syphilis surveillance, conducting rapid
outbreak investigations, expanding
clinical and laboratory services, and
enhancing health promotion.
Landmark studies and improved
testing technology in the 1990s greatly
increased knowledge of the incidence
and prevalence of genital herpes and
HPV. These viral STDs affect far more
Americans than were documented by
Healthy People 2000 baselines in 1988
(163,000 first-time consultations for
genital herpes and 290,000 first-time
consultations for genital warts). An
estimated 45 million Americans—more
than one in five adults—are infected
with genital herpes (9). Herpes
prevalence increased by 30 percent from
the late 1970s to the early 1990s. An
estimated 1 million Americans acquire
genital herpes infections annually (4).
Preliminary data suggest that the
number of persons infected with genital
herpes has remained relatively stable
during the 1990s (10). Recent data
suggest that HPV is much more
common than originally believed and is
likely the most common STD among
young, sexually active persons. Concern
about HPV has increased in recent years
after studies showed that some types of
HPV infection cause cervical cancer. An
estimated 5.5 million Americans become
infected with HPV each year (4). An
estimated 20 million Americans are
currently infected. Research indicates
that about 1 percent of sexually active
adults in the United States have genital
warts caused by HPV (11).
Transition to Healthy People
2010
Healthy People 2010 contains 19
objectives in the Sexually Transmitted
Diseases focus area. Overall, Healthy
People 2010 health status objectives are
similar to Healthy People 2000
objectives. There is expanded emphasis
on reducing STD complications
affecting women (including infertility
and heterosexually transmitted HIV
infection) and reducing neonatal
consequences of maternal STD infection
Healthy People 2000 Final Review 269
(including chlamydia pneumonia,
gonococcal and chlamydial eye
infections, laryngeal papillomatosis,
neonatal herpes, and preterm birth and
low birthweight associated with bacterial
vaginosis). Behavioral objectives include
those for increased responsible
adolescent sexual behavior (abstinence
or condom use) and for responsible
sexual behavior messages on television.
Because expanded services and venues
for STD prevention proved successful in
the 1990s, Healthy People 2010
objectives call for administration of
hepatitis B vaccine in STD clinics,
increased screening for STDs among
high-risk persons in youth detention
facilities and jails, and closer ties
between public STD clinics and
community managed-care organizations.
Personal health services objectives for
Healthy People 2010 are similar to
those for Healthy People 2000. Greater
emphasis is placed on annual screening
of sexually active young women for
chlamydia and on STD testing of
pregnant women during prenatal visits.
The link between STDs and other
preventable health outcomes strengthens
the relationship between the Healthy
People 2010 STD chapter and objectives
in other chapters, such as: Access to
Quality Health Services (e.g., counseling
about health behaviors; clinical skills for
providers); Cancer (e.g., cervical cancer
deaths; Pap tests); Family Planning
(e.g., abstinence among adolescents;
dual protection against pregnancy and
STDs; infertility services); HIV (e.g.,
condom use; STD services in HIV
clinics); Immunization and Infectious
Diseases (e.g., expanded hepatitis B
immunization); Injury and Violence
Prevention (e.g., sexual assault); and
Maternal, Infant, and Child Health (e.g.,
fetal deaths; prenatal care; low
birthweight; pre-term birth). These links
are highlighted as related objectives at
the end of the STD chapter in Healthy
People 2010.
Responsible sexual behavior is the
topic of 1 of the 10 Leading Health
Indicators (LHIs), which Healthy People
2010 introduces to serve as a barometer
of the Nation’s health. One objective
from the Sexually Transmitted Diseases
focus area—adolescents who abstain
from sexual intercourse or use condoms
if currently sexually active—is used to
measure this LHI.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the differences between the
two decades of objectives, reflecting
new knowledge and directions in this
area.
Data Issues
Definitions
In January 1988, the Centers for
Disease Control and Prevention (CDC)
issued new guidelines for classifying
and reporting cases of congenital
syphilis (19.4). The new surveillance
case definition is more useful for public
health surveillance; the previous
definition involved physical
examination, laboratory and radiographic
results, and follow-up serological data
(12). Followup information was often
difficult to obtain and led to delayed
and incomplete reporting. In addition,
the clinical criteria excluded stillbirths
to mothers with untreated syphilis. The
new case definition includes criteria for
presumptive and confirmed cases of
syphilis in infants and children and
includes stillbirths. A presumptive case
includes all infants whose mothers have
untreated or inadequately treated
syphilis at delivery (13). The number of
cases increased dramatically during
1989–91, partly as a result of the new
case definition. The case definition was
fully implemented in all States on
January 1, 1992; trends after this point
more accurately reflect changes in the
true incidence of congenital syphilis.
For objective 19.15, provider
referral (previously called contact
tracing) is the process whereby health
department personnel directly notify the
sexual partners of infected individuals of
their exposure to an infected individual
for the purpose of education, counseling,
and referral to health care services.
Data Sources
Data for objective 19.6 come from
the National Hospital Discharge Survey
(NHDS) maintained by the National
Center for Health Statistics, CDC. Data
for the survey are obtained from
approximately 480 hospitals throughout
the United States. Data on race are not
reported by many hospitals due to
omission of a race field on hospital
discharge reporting forms (UB–82 and
UB–92). Automation of the hospital
discharge systems has led to an increase
in the use of these forms in recent years.
A comparison of NHDS data with
National Health Interview Survey
(NHIS) data for people who reported
being hospitalized indicated that
underreporting for whites was roughly
22 percent in 1991; the difference in
reporting for blacks was negligible
(NHIS data were adjusted to exclude
hospitalizations of 1 day or less) (14).
The baseline data on counseling to
prevent HIV and other STDs for
objective 19.14 are from the Primary
Care Provider Surveys (PCPS). The
sample was drawn from the membership
rolls of professional organizations for
pediatricians, nurse practitioners, family
physicians, obstetricians/gynecologists,
and internists. Response rates varied
from 50 to 80 percent across these
groups. The data on counseling refer to
the proportion of providers who
routinely delivered these services to
81–100 percent of their patients who
needed the services.
The Prevention in Primary Care
Study (PPCS) was conducted in
1997–98 to update data from the PCPS.
The design and items included in the
1997–98 study were similar to the
PCPS, but a slightly different sampling
frame was used and some items
included in the 1992 surveys were not
included in the PPCS. The professionals
were sampled from listings of all
licensed, active practitioners in the
United States whose practices were at
least 50–80 percent primary care.
Because of low response rates from the
other provider groups, updates are
available only for nurse practitioners.
Data Comparability
Estimates of chlamydia prevalence
among females 15–24 years of age
(19.2) are obtained from the Sexually
Transmitted Disease Surveillance
System. Surveillance of chlamydial
infections is incomplete in many areas
of the United States; however,
surveillance is improving and, in 1994,
chlamydia became a nationally
notifiable condition (12). Baseline and
update data differ in data collection
methodology, which has improved, and
in the number of regions from which
rates are derived, which has increased.
Baseline and 1995 data for ‘‘all
females’’ for objective 19.9 (adolescent
postponement of sexual intercourse), for
‘‘females 15–44 years of age’’ and for
‘‘sexually active females 15–19 years of
age’’ for objective 19.10 (condom use at
last sexual intercourse), and for ‘‘all
females’’ for objective 19.16 (adolescent
270 Healthy People 2000 Final Review
abstinence) are from the National
Survey of Family Growth (NSFG).
Baseline and 1995 data for ‘‘all males’’
for these objectives are from the
National Survey of Adolescent Males
(NSAM). Biennial tracking data from
the Youth Risk Behavior Survey
(YRBS) are also displayed for these
objectives, but are not directly
comparable to the baselines or the
targets. The YRBS is a school-based
survey and thus does not include
teenagers who are not in school and
who are potentially at higher risk of
these behaviors (15). YRBS data, shown
by age in this report, are published by
grade only in other publications. (See
text for Priority Area 5 for more
information.)
References
1.Department of Health and Human Services.
Healthy People 2010. 2d ed. Washington: U.S.
Government Printing Office. Nov. 2000.
2. Institute of Medicine. Eng TR, Butler WT, eds.
The Hidden Epidemic: Confronting sexually
transmitted diseases. Washington: National
Academy Press. 1997.
3. Centers for Disease Control and Prevention,
Division of STD Prevention. Sexually transmitted
disease surveillance, 1999. Atlanta, Georgia:
Department of Health and Human Services, Public
Health Service. 2000.
4. American Social Health Association. Sexually
transmitted diseases in America: How many cases
and at what cost? Menlo Park, California: Kaiser
Family Foundation. 1998.
5. Wasserheit, J.N, Aral, SO. The dynamic
topology of sexually transmitted disease
epidemics. J Inf Dis 174 (suppl 2):201–13. 1996.
6. Centers for Disease Control and Prevention. Ten
leading national notifiable infectious
diseases—United States, 1995. MMWR 41:883–4.
1996.
7. Division of STD Prevention. Sexually
Transmitted Disease Surveillance, 1999.
Department of Health and Human Services,
Atlanta: Centers for Disease Control and
Prevention (CDC), Sept. 2000.
8. Centers for Disease Control and Prevention,
Division of STD Prevention. National plan to
eliminate syphilis from the United States. Atlanta,
Georgia: Department of Health and Human
Services, Public Health Service. 1999.
9. Fleming DT, McQuillan GM, Johnson RE, et al.
Herpes simplex virus type 2 in the United States,
1976 to 1994. N Engl J Med 337:1105–11. 1997.
10. McQuillan GM. Implications of a national
survey for STDs: Results from the NHANES
survey. Paper presented at the 2000 Infectious
Disease Society of America Conference. Sept.
7–10, New Orleans. 2000.
11. Koutsky L. Epidemiology of genital human
papillomavirus infection. Am J Med 102(suppl
5A):3–8. 1997.
12. Zenker P. New case definition for congenital
syphilis reporting. Sex Transm Dis 18:44–5. 1991.
13. Centers for Disease Control and Prevention.
Health risk behaviors among adolescents who do
and do not attend schools: United States, 1992.
MMWR 43:129–32. 1994.
14. Kozak LJ. Underreporting of race in the
National Hospital Discharge Survey. Advance data
from vital and health statistics; no 265.
Hyattsville, Maryland: National Center for Health
Statistics. 1995.
15. Centers for Disease Control and Prevention.
Youth Risk Behavior Surveillance—United States,
1999. MMWR 49:(SS–5). 2000.
Healthy People 2000 Final Review 271

Table 19. Sexually Transmitted Diseases objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
19.1 Gonorrhea (per 100,000) ........ 1989 300 277 247 197 173 166 149 124 123 133 133 100
a. Black (non-Hispanic)......... 1989 1,990 1,941 1,714 1,408 1,175 1,163 1,046 817 802 851 849 650
b. Adolescents 15-19 years ..... 1989 1,123 1,114 1,031 869 728 734 671 544 522 547 534 375
c. Female 15-44 years ......... 1989 501 495 417 364 309 316 299 259 252 282 283 175
19.2 Chlamydia prevalence among
females 15–24 years
Female 15-19 years ............ 1988 12.2% - - - - - - - - - - - - - - - 6.7% 5.4% - - -
1
6.9% 6.6% 5%
Female 20-24 years ............ 1988 8.5% - - - - - - - - - - - - - - - 4.2% 3.4% - - -
1
4.4% 4.5% 5%
19.3 Primary and secondary syphilis
(per 100,000) ................ 1989 18.1 20.3 17.0 13.3 10.3 7.9 6.3 4.3 3.2 2.6 2.5 4
a. Black .................... 1989 118 143 122 97 75 57 45 30 22 17 15 30
19.4 Congenital syphilis among infants
aged less than 1 year (per
100,000 live births) ............ 1990 91.0 . . . 107.3 94.7 80.9 55.8 47.6 32.9 27.7 21.6 14.3 40
a. Black .................... 1992
§
415.9 . . . . . . . . . 373.2 252.8 213.5 148.8 122.4 90.3 57.9 175
b. Hispanic .................. 1992
§
134.6 . . . . . . . . . 101.6 73.8 61.6 38.5 33.4 28.7 20.4 50
19.5 Annual number of first time
consultations
2
Genital herpes ................ 1988 163,000
3
172,000 235,000 139,000 172,000 142,000 160,000 208,000 176,000 188,000 224,000 138,500
Genital warts ................. 1988 290,000
3
275,000 282,000 218,000 167,000 238,000 253,000 191,000 145,000 211,000 240,000 246,500
19.6 Pelvic inflammatory disease
Hospitalizations per 100,000 females
15-44 years ................. 1988 311 261 233 212 196 177 162 164 157 153 - - - 100
Initial visits to physicians (number of
visits)
2
..................... 1988 430,800 357,522 376,540 334,793 386,860 312,000 262,000 286,000 261,000 234,000 251,000 290,000
Hospitalizations per 100,000 females
a. Black 15-44 years .......... 1988 655 567 523 539 399 378 296 320 281 266 - - - 150
b. Adolescents 15-19 years ..... 1988 342 279 239 205 159 184 141 168 186 158 - - - 110
19.7* Sexually transmitted Hepatitis B
(number of cases)............. 1987 47,593 47,811 58,393 52,882 35,849 35,077 29,446 36,794 34,568 32,700
4
24,527 30,500
19.8 Repeat gonorrhea infection ..... 1987 20% - - - - - - 16.7% 16.1% 13.8% 18.4% 18.5% 17.0% 17.5% 17.2% 15%
a. Black .................... 1992 21.3% . . . . . . . . . 19.9% 15.6% 20.1% 19.8% 18.3% 18.6% 19.2% 17%
19.9* Adolescents who ever engaged in
sexual intercourse
Adolescents 15 years
All females ................. 1988 27% - - - - - - - - - - - - - - - 22% - - - - - - - - - - - - 15%
In-school females........... ... --- 35% 36% --- 37% --- 38% --- 44% --- 43% ...
All males ................... 1988 33% - - - - - - - - - - - - - - - 27% - - - - - - - - - - - - 15%
In-school males ............ ... --- 48% 44% --- 45% --- 42% --- 42% --- 34% ...
a. All black males ............ 1988 69% - - - - - - - - - - - - - - - 58% - - - - - - - - - - - - 15%
In-school non-Hispanic black
males................... ... --- --- 79% --- 82% --- 77% --- 75% --- 68% ...
Adolescents 17 years
All females ................. 1988 50% - - - - - - - - - - - - - - - 51% - - - - - - - - - - - - 40%
In-school females........... ... --- 62% 66% --- 66% --- 67% --- 62% --- 40% ...
See footnotes and key at end of table.
272 Healthy People 2000 Final Review

Table 19. Sexually Transmitted Diseases objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
All males ................... 1988 66% - - - - - - - - - - - - - - - 53% - - - - - - - - - - - - 40%
In-school males ............ ... --- 73% 68% --- 68% --- 65% --- 60% --- 44% ...
b. All black males ............. 1988 90% - - - - - - - - - - - - - - - 79% - - - - - - - - - - - - 40%
In-school non-Hispanic black
males ................. ... --- --- 90% --- 92% --- 88% --- 85% --- 82% ...
c. All black females 15-17 years . 1988 66% - - - - - - - - - - - - - - -
5
4 8 % --- --- --- --- 4 0 %
In-school non-Hispanic black
females................ ... --- --- 84% --- 80% --- 75% --- 73% --- 73% ...
19.10 Condom use at last sexual
intercourse
Sexually active unmarried females
15-44 years (by their partners) . . . 1988 19% - - - - - - - - - - - - - - - 25% - - - - - - - - - - - - 50%
a. Sexually active females
15–19 years (by their
partners) ................. 1988 26% - - - - - - - - - - - - - - - 37% - - - - - - - - - - - - 60%
Sexually active females 15-19 years
in grades 9-12 (by their partners) . . . . - - - 40% 38% - - - 46% - - - 49% - - - 51% - - - 51% . . .
b. Sexually active males
15-19 years .............. 1988 57% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 75%
Sexually active males 15-19 years
in grades 9-12................ ... --- 49% 54% --- 59% --- 61% --- 63% --- 66% ...
c. Injecting drug users ......... 1992
6
34% ... ... ... --- --- --- --- --- --- --- 75%
d. Black females 15-44 years (by
their partners) ............. 1988 12.4% - - - - - - - - - - - - - - -
5
25% --- --- --- --- 75%
19.11* Clinic services for HIV and other
sexually transmitted diseases . . ... --- --- --- --- --- --- --- --- --- --- --- 50%
Family planning clinics .......... 1989 40% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - . . .
Title X funded family planning clinics
STD testing (excluding HIV)..... ... --- --- --- --- --- 95% --- --- --- --- --- ...
STD counseling (excluding HIV) . . . . - - - - - - - - - - - - - - - 98% - - - - - - - - - - - - - - - . . .
STD treatment (excluding HIV) . . . . . - - - - - - - - - - - - - - - 93% - - - - - - - - - - - - - - - . . .
Gonorrhea
Client testing
7
............... ... --- 97% --- --- --- --- --- --- --- --- --- ...
Client treatment.............. ... --- 82% --- --- --- --- --- --- --- --- --- ...
Partner notification
8
........... ... --- 23% --- --- --- --- --- --- --- --- --- ...
Partner testing............... ... --- 60% --- --- --- --- --- --- --- --- --- ...
Partner treatment ............ ... --- 62% --- --- --- --- --- --- --- --- --- ...
Syphilis
Client testing
7
................. ... --- 86% --- --- --- --- --- --- --- --- --- ...
Client treatment.............. ... --- 48% --- --- --- --- --- --- --- --- --- ...
Partner notification
8
........... ... --- 29% --- --- --- --- --- --- --- --- --- ...
Partner testing............... ... --- 57% --- --- --- --- --- --- --- --- --- ...
Partner treatment ............ ... --- 40% --- --- --- --- --- --- --- --- --- ...
See footnotes and key at end of table.
Healthy People 2000 Final Review 273

Table 19. Sexually Transmitted Diseases objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Chlamydia
Client testing
7
............... ... --- 66% --- --- --- --- --- --- --- --- --- ...
Client treatment.............. ... --- 73% --- --- --- --- --- --- --- --- --- ...
Partner notification
8
........... ... --- 15% --- --- --- --- --- --- --- --- --- ...
Partner testing............... ... --- 29% --- --- --- --- --- --- --- --- --- ...
Partner treatment ............ ... --- 50% --- --- --- --- --- --- --- --- --- ...
HIV
Client pretest counseling ....... ... --- 66% --- --- --- 82% --- --- --- --- --- ...
Client testing ................ ... --- 60% --- --- --- 74% --- --- --- --- --- ...
19.12* HIV and other STD education
curricula
Schools offering at least one STD
class....................... 1988 95% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 95%
Proportion of middle and senior high
schools:
With HIV prevention in required
courses ................... ... --- --- --- --- --- 86% --- --- --- --- --- ...
With STD prevention in required
courses ................... ... --- --- --- --- --- 84% --- --- --- --- --- ...
19.13 Correct management of sexually
transmitted disease cases by
primary care providers ........ 1988 70% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 90%
19.14* Clinician counseling to prevent
HIV and other sexually
transmitted diseases.......... 1987
9
10% --- --- --- --- --- --- --- --- --- --- 75%
Percent of clinicians routinely
providing service to 81-100% of
patients
a. Providers practicing in high
incidence areas ............ ... --- --- --- --- --- --- --- --- --- --- --- 90%
b. Family physicians ........... 1992 27% . . . . . . . . . - - - - - - - - - - - - - - -
10,11
--- --- 75%
c. Internists.................. 1992 30% . . . . . . . . . - - - - - - - - - - - - - - -
10,11
--- --- 75%
d. Nurse practitioners .......... 1992 50% . . . . . . . . . - - - - - - - - - - - - - - -
10
45% - - - 75%
e. Obstetricians/gynecologists.... 1992 46% . . . . . . . . . - - - - - - - - - - - - - - -
10,11
--- --- 75%
f. Pediatricians ............... 1992 46% . . . . . . . . . - - - - - - - - - - - - - - -
10,11
--- --- 75%
g. Mental health care providers. . . . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 75%
19.15 Partner notification of exposure to
sexually transmitted diseases
Patients with bacterial sexually
transmitted diseases ........... 1988 20% - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 50%
19.16* Adolescent abstinence from
sexual intercourse for previous
3 months
All sexually active females 15-17
years ....................... 1988 23.6% - - - - - - - - - - - - - - - 27% - - - - - - - - - - - - 40%
See footnotes and key at end of table.
274 Healthy People 2000 Final Review

Table 19. Sexually Transmitted Diseases objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
In-school sexually active females
15-17 years ............... ... --- 2 4 % 2 5 % --- 2 5 % --- 2 3 % --- 2 3 % --- 2 5 % ...
All sexually active males 15-17
years ...................... 1988 33% - - - - - - - - - - - - - - - 37% - - - - - - - - - - - - 40%
In-school sexually active males
15-17 years ................. ... --- 3 0 % 3 6 % --- 3 3 % --- 3 4 % --- 3 2 % --- 3 2 % ...
19.17* HIV and STD education for
students at colleges and
universities
Students 18 years and over given:
AIDS or HIV infection prevention
information ................ 1995 49.1% . . . . . . . . . . . . . . . . . . - - - - - - - - - - - - 90%
STD prevention information....... 1995 43.4% . . . . . . . . . . . . . . . . . . - - - - - - - - - - - - 90%
Students 18 years and over taught
about AIDS or HIV in a college
class....................... 1995 41.4% . . . . . . . . . . . . . . . . . . - - - - - - - - - - - - 90%
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
1
Positivity measure is not adjusted for changes in laboratory test methods and associated increases in the test sensitivity.
2
As measured by first-time visits to physicians’ offices.
3
1989 data.
4
Data are provisional.
5
Data are for non-Hispanic black females.
6
Data are for male and female injecting drug users (married and unmarried) who report having vaginal sex within the last 6 months and reported using a condom ‘‘always’’ or ‘‘sometimes.’’
7
Includes testing at initial visit, at annual visit, or if symptomatic.
8
By family planning clinic staff via telephone or mail.
9
Data are for new patients.
10
1997–98 data.
11
Response rate for this group was too low to produce reliable estimates.
NOTES: Data include revisions and, therefore, may differ from data previously published in these reports and other publications. HIV is human immunodeficiency virus. STD is sexually
transmitted disease.
Objective number Data source
19.1, 19a-c Sexually Transmitted Disease Surveillance System, CDC, NCHSTP.
19.2 Sexually Transmitted Disease Surveillance System, CDC, NCHSTP.
19.3, 19.3a Sexually Transmitted Disease Surveillance System, CDC, NCHSTP.
19.4 Sexually Transmitted Disease Surveillance System, CDC, NCHSTP.
19.5 National Disease and Therapeutic Index, IMS America, Ltd.
19.6, 19.6a-b For hospitalizations, National Hospital Discharge Survey, CDC, NCHS.
For number of visits, National Disease and Therapeutic Index, IMS America, Ltd.
19.7* Viral Hepatitis Surveillance System, CDC, NCID.
19.8 Gonococcal Isolate Surveillance Project, CDC, NCHSTP.
Met
Toward
Mixed/ no change
Away
Cannot assess
Healthy People 2000 Final Review 275

Objective number Data source
19.9* Baseline and updates for all females and all black females: National Survey of Family Growth, CDC, NCHS.
Baseline and updates for all males and all black males: National Survey of Adolescent Males, NIH, NICHD.
1990–99 data for in-school females and males: Youth Risk Behavior Survey, CDC, NCCDPHP.
19.10*, 19.10d National Survey of Family Growth, CDC, NCHS.
19.10a Baseline: National Survey of Family Growth, CDC, NCHS.
1990–99 data for in-school females and males: Youth Risk Behavior Survey, CDC, NCCDPHP.
19.10b Baseline: National Survey of Adolescent Males, NIH, NICHD.
1990–99 data for in-school males and females: Youth Risk Behavior Survey, CDC, NCCDPHP.
19.10c National AIDS Demonstration Research Program, NIH, NIDA.
19.11* Baseline:State Family Planning Directors.
1990 data: National Questionnaire on Provision of STD and HIV Services by Family Planning Clinics, OPA.
1994 data: The Urban Institute. Family planning clinics: Current status and recent changes in services, clients, staffing, and income sources. March 1994.
19.12* Baseline: AIDS education: Public school programs require more student information and teacher training, GAO, 1990.
1994 data: School Health Policies and Programs Study, CDC, NCCDPHP.
19.13 National Disease and Therapeutic Index, IMS America, Ltd.
19.14* 1987 baseline: Sexual history-taking and counseling practices of primary care physicians, Lewis CE and Freeman HE. Western Journal of Medicine,
147: 165–7. 1987.
1992 baseline: Primary Care Provider Surveys, OPHS, ODPHP.
Update: Prevention in Primary Care Study, American College of Preventive Medicine.
19.15 Sexually Transmitted Disease Surveillance System, CDC, NCHSTP.
19.16* Baseline and update for all females: National Survey of Family Growth, CDC, NCHS.
Baseline and update for all males: National Survey of Adolescent Males, NIH, NICHD.
1990–99 data for in-school females and males: Youth Risk Behavior Survey, CDC, NCCDPHP.
19.17* National College Health Risk Behavior Survey, CDC, NCCDPHP.
* Duplicate objective. See full text of objective following this table.
276 Healthy People 2000 Final Review
Sexually Transmitted
Diseases Objectives
19.1: Reduce gonorrhea to an incidence
of no more than 100 cases per 100,000
people.
19.1a: Reduce gonorrhea among
blacks to an incidence of no more
than 650 cases per 100,000.
19.1b: Reduce gonorrhea among
adolescents aged 15–19 to an
incidence of no more than 375
cases per 100,000.
19.1c: Reduce gonorrhea among
women aged 15–44 to an incidence
of no more than 175 cases per
100,000.
19.2: Reduce the prevalence of
Chlamydia trachomatis infections among
young women (under the age of 25
years) to no more than 5 percent.
19.3: Reduce primary and secondary
syphilis to an incidence of no more than
4 cases per 100,000 people.
19.3a: Reduce primary and
secondary syphilis among blacks to
an incidence of no more 30 cases
per 100,000.
19.4: Reduce congenital syphilis to an
incidence of no more than 40 cases per
100,000 live births.
19.4a: Reduce congenital syphilis
among blacks to an incidence of no
more than 175 cases per 100,000
live births.
19.4b: Reduce congenital syphilis
among Hispanics to an incidence of
no more than 50 cases per 100,000
live births.
19.5: Reduce genital herpes and genital
warts, as measured by a reduction to
138,500 and 246,500, respectively, in
the annual number of first-time
consultations with a physician for the
conditions.
19.6: Reduce the incidence of pelvic
inflammatory disease, as measured by a
reduction in hospitalizations for pelvic
inflammatory disease, to no more than
100 per 100,000 women aged 15–44
and a reduction in the number of initial
visits to physicians for pelvic
inflammatory disease to no more than
290,000.
19.6a: Reduce the incidence of
pelvic inflammatory disease among
blacks, as measured by a reduction
in hospitalizations for pelvic
inflammatory disease, to no more
than 150 per 100,000 women aged
15–44.
19.6b: Reduce the incidence of
pelvic inflammatory disease among
adolescents, as measured by a
reduction in hospitalizations for
pelvic inflammatory disease, to no
more than 110 per 100,000 females
aged 15–19.
19.7*: Reduce sexually transmitted
hepatitis B infection to no more than
30,500 cases.
Duplicate objectives: 20.03b and 20.03c,
combined
19.8: Reduce the rate of repeat
gonorrhea infection to no more than
15 percent within the previous year.
19.8a: Reduce the rate of repeat
gonorrhea infection among blacks to
no more than 17 percent within the
previous year.
19.9*: Reduce the proportion of
adolescents who have engaged in sexual
intercourse to no more than 15 percent
by age 15 and no more than 40 percent
by age 17.
Duplicate objectives: 5.4 and 18.3
19.9a*: Reduce the proportion of
black males aged 15 years who
have engaged in sexual intercourse
to no more than 15 percent.
Duplicate objectives: 5.4a and 18.3a
19.9b*: Reduce the proportion of
black males aged 17 years who
have engaged in sexual intercourse
to no more than 40 percent.
Duplicate objectives: 5.4b and 18.3b
19.9c*: Reduce the proportion of
black females aged 17 years who
have engaged in sexual intercourse
to no more than 40 percent.
Duplicate objectives: 5.4c and 18.3c
19.10*: Increase to at least 50 percent
the proportion of sexually active,
unmarried people who used a condom at
last sexual intercourse.
Duplicate objective: 18.4
19.10a*: Increase to at least
60 percent the proportion of
sexually active, unmarried young
women aged 15–19 whose partner
used a condom at last sexual
intercourse.
Duplicate objective: 18.4a
19.10b*: Increase to at least
75 percent the proportion of
sexually active, unmarried young
men aged 15–19 who used a
condom at last sexual intercourse.
Duplicate objective: 18.4b
19.10c*: Increase to at least
60 percent the proportion of
intravenous drug users who used a
condom at last sexual intercourse.
Duplicate objective: 18.4c
19.10d*: Increase to at least
75 percent the proportion of black
women aged 15–44 whose partner
used a condom at last sexual
intercourse.
Duplicate objective: 18.4d
19.11*: Increase to at least 50 percent
the proportion of family planning
clinics, maternal and child health clinics,
sexually transmitted disease clinics,
tuberculosis clinics, drug treatment
centers, and primary care clinics that
provide on site primary prevention and
provide or refer for secondary
prevention services for HIV infection
and bacterial sexually transmitted
diseases (gonorrhea, syphilis, and
Chlamydia) to high-risk individuals and
their sex or needle-sharing partners.
Duplicate objectives: 5.11 and 18.13
19.12*: Increase to at least 95 percent
the proportion of schools that have
appropriate HIV and other STD
education curricula for students in
4th–12th grade, preferably as part of
comprehensive school health education,
based upon scientific information that
includes the way HIV and other STDs
are prevented and transmitted. Duplicate
objective: 18.10
19.13: Increase to at least 90 percent the
proportion of primary care providers
treating patients with sexually
transmitted diseases who correctly
manage cases, as measured by their use
of appropriate types and amounts of
therapy.
19.14*: Increase to at least 75 percent
the proportion of primary care and
mental health care providers who
provide appropriate counseling on the
Healthy People 2000 Final Review 277
prevention of HIV and other sexually
transmitted diseases.
Duplicate objective: 18.9
19.14a*: Increase to at least
90 percent the proportion of primary
care and mental health care
providers who practice in areas of
high AIDS and sexually transmitted
disease incidence who provide
appropriate counseling on the
prevention of HIV and other
sexually transmitted diseases.
Duplicate objective: 18.9a
19.14b*: Increase to at least
75 percent the proportion of family
physicians who provide appropriate
counseling on the prevention of
HIV and other sexually transmitted
diseases.
Duplicate objective: 18.9b
19.14c*: Increase to at least
75 percent the proportion of
internists who provide appropriate
counseling on the prevention of
HIV and other sexually transmitted
diseases.
Duplicate objective: 18.9c
19.14d*: Increase to at least
75 percent the proportion of nurse
practitioners who provide
appropriate counseling on the
prevention of HIV and other
sexually transmitted diseases.
Duplicate objective: 18.9d
19.14e*: Increase to at least
75 percent the proportion of
obstetricians/gynecologists who
provide appropriate counseling on
the prevention of HIV and other
sexually transmitted diseases.
Duplicate objective: 18.9e
19.14f*: Increase to at least
75 percent the proportion of
pediatricians who provide
appropriate counseling on the
prevention of HIV and other
sexually transmitted diseases.
Duplicate objective: 18.9f
19.14g*: Increase to at least
75 percent the proportion of mental
health care providers who provide
appropriate counseling on the
prevention of HIV and other
sexually transmitted diseases.
Duplicate objective: 18.9g
19.15: Increase to at least 50 percent the
proportion of all patients with bacterial
sexually transmitted diseases (gonorrhea,
syphilis, and chlamydia) who are offered
provider referral services.
19.16*: Increase to at least 40 percent
the proportion of ever sexually active
adolescents aged 17 and younger who
have not had sexual intercourse for the
previous 3 months.
Duplicate objective: 5.5 and 18.15
19.17*: Increase to at least 90 percent
the proportion of students who received
HIV and other STD information,
education, or counseling on their college
or university campus.
Duplicate objective: 18.11
*Duplicate objective.
278 Healthy People 2000 Final Review

Priority Area 20
Immunization and
Infectious Diseases
Background
During the 20th century, the United
States made great strides in the
reduction in incidence of infectious
disease, yet, despite the progress,
infectious diseases remain an important
cause of illness and death in the United
States. Each of the causative agents of
infectious diseases, even those that are
currently rare, poses a potential threat of
producing disease, as evidenced by the
resurgence of tuberculosis (TB) that was
complicated by multiple-drug resistance.
The emergence of drug resistance to TB
and other infectious diseases threatens to
reverse the progress prompted by the
discovery of penicillin and other miracle
drugs. Even with these miracle drugs,
infectious diseases are a leading cause
of death worldwide, and serious
infections were the third leading cause
of death in the United States in 1992
(1).
The development and widespread
use of vaccines, combined with
increased funding and enhancement of
infrastructure, have been instrumental in
reducing the incidence of many
infectious diseases, particularly
childhood diseases. Approximately
80 percent of childhood vaccine doses
are recommended for administration
before the second birthday; and
vaccination coverage among children in
the United States is at record high levels
(2). Protecting children against
vaccine-preventable diseases has become
a national priority. One of the greatest
challenges the country faces is
extending the success in immunization
of children to the adult population. The
public health and financial burdens due
to the occurrence of vaccine-preventable
diseases among adults in the United
States is staggering; for example,
moderately severe outbreaks of
influenza may cost society more than
$10 billion (3). Although the severity of
influenza seasons varies, an annual
average of approximately 20,000 deaths
and 110,000 pneumonia and influenza
(P&I) hospitalizations result from
influenza infections (4–6). In addition,
over 6,000 deaths occur annually from
invasive pneumococcal infections, and
an estimated 13,000 adults die every
year from chronic liver disease related
to hepatitis B virus (HBV) infection or
hepatitis C virus (HCV) infection.
Data Summary
Highlights
Vaccination levels among children
are the highest ever recorded in the
United States (20.11). The proportion of
children 19–35 months fully vaccinated
against Haemophilus influenza type b
(Hib) increased to 94 percent in 1999
from 2 percent in 1991 when the
recommendations were published; the
proportion of children vaccinated against
polio increased 76 percent between 1991
(53 percent) and 1999 (90 percent). The
proportion of children who have
received a series of vaccinations
measured by having four doses of
diphtheria-tetanus-pertussis vaccine,
three doses of polio vaccine, and one
dose of measles-containing vaccine
(MCV) increased from 55 percent in
1992 to 80 percent in 1999. From 1989
to 1998, influenza and pneumococcal
vaccination levels among people 65
years of age and over (20.11) also
continued to increase for the total
population, African Americans (20.11a),
and Hispanics (20.11b). However, racial
and ethnic disparities in vaccination
levels persist among Hispanics and
non-Hispanic African Americans (7).
The incidence of almost all
vaccine-preventable diseases (20.1)in
children continued to be low during
1999. There were no cases of diphtheria
or polio due to wild virus, fewer than 6
cases of tetanus among persons 25 years
of age and under, and only 6 cases of
reported congenital rubella syndrome.
The number of rubella cases in 1999
(267) dropped 76 percent from the
number of cases reported in 1990
(1,125). An interruption of indigenous
measles transmission likely occurred in
the fall of 1993, although importation of
the virus resulted in moderate measles
outbreaks in 1994 primarily among
groups that refused vaccination. The
number of measles cases decreased
99.6 percent from 1990 (26,527) to 1999
(100). However, pertussis incidence,
which had declined by 15 percent in
1994 from the 20–year high reported in
1993, increased to an even higher level
in 1996, with 7,796 cases, and remained
high in 1999 (7,298).
The incidence rate of hepatitis B
(HBV) (20.3) has continued to decline
since the start of the decade, and in
1999 was far below the baseline rate
reported in 1987 and far below the
target rate set for the year 2000.
Although cases of HBV infection in
children have become rare, as a result of
high levels of hepatitis B vaccine
coverage in younger age groups, a
substantial number (estimated 180,000)
of adults continue to be infected with
HBV because of low levels of vaccine
coverage in older age groups. The rate
of hepatitis A (20.3) has decreased by
approximately 50 percent since 1995,
and in 1999 was the lowest yet
recorded, reflecting dramatic declines in
the rates of disease among Native
Americans (20.3j) and other high-risk
groups. A dramatic decline of more than
80 percent in the incidence of hepatitis
C (HCV) (20.3) has occurred since 1989
and is associated with a decrease in
cases occurring among injecting-drug
users.
The incidence of tuberculosis (20.4)
declined after 1992 to 6.4 cases per
100,000 persons in 1999, well below the
1988 baseline of 9.1. Also by 1999, the
percent of multi-drug resistant
tuberculosis cases was reduced to
1.1 percent from a 1992 high of almost
3 percent.
In 1998, preliminary data show that
the surgical wound infection rate (20.5)
targets were surpassed for all groups
except low-risk patients.
The rate of infection with bacterial
meningitis (20.7) decreased from 6.5
cases per 100,000 persons in 1986 to
2.2 in 1999, far surpassing the year
2000 target of 4.7. Among Alaska
Natives (20.7a), a group particularly at
risk, the infection rate fell from 33 cases
per 100,000 in 1987 to 5.7 in 1999, also
surpassing the target of 8 cases per
100,000.
The number of restricted activity
days due to ear infections per 100
children 4 years and under (20.9)
dropped to 103.4 in 1996, a rate below
the Healthy People 2000 target of 105.0
restricted activity days per 100 children.
Summary of Progress
Data are available to assess progress
for 17 of the 19 objectives in the
Immunization and Infectious Diseases
Priority Area. Three objectives (20.2,
20.7, and 20.9) met or exceeded the
year 2000 targets. For seven objectives
(20.3, 20.4, 20.11, 20.13, 20.14, 20.16,
Healthy People 2000 Final Review 279

20.1 Vaccine-preventable disease: Diphtheria
Tetanus
Polio (wild-type virus)
Measles
Rubella
Congenital Rubella Syndrome
Mumps
Pertussis
20.2 Epidemic-related pneumonia and influenza deaths
20.3 Viral hepatitis: Hepatitis B
Hepatitis A
Hepatitis C
20.4 Tuberculosis cases
Urinary tract infections: Medical/coronary ICUs
20.5 Surgical wound/nosocomial infections,
via bloodstream: Medical/coronary ICUs
Pediatric ICUs
Surgical/medical-surgical ICUs
Surgical/medical-surgical ICUs
Pediatric ICUs
Pneumonia: Medical/coronary ICUs
Surgical/medical-surgical ICUs
Pediatric ICUs
Medium-low risk patients
Medium-high risk patients
20.6 Illness international travelers: Typhoid fever
High-risk patients
Surgical wound infection rates: Low-risk patients
Hepatitis A
Malaria
20.7 Bacterial meningitis cases
20.9 Ear infections
Moved away from target Moved toward target Met target
Chart continues onto next page- see notes at the end of the chart.
78%
-80%
-67%
-19%
-73%
No change
2%
11%
Percent of target achieved
-100 -80 -60 -40 -20 0 20 40 60 80 100
100%
100%
103%
157%
327%
391%
300%
492%
233%
167%
367%
507%
239%
105%
103%
198%
346%
48%
83%
-157%
-120%
-600%
97%
84%
280 Healthy People 2000 Final Review
Figure 20. Final status of Immunization and Infectious Diseases objectives

Figure 20. Final status of Immunization and Infectious Diseases objectives—Con.
20.10 Pneumonia-related restricted activity days,
persons 65 years and over
Children 4 years and under
20.11 Basic Immunization Series, children 19-35 months:
DTP (3 or more doses)
Polio (3 or more doses)
Measles-containing
Hib (3 or more doses)
Hep B (3 or more doses)
4DTP/3Polio/1MMR
Pneumococcal vaccine - Institutionalized
chronically ill or older people
Pneumococcal vaccine - Noninstitutionalized
65 years and over
Influenza immunization - Institutionalized chronically ill
or older people
Influenza immunization - Noninstitutionalized
65 years and over
20.12 Post exposure rabies treatments
20.13 Immunization laws
20.15 Financial barriers to immunization: Conventional
insurance plans
PPOs
HMOs
20.16 Public health department provision of
immunizations: Pneumococcal vaccine
Influenza vaccine
20.19 Lab-diagnosed influenza: Tertiary care hospital labs
20.18 Infected people completing tuberculosis therapy
Secondary care hospital labs
HMO labs
Moved away from target Moved toward target Met target
NOTES: Complete tracking data are shown in table 20. Progress quotients are not calculated for objectives 20.8, 20.14, and 20.17.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
ICU is intensive care unit. DTP is diphtheria-tetanus-pertussis. Hib is
Haemophilus influenzae
type b vaccine. MMR is measles-mumps-rubella.
PPO is preferred provider organization. HMO is health maintenance organization.
50%
58%
15%
8%
-9%
No change
21%
20%
18%
Percent of target achieved
-100 -80 -60 -40 -20 0 20 40 60 80 100
228%
129%
100%
125%
105%
111%
103%
69%
16%
10%
71%
-133%
-150%
-1,540%
97%
85%
Healthy People 2000 Final Review 281
and 20.19), progress was made toward
achieving the targets. The progress of
objective 20.14 is based on nurse
practitioner data only. Trends for two
objectives (20.12 and 20.18) indicate
movement away from the year 2000
targets. Mixed results are shown for five
objectives (20.1, 20.5, 20.6, 20.10, and
20.15). Data are not available to provide
measures after baseline for two
objectives (20.8 and 20.17). See table 20
for the tracking data for the objectives
in this priority area and figure 20 for a
quantitative assessment of progress.
Discussion
The reduction in incidence of
infectious diseases is a significant public
health achievement of the 20th century.
Record levels of vaccination coverage
have greatly contributed to this
reduction. For instance, the dramatic
decrease in cases of bacterial meningitis
is due to the licensure of Hib
polysaccharide-protein conjugate
vaccines in 1988. Since the vaccines
were licensed, the number of reported
Hib invasive disease among children
under 5 years of age has declined
99 percent.
High levels of hepatitis B
vaccination coverage in children have
resulted in decreasing rates of hepatitis
B among younger age groups, reducing
the burden of chronic liver disease due
to childhood HBV infection and
eventually resulting in decreases in the
overall rate of hepatitis B. However,
until there is a nationwide program to
vaccinate adults at increased risk for
HBV infection, transmission cannot be
eliminated and disease rates will most
likely remain elevated for decades.
The sharp decrease in hepatitis A
rates among high-risk populations such
as Native Americans is attributed to the
implementation of hepatitis A
vaccination programs in those
populations. However, cyclic peaks in
hepatitis A rates have been observed
approximately every 10 years and future
increases in rates may yet occur.
Widespread hepatitis A vaccination of
children, particularly in States and
communities with consistently elevated
rates of hepatitis A, is needed to achieve
sustained reductions in hepatitis A rates.
Although the incidence of HCV
infection has decreased dramatically
over the decade, 1988–94
seroprevalence data indicate that
approximately 4 million people in the
United States have been infected with
HCV, most of whom are chronically
infected (8). Reducing the burden of
HCV infection and HCV-related disease
in the United States requires
implementation of primary prevention
activities to reduce the risk for
contracting HCV infection and
secondary prevention activities to reduce
the risk of liver and other chronic
diseases in HCV-infected persons.
High vaccination coverage levels
have been achieved for children ages
19–35 months. In 1991, in an attempt to
achieve 90 percent coverage by the year
2000, Congress required States to
prepare Immunization Action Plans that
focused on expanding the public health
infrastructure for vaccination service
delivery and allowed Federal grant
funds to support both service delivery
(for example, salaries of nurses and
clinic supplies) and vaccine costs. In
1994, the Vaccines for Children (VFC)
program was initiated. VFC is an
entitlement program that has removed
vaccine cost as a barrier to vaccination
for America’s neediest children, and
provides a more secure source of
funding. Seventy-five percent of
preschool-aged children in the United
States are vaccinated in one of the
50,000 provider sites participating in
this program. As a result of increased
vaccination coverage levels, the
incidence of vaccine-preventable
diseases decreased. While vaccination
levels have progressed toward and
reached many of the year 2000 coverage
goals, continued diligence is needed to
sustain and improve our progress.
Timely, age-appropriate delivery of
vaccines to infants and children is the
backbone of the Nation’s immunization
program. Yet, despite recent dramatic
gains in childhood vaccination coverage,
more than 20 percent of 2 year olds in
the United States have still not received
all recommended doses of vaccine. The
addition of new and improved vaccines
to an already complex childhood
vaccination schedule makes it
increasingly challenging to ensure
complete and age-appropriate
vaccination.
While an increasing number of
adults 65 years of age and over have
taken advantage of the health benefits of
vaccines against influenza and
pneumococcal disease, more work needs
to be done to protect older Americans
from these preventable diseases,
particularly to eliminate racial and
ethnic disparities, and to ensure high
levels of coverage among seniors who
live in nursing homes.
From a rate of 9.1 cases per
100,000 persons in the 1988 baseline
year, the United States tuberculosis case
rate rose to 10.5 in 1992. The national
resurgence of tuberculosis between 1985
and 1992 was associated with the HIV
epidemic, imported cases among
immigrants from tuberculosis-endemic
areas, and the occurrence of multi-drug
resistant strains of the disease. The
resurgence necessitated a rebuilding of
the network for diagnosis, treatment,
and follow-up of tuberculosis cases and
contacts. As a consequence, there has
been a renewed decline in the United
States case rate that was reduced to 6.4.
per 100,000 by 1999. Although the 1999
rate is still almost twice the year 2000
target of 3.5 cases per 100,000 persons,
it is an historic low annual rate for the
United States since nationwide reporting
began in 1953. In 1999, the rate for
blacks was 16.8, for Hispanics 12.4, for
American Indians/Alaska Natives 11.8,
for Asians and Pacific Islanders 35.3;
rates for all of these groups are now
below their 1988 baselines. The targets
for each of these population groups was
10.0, 5.0, 5.0, and 15.0, respectively.
The proportion of persons who
completed a prescribed course of
treatment for latent tuberculosis
infection has changed little since the
1987 baseline of 66.3 percent and the
latest report was 64.6 percent in 1998.
During and after the resurgence of
tuberculosis, many health departments
necessarily placed priority on controlling
outbreaks, finding cases, ensuring the
completion of therapy by patients,
examining exposed persons, and halting
the spread of infection. Resources were
not adequate in many areas to identify
persons with latent TB infection and
ensure completion of preventive therapy.
Transition to Healthy People
2010
Although there has been notable
progress in efforts to prevent and
control infectious diseases, reemerging
and antimicrobial resistant infectious
diseases present major public health
issues. There is a continuing need to
improve the capacity to address the
challenges posed by infectious diseases.
The scope of the Healthy People 2010
Immunization and Infectious Diseases
282 Healthy People 2000 Final Review
chapter (Focus Area 14) has been
expanded from Healthy People 2000 to
reflect new prevention opportunities
resulting from new scientific knowledge
and technology. Healthy People 2010
includes the addition of objectives to
encourage appropriate use of
antimicrobials and prophylaxis, plus
new vaccines such as varicella. The
chapter also places a greater emphasis
on preventing disease among high-risk
persons of all ages. For example, in
recognition of the vaccine-preventable
disease burden among adolescents and
adults, several Healthy People 2010
immunization-related objectives address
these age groups.
Several of the immunization-related
objectives will continue to be measured
in Healthy People 2010. These include
incidence of vaccine-preventable
diseases and vaccination coverage levels
for specific vaccines. Some new
objectives have also been added,
including one to assess how well the
service delivery system is meeting the
vaccination needs of young children and
adolescents. Other new objectives
include increasing the proportion of
providers who measure coverage levels
in their practice and increasing the
proportion of children under 6 years of
age who participate in fully operational,
State- and population-based
immunization registries. Two vaccine
safety-related objectives have also been
included in Healthy People 2010,
reducing vaccine-associated adverse
events, and increasing the number of
persons under active surveillance for
vaccine safety via large linked
databases.
In order to better track progress in
preventing and controlling viral hepatitis
in the United States, the one hepatitis
objective in Healthy People 2000 (20.3)
has been expanded into six objectives in
Healthy People 2010. To better assess
the disease burden due to HCV, Healthy
People 2010 includes a developmental
objective to increase the proportion of
persons with chronic HCV infection
identified by health departments. Several
of the infectious diseases objectives
from Healthy People 2000 were revised
to reflect a growing problem of
antimicrobial resistance. Recognizing
that completion of therapy helps to
reduce the TB case rate by preventing
transmission of infection as well as
outbreaks and development and spread
of multi-drug resistant TB, an objective
has been added, which seeks to increase
the percent of all tuberculosis patients
who complete therapy for tuberculosis
disease within 12 months.
Immunizations is the topic of 1 of
the 10 Leading Health Indicators
(LHIs), which Healthy People 2010
introduces to serve as a barometer of the
Nation’s health. Two objectives from the
Immunizations and Infectious Diseases
focus area—pneumococcal and influenza
vaccinations of adults at high risk and
fully vaccinated young children—are
used to measure this LHI.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the differences between the
two decades of objectives, reflecting
new knowledge and directions in this
area.
Data Issues
Definitions
Operational definitions and data
collection specifications for all Healthy
People 2000 objectives in Priority Area
20 have been published in the National
Center for Health Statistics, Healthy
People Statistical Notes series (9). Data
issues are discussed and references are
cited for expanded discussions of the
data systems that provide data for the
national objectives. When appropriate,
the text of questionnaire items used to
measure the objectives is also provided.
See Appendix table VI for more
information.
For objective 20.2, epidemic-related
pneumonia and influenza deaths are
defined as those that occur above and
beyond the normal yearly fluctuations of
mortality. Because of the extreme
variability in epidemic-related deaths
from year to year, the objective was
measured using a 3-year average. The
data cannot be obtained directly from
published mortality figures. Each year
expected numbers of pneumonia and
influenza deaths are calculated through a
cyclical regression model using data for
previous years but excluding data for
the periods when mortality was known
to be raised by influenza epidemics (5).
Epidemic-related deaths are defined as
those that exceed the predicted number
during epidemic periods based on the
model.
Data Sources
The National Notifiable Disease
Surveillance System (NNDSS) is the
data source for tracking cases of
vaccine-preventable diseases (20.1).
Detailed epidemiologic analyses of data
from NNDSS are sometimes published
in special surveillance reports. Data in
these reports may not agree exactly with
reports published in the Morbidity and
Mortality Weekly Report because of
differences in timing or refinements in
case definitions. The NNDSS is the data
source for specific disease surveillance
systems, such as the Tuberculosis
Morbidity Data System (20.4).
Additional hepatitis surveillance data
besides that provided through NNDSS
are collected through the Viral Hepatitis
Surveillance Program and the Sentinel
Counties Study of Acute Viral Hepatitis
(20.3). Although cases of congenital
rubella syndrome are reported through
NNDSS, the actual source of the data is
the National Congenital Rubella
Syndrome Registry (NCRSR).
The baseline data on provision of
immunizations by physicians for
objective 20.14 are from the Primary
Care Provider Surveys (PCPS). The
sample was drawn from the membership
rolls of professional organizations for
pediatricians, nurse practitioners, family
physicians, obstetricians/gynecologists,
and internists. The data show the
proportion of primary care providers
who provided vaccination to
81–100 percent of their patients. The
Prevention in Primary Care Study
(PPCS) was conducted in 1997–98 to
update data from the PCPS. The design
and items included in the 1997–98 study
were similar to the PCPS, but a slightly
different sampling frame was used and
some items included in the 1992 surveys
were not included in the PPCS. The
providers were sampled from listings of
all licensed, active practitioners in the
United States whose practices were at
least 50 percent primary care. Because
of low response rates from the other
provider groups, updates are available
only for nurse practitioners.
Baseline data for objective 20.17 is
based on data from the local health
departments collected by the National
Profile of Local Health Departments,
NACCHO. Local health department
refers to any local component of the
public health system, defined as an
administrative and service unit of local
or State government concerned with
health and carrying some responsibility
for the health of a jurisdiction smaller
than a State.
Healthy People 2000 Final Review 283
Data Comparability
Data sources on vaccination
coverage levels have changed over the
years. The childhood vaccination
baseline data (20.11) were obtained from
the 1985 United States Immunization
Survey (USIS) and show the range of
antigen-specific vaccination levels at the
time of the interview among children
24–35 months of age. From 1991 to
1994, the source of the data was the
National Health Interview Survey
(NHIS) and the age included in the data
set was expanded to children 19–35
months of age. In 1992, the NHIS
questions on childhood immunizations
were modified. Therefore, the 1991 data
are not directly comparable to data for
subsequent years. The 1992 data are
now considered the baseline data for
estimates from the NHIS. The 1994
NHIS data have been provider-verified
and adjusted. Providers were contacted
and asked to provide vaccination
information for each child in the sample.
One of the limitations of the NHIS
is that it provides only national
estimates. In contrast, the National
Immunization Survey (NIS) provides
comparable national, State, and local
vaccination coverage estimates.
Therefore, since 1995, vaccination
coverage data have been obtained from
the NIS. The NIS, first fielded in 1994,
is an ongoing survey that provides the
first population-based State and urban
area-specific estimates of vaccination
coverage by a standard methodology for
the United States for children 19–35
months of age.
For influenza and pneumococcal
vaccination (20.11), baseline data for the
noninstitutionalized population were
obtained from the NHIS starting in
1989; for the institutionalized
population, national estimates were first
available from the National Nursing
Home Survey (NNHS) in 1995. NHIS
estimates are based on self-report (not
provider-verified), while the NNHS
estimates are based on records available
to nursing home staff completing the
survey, and may be incomplete.
The baseline data for objective
20.11 (hepatitis immunizations among
occupationally exposed workers) were
collected by OSHA’s Regulatory Impact
Analysis; the updates are from CDC’s
National Center for Infectious Diseases.
References
1. Pinner RW, Teutsch SM, Simonsen L, et al.
Trends in infectious diseases mortality in the
United States. JAMA 1996; 275:189–93.
2. Centers for Disease Control and Prevention.
Reported Vaccine-Preventable Diseases—United
States, 1993, and the Childhood Immunization
Initiative. MMWR 43(4):57–60. 1994.
3. National Vaccine Advisory Committee. Adult
Immunization. Atlanta, GA: Centers for Disease
Control and Prevention, National Immunization
Program. Jan. 1994.
4. Simonson L, Schonberger LB, Stroup DF, et al.
The impact of influenza on mortality in the USA.
In: Brown LE, Hampson AW, Webster RB, eds.
Options for the control of influenza III.
Amsterdam, The Netherlands: Elsevier Science
BV, 26–33. 1996.
5. Lui K-J, Kendal AP. Impact of influenza
epidemics on mortality in the United States from
October 1972 to May 1985. Am J Public Health
77:712–6. 1987.
6. Simonsen L, Fukuda K, Schonberger LB, et al.
The impact of influenza epidemics on
hospitalizations. J Infect Dis 2000;181:831–7.
7. Centers for Disease Control and Prevention.
Influenza and pneumococcal vaccination levels
among adults aged greater than or equal to 65
years, United States. MMWR 47(38):797–802.
1998.
8. Alter ML, Kruszon-Moran D, Nainan O, et al.
Prevalence of hepatitis C virus infection in the
United States: Results of the third National Health
and Nutrition Examination Survey (NHANES III)
1988–1994. N Engl J Med. 341:556–62. 1999.
9. Ryan C, Schober S, Turczyn K. Operational
definitions for year 2000 objectives: Priority area
20, immunization and infectious diseases. Healthy
people statistical notes; no 11. Hyattsville,
Maryland: National Center for Health Statistics.
1997.
284 Healthy People 2000 Final Review

Table 20. Immunization and Infectious Diseases objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
20.1 Vaccine-preventable diseases (number
of cases)
Diphtheria among people 25 years and
under .........................
1988 1 223020031 00
Tetanus among people 25 years and
under .........................
1988 3 647455399 50
Polio (wild-type virus) .............. 1988 0 000000000 00
Measles ........................ 1988
§
3,396 26,527 9,411 2,237 312 963 309 508 138 100 100 0
Rubella......................... 1988 225 1,125 1,401 160 192 227 128 238 181 364 267 0
Congenital Rubella Syndrome ........ 1988 6 11 47 11 576457 60
Mumps ......................... 1988 4,866 5,292 4,264 2,572 1,692 1,537 906 751 683 666 387 500
Pertussis........................ 1988 3,450 4,570 2,719 4,083 6,586 4,617 5,137 7,796 6,564 7,405 7,298 1,000
20.2 Epidemic-related pneumonia and
influenza deaths among people
65 years and over (per 100,000) .... 1979-87
1
19.9
2
22.6
3
18.6
4
20.0
5
15.7
6
21.0
7
19.2
8
17.3
9
15.8 - - - - - - 15.9
20.3* Viral hepatitis cases (per 100,000)
Hepatitis B ...................... 1987 63.5 50.6 42.6 37.7 30.9 28.7 25.0 23.9 23.6 22.5 16.9 40.0
Hepatitis A ...................... 1987 33.0 37.9 29.0 27.2 28.2 30.9 36.4 31.1 33.9 25.8 18.8 16.1
Hepatitis C ...................... 1987 18.3 13.1 8.3 5.6 4.4 4.1 2.7 2.9 2.4 2.4 - - - 13.7
Hepatitis B (number of cases)
a. Injecting drug users ............ 1987 44,348 17,615 12,666 10,576 15,136 14,180 10,216 9,199 11,132 11,506
p
8,630 7,932
b. Heterosexually active people ..... 1987 33,995 33,971 43,795 46,152 26,289 25,375 19,831 25,659 23,436 21,800
p
16,351 22,663
c. Men who have sex with men...... 1987 13,598 13,840 14,598 6,730 9,560 9,702 9,615 11,135 11,132 10,900
p
8,176 4,568
d. Children of Asians/Pacific Islanders. 1987 10,817 8,807 7,514 6,730 5,576 5,224 4,207 4,440 4,347 4,281 3,211 1,500
e. Occupationally exposed workers . . . 1987 3,090 1,258 2,576 1,923 727 506 407 391 383 377 243 623
f. Infants (chronic infections) ....... 1987 6,012 3,003 2,235 2,464 2,464 1,682 1,682 1,682 1,046 1,046 1,046 1,111
g. Alaska Native (number of new
carriers)..................... 1987 15 15 15 15 100000 01
Hepatitis B (cases per 100,000)
h. Black ....................... 1992 52.8 . . . . . . . . . 57.0 52.3 45.3 41.5 39.5 32.4 24.7 40
Hepatitis A (cases per 100,000)
i. Hispanic ..................... 1992 53.8 . . . . . . . . . 50.6 61.9 44.9 62.9 72.6 41.0 37.9
§
26.9
j. American Indian/Alaska Native..... 1992 256.0 . . . . . . . . . 192.7 363.7 240.7 142.3 69.4 30.0 19.7 128
Hepatitis C (cases per 100,000)
k. Hispanic ..................... 1992 17.2 . . . . . . . . . 11.1 6.7 3.9 7.7
10
---
10
---
10
---
§
13.7
20.4 Tuberculosis new cases (per 100,000) 1988 9.1 10.3 10.4 10.5 9.8 9.4 8.7 8.0 7.4 6.8 6.4 3.5
a. Asians/Pacific Islanders ......... 1988 36.3 41.6 41.8 46.6 44.5 45.3 45.9 41.6 40.6 36.6 35.3 15.0
b. Black ....................... 1988 28.3 33.0 31.9 31.7 29.1 26.8 23.9 22.3 20.5 17.8 16.8 10.0
c. Hispanic ..................... 1988 18.3 21.4 22.8 22.4 20.6 19.5 18.0 16.0 14.4 13.6 12.4 5.0
d. American Indian/Alaska Native .... 1988 18.1 18.9 16.3 16.3 14.6 17.4 16.5 14.5 13.4 12.6 11.8 5.0
20.5 Surgical wound and nosocomial
infections
See footnotes and key at end of table.
Healthy People 2000 Final Review 285

Table 20. Immunization and Infectious Diseases objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Device-associated nosocomial infection
rates in ICU patients (per 1,000
device-days)
Bloodstream Infections
Medical/coronary ICUs .......... 1986-90 6.9 . . . - - - 6.5 5.9 5.4 5.7 5.7 5.9 5.8
p
5.0 6.2
Surgical/medical-surgical ICUs .... 1986-90 5.3 . . . - - - 5.8 4.9 4.6 4.3 4.9 5.4 5.7
p
5.0 4.8
Pediatric ICUs ................ 1986-90 11.4 . . . - - - 7.9 8.3 8.0 7.8 7.5 7.8 7.8
p
6.4 10.3
Urinary Tract Infections
Medical/coronary ICUs .......... 1986-90 10.7 . . . - - - 10.1 8.5 7.9 7.3 6.9 6.7 6.4
p
5.7 9.6
Surgical/medical-surgical ICUs .... 1986-90 7.6 . . . - - - 6.3 5.9 5.8 4.9 5.1 4.9 5.2
p
5.4 6.8
Pediatric ICUs ................ 1986-90 5.8 . . . - - - 5.4 5.1 5.2 5.9 4.9 4.9 5.3
p
4.1 5.2
Pneumonia
Medical/coronary ICUs .......... 1986-90 12.8 . . . - - - 9.0 9.5 8.6 10.1 9.2 7.5 6.4
p
6.6 11.5
Surgical/medical-surgical ICUs .... 1986-90 17.6 . . . - - - 15.1 14.1 13.6 12.9 12.7 13.7 13.4
p
12.7 15.8
Pediatric ICUs ................ 1986-90 4.7 . . . - - - 6.7 5.8 5.7 5.5 5.3 5.5 5.3
p
3.9 4.2
Surgical wound infection rates (per
100 operations)
Low-risk patients .............. 1986-90 1.1 . . . - - - 1.2 1.2 1.1 1.2 1.0 1.6 1.7
p
1.4 1.0
Medium-low risk patients ........ 1986-90 3.2 . . . - - - 3.2 3.2 3.1 3.4 3.2 2.7 2.7
p
2.3 2.9
Medium-high risk patients........ 1986-90 6.3 . . . - - - 6.4 5.8 6.1 5.9 5.8 4.6 4.1
p
3.9 5.7
High-risk patients .............. 1986-90 14.4 . . . - - - 12.1 11.0 11.0 10.1 10.3 6.4 7.3
p
5.7 13.0
20.6 Illness among international travelers
(number of cases)
Typhoid fever .................... 1987 280 386 351 299 308 309 258 352 279 306 277 140
Hepatitis A ...................... 1987 4,475 3,962 3,814 3,814 4,581 6,602 7,815 6,331 6,575 6,411 4,112 1,119
Malaria ......................... 1987 932 1,098 1,046 910 1,275 1,014 1,167 1,392 1,544 1,225 1,065 750
20.7 Bacterial meningitis cases (per
100,000) ....................... 1986 6.5 - - - - - - - - - - - - - - - 1.9 2.3 2.1 1.9 2.2 4.7
a. Alaska Native ................. 1987 33 22 17 6.5 7.4 6.2 3.1 6.0 6.9 6.7
11
5.7 8
20.8 Infectious diarrhea among children in
child care centers
Children 0–5 years ................ 1991 32% . . . . . . - - - - - - - - - - - - - - - - - - - - - - - - 24%
Children 0–3 years ................ 1991 38% . . . . . . - - - - - - - - - - - - - - - - - - - - - - - - 28%
20.9 Ear infections among children 4 years
and under (restricted activity days
per 100 children)................. 1987 135.4 125.0 155.7 155.2 196.3 137.0 134.4 103.4 - - - - - - - - - 105.0
20.10 Pneumonia-related restricted activity
days (per 100 people)
People 65 years and over ........... 1987 19.1 46.2 78.5 63.5 45.1 71.3 58.8 80.7 - - - - - - - - - 15.1
Children 4 years and under.......... 1987 29.4 51.3
12
24.1
12
19.4
12
22.5 39.5
12
23.2
12
17.1 - - - - - - - - - 24.0
20.11 Immunization (percent immunized)
Basic immunization series among
children
See footnotes and key at end of table.
286 Healthy People 2000 Final Review

Table 20. Immunization and Infectious Diseases objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Children 2 years and under ........ 1985
13,14
54–
65% --- --- --- --- --- --- --- --- --- --- 90%
Children 19-35 months
Diphtheria-tetanus-pertussis (3 or
more doses)................. ... --- --- 69% 83% 88% 90% 95% 95% 95% 96% 96% ...
Polio (3 or more doses) ......... ... --- --- 53% 72% 79% 79% 88% 91% 91% 91% 90% ...
Measles-containing ............ ... --- --- 82% 83% 84% 90% 90% 91% 91% 92% 92% ...
Haemophilus influenzae type b (3 or
more doses)................. ... --- --- 2% 28% 55% 75% 92% 92% 93% 93% 94% ...
Hepatitis B (3 or more doses). . . . . . - - - - - - - - - - - - 16% 34% 68% 82% 84% 87% 88% . . .
4DTP/3Polio/1MMR ............ ... --- --- --- 55% 67% 68% 76% 78% 78% 81% 80% ...
Children in licensed child care
facilities
13,14
................... 1987-88
15
94–
95%
16
94–
96%
17
94–
96%
18
94–
96%
19
95–
98%
20
97–
98%
21
98–
99%
22
95%
23
93–
95%
24
93–
96%
25
93–
95% 95%
Children in kindergarten through
postsecondary education
institutions
13,14
................. 1987-88
15
97–
98%
16
97–
98%
17
96–
98%
18
96–
98%
19
92–
94%
20
93–
94%
21
94–
95%
22
98–
99%
23
95–
98%
24
95–
97%
25
96–
99% 95%
Hepatitis B immunizations
Infants of antigen-positive mothers. . . 1991 40% . . . . . . 71% 71% 78% 78% 79% 82% 81% - - - 90%
Occupationally exposed workers
26
. . . 1989 37% - - - - - - 50% - - - 67% - - - - - - - - - - - - - - - 90%
Injecting drug users in drug treatment
programs..................... ... --- --- --- --- --- --- --- --- --- --- --- 50%
Men who have sex with men ....... 1992-93 3% . . . . . . . . . . . . - - - - - - - - - - - -
27
9% - - - 50%
Pneumococcal immunizations
Institutionalized chronically ill people
or older people ................ 1995
28
22% ... ... ... ... ... ... ---
29
28% --- --- 80%
Noninstitutionalized people 65 years
and over ..................... 1989
§
15% - - - 21% - - - 28% 30% 34% - - - 43% 46% - - - 60%
a. Black 65 years and over ..... 1989
§
6% - - - 14% - - - 14% 15% 23% - - - 22% 26% - - - 60%
b. Hispanic 65 years and over . . . 1989
§
11% - - - 12% - - - 12% 14% 23% - - - 23% 23% - - - 60%
Influenza immunizations (in last 12
months)
Institutionalized chronically ill people
or older people............... 1995
30
61% ... ... ... ... ... ... ---
31
64% --- --- 80%
Noninstitutionalized people
65 years and over ............ 1989
§
33% - - - 42% - - - 52% 55% 58% - - - 63% 63% - - - 60%
a. Black 65 years and over ..... 1989
§
20% - - - 27% - - - 33% 39% 40% - - - 45% 46% - - - 60%
b. Hispanic 65 years and over . . . 1989
§
28% - - - - - - - - - 47% 38% 50% - - - 53% 50% - - - 60%
20.12 Postexposure rabies treatments
(number)....................... 1987 18,000 - - - 18,800 24,700 25,000– 22,000– - - - 16,000– 20,000– - - - - - - 9,000
43,000 43,000 39,000 40,000
20.13 Immunization laws (number of
States)
32
....................... 1989 10-49 - - - - - - 34–50 - - - - - -
33
42–50 - - -
34
44–50 - - - 44–50 50
20.14 Provision of immunizations by
clinicians ...................... ... --- --- --- --- --- --- --- --- --- --- --- 90%
See footnotes and key at end of table.
Healthy People 2000 Final Review 287

Table 20. Immunization and Infectious Diseases objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Percent of clinicians routinely providing
service to 81-100% of patients
Children:
DTP vaccination
Pediatricians ................... 1992 86% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Nurse practitioners .............. 1992 76% . . . . . . . . . - - - - - - - - - - - - - - -
35
78% - - - 90%
Family physicians ............... 1992 89% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Oral polio vaccination
Pediatricians ................... 1992 87% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Nurse practitioners .............. 1992 76% . . . . . . . . . - - - - - - - - - - - - - - -
35
79% - - - 90%
Family physicians ............... 1992 89% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Tetanus-diphtheria booster (under
18 years)
Pediatricians ................... 1992 79% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Nurse practitioners .............. 1992 71% . . . . . . . . . - - - - - - - - - - - - - - -
35
72% - - - 90%
Family physicians ............... 1992 70% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Hib vaccination
Pediatricians ................... 1992 85% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Nurse practitioners .............. 1992 68% . . . . . . . . . - - - - - - - - - - - - - - -
35
74% - - - 90%
Family physicians ............... 1992 74% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Tetanus-diphtheria booster (18 years
and over)
Nurse practitioners .............. 1992 38% . . . . . . . . . - - - - - - - - - - - - - - -
35
40% - - - 90%
Obstetricians/gynecologists ........ 1992 4% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Internists ...................... 1992 29% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Family physicians ............... 1992 28% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Influenza vaccination (65 years and
over)
Nurse practitioners .............. 1992 42% . . . . . . . . . - - - - - - - - - - - - - - -
35
47% - - - 90%
Obstetricians/gynecologists ........ 1992 6% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Internists ...................... 1992 49% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Family physicians ............... 1992 31% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Pneumococcal vaccination (65 years
and over)
Nurse practitioners .............. 1992 33% . . . . . . . . . - - - - - - - - - - - - - - -
35
43% - - - 90%
Obstetricians/gynecologists ........ 1992 5% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Internists ...................... 1992 40% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
Family physicians ............... 1992 25% . . . . . . . . . - - - - - - - - - - - - - - -
35,36
--- --- 90%
20.15 Financial barriers to immunization
Employment-based insurance plans that
provide coverage for immunizations
Conventional insurance plans ...... 1989 45% 47% - - - 53% - - - - - - - - - - - - - - - - - - - - - 100%
See footnotes and key at end of table.
288 Healthy People 2000 Final Review

Table 20. Immunization and Infectious Diseases objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Preferred Provider Organization
plans ........................ 1989 62% 65% - - - 65% - - - - - - - - - - - - - - - - - - - - - 100%
Health Maintenance Organization
plans ........................ 1989 98% 98% - - - 95% - - - - - - - - - - - - - - - - - - - - - 100%
20.16
Public health department provision of
immunizations
Pneumococcal vaccine............. 1990 37% . . . - - - - - -
33
48% --- --- --- --- --- --- 90%
Influenza vaccine ................. 1990 60% . . . - - - - - -
33
91% --- --- --- --- --- --- 90%
Tetanus/Diphtheria vaccine .......... 1990 70% . . . - - - - - - - - - - - - - - - - - - - - - - - - - - - 90%
Tetanus ....................... ... --- --- --- ---
33
85% --- --- --- --- --- --- 90%
Diphtheria ..................... ... --- --- --- ---
33
77% --- --- --- --- --- --- 90%
Hepatitis B vaccine ................ 1992-93 77% . . . . . . . . . . . . - - - - - - - - - - - - - - - - - - 90%
20.17
Local health department programs
that identify tuberculosis cases .... 1992-93 80% . . . . . . . . . . . . - - - - - - - - - - - - - - - - - - 90%
20.18
Preventive therapy for tuberculosis
(percent of infected people completing
therapy) ....................... 1987 66.3% 63.0% 64.9% 66.3% 65.3% 65% 65.4% 65.2% 62.2% 64.6% - - - 85%
20.19
Laboratory capability for influenza
diagnosis
Tertiary care hospital laboratories . . . 1993 52% . . . . . . . . . . . . - - - 57% - - - 58% - - -
p
64% 85%
Secondary care hospital
laboratories ................... 1993 45% . . . . . . . . . . . . - - - 46% - - - 46% - - -
p
51% 50%
Health maintenance organization
laboratories ................... 1993
§
68% ... ... ... ... --- 56% --- 68% ---
p
67% 50%
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline and/or target have been revised.
p
Preliminary data.
1
1979–80 influenza season through 1986-87 influenza season.
2
1987–88 influenza season through 1989-90 influenza season.
3
1988–89 influenza season through 1990-91 influenza season.
4
1989–90 influenza season through 1991-92 influenza season.
5
1990–91 influenza season through 1992-93 influenza season.
6
1991–92 influenza season through 1993-94 influenza season.
7
1992–93 influenza season through 1994-95 influenza season.
8
1993–94 influenza season through 1995-96 influenza season.
9
1994–95 influenza season through 1996-97 influenza season.
10
Data are unreliable. Number of cases is too small to make reliable estimates.
11
Data are based on cases of Haemophilus influenzae type b and Streptococcus pneumoniae only.
12
Data are unreliable. Numerator has a relative standard error of more than 30%.
13
Range of antigen-specific immunization levels.
14
Three or more doses for DTP and polio.
15
1987-88 school year.
16
1989-90 school year.
17
1990-91 school year
18
1991-92 school year.
19
1992-93 school year.
20
1993-94 school year.
Met
Toward
Mixed/ no change
Away
Cannot assess
Healthy People 2000 Final Review 289

21
1994-95 school year.
22
1995-96 school year.
23
1996-97 school year.
24
1997-98 school year.
25
1998-99 school year.
26
Health care workers only.
27
1994–98 data.
28
43% of nursing home residents surveyed in 1995 had unknown pneumococcal vaccination status and were counted as unvaccinated.
29
44% of nursing home residents surveyed in 1997 had unknown pneumococcal vaccination status and were counted as unvaccinated.
30
21% of nursing home residents surveyed in 1995 had unknown influenza vaccination status and were counted as unvaccinated.
31
22% of nursing home residents surveyed in 1997 had unknown influenza vaccination status and were counted as unvaccinated.
32
Data represent the range in the number of States with laws on each of the nine antigen types (diphtheria, tetanus, pertussis, measles, mumps, rubella, polio, and Haemophilus) and by the
two types of facilities (schools with grades K-12 and day care centers). Hepatitis B vaccine in schools with grades K-12 and day care centers are not included in the range. The number of
States with Hepatitis B immunization laws for grades K-12 was 2 in 1994–95 and 9 in 1998-99.
33
1994-95 data.
34
1996-97 data.
35
1997-98 data.
36
Response rate for this group was too low to produce reliable estimates.
NOTES: Data include revisions and, therefore, may differ from those previously published in these reports and other publications. ICU is intensive care unit. DTP is diphtheria-tetanus-pertusis.
MMR is measles-mumps, rubella. Hib is Haemophilus influenzae type b.
Objective number Data source
20.1 National Notifiable Disease Surveillance System, CDC, EPO.
20.2 CDC, NCID.
National Vital Statistics System, CDC, NCHS.
20.3*, 20.3a-c, i-k National Notifiable Disease Surveillance System, CDC, EPO.
Sentinel Counties Study of Acute Viral Hepatitis, Viral Hepatitis Surveillance Program, CDC, NCID.
20.3d CDC, NCID.
20.3e National Notifiable Disease Surveillance System, CDC, EPO.
Sentinel Counties Study of Acute Viral Hepatitis, Viral Hepatitis Surveillance Program, CDC, NCID.
20.3f National Notifiable Disease Surveillance System, CDC, EPO.
Margolis, HS. Estimates and reported cases of hepatitis B infection and its sequelae in Alaskan Natives.
Lancet (1987) 2: 1134-6.
20.3g Alaskan Registry, IHS.
20.3h National Notifiable Disease Surveillance System, CDC, EPO.
20.4, 20.4a-d Tuberculosis Morbidity Data, CDC, NCHSTP.
20.5 National Nosocomial Infection Surveillance System, CDC, NCID.
20.6 Typhoid Surveillance System, CDC, NCID.
Sentinel Counties Study of Acute Viral Hepatitis, Viral Hepatitis Surveillance Program, CDC, NCID; National Notifiable Disease Surveillance System, CDC,
EPO.
Malaria Surveillance System, CDC, NCID.
20.7 Bacterial Meningitis Surveillance System, CDC, NCID.
20.7a Arctic Investigations Laboratory, CDC, NCID.
20.8 National Health Interview Survey, CDC, NCHS.
20.9 National Health Interview Survey, CDC, NCHS.
20.10 National Health Interview Survey, CDC, NCHS.
290 Healthy People 2000 Final Review

Objective number Data source
20.11 Basic immunization series among children:
Baseline for children 2 years and under: United States Immunization Survey, CDC, NCHSTP.
Children 19–35 months: 1991–1994 data: National Health Interview Survey, CDC, NCHS.
1995–96 data: National Immunization Survey, CDC, NIP.
Immunizations among children in licensed child care facilities and in schools: State Immunization Survey, CDC, NCHSTP.
Hepatitis B immunizations among infants of antigen-positive women:
Perinatal Hepatitis B Prevention Program, CDC, NIP.
Hepatitis B immunizations among occupationally exposed workers:
Baseline: Regulatory Impact Analysis of OSHA Final Rule on Occupational Exposure to Bloodborne Pathogens, DOL, OSHA, ORA.
Updates: CDC, NCID.
Hepatitis B immunizations among men who have sex with men:
Baseline: Young Men’s Survey, San Francisco Department of Public Health. MMWR Vol. 45 No.10: March 15, 1996.
Update: Young Men’s Survey, CDC, NCHSTP.
Pneumococcal and influenza immunizations among noninstitutionalized people:
National Health Interview Survey, CDC, NCHS.
Pneumococcal and influenza immunizations among institutionalized people:
National Nursing Home Survey, CDC, NCHS.
20.11a, b National Health Interview Survey, CDC, NCHS.
20.12 Rabies Vaccine and Immune Globulin Manufacturers Sales Data, CDC, NCID.
20.13 Survey of Immunization Laws, CDC, NIP.
20.14 Baseline: Primary Care Provider Surveys, OPHS, ODPHP.
Update: Prevention in Primary Care Study, American College of Preventive Medicine.
20.15 Health Insurance Association of America Employer Survey, Health
Insurance Association of America.
20.16 Baseline: Immunization Grant Program Profiles, CDC, NCPS.
Update: National Profile of Local Health Departments, National Association of County and City Health Officials.
20.17 National Profile of Local Health Departments, National Association of County and City Health Officials.
20.18 Tuberculosis Program Management Report Data on Completion of Preventive Therapy, CDC, NCHSTP.
20.19 Survey of Laboratories using Rapid Viral Diagnosis of Influenza, CDC, NCID.
* Duplicate objective. See full text of objective following this table.
Healthy People 2000 Final Review 291
Immunization and
Infectious Diseases
Objectives
20.1: Reduce indigenous cases of
vaccine-preventable diseases as follows:
Disease 2000 target
Diphtheria among people
aged 25 and younger 0
Tetanus among people
aged 25 and younger 0
Polio (wild-type virus) 0
Measles (indigenous) 0
Rubella 0
Congenital Rubella
Syndrome 0
Mumps 500
Pertussis 1,000
20.2: Reduce epidemic-related
pneumonia and influenza deaths among
people aged 65 and older to no more
than 15.9 per 100,000 people.
20.3*: Reduce viral hepatitis as follows:
Hepatitis B: 40 per 100,000 people
Hepatitis A: 16.1 per 100,000 people
Hepatitis C: 13.7 cases per 100,000
people
20.3a: Reduce hepatitis B among
injecting drug users to no more than
7,932 cases.
20.3b*: Reduce hepatitis B among
heterosexually active people to no
more than 22,663 cases.
Duplicate objective: 19.7
20.3c*: Reduce hepatitis B among
men who have sex with men to no
more than 4,568 cases.
Duplicate objective: 19.7
20.3d: Reduce hepatitis B among
children of Asian and Pacific
Islanders to no more than 1,500
cases.
20.3e*: Reduce hepatitis B among
occupationally exposed workers to
no more than 623 cases.
Duplicate objective: 10.5
20.3f: Reduce hepatitis B among
infants to no more than 1,111
chronic infections.
20.3g: Reduce hepatitis B among
Alaska Natives to no more than 1
new chronic infection.
20.3h: Reduce hepatitis B among
blacks to no more than 40 cases per
100,000 people.
20.3i: Reduce hepatitis A among
Hispanics to no more than 26.9
cases per 100,000 people.
20.3j: Reduce hepatitis A among
American Indians and Alaska
Natives to no more than 128 cases
per 100,000 people.
20.3k: Reduce hepatitis C among
Hispanics to no more than 13.7
cases per 100,000 people.
20.4: Reduce tuberculosis to an
incidence of no more than 3.5 cases per
100,000 people.
20.4a: Reduce tuberculosis among
Asians and Pacific Islanders to an
incidence of no more than 15 cases
per 100,000 people.
20.4b: Reduce tuberculosis among
blacks to an incidence of no more
than 10 cases per 100,000 people.
20.4c: Reduce tuberculosis among
Hispanics to an incidence of no
more than 5 cases per 100,000
people.
20.4d: Reduce tuberculosis among
American Indians and Alaska
Natives to an incidence of no more
than 5 cases per 100,000 people.
20.5: Reduce by at least 10 percent the
incidence of surgical wound infections
and nosocomial infections in intensive
care patients.
20.6: Reduce selected illness among
international travelers, as follows:
Typhoid fever: 140 cases
Hepatitis A: 1,119 cases
Malaria: 750 cases
20.7: Reduce bacterial meningitis to no
more than 4.7 cases per 100,000 people.
20.7a: Reduce bacterial meningitis
among Alaska Natives to no more
than 8 cases per 100,000 people.
20.8: Reduce infectious diarrhea by at
least 25 percent among children in
licensed child care centers and children
in programs that provide an
Individualized Education Program (IEP)
or Individualized Health Plan (IHP).
20.9: Reduce acute middle ear infections
among children aged 4 and younger, as
measured by days of restricted activity
or school absenteeism, to no more than
105 days per 100 children.
20.10: Reduce pneumonia-related days
of restricted activity as follows:
15.1 days per 100 people aged 65
and older
24 days per 100 children aged 4
and younger
20.11: Increase immunization levels as
follows:
Basic immunization series among
children under age 2: at least 90 percent.
Basic immunization series among
children in licensed child care facilities
and kindergarten through post-secondary
education institutions: at least
95 percent.
Hepatitis B immunization among
high-risk populations, including infants
of hepatitis B surface antigen-positive
mothers to at least 90 percent;
occupationally exposed workers to at
least 90 percent; injecting drug users in
drug treatment programs to at least
50 percent; and men who have sex with
men to at least 50 percent.
Pneumococcal pneumonia and influenza
immunization among institutionalized
chronically ill or older people: at least
80 percent.
Pneumococcal pneumonia and influenza
immunization among
noninstitutionalized, high-risk
populations, as defined by the
Immunization Practices Advisory
Committee: at least 60 percent.
Duplicate objective for occupationally
exposed workers: 10.9
20.11a: Increase pneumococcal
pneumonia and influenza
immunization among blacks aged
65 years and older to 60 percent.
20.11b: Increase pneumococcal
pneumonia and influenza
immunization among Hispanics
aged 65 years and older to
60 percent.
20.12: Reduce postexposure rabies
treatments to no more than 9,000 per
year.
20.13: Expand immunization laws for
schools, preschools, and day care
settings to all States for all antigens.
20.14: Increase to at least 90 percent the
proportion of primary care providers
who provide information and counseling
292 Healthy People 2000 Final Review
about immunizations and offer
immunizations as appropriate for their
patients.
20.15: Improve the financing and
delivery of immunizations for children
and adults so that virtually no American
has a financial barrier to receiving
recommended immunizations.
20.16: Increase to at least 90 percent the
proportion of public health departments
that provide adult immunization for
influenza, pneumococcal disease,
hepatitis B, tetanus, and diphtheria.
20.17: Increase to at least 90 percent the
proportion of local health departments
that have ongoing programs for actively
identifying cases of tuberculosis and
latent infection in populations at high
risk for tuberculosis.
20.18: Increase to at least 85 percent the
proportion of people found to have
tuberculosis infection who completed
courses of preventive therapy.
20.19: Increase to at least 85 percent the
proportion of tertiary care hospital
laboratories and to at least 50 percent
the proportion of secondary care
hospital and health maintenance
organization laboratories possessing
technologies for rapid viral diagnosis of
influenza.
*Duplicate objective.
Healthy People 2000 Final Review 293

Priority Area 21
Clinical Preventive
Services
Background
Achieving access to preventive
services for all Americans was one of
three overarching goals of Healthy
People 2000. The Clinical Preventive
Services priority area directly addresses
this goal and covers a range of
recommended screening services
delivered to individuals in a health care
setting. The services include
immunizations, screening for the early
detection of disease or risk factors,
clinician counseling about modifiable
risk factors such as smoking and
physical activity, and use of medication
to prevent disease in healthy individuals.
Clinical preventive services are not only
an essential component of national
efforts to reduce disease and disability,
but also are an important barometer of
the effectiveness and functioning of the
Nation’s health care system. Failure to
receive recommended, needed
immunizations or screening tests often
reflects fundamental problems in access
to essential health care services or in the
quality of available care. The objectives
in this priority area acknowledge the
importance of specific barriers in the
Nation’s efforts to improve the
preventive care and the overall health of
our population. Lack of health insurance
remains a fundamental problem for a
substantial number of persons, and
uninsured patients are less likely to get
needed services and more likely to
suffer poor health. Having insurance,
however, is not sufficient to ensure that
individuals get the preventive care they
need. Having a regular source of care
may be an equally important factor for
prevention. Promoting regular
preventive care will require steps to
ensure an adequate number of health
care providers, especially for
underserved rural and urban areas, along
with efforts to expand health care
coverage.
Health insurance, access to care,
and receipt of preventive care are
especially a problem in racial and ethnic
minority populations (1), as well as
other unserved or underserved
populations. One important step toward
improving care for these populations is
to improve their representation among
those individuals entering the health
professions.
Over the last decade, there has been
a growing recognition among clinicians,
patients, and policymakers of the
importance of preventive services as a
part of primary health care. Much of
this awareness can be attributed to the
increasing evidence of the effectiveness
of clinical preventive services in
improving health outcomes and the cost-
effectiveness of a range of identified
services, including immunizations,
screening for cancer and cardiovascular
risk factors, and counseling about
smoking and other important behavioral
risk factors (2,3).
Data Summary
Highlights
Substantial progress has been made
in the delivery of effective preventive
care to Americans. The average number
of years of healthy life for the total
population (21.1) increased from a
baseline of 64 years in 1990 to 65.2
years in 1998, exceeding the Healthy
People 2000 target. Among special
populations targeted by the objective,
the Hispanics have met the year 2000
target, while progress was made for
blacks and older adults. Receipt of
individual preventive services (21.2)
increased steadily across a variety of
service and patient populations, and
reached the year 2000 targets for several
in 1998. Of the five universally
recommended childhood immunizations,
four had rates that exceeded the year
2000 target of 90 percent and narrowly
missed the target for the fifth
immunization series, hepatitis B, with a
rate of 88 percent. Receipt of
mammograms and clinical breast exams
in the past 2 years was reported by
64 percent of women over 50 years of
age, up from 51 percent in 1992, and
63 percent of adults over 65 years of
age received an influenza immunization.
By 1998, additional services made
important progress toward the year 2000
targets: rates of pneumococcal
immunization in older persons more
than doubled to 46 percent, rates of
recent tetanus booster, cholesterol
screening, and Pap test increased to
57 percent, 67 percent, and 79 percent,
respectively.
The proportion of the adult
population with a usual source of care
increased from 80 percent in 1991 to
85 percent in 1998 (21.3). Gains were
evident in all targeted racial and ethnic
groups and among persons of low
income.
Little progress was made in rates of
insurance coverage (21.4), although
promising recent trends are evident for
children under 18 years of age. Overall,
the proportion of adults under 65 years
of age without health care coverage
increased slightly from 15.7 percent in
1989 to 16.6 percent in 1998. Rates for
uninsured persons among Hispanics
under 65 years of age (34 percent) and
those below the poverty level
(33 percent) were close to double that of
the general population. Similar
disparities exist in access to a usual
source of care.
Summary of Progress
Data are available to assess the
progress for six of the eight objectives
for Clinical Preventive Services. One
objective (21.1) met its year 2000 target.
Two objectives (21.3 and 21.8) moved
toward the year 2000 targets. One
objective (21.4) moved away from its
target. Progress was mixed for two
objectives (21.2 and 21.6). For objective
21.6, the assessment of progress is
based on nurse-practitioner data only.
Data beyond baseline are not available
for two objectives (21.5 and 21.7). See
table 21 for the tracking data for the
objectives in this priority area and
figure 21 for a quantitative assessment
of progress.
Discussion
Important progress has been made
in delivering effective preventive
services to persons in the United States.
This is reflected in generally increasing
delivery rates for individual services,
better coverage of preventive services
by public and private insurers, and
growing attention to prevention within
the clinical community and the public.
At the same time, substantial barriers
remain. While insurance coverage of
preventive services has increased
significantly, a substantial and steady
number of persons have no health care
coverage and/or no usual source of care.
One isolated pocket of recent progress,
however, is insurance for children. Over
the past 2 years, the Child Health
Insurance Program, through Federal
support to States, has reduced the
294 Healthy People 2000 Final Review

21.1 Years of healthy life
21.2 Receipt of recommended services Children
19-35 months:Basic immunization series, 3DTP
3Polio
1MMR
3Hib
3HepB
4DTP/3Polio/1MMR
Adults: Routine checkup, 18-64 years
Cholesterol screened in past 5 years, 18 years and over
Cholesterol ever screened, 18 years and over
Cholesterol screened in past 2 years, 18 years and over
Tetanus booster, 18 years and over
Influenza vaccination, 65 years and over
Pneumococcal vaccination, 65 years and over
Pap test in past 3 years, 18 years and over
Breast exam and mammography in past 2 years,
50 years and over
Screening questions at routine checkup,
18 years and over
21.3 Usual source of care
21.4 No health insurance
21.8 Racial/ethnic representation in health
professions, degrees: Black
Hispanic
American Indian/Alaska Native
Enrolled in nursing school: Black
Hispanic
Asian or Pacific Islander
American Indian/Alaska Native
Moved away from target Moved toward target Met target
NOTES: Complete tracking data are shown in table 21. Progress quotients are not calculated for objectives 21.5, 21.6, and 21.7.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
DTP is diphtheria-tetanus-pertussis. Hib is
Haemophilus influenzae
type b vaccine. MMR is measles-mumps-rubella.
-12%
71%
12%
47%
75%
28%
50%
64%
46%
33%
33%
41%
70%
44%
52%
No change
Percent of target achieved
-100 -80 -60 -40 -20
0
20 40 60 80 100
97%
107%
117%
129%
100%
186%
120%
144%
100%
83%
Healthy People 2000 Final Review 295
Figure 21. Final status of Clinical Preventive Services objectives
proportion of children under 18 years of
age without insurance. Insurance
coverage for counseling services such as
smoking cessation continues to lag far
behind that for screening tests. Finally,
although several measures have been
shown consistently to improve
preventive care—reminder systems for
patients and clinicians, audit, and
feedback to clinicians, and reducing
financial barriers for patients
(2–5)—only a minority of clinicians
practice in health systems where these
measures can be easily implemented
through integrated data systems and
computerized medical records.
An important potential force for
change is the drive to make health plans
accountable for delivering quality care,
including appropriate preventive care.
The National Committee for Quality
Assurance (NCQA), through its Health
Plan Employer Data and Information Set
(HEDIS) (6), collects data on the
provision of a number of clinical
preventive services, including child and
adult immunizations, smoking cessation
advice, and breast and cervical cancer
screening (2,3). The expectation,
supported in part by preliminary data, is
that increasing accountability among
competing health plans will encourage
them to invest in improving preventive
care and in building the information
infrastructure that could facilitate further
gains (2,3,6). At the same time,
however, these efforts are impeded by
increasing turnover of patients within
plans, fragmentation of care, and
heightened economic pressures on the
health care system.
Transition to Healthy People
2010
The 1990s were marked by growing
attention to improving the quality of
health care and reducing medical errors
(7). Several issues emerged that will
demand the efforts of the public health
community over the next decade, for
example, monitoring and reporting on
health care quality (8). Other issues that
surfaced, particularly with respect to
increasing access to needed services,
include fostering community-based,
integrated systems of care that better
respond to the community’s needs and
supporting the development of a health
work force that reflects and responds to
the cultural diversity of populations
served.
To address these issues, Healthy
People 2010 focus area 1, Access to
Quality Health Services, expands on the
Healthy People 2000 priority area on
clinical preventive services. The revised
focus area aims to track access to
services for four important sectors of the
health care system—clinical preventive
care, primary care, emergency services,
and long-term care and rehabilitative
services.
In addition, some objectives
covering clinical preventive care have
been moved to other Healthy People
2010 focus areas: focus area 14,
Immunization and Infectious Diseases
includes objectives on childhood
immunizations, adult immunizations;
focus area 12, Heart Disease and Stroke
addresses cholesterol screening; and
focus area 3, Cancer covers women’s
receipt of breast exams and
mammograms and Pap tests as well as
primary care provider counseling about
mammograms and Pap tests.
Two objectives from the access to
quality health services focus area (health
insurance coverage and usual source of
care) are used to measure the Healthy
People 2010 Leading Health Indicator
on access to health care.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the differences between the
two decades of objectives, reflecting
new knowledge and directions in this
area.
Data Issues
Years of Healthy Life
Years of healthy life is a summary
measure of health that combines
mortality (quantity of life) and
morbidity and disability (quality of life)
into a single measure. The concept of
increasing the span of healthy life is one
of the three Healthy People 2000 goals
and a specific measure has been
developed to track this objective in three
priority areas (8.1, 17.1, and 21.1). The
data used to track the objective come
from the National Vital Statistics System
(mortality) and the National Health
Interview Survey (NHIS) (morbidity).
The NHIS was redesigned in 1997 and,
therefore, data for 1997–98 may not be
comparable with those from previous
years. See the Appendix for a discussion
of the changes to the NHIS. The
methodology for the Healthy People
2000 years of healthy life measure,
developed by NCHS and outside
consultants is published in Healthy
People Statistical Notes series (9).
Definitions
Operational definitions and data
collection specifications for all Healthy
People 2000 objectives in Priority Area
21 have been published in the National
Center for Health Statistics, Healthy
People Statistical Notes series (10). Data
issues are discussed and references are
cited for expanded discussions of the
data systems that provide data for the
national objectives. When appropriate,
the text of questionnaire items used to
measure the objectives is also provided.
See Appendix table VI for more
information.
Data to determine the level of
receipt of clinical preventive services
among adults (21.2) are obtained
through periodic supplements to the
National Health Interview Survey
(NHIS). The supplements provide
limited information on counseling, and
recommendations for high-risk groups
are not addressed. Respondents were
asked if they had been asked about at
least one behavior that indicates the
need for counseling at their last routine
checkup. A positive response was used
as an indication that the person had
received at least one recommended
counseling service.
For objective 21.7, a local health
department refers to any local
component of the public health system,
defined as an administrative and service
unit of local or State government
concerned with health and carrying
some responsibility for the health of a
jurisdiction smaller than a State.
Data Sources
Baseline data for objective 21.6
(provision of recommended services) are
from the Primary Care Provider Surveys
(PCPS). The sample was drawn from
the membership rolls of professional
organizations for pediatricians, nurse
practitioners, family physicians,
obstetricians/gynecologists, and
internists. Response rates varied from 50
to 80 percent across these groups. The
data on provision of recommended
services represent the proportion of
providers who report that they routinely
provided 81–100 percent of eligible
patients with the recommended services.
The Prevention in Primary Care Study
296 Healthy People 2000 Final Review
(PPCS) was conducted in 1997–98 to
update data from the PCPS. The design
and items included in the 1997–98 study
were similar to the PCPS, but a slightly
different sampling frame was used and
some items included in the 1992 surveys
were not included in the PPCS. The
professionals were sampled from listings
of all licensed, active practitioners in the
United States whose practices were at
least 50 percent primary care. Because
of low response rates from the other
provider groups, updates are available
only for nurse practitioners.
Data Comparability
Data on the proportion of people
who have a usual source of care (21.3)
are obtained from the NHIS. In 1991
and 1992, information on source of
primary care was received from one
adult randomly selected from among
household members. Beginning in 1993,
a knowledgeable adult respondent
provided information for all members of
the household.
Data on the proportion of people
under 65 years of age who do not have
health care coverage (21.4) are from the
NHIS. The 1989 baseline data and
tracking data from 1992 through 1994
are not directly comparable because of
questionnaire changes. Also, beginning
with 1995 data, persons receiving public
assistance other than Medicaid are
considered to have health care coverage.
Prior to 1995 they were considered to
not have health care coverage. In 1996,
0.4 percent of the population under 65
years of age were covered by public
assistance other than Medicaid.
Proxy Data
The proportion of the U.S.
population under 65 years of age
(age-adjusted to the 1970 U.S. civilian
noninstitutionalized population) that
does not have health care coverage
(private insurance, Medicare, Medicaid,
or a military plan) is used to measure
progress for objective 21.4, financial
barriers to receiving recommended
clinical preventive services. This is only
a partial measure. Many health
insurance plans do not provide full
coverage for preventive health care;
however, overall coverage for preventive
care services is improving. In 1997,
89 percent of employer-sponsored health
insurance plans covered periodic
physical examinations, 88 percent
covered well-baby care, and 92 percent
covered periodic gynecological
examinations (11).
Beginning with 1996 data, persons
receiving public assistance other than
Medicaid are considered to have health
care coverage. Prior to 1996, they were
considered to not have health care
coverage. In 1996, the age-adjusted
percent of the population under 65 years
of age covered by Medicaid was
11.3 percent, and 0.4 percent were
covered by other public assistance (12).
References
1. Weinick RM, Zuvekas SH, Drilea SK. Access
to health care—sources and barriers, 1996. MEPS
Research Findings No. 3. Rockville, Maryland:
Agency for Health Care Policy and Research.
1997.
2. U.S. Preventive Services Task Force. Guide to
clinical preventive services: An assessment of the
effectiveness of 169 interventions. Report of the
U.S. Preventive Services Task Force. Baltimore,
Maryland: Williams and Wilkins. 1989.
3. U.S. Preventive Services Task Force. Guide to
clinical preventive services, 2d ed. Baltimore,
Maryland: Williams and Wilkins. 1996.
4. Centers for Disease Control and Prevention.
Vaccine-preventable diseases: Improving
vaccination coverage in children, adolescents, and
adults. A report on recommendations of the Task
Force on Community Preventive Services.
MMWR 48(RR–8):1–16. 1999.
5. National Health Service, Center for Reviews
and Dissemination. Getting evidence into practice.
Effective Health Care 5(1). 1999.
6. National Committee on Quality Assurance.
Health Plan Employer Data and Information Set
(HEDIS 3.0). Washington: National Committee on
Quality Assurance. 1997.
7. Kohn L, Corrigan J, Donaldson M, eds.
Committee on Quality of Health Care in America.
To err is human: Building a safer health system.
Washington: Institute of Medicine. 1999.
8. President’s Advisory Commission on Consumer
Protection and Quality in the Health Care Industry.
Quality first: Better health care for all Americans:
Final report to the President of the United States.
Washington: U.S. Government Printing Office.
1998.
9. Erickson P, Wilson R, Shannon I. Years of
Healthy Life. Healthy people statistical notes no 7.
Hyattsville, Maryland: National Center for Health
Statistics. 1995.
10. Ryan C, Klein R, Wagener D. Operational
definitions for year 2000 objectives: Priority area
21, clinical preventive services. Healthy people
statistical notes no 17. Hyattsville, Maryland:
National Center for Health Statistics. 1998.
11. Partnership for Prevention/William Mercer
Survey of Employer-Sponsored Health Plans.
Washington, DC. 1999.
12. National Center for Health Statistics. Health,
United States, 1998, with socioeconomic status
and health chartbook. Hyattsville, Maryland:
Public Health Service. 1998.
Healthy People 2000 Final Review 297

Table 21. Clinical Preventive Services objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
21.1* Years of healthy life ..................... 1990 64.0 . . . 63.9 63.7 63.5 63.8 63.9 64.2
‡
64.8
‡
65.2 - - - 65
a. Black .............................. 1990 56.0 . . . 56.0 55.6 55.2 55.6 56.0 56.5
‡
57.4
‡
57.8 - - - 60
b. Hispanic
1
........................... 1990 64.8 . . . 63.6
2
64.0 63.2 64.2 64.0 64.7
‡
65.8
‡
66.3 - - - 65
c. People 65 years and over
3
.............. 1990 11.9 . . . 11.8 11.9 11.9 12.1 12.0 12.2
‡
12.0
‡
12.2 - - - 14
21.2 Receipt of recommended services
Children 19-35 months
Basic immunization series.................. ... --- --- --- --- --- --- --- --- --- --- --- 90%
DTP (3 or more doses) .................. 1992 83% . . . . . . . . . 88% 90% 95% 95% 95% 96% 96% . . .
Polio (3 or more doses) .................. 1992 72% . . . . . . . . . 79% 79% 88% 91% 91% 91% 90% . . .
Measles/Mumps/Rubella (1 dose) .......... 1992 83% . . . . . . . . . 84% 90% 90% 91% 91% 92% 92% . . .
Haemophilus influenza type b (3 or more
doses) .............................. 1992 28% . . . . . . . . . 55% 75% 92% 92% 93% 93% 94% . . .
Hepatitis B (3 or more doses) ............. 1993 16% . . . . . . . . . . . . 34% 68% 82% 84% 87% 88% . . .
4DTP/3Polio/1MMR ..................... 1992 55% . . . . . . . . . 67% 68% 76% 78% 78% 81% 80% . . .
People 18 years and over
Routine checkup, people 18–64 years
4
...... 1991 74% . . . . . . - - - 78% 70% 81% - - - - - - 76% - - - 91%
Routine checkup, people 65 years and over . . 1991 67% . . . . . . - - - 73% 62% 74% - - - - - - 77% - - - . . .
Cholesterol checked in last 5 years........... 1993 60% . . . . . . . . . . . . - - - - - - - - - - - - 67% - - - 75%
Cholesterol ever checked .................. 1991 63% . . . . . . - - - 71% - - - - - - - - - - - - 72% - - - . . .
People with low income
5
................. 1991 46% . . . . . . - - - 55% - - - - - - - - - - - - 55% - - - . . .
Black................................ 1991 56% . . . . . . - - - 72% - - - - - - - - - - - - 67% - - - . . .
Hispanic ............................. 1991 51% . . . . . . - - - 62% - - - - - - - - - - - - 56% - - - . . .
American Indian/Alaska Native ............ 1991 46% . . . . . . - - - 60% - - - - - - - - - - - - 53% - - - . . .
Cholesterol checked in last 2 years........... 1991 50% . . . . . . - - - 54% - - - - - - - - - - - - 57% - - - . . .
People with low income
5
................. 1991 37% . . . . . . - - - 41% - - - - - - - - - - - - 43% - - - . . .
Hispanic ............................. 1991 42% . . . . . . - - - 47% - - - - - - - - - - - - 45% - - - . . .
Asian/Pacific Islander ................... 1991 45% . . . . . . - - - 44% - - - - - - - - - - - - 54% - - - . . .
American Indian/Alaska Native ............ 1991 38% . . . . . . - - - 49% - - - - - - - - - - - - 42% - - - . . .
Tetanus booster in last 10 years ............. 1991 52% . . . . . . - - - 57% 56% 59% - - - - - - 57% - - - 62%
People 65 years and over ................ 1991 29% . . . . . . - - - 34% 32% 40% - - - - - - 41% - - - . . .
Hispanic ............................. 1991 45% . . . . . . - - - 48% 51% 51% - - - - - - 48% - - - . . .
Asian/Pacific Islander ................... 1991 40% . . . . . . - - - 45% 43% 40% - - - - - - 45% - - - . . .
People with disabilities................... 1991 47% . . . . . . - - - 51% 52% 56% - - - - - - - - - - - - . . .
Pneumococcal vaccine in lifetime (people
65 years and over) ...................... 1991 21% . . . . . . - - - 28% 30% 34% - - -
‡
42%
‡
46% - - - 60%
People with low income
5
................. 1991 17% . . . . . . - - - 18% 19% 25% - - -
‡
31%
‡
34% --- ...
Black................................ 1991 14% . . . . . . - - - 14% 15% 23% - - -
‡
22%
‡
26% --- ...
Hispanic ............................. 1991 12% . . . . . . - - - 13% 14% 23% - - -
‡
23%
‡
23% --- ...
Asian/Pacific Islander ................... 1991 15% . . . . . . - - - 21% 14% 22% - - -
‡
26%
‡
37% --- ...
Influenza vaccine in last 12 months (people
65 years and over) ...................... 1991 42% . . . . . . - - - 52% 55% 58% - - -
‡
63%
‡
63% - - - 60%
People with low income
5
................. 1991 36% . . . . . . - - - 41% 44% 46% - - -
‡
52%
‡
54% --- ...
Black................................ 1991 27% . . . . . . - - - 33% 39% 40% - - -
‡
45%
‡
46% --- ...
See footnotes and key at end of table.
298 Healthy People 2000 Final Review

Table 21. Clinical Preventive Services objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Hispanic ............................. 1991 34% . . . . . . - - - 47% 38% 50% - - -
‡
53%
‡
50% --- ...
Asian/Pacific Islander ................... 1991 29% . . . . . . - - - 54% 43% 51% - - -
‡
53%
‡
68% --- ...
Pap test in last 3 years (female 18 years and
over)................................. 1992 74% . . . . . . . . . 78% 77% - - - - - - - - - 79% - - - 85%
Females 65 years and over ............... 1992 51% . . . . . . . . . 58% 57% - - - - - - - - - 64% - - - . . .
Asian/Pacific Islander ................... 1992 62% . . . . . . . . . 69% 66% - - - - - - - - - 68% - - - . . .
American Indian/Alaska Native ............ 1992 64% . . . . . . . . . 78% 73% - - - - - - - - - 75% - - - . . .
Females with disabilities ................. 1992 65% . . . . . . . . . 69% 69% - - - - - - - - - - - - - - - . . .
Breast exam and mammogram in past 2 years
(female 50 years and over) ................ 1992 51% . . . . . . . . . 55% 56% - - - - - - - - - 64% - - - 60%
Females 65 years and over ............... 1992 43% . . . . . . . . . 49% 49% - - - - - - - - - 58% - - - . . .
Females with low income
5
................ 1992 30% . . . . . . . . . 39% 38% - - - - - - - - - 48% - - - . . .
Asian/Pacific Islander ................... 1992 38% . . . . . . . . . 53% 46% - - - - - - - - - 55% - - - . . .
American Indian/Alaska Native ............ 1992 31% . . . . . . . . . 38% 53% - - - - - - - - -
6
--- --- ...
Females with disabilities ................. 1992 44% . . . . . . . . . 51% 50% - - - - - - - - - - - - - - - . . .
Asked at least one screening question at routine
checkup
7
.............................. 1991 56% . . . . . . - - - 63% 56% - - - - - - - - - 76% - - - 80%
People 65 years and over ................ 1991 42% . . . . . . - - - 48% 38% - - - - - - - - - 77% - - - . . .
Asian/Pacific Islander ................... 1991 51% . . . . . . - - - 60% 48% - - - - - - - - - 76% - - - . . .
21.3 Usual source of care
Total population (18 years and over) .......... 1991 80% . . . . . . 78% 83% 84% 85% 85%
‡
84%
‡
85% - - - 95%
a. Hispanic ............................ 1991 63% . . . . . . 64% 71% 71% 74% 73%
‡
71%
‡
72% - - - 95%
Mexican American .................... 1991 57% . . . . . . 62% 69% 69% 72% 70%
‡
66%
‡
66% - - - 95%
b. Black .............................. 1991 78% . . . . . . 75% 79% 82% 82% 83%
‡
81%
‡
83% - - - 95%
c. Low-income people (family income below
poverty level)........................ 1991 71% . . . . . . 71% 72% 73% 76% 76%
‡
75%
‡
76% - - - 95%
d. American Indian/Alaska Native ........... 1991 70% . . . . . . 85% 82% 81% 84% 83%
‡
83%
‡
77% - - - 95%
e. Asian/Pacific Islander .................. 1991 70% . . . . . . 71% 74% 78% 81% 79%
‡
80%
‡
80% - - - 95%
21.4 Financial barriers to receipt of clinical
preventive services
Proportion of people under 65 years without
health care coverage
8
(age adjusted) ........ 1989
§
15.7% - - - - - - 17.2% 17.3% 17.8% 15.6% 16.1%
‡
17.5%
‡,p
16.6 - - - 0%
a. American Indian/Alaska Native ........... 1989
§
36.1% - - - - - - - - - 34.2% 39.0% 33.9% 33.9%
‡
38.1%
‡,p
38.7 - - - 0%
b. Hispanic ............................ 1989
§
31.3% - - - - - - 34.0% 34.2% 32.9% 30.8% 31.6%
‡
34.5%
‡,p
34.1 - - - 0%
Mexican American .................... 1989
§
38.1% - - - - - - 37.8% 39.5% 37.2% 35.4% 36.7%
‡
39.4%
‡,p
40.1 - - - 0%
Puerto Rican ........................ 1989
§
21.4% - - - - - - 18.3% 21.0% 17.4% 17.8% 14.4%
‡
19.0%
‡,p
18.9 - - - 0%
Cuban ............................. 1989
§
20.7% - - - - - - 20.1% 16.9% 27.4% 21.6% 17.6%
‡
21.1%
‡,p
18.5 - - - 0%
c. Black .............................. 1989 22.0% - - - - - - 22.3% 23.2% 21.5% 17.9% 19.0%
‡
20.1%
‡,p
20.4 - - - 0%
See footnotes and key at end of table.
Healthy People 2000 Final Review 299

Table 21. Clinical Preventive Services objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
21.5 Clinical preventive services from publicly
funded programs (proportion of eligible
people)
Federal programs
Screening .......................... 1991-92 10-100% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 90%
Counseling.......................... 1991-92 40-100% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 90%
Immunizations ....................... 1991-92 10-100% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - 90%
21.6 Provision of recommended services by
primary care providers .................. ... --- --- --- --- --- --- --- --- --- --- --- 50%
Percent of clinicians routinely providing service to
81–100% of patients
Preventive services for children:
Hemoglobin/hematocrit
Pediatricians .......................... 1992 78% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - . . .
Nurse practitioners ..................... 1992 77% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - . . .
Family physicians ...................... 1992 52% . . . . . . . . . - - - - - - - - - - - - - - - - - - - - - . . .
Eye exam (for strabismus and amblyopia)
Pediatricians .......................... 1992 64% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Nurse practitioners ..................... 1992 67% . . . . . . . . . - - - - - - - - - - - - - - -
9
51% --- ...
Family physicians ...................... 1992 53% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Blood pressure
Pediatricians .......................... 1992 78% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Nurse practitioners ..................... 1992 71% . . . . . . . . . - - - - - - - - - - - - - - -
9
65% --- ...
Family physicians ...................... 1992 42% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Height and weight
Pediatricians .......................... 1992 96% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Nurse practitioners ..................... 1992 88% . . . . . . . . . - - - - - - - - - - - - - - -
9
85% --- ...
Family physicians ...................... 1992 89% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
DTP vaccination
Pediatricians .......................... 1992 86% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Nurse practitioners ..................... 1992 76% . . . . . . . . . - - - - - - - - - - - - - - -
9
78% --- ...
Family physicians ...................... 1992 89% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Oral polio vaccination
Pediatricians .......................... 1992 87% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Nurse practitioners ..................... 1992 76% . . . . . . . . . - - - - - - - - - - - - - - -
9
79% --- ...
Family physicians ...................... 1992 89% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Tetanus-diphtheria booster
Pediatricians .......................... 1992 79% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Nurse practitioners ..................... 1992 71% . . . . . . . . . - - - - - - - - - - - - - - -
9
72% --- ...
Family physicians ...................... 1992 70% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Hib vaccination
Pediatricians .......................... 1992 85% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Nurse practitioners ..................... 1992 68% . . . . . . . . . - - - - - - - - - - - - - - -
9
74% --- ...
Family physicians ...................... 1992 74% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
300 Healthy People 2000 Final Review

Table 21. Clinical Preventive Services objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
Preventive services for adults:
Tetanus-diphtheria booster (18 years and over)
Nurse practitioners ..................... 1992 38% . . . . . . . . . - - - - - - - - - - - - - - -
9
40% --- ...
Obstetricians/gynecologists ............... 1992 4% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Internists ............................. 1992 29% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Family physicians ...................... 1992 28% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Influenza vaccination (65 years and over)
Nurse practitioners ..................... 1992 42% . . . . . . . . . - - - - - - - - - - - - - - -
9
47% --- ...
Obstetricians/gynecologists ............... 1992 6% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Internists ............................. 1992 49% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Family physicians ...................... 1992 31% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Pneumococcal vaccination (65 years and over)
Nurse practitioners ..................... 1992 33% . . . . . . . . . - - - - - - - - - - - - - - -
9
43% --- ...
Obstetricians/gynecologists ............... 1992 5% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Internists ............................. 1992 40% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Family physicians ...................... 1992 25% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Blood pressure
Nurse practitioners ..................... 1992 82% . . . . . . . . . - - - - - - - - - - - - - - -
9
90% --- ...
Obstetricians/gynecologists ............... 1992 88% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Internists ............................. 1992 92% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Family physicians ...................... 1992 89% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Cholesterol level
Nurse practitioners ..................... 1992 45% . . . . . . . . . - - - - - - - - - - - - - - -
9
55% --- ...
Obstetricians/gynecologists ............... 1992 36% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Internists ............................. 1992 80% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Family physicians ...................... 1992 61% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Breast exam (by clinician)
Nurse practitioners ..................... 1992 78% . . . . . . . . . - - - - - - - - - - - - - - -
9
67% --- ...
Obstetricians/gynecologists ............... 1992 92% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Internists ............................. 1992 76% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Family physicians ...................... 1992 62% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Pap smear
Nurse practitioners ..................... 1992 77% . . . . . . . . . - - - - - - - - - - - - - - -
9
65% --- ...
Obstetricians/gynecologists ............... 1992 92% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Internists ............................. 1992 67% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Family physicians ...................... 1992 62% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Mammogram
Nurse practitioners 1992 63% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Patients 40-49 years .................. ... --- --- --- --- --- --- --- --- ---
9
58% --- ...
Patients 50 years and over.............. ... --- --- --- --- --- --- --- --- ---
9
60% --- ...
Obstetricians/gynecologists ............... 1992 85% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Internists ............................. 1992 67% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
Family physicians ...................... 1992 53% . . . . . . . . . - - - - - - - - - - - - - - -
6,9
--- --- ...
See footnotes and key at end of table.
Healthy People 2000 Final Review 301

Table 21. Clinical Preventive Services objectives—Con.
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
21.7 Local health department assurance of access
to essential clinical preventive services
Proportion of people served ................ ... --- --- --- --- --- --- --- --- --- --- --- 90%
Proportion of local health departments that:
Assess the extent to which screening,
immunization, and counseling services are
provided to the local population ........... 1992-93 76% . . . . . . . . . . . . - - - - - - - - - - - - - - - - - - . . .
Collect data to document the number of
providers of clinical preventive services in
their jurisdiction ....................... 1992-93 45% . . . . . . . . . . . . - - - - - - - - - - - - - - - - - - . . .
Evaluate to determine whether a gap exists
between available clinical preventive services
and the need for those services ........... 1992-93 57% . . . . . . . . . . . . - - - - - - - - - - - - - - - - - - . . .
(Of those that assess gaps) Provide clinical
preventive services to fill gaps ............ 1992-93 83% . . . . . . . . . . . . - - - - - - - - - - - - - - - - - - . . .
21.8 Racial/ethnic minority representation in the
health professions
Degrees awarded:
Black................................ 1985–86 5.0% - - -
10
5.7%
11
5.7%
12
5.9%
13
5.9%
14
6.0
15
6.6
16
6.7
17
7.1 - - - 8.0%
Hispanic ............................. 1985–86 3.0% - - -
10
4.3%
11
4.8%
12
4.8%
13
4.3%
14
4.1
15
3.8
16
4.0
17
4.4 - - - 6.4%
American Indian/Alaska Native ............ 1985–86 0.3% - - -
10
0.4%
11
0.5%
12
0.4%
13
0.4%
14
0.4
15
0.5
16
0.6
17
0.6 - - - 0.6%
a. Enrolled in schools of nursing:
Black ..............................
11
1991–
92 9.1% . . . . . . . . .
12
8.6%
13
8.7%
14
9.0%
15
9.4 - - - - - - - - - 10.0%
Hispanic............................
11
1991–
92 3.1% . . . . . . . . .
12
3.0%
13
3.0%
14
3.2%
15
3.5 - - - - - - - - - 4.0%
Asian/Pacific Islander ..................
11
1991–
92 2.9% . . . . . . . . .
12
3.2%
13
3.3%
14
3.6%
15
4.0 - - - - - - - - - 5.0%
American Indian/Alaska Native ...........
11
1991–
92 0.7% . . . . . . . . .
12
0.6%
13
0.7%
14
0.7%
15
0.7 - - - - - - - - - 1.0%
- - - Data not available. Final objective status:
. . . Category not applicable.
§
Baseline has been revised.
p
Preliminary data.
‡
The NHIS was redesigned in 1997. Data may not be comparable with previous years; see Appendix.
1
Estimate based on preliminary data. Excludes mortality data from States lacking an Hispanic-origin item on their death certificate or for which Hispanic-origin data were not of sufficient quality.
See Appendix.
2
Estimate derived from 1991-93 health status data and 1992 mortality data.
3
Years of healthy life remaining for those surviving to age 65.
4
In the last 3 years for people 18-64 and in the last year for people 65 years and over.
5
Data are for persons with family incomes below the Census poverty threshold.
6
Response rates for this group were too low to produce reliable estimates.
7
For people 18-64 years, a screening question was asked during a routine checkup in the past 3 years on at least one of the following: diet, physical activ ity, tobacco use, alcohol use, drug
use, sexually transmitted diseases, or contraceptive use. For people 65 years and over, a screening question was asked during a routine checkup in the past year about at least one of the
following: diet, physical activity, tobacco use, or alcohol use.
8
Beginning with 1996 data, persons receiving public assistance other than Medicaid are considered to have health care coverage. Prior to 1996 they were considered not to have health care
coverage.
9
1997-98 data.
Met
Toward
Mixed/ no change
Away
Cannot assess
302 Healthy People 2000 Final Review

10
Academic year 1990-91.
11
Academic year 1991-92.
12
Academic year 1992-93.
13
Academic year 1993-94.
14
Academic year 1994-95.
15
Academic year 1995-96.
16
Academic year 1996-97.
17
Academic year 1997-98.
NOTE: Data include revisions and, therefore, may differ from those previously published in these reports and other publications. DTP is diphtheria-tetanus-pertusis. MMR is measles-mumps-
rubella. Hib is Haemophilus influenzae type B.
Objective number Data source
21.1*, 21.1a-c National Vital Statistics System, CDC, NCHS; National Health Interview Survey, CDC, NCHS.
21.2 Excluding basic immunization series among children: National Health Interview Survey, CDC, NCHS.
Basic immunization series among children:
Baseline for children 2 years and under: United States Immunization Survey, CDC, NCHSTP.
Children 19–35 months: 1991–1994 data: National Health Interview Survey, CDC, NCHS.
1995–1999 data: National Immunization Survey, CDC, NIP.
21.3, 21.3a-e National Health Interview Survey, CDC, NCHS.
21.4, 21.4a-c National Health Interview Survey, CDC, NCHS.
21.5 For Community/Migrant Health Centers: Bureau of Primary Health Care Survey, HRSA, OPEL
For other publicly funded programs: Survey of Federal Programs, HRSA, OPEL.
21.6 Baseline: Primary Care Providers Survey, OPHS, ODPHP.
Update: Prevention in Primary Care Study, American College of Preventive Medicine.
21.7 National Profile of Local Health Departments, National Association of County and City Health Officials.
21.8 Minorities and Women in the Health Fields, HRSA, BHPr.
21.8a National League for Nursing, Nursing Data Source.
*Duplicate objective. See full text of objective following this table.
Healthy People 2000 Final Review 303
Clinical Preventive
Services Objectives
21.1*: Increase years of healthy life to
at least 65 years.
Duplicate objectives: 8.1 and 17.1
21.1a*: Increase years of healthy
life among blacks to at least 60
years.
Duplicate objectives: 8.1 and 17.1a
21.1b*: Increase years of healthy
life among Hispanics to at least 65
years.
Duplicate objectives: 8.1b and 17.1b
21.1c*: Increase years of healthy
life among people aged 65 and
older to at least 14 years remaining.
Duplicate objectives: 8.1c and 17.1c
21.2: Increase the proportion of people
who have received selected clinical
preventive screening and immunization
services and at least one of the
counseling services appropriate for their
age and gender as recommended by the
U.S. Preventive Services Task Force.
Receipt of selected clinical 2000
preventive and counseling target
services (percent)
Basic Immunization Series 90
Routine check-up 91
Cholesterol checked in last 5 years 75
Cholesterol ever checked 75
Cholesterol checked in last 2 years 75
Tetanus booster in last 10 years 62
Pneumococcal vaccine in lifetime
(aged 65 and over) 60
Influenza vaccine in last year
(aged 65 and over) 60
Pap test in last 3 years 85
Breast exam and mammogram in
past 2 years 60
Counseling services 80
21.3: Increase to at least 95 percent the
proportion of people who have a
specific source of ongoing primary care
for coordination of their preventive and
episodic health care.
21.3a: Increase to at least
95 percent the proportion of
Hispanics and the proportion of
Mexican-Americans who have a
specific source of ongoing primary
care for coordination of their
preventive and episodic health care.
21.3b: Increase to at least
95 percent the proportion of blacks
who have a specific source of
ongoing primary care for
coordination of their preventive and
episodic health care.
21.3c: Increase to at least 95 percent
the proportion of low-income
people who have a specific source
of ongoing primary care for
coordination of their preventive and
episodic health care.
21.3d: Increase to at least
95 percent the proportion of
American Indians and Alaska
Natives who have a specific source
of ongoing primary care for
coordination of their preventive and
episodic health care.
21.3e: Increase to at least 95 percent
the proportion of Asians and Pacific
Islanders who have a specific source
of ongoing primary care for
coordination of their preventive and
episodic health care.
21.4: Improve financing and delivery of
clinical preventive services so that
virtually no American has a financial
barrier to receiving, at a minimum, the
screening, counseling, and immunization
services recommended by the U.S.
Preventive Services Task Force.
21.4a: Decrease to 0 percent the
proportion of American Indians and
Alaska Natives under 65 years
without health care coverage.
21.4b: Decrease to 0 percent the
proportion of Hispanics under 65
years, and Mexican-Americans,
Puerto Ricans, and Cubans under 65
years without health care coverage.
21.4c: Decrease to 0 percent the
proportion of blacks under 65 years
without health care coverage.
21.5: Ensure that at least 90 percent of
people for whom primary care services
are provided directly by publicly funded
programs are offered, at a minimum, the
screening, counseling, and immunization
services recommended by the U.S.
Preventive Services Task Force.
21.6: Increase to at least 50 percent the
proportion of primary care providers
who provide their patients with the
screening, counseling, and immunization
services recommended by the U.S.
Preventive Services Task Force.
21.7: Increase to at least 90 percent the
proportion of people who are served by
a local health department that assesses
and assures access to essential clinical
preventive services.
21.8: Increase the proportion of all
degrees in the health professions and
allied and associated health profession
fields awarded to members of
underrepresented racial and ethnic
minority groups as follows:
2000 target
Degrees awarded to— (percent)
Blacks 8.0
Hispanics 6.4
American Indians and
Alaska Natives 0.6
21.8a: Increase the proportion of
individuals from underrepresented
racial and ethnic minority groups
enrolled in U.S. schools of nursing.
2000
Proportion enrolled target
in fall academic year (percent)
Blacks 10
Hispanics 4
Asians and Pacific
Islanders 5
American Indians and
Alaska Natives 1
*Duplicate objective.
304 Healthy People 2000 Final Review

Priority Area 22
Surveillance and
Data Systems
Background
Public health surveillance is the
systematic collection, analysis, and use
of health information. It is essential to
understanding the health status of a
population and to planning effective
programs and interventions. The
Institute of Medicine identifies
surveillance as one of the core functions
of public health (1). Surveillance and
health data systems provide information
on illness, disability, and death from
acute to chronic conditions; injuries,
personal, environmental, and
occupational risk factors; preventive and
treatment services; and costs. To be
most useful, public health data must be
accessible, accurate, timely, and clearly
stated and must adhere to strict
confidentiality standards. Surveillance or
health data systems must be linked with
other data systems as well as linked and
integrated at Federal, Tribal, State, and
local levels.
While the collection of national
public health data usually rests with
Federal agencies, data collection,
analysis, and use depend on the active
involvement of many partners including
Tribal, State, and local agencies. For
example, the Vital Statistics Cooperative
program (VSCP) collects information on
births, deaths, marriages, and divorces
from many sources in local
communities, including hospitals,
funeral directors, medical examiners,
coroners, hospitals, religious authorities,
and justices of the peace. VSCP gets
information from all 50 States, the
District of Columbia, Puerto Rico, the
U.S. Virgin Islands, and Guam. Other
data collection systems, based on sample
surveys rather than reports, depend on
the participation of thousands of private
citizens nationwide. Still others rely on
the administrative records of public and
private health care organizations.
Having appropriate surveillance and
data systems in place also provides the
ability to measure the health status of
special populations. Morbidity, mortality,
health behaviors, and access to and use
of health services vary markedly by age,
race, sex, and socioeconomic status.
There are 319 Healthy People 2000
subobjectives that are targeted toward
racial and ethnic minorities, elderly
people, people with chronic disabilities,
people with less than a high school
education, people with low incomes, and
others.
At the beginning of the 1990s,
23 percent of the Healthy People 2000
objectives could not be assessed because
of the unavailability of national data.
The lack of data at the State and local
levels was of even greater concern.
During the decade, almost all the States
adopted their own State plans or
objectives. Data have been needed to
monitor progress toward achieving those
objectives and to compare that progress
with the national progress. Thus, several
objectives in Priority Area 22 were
directed toward enhancing data systems
in States and communities. Similarly,
some objectives addressed the
identification of and response to data
gaps related to minorities and other
special populations.
Data Summary
Two (22.1 and 22.6) of the seven
objectives in the Surveillance and Data
Systems priority area have been met.
Four (22.2–22.5) objectives moved
toward the target, and one objective
(22.7) showed mixed progress.
Objective 22.1, to develop and
establish use of Health Status Indicators
(HSI’s), has been achieved. The first
part of the objective was achieved early
in the decade when the consensus set of
18 indicators was published in July
1991 (2). National data for the HSI’s
were first published in October 1992
(3). National trends in the HSI data for
the past 3 years are shown in the Health
Status Indicators/Priority Data Needs
section in table B. Table C presents the
indicators for the major racial and ethnic
groups for 1998, the most recent data
year. A detailed discussion of HSI
differentials by race and Hispanic origin
was published in September 1995 (4).
An assessment of the progress achieved
by each State and the United States was
published in November 2000 (5).
National data for Priority Data Needs
(6), a set of indicators of community
health status identified as important for
evaluating the health of a population but
lacking data at all levels of government,
are presented in table D.
The achievement of the second part
of this objective, to establish use of the
HSI’s in at least 40 States, was
measured by tracking their use by State
and local health departments. All States,
including the District of Columbia, are
monitoring some of the HSI’s. At least
nine States have published the HSI data
for county, region, or health department
district (an update of Statistics and
Surveillance number 8) (7). The 1997
State and Local Capacity Survey
developed and administered by the
Public Health Foundation found that 41
of 42 responding States provided
information about some HSI’s to local
health departments (8). Thirty-nine of
forty-six responding States have
produced a report that includes over
one-half the HSI’s and 31 of these
States indicated that the reports included
information specific to areas within the
State (such as counties, regions, and
health districts). HSI data by State can
be accessed via FTP (file transfer
protocol) on the Internet at
http://ftp.cdc.gov/pub/Health_Statistics/
NCHS/Datasets/Healthy_People_2000.
See the section on Health Status
Indicators/Priority Data Needs for more
information.
Objective 22.2 was almost achieved
with 97 percent of all Healthy People
2000 objectives having ongoing data
sources. Overall, nine objectives lacked
baseline data (7.12, 7.14, 7.17, 9.11,
9.20, 14.14, 14.16, 17.20, and 18.17).
By 1997, 32 of the 44 States and the
District of Columbia with Healthy
People 2000 plans had included
surveillance and data systems objectives
in their plans.
Objective 22.3 has three parts:
developing, disseminating, and
incorporating into Public Health Service
data collection systems the procedures
for collecting comparable data for each
of the Healthy People 2000 national
health objectives among Federal, State,
and local agencies. The baseline shows
the percent of objectives that are tracked
with vital statistics data, which are
comparable at all levels of measurement.
Comparable procedures for monitoring
population-based nutrition objectives
among Federal surveys were included
for 1992. Objectives measured by
systems that have comparable data at
the Federal, State, or local levels such
as those monitored with the Youth Risk
Behavior Surveillance System (YRBSS),
the National Notifiable Disease
Surveillance System, the Fatality
Analysis Reporting System (formerly
the Fatal Accident Reporting System),
and several other national systems that
Healthy People 2000 Final Review 305

Figure 22. Final status of Surveillance and Data Systems objectives
22.1 Health status indicators: Develop
Establish use
22.2 National data sources
BRFSS data
22.5 Analysis/publication of data: Vital statistics
22.3 Comparable data collection procedures
Hospital discharge data
YRBSS data
22.6 Data transfer systems: NETSS
22.7 Timely release of national data:
Within 1 year of collection
Between 1-2 years of collection
DATA2000
PHLIS
Moved away from target Moved toward target Met target
NOTES: Complete tracking data are shown in table 22. Progress quotients are not calculated for objective 22.4.
See the section on Measuring Progress Toward the
Healthy People 2000
Targets in the Appendix for more information.
BRFSS is Behavioral Risk Factor Surveillance System. YRBSS is Youth Risk Behavior Surveillance System. NETSS is National Electronic
Telecommunications System for Surveillance. PHLIS is Public Health Laboratory Information System.
Percent of target achieved
-100 -80 -60 -40 -20
0
20 40 60 80 100
100%
100%
100%
100%
100%
100%
100%
20%
69%
79%
10%
87%
-106%
306 Healthy People 2000 Final Review
depend on State data were included for
1994. Objectives monitored with the
National Health Interview Survey were
also included in 1994 if the State
Behavioral Risk Factor Surveillance
System (BRFSS) asked questions that
were similar in wording and conceptual
approach (some objectives, for example,
physical activity and current smoking,
are being monitored by the BRFSS but
not with methods comparable with
national methods).
The denominator for the proportion
of objectives with comparable data is
the total number of objectives, which
underrepresents Federal-State-local
objective measurement comparability.
Some objectives are out of scope for the
purposes of tracking objective 22.3,
including most Services and Protection
objectives, which do not involve
traditional data collection comparability
issues, although they may involve what
is considered appropriate protocol.
These objectives include patient
education and counseling, employer- and
community-based prevention programs,
development and implementation of
quality standards, conformance with
national guidelines, and enactment of
national or State laws. Therefore, the
1994 estimate of 21 percent is an
underestimate of the actual proportion of
all objectives with comparable data
collection procedures.
Developing and disseminating
comparable data collection procedures
involves systematically documenting the
methods that are currently being used
and accepted (as well as changes in
measurement methods over time). A
series of reports was initiated as part of
the Healthy People Statistical Notes
series that documented methods for
objectives in specific priority areas of
Healthy People 2000. For a priority
area, the report describes how each
objective is being measured at the
Federal level and the relevant data
issues involved. Six of these Statistical
Notes were published (see Appendix
table VI). For Healthy People 2010,a
separate report, Tracking Healthy People
2010, was published in November 2000
(9). This report contains operational
definitions for the Healthy People 2010
objectives that have baseline data (595
of 824 measures—72 percent—have
complete baseline data and operational
definitions). The report also provides
information on the major national data
sources and State data sources (if
available) used for Healthy People 2010
objectives and discusses cross-cutting
statistical issues.
Objective 22.4 (duplicated as
objective 17.22 during the midcourse
review) addresses the development and
implementation of a national process to
identify significant gaps in the Nation’s
disease prevention and health promotion
data. Progress was made, although it is
difficult to quantify. There are two parts
to this objective: the identification of
data gaps in broad areas of public health
where insufficient data exist to develop
objectives, and the identification of data
gaps connected with special populations.
In 1993, first steps to identify significant
gaps in broad areas of the Nation’s
disease prevention and health promotion
data were taken. The National
Committee on Vital and Health Statistics
Subcommittee on State and Community
Health Statistics recommended the
development of a coordinated Federal,
State, and community health statistics
system that should include the following
data sets in order to carry out the
functions of assessment and policy
development: vital statistics, in-patient
hospitalization utilization, ambulatory
care, long-term care, incidence and
prevalence of disease and disability,
health care resources, health care costs
and expenditures, demographic profiles
of populations served, access to basic
health care and preventive services,
health risk behaviors and attitudes, and
environmental health risks.
The process of the Healthy People
2000 midcourse review (10) brought the
Nation closer to achieving the latter part
of objective 22.4. During the midcourse
review, considerable attention was given
to population groups that are at highest
risk for premature death, disease, or
disability, and 120 new subobjectives
were proposed by the lead PHS
agencies; 111 new subobjectives were
eventually added (see the section on the
midcourse review in the Appendix).
The importance of identifying data
gaps connected with special populations
is further emphasized in Healthy People
2010 where objectives that utilize
population-based measures display the
baseline status of multiple population
groups. The minimum breakout set of
groups includes race and ethnicity,
gender, and measures of socioeconomic
status. However, many objectives
include additional population groups
such as age, geographic location, health
insurance status, disability status, sexual
orientation, or people with specific
health conditions. Baseline data were
available for 74 percent of the 396
measures in Healthy People 2010 that
listed population groups (11).
Progress toward objective 22.5, the
number of States that periodically
analyze and publish data needed to
measure progress toward the national
health objectives, was assessed by the
number of States that publish data from
major databases including vital statistics,
the BRFSS, hospital discharge systems,
and the YRBSS. The Health Care Cost
and Utilization Project (HCUP) has been
developed to build comparable hospital
discharge data sets among States; HCUP
contains a subset of the States reporting
inpatient hospital discharge data. In
1997, 22 States participated in the
national database (12); 26 participated in
1999. The number of States with at least
one racial or ethnic group that
comprises at least 10 percent of their
population that publish vital statistics
data for each of these groups is also
being tracked. There are 27 States
whose populations include at least
10 percent racial and ethnic minorities.
In 1999, data on major racial and ethnic
groups were published for all of these
States, exceeding the Healthy People
2000 target of 25 States.
Data to measure objective 22.6 (to
expand in all States systems for the
transfer of year 2000 data among
Federal, State, and local agencies) are
available for three data systems. All
three data systems have met the Healthy
People 2000 target of providing
electronic data transfer systems for all
States. The National Electronic
Telecommunications System for
Surveillance (NETSS) is operating in all
States and the District of Columbia. In
1995, the Public Health Laboratory
Information System (PHLIS) became
available in all States and the District of
Columbia as well. Electronic access to
national monitoring data for Healthy
People was provided through
DATA2000 on the Centers for Disease
Control and Prevention WONDER
system from 1995 to 2000. In 2000,
DATA2000 was replaced on the CDC
WONDER system by DATA2010.
DATA2010 provides national and State
data to monitor Healthy People 2010
objectives and represents the expanded
data monitoring agenda of the next
decade.
The National Center for Health
Statistics Internet homepage contains
Healthy People 2000 Final Review 307
links to State data for a number of
Healthy People 2000 objectives
(http://www.cdc.gov/nchs). Since 1997,
mortality data have been available
through the CDC FTP server; in 1998
Healthy People 2000 data for natality
objectives by State became available.
Since 2000, mortality data by State for
the years 1989–97 are available in the
Data Warehouse section reached through
the NCHS homepage
(http://www.cdc.gov/nchs/datawh/ftpserv/
hstatus/hstatus.htm); race and ethnicity
data are provided for 19 causes of death
targeted in Healthy People 2000 and for
HIV mortality; and natality data are
available from 1993 to 1997. The
National Center for Chronic Disease
Prevention and Health Promotion has
made State data available on the Internet
from the BRFSS
(http://www2.cdc.gov/nccdphp/brfss/
index.asp). Much of these data relate to
Healthy People 2000 objectives.
Achieving the timely release of
national surveillance and survey data to
measure progress toward the national
health objectives (22.7), is measured by
percent of objectives with data released
within 1 year and between 1 and 2 years
of data collection. The actual
measurement of this objective involves
counting the objectives that have
updates for a particular year. For the
Healthy People 2000 Final Review, data
collected in 1998 or later are counted as
being released within 1 year. Data for
1997 are counted as being released
between 1 and 2 years of data
collection.
Because all objectives are included
in the denominator, the data monitoring
objective 22.7 represents an
underestimate of the ‘‘true’’ percent of
objectives with timely data. Some
objectives are not applicable because of
achievement through legislation or some
other means earlier in the decade,
leading to an underestimate of
timeliness. In addition, the measurement
of progress for this objective is also
affected by the periodicity of data
collection. Some objectives are tracked
with data available annually, such as
data from the National Vital Statistics
System for objectives targeting mortality
rates and the National Notifiable
Disease Surveillance System for
vaccine-preventable diseases (objective
20.1). Other objectives are tracked with
biennial data, such as data from the
YRBSS for objectives targeting
adolescents. Other objectives are
monitored with data from periodic
surveys such as the National Health and
Nutrition Examination Surveys for
measures of overweight and prevalence
of high blood cholesterol. For these
surveys, data are counted as timely for
years close to the years of data
collection only, even though the data
may have been released soon after
collection.
See table 22 for the tracking data
for the objectives in this priority area
and figure 22 for a quantitative
assessment of progress.
Transition to Healthy People
2010
The Healthy People initiative is
now in its third decade with the
publication of Healthy People 2010
(11). Healthy People activities during
the 1980s and 1990s have demonstrated
the central role of data, focused
attention on what is important to
measure, and stimulated the
development of new data systems. The
past 20 years in monitoring and
reporting on the Healthy People
objectives have provided a means for
assessing improvements in national
health data systems (13). The focus on
data and information systems continues
in Healthy People 2010 as a pivotal part
of improving the Nation’s public health
infrastructure, that is, the resources
needed to deliver the essential public
health services to every community.
Seven objectives in the Data and
Information Systems section of the
Healthy People 2010 Public Health
Infrastructure chapter focus on data
issues. The need for data on special
population groups, availability of data at
subnational levels on important
indicators, regular collection and
publication of data, and timely release
of data are continued as objectives for
Healthy People 2010. New objectives
have been added that focus on the use
of geocoding to promote nationwide use
of geographic information systems
(GIS), and the provision of Internet and
e-mail access to public health
employees.
Appendix table III, a crosswalk
between Healthy People 2000 and
Healthy People 2010 objectives,
summarizes the differences between the
two decades of objectives, reflecting
new knowledge and direction in the area
of surveillance and data systems.
References
1. Institute of Medicine. The future of public
health. Washington: National Academy Press.
1988.
2. Centers for Disease Control and Prevention.
Consensus set of health indicators for the general
assessment of community health status, United
States. MMWR 40(27):449–51. 1991.
3. Klein RJ, Hawk SA. Health status indicators:
Definitions and national data. Healthy People
statistical notes; vol 1 no 3. Hyattsville, Maryland:
National Center for Health Statistics. 1992.
4. Plepys C, Klein R. Health status indicators:
Differentials by race and Hispanic origin. Healthy
People statistical notes; no 10. Hyattsville,
Maryland: National Center for Health Statistics.
1995.
5. Keppel KG and Pearcy JN. Healthy People
2000: An assessment based on the health status
indicators for the United States and each state.
Healthy people statistical notes; no 19. Hyattsville,
Maryland: National Center for Health Statistics.
2000.
6. Kim I and Keppel KG. Priority data needs:
Sources of national, state, and local-level data and
data collection systems. Healthy people statistical
notes; no 15. Hyattsville, Maryland: National
Center for Health Statistics. 1997.
7. National Center for Health Statistics. Healthy
people statistics and surveillance; no 8.
Hyattsville, Maryland: National Center for Health
Statistics. 1996.
8. Public Health Foundation. Measuring health
objectives and indicators: 1997 state and local
capacity survey. Washington, D.C. 1998.
9. Department of Health and Human Services.
Tracking healthy people 2010. Washington: U.S.
Government Printing Office. 2000.
10. Department of Health and Human Services.
Healthy people 2000 midcourse review and 1995
revisions. Washington: Public Health Service.
1995.
11. Department of Health and Human Services.
Healthy People 2010. 2d ed. With understanding
and improving health and objectives for improving
health. 2 vols. Washington: U.S. Government
Printing Office. 2000.
12. For information on the Healthcare Cost and
Utilization Project (HCUP–3), see
http://www.ahrq.gov or contact the Agency for
Healthcare Research and Quality at (301)
594–1400.
13. Maiese DR. Data challenges and successes
with healthy people. Healthy people statistics and
surveillance; no 9. Hyattsville, Maryland: National
Center for Health Statistics. 1998.
308 Healthy People 2000 Final Review

Table 22. Surveillance and Data Systems objectives
Final
status Objective
Baseline
year Baseline 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999
Target
2000
22.1 Health status indicators (HSI)
Develop (indicators selected) .................... 1991 100% 100% 100% 100% 100% 100% 100% 100% 100% 100% 100% 100%
Establish use (number of States)
Monitoring some indicators .................... ... --- --- --- 48
1
51
1
51
1
51
1
51
1
51
1
51
1
51 40
Providing HSI data to local health departments ..... ... --- --- --- 36 --- --- --- --- 39 --- --- 40
22.2 National data sources......................... 1990
2
77% ... --- --- 93% 93%
3
96% 97%
4
97%
4
97%
4
97% 100%
a. State level data for at least two-thirds of State
objectives (number of States)
1,5
............... 1995 42 . . . . . . . . . . . . . . . . . . - - - 45 - - - - - - 50
22.3 Comparable data collection procedures
Federal, State, and local agencies ................ 1990 12% . . . - - - 14% - - - 21% - - - - - - - - - - - - - - - 100%
Publication of operational definitions and sources for
national data................................ ... --- --- --- --- --- --- --- ---
6
29% - - -
7
72% . . .
22.4* Identify gaps in health data .................... 1990
None
identified - - - - - - - - - - - -
8
---
9
---
10
---
10
---
11
---
11
--- ...
Establish mechanisms to meet needs .............. 1990
None
established - - - - - - - - - - - -
8
---
9
---
10
---
10
---
11
---
11
--- ...
22.5 Periodic analysis and publication of data
(number of States) ............................ 1989 20 - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 50
Vital statistics
1
............................... ... ---
12
51 51 51 51 51 51 51 51 51 51 50
Behavioral Risk Factor Surveillance System data
1
..... ... ---
12
40 - - - 49 50 51 51 51 51 51 51 50
Hospital discharge data
13
....................... ... ---
12
22 --- --- 39 39 39 --- --- --- 44 50
Youth Risk Behavior Survey data
1
................. ... --- 24 27 --- 41 ... 41 ... 39 ... 42 50
a. Analysis for racial and ethnic groups
(number of States)
1,14
.......................... 1992
15
19 ... ... ...
15
23 --- --- 22 --- ---
16
27 25
22.6 Number of States with data transfer system data
1
.. 1989 30 - - - - - - - - - - - - - - - - - - - - - - - - - - - - - - 50
National Electronic Telecommunications System for
Surveillance (NETSS)
1
........................ ... --- --- --- 51 51 51 51 51 51 51 51 50
Public Health Laboratory Information System (PHLIS) . . . . . - - - - - - - - - 37
1
44
1
51
1
51
1
51
1
51
1
51
1
51 50
DATA2000 on CDC WONDER/PC
1
................ ... --- --- --- --- --- --- 51 51 51 51 51 50
22.7 Timely release of national data (percent of
objectives)
Data released within 1 year of collection ............ 1994 65% . . . . . . . . . . . . . . . 67% 46% 49% 23% 28% 100%
Data released between 1–2 years of collection ....... 1994 24% . . . . . . . . . . . . . . . 24% 16% 18% 31% 39% . . .
- - - Data not available. Final objective status:
. . . Category not applicable.
1
Includes the District of Columbia.
2
Total number of objectives is 300.
3
Number of objectives changed from 300 to 319 during the midcourse review.
4
Baselines were not obtained for 9 of 319 objectives.
5
States that have adopted Healthy People 2000 plans.
6
6of21Statistical Notes have been published. See appendix table VIII.
7
Tracking Healthy People 2010 includes 823 measures, 595 of which have complete baseline data and operational definitions.
8
The National Committee on Vital and Health Statistics established a Subcommittee on State and Community Health Statistics. The Subcommittee’s charge (in part) is to work with Federal and
State agencies and appropriate private agencies to review and identify gaps in current health statistics.
9
The Healthy People 2000 Midcourse Review added 111 additional subobjectives for major population groups at highest risk for disease, injury, and disability.
10
As part of the planning process for 2010, data gaps are being identified and mechanisms to address these gaps are being considered.
Met
Toward
Mixed/ no change
Away
Cannot assess
Healthy People 2000 Final Review 309

11
Healtlhy People 2010 is addressing data gaps by requiring data for race, ethnicity, socioeconomic status, and disability measures for population-based objectives and by listing developmental
objectives.
12
1989 data.
13
States that collect inpatient hospital discharge data. The number of States with legislative mandates to collect hospital discharge data was 33 in 1999.
14
Twenty-seven States have at least one racial/ethnic group comprising at least 10 percent of their population; data show number of States that published vital statistics data for these
racial/ethnic groups.
15
Number of all States that published vital statistics data for racial/ethnic groups; racial/ethnic groups may comprise less than 10 percent of State population.
16
Twenty-seven State Health Profiles provide race/ethnic data for some vital statistics measures.
NOTE: Data include revisions and, therefore, may differ from data previously published in these reports and other publications.
Objective number Data source
22.1 CDC, NCHS.
22.2, 22.2a Baseline: National data: OPHS, ODPHP.
State data: Public Health Foundation.
Updates: CDC, NCHS; OPHS, ODPHP.
22.3 CDC, NCHS.
22.4* Subcommittee on State and Community Health Statistics, NCVHS.
CDC, NCHS; OPHS, ODPHP.
22.5 1990 data: PHF.
1989 baselines and updates:
Vital statistics: CDC, NCHS.
Behavioral Risk Factor Surveillance System: CDC, NCCDPHP.
Hospital discharge data: National Association of Health Data Organizations.
Youth Risk Behavior Survey: CDC, NCCDPHP.
22.5a CDC, NCHS.
22.6 1989 baseline: PHF.
1992–97 data:
NETSS: CDC, EPO.
PHLIS: CDC, NCID.
CDC WONDER: CDC, EPO; CDC, NCHS.
22.7 CDC, NCHS.
*Duplicate objective. See full text of objectives following this table.
310 Healthy People 2000 Final Review
Surveillance and
Data Systems
Objectives
22.1: Develop a set of health status
indicators appropriate for Federal, State,
and local health agencies, and establish
use of the set in at least 40 States.
22.2: Identify, and create where
necessary, national data sources to
measure progress toward each of the
year 2000 national health objectives.
22.2a: Identify, and create where
necessary, State-level data for at
least two-thirds of the objectives in
State year 2000 plans in all 50
States.
22.3: Develop and disseminate among
Federal, State, and local agencies
procedures for collecting comparable
data for each of the year 2000 national
health objectives and incorporate these
into Public Health Service data
collection systems.
22.4*: Develop and implement a
national process to identify significant
gaps in the Nation’s disease prevention
and health promotion data, including
data for racial and ethnic minorities,
people with low incomes, and people
with disabilities, and establish
mechanisms to meet these needs.
Duplicate objective: 17.22
22.5: Implement in all States periodic
analysis and publication of data needed
to measure progress toward objectives
for at least 10 of the priority areas of
the national health objectives.
22.5a: Implement in 25 States
periodic analysis and publication of
data needed to measure State
progress toward the national or
State-specific health objectives for
each racial or ethnic group that
makes up at least 10 percent of the
State population.
22.6: Expand in all States systems for
the transfer of health information related
to the national health objectives among
Federal, State, and local agencies.
22.7: Achieve timely release of national
surveillance and survey data needed by
health professionals and agencies to
measure progress toward the national
health objectives.
Healthy People 2000 Final Review 311

Appendix

Appendix
Contents
Technical Notes 316
Age Adjustment 316
Cause-of-Death Terminology and Codes
316
Census Poverty Threshold
316
Data Source Comparability 316
Disparity Measurement
316
Geographic Coverage 317
Hispanic Vital Statistics 317
Measuring Progress Toward the Healthy People 2000 Targets
317
Mortality Data by Race and Hispanic Origin
318
National Health Interview Survey Redesign 318
Sources of Monitoring Data and Information
319
Appendix Tables
I. Healthy People 2010 Lead Agencies and Work Group Coordinators 320
II. Acronyms used in Healthy People 2000 Final Review
328
III. Crosswalk between objectives from Healthy People 2000 to Healthy People 2010
330
IV. Mortality objective cause-of-death categories
340
V. Morbidity codes used for objectives tracked by the National Hospital Discharge Survey
342
VI. Published issues of Healthy People Statistical Notes
343
VII. Special population objectives included in Healthy People 2000
344
Healthy People 2000 Final Review 315

Appendix
Technical Notes
Age Adjustment
Most of the baselines and
monitoring data for the population-based
Healthy People 2000 mortality
objectives are derived from the National
Vital Statistics System (NVSS) operated
by the Centers for Disease Control and
Prevention’s (CDC) National Center for
Health Statistics (NCHS) and are age
adjusted to the 1940 population (see
Appendix table III) (1). Exceptions are
objectives 4.1, 9.3 (except 9.3d), 10.1,
and 10.16. Data for 4.1 and 9.3 (except
9.3d) are crude rates from the National
Highway and Traffic Safety
Administration’s Fatality Analysis
Reporting System (formerly the Fatal
Accident Reporting System—FARS).
Data for 10.1 are crude rates from the
Department of Labor’s Annual Survey
of Occupational Injuries and Illnesses
and Census of Fatal Occupational
Injuries. Baseline data for 10.16,an
objective added during the midcourse
review, are crude rates from the
National Traumatic Occupational
Fatality Surveillance System, CDC.
Update data are crude rates from the
Census of Fatal Occupational Injuries.
For Healthy People 2010, data for
most of the population-based objectives
(including mortality) are age adjusted to
the year 2000 standard population.
Because this is a different standard than
what was used in Healthy People 2000,
it is difficult to compare the
age-adjusted data in Healthy People
2010 objectives to Healthy People 2000
objectives with data that are otherwise
comparable. A complete discussion on
age adjustment to the 2000 standard is
available in Tracking Healthy People
2010 (2).
Cause-of-Death Terminology and
Codes
Twenty-six objectives (excluding
duplicates) in Healthy People 2000 are
tracked using mortality data. For most
of these objectives, the cause-of-death
terminology used in Healthy People
2000 is different from that used in
Health, United States; Vital Statistics of
the United States, Mortality, and other
NCHS publications; in some cases, the
International Classification of Diseases;
Ninth Revision (ICD–9) codes are
different as well (3) (see Appendix
table III).
For five objectives, the terminology
and the codes are different from those
used for similar cause-of-death
categories in NCHS publications. One
example, objective 7.1, concerns
reduction of homicides. Progress toward
this objective is measured using ICD–9
codes E960–E969. NCHS generally uses
‘‘homicide and legal intervention’’
(ICD–9 codes E960–E978), which
includes ‘‘legal intervention’’ or ‘‘police
action.’’ For 14 objectives, only the
terminology differs; the defining ICD–9
identifying codes are the same. For
example, objective 15.2 calls for
reduction in mortality from ‘‘stroke’’;
NCHS tabulation lists use the term
‘‘cerebrovascular diseases’’ (both use
ICD–9 numbers 430–438). Only one
objective, suicide, has the same title and
the same code structure in both uses.
The remaining six mortality objectives
have no comparable category in NCHS
publications. With the exception of heart
disease, the differences between
mortality rates defined by the Healthy
People 2000 ICD–9 categories and those
defined by NCHS rubrics are relatively
small, if not trivial.
Healthy People 2010 baselines use
the International Classification of
Diseases; Ninth Revision (ICD–9,
ICD–9–CM) codes to define baseline
measures. Future tracking data for
Healthy People 2010 will incorporate
the International Classification of
Diseases; Tenth Revision (ICD–10,
ICD–10–CM). Once implemented,
revised coding guidelines, training
materials, and crosswalks between
ICD–9/ICD–9–CM and
ICD–10/ICD–10–CM will be made
available on the NCHS Web site
(http://www.cdc.gov/nchs).
Census Poverty Threshold
Data for subobjectives targeting
family income below the poverty level
are based on definitions originally
developed by the Social Security
Administration. They include a set of
income thresholds that vary by family
size and composition. Families or
individuals with income below the
appropriate thresholds are classified as
below the poverty level. These
thresholds are updated annually by the
U.S. Bureau of the Census. The
weighted-average poverty threshold for
a family of four was $14,335 in 1992,
$14,764 in 1993, $15,141 in 1994,
$15,569 in 1995, $16,036 in 1996,
$16,400 in 1997, $16,660 in 1998, and
$17,029 in 1999 (4).
Data Source Comparability
For some objectives the baseline
data source differs from the source used
to monitor progress. Comparability
between different data sources or even
within the same data source for different
years is not ensured. Comparability can
be compromised by changes in survey
questions, survey systems, survey
methodology, operational definitions,
and analytic techniques. Some of the
most important comparability issues
related to specific objectives are
discussed in the Data Issues section of
the priority area chapters. Other issues
related to tracking the objectives are
addressed in Healthy People Statistical
Notes Number 4, Issues Related to
Monitoring the Year 2000 Objectives
(5). The data source for each Healthy
People 2000 objective is shown at the
end of the summary data table in each
priority area chapter.
Disparity Measurement
A major goal of Healthy People
2000 is to reduce disparity in health
status between special populations (SP)
at high risk for death, disease, or
disability, and the population as a
whole. As appropriate, the 319 main
objectives identified these special
populations as subobjectives. During the
midcourse review and 1995 revisions,
additional SP objectives were included
(6). For these special population
subobjectives, the statistic used to track
the overall objective is, then, considered
the measure for the reference population
(RP). Disparity is a function of the
difference between the relative rates of
change for the SP and RP. In many
instances, different targets were set for
the SP and RP. These targets were set
with the aim of reducing the gap
between these racial and ethnic groups
and the total population, that is,
targeting a greater percent change for
the SP.
A method for measuring disparity
and change in disparity was not
specified in Healthy People 2000 (7). In
preparation for this Final Review,a
working group of agency representatives
from the U.S. Department of Health and
Human Services evaluated several
methods for measuring change in
disparity. In some instances the work
316 Healthy People 2000 Final Review

group found that different methods used
for the same data yielded different
conclusions regarding the widening or
narrowing of disparity.
Ultimately, the ratio of percent
changes was chosen to measure the
change in disparity for several reasons.
As noted above, the targets for SP and
RP were chosen to decrease disparity.
Therefore, if both targets were attained,
the ratio of the percent changes would
be less than 1.0 and the disparity would
be narrowed. The ratio was also chosen
because it is widely used to describe
relative change between two
populations. In measuring disparity for
the special population groups, objectives
that were duplicated in more than one
chapter were counted only once.
The ratio of percent change is
calculated as:
冉
RP
most recent
–RP
baseline
RP
baseline
冊
冉
SP
most recent
–SP
baseline
SP
baseline
冊
The ratio of percent change is not a
measure of the direction of change; it is
a measure of relative change.
Consequently, the ratio can be less than
1.0, indicating a narrowing of the
disparity, if data for the SP indicate that:
SP is moving toward the SP target
at a relative rate greater than the RP is
moving toward the RP target;
SP progress is toward the SP target
but RP is moving away from the RP
target; or
SP is moving away from the SP
target at a relative rate less than the RP
movement.
A conclusion of a widening
disparity, that is, a ratio greater than 1.0,
can result if data for the SP indicate
that:
SP is moving away from the SP
target although the RP is moving toward
the RP target;
SP is moving toward the SP target
at a relative rate less than the RP
movement; or
SP is moving away from the SP
target at a relative rate larger than the
RP movement.
Some special population objectives
or subobjectives were not constructed to
assess disparity. In these cases, disparity
measurement was not applicable. These
include:
SP measure was part of a main
objective and had no RP. Examples of
this include attempting to measure
disparities for women when the main
objective targeted only women, such as
mammogram screening. There is no
reference population.
SP was better than RP at baseline.
In most cases, these objectives are
excluded from the disparity assessment.
Some of these objectives did show a
disparity at the end of the decade.
These disparities will be tracked by
Healthy People 2010 in support of its
goal to eliminate health disparities.
For the other special population
objectives or subobjectives, disparity
measurement was applicable, but in
several instances could not be assessed
(due to data limitations):
SP or RP measure had missing or
incomplete baseline, update, and/or
target data;
RP measure was not comparable to
the SP measure.
To obtain a full picture of the
progress of the SP subobjectives, the
disparity measure can be combined with
the progress quotient calculated for the
subobjective separately. This combined
picture is presented for each SP in the
charts of the ‘‘Reducing Disparity’’
section of the Review. In that section,
the summary of progress is presented
for nine of the special populations:
adolescents and young adults, American
Indian/Alaska Native, Asian or Pacific
Islander, black or African American,
Hispanic or Latino, older adults, people
with disabilities, people with low
socioeconomic status, and women. In
each chart, the pie indicates the
proportion of objectives for which the
disparity of the special population was
eliminated or narrowing, showed no
change, was widening, or could not be
assessed using the ratio of percent
change statistic. Progress toward or
away from the target was then assessed
for those objectives in which the
disparity was either eliminated or
narrowing. The progress is displayed in
the chart as a bar showing the
proportion of these objectives for which
data indicate the special population met
its target, moved either toward or away
from the target, or made no change.
Geographic Coverage
Data used to track the Healthy
People 2000 objectives are, where
available, for the 50 States and the
District of Columbia. In cases where
complete U.S. data are not available,
subnational data (if they exist) are used
as a proxy. For all objectives, data for
U.S. territories are excluded.
Hispanic Vital Statistics
There are nine subobjectives
targeting mortality reduction for
Hispanic populations (4.2c, 4.3b, 7.1d,
9.1d, 9.3g, 9.6g, 16.4b, 17.9c, and
17.9d). For objective 7.1d, the only
subobjective with pre-1990 data, the
1987–89 baseline and tracking data are
based on deaths to residents of selected
States that had data that were at least
90 percent complete on a
place-of-occurrence basis and considered
to be sufficiently comparable. Beginning
with data for 1990 for all Hispanic
subobjectives, the criterion was changed
to include States with data that were at
least 80 percent complete. The number
of States in the mortality reporting area
increased from 18 States and the District
of Columbia in 1987 to 49 States and
the District of Columbia in 1993.
Hispanic origin data for low/very
low birth weight (14.5c,d) and prenatal
care in the first trimester (14.11c) are
based on States that reported Hispanic
parentage on the birth certificate. The
number of States in the natality
reporting area increased from 23 States
and the District of Columbia in 1987 to
all 50 States and the District of
Columbia in 1993. The reporting area
for infant mortality data from the
national linked file of live births and
infant deaths for Puerto Ricans (14.1c, f,
and j) increased from 23 States and the
District of Columbia in 1984 to 49
States and the District of Columbia in
1991. Since 1991, only Oklahoma does
not report Hispanic origin for deaths.
A listing of the States included in
the reporting areas for each year and
more information can be found in
another publication (8).
Measuring Progress Toward the
Healthy People 2000 Targets
A primary purpose of the Healthy
People 2000 Final Review is reporting
the progress the 319 objectives made in
reaching their year 2000 targets by the
end of the decade. The priority area
chapters in this Review contain a bar
chart illustrating the status of each
objective measure in reference to its
year 2000 target. Progress in these
charts is measured using the percentage
of targeted change that was achieved, or
Healthy People 2000 Final Review 317

the ‘‘progress quotient.’’ This is the
same progress measure used in the
Healthy People 2000 Midcourse Review
and 1995 Revisions (6).
The formula for the progress
quotient is:
冉
most recent value – baseline value
year 2000 target – baseline value
冊
⳯ 100
The progress quotient allows for
comparisons to be made between
objectives and between priority areas
regarding the degree of change attained
compared with the change desired, as
reflected in the target set in 1990. There
are, however, some limitations to the
interpretation of these statistics. The
progress quotient reflects the difference
between the data for the baseline year
and the most recent data year only; thus,
fluctuations in the measure over the
decade are not reflected. Also, the
amount of change required to attain the
target differed among objectives.
Therefore, equal progress quotients do
not reflect equal percent changes from
the baseline, which would be calculated
using only the baseline data in the
denominator. Finally, because
information on variability of the
measures was not available for all
objectives, the actual change toward the
target is used to determine progress.
Some of these changes are relatively
small and could be within what could be
expected on the basis of sampling or
random variation. This is particularly
true for small subpopulations, such as
Asians and Pacific Islanders and
American Indians/Alaska Natives.
The priority area bar charts display
progress quotients for each objective
measure. If more than one measure was
used to evaluate an objective, the status
of each measure is displayed separately.
Depending on the data, bar charts depict
progress in one of the following ways:
When the most recent data were the
same as the data from the baseline year,
there was no progress and the progress
quotient is 0 percent. This is noted in
the bar charts as ‘‘no change.’’
When the most recent data fall
between the baseline year data and the
year 2000 target, there was progress,
although the target was not attained. In
this case, the progress quotient is
between 0 and 100 percent, and the bar
is displayed as light green.
When the most recent data are
equal to or better than the year 2000
target, the objective was met or
exceeded. In this case, the progress
quotient is at least 100 percent, and the
bar is displayed as dark green. If the
progress quotient is greater than
100 percent, the actual percent is printed
on the bar; however, graphically the bar
is drawn as 100 percent.
When the most recent data are
worse than the baseline data, the
objective changed in a direction away
from the target, and the progress
quotient is negative. In this case, the bar
is displayed as burgundy. If the negative
change is less than 100 percent, the
actual percent is printed on the bar;
graphically, however, the bar is drawn
as –100 percent.
In several situations, the progress
quotient was not calculated. These
include when:
The baseline year data and/or more
recent data were not available.
Objectives have targets stated as
‘‘slow the rise’’ of the rate of a
particular condition or equal to the
baseline rate. In these cases, a
meaningful progress quotient could not
be calculated; however, in the bar chart
such measures are labeled as ‘‘met’’ or
‘‘away’’ depending on the direction of
the change.
Objectives had a baseline value that
was already ‘‘better’’ than the target
value.
Objectives contained multiple parts
for which data were unavailable. For
instance, for the objectives tracking
clinical counseling, data more recent
than the baseline were available only for
nurse practitioners. Tracking data were
unavailable for the other provider
specialities.
Specific discussion of some of the
objectives for which progress quotients
were not calculated can be found in the
Data Issues section of each priority area
chapter.
Mortality Data by Race and
Hispanic Origin
Death rates by race and Hispanic
origin are based on information from
death certificates (numerators of the
rates) and on population estimates from
the Census Bureau (denominator). Race
and ethnicity information on the death
certificate are reported by the funeral
director as provided by an informant,
often the surviving next of kin, or, in
the absence of an informant, on the
basis of observation. Race and ethnicity
information from the census is by
self-report. To the extent that race and
Hispanic origin are inconsistent between
these two data sources, death rates will
be biased. Studies have shown that
persons self-reported as American
Indian, Asian, or Hispanic on census
and survey records may sometimes be
reported as white or non-Hispanic on
the death certificate, resulting in an
underestimation of deaths and death
rates for the American Indian, Asian,
and Hispanic groups. Bias also results
from undercounts of some population
groups in the census, particularly young
black and white males and elderly
persons, resulting in an overestimation
of death rates. The net effects of
misclassification and under coverage
result in overstated death rates for the
white population and the black
population are estimated to be 1 percent
and 5 percent, respectively; understated
death rates for other population groups
are estimated as follows: American
Indians, 21 percent; Asian or Pacific
Islanders, 11 percent; and Hispanics,
2 percent (9).
Vital event rates for the American
Indian/Alaska Native population shown
in this book are based on the total U.S.
resident population of American Indians
and Alaska Natives as enumerated by
the U.S. Bureau of Census. In contrast
the Indian Health Service calculates
vital event rates for this population
based on U.S. Bureau of Census county
data for American Indians and Alaska
Natives who reside on or near
reservations.
Racial and ethnic classification for
infant deaths is substantially improved
by using the linked birth and infant
death file, which uses the race of the
mother as self-reported on the birth
certificate instead of the race of child as
reported on the death certificate. The
infant mortality rates for Puerto Ricans
and American Indians/Alaska Natives
shown in objectives 14.1b, c, f, i, and j
used data from the linked file.
National Health Interview Survey
Redesign
The National Health Interview
Survey (NHIS) is a continuing
nationwide sample survey in which data
are collected through personal household
interviews. The NHIS is the primary
data source for Healthy People 2000,
tracking 20 percent of the objectives.
In 1997, the collection methodology
changed from paper and pencil
questionnaires to computer-assisted
personal interviewing (CAPI). The
318 Healthy People 2000 Final Review
NHIS questionnaire was also revised
extensively in 1997. The survey changed
from two components (Core and Topic
Supplements) to now consisting of three
components (Family Core, Sample Adult
Core, Sample Child Core) and periodic
and topical modules. In some instances,
basic concepts measured in the NHIS
changed and in other instances the same
concepts were measured in a different
way. While some questions remain the
same over time, they may be preceded
by different questions or topics. For
some questions, there was a change in
the reference period for reporting an
event or condition (8).
Because of this extensive redesign
of the questionnaire in 1997 and the
introduction of the CAPI method of data
collection, data from 1997 and later
years may not be comparable with
earlier years. When data are not
comparable because of these survey
revisions, the data from 1997 and later
years have been omitted and denoted
with a footnote ‘‘‡.’’
Sources of Monitoring Data and
Information
Relevant Internet sites include the
following:
The Healthy People 2000 home
page
(http://odphp.osophs.dhhs.gov/pubs/
hp2000): Contains connections to many
activities related to Healthy People 2000
including lead agency contacts for the
priority areas, progress review reports,
the Healthy People 2000 Consortium,
and more.
The National Health Information
Center (NHIC) home page
(http://nhic-nt.health.org): Serves as a
health information referral service that
enables health professionals and
consumers who have health questions to
contact organizations that are best able
to provide answers. Many documents
related to Healthy People 2000 can be
located at this site. NHIC was
established in 1979 by the Office of
Disease Prevention and Health
Promotion (ODPHP), Office of Public
Health and Science, Office of the
Secretary, U.S. Department of Health
and Human Services.
The NCHS home page
(http://www.cdc.gov/nchs) and the
NCHS Healthy People home page
(http://www.cdc.gov/nchs/hphome.htm):
The NCHS site provides statistical
information on vital events as well as
information on health status, lifestyle
and exposure to unhealthy influences,
the onset and diagnosis of illness and
disability, and the use of health care.
The NCHS Healthy People site provides
links to previous Healthy People 2000
Reviews, data presentations from past
Healthy People 2000 Progress Reviews,
and provides other related links to
organizations and data systems
associated with monitoring Healthy
People 2000. Data for recent years are
available for the Health Status Indicators
(HSIs) and the Priority Data Needs
(PDNs) in files that can be downloaded
through the CDC FTP-server at
http://www.cdc.gov/nchswww/datawh/
datawh.htm.
The Healthy People 2010 home
page
(http://www.health.gov/healthypeople):
Contains links to many Healthy People
2010 activities including related
publications (Healthy People 2010 with
Understanding and Improving Health
and Objectives for Improving Health,
and Tracking Healthy People 2010),
Leading Health Indicators, lead agency
contacts for the focus areas, and the
Healthy People Consortium.
The DATA2010 home page
(http://wonder.cdc.gov/data2010):
Contains the most recent data tracking
the Healthy People 2010 objectives,
provides data by State, race and
ethnicity, gender, socioeconomic status,
disability status, and geographic
location.
A valuable resource for public
health professionals wishing to measure
and track data comparable to the
national Healthy People 2000 objectives
is presented as a series of publications
entitled Healthy People Statistical
Notes. This series contains information
on the HSIs, monitoring issues,
operational definitions, and other issues
related to tracking the Healthy People
2000 objectives. For a list of these
publications, see Appendix table VI. The
full text of Healthy People Statistical
Notes numbers 6 through the present
can be found on the NCHS Healthy
People home page
(http://www.cdc.gov/nchs/products/pubs/
pubd/hp2k/hp2k.htm#Statistical Notes).
NCHS has also presented a course
entitled ‘‘Measuring the Healthy People
2000 Objectives’’ through the Applied
Statistics Training Institute (ASTI) in
several locations around the country.
This course targeted health professionals
and others working in areas of public
health in government agencies and
private organizations, addressed specific
measurement issues related to
monitoring progress toward selected
Healthy People 2000 objectives and
HSIs. A number of objectives that
presented unusual problems or required
the use of complex algorithms were
discussed (for example, years of healthy
life; light, moderate, vigorous physical
activity; overweight prevalence; and air
quality). Data comparability, the
International Classification of Disease
(ICD) codes for mortality data,
computation of age-adjusted death rates,
and a demonstration of using statistical
data on the Internet were also included.
References
1. Curtain LR, Klein RJ. Direct standardization
(age-adjusted death rates). Healthy people
statistical notes; no 6. Hyattsville, Maryland:
National Center for Health Statistics. 1995.
2. Department of Health and Human Services.
Tracking healthy people 2010. Washington: U.S.
Government Printing Office. 2000.
3. World Health Organization. Manual of the
International Statistical Classification of Diseases,
Injuries, and Causes of Death, based on the
recommendations of the Ninth Revision
Conference, 1975. Geneva: World Health
Organization. 1977.
4. Bureau of the Census. Current population
reports, Series P60. Income, poverty, and
validation of non-cash benefits. Washington.
Published annually.
5. Wilson R, Freedman MA, Klein RJ. Issues
related to monitoring the healthy people 2000
objectives. Healthy people statistical notes; no 4.
Hyattsville, Maryland: National Center for Health
Statistics. 1993.
6. Department of Health and Human Services.
Healthy people 2000 midcourse review and 1995
revisions. Washington: Public Health Service.
1995.
7. Department of Health and Human Services.
Healthy people 2000: National health promotion
and disease prevention objectives. Washington:
Public Health Service. 1991.
8. National Center for Health Statistics. Health,
United States, 2000. Hyattsville, Maryland: Public
Health Service. 2000.
9. Rosenberg HM, Maurer JD, Sorlie PD, Johnson
NJ, et al. Quality of death rates by race and
Hispanic origin: A summary of current research,
1999. National Center for Health Statistics. Vital
Health Stat 2(128). 1999.
Healthy People 2000 Final Review 319

Table I. Healthy People 2010 Lead Agencies and Work Group Coordinators
1. Access to Quality Health Services
David Atkins, M.D., M.P.H.
Medical Officer
Center for Practice and Technology Assessment
Agency for Healthcare Research and Quality
6010 Executive Boulevard, Suite 300
Rockville, MD 20852
(301) 594-4016/Fax (301) 594-4027
E-mail: [email protected]
Melissa Clarke, M.P.A.
Senior Health Policy and Program Analyst
Healthy People 2000/2010 Coordinator
Office of Planning, Evaluation, and Legislation
Health Resources and Services Administration
Parklawn Building, Room 14-33
5600 Fishers Lane
Rockville, MD 20857
(301) 443-5277/Fax (301) 443-9270
E-mail: [email protected]
Paul Nannis, M.S.W.
Director
Office of Planning, Evaluation and Legislation
Health Resources and Services Administration
Parklawn Building, Room 14-33
5600 Fishers Lane
Rockville, MD 20857
(301) 443-2460/Fax (301) 443-9270
E-mail: [email protected]
Kathryn Rickard, M.P.A.
Program Analyst
Center for Practice and Technology Assessment
Agency for Healthcare Research and Quality
6010 Executive Boulevard, Suite 300
Rockville, MD 20852
(301) 594-2431/Fax (301) 594-4027
E-mail: [email protected]
2. Arthritis, Osteoporosis, and Chronic Back Conditions
Charles G. Helmick, M.D.
Medical Epidemiologist
Health Care and Aging Studies Branch
Division of Adult and Community Health
National Center for Chronic Disease Prevention and Health Promotion
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K45
Atlanta, GA 30341
(770) 488-5456/Fax (770) 488-5964
E-mail: [email protected]
Reva Lawrence, M.P.H.
Epidemiologist/Data Systems Program Officer
National Institute of Arthritis and Musculoskeletal and Skin Diseases
National Institutes of Health
45 Center Drive, MS 6500 Bethesda, MD 20892
(301) 594-5014/Fax (301) 402-2406
E-mail: [email protected]
320 Healthy People 2000 Final Review
Paul Scherr, Ph.D., D.Sc.
Supervisory Epidemiologist
National Center for Chronic Disease
Prevention and Health Promotion
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K45
Atlanta, GA 30341
(770) 488-5454/Fax (770) 488-5964
E-mail: [email protected]
3. Cancer
Barry Portnoy, Ph.D.
Cancer Planning and Program Officer
National Cancer Institute
National Institutes of Health
Building 31, Room 10A49
31 Center Drive, MSC 2580
Bethesda, MD 20892-2580
(301) 496-9569/Fax (301) 496-9931
E-mail: [email protected]
Karen Richard, M.P.A.
Public Health Advisor
National Center for Chronic Disease
Prevention and Health Promotion
Division of Cancer Prevention and Control
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K64
Atlanta, GA 30341
(770) 488-4737/Fax (770) 488-4760
E-mail: [email protected]
4. Chronic Kidney Disease
Lawrence Agodoa, M.D.
Program Director
Division of Kidney Urologic and Hematologic Diseases
National Institute of Diabetes and Digestive and Kidney
Diseases
National Institutes of Health
Building 45, Room 6AS-13B
45 Center Drive
Bethesda, MD 20892-6600
(301) 594-7717/Fax (301) 480-3510
E-mail: [email protected]
5. Diabetes
Benjamin Burton, Ph.D.
Emeritus Scientist
National Institute of Diabetes and Digestive and Kidney
Diseases
National Institutes of Health
Building 45, Room 6AN38J
45 Center Drive
Bethesda, MD 20892
(301) 594-8867/Fax (301) 480-4237
Email: [email protected]

Table I. Healthy People 2010 Lead Agencies and Work Group Coordinators—Con.
Bill Foster, M.D.
Senior Staff Physician
National Institute of Diabetes and Digestive and Kidney
Diseases
National Institutes of Health
Building 31, Room 9A21
45 Center Drive
Bethesda, MD 20892
(301) 435-2991/Fax (301) 396-2830
E-mail: [email protected]
Frank M. Vinicor, M.D.
Director
Division of Diabetes Translation
National Center for Chronic Disease
Prevention and Health Promotion
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K10
Atlanta, GA 30341-3724
(770) 488-5000/Fax (770) 488-5966
E-mail: [email protected]
6. Disability and Secondary Conditions
Chris Kochtitzky, M.S.P.
Policy Analyst
National Center for Environmental Health
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS F29
Atlanta, GA 30341-3724
(770) 488-7114/Fax (770) 488-7024
E-mail: [email protected]
Don Lollar, Ed.D.
Director
Office on Disability and Health
National Center for Environmental Health
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS F29
Atlanta, GA 30341-3724
(770) 488-7094/Fax (770) 488-7075
E-mail: [email protected]
Katherine Seelman, Ph.D.
Director
National Institute on Disability and Rehabilitation Research
U.S. Department of Education
Switzer Building, Room 3060
330 C Street, S.W.
Washington, DC 20202
(202) 205-8134/Fax (202) 205-8997
E-mail: [email protected]
Lisa Sinclair, M.P.H.
Health Policy Analyst
Office on Disability and Health
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS F29
Atlanta, GA 30341-3724
(770) 488-7667
Email: [email protected]
Healthy People 2000 Final Review 321
7. Educational and Community-Based Programs
Melissa Clarke, M.P.A.
Senior Health Policy and Program Analyst
Healthy People 2000/2010 Coordinator
Office of Planning, Evaluation and Legislation
Health Resources and Services Administration
Parklawn Building, Room 14-33
5600 Fishers Lane
Rockville, MD 20857
(301) 443-5277/Fax (301) 443-9270
E-mail: [email protected]
Catherine A. Hutsell, M.P.H.
Health Education Specialist
Community Health and Program Services
National Center for Chronic Disease
Prevention and Health Promotion
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K30
Atlanta, GA 30341-3717
(770) 488-5438/FAX (770) 488-5974
E-mail: [email protected]
Brick Lancaster, M.A., CHES
Associate Director for Health Education
Practice and Policy
National Center for Chronic Disease Prevention and Health
Promotion
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K45
Atlanta, GA 30341-3724
(770) 488-5269/Fax (770) 488-5964
E-mail: [email protected]
Paul Nannis, M.S.W.
Director
Office of Planning, Evaluation, and Legislation
Health Resources and Services Administration
Parklawn Building, Room 14-33
5600 Fishers Lane
Rockville, MD 20857
(301) 443-2460/Fax (301) 443-9270
E-mail: [email protected]
8. Environmental Health
David Evans
Senior Program Analyst
Office of Policy and External Affairs
Agency for Toxic Substances and Disease Registry
1600 Clifton Road, N.E., MS E60
Atlanta, GA 30333
(404) 639-0500/Fax (404) 639-0522
E-mail: [email protected]
David Homa, Ph.D., M.P.H.
Epidemiologist
Air Pollution and Respiratory Health Branch
Division of Environmental Hazards and Health Effects
National Center for Environmental Health
Centers for Disease Control and Prevention
1600 Clifton Road, N.E., MS E17
Atlanta, GA 30333
(404) 639-2544/Fax (404) 639-2560
E-mail: [email protected]

Table I. Healthy People 2010 Lead Agencies and Work Group Coordinators—Con.
William Jirles, M.P.H.
Program Analyst
Office of Policy, Planning, and Evaluation
National Institute of Environmental Health Sciences
National Institutes of Health
Building 101, Room B224
111 Alexander Drive, MS B2-08
Research Triangle Park, NC 27709
(919) 541-2637/Fax (919) 541-4737
E-mail: [email protected]
Chris Kochtitzky, M.S.P.
Policy Analyst
Office of Planning, Evaluation, and Legislation
National Center for Environmental Health
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS F29
Atlanta, GA 30341-3724
(770) 488-7114/Fax (770) 488-7024
E-mail: [email protected]
Mark McClanahan, Ph.D.
Health Studies Branch
Division of Environmental Hazards and Health Effects
National Center for Environmental Health
Centers for Disease Control and Prevention
1600 Clifton Road, N.E., MS E23
Atlanta, GA 30333
(404) 639-2562/Fax (404) 639-2565
E-mail: [email protected]
Sheila Newton, Ph.D.
Director
Office of Policy, Planning, and Evaluation
National Institute of Environmental Health Sciences
National Institutes of Health
Building 101, Room B250
111 Alexander Drive, MS B2-08
Research Triangle Park, NC 27709
(919) 541-3484/Fax (919) 541-4737
E-mail: [email protected]
Carol Pertowski, M.D.
Associate Director for Science
Division of Environmental Hazards and Health Effects
National Center for Environmental Health
Centers for Disease Control and Prevention
1600 Clifton Road, N.E., MS G317
Atlanta, GA 30333
(404) 639-2500/Fax (404) 639-2555
E-mail: [email protected]
John Schelp, M.P.A.
Office of Policy, Planning, and Evaluation
National Institute of Environmental Health Sciences
National Institutes of Health
Building 101, Room B218
111 Alexander Drive, MS B2-08
Research Triangle Park, NC 27709
(919) 541-5723/Fax (919) 541-2260
E-mail: [email protected]
322 Healthy People 2000 Final Review
9. Family Planning
Evelyn Kappeler
Senior Policy Analyst
Office of Population Affairs
Office of Public Health and Science
Suite 200 West
4350 East West Highway
Bethesda, MD 20814
(301) 594-7608/Fax (301) 594-5980
E-mail: [email protected]
10. Food Safety
Elisa L. Elliot, Ph.D.
Microbiologist
Center for Food Safety and Applied Nutrition
Food and Drug Administration
200 C Street, S.W., HFS-615
Washington, DC 20204
(202) 205-4018/Fax (202) 260-0136
E-mail: [email protected]
Ruth Etzel, M.D., Ph.D.
Director
Division of Epidemiology and Risk Assessment
Food Safety and Inspection Service
U.S. Department of Agriculture
Franklin Court Building, Room 3718
1400 Independence Avenue, S.W.
Washington, DC 20250
(202) 501-7373/Fax (202) 501-6982
E-mail: [email protected]
11. Health Communication
Cynthia Baur, Ph.D.
Policy Advisor, Interactive Health
Communication
Office of Disease Prevention and Health Promotion
Office of Public Health and Science
200 Independence Avenue, S.W., Room 738G
Washington, DC 20201
(202) 205-2311/Fax (202) 205-0463
E-mail: [email protected]
Mary Jo Deering, Ph.D.
Director
Health Communication and Telehealth Staff
Office of Disease Prevention and Health Promotion
Office of Public Health and Science
200 Independence Avenue, S.W., Room 738G
Washington, DC 20201
(202) 260-2652/Fax (202) 205-0463
E-mail: [email protected]

Table I. Healthy People 2010 Lead Agencies and Work Group Coordinators—Con.
12. Access to Quality Health Services
Robinson Fulwood, Ph.D., M.S.P.H.
Senior Manager for Public Health Program Development
Office of Prevention, Education, and Control
National Heart, Lung, and Blood Institute
National Institutes of Health
Building 31, Room 4A03
31 Center Drive MSC 2480
Bethesda, MD 20892-2480
(301) 496-0554/Fax (301) 480-4907
E-mail: [email protected]
Kurt Greenlund, Ph.D.
National Center for Chronic Disease
Prevention and Health Promotion
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K47
Atlanta, GA 30341-3717
(770) 488-2572/Fax (770) 488-8151
E-mail: [email protected]
Claude Lenfant, M.D.
Director
National Heart, Lung, and Blood Institute
National Institutes of Health
31 Center Drive MSC 2486
Bethesda, MD 20892-2486
(301) 496-5166/Fax (301) 480-4907
Zhi-Jie Zheng, M.D., Ph.D.
National Center for Chronic Disease
Prevention and Health Promotion
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K47
Atlanta, GA 30341-3717
(770) 488-8058/Fax (770) 488-8151
E-mail: [email protected]
13. HIV
Emily DeCoster, M.P.H.
Office of Policy and Program Development
HIV/AIDS Bureau
Health Resources and Services Administration
Parklawn Building, Room 7-20
5600 Fishers Lane
Rockville, MD 20857
(301) 443-1381/Fax (301) 443-3323
E-mail: [email protected]
Gena Hill, M.P.H.
Program Analyst
Office of Planning, Policy, and Coordination
National Center for HIV, STD, and TB Prevention
Centers for Disease Control and Prevention
1600 Clifton Road, N.E., MS E07
Atlanta, GA 30333
(404) 639-8008/Fax (404) 639-8600
E-mail: [email protected]
Healthy People 2000 Final Review 323
Eva Margolies-Seiler
Associate Director for Planning, Policy, and Coordination
National Center for HIV, STD, and TB Prevention
Centers for Disease Control and Prevention
1600 Clifton Road, N.E., MS E07
Atlanta, GA 30333
(404) 639-8008/Fax (404) 639-8600
E-mail: [email protected]
14. Immunization and Infectious Diseases
Jennifer Brooks, M.P.H.
Program Analyst
Office of Policy, Planning, and Legislation
National Center for Infectious Diseases
Centers for Disease Control and Prevention
1600 Clifton Road, N.E., MS C12
Atlanta, GA 30333
(404) 639-4915/Fax (404) 639-2715
E-mail: [email protected]
James Hughes, M.D.
Director
National Center for Infectious Diseases
Centers for Disease Control and Prevention
1600 Clifton Road, N.E., MS C12
Atlanta, GA 30333
(404) 639-3401/Fax (404) 639-3039
E-mail: [email protected]
Martin Landry
Associate Director
Planning, Evaluation, and Legislation
National Immunization Program
Centers for Disease Control and Prevention
1600 Clifton Road, N.E., MS E05
Atlanta, GA 30333
(404) 639-8200/Fax (404) 639-8626
E-mail: [email protected]
Walter Orenstein, M.D.
Director
National Immunization Program
Centers for Disease Control and Prevention
1600 Clifton Road, N.E., MS E05
Atlanta, GA 30333
(404) 639-8200/Fax (404) 639-8626
E-mail: [email protected]
Rosemary Ramsey
Associate Director
Policy, Planning, and Legislation
National Center for Infectious Diseases
Centers for Disease Control and Prevention
1600 Clifton Road, N.E., MS C12
Atlanta, GA 30333
(404) 639-3484/Fax (404) 639-2715
E-mail: [email protected]

Table I. Healthy People 2010 Lead Agencies and Work Group Coordinators—Con.
Nicole Smith, M.P.H., M.P.P.
Program Analyst
Office of Planning, Evaluation, and Legislation
National Immunization Program
Centers for Disease Control and Prevention
1600 Clifton Road, N.E., MS E05
Atlanta, GA 30333
(404) 639-8711/Fax (404) 639-8626
E-mail: [email protected]
15. Injury and Violence Prevention
LaTanya Butler
Deputy Director
Division of Unintentional Injury
National Center for Injury Prevention and Control
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K63
Atlanta, GA 30341-3724
(770) 488-4652/Fax (770) 488-1317
E-mail: [email protected]
Alex Crosby, M.D., M.P.H.
Medical Epidemiologist
Division of Violence Prevention
National Center for Injury Prevention and Control
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K60
Atlanta, GA 30341
(770) 488-4272/Fax (770) 488-4349
E-mail: [email protected]
Tim W. Groza, M.P.A.
Public Health Advisor
National Center for Injury Prevention and Control
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K63
Atlanta, GA 30341-3724
(770) 488-4676/Fax (770) 488-1317
E-mail: [email protected]
Martha Highsmith
Program Analyst
Division of Violence Prevention
National Center for Injury Prevention and Control
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K60
Atlanta, GA 30341-3724
(770) 488-4276/Fax (770) 488-4349
E-mail: [email protected]
Laura Yerdon Martin
Assistant Deputy for Policy and Planning
Division of Violence Prevention
National Center for Injury Prevention and Control
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K60
Atlanta, GA 30341-3724
(770) 488-4276/Fax (404) 488-4349
E-mail: [email protected]
324 Healthy People 2000 Final Review
16. Maternal, Infant, and Child Health
Isabella Danel, M.D., M.Sc.
Deputy Chief
Pregnancy and Infant Health Branch
Division of Reproductive Health
National Center for Chronic Disease Prevention and Health
Promotion
4770 Buford Highway, N.E.
Atlanta, GA 30341
(770) 488-5187/Fax (770) 488-5628
E-mail: [email protected]
Charlotte Dickinson, Ph.D.
Planning and Communications Officer
National Center for Environmental Health
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS F34
Atlanta, GA 30341-3724
(770) 488-7155/Fax (770) 488-7156
E-mail: [email protected]
Stella Yu, Sc.D., M.P.H.
Statistician
Maternal and Child Health Bureau
Health Resources and Services Administration
Parklawn Building, Room 18A-55
5600 Fishers Lane
Rockville, MD 20857
(301) 443-0695/Fax (301) 443-4842
E-mail: [email protected]
17. Medical Product Safety
Linda Brophy, R.N., M.N., M.P.H.
Special Assistant
Office of Post-Marketing Drug Risk Assessment
Center for Drug Evaluation and Research
Food and Drug Administration
Parklawn Building, MS HFD 400
5600 Fishers Lane
Rockville, MD 20857
(301) 827-3193/Fax (301) 443-9664
E-mail: brophyl@cder.fda.gov
18. Mental Health and Mental Disorders
Michele Edwards, M.S., A.C.S.W.
Healthy People Coordinator
Center for Mental Health Services
Substance Abuse and Mental Health Services Administration
Parklawn Building, Room 17C-06
5600 Fishers Lane
Rockville, MD 20857
(301) 443-7790 x8/Fax (301) 443-7912
E-mail: [email protected]

Table I. Healthy People 2010 Lead Agencies and Work Group Coordinators—Con.
Doreen S. Koretz, Ph.D.
Associate Director for Prevention
Chief, Developmental Psychopathology
and Prevention Research Branch
National Institute of Mental Health
National Institutes of Health
Neuroscience Center Building, Room 6196
6001 Executive Boulevard
Bethesda, MD 20892-9617
(301) 443-5944/Fax (301) 480-4415
E-mail: [email protected]
19. Nutrition and Overweight
Nancy T. Crane, M.S., M.P.H., R.D.
Nutritionist
Division of Nutrition Science and Policy
Office of Nutritional Products, Labeling, and
Dietary Supplements (HFS-830)
Center for Food Safety and Applied Nutrition
Food and Drug Administration
200 C Street, S.W.
Washington, DC 20204
(202) 205-5615/Fax (202) 205-5532
E-mail: [email protected]
Van S. Hubbard, M.D., Ph.D.
Director, NIH Division of Nutrition
Research Coordination, and Chief,
Nutritional Sciences Branch
National Institute of Diabetes and
Digestive and Kidney Diseases
National Institutes of Health
Rockledge 1, Suite 8048
6705 Rockledge Drive MSC 7973
Bethesda, MD 20892-7973
(301) 594-8822/Fax (301) 480-3768
Christine J. Lewis, Ph.D., R.D.
Director
Office of Nutritional Products, Labeling, and
Dietary Supplements (HFS-800)
Center for Food Safety and Applied Nutrition
Food and Drug Administration
200 C Street, S.W.
Washington, DC 20204
(202) 205-4561/Fax (202) 205-5295
E-mail: [email protected]
Pamela E. Starke-Reed, Ph.D.
Deputy Director
NIH Division of Nutrition Research
Coordination
National Institute of Diabetes and Digestive
and Kidney Diseases
National Institutes of Health
Rockledge 1, Suite 8048
6705 Rockledge Drive, MSC 7973
Bethesda, MD 20892-7973
(301) 594-8805/Fax (301) 480-3768
E-mail: [email protected]
Healthy People 2000 Final Review 325
20. Occupational Safety and Health
Regina Pana-Cryan
Senior Service Fellow
Office of Policy and Legislation
National Institute for Occupational Safety and Health
Centers for Disease Control and Prevention
200 Independence Avenue, S.W., Room 715H
Washington, DC 20201
(202) 401-3741/Fax (202) 260-4464
E-mail: [email protected]
21. Oral Health
Alice M. Horowitz, Ph.D.
Senior Scientist
National Institute of Dental and Craniofacial Research
National Institutes of Health
Building 45, Room 3AN-44B
45 Center Drive, MSC 6401
Bethesda, MD 20892-6401
(301) 594-5391/Fax (301) 480-8254
E-mail: [email protected]
Candace M. Jones, M.P.H.
Health Resources and Services Administration
Parklawn Building, Room 6A-30
5600 Fishers Lane
Rockville, MD 20857
(301) 443-1106/Fax (301) 594-6610
E-mail: [email protected]
Stuart A. Lockwood, D.M.D., M.P.H.
Dental Officer
Surveillance Investigation Research Branch
Division of Oral Health
National Center for Chronic Disease
Prevention and Health Promotion
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS F10
Atlanta, GA 30341
(770) 488-6067/Fax (770) 488-6080
E-mail: [email protected]
22. Physical Activity and Fitness
Carol Macera, Ph.D.
Senior Epidemiologist
Division of Nutrition and Physical Activity
National Center for Chronic Disease Prevention and Health
Promotion
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K46
Atlanta, GA 30341-3724
(770) 488-5018/Fax (770) 488-5473
E-mail: [email protected]
Christine Spain
Director
Research, Planning, and Special Projects
President’s Council on Physical Fitness and Sports
Office of Public Health and Science
200 Independence Avenue, S.W., Room 731
Washington, DC 20201
(202) 690-5148/Fax (202) 690-5211
E-mail: [email protected]

Table I. Healthy People 2010 Lead Agencies and Work Group Coordinators—Con.
23. Public Health Infrastructure
Carol Roddy, J.D.
Senior Advisor to the Administrator
Center for Public Health Practice
Health Resources and Services Administration
Parklawn Building, Room 14-15
5600 Fishers Lane
Rockville, MD 20857
(301) 443-4034/Fax (301) 443-0192
E-mail: [email protected]
Diane Rodill, Ph.D.
Center for Public Health Practice
Health Resources and Services Administration
Parklawn Building, Room 14-15
5600 Fishers Lane
Rockville, MD 20857
(301) 443-0062/Fax (301) 443-0192
E-mail: [email protected]
Pomeroy Sinnock, Ph.D.
Division of Public Health
Public Health Practice Program Office
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K39
Atlanta, GA 30341-3724
(770) 488-2469/Fax (770) 488-2489
E-mail: [email protected]
24. Respiratory Diseases
Robinson Fulwood, Ph.D., M.S.P.H.
Senior Manager for Public Health Program Development
Office of Prevention, Education, and Control
National Heart, Lung, and Blood Institute
National Institutes of Health
Building 31, Room 4A03
31 Center Drive MSC 2480
Bethesda, MD 20892-2480
(301) 496-0554/Fax (301) 480-4907
E-mail: [email protected]
William Jirles, M.P.H.
Office of Policy, Planning, and Evaluation
National Institute of Environmental Health Sciences
National Institutes of Health
Building 101, Room B224
111 Alexander Drive, MS B2-08
Research Triangle Park, NC 27709
(919) 541-2637/Fax (919) 541-4737
E-mail: [email protected]
Carol Johnson, M.P.H.
Air Pollution and Respiratory Health Branch
Division of Environmental Hazards and Health Effects
National Center for Environmental Health
Centers for Disease Control and Prevention
1600 Clifton Road, N.E., MS E17
Atlanta, GA 30333
(404) 639-2556/Fax (404) 639-2560
E-mail: [email protected]
326 Healthy People 2000 Final Review
Marshall Plaut, M.D.
Chief
Allergic Mechanisms Section
Division of Allergy, Immunology, and Transplantation
National Institute of Allergy and Infectious Diseases
National Institutes of Health
6700-B Rockledge Drive, Room 5146
Bethesda, MD 20892-7640
(301) 496-8973/Fax (301) 402-2571
E-mail: [email protected]
Stephen Redd, M.D.
Chief
Air Pollution and Respiratory Health Branch
Division of Environmental Hazards and Health Effects
National Center for Environmental Health
Centers for Disease Control and Prevention
1600 Clifton Road, N.E., MS E17
Atlanta, GA 30341
(404) 639-2549/Fax (404) 639-2560
E-mail: [email protected]
Daniel Rotrosen, M.D.
Director
Division of Allergy, Immunology, and Transplantation
National Institute of Allergy and Infectious Diseases
National Institutes of Health
6700-B Rockledge Drive, Room 5142
Bethesda, MD 20892-7640
(301) 496-1886/Fax (301) 402-2571
E-mail: [email protected]
25. Sexually Transmitted Diseases
Dana M. Shelton
Assistant Director for Policy, Planning, and External Relations
Division of STD Prevention
National Center for HIV, STD, and TB Prevention
Centers for Disease Control and Prevention
1600 Clifton Road, N.E., MS E02
Atlanta, GA 30333
(404) 639-8260/Fax (404) 639-8608
E-mail: [email protected]
26. Substance Abuse
Wendy Davis
Special Assistant to the Director
Office of Policy and Planning
Center for Substance Abuse Prevention
Substance Abuse and Mental Health Services Administration
Rockwall II Building, Room 950
5600 Fishers Lane
Rockville, MD 20857
(301) 443-9913/Fax (301) 443-6394
E-mail: [email protected]
Susan Farrell, Ph.D.
Acting Deputy Director
Division of Biometry and Epidemiology
National Institute on Alcohol Abuse and Alcoholism
National Institutes of Health
Willco Building, Room 514
6000 Executive Boulevard
Bethesda, MD 20892-7005
(301) 443-1274/Fax (301) 443-7043
E-mail: [email protected]

Table I. Healthy People 2010 Lead Agencies and Work Group Coordinators—Con.
James D. Colliver, Ph.D.
Epidemiology Research Branch, DESPR
National Institute on Drug Abuse
National Institutes of Health
Suite 5153 MSC 9589
6001 Executive Boulevard
Bethesda, MD 20892-9589
(301) 402-1846/Fax (301) 443-2636
E-mail: [email protected]
Ann Mahony, M.P.H.
Senior Program Management Officer
Office of Policy, Coordination, and Planning
Center for Substance Abuse Treatment
Substance Abuse and Mental Health
Services Administration
Rockwall II Building, Room 619
5600 Fishers Lane
Rockville, MD 20857
(301) 443-7924/Fax (301) 480-6077
E-mail: [email protected]
27. Tobacco Use
Karil Bialostosky, M.S.
Senior Health Policy Analyst
Office on Smoking and Health
Centers for Disease Control and Prevention
200 Independence Ave, S.W., Room 317-B
Washington, DC 20201
(202) 205-9231/Fax (202) 205-8313
E-mail: [email protected]
28. Vision and Hearing
Marin P. Allen, Ph.D.
Chief
Office of Health Communication
and Public Liaison
National Institute on Deafness
and Other Communication Disorders
National Institutes of Health
Building 31, Room 3C35
31 Center Drive, MSC 2320
Bethesda, MD 20892-2320
(301) 496-7243 or 402-0252/Fax (301) 402-0018
Email: [email protected]
Judith Cooper, Ph.D.
Chief, Scientific Programs Branch
National Institute on Deafness and Other
Communication Disorders
National Institutes of Health
EPS400C, MSC 7180
6120 Executive Boulevard
Bethesda, MD 20892-7180
(301) 496-5061/Fax (301) 402-6251
E-mail: [email protected]
Michael Davis
Associate Director for Science Policy and Legislation
National Eye Institute
National Institutes of Health
Building 31, Room 6A23
9000 Rockville Pike
Bethesda, MD 20892-2510
(301) 496-4308/Fax (301) 402-3799
E-mail: [email protected]
Healthy People 2000 Final Review 327
Rosemary Janiszewski, M.S., CHES.
Chief
Scientific Programs Branch
National Eye Institute
National Institutes of Health
Building 31, Room 6A32
9000 Rockville Pike
Bethesda, MD 20892-2510
(301) 496-5248/Fax (301) 402-1065
E-mail: [email protected]
Adolescent Health
Trina Anglin, M.D., Ph.D.
Chief, Office of Adolescent Health
Maternal and Child Health Bureau
Health Resources and Services Administration
Parklawn Building, Room 18A-39
5600 Fishers Lane
Rockville, MD 20857
(301) 443-4291/Fax (301) 443-1296
Email: [email protected]
Casey Hannan, M.P.H.
Health Education Specialist
Division of Adolescent and School Health
National Center for Chronic Disease
Prevention and Health Promotion
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K29
Atlanta, GA 30341-3724
(770) 488-3190/Fax (770) 488-3110
Email: [email protected]
Lloyd J. Kolbe, Ph.D.
Director, Division of Adolescent and School Health
National Center for Chronic Disease
Prevention and Health Promotion
Centers for Disease Control and Prevention
4770 Buford Highway, N.E., MS K29
Atlanta, GA 30341-3724
(770) 488-3254/Fax (770) 488-3110
Email: [email protected]
Women’s Health
Theresa Brown
Staff Assistant
DHHS Office on Women’s Health
200 Independence Avenue, S.W., Room 728F
Washington, DC 20201
(202) 690-7650/Fax (202) 260-6537
Email: [email protected]
Miryam Granthon
Healthy People Consortium Coordinator
Office of Disease Prevention and Health Promotion
200 Independence Avenue, S.W., Room 738G
Washington, DC 20201
(202) 690-6245/Fax (202) 205-9478
Email: [email protected]
Suzanne Haynes, Ph.D.
Assistant Director for Science
DHHS Office on Women’s Health
200 Independence Avenue, S.W., Room 728F
Washington, DC 20201
(202) 690-7650/Fax (202) 260-6537
Email: [email protected]

Table II. Acronyms used in Healthy People 2000 Final Review
Acronyms Agency/Organization
ACS................................ American Cancer Society
AHA................................ American Hospital Association
AIRS................................ Aerometric Information Retrieval System
ALA ................................ American Lung Association
ATSDR .............................. Agency for Toxic Substances and Disease Registry
BHPr ............................... Bureau of Health Professions
BJS ................................ Bureau of Justice Statistics
BLS ................................ Bureau of Labor Statistics
CDC................................ Centers for Disease Control and Prevention
CFSAN.............................. Center for Food Safety and Applied Nutrition
CPSC ............................... Consumer Product Safety Commission
CSAT............................... Center for Substance Abuse Treatment
DoD................................ Department of Defense
DOJ................................ Department of Justice
DOL................................ Department of Labor
DOT................................ Department of Transportation
EPA................................ Environmental Protection Agency
EPO................................ Epidemiology Program Office
FARS............................... Fatality and Analysis Reporting System
FDA................................ Food and Drug Administration
FEMA ............................... Federal Emergency Management Administration
GAO................................ Government Accounting Office
HCFA............................... Health Care Financing Administration
HRSA ............................... Health Resources and Services Administration
IHS................................. Indian Health Service
IRMO ............................... Information Resources Management Office
NCCAN ............................. National Center for Child Abuse and Neglect
NCCDPHP ........................... National Center for Chronic Disease Prevention and Health Promotion
NCEH............................... National Center for Environmental Health
NCHS............................... National Center for Health Statistics
NCHSR ............................. National Center for Health Services Research
NCHSTP............................. National Center for HIV, STD, and TB Prevention
NCI................................. National Cancer Institute
NCID ............................... National Center for Infectious Disease
NCIPC .............................. National Center for Injury Prevention and Control
NCPS ............................... National Center for Prevention Services
NCVHS.............................. National Committee on Vital and Health Statistics
NHLBI .............................. National Heart, Lung, and Blood Institute
NHTSA.............................. National Highway Traffic Safety Administration
NIAAA .............................. National Institute on Alcoholism and Alcohol Abuse
NICHD .............................. National Institute for Child Health and Human Development
NIDA ............................... National Institute on Drug Abuse
NIDR ............................... National Institute of Dental Research
NIH................................. National Institutes of Health
NIMH ............................... National Institute for Mental Health
NIOSH .............................. National Institute for Occupational Safety and Health
NIP................................. National Immunization Program
NSBA ............................... National School Boards Association
OAR................................ Office of Air and Radiation
OAS................................ Office of the Assistant Secretary
OASD............................... Office of the Assistant Secretary of Defense
OASH............................... Office of the Assistant Secretary for Health
ODPHP ............................. Office of Disease Prevention and Health Promotion
OPA................................ Office of Population Affairs
OPEL ............................... Office of Planning, Evaluation, and Legislation
OPHS............................... Office of Public Health and Science
OPPTS.............................. Office of Pollution, Prevention, and Toxic Substance
ORA................................ Office of Research and Analysis
OSH................................ Office of the Secretary of Health
328 Healthy People 2000 Final Review

Table II. Acronyms used in Healthy People 2000 Final Review—Con.
Acronyms Agency/Organization
OSHA............................... Occupational Safety and Health Administration
OSWER ............................. Office of Solid Waste Enforcement and Remediation
PHF................................ Public Health Foundation
PHS................................ Public Health Service
SAMHSA ............................ Substance Abuse and Mental Health Services Administration
USDA ............................... United States Department of Agriculture
Healthy People 2000 Final Review 329

Table III. Crosswalk between objectives from Healthy People 2000
to Healthy People 2010
Healthy People 2000 Healthy People 2010
Objective
number
Duplicate objective
number(s)
Objective
number
Priority Area 1: Physical Activity and Fitness
1.1 2.1, 3.1, 15.1 12-1*
1.2 2.3, 15.10, 17.12 19-2,* 19-3*
1.3 15.11, 17.13 22-6*
1.3 15.11, 17.13 22-2*
1.4 22-3*
1.4 22-7*
1.5 22-1*
1.6 22-4*
1.6 22-5*
1.7 2.7
1.8 22-9
1.8
1.9 22-10
1.1 22-13*
1.11
1.12 1-3a*
1.12 3-10h*
1.13 17.3
Priority Area 2: Nutrition
2.1 1.1, 3.1, 15.1 12-1*
2.2 16.1 3-1*
2.3 1.2, 15.10, 17.12 19-2*
2.3 1.2, 15.10, 17.12 19-3a-c*
2.4 19-4
2.5 15.9, 16.7 19-8*
2.5 19-9*
2.6 16.8 19-5*
2.6 16.8 19-6*
2.6 16.8 19-7*
2.7 1.7
2.8 19-11*
2.9 19-10*
2.10 19-12a-c*
2.10 19-13*
2.11 14.9 16-19a-b
2.11 14.9 16-19c*
2.12 13.11
2.13
2.14
2.15
2.16
2.17 19-15*
2.18
2.19 7-2h*
2.20 19-16*
2.21 19-17*
2.21 1-3b*
2.22 3.18, 15.2 12-7*
2.23 16.5 3-5*
2.24 17.11 5-2*
2.24 17.11 5-3*
2.25 15.7 12-14*
2.26 15.4 12-10*
2.27 15.6 12-13*
330 Healthy People 2000 Final Review
Table III. Crosswalk between objectives from Healthy People 2000
to Healthy People 2010—Con.
Healthy People 2000 Healthy People 2010
Objective
number
Duplicate objective
number(s)
Objective
number
Priority Area 3: Tobacco
3.1 1.1, 2.1, 15.1 12-1*
3.2 16.2 3-2*
3.3 24-10*
3.4 15.12, 16.6 27-1a*
3.5 27-3*
3.6 27-5*
3.7 27-6*
3.7 27-7
3.8 11.17 27-9
3.9 13.17 27-2a*
3.9 13.17 27-2c*
3.10 27-11
3.11 10.18 27-12*
3.12 10.19 27-13a-e
3.12 10.19 27-13f*
3.13 27-14a-b*
3.14
3.15
3.16 1-3c*
3.16 3-10a-c*
3.17 13.7, 16.17 3-6*
3.18 2.22, 15.2 12-7*
3.19 4.5 26-9a
3.19 4.5 26-9b
3.19 4.5 26-10a,c
3.19 4.5 27-4a
3.19 4.5 27-4b*
3.20 4.6 26-10b
3.20 4.6 27-2a*
3.20 4.6 27-2b*
3.21 4.9 26-16a-f*
3.21 4.9 27-17a-b*
3.21 4.9 27-17c
3.22 4.10 26-17a-c
3.23 27-21a-b*
3.24 27-8a-c*
3.25 10.20 27-19*
3.26
Priority Area 4: Substance Abuse: Alcohol and Other Drugs
4.1 9.23 26-1a
4.2 26-2*
4.3 26-3*
4.4 26-4*
4.5 3.19 26-9a-b
4.5 3.19 26-10a,c
4.5 3.19 27-4a
4.6 3.20 26-10b
4.6 3.20 27-2b
4.7 26-11a-b
4.7 26-11c-d*
4.8 26-12
4.9 3.21 26-16a-f*
4.9 3.21 27-17a-b*
4.9 3.21 27-17c
4.10 3.22 26-17a-c
Healthy People 2000 Final Review 331

Table III. Crosswalk between objectives from Healthy People 2000
to Healthy People 2010—Con.
Healthy People 2000 Healthy People 2010
Objective
number
Duplicate objective
number(s)
Objective
number
4.11 26-14a-c*
4.12
4.13
4.14
4.15 26-24
4.16
4.17
4.18 26-25
4.19 1-3d*
4.20
Priority Area 5: Family Planning
5.1 9-7
5.2 9-1*
5.3 9-12*
5.4 18.3, 19.9 9-8a*
5.4 18.3, 19.9 9-8b*
5.4 18.3, 19.9 9-9a*
5.4 18.3, 19.9 9-9b*
5.5 18.15, 19.16 25-11*
5.6 9-10a-d, g-h
5.6 25-11*
5.7 9-4
5.8 9-11a-c*
5.9
5.10 14.12 1-3f*
5.11 18.13, 19.11
5.12 9-3
Priority Area 6: Mental Health and Mental Disorders
6.1 7.2 18-1*
6.2 7.8 18-2
6.3
6.4
6.5
6.6
6.7 18-9b*
6.8
6.9
6.10 7.18
6.11 20-9
6.12
6.13
6.14
6.15
Priority Area 7: Violent and Abusive Behavior
7.1 15-32*
7.2 6.1 18-1*
7.3 15-3*
7.4 15-33a–b*
7.5 15-34*
7.6 15-37*
7.7 15-35*
7.8 6.2 18-2
7.9 15-38*
7.10 15-39*
7.11 15-4*
332 Healthy People 2000 Final Review

Table III. Crosswalk between objectives from Healthy People 2000
to Healthy People 2010—Con.
Healthy People 2000 Healthy People 2010
Objective
number
Duplicate objective
number(s)
Objective
number
7.12
7.13
7.14
7.15
7.16
7.17
7.18 6.10
7.19 9.25
Priority Area 8: Educational and Community-Based Programs
8.1 17.1, 21.1
8.2 7-1
8.3
8.4 7-2a-j*
8.5 7-3
8.6 7-5a-f*
8.7 7-6*
8.8 7-12
8.9
8.10 7-10*
8.11 7-11a-b, d-f, j-l, p-q, z, bb*
8.11 7-11c, g-i, m-o, r, t-v, y, aa
8.12 7-7*
8.12 7-9*
8.13
8.14
Priority Area 9: Unintentional Injuries
9.1 15-13*
9.2 15-14*
9.3 15-15a*
9.3 15-15b
9.3f 15–16
9.4 15-27*
9.5 15-29*
9.6 15-25*
9.7 15-28a-b*
9.8 15-7*
9.9 15-1*
9.10 15-2*
9.11
9.12 15-19
9.12 15-20
9.13 15-21
9.13 15-23
9.14
9.15
9.16
9.17 15-26a-b*
9.18
9.19 13.16 15-31*
9.20
9.21 1-3e*
9.22
9.23 4.1 26-1a
9.24 15-24
9.25 7.19
9.26 15-22
Healthy People 2000 Final Review 333

Table III. Crosswalk between objectives from Healthy People 2000
to Healthy People 2010—Con.
Healthy People 2000 Healthy People 2010
Objective
number
Duplicate objective
number(s)
Objective
number
Priority Area 10: Occupational Safety and Health
10.1 20-1a
10.1a 20-1b
10.1b 20-1c
10.1c 20-1d
10.1d 20-1e
10.2 20-2a
10.2 20-2g*
10.2a 20-2b
10.2b 20-2c
10.2c 20-2d
10.2d 20-2e
10.2 20-2f
10.2f 20-2h
10.3 20-3
10.4 20-8
10.5 20.3e 14-3g
10.6
10.7 20-11*
10.8 20-7*
10.9 20.11 14-28c*
10.10
10.11
10.12
10.13
10.14
10.15
10.16 20-5
10.17 20-4
10.18 3.11 27-12*
10.19 3.12 27-13b*
10.20 3.25 27-19*
Priority Area 11: Environmental Health
11.10 24-2a-c*
11.1b 1.9a
11.2 17.8
11.3 8-6
11.4 8-11*
11.5 8-1a-g*
11.6 8-18*
11.7 8-4*
11.8 8-15*
11.9 8-5*
11.10 8-8*
11.11 8-22*
11.12 8-19*
11.13
11.14 8-12a*
11.15
11.16 8-27a-j
11.17 3.8 27-9
Priority Area 12: Food and Drug Safety
12.1 10-1a-d
12.2 10-2a*
12.2 10-2b
12.3 10-5*
334 Healthy People 2000 Final Review

Table III. Crosswalk between objectives from Healthy People 2000
to Healthy People 2010—Con.
Healthy People 2000 Healthy People 2010
Objective
number
Duplicate objective
number(s)
Objective
number
12.4
12.5
12.6 17-3*
12.7
12.8 17-4*
12.8 17-5a*
12.8 17-5b*
Priority Area 13: Oral Health
13.1 21-1a*
13.1 21-1b
13.1 21-1c
13.2 21-2a,d*
13.2 21-2b-c
13.3 21-3
13.4 21-4*
13.5 21-5a*
13.6 21-5b*
13.7 3.17, 16.17 3-6*
13.8 21-8a
13.8 21-8b*
13.9 21-9
13.10
13.11 2.12
13.12
13.13
13.14 21-10*
13.15 21-15*
13.16 9.19 15-31
13.17 3.9 27-2c*
Priority Area 14: Maternal and Infant Health
14.1 16-1c
14.1d 16-1d
14.1g 16-1
14.2 16-1a
14.3 16-4
14.4 16-18*
14.5 16-10a
14.5 16-10b
14.6 16-12*
14.7 16-5a*
14.8a 16-9a*
14.8b 16-9b*
14.9 2.11 16-19a-b
14.9 2.11 16-19c*
14.10 16-17a-b, d*
14.10 16-17c
14.11 16-6a
14.12 5.1 1-3f*
14.13
14.14 16-8*
14.15 16-20a-c*
14.16
14.17 16-15*
Healthy People 2000 Final Review 335

Table III. Crosswalk between objectives from Healthy People 2000
to Healthy People 2010—Con.
Healthy People 2000 Healthy People 2010
Objective
number
Duplicate objective
number(s)
Objective
number
Priority Area 15: Heart Disease and Stroke
15.1 1.1, 2.1, 3.1 12-1*
15.2 2.22, 3.18 12-7*
15.3
15.4 2.26 12-10*
15.5 12-11*
15.6 2.27 12-13
15.7 2.25 12-14
15.8
15.9 2.5, 16.7 19.8
15.1 1.2, 2.3, 17.12 19-2, 19-3*
15.11 1.3, 17.13 22-6*
15.12 3.4, 16.6
15.13 12-12*
15.14 12-15*
15.15
15.16 7-5b
15.17
Priority Area 16: Cancer
16.1 2.2 3-1*
16.2 3.2 3-2*
16.3 3-3*
16.4 3-4*
16.5 2.23 3-5*
16.6 3.4, 15.12
16.7 2.5, 15.9 19-8
16.8 2.6 19-5
16.8 2.6 19-6
16.8 2.6 19-7
16.9 3-9b*
16.10 3-10a-h*
16.11 3-13*
16.12 3-11a*
16.12 3-11b*
16.13 3-12a*
16.13 3-12b*
16.14 21-7*
16.15
16.16
16.17 3.17, 13.7 3-6*
Priority Area 17: Diabetes and Chronic Disabling Conditions
17.1 8.1, 21.1
17.2
17.3 1.13
17.4 24-4*
17.5
17.6
17.7 28-4*
17.8 11.2
17.9 5-5*
17.10 4-7*
17.10 5-10*
17.10 28-5
17.11 2.24 5-2*
336 Healthy People 2000 Final Review

Table III. Crosswalk between objectives from Healthy People 2000
to Healthy People 2010—Con.
Healthy People 2000 Healthy People 2010
Objective
number
Duplicate objective
number(s)
Objective
number
17.11 2.24 5-3*
17.12 1.2, 2.3, 15.10 19-2, 19-3*
17.13 1.3, 15.11 22-6
17.14 5-1*
17.14 24-6*
17.15
17.16
17.17
17.18 1-3h*
17.19
17.20 16-23*
17.21 14-17*
17.22 22.4
17.23 5-13*
Priority Area 18: HIV Infection
18.1
18.2 13-1
18.2 13-4*
18.2 13-5*
18.2a 13-2
18.2b 13-3
18.3 5.4, 19.9 9-8a*
18.3 5.4, 19.9 9-8b*
18.3 5.4, 19.9 9-9a*
18.3 5.4, 19.9 9-9b*
18.4 19.10 13-6*
18.4 19.10 25-11*
18.4a 19.10a 9-10e*
18.4b 19.10b 9-10f*
18.5
18.6
18.7
18.8 13-7*
18.9 19.14 1-3g
18.10 19.12 7-2g*
18.11 19.17 7-3*
18.12
18.13 5.11, 19.11 25-18*
18.14
18.15 5.5, 19.16 25-11*
18.16
18.17
Priority Area 19: Sexually Transmitted Diseases
19.1 25-2
19.2 25-1a-c*
19.3 25-3
19.4 25-9
19.5 25-4*
19.5 25-5*
19.6 25-6*
19.7 20.3b–c 14-3e-f
19.8
19.9 5.4, 18.3 9-8a*
19.9 5.4, 18.3 9-8b*
19.9 5.4, 18.3 9-9a*
19.9 5.4, 18.3 9-9b*
Healthy People 2000 Final Review 337
Table III. Crosswalk between objectives from Healthy People 2000
to Healthy People 2010—Con.
Healthy People 2000 Healthy People 2010
Objective
number
Duplicate objective
number(s)
Objective
number
19.10 18.4 13-6
19.10a 18.4a 9-10e*
19.10b 18.4b 9-10f*
19.11 5.11, 18.13
19.12 18.10 7-2g*
19.13 25-18*
19.14 18.9 1-3g*
19.15 25-15*
19.15 25-19*
19.16 5.5, 18.15 25-11*
19.17 18.11 7-3*
Priority Area 20: Immunization and Infectious Diseases
20.1 14-1a, e-f, h-i
20.1 14-1b-d, g, j*
20.2
20.3 10.5 14-3a-c*
20.3 10.5 14-6*
20.3 10.5 14-9
20.3a 14-3d
20.3b 19.7 14-3e
20.3c 19.7 14-3f
20.3 10.5 14-3g
20.3f 14-2
20.4 14-11
20.5 14-20a-e*
20.5 14-21*
20.6 14-15*
20.7 14-4*
20.7 14-7*
20.8
20.9 14-18*
20.10 14-5a-d*
20.11 14-22a-e*
20.11 14-23a–c,f-h
20.11 14-24a*
20.11 14-27a-d*
20.11 14-28b
20.11 10.9 14-28c
20.11 14-29a-f*
20.12
20.13
20.14
20.15
20.16
20.17
20.18 14-13*
20.19
338 Healthy People 2000 Final Review

Table III. Crosswalk between objectives from Healthy People 2000
to Healthy People 2010—Con.
Healthy People 2000 Healthy People 2010
Objective
number
Duplicate objective
number(s)
Objective
number
Priority Area 21: Clinical Preventive Services
21.1 8.1, 17.1
21.2 20.11 †
21.3 1-4a*
21.3 1-4b*
21.3 1-4c
21.4 1-1*
21.4 1-2*
21.5
21.6
21.7
21.8 1-8a-d
21.8 1-8i-t*
21.8a 1-8e-h*
Priority Area 22: Surveillance and Data Systems
22.1
22.2
22.3
22.4 17.22
22.5
22.6
22.7 23-7*
Healthy People 2000 Final Review 339
* Healthy People 2010 objective is modified from the comparable Healthy People 2000 objective;
see Tracking Healthy People 2010, Part B: Operational Definitions for a description of changes.
†This is a compound Healthy People 2000 objective with several measures. In Healthy People
2010, these measures were separated and reassigned to appropriate focus areas. See chapter
21, section ‘‘Transition to Healthy People 2010’’ for more information.

Table IV. Mortality objective cause-of-death categories
Objective
number
Healthy People 2000 Mortality tabulation lists
Cause of death
1
ICD-9 identifying codes Cause of death ICD-9 identifying codes
1.1 Coronary heart disease 402, 410-414, 429.2 Diseases of heart 390-398, 402, 404-429
2.1 See 1.1
2.2 Cancer (all sites) 140-208 Malignant neoplasms, including
neoplasms of lymphatic hematopoietic
tissues
(Same as HP2000)
2.22 Stroke 430-438 Cerebrovascular diseases (Same as HP2000)
2.23 Colorectal cancer 153.0-154.3, 154.8, 159.0 Malignant neoplasms of colon, rectum, 153, 154
rectosigmoid junction, and anus
3.1 See 1.1
3.2 Lung cancer 162.2-162.9 Malignant neoplasms of trachea,
bronchus, and lung
162
3.3 Chronic obstructive pulmonary disease 490-496 Chronic obstructive pulmonary diseases
and allied conditions
(Same as HP2000)
3.17 Cancer of the oral cavity and pharynx 140-149 Malignant neoplasms of lip, oral cavity,
and pharynx
(Same as HP2000)
3.18 See 2.22
4.1 Alcohol-related motor vehicle crashes E810-E819
2
No comparable category . . .
4.2 Cirrhosis 571 Chronic liver disease and cirrhosis (Same as HP2000)
4.3 Drug-related deaths 292, 304, 305.2-305.9, Drug-induced causes (Same as HP2000)
E850-E858, E950.0–
E950.5, E962.0, E980.0–
E980.5
6.1 Suicides E950–E959 (Same as HP2000) (Same as HP2000)
7.1 Homicides E960–E969 Homicide and legal intervention E960-E978
7.2 See 6.1
7.3 Firearm-related deaths E922.0–E922.3, E922.8– Injury by firearms (Same as HP2000)
E922.9, E955.0-E955.4,
E965.0–E965.4, E970,
E985.0–E985.4
9.1 Unintentional injuries E800–E949 Accidents and adverse effects (Same as HP2000)
9.3, 9.3a–c, e–f Motor vehicle crashes E810–E819 Motor vehicle traffic accidents (Same as HP2000)
9.3d,g Motor vehicle crashes E810–E825 Motor vehicle accidents (Same as HP2000)
9.4 Falls and fall-related injuries E880–E888 Accidental falls (Same as HP2000)
9.5 Drowning E830, E832, E910 Accidental drowning and submersion E910
9.6 Residential fires E890–E899 Accidents caused by fire and flames (Same as HP2000)
9.23 See 4.1
10.1 Work-related injuries
3
E800–E999 No comparable category . . .
10.16 Work-related homicides E960–E969 No comparable category . . .
10.17 Occupational lung diseases
3
500–505 No comparable category . . .
13.7 See 3.17
14.3 Maternal mortality 630–676 Complications of pregnancy, childbirth,
and the puerperium or maternal
mortality
(Same as HP2000)
See footnotes and key at end of table.
340 Healthy People 2000 Final Review

Table IV. Mortality objective cause-of-death categories—Con.
Objective
number
Healthy People 2000 Mortality tabulation lists
Cause of death
1
ICD–9 identifying codes Cause of death ICD–9 identifying codes
15.1 See 1.1
15.2 See 2.22
16.1 See 2.2
16.2 See 3.2
16.3 Breast cancer in women 174 Malignant neoplasm of female breast (Same as HP2000)
16.4 Cancer of the uterine cervix 180 Malignant neoplasm of cervix uteri (Same as HP2000)
16.5 See 2.23
16.17 See 3.17
17.9 Diabetes-related deaths
3
250 Diabetes mellitus (Same as HP2000)
20.2 Epidemic-related pneumonia
and influenza deaths for ages 65 years
and over
480-487 No comparable category . . .
. . . Category not applicable.
1
Unless otherwise specified, Healthy People 2000 uses underlying cause-of-death data.
2
Includes only those deaths assigned to E810–E819 that were alcohol related; see Priority Area 4, Substance Abuse: Alcohol and Other Drugs.
3
Healthy People 2000 uses multiple cause-of-death data.
Healthy People 2000 Final Review 341

Table V. Morbidity codes used for objectives tracked by the National Hospital Discharge Survey
1
Objective
number Subject ICD-9-CM diagnosis codes Comment
9.2 All nonfatal injuries 800-959 Because of limited reporting of external causes (E-codes) in hospital
discharge systems, the data include injuries that are unintentional,
intentional, and where the intent is unknown.
9.7 Hip fractures 820
9.9 Nonfatal head injuries 800-801, 803-804, 850-854,
870-873, 925
9.10 Nonfatal spinal cord injuries 806, 952
11.1 Asthma 493
14.7 Severe complications of pregnancy 630-634, 636-677 First listed code cannot equal V27 (which would indicate a delivery)
14.8 Cesarean births — total V27 Procedure code 74, excluding 74.3 and 74.91
14.8a Cesarean births — primary V27 first listed and 654.2 not listed Procedure code 74, excluding 74.3 and 74.91
14.8b Cesarean births — repeat V27 first listed and 654.2 (any listed) Procedure code 74, excluding 74.3 and 74.91
17.10 Lower extremity amputation 250 (any listed) and Procedure codes 84.11–84.12
among people with diabetes 895-897 are not listed
19.16 Pelvic inflammatory disease 614.0-614.5, 614.7-614.9,
615.0, 615.1, 615.9, 98.10
098.16, 098.17, 098.30,
098.36-098.37, 098.39,
098.86 (any listed)
1
First listed diagnoses are used for all objectives, unless otherwise stated. Because of the hierarchical nature of ICD–9–CM coding, three digit code s (such as 493) include all the more detailed
four- and five-digit codes with the same first three digits (for example, 493 includes 493.0-493.9).
342 Healthy People 2000 Final Review

Table VI. Published issues of Healthy People Statistical Notes
Number Title Date of Issue
Healthy People 2000
1 Health Status Indicators for the Year 2000 Fall 1991
2 Infant Mortality Winter 1991
3 Health Status Indicators: Definitions and National Data Spring 1992
4 Issues Related to Monitoring the Year 2000 Objectives Summer 1993
5 Revisions to Healthy People 2000 Baselines July 1993
6 Direct Standardization (Age-Adjusted Death Rates) March 1995
7 Years of Healthy Life April 1995
8 Evaluating Public Health Data Systems: A Practical Approach June 1995
9 Monitoring Air Quality in Healthy People 2000 September 1995
10 Health Status Indicators: Differentials by Race and Hispanic Origin September 1995
11 Operational Definitions for Year 2000 Objectives: Priority Area 20, Immunization and Infectious
Diseases February 1997
12 Operational Definitions for Year 2000 Objectives: Priority Area 13, Oral Health May 1997
13 Healthy People 2000 Midcourse Revisions: A Compendium August 1997
14 Operational Definitions for Year 2000 Objectives: Priority Area 14, Maternal and Infant Health December 1997
15 Priority Data Needs: Sources of National, State, and Local-level Data and Data Collection Systems December 1997
16 Operational Definitions for Year 2000 Objectives: Priority Area 6, Mental Health and Mental Disorders February 1998
17 Operational Definitions for Year 2000 Objectives: Priority Area 21, Clinical Preventive Services December 1998
18 Operational Definitions for Year 2000 Objectives: Priority Area 1, Physical Activity and Fitness December 1998
19 Healthy People 2000: An Assessment Based on the Health Status Indicators for the United States
and Each State November 2000
Healthy People 2010
20 Age Adjustment Using the 2000 Projected U.S. Population January 2001
Healthy People 2000 Final Review 343

Table VII. Special population objectives included in Healthy People 2000
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
American Indian/Alaska Native
1.2d 2.3d, 15.10d, 17.12d Overweight prevalence: 20 years and over Narrowing Toward
1.5f Sedentary lifestyle: 18 years and over Narrowing Toward
2.10d Anemia prevalence: Children 1-5 years Not applicable Away
2.11d 14.9d Breastfeeding: During early postpartum period Narrowing Toward
2.11d 14.9d Breastfeeding: At age 6 months Widening Away
2.12b 13.11b Baby bottle tooth decay Cannot assess Cannot assess
2.24a 17.11a Diabetes prevalence: 15 years and over Narrowing Away
3.4f 15.12f, 16.6f Cigarette smoking prevalence: 18 years and over Narrowing Toward
3.9a 13.17a Smokeless tobacco use: 18-24 years Cannot assess Cannot assess
4.1a 9.23a Alcohol-related motor vehicle deaths: Male Widening Met
4.2b Cirrhosis deaths Widening Away
6.1d 7.2d Suicide: Male Widening Away
7.1f Homicide Widening Toward
8.11 Counties with programs for racial/ethnic minority
groups Not applicable Cannot assess
9.1a Unintentional injury deaths Narrowing Toward
9.3d Motor vehicle crash deaths Widening Met
9.4d Fall-related deaths Widening Away
9.5d Drowning deaths Narrowing Toward
9.6f Residential fire deaths Widening Toward
13.1b Dental caries: Children 6-8 years Widening Away
13.1d Dental caries: Adolescents 15 years Widening Toward
13.2b Untreated dental caries: Children 6-8 years Widening Away
13.2f Untreated dental caries: Adolescents 15 years Narrowing Toward
13.4b Complete tooth loss: 65 years and over Eliminate Toward
13.5b Gingivitis: 35-44 years Narrowing Away
14.1b Infant mortality Narrowing Toward
14.1i Postneonatal mortality Narrowing Toward
14.4a Fetal alcohol syndrome Narrowing Away
14.11b Prenatal care in the first trimester Narrowing Toward
15.14c Blood cholesterol checked: Ever, 18 years and over Widening Toward
15.14e Blood cholesterol checked: Past 2 years, 18 years
and over Narrowing Toward
17.2b Limitation in major activity due to chronic conditions Widening Away
17.9b Diabetes-related deaths Widening Away
17.10b End-stage renal disease due to diabetes Widening Away
17.22 22.4 Identify gaps in health data Not applicable Toward
17.22 22.4 Establish mechanisms to meet data needs Not applicable Toward
20.3g Hepatitis B cases Cannot assess Met
20.3j Hepatitis A cases Narrowing Met
20.4d Tuberculosis new cases Narrowing Toward
20.7a Bacterial meningitis cases Narrowing Met
21.2 Receipt of recommended services: Cholesterol ever
checked, adults 18 years and over Narrowing Toward
21.2 Receipt of recommended services: Cholesterol
checked in last 2 years, adults 18 years and over Widening Toward
21.2 Receipt of recommended services: Pap test in last
3 years, women 18 years and over Narrowing Toward
21.2 Receipt of recommended services: Breast exam and
mammogram in past 2 years, women 50 years and
over Narrowing Toward
21.3d Usual source of care: 18 years and over Narrowing Toward
21.4a People without health care coverage: Under 65 years Narrowing Away
344 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
American Indian/Alaska Native—Con.
21.8 Racial/ethnic minority representation in the health
professions: Degrees awarded Not applicable Met
21.8a Racial/ethnic minority representation in the health
professions: Enrolled in schools of nursing No change No change
22.5a Periodic analysis and publication of data for racial
and ethnic group Narrowing
Met
* For method of calculation, see Appendix Technical Notes.
Special population objectives included in Healthy People 2000: American Indian/Alaska Native
Objective status
Disparity Measure
Eliminated
Narrowing
gap
No
change
Widening
gap
Cannot
assess
Disparity
total
Not a disparity
measure
Overall
total
Met..................... 0 3 0 2 1 6 1 7
Toward .................. 1 16 0 5 0 22 2 24
Mixed/No change .......... 0 0 1 0 0 1 0 1
Away ................... 0 4 0 9 0 13 1 14
Cannot assess ............ 0 0 0 0 2 2 1 3
TOTAL.................. 1 23 1 16 3 44 5 49
Healthy People 2000 Final Review 345

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Asian/Pacific Islander
2.4d Growth retardation: Low-income children 1 year Cannot assess Met
2.4e Growth retardation: Low-income children 2-4 years Cannot assess Met
3.4g 15.12g, 16.6g Cigarette smoking prevalence: Male 18 years and
over Narrowing Toward
8.11 Counties with programs for racial/ethnic minority
groups Not applicable Cannot assess
15.14f Blood cholesterol checked: Past 2 years, 18 years
and over Narrowing Toward
17.22 22.4 Identify gaps in health data Not applicable Toward
17.22 22.4 Establish mechanisms to meet data needs Not applicable Toward
20.3d Hepatitis B cases: Children Cannot assess Toward
20.4a Tuberculosis new cases Widening Toward
21.2 Receipt of recommended services, adults 18 years
and over: Cholesterol checked in last 2 years Narrowing Toward
21.2 Receipt of recommended services, adults 18 years
and over: Tetanus booster in last 2 years Narrowing Toward
21.2 Receipt of recommended services, adults 65 years
and over: Pneumococcal vaccine in lifetime Narrowing Toward
21.2 Receipt of recommended services, adults 65 years
and over: Influenza vaccine in last 12 months Eliminate Met
21.2 Receipt of recommended services, women 18 years
and over: Pap test in last 3 years Narrowing Toward
21.2 Receipt of recommended services, women 50 years
and over: Breast exam and mammogram in past
2 years Narrowing Toward
21.2 Receipt of recommended services, adults 18 years
and over: Asked at least one screening question at
routine checkup Narrowing Toward
21.3e Usual source of care: 18 years and over Narrowing Toward
21.8a Racial/ethnic minority representation in the health
professions: Enrolled in schools of nursing Narrowing Toward
22.5a Periodic analysis and publication of data for racial
and ethnic groups Narrowing
Met
* For method of calculation, see Appendix Technical Notes.
Special population objectives included in Healthy People 2000: Asian/Pacific Islander
Objective status
Disparity Measure
Eliminated
Narrowing
gap
No
change
Widening
gap
Cannot
assess
Disparity
total
Not a disparity
measure
Overall
total
Met..................... 1 1 0 0 2 4 0 4
Toward .................. 0 10 0 1 1 12 2 14
Mixed/No change .......... 0 0 0 0 0 0 0 0
Away ................... 0 0 0 0 0 0 0 0
Cannot assess ............ 0 0 0 0 0 0 1 1
TOTAL.................. 1 11 0 1 3 16 3 19
346 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Black or African American
1.1a 2.1a, 3.1a, 15.1a Coronary heart disease deaths Widening Toward
1.2b 2.3b, 15.10b, 17.12b Overweight prevalence: Female Narrowing Away
1.4b Vigorous physical activity: 18 years and over Not applicable Away
1.5d Sedentary lifestyle: 18 years and over Widening Away
1.13b 17.3b Difficulty performing self-care activities: 70 years and
over Widening Away
2.2a 16.1a Cancer deaths Narrowing Met
2.4a Growth retardation: Low-income children under
1 year Cannot assess No change
2.10e Anemia prevalence: Low-income pregnant female Not applicable Away
2.11b 14.9b Breastfeeding: During early postpartum period Narrowing Toward
2.11b 14.9b Breastfeeding: At age 6 months Narrowing Toward
2.12c 13.11c Baby bottle tooth decay Narrowing Toward
2.22a 3.18a, 15.2a Stroke deaths Narrowing Toward
2.23a 16.5a Colorectal cancer deaths Widening Toward
2.24e 17.11 Diabetes prevalence Widening Away
3.2b 16.2b Slow the rise in lung cancer deaths: Male Narrowing Met
3.4d 15.12d, 16.6d Cigarette smoking prevalence: 18 years and over Narrowing Toward
3.17a 13.7a, 16.17a Oral cancer deaths: Male 45-74 years Narrowing Met
3.17b 13.7b, 16.17b Oral cancer deaths: Female 45-74 years Narrowing Met
4.2a Cirrhosis deaths: Male Narrowing Toward
4.3a Drug-related deaths Narrowing Away
5.1a Adolescent pregnancy: 15-19 years Narrowing Toward
5.2a Unintended pregnancy: 15-44 years Widening Toward
5.3a Infertility: Married couples with wives 15-44 years Narrowing Toward
5.4a 18.3a,19.9a Adolescents who ever engaged in sexual
intercourse: Male 15 years Widening Toward
5.4b 18.3b,19.9b Adolescents who ever engaged in sexual
intercourse: Male 17 years Widening Toward
5.4c 18.3c, 19.9c Adolescents who ever engaged in sexual
intercourse: Female 15-17 years Eliminate Toward
5.7a Failure of contraceptive method: Female 15-44 years Widening Away
5.12a Contraception use: Female 15-44 years Narrowing Toward
7.1c Homicide: Male 15-34 years Widening Toward
7.1e Homicide: Female 15-34 years Narrowing Met
7.3a Firearm related deaths Narrowing Met
7.9a Physical fighting: Adolescent male 14-17 years Narrowing Met
7.10a Weapon-carrying: Adolescent 14-17 years Narrowing Met
8.1a 17.1a, 21.1a Years of healthy life Narrowing Toward
8.2b Completion of high school: 18-24 years Widening Away
8.11 Counties with programs for racial/ethnic minority
groups Not applicable Cannot assess
9.1b Unintentional injury deaths: Male Narrowing Toward
9.2a Unintentional injury hospitalizations: Male Narrowing Met
9.4c Fall-related deaths: Male 30–69 years Narrowing Met
9.5c Drowning deaths: Male Narrowing Toward
9.6c Residential fire deaths: Male Narrowing Met
9.6d Residential fire deaths: Female Narrowing Met
11.1a Asthma hospitalizations Widening Toward
11.4a Blood lead levels: Inner-city low-income children
6 months to 5 years, Levels exceeding 15 ug/dL Widening Toward
11.4a Blood lead levels: Inner-city low-income children
6 months to 5 years, Levels exceeding 25 ug/dL Widening Toward
13.1c Dental caries: Children 6-8 years Eliminate Toward
13.2c Untreated dental caries: Children 6-8 years Narrowing Toward
Healthy People 2000 Final Review 347

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Black or African American—Con.
13.2g Untreated dental caries: Adolescents 15 years Narrowing Toward
13.8a Protective sealants: Children 8 years Narrowing Toward
13.8b Protective sealants: Adolescents 14 years Widening Toward
13.12a Visited the dentist in the past year: Children 5 years Narrowing Toward
13.14c Regular dental visits: 35 years and over Narrowing Toward
14.1a Infant mortality Widening Toward
14.1e Neonatal mortality Widening Toward
14.1h Postneonatal mortality Widening Toward
14.2a Fetal deaths Widening Toward
14.3a Maternal mortality Widening Away
14.4b Fetal alcohol syndrome Widening Away
14.5a Lowbirth weight Narrowing No change
14.5b Very low birthweight Narrowing Away
14.7a Severe complications of pregnancy Widening Toward
14.11a Prenatal care in the first trimester Narrowing Toward
14.15 Newborn screening: Sickle cell anemia Not applicable Toward
15.3a End-stage renal disease Narrowing Away
15.5b Taking action to control blood pressure: Hypertensive
male 18-34 years Widening Toward
15.14a Blood cholesterol checked: ever, 18 years and over Widening Toward
16.3a Female breast cancer deaths Widening Toward
16.4a Cervical cancer deaths Narrowing Toward
16.11e Breast exam and mammogram within past 2 years:
50 years and over Narrowing Met
17.2c Limitation in major activity due to chronic conditions Widening Away
17.4a People with asthma with activity limitation due to
asthma Narrowing Toward
17.9a Diabetes-related deaths Widening Away
17.10a End-stage renal disease due to diabetes Widening Away
17.10c People with diabetes: Lower extremity amputations Widening Away
17.14c Patient education for people 18 years and over with
diabetes Not applicable Toward
17.16a Early detection of significant hearing impairment in
children Cannot assess Cannot assess
17.22 22.4 Identify gaps in health data Not applicable Toward
17.22 22.4 Establish mechanisms to meet data needs Not applicable Toward
18.1b Acquired immunodeficiency syndrome (AIDS)
incidence: 18 years and over Widening Met
18.4d 19.10d Condom use at last sexual intercourse: Female
15-44 years Narrowing Toward
19.1a Gonorrhea Narrowing Toward
19.3a Primary and secondary syphilis Narrowing Met
19.4a Congenital syphilis: Infants under 1 year Narrowing Met
19.6a Pelvic inflammatory disease hospitalizations:
15-44 years Narrowing Toward
19.8a Repeat gonorrhea infection Widening Toward
20.3h Hepatitis B cases Not applicable Met
20.4b Tuberculosis new cases Narrowing Toward
20.11a Pneumococcal immunizations: Noninstitutionalized
people 65 years and over Narrowing Toward
20.11a Influenza immunizations: Noninstitutionalized people
65 years and over Narrowing Toward
21.2 Receipt of recommended services: Cholesterol ever
checked, adults 18 years and over Narrowing Toward
21.2 Receipt of recommended services: Pneumococcal
vaccine in lifetime, adults 65 years and over Widening Toward
348 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Black or African American—Con.
21.2 Receipt of recommended services: Influenza vaccine
in last 12 months, adults 65 years and over Narrowing Toward
21.3b Usual source of care: 18 years and over Narrowing Toward
21.4c People without health care coverage: Under 65 years Narrowing Toward
21.8 Racial/ethnic minority representation in the health
professions: Degrees awarded Narrowing Toward
21.8a Racial/ethnic minority representation in the health
professions: Enrolled in schools of nursing Narrowing Toward
22.5a Periodic analysis and publication of data for racial
and ethnic groups Narrowing
Met
* For method of calculation, see Appendix Technical Notes.
Special population objectives included in Healthy People 2000: Black or African American
Objective status
Disparity Measure
Eliminated
Narrowing
gap
No
change
Widening
gap
Cannot
assess
Disparity
total
Not a disparity
measure
Overall
total
Met..................... 0 16 0 1 0 17 1 18
Toward .................. 2 32 0 20 0 54 4 58
Mixed/No change .......... 0 1 0 0 1 2 0 2
Away ................... 0 4 0 11 0 15 2 17
Cannot assess ............ 0 0 0 0 1 1 1 2
TOTAL.................. 2 53 0 32 2 89 7 97
Healthy People 2000 Final Review 349

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Hispanic or Latino
1.2c 2.3c, 15.10c, 17.12c Overweight prevalence: Mexican American female Narrowing Away
1.2h 2.3h, 15.10h, 17.12h Overweight prevalence: Mexican American male Narrowing Away
1.3a 15.11a, 17.13a Light to moderate physical activity, 5 or more times
per week: 18 years and over Narrowing Met
1.4c Vigorous physical activity: 18 years and over Widening Away
1.5e Sedentary lifestyle: 18 years and over Widening Away
1.7a 2.7a Sound weight loss practices: Overweight male
18 years and over Narrowing Away
1.7b 2.7b Sound weight loss practices: Overweight female
18 years and over Narrowing Away
2.4b Growth retardation: Low-income children under
1 year Cannot assess Met
2.4c Growth retardation: Low-income children 1 year Cannot assess Met
2.11c 14.9c Breastfeeding: During early postpartum period Eliminate Toward
2.11c 14.9c Breastfeeding: At age 6 months Narrowing Toward
2.12d 13.11d Baby bottle tooth decay Widening Toward
2.24b 17.11b Diabetes prevalence: Puerto Rican 20-74 years Cannot assess Cannot assess
2.24c 17.11c Diabetes prevalence: Mexican American 20-74 years Narrowing Away
2.24d 17.11d Diabetes prevalence: Cuban American 20-74 years Cannot assess Cannot assess
2.26b 15.4b Controlled high blood pressure: Mexican American
with high blood pressure 18-74 years
Cannot assess Cannot assess
3.4e 15.12e, 16.6e Cigarette smoking prevalence: 18 years and over Not applicable Toward
3.20 4.6 Use in past month: Alcohol, 12-17 years Not applicable Toward
3.20 4.6 Use in past month: Cocaine, 12-17 years Widening No change
3.20 4.6 Use in past month: Cocaine, 18-25 years Widening Away
4.2c Cirrhosis deaths Widening Toward
4.3b Drug-related deaths Eliminate Away
5.1b Adolescent pregnancy: 15-19 years Widening Away
5.2b Unintended pregnancy: 15-44 years Not applicable Toward
5.3b Infertility: Married couples with wives 15-44 years Eliminate Met
5.7b Failure of contraceptive method: Female 15-44 years Widening Toward
7.1d Homicide: Male 15-34 years Narrowing Toward
8.1b 17.1b, 21.1b Years of healthy life Not applicable Met
8.2a Completion of high school: 18-24 years Narrowing Toward
8.11 Counties with programs for racial/ethnic minority
groups
Not applicable Cannot assess
9.1d Unintentional injury deaths: Mexican American male Narrowing Toward
9.3g Motor vehicle crash deaths: Mexican American Widening Met
9.6g Residential fire deaths: Puerto Rican Eliminate Met
13.2d Untreated dental caries: Children 6-8 years Widening Away
13.2h Untreated dental caries: Adolescents 15 years Narrowing Toward
13.5c Gingivitis: Mexican American 35-44 years Narrowing Toward
13.5c Gingivitis: Cuban 35-44 years Cannot assess Cannot assess
13.5c Gingivitis: Puerto Rican 35-44 years Cannot assess Cannot assess
13.8c Protective sealants: Children 8 years Widening Away
13.8d Protective sealants: Adolescents 14 years Widening Toward
13.12b Visited the dentist in the past year: Children 5 years Narrowing Toward
13.14d Regular dental visits: Mexican American 35 years
and over
Narrowing Toward
13.14e Regular dental visits: Puerto Rican 35 years and
over
Widening Toward
14.1c Infant mortality: Puerto Rican Narrowing Met
14.1f Neonatal mortality: Puerto Rican Narrowing Met
14.1j Postneonatal mortality: Puerto Rican Narrowing Met
14.5c Low birthweight: Puerto Rican Narrowing Away
350 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Hispanic or Latino—Con.
14.5d Very low birthweight: Puerto Rican Widening Away
14.11c Prenatal care in the first trimester Narrowing Toward
15.13a Know blood pressure values: Mexican American
male 18 years and over Widening Away
15.14b Blood cholesterol checked: ever, Mexican American
18 years and over Widening Toward
15.14d Blood cholesterol checked: Past 2 years: Mexican
American 18 years and over Narrowing Toward
16.4b Cervical cancer deaths Narrowing Toward
16.11a Breast exam and mammogram within past 2 years:
50 years and over Narrowing Met
16.12a Pap test ever: 18 years and over Narrowing Toward
16.12a Pap test within past 3 years: 18 years and over Narrowing Toward
17.2d Limitation in major activity due to chronic conditions:
Puerto Rican Widening Away
17.4b People with asthma with activity limitation due to
asthma: Puerto Rican Cannot assess Cannot assess
17.9c Diabetes-related deaths: Mexican American Widening Away
17.9d Diabetes-related deaths: Puerto Rican Widening Away
17.14d Patient education for people 18 years and over with
diabetes Widening Toward
17.22 22.4 Identify gaps in health data Not applicable Toward
17.22 22.4 Establish mechanisms to meet data needs Not applicable Toward
18.1c AIDS incidence: 18 years and over Narrowing Met
19.4b Congenital syphilis Narrowing Met
20.11b Influenza immunizations: Noninstitutionalized people
65 years and over Widening Toward
20.11b Pneumococcal immunizations: Noninstitutionalized
people 65 years and over Widening Toward
20.3i Hepatitis A cases Widening Toward
20.3k Hepatitis C cases Not applicable Met
20.4c Tuberculosis new cases Narrowing Toward
21.2 Receipt of recommended services: Cholesterol ever
checked, adults 18 years and over Widening Toward
21.2 Receipt of recommended services: Cholesterol
checked in last 2 years, adults 18 years Widening Toward
21.2 Receipt of recommended services: Tetanus booster
in last 10 years Widening Toward
21.2 Receipt of recommended services: Pneumococcal
vaccine in lifetime, adults 65 years and over Widening Toward
21.2 Receipt of recommended services: Influenza vaccine
in last 12 months, adults 65 years and over Widening Toward
21.3a Usual source of care: Mexican American 18 years
and over Narrowing Toward
21.3a Usual source of care: 18 years and over Narrowing Toward
21.4b People without health care coverage: Under 65 years Narrowing Away
21.4b People without health care coverage: Mexican
American under 65 years Narrowing Away
21.4b People without health care coverage: Puerto Rican
under 65 years Narrowing Toward
21.4b People without health care coverage: Cuban under
65 years Narrowing Away
21.8 Racial/ethnic minority representation in the health
professions: Degrees awarded Narrowing Toward
21.8a Racial/ethnic minority representation in the health
professions: Enrolled in schools of nursing Narrowing Toward
Healthy People 2000 Final Review 351

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
American Indian/Alaska Native—Con.
21.8 Racial/ethnic minority representation in the health
professions: Degrees awarded Not applicable Met
21.8a Racial/ethnic minority representation in the health
professions: Enrolled in schools of nursing No change No change
22.5a Periodic analysis and publication of data for racial
and ethnic group Narrowing
Met
* For method of calculation, see Appendix Technical Notes.
Special population objectives included in Healthy People 2000: American Indian/Alaska Native
Objective status
Disparity Measure
Eliminated
Narrowing
gap
No
change
Widening
gap
Cannot
assess
Disparity
total
Not a disparity
measure
Overall
total
Met..................... 0 3 0 2 1 6 1 7
Toward .................. 1 16 0 5 0 22 2 24
Mixed/No change .......... 0 0 1 0 0 1 0 1
Away ................... 0 4 0 9 0 13 1 14
Cannot assess ............ 0 0 0 0 2 2 1 3
TOTAL.................. 1 23 1 16 3 44 5 49
Healthy People 2000 Final Review 345

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Women
1.2 2.3, 15.10, 17.12 Overweight prevalence Widening Away
1.2a 2.3a, 15.10a, 17.12a Overweight prevalence: Low-income 20–74 years Narrowing Away
1.2b 2.3b, 15.10b, 17.12b Overweight prevalence: Black Narrowing Away
1.2c 2.3c, 15.10c, 17.12c Overweight prevalence: Mexican American Narrowing Away
1.2f 2.3f, 15.10f, 17.12f Overweight prevalence: Females with high blood
pressure 20–74 years Cannot assess Cannot assess
1.7 2.7 Sound weight loss practices: Overweight, 18 years
and over Not applicable Away
1.7b 2.7b Sound weight loss practices: Overweight, Hispanic
18 years and over Not applicable Away
2.8 Consumption of foods rich in calcium: Pregnant and
lactating Not applicable Away
2.8a Consumption of foods rich in calcium: Meeting
average daily goal, 11–24 years Widening Away
2.10 Iron deficiency prevalence Not applicable Away
2.10 Iron deficiency prevalence: 20–44 years Not applicable Away
2.10c Iron deficiency prevalence: Low-income 20–44 years Not applicable Away
2.10e Anemia prevalence: Black, low-income, pregnant Not applicable Away
2.11 14.9 Breastfeeding: During early postpartum period Not applicable Toward
2.11a 14.9a Breastfeeding: During early postpartum period,
low-income Not applicable Toward
2.11b 14.b Breastfeeding: During early postpartum period, Black Not applicable Toward
2.11c 14.9c Breastfeeding: During early postpartum period,
Hispanic Not applicable Toward
2.11d 14.9d Breastfeeding: During early postpartum period,
American Indian/Alaska Native Not applicable Toward
2.11 14.9 Breastfeeding: At age 6 months Not applicable Toward
2.11a 14.9a Breastfeeding: At age 6 months, low-income Not applicable Toward
2.11b 14.9b Breastfeeding: At age 6 months, Black Not applicable Toward
2.11c 14.9c Breastfeeding: At age 6 months, Hispanic Not applicable Toward
2.11d 14.9d Breastfeeding: At age 6 months, American
Indian/Alaska Native Not applicable Away
2.25 15.7 High blood cholesterol prevalence: 20-74 years Narrowing Met
2.26c 15.4c Controlled high blood pressure: 70 years and over Cannot assess Cannot assess
2.27 15.6 Mean serum cholesterol level: 20-74 years Narrowing Toward
3.2a 16.2a Slow the rise in lung cancer deaths Widening Met
3.4 15.12, 16.6 Cigarette smoking prevalence: 18 years and over Not applicable Toward
3.4h 15.12h, 16.6h Cigarette smoking prevalence: 18-44 years Not applicable Toward
3.4i 15.12i, 16.6i Cigarette smoking prevalence: Pregnant Not applicable Toward
3.4j 15.12j, 16.6j Cigarette smoking prevalence: Use oral
contraceptives Eliminate Toward
3.7 Smoking cessation during pregnancy Not applicable Away
3.7a Smoking cessation during pregnancy: Less than a
high school education Not applicable Cannot assess
3.17 13.7, 16.17 Oral cancer deaths: 45-74 years Not applicable Met
3.17b 13.7b, 16.17b Oral cancer deaths: Black: 45-74 years Not applicable Met
5.1 Adolescent pregnancy: 15-17 years Not applicable Toward
5.1a Adolescent pregnancy: Black 15-19 years Not applicable Toward
5.1b Adolescent pregnancy: Hispanic 15-19 years Not applicable Away
5.2 Unintended pregnancy: 15-44 years Not applicable Toward
5.2a Unintended pregnancy: Black 15-44 years Not applicable Toward
5.2b Unintended pregnancy: Hispanic 15-44 years Not applicable Toward
5.3 Infertility: Married couples with wives 15-44 years Not applicable Toward
5.3a Infertility: Married couples with wives 15-44 years,
Black Not applicable
Toward
Healthy People 2000 Final Review 353

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Women—Con
5.3b Infertility: Married couples with wives 15–44 years,
Hispanic Not applicable Met
5.4 18.3, 19.9 Adolescents who ever engaged in sexual
intercourse: 15 years Not applicable Toward
5.4 18.3, 19.9 Adolescents who ever engaged in sexual
intercourse: 17 years Not applicable Away
5.4c 18.3c, 19.9c Adolescents who ever engaged in sexual
intercourse: Black 15-17 years Not applicable Toward
5.5 18.15, 19.16 Adolescent abstinence from sexual intercourse for
previous 3 months: Sexually active 15-17 years Not applicable Toward
5.6 Contraception use: First intercourse,15-19 years Not applicable Toward
5.6 Contraception use: Recent intercourse,15-19 years Not applicable Away
5.6 Contraception use: Oral contraceptive and the
condom at most recent intercourse, 15-19 years Not applicable Toward
5.7 Failure of contraceptive method: 15-44 years Not applicable Toward
5.7a Failure of contraceptive method: Black 15-44 years Not applicable Away
5.7b Failure of contraceptive method: Hispanic
15-44 years Not applicable Toward
5.8 Persons 13-18 years who have discussed sexuality
with parents Not applicable Toward
5.9 Family planning counseling Not applicable Cannot assess
5.10 14.12 Age-appropriate preconception counseling by
clinicians: Inquiry about family planning, nurse
practitioners Not applicable Away
5.10 14.12 Age-appropriate preconception counseling by
clinicians: Counseling about family planning, nurse
practitioners Not applicable Away
5.10 14.12 Age-appropriate preconception counseling by
clinicians: Counseling about family planning,
obstetricians/gynecologists Not applicable Met
5.11 18.13, 19.11 Clinic services for human immunodeficiency virus
(HIV) and other sexually transmitted diseases
(STDs): HIV client pretest counseling Not applicable Toward
5.11 18.13, 19.11 Clinic services for HIV and other STDs: HIV client
testing Not applicable Toward
5.12 Contraception use: 15-44 years Not applicable Toward
5.12a Contraception use: Black 15-44 years Not applicable Toward
5.12b Contraception use: 15-44 years, under 100%
poverty Not applicable Toward
5.12c Contraception use: 15-19 years, under 200%
poverty Not applicable Toward
6.2a 7.8a Suicide attempts: 14–17 years Widening Away
6.15a Prevalence of depression: 18–54 years (1-year
prevalence) Not applicable Toward
7.1e Homicide: Black 15–34 years Narrowing Met
7.5 Partner abuse Not applicable Toward
7.7 Rape and attempted rape: 12 years and over Not applicable Toward
7.7a Rape and attempted rape: 12–34 years Not applicable Toward
7.7 Sexual assaults: 12 years and over Not applicable Toward
7.12 Emergency room protocols for victims of violence Not applicable Cannot assess
7.15 Battered women turned away from shelters Not applicable Away
9.6d Residential fire deaths: Black Narrowing Met
9.7a Hip fractures: White 85 years and over Narrowing Toward
11.1c Asthma hospitalizations: 25 years and over Widening Toward
14.1 Infant mortality Not applicable Toward
14.1a Infant mortality: Black Not applicable Toward
14.1b Infant mortality: American Indian/Alaska Native Not applicable Toward
354 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Women—Con.
14.1c Infant mortality: Puerto Rican Not applicable Met
14.1d Neonatal mortality Not applicable Toward
14.1e Neonatal mortality: Black Not applicable Toward
14.1f Neonatal mortality: Puerto Rican Not applicable Met
14.1g Postneonatal mortality Not applicable Met
14.1h Postneonatal mortality: Black Not applicable Toward
14.1i Postneonatal mortality: American Indian/Alaska
Native Not applicable Toward
14.1j Postneonatal mortality: Puerto Rican Not applicable Met
14.2 Fetal deaths Not applicable Toward
14.2a Fetal deaths: Black Not applicable Toward
14.3 Maternal mortality Not applicable Away
14.3a Maternal mortality: Black Not applicable Away
14.4 Fetal alcohol syndrome Not applicable Away
14.4a Fetal alcohol syndrome: American Indian/Alaska
Native Not applicable Away
14.4b Fetal alcohol syndrome: Black Not applicable Away
14.5 Low birthweight Not applicable Away
14.5 Very low birthweight Not applicable Away
14.5a Low birthweight: Black Not applicable No change
14.5b Very low birthweight: Black Not applicable Away
14.5c Low birthweight: Puerto Rican Not applicable Away
14.5d Very low birthweight: Puerto Rican Not applicable Away
14.6 Recommended weight gain during pregnancy Not applicable Toward
14.7 Severe complications of pregnancy Not applicable Met
14.7a Severe complications of pregnancy: Black Not applicable Toward
14.8 Cesarean delivery Not applicable Toward
14.8a Cesarean delivery: Primary Not applicable Toward
14.8b Cesarean delivery: Repeat Not applicable Toward
14.10 Abstinence during pregnancy: Tobacco Not applicable Toward
14.10 Abstinence during pregnancy: Alcohol Not applicable Toward
14.10 Abstinence during pregnancy: Cocaine Not applicable No change
14.10 Abstinence during pregnancy: Marijuana Not applicable Away
14.11 Prenatal care in the first trimester Not applicable Toward
14.11a Prenatal care in the first trimester: Black Not applicable Toward
14.11b Prenatal care in the first trimester: American
Indian/Alaska Native Not applicable Toward
14.11c Prenatal care in the first trimester: Hispanic Not applicable Toward
14.13 Screening for fetal abnormalities Not applicable Toward
14.14 Pregnant women and infants receiving
risk-appropriate care Not applicable Cannot assess
14.15 Newborn screening: Sickle cell anemia Not applicable Toward
14.15 Newborn screening: Sickle cell anemia, black Not applicable Toward
14.15 Newborn screening: Galactosemia Not applicable Met
14.15 Newborns diagnosed positive for sickle cell anemia
receiving treatment Not applicable Met
14.15 Newborns diagnosed positive for galactosemia
receiving treatment Not applicable Met
14.16 Babies receiving primary care Not applicable Cannot assess
14.17 Spina bifida and other neural tube defects Not applicable No change
15.4 18.3, 19.9 Controlled high blood pressure: 18–74 years Not applicable Toward
16.3 Female breast cancer deaths Not applicable Met
16.3a Female breast cancer deaths: Black Not applicable Toward
16.4 Cervical cancer deaths Not applicable Toward
16.4a Cervical cancer deaths: Black Not applicable Toward
Healthy People 2000 Final Review 355

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Women—Con.
16.4b Cervical cancer deaths: Hispanic Not applicable Toward
16.11 Breast exam and mammogram within past 2 years:
50 years and over Not applicable Met
16.11a Breast exam and mammogram within past 2 years:
Hispanic 50 years and over Not applicable Met
16.11b Breast exam and mammogram within past 2 years:
Annual family income less than $10,000, 50 years
and over Not applicable Toward
16.11c Breast exam and mammogram within past 2 years:
Less than high school education, 50 years and over Not applicable Toward
16.11d Breast exam and mammogram within past 2 years:
70 years and over Not applicable Toward
16.11e Breast exam and mammogram within past 2 years:
Black 50 years and over Not applicable Met
16.12 Pap test ever: 18 years and over Not applicable Toward
16.12a Pap test ever: Hispanic 18 years and over Not applicable Toward
16.12b Pap test ever: 70 years and over Not applicable Toward
16.12c Pap test ever: Less than high school education,
18 years and over Not applicable Toward
16.12d Pap test ever: Annual family income less than
$10,000, 18 years and over Not applicable Toward
16.12 Pap test within past 3 years: 18 years and over Not applicable Toward
16.12a Pap test within past 3 years: Hispanic 18 years and
over Not applicable Toward
16.12b Pap test within past 3 years: 70 years and over Not applicable Toward
16.12c Pap test within past 3 years: Less than high school
education, 18 years and over Not applicable Toward
16.12d Pap test within past 3 years: Annual family income
less than $10,000, 18 years and over Not applicable Toward
16.15 Pap test quality: Monitoring cytology laboratory Not applicable Met
16.16 FDA-certified mammography facilities Not applicable Toward
17.18 Counseled about estrogen replacement therapy:
40-60 years Not applicable Toward
17.18 Counseled about estrogen replacement therapy:
40-49 years Not applicable Toward
17.18 Counseled about estrogen replacement therapy:
50-60 years Not applicable Met
18.1d AIDS incidence: 18 years and over Widening Met
18.2c HIV prevalence: females 15-44 years, giving birth to
live-born infants No change No change
18.4 19.10 Condom use at last sexual intercourse: 15-44 years Not applicable Toward
18.4a 19.10a Condom use at last sexual intercourse: 15-19 years Not applicable Toward
18.4d 19.10d Condom use at last sexual intercourse: Black
15-44 years Not applicable Toward
19.1c Gonorrhea: 15-44 years Widening Toward
19.2 Chlamydia prevalence: 15-19 years Not applicable Toward
19.2 Chlamydia prevalence: 20-24 years Not applicable Met
19.6 Pelvic inflammatory disease hospitalizations:
15-44 years Not applicable Toward
19.6a Pelvic inflammatory disease hospitalizations: Black
15-44 years Not applicable Toward
19.6b Pelvic inflammatory disease hospitalizations:
15-19 years Not applicable Toward
19.6 Pelvic inflammatory disease initial visits to physicians Not applicable Met
21.2 Receipt of recommended services: Pap test in last
3 years, 18 years and over Not applicable Toward
356 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Women—Con.
21.2 Receipt of recommended services: Pap test in last
3 years, 65 years and over Not applicable Toward
21.2 Receipt of recommended services: Pap test in last
3 years, Asian/Pacific Islander 18 years and over Not applicable Toward
21.2 Receipt of recommended services: Pap test in last
3 years, American Indian/Alaska Native 18 years
and over Not applicable Toward
21.2 Receipt of recommended services: Pap test in last
3 years, females with disabilities 18 years and over Not applicable Toward
21.2 Receipt of recommended services: Breast exam and
mammogram in past 2 years, 50 years and over Not applicable Met
21.2 Receipt of recommended services: Breast exam and
mammogram in past 2 years, 65 years and over Not applicable Toward
21.2 Receipt of recommended services: Breast exam and
mammogram in past 2 years, females with low
income 50 years and over Not applicable Toward
21.2 Receipt of recommended services: Breast exam and
mammogram in past 2 years, Asian/Pacific
Islander 50 years and over Not applicable Toward
21.2 Receipt of recommended services: Breast exam and
mammogram in past 2 years, American
Indian/Alaska Native 50 years and over Not applicable Toward
21.2 Receipt of recommended services: Breast exam and
mammogram in past 2 years, females with
disabilities 50 years and over Not applicable Toward
21.6 Provision of recommended services: Breast exam,
nurse practitioners Not applicable Away
21.6 Provision of recommended services: Breast exam,
obstetricians/gynecologists Not applicable Cannot assess
21.6 Provision of recommended services: Breast exam,
internists Not applicable Cannot assess
21.6 Provision of recommended services: Breast exam,
family physicians Not applicable Cannot assess
21.6 Provision of recommended services: Pap smear,
nurse practitioners Not applicable Away
21.6 Provision of recommended services: Pap smear,
obstetricians/gynecologists Not applicable Cannot assess
21.6 Provision of recommended services: Pap smear,
internists Not applicable Cannot assess
21.6 Provision of recommended services: Pap smear,
family physicians Not applicable Cannot assess
21.6 Provision of recommended services: Mammogram,
nurse practitioners Not applicable Cannot assess
21.6 Provision of recommended services: Mammogram,
nurse practitioners, patients 40–49 years Not applicable Cannot assess
21.6 Provision of recommended services: Mammogram,
nurse practitioners, patients 50 years and over Not applicable Cannot assess
21.6 Provision of recommended services: Mammogram,
obstetricians/gynecologists Not applicable Cannot assess
21.6 Provision of recommended services: Mammogram,
internists Not applicable Cannot assess
21.6 Provision of recommended services: Mammogram,
family physicians Not applicable
Cannot assess
* For method of calculation, see Appendix Technical Notes.
Healthy People 2000 Final Review 357

Special population objectives included in Healthy People 2000: Women
Objective status
Disparity Measure
Eliminated
Narrowing
gap
No
change
Widening
gap
Cannot
assess
Disparity
total
Not a disparity
measure
Overall
total
Met..................... 0 3 0 2 0 5 21 26
Toward .................. 1 2 0 2 0 5 98 103
Mixed/No change .......... 0 0 1 0 0 1 3 4
Away ................... 0 3 0 3 0 6 29 35
Cannot assess ............ 0 0 0 0 2 2 17 19
TOTAL.................. 1 8 1 7 2 19 168 187
358 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
People with low socioeconomic status
1.2a, 2.3a, 15.10a Overweight prevalence: Female 20–74 years Narrowing Away
1.4a Vigorous physical activity: 18 years and over Narrowing Toward
1.5c Sedentary lifestyle: 18 years and over Narrowing Toward
2.4 Growth retardation: Children 5 years and under Not applicable Met
2.4a Growth retardation: Black children under 1 year Not applicable No change
2.4b Growth retardation: Hispanic children under 1 year Not applicable Met
2.4c Growth retardation: Hispanic children 1 year Not applicable Met
2.4d Growth retardation: Asian/Pacific Islander children
1 year Not applicable Met
2.4e Growth retardation: Asian/Pacific Islander children
2-4 years Not applicable Met
2.10a Iron deficiency prevalence: Children 1-2 years Cannot assess Toward
2.10b Iron deficiency prevalence: Children 3-4 years Cannot assess Toward
2.10c Iron deficiency prevalence: Female 20-44 years Narrowing Away
2.10e Anemia prevalence: Black pregnant female Not applicable Away
2.11a 14.9a Breastfeeding: During early postpartum period Narrowing Toward
2.11a 14.9a Breastfeeding: At age 6 months Narrowing Toward
2.12a 13.11a Baby bottle tooth decay: Less than high school
education Widening Toward
3.4a 15.12a Cigarette smoking prevalence: People with high
school education or less, 18 years and over Widening Toward
3.4b 15.12b Cigarette smoking prevalence: Blue-collar workers
18 years and over Widening Toward
3.5a Smoking initiation: Ages 20-24 years with high
school education or less Cannot assess Away
3.7a Smoking cessation during pregnancy: Less than high
school education Cannot assess Cannot assess
4.12 States with access to treatment programs,
underserved Not applicable Met
5.12b Contraception use: Female 15-44 years under 100%
of poverty Narrowing Toward
5.12c Contraception use: Female 15-19 years under 200%
of poverty Narrowing Toward
8.2 Completion of high school: 18-24 years Not applicable Away
8.2a Completion of high school: Hispanic 18-24 years Not applicable Toward
8.2b Completion of high school: Black 18-24 years Not applicable Away
8.3 Eligible children 4 years afforded opportunity to enroll
in Head Start Not applicable Toward
8.3 Low-income children receiving 1 year of Head Start
services prior to entering kindergarten or first grade Not applicable Away
8.7 Hourly workers participating in health promotion
activities: 18 years and over Not applicable Met
11.4a Blood lead levels: Inner-city low-income black
children 6 months-5 years, levels exceeding
15ug/dL Widening Toward
11.4a Blood lead levels: Inner-city low-income black
children 6 months-5 years, levels exceeding
25ug/dL Widening Toward
13.1a Dental caries: Parents who have less than a high
school education, children 6-8 years Narrowing Toward
13.2a Untreated dental caries: Parents who have less than
a high school education, children 6-8 years Narrowing Away
13.2e Untreated dental caries: Parents who have less than
high school education, adolescents 15 years Narrowing Toward
13.4a Complete tooth loss: Annual family income less than
$15,000, 65 years and over Widening Toward
Healthy People 2000 Final Review 359

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
People with low socioeconomic status—Con.
13.5a Gingivitis: Annual family income less than $12,500,
35-44 years Widening Away
16.11b Breast exam and mammogram within past 2 years:
Annual family income less than $10,000, 50 years
and over Narrowing Toward
16.11c Breast exam and mammogram within past 2 years:
Less than high school education, 50 years and over Narrowing Toward
16.12c Pap test ever: Less than high school education,
18 years and over Narrowing Toward
16.12d Pap test ever: Annual family income less than
$10,000, 18 years and over Narrowing Toward
16.12c Pap test within past 3 years: Less than high school
education,18 years and over Narrowing Toward
16.12d Pap test within past 3 years: Annual family income
less than $10,000, 18 years and over Narrowing Toward
17.2a Limitation in major activity due to chronic conditions:
Annual family income less than $10,000, 18 years
and over Widening Away
17.2a Limitation in major activity due to chronic conditions:
Below poverty level Widening Away
17.22 22.4 Identify gaps in health data Not applicable Toward
17.22 22.4 Establish mechanisms to meet data needs Not applicable Toward
20.15 Employment-based conventional insurance plans for
immunizations Not applicable Toward
20.15 Employment-based preferred provider organization
plans for immunizations Not applicable Toward
20.15 Employment-based health maintenance organization
plans for immunizations Not applicable Away
21.2 Receipt of recommended services, adults 65 years
and over: Pneumococcal vaccine in lifetime Widening Toward
21.2 Receipt of recommended services, adults 18 years
and over: Cholesterol ever checked Narrowing Toward
21.2 Receipt of recommended services, women 50 years
and over: Breast exam and mammogram in past
2 years Narrowing Toward
21.2 Receipt of recommended services, adults 65 years
and over: Influenza vaccine in 12 months No change Toward
21.2 Receipt of recommended services, adults 18 years
and over: Cholesterol checked in last 2 years Narrowing Toward
21.3c Usual source of care, 18 years and over: Family
income below poverty line Narrowing Toward
21.4 People without health care coverage: Under 65 years Not applicable Away
21.4a People without health care coverage: American
Indian/Alaska Native, under 65 years Not applicable Away
21.4b People without health care coverage: Hispanic, under
65 years Not applicable Away
21.4b People without health care coverage: Mexican
American, under 65 years Not applicable Away
21.4b People without health care coverage: Puerto Rican,
under 65 years Not applicable Toward
21.4b People without health care coverage: Cuban, under
65 years Not applicable Away
21.4c People without health care coverage: Black, under
65 years Not applicable Toward
21.5 Clinical preventive services from Federal programs:
Immunizations Not applicable
Cannot assess
360 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
People with low socioeconomic status—Con.
21.5 Clinical preventive services from Federal programs:
Counseling Not applicable Cannot assess
21.5 Clinical preventive services from Federal programs:
Screening Not applicable
Cannot assess
* For method of calculation, see Appendix Technical Notes.
Special population objectives included in Healthy People 2000: People with low socioeconomic
status
Objective status
Disparity Measure
Eliminated
Narrowing
gap
No
change
Widening
gap
Cannot
assess
Disparity
total
Not a disparity
measure
Overall
total
Met..................... 0 0 0 0 0 0 7 7
Toward .................. 0 18 1 7 2 28 8 36
Mixed/No change .......... 0 0 0 0 0 0 1 1
Away ................... 0 3 0 3 1 7 10 17
Cannot assess ............ 0 0 0 0 1 1 3 4
TOTAL.................. 0 21 1 10 4 36 29 65
Healthy People 2000 Final Review 361

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
People with disabilities
1.2e 2.3e, 15.10e, 17.12e Overweight prevalence: 20 years and over Narrowing Away
1.5b Sedentary lifestyle: 18 years and over Narrowing Toward
1.13 17.3 Difficulty performing self-care activities: 70 years and
over Not applicable Away
1.13a 17.3a Difficulty performing self-care activities: 85 years and
over Not applicable Away
1.13b 17.3b Difficulty performing self-care activities: Black 70
years and over Not applicable Away
6.5a Adverse health effects from stress: 18 years and
over Widening Toward
6.6 Use of community support among people 18 years
and over with severe mental disorders Not applicable Met
6.7 Treatment for depression: 18–54 years Not applicable Away
6.8a People 18 and over seeking help with
emotional/personal problems Not applicable Toward
6.15 Prevalence of depression: 18–54 years Not applicable Away
6.15a Prevalence of depression: Female 18–54 years Not applicable Toward
8.1 17.1, 21.1 Years of healthy life Not applicable Met
8.1a 17.1a, 21.1a Years of healthy life: Black Not applicable Toward
8.1b 17.1b, 21.1b Years of healthy life: Hispanic Not applicable Met
8.1c 17.1c, 21.1c Years of healthy life: 65 years and over Not applicable Toward
8.3 Preschool child development programs: Children 3–5
years enrolled in preschool Not applicable Toward
9.9 Nonfatal head injuries Not applicable Met
9.10 Nonfatal spinal cord injuries Not applicable Met
9.10a Nonfatal spinal cord injuries: Male Not applicable Toward
9.11 Incidence of secondary conditions associated with
traumatic spinal cord injuries Not applicable Cannot assess
9.22 States with linked emergency medical services and
trauma systems Not applicable Met
11.2 17.8 Serious mental retardation: Children 10 years with IQ
less than 50 Not applicable Away
14.4 Fetal alcohol syndrome Not applicable Away
14.4a Fetal alcohol syndrome: American Indian/Alaska
Native Not applicable Away
14.4b Fetal alcohol syndrome: Black Not applicable Away
14.15 Newborn screening: Sickle cell anemia Not applicable Toward
14.15 Newborn screening: Sickle cell anemia, black Not applicable Toward
14.15 Newborn screening: Galactosemia Not applicable Met
14.15 Newborns diagnosed positive for galactosemia
receiving treatment Not applicable Met
14.15 Newborns diagnosed positive for sickle cell anemia
receiving treatment Not applicable Met
14.17 Spina bifida and other neural tube defects Not applicable No change
17.2 Limitation in major activity due to chronic conditions Not applicable Away
17.2a Limitation in major activity due to chronic conditions:
Annual family income less than $10,000 Not applicable Away
17.2a Limitation in major activity due to chronic conditions:
Below poverty level Not applicable Away
17.2b Limitation in major activity due to chronic conditions:
American Indian/Alaska Native Not applicable Away
17.2c Limitation in major activity due to chronic conditions:
Black Not applicable Away
17.2d Limitation in major activity due to chronic conditions:
Puerto Rican Not applicable
Away
362 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
People with disabilities—Con.
17.4 People with asthma with activity limitation due to
asthma Not applicable Away
17.4a People with asthma with activity limitation due to
asthma: Black Not applicable Toward
17.4b People with asthma with activity limitation due to
asthma: Puerto Ricans Not applicable Cannot assess
17.5 Activity limitation due to chronic back conditions Not applicable Away
17.6 Significant hearing impairment Not applicable Toward
17.6a Significant hearing impairment: People 45 years and
over Not applicable Toward
17.7 Significant visual impairment Not applicable Toward
17.7a Significant visual impairment: People 65 years and
over Not applicable Toward
17.10 Diabetes-related complications: End-stage renal
disease Not applicable Away
17.10 People with diabetes: Blindness Not applicable No change
17.10 People with diabetes: Lower extremity amputation Not applicable Toward
17.10a End-stage renal disease due to diabetes: Black with
diabetes Not applicable Away
17.10b End-stage renal disease due to diabetes: American
Indian/Alaska Native with diabetes Not applicable Away
17.10c Lower extremity amputations due to diabetes: Black Not applicable Away
17.14a Patient education for people 18 years and over with
diabetes Not applicable Toward
17.14b Patient education for people 18 years and over with
asthma Not applicable Away
17.14c Patient education for people 18 years and over with
diabetes: Black Not applicable Toward
17.14d Patient education for people 18 years and over with
diabetes: Hispanic Not applicable Toward
17.15 Clinician assessment of childhood development,
nurse practitioners Not applicable Away
17.16 Early detection of significant hearing impairment in
children Not applicable Away
17.16a Early detection of significant hearing impairment in
children: Black Not applicable Cannot assess
17.17 Clinician assessment in adults 65 years and over:
Visual acuity testing, nurse practitioners Not applicable Away
17.17 Clinician assessment in adults 65 years and over:
Prescription of mobility aids/modification, nurse
practitioners Not applicable Toward
17.19 Worksites with policies for hiring people with
disabilities Not applicable Met
17.20 Service systems for children with or at risk of chronic
and disabling conditions Not applicable Cannot assess
17.22 22.4 Identify gaps in health data Not applicable Toward
17.22 22.4 Establish mechanisms to meet data needs Not applicable Toward
17.23 People with diabetes who had a dilated eye exam in
the past year Not applicable Toward
20.11 Basic immunization series among children: Children
2 years and under Not applicable Cannot assess
20.11 Basic immunization series among children
19–35 months: Diphtheria-tetanus-pertussis Not applicable Met
20.11 Basic immunization series among children
19–35 months: Polio Not applicable Met
20.11 Basic immunization series among children
19–35 months: Measles-containing Not applicable Met
Healthy People 2000 Final Review 363

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
People with disabilities—Con.
20.11 Basic immunization series among children
19–35 months: Haemophilus influenza type b Not applicable Met
20.11 Basic immunization series among children
19–35 months: Hepatitis B Not applicable Toward
20.11 Basic immunization series among children
19–35 months: 4DTP/3Polio/1MMR Not applicable Toward
20.11 Basic immunization series among children: Children
in licensed child care facilities Not applicable Met
20.11 Basic immunization series among children: Children
in kindergarten through postsecondary education
institutions Not applicable Met
20.11 Hepatitis B immunizations: Infants of antigen-positive
mothers Not applicable Toward
20.11 Hepatitis B immunizations: Occupationally exposed
workers Not applicable Toward
20.11 Hepatitis B immunizations: Injecting drug users in
drug treatment programs Not applicable Cannot assess
20.11 Hepatitis B immunizations: Men who have sex with
men Not applicable Toward
20.11 Pneumococcal immunizations: Institutionalized
chronically ill people or older people Not applicable Toward
20.11 Pneumococcal immunizations: Noninstitutionalized
people 65 years and over Not applicable Toward
20.11a Pneumococcal immunizations, noninstitutionalized
people 65 years and over: Black Not applicable Toward
20.11b Pneumococcal immunizations, noninstitutionalized
people 65 years and over: Hispanic Not applicable Toward
20.11 Influenza immunizations: Institutionalized chronically
ill people or older people Not applicable Toward
20.11 Influenza immunizations: Noninstitutionalized people
65 years and over Not applicable Met
20.11a Influenza immunizations, noninstitutionalized people
65 years and over: Black Not applicable Toward
20.11b Influenza immunizations, noninstitutionalized people
65 years and over: Hispanic Not applicable Toward
21.2 Receipt of recommended services: Tetanus booster
in last 10 years, 18 years and over Narrowing Toward
21.2 Receipt of recommended services: Pap test in last
3 years, women 18 years and over Widening Toward
21.2 Receipt of recommended services: Breast exam and
mammogram in past 2 years, women 50 years and
over Widening
Toward
* For method of calculation, see Appendix Technical Notes.
Special population objectives included in Healthy People 2000: People with disabilities
Objective status
Disparity Measure
Eliminated
Narrowing
gap
No
change
Widening
gap
Cannot
assess
Disparity
total
Not a disparity
measure
Overall
total
Met..................... 0 0 0 0 0 0 17 17
Toward .................. 0 2 0 3 0 5 33 38
Mixed/No change .......... 0 0 0 0 0 0 2 2
Away ................... 0 1 0 0 0 1 25 26
Cannot assess ............ 0 0 0 0 0 0 6 6
TOTAL.................. 0 3 0 3 0 6 83 89
364 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Older adults
1.1 2.1, 3.1, 15.1 Coronary heart disease deaths Not applicable Met
1.1a 2.1a, 3.1a, 15.1a Coronary heart disease deaths: Black Not applicable Toward
1.2 2.3, 15.10, 17.12 Overweight prevalence Not applicable Away
1.2 2.3, 15.10, 17.12 Overweight prevalence: Male Not applicable Away
1.2 2.3, 15.10, 17.12 Overweight prevalence: Female Not applicable Away
1.2a 2.3a, 15.10a, 17.12a Overweight prevalence: Low-income female
20–74 years Not applicable Away
1.2b 2.3b, 15.10b, 17.12b Overweight prevalence: Black female Not applicable Away
1.2c 2.3c, 15.10c, 17.12c Overweight prevalence: Mexican American female Not applicable Away
1.2d 2.3d, 15.10d, 17.12d Overweight prevalence: American Indian/Alaska
Native 20 years and over Not applicable Toward
1.2e 2.3e, 15.10e, 17.12e Overweight prevalence: People with disabilities
20 years and over Not applicable Away
1.2f 2.3f, 15.10f, 17.12f Overweight prevalence: Females with high blood
pressure 20–74 years Not applicable Cannot assess
1.2g 2.3g, 15.10g, 17.12g Overweight prevalence: Males with high blood
pressure 20–74 years Not applicable Cannot assess
1.2h 2.3h, 15.10h, 17.12h Overweight prevalence: Mexican American male Not applicable Away
1.3 15.11, 17.13 Light to moderate physical activity, 5 or more times
per week: 18 years and over Not applicable Met
1.3 15.11, 17.13 Light to moderate physical activity, 7 or more times
per week: 18 years and over Not applicable Toward
1.3a 15.11a, 17.13a Light to moderate physical activity, 5 or more times
per week: Hispanic 18 years Not applicable Met
1.5a Sedentary lifestyle: 65 years and over Eliminate Met
1.13 17.3 Difficulty performing self-care activities: 70 years and
over Not applicable Away
1.13a 17.3a Difficulty performing self-care activities: People
85 years and over Widening Away
1.13b 17.3b Difficulty performing self-care activities: Black
70 years and over Not applicable Away
2.2 16.1 Cancer deaths Not applicable Met
2.2a 16.1a Cancer deaths: Black Not applicable Met
2.5 15.9, 16.7 Dietary fat intake, National Health and Nutrition
Examination Survey (NHANES): Average percent of
calories from total fat Not applicable Toward
2.5 15.9, 16.7 Dietary fat intake, NHANES: Average percent of
calories from saturated fat Not applicable Toward
2.5 15.9, 16.7 Dietary fat intake, Continuing Survey of Food Intakes
by Individuals (CSFII): Average percent of calories
from total fat Not applicable Toward
2.5 15.9, 16.7 Dietary fat intake, CSFII: Met goal for fat Not applicable Toward
2.5 15.9, 16.7 Dietary fat intake, CSFII: Average percent of calories
from saturated fat Not applicable Toward
2.5 15.9, 16.7 Dietary fat intake, CSFII: Met goal for saturated fat Not applicable Toward
2.6 16.8 Average daily intake of vegetables, fruits, and grain
products Not applicable Toward
2.6 16.8 Average number of daily servings: Vegetables and
fruits Not applicable Toward
2.6 16.8 Average number of daily servings: Grain products Not applicable Met
2.6 16.8 Met Dietary Guidelines goal: Vegetables and fruits Not applicable Toward
2.6 16.8 Met Dietary Guidelines goal: Grain products Not applicable Met
2.8 Consumption of foods rich in calcium: 25 years
and over Not applicable No change
2.18 Receipt of home-delivered meals: People 65 years
and over in need Not applicable
Toward
Healthy People 2000 Final Review 365

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Older adults—Con.
2.22 3.18, 15.2 Stroke deaths Not applicable Toward
2.22a 3.18a, 15.2a Stroke deaths: Black Not applicable Toward
2.23 16.5 Colorectal cancer deaths Not applicable Met
2.23a 16.5a Colorectal cancer deaths: Black Not applicable Toward
2.24 17.11 Diabetes incidence Not applicable Away
2.24 17.11 Diabetes prevalence Not applicable Away
2.24a 17.11a Diabetes prevalence: American Indian/Alaska Native
15 years and over Not applicable Away
2.24b 17.11b Diabetes prevalence: Puerto Rican 20–74 years Not applicable Cannot assess
2.24c 17.11c Diabetes prevalence: Mexican American 20–74 years Not applicable Away
2.24d 17.11d Diabetes prevalence: Cuban American 20–74 years Not applicable Cannot assess
2.24e 17.11e Diabetes prevalence: Black Not applicable Away
2.26c 15.4c Controlled high blood pressure: Females with high
blood pressure 70 years and over Cannot assess Cannot assess
3.2 16.2 Slow the rise in lung cancer deaths Not applicable Met
3.2a 16.2a Slow the rise in lung cancer deaths: Female Not applicable Met
3.2b 16.2b Slow the rise in lung cancer deaths: Black male Not applicable Met
3.17 13.7, 16.17 Oral cancer deaths: Male 45–74 years Not applicable Met
3.17 13.7, 16.17 Oral cancer deaths: Female 45–74 years Not applicable Met
3.17a 13.7a, 16.17a Oral cancer deaths: Black male 45–74 years Not applicable Met
3.17b 13.7b, 16.17b Oral cancer deaths: Black female 45–74 years Not applicable Met
6.1c 7.2c Suicide: White male 65 years and over Narrowing Met
8.1 17.1, 21.1 Years of healthy life Not applicable Met
8.1a 17.1a, 21.1a Years of healthy life: Black Not applicable Toward
8.1b 17.1b, 21.1b Years of healthy life: Hispanic Not applicable Met
8.1c 17.1c, 21.1c Years of healthy life: 65 years and over Narrowing Toward
8.8 Health promotion programs for adults 65 years and
over Not applicable No change
9.3c Motor vehicle crash deaths: 70 years and over Widening Toward
9.4a Fall-related deaths: 65–84 years Widening Away
9.4b Fall-related deaths: 85 years and over Widening Away
9.6b Residential fire deaths: 65 years and over Widening Met
9.7 Hip fractures: 65 years and over Not applicable Away
9.7a Hip fractures: White females 85 years and over Narrowing Toward
12.6 Providers reviewing medication, maintenance of
current medication list: Nurse practitioners Not applicable Toward
12.6 Providers reviewing medication, review of
medications when prescribing: Nurse practitioners Not applicable Toward
13.4 Complete tooth loss: 65 years and over Not applicable Toward
13.4a Complete tooth loss: Annual family income less than
$15,000, 65 years and over Not applicable Toward
13.4b Complete tooth loss: American Indian/Alaska Native
65 years and over Not applicable Toward
13.13 Oral health care at institutional facilities: Nursing
facilities Not applicable Toward
13.14 Regular dental visits: 35 years and over Not applicable Toward
13.14a Regular dental visits: Edentulous people, 35 years
and over Not applicable Toward
13.14b Regular dental visits: 65 years and over Narrowing Toward
13.14c Regular dental visits: Black 35 years and over Not applicable Toward
13.14d Regular dental visits: Mexican American 35 years
and over Not applicable Toward
13.14e Regular dental visits: Puerto Rican 35 years and
over Not applicable Toward
16.3 Female breast cancer deaths Not applicable Met
366 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Older adults—Con.
16.3a Female breast cancer deaths: Black Not applicable Toward
16.4 Cervical cancer deaths Not applicable Toward
16.4a Cervical cancer deaths: Black Not applicable Toward
16.4b Cervical cancer deaths: Hispanic Not applicable Toward
16.11 Breast exam and mammogram within past 2 years:
50 years and over Not applicable Met
16.11a Breast exam and mammogram within past 2 years:
Hispanic 50 years and over Not applicable Met
16.11b Breast exam and mammogram within past 2 years:
Annual family income less than $10,000, 50 years
and over Not applicable Toward
16.11c Breast exam and mammogram within past 2 years:
Less than high school education, 50 years and over Not applicable Toward
16.11d Breast exam and mammogram within past 2 years:
70 years and over Not applicable Toward
16.11e Breast exam and mammogram within past 2 years:
Black 50 years and over Not applicable Met
16.12b Pap test ever: 70 years and over Narrowing Toward
16.12b Pap test within past 3 years: 70 years and over Narrowing Toward
16.13 Colon screen, fecal occult blood test within past
2 years: 50 years and over Not applicable Toward
16.13 Colon screen sigmoidoscopy ever: 50 years and over Not applicable Toward
16.14 Oral exam within past year: 50 years and over Not applicable Toward
16.14 Skin exam within past year: 50 years and over Not applicable Away
16.14 Digital rectal exam within past year: 50 years and
over Not applicable Toward
17.2 Limitation in major activity due to chronic conditions Not applicable Away
17.2a Limitation in major activity due to chronic conditions:
Annual family income less than $10,000 Not applicable Away
17.2a Limitation in major activity due to chronic conditions:
Below poverty level Not applicable Away
17.2b Limitation in major activity due to chronic conditions:
American Indian/Alaska Native Not applicable Away
17.2c Limitation in major activity due to chronic conditions:
Black Not applicable Away
17.2d Limitation in major activity due to chronic conditions:
Puerto Rican Not applicable Away
17.4 People with asthma with activity limitation due to
asthma Not applicable Away
17.4a People with asthma with activity limitation due to
asthma: Black Not applicable Toward
17.4b People with asthma with activity limitation due to
asthma: Puerto Rican Not applicable Cannot assess
17.5 Activity limitation due to chronic back conditions Not applicable Away
17.6 Significant hearing impairment Not applicable Toward
17.6a Significant hearing impairment: People 45 years and
over Widening Toward
17.7 Significant visual impairment Not applicable Toward
17.7a Significant visual impairment: People 65 years and
over Widening Toward
17.9 Diabetes-related deaths Not applicable Away
17.9a Diabetes-related deaths: Black Not applicable Away
17.9b Diabetes-related deaths: American Indian/Alaska
Native Not applicable Away
17.9c Diabetes-related deaths: Mexican American Not applicable Away
17.9d Diabetes-related deaths: Puerto Rican Not applicable Met
Healthy People 2000 Final Review 367

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Older adults—Con.
17.10 People with diabetes: End-stage renal disease Not applicable Away
17.10 People with diabetes: Blindness Not applicable No change
17.10 People with diabetes: Lower extremity amputation Not applicable Toward
17.10a End-stage renal disease due to diabetes: Black Not applicable Away
17.10b End-stage renal disease due to diabetes: American
Indian/Alaska Native Not applicable Away
17.10c Lower extremity amputations due to diabetes: Black Not applicable Away
17.17 Clinician assessment in adults 65 years and over:
Visual acuity testing, nurse practitioners Not applicable Away
17.17 Clinician assessment in adults 65 years and over:
Prescription of mobility aids/modification, nurse
practitioners Not applicable Toward
17.18 Counseled about estrogen replacement therapy:
Female 40-60 years Not applicable Toward
17.18 Counseled about estrogen replacement therapy:
Female 40-49 years Not applicable Toward
17.18 Counseled about estrogen replacement therapy:
Female 50-60 years Not applicable Met
20.2 Epidemic-related pneumonia and influenza deaths:
65 years and over Not applicable Met
20.10 Pneumonia-related restricted activity days: 65 years
and over Not applicable Away
20.11 Pneumococcal immunizations: Institutionalized
chronically ill people or older people Not applicable Toward
20.11 Pneumococcal immunizations: Noninstitutionalized
people 65 years and over Not applicable Toward
20.11 Influenza immunizations: Institutionalized chronically
ill people or older people Not applicable Toward
20.11 Influenza immunizations: Noninstitutionalized people
65 years and over Not applicable Met
20.11a Pneumococcal immunizations: Noninstitutionalized
black people 65 years and over Not applicable Toward
20.11b Pneumococcal immunizations: Noninstitutionalized
Hispanic people 65 years and over Not applicable Toward
20.11a Influenza immunizations: Noninstitutionalized black
people 65 years and over Not applicable Toward
20.11b Influenza immunizations: Noninstitutionalized
Hispanic people 65 years and over Not applicable Toward
20.14 Influenza vaccination: Nurse practitioners Not applicable Toward
20.14 Influenza vaccination: Obstetricians/gynecologists Not applicable Cannot assess
20.14 Influenza vaccination: Internists Not applicable Cannot assess
20.14 Influenza vaccination: Family physicians Not applicable Cannot assess
20.14 Pneumococcal vaccination: Nurse practitioners Not applicable Toward
20.14 Pneumococcal vaccination:
Obstetricians/gynecologists Not applicable Cannot assess
20.14 Pneumococcal vaccination: Internists Not applicable Cannot assess
20.14 Pneumococcal vaccination: Family physicians Not applicable Cannot assess
21.2 Receipt of recommended services: Routine check-up Not applicable Toward
21.2 Receipt of recommended services, adults 65 years
and over: Tetanus booster in last 10 years Narrowing Toward
21.2 Receipt of recommended services, adults 65 years
and over: Pneumococcal vaccine in lifetime Not applicable Toward
21.2 Receipt of recommended services, adults 65 years
and over: Pneumococcal vaccine in lifetime, people
with low income Not applicable Toward
21.2 Receipt of recommended services, pneumococcal
vaccine in lifetime, adults 65 years and over: Black Not applicable Toward
368 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Older adults—Con.
21.2 Receipt of recommended services, pneumococcal
vaccine in lifetime, adults 65 years and over:
Hispanic Not applicable Toward
21.2 Receipt of recommended services, pneumococcal
vaccine in lifetime, adults 65 years and over:
Asian/Pacific Islander Not applicable Toward
21.2 Receipt of recommended services, adults 65 years
and over: Influenza vaccine in last 12 months Not applicable Met
21.2 Receipt of recommended services: Influenza vaccine
in last 12 months, People with low income Not applicable Toward
21.2 Receipt of recommended services, influenza vaccine
in last 12 months, adults 65 years and over: Black Not applicable Toward
21.2 Receipt of recommended services, influenza vaccine
in last 12 months, adults 65 years and over:
Hispanic Not applicable Toward
21.2 Receipt of recommended services, influenza vaccine
in last 12 months, adults 65 years and over:
Asian/Pacific Islander Not applicable Met
21.2 Receipt of recommended services, women 18 years
and over: Pap test in last 3 years Narrowing Toward
21.2 Receipt of recommended services, women 50 years
and over: Breast exam and mammogram in past
2 years Not applicable Met
21.2 Receipt of recommended services, women 65 years
and over: Breast exam and mammogram in past
2 years Narrowing Toward
21.2 Receipt of recommended services: Breast exam and
mammogram in past 2 years, females with low
income 50 years and over Not applicable Toward
21.2 Receipt of recommended services, women 50 years
and over, breast exam and mammogram in past
2 years: Asian/Pacific Islander Not applicable Toward
21.2 Receipt of recommended services, women 50 years
and over, breast exam and mammogram in past 2
years: American Indian/Alaska Native Not applicable Toward
21.2 Receipt of recommended services, women 50 years
and over, breast exam and mammogram in past
2 years: Females with disabilities Not applicable Toward
21.2 Receipt of recommended services: Asked at least
one screening question at routine checkup Eliminate Toward
21.6 Provision of recommended services, adults 65 years
and over: Influenza vaccination nurse practitioners Not applicable Toward
21.6 Provision of recommended services, adults 65 years
and over: Influenza vaccination, obstetricians/
gynecologists Not applicable Cannot assess
21.6 Provision of recommended services, adults 65 years
and over: Influenza vaccination, internists Not applicable Cannot assess
21.6 Provision of recommended services, adults 65 years
and over: Influenza vaccination, family physicians Not applicable Cannot assess
21.6 Provision of recommended services, adults 65 years
and over: Pneumococcal vaccination, nurse
practitioners Not applicable Toward
21.6 Provision of recommended services, adults 65 years
and over: Pneumococcal vaccination,
obstetricians/gynecologists Not applicable Cannot assess
21.6 Provision of recommended services, adults 65 years
and over: Pneumococcal vaccination, internists Not applicable
Cannot assess
Healthy People 2000 Final Review 369

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Older adults—Con.
21.6 Provision of recommended services, adults 65 years
and over: Pneumococcal vaccination, family
physicians Not applicable
Cannot assess
* For method of calculation, see Appendix Technical Notes.
Special population objectives included in Healthy People 2000: Older adults
Objective status
Disparity Measure
Eliminated
Narrowing
gap
No
change
Widening
gap
Cannot
assess
Disparity
total
Not a disparity
measure
Overall
total
Met..................... 1 1 0 1 0 3 28 31
Toward .................. 1 8 0 3 0 12 71 83
Mixed/No change .......... 0 0 0 0 0 0 3 3
Away ................... 0 0 0 3 0 3 35 38
Cannot assess ............ 0 0 0 0 1 1 17 18
TOTAL.................. 2 9 0 7 1 19 154 173
370 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Adolescents and young adults
1.2 2.3, 15.10, 17.12 Overweight prevalence Cannot assess Away
1.3 15.11,17.13 Light to moderate physical activity: 5 or more times
per week Not applicable Met
1.3 15.11,17.13 Light to moderate physical activity: 7 or more times
per week Not applicable Toward
1.4 Vigorous physical activity: 10-17 years Not applicable Cannot assess
1.4 Vigorous physical activity: Grades 9-12 Not applicable Toward
1.6 Strengthening 4 or more times per week: Grades
9–12 Cannot assess Met
1.6 Stretching 4 or more times per week: Grades 9-12 Cannot assess Met
1.8 Daily school physical education: Grades 1-12 Not applicable Cannot assess
1.8 Daily school physical education: Grades 9-12 Not applicable Away
1.9 Active physical education class time: Grades 9-12,
21 or more minutes, 3-5 times per week Not applicable Toward
1.9 Active physical education class time: Grades 9-12,
30 or more minutes, 1 or more times per week Not applicable Toward
1.9 Active physical education class time: All students Not applicable Cannot assess
2.8 Consumption of foods rich in calcium: 11-24 years Not applicable Away
2.8 Consumption of foods rich in calcium: Pregnant and
lactating females Not applicable Away
2.8a Consumption of foods rich in calcium: Meeting
average daily goal, female 11-24 years Not applicable Away
2.13 Use of food labels: 18 years and over Not applicable Toward
3.5 Smoking initiation: Proxy 20-24 years Not applicable Toward
3.5a Smoking initiation: Proxy 20-24 years with a high
school education or less Not applicable Away
3.9 13.17 Smokeless tobacco use: Male 12-17 years Not applicable Met
3.9 13.17 Smokeless tobacco use: Male 18-24 years Not applicable Toward
3.9a 13.17a Smokeless tobacco use: American Indian/Alaska
Native 18-24 years Not applicable Cannot assess
3.15 Tobacco product advertising and promotion to youth Not applicable Cannot assess
3.19 4.5 Average age of first use: Cigarettes Not applicable Toward
3.19 4.5 Average age of first use: Alcohol Not applicable No change
3.19 4.5 Average age of first use: Marijuana Not applicable Toward
3.20 4.6 Use in past month: Alcohol, 12-17 years Not applicable Toward
3.20 4.6 Use in past month: Alcohol, 18-20 years Not applicable Toward
3.20 4.6 Use in past month: Alcohol, Hispanic 12-17 years Not applicable Toward
3.20 4.6 Use in past month: Marijuana, 12-17 years Not applicable Away
3.20 4.6 Use in past month: Marijuana, 18-25 years Not applicable Toward
3.20 4.6 Use in past month: Cocaine, 12-17 years Not applicable Toward
3.20 4.6 Use in past month: Cocaine, 18-25 years Not applicable Met
3.20 4.6 Use in past month: Cocaine, Hispanic 12-17 years Not applicable No change
3.20 4.6 Use in past month: Cocaine, Hispanic 18-25 years Not applicable Away
3.20 4.6 Use in past month: Cigarettes, 12-17 years Not applicable Toward
3.21 4.9 Perception of social disapproval by high school
seniors: Heavy use of alcohol Not applicable Toward
3.21 4.9 Perception of social disapproval by high school
seniors: Occasional use of marijuana Not applicable Away
3.21 4.9 Perception of social disapproval by high school
seniors: Trying cocaine once or twice Not applicable Away
3.21 4.9 Perception of social disapproval by high school
seniors: Smoking one or more packs of cigarettes
per day Not applicable Away
3.22 4.10 Perception of harm by high school seniors: Heavy
use of alcohol Not applicable
Away
Healthy People 2000 Final Review 371

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Adolescents and young adults—Con.
3.22 4.10 Perception of harm by high school seniors: Regular
use of marijuana Not applicable Away
3.22 4.10 Perception of harm by high school seniors: Trying
cocaine once or twice Not applicable Away
3.22 4.10 Perception of harm by high school seniors: Smoking
one or more packs of cigarettes per day Not applicable Toward
3.22 4.10 Perception of harm by high school seniors: Using
smokeless tobacco regularly Not applicable Toward
4.1b 9.23 Alcohol-related motor vehicle deaths: 15-24 years Narrowing Met
4.7 Heavy drinking in past 2 weeks: High school seniors Not applicable Toward
4.7 Heavy drinking in past 2 weeks: College students Not applicable Toward
4.8 Alcohol consumption: 14 years and over Not applicable Toward
4.11 Anabolic steroid use: Male high school seniors Not applicable Met
4.13 Alcohol and drug education in schools Not applicable Cannot assess
4.18 States with zero tolerance blood alcohol levels:
Under 21 years Not applicable Met
5.1 Pregnancy: 15-17 years Not applicable Toward
5.1a Pregnancy: Black 15-19 years Not applicable Toward
5.1b Pregnancy: Hispanic 15-19 years Not applicable Away
5.4 18.3, 19.9 Ever engaged in sexual intercourse: Female
15 years Not applicable Toward
5.4 18.3, 19.9 Ever engaged in sexual intercourse: Male 15 years Not applicable Toward
5.4 18.3, 19.9 Ever engaged in sexual intercourse: Female
17 years Not applicable Away
5.4 18.3, 19.9 Ever engaged in sexual intercourse: Male 17 years Not applicable Toward
5.4a 18.3a,19.9a Ever engaged in sexual intercourse: Black male
15 years Not applicable Toward
5.4b 18.3b,19.9b Ever engaged in sexual intercourse: Black male
17 years Not applicable Toward
5.4c 18.3c,19.9c Ever engaged in sexual intercourse: Black female
15-17 years Not applicable Toward
5.5 18.15,19.16 Abstinence from sexual intercourse for previous
3 months: Female 15-17 years Not applicable Toward
5.5 18.15,19.16 Abstinence from sexual intercourse for previous
3 months: Male 15-17 years Not applicable Toward
5.6 Contraception use: First intercourse, female
15-19 years Not applicable Toward
5.6 Contraception use: Recent intercourse, female
15-19 years Not applicable Away
5.6 Contraception use: Oral contraceptive and condom
at most recent intercourse, female 15-19 years Not applicable Toward
5.6 Contraception use: First intercourse, male
15-19 years Not applicable Toward
5.6 Contraception use: Birth control pills and condoms at
most recent intercourse, in school, male
15-17 years Not applicable Toward
5.6 Contraception use: Condom and pill use at last
intercourse, male 17-19 years Not applicable Toward
5.8 Persons 13-18 years who have discussed sexuality
with parents Not applicable Toward
5.10 14.12 Age-appropriate preconception counseling by
clinicians: Inquiry about family planning, nurse
practitioners Not applicable Away
5.10 14.12 Age-appropriate preconception counseling by
clinicians: Counseling about family planning, nurse
practitioners Not applicable
Away
372 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Adolescents and young adults—Con.
5.10 14.12 Age-appropriate preconception counseling by
clinicians: Counseling about family planning,
obstetricians/gynecologists Not applicable Met
5.12 Contraception use: Female 15-44 years Not applicable Toward
5.12c Contraception use: Under 200% poverty, female
15-19 years Narrowing Toward
6.1a 7.2a Suicide: 15-19 years Not applicable Toward
6.1b 7.2b Suicide: Male 20-34 years Widening Toward
6.2 7.8 Suicide attempts:14-17 years Not applicable Away
6.2a 7.8a Suicide attempts: Female 14-17 years Not applicable Away
6.3 Mental disorders: 18 years and under Not applicable Cannot assess
6.8 People 18 years and over seeking help with
emotional/personal problems Not applicable Met
7.1b Homicide: Spouse 15-34 years Not applicable Met
7.1c Homicide: Black male 15-34 years Widening Toward
7.1d Homicide: Hispanic male 15-34 years Narrowing Toward
7.1e Homicide: Black female 15-34 years Narrowing Met
7.4 Child abuse and neglect Not applicable Away
7.4a Child abuse and neglect: Physical abuse Not applicable Away
7.4b Child abuse and neglect: Sexual abuse Not applicable Away
7.4c Child abuse and neglect: Emotional abuse Not applicable Away
7.4d Child abuse and neglect: Neglect Not applicable Away
7.7a Rape and attempted rape: Female 12-34 years Widening Toward
7.9 Physical fighting: 14-17 years Not applicable Met
7.9a Physical fighting: Non-Hispanic black male
14-17 years Not applicable Met
7.10 Weapon-carrying: 14-17 years Not applicable Met
7.10a Weapon-carrying: Non-Hispanic black 14-17 years Not applicable Met
7.16 Conflict resolution in a required course: Middle/junior
and senior high schools Not applicable Cannot assess
8.2 Completion of high school: 18-24 years Not applicable Away
8.2a Completion of high school: Hispanic 18-24 years Not applicable Toward
8.2b Completion of high school: Black 18-24 years Not applicable Away
8.4 Schools with comprehensive school health education:
All eight criteria met Not applicable Cannot assess
8.5 Health promotion in postsecondary institutions:
Higher education institutions Not applicable Cannot assess
8.9 Family discussion of health issues: 10 years and
over Not applicable Away
8.9 Family discussion of health issues: Engaging in
family discussion of HIV/AIDS, grades 9-12 Not applicable Toward
8.10 States with community health programs addressing
at least three Healthy People 2000 objectives Not applicable Cannot assess
8.13 Television partnerships with community organizations
for health promotion Not applicable Met
9.3b Motor vehicle crash deaths: 15-24 years Narrowing Toward
9.5b Drowning deaths: Male 15-34 years Narrowing Toward
9.18 Injury prevention instruction in required course:
Middle/junior and senior high schools Not applicable Met
9.19 13.16 Protective equipment: Baseball/softball, Mouth guard Not applicable Toward
9.19 13.16 Protective equipment: Baseball/softball, Headgear Not applicable Toward
9.19 13.16 Protective equipment: Football, Mouth guard Not applicable Toward
9.19 13.16 Protective equipment: Football, Headgear Not applicable Toward
9.19 13.16 Protective equipment: Soccer, Mouth guard Not applicable Toward
9.19 13.16 Protective equipment: Soccer, Headgear Not applicable Toward
10.2f Nonfatal work-related injuries: 15-17 years Not applicable Toward
Healthy People 2000 Final Review 373

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Adolescents and young adults—Con.
11.1b Asthma hospitalizations: 14 years and under Widening Toward
13.1 Dental caries: 15 years Not applicable Toward
13.1d Dental caries: American Indian/Alaska Native
15 years
Not applicable Toward
13.2 Untreated dental caries: 15 years Not applicable Toward
13.2e Untreated dental caries: Parents who have less than
a high school education, 15 years
Not applicable Toward
13.2f Untreated dental caries: American Indian/Alaska
Native 15 years
Not applicable Toward
13.2g Untreated dental caries: Black 15 years Not applicable Toward
13.2h Untreated dental caries: Hispanic 15 years Not applicable Toward
13.8 Protective sealants: 14 years Not applicable Toward
13.8b Protective sealants: Black 14 years Not applicable Toward
13.8d Protective sealants: Hispanic 14 years Not applicable Toward
17.20 Service systems for children with or at risk of chronic
and disabling conditions Not applicable Cannot assess
17.22 22.4 Identify gaps in health data Not applicable Toward
17.22 22.4 Establish mechanisms to meet data needs Not applicable Toward
18.1 AIDS incidence: 18 years and over Not applicable Met
18.1b AIDS incidence: Black (non-Hispanic) 18 years and
over Not applicable Met
18.1c AIDS incidence: Hispanic 18 years and over Not applicable Met
18.1d AIDS incidence: Female 18 years and over Not applicable Met
18.2 HIV prevalence: 13 years and over Not applicable Met
18.2c HIV prevalence: Females 15–44 years giving birth to
live-born infants Not applicable No change
18.4a 19.10a Condom use at last sexual intercourse: Female
15-19 years Not applicable Toward
18.4b 19.10b Condom use at last sexual intercourse: Male
15-19 years Not applicable Cannot assess
18.4d 19.10d Condom use at last sexual intercourse: Black female
15-44 years Not applicable Toward
18.10 19.12 HIV and other STD education curricula: Schools
offering at least one STD class Not applicable Met
18.10 19.12 HIV and other STD education curricula: Middle and
senior high schools, HIV prevention in required
courses Not applicable Cannot assess
18.10 19.12 HIV and other STD education curricula: Middle and
senior high schools, STD prevention required
courses Not applicable Cannot assess
18.11 19.17 HIV and STD education for students at colleges and
universities: Given AIDS or HIV infection prevention
information Not applicable Cannot assess
18.11 19.17 HIV and STD education for students at colleges and
universities: Given STD prevention information Not applicable Cannot assess
18.11 19.17 HIV and STD education for students at colleges and
universities: AIDS or HIV in a college class Not applicable Cannot assess
19.1b Gonorrhea: 15-19 years Widening Toward
19.1c Gonorrhea: Female 15-44 years Not applicable Toward
19.2 Chlamydia prevalence: Female 15-19 years Not applicable Toward
19.2 Chlamydia prevalence: Female 20-24 years Not applicable Met
19.3 Primary and secondary syphilis Not applicable Met
19.3a Primary and secondary syphilis: Black Not applicable Met
19.4 Congenital syphilis in infants under 1 year Not applicable Met
19.4a Congenital syphilis in infants under 1 year: Black Not applicable Met
19.4b Congenital syphilis in infants under 1 year: Hispanic Not applicable Met
374 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Adolescents and young adults—Con.
19.5 First time consultations: Genital herpes Not applicable Away
19.5 First time consultations: Genital warts Not applicable Met
19.6 Pelvic inflammatory disease hospitalizations:
15-44 years Not applicable Toward
19.6 Pelvic inflammatory disease initial visits to physicians Not applicable Met
19.6a Pelvic inflammatory disease hospitalizations: Black
15-44 years Not applicable Toward
19.6b Pelvic inflammatory disease hospitalizations:
15-19 years Narrowing Toward
19.7 20.3b, 20.3c Sexually transmitted Hepatitis B Not applicable Met
19.8 Repeat gonorrhea infection Not applicable Toward
19.8a Repeat gonorrhea infection: Black Not applicable Toward
20.1 Vaccine-preventable diseases: Diphtheria 25 years
and under Not applicable Met
20.1 Vaccine-preventable diseases: Tetanus 25 years and
under Not applicable Away
20.1 Vaccine-preventable diseases: Polio Not applicable Met
20.1 Vaccine-preventable diseases: Measles Not applicable Toward
20.1 Vaccine-preventable diseases: Rubella Not applicable Away
20.1 Vaccine-preventable diseases: Congenital Rubella
Syndrome Not applicable No change
20.1 Vaccine-preventable diseases: Mumps Not applicable Met
20.1 Vaccine-preventable diseases: Pertussis Not applicable Away
20.11 Basic immunization series among children in
kindergarten through postsecondary education
institutions Cannot assess Met
21.3 Usual source of care: 18 years and over Not applicable Toward
21.3a Usual source of care: Hispanic 18 years and over Not applicable Toward
21.3a Usual source of care: Mexican American 18 years
and over Not applicable Toward
21.3b Usual source of care: Black 18 years and over Not applicable Toward
21.3c Usual source of care: Low-income people 18 years
and over Not applicable Toward
21.3d Usual source of care: American Indian/Alaska Native
18 years and over Not applicable Toward
21.3e Usual source of care: Asian/Pacific Islander 18 years
and over Not applicable Toward
21.4 People without health care coverage: Under 65 years Not applicable Away
21.4a People without health care coverage: American
Indian/Alaska Native under 65 years Not applicable Away
21.4b People without health care coverage: Hispanic under
65 years Not applicable Away
21.4b People without health care coverage: Mexican
American under 65 years Not applicable Away
21.4b People without health care coverage: Puerto Rican
under 65 years Not applicable Toward
21.4b People without health care coverage: Cuban under
65 years Not applicable Away
21.4c People without health care coverage: Black under
65 years Not applicable Toward
21.5 Clinical preventive services from Federal programs:
Screening Not applicable Cannot assess
21.5 Clinical preventive services from Federal programs:
Counseling Not applicable Cannot assess
21.5 Clinical preventive services from Federal programs:
Immunizations Not applicable Cannot assess
Healthy People 2000 Final Review 375

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Adolescents and young adults—Con.
21.6 Provision of recommended services, children:
Hemoglobin/hematocrit, Pediatricians Not applicable Cannot assess
21.6 Provision of recommended services, children:
Hemoglobin/hematocrit, Nurse practitioners Not applicable Cannot assess
21.6 Provision of recommended services, children:
Hemoglobin/hematocrit, Family physicians Not applicable Cannot assess
21.6 Provision of recommended services, children: Eye
exam, Pediatricians Not applicable Cannot assess
21.6 Provision of recommended services, children: Eye
exam, Nurse practitioners Not applicable Away
21.6 Provision of recommended services, children: Eye
exam, Family physicians Not applicable Cannot assess
21.6 Provision of recommended services, children: Blood
pressure, Pediatricians Not applicable Cannot assess
21.6 Provision of recommended services, children: Blood
pressure, Nurse practitioners Not applicable Away
21.6 Provision of recommended services, children: Blood
pressure, Family physicians Not applicable Cannot assess
21.6 Provision of recommended services, children: Height
and weight, Pediatricians Not applicable Cannot assess
21.6 Provision of recommended services, children: Height
and weight, Nurse practitioners Not applicable Away
21.6 Provision of recommended services, children: Height
and weight, Family physicians Not applicable Cannot assess
21.6 Provision of recommended services, children:
Diphtheria-tetanus-pertussis (DTP) vaccination,
Pediatricians Not applicable Cannot assess
21.6 Provision of recommended services, children: DTP
vaccination, Nurse practitioners Not applicable Toward
21.6 Provision of recommended services, children: DTP
vaccination, Family physicians Not applicable Cannot assess
21.6 Provision of recommended services, children: Oral
polio vaccination, Pediatricians Not applicable Cannot assess
21.6 Provision of recommended services, children: Oral
polio vaccination, Nurse practitioners Not applicable Toward
21.6 Provision of recommended services, children: Oral
polio vaccination, Family physicians Not applicable Cannot assess
21.6 Provision of recommended services, children:
Tetanus-diphtheria booster, Pediatricians Not applicable Cannot assess
21.6 Provision of recommended services, children:
Tetanus-diphtheria booster, Nurse practitioners Not applicable Toward
21.6 Provision of recommended services, children:
Tetanus-diphtheria booster, Family physicians Not applicable Cannot assess
21.6 Provision of recommended services, children:
Haemophilus influenzae type b (Hib) vaccination,
Pediatricians Not applicable Cannot assess
21.6 Provision of recommended services, children: Hib
vaccination, Nurse practitioners Not applicable Toward
21.6 Provision of recommended services, children: Hib
vaccination, Family physicians Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Tetanus-diphtheria booster, Nurse
practitioners Not applicable Toward
21.6 Provision of recommended services, adults 18 years
and over: Tetanus-diphtheria booster,
Obstetricians/gynecologists Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Tetanus-diphtheria booster, Internists Not applicable
Cannot assess
376 Healthy People 2000 Final Review

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Adolescents and young adults—Con.
21.6 Provision of recommended services, adults 18 years
and over: Tetanus-diphtheria booster, Family
physicians Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Blood pressure, Nurse practioners Not applicable Toward
21.6 Provision of recommended services, adults 18 years
and over: Blood pressure,
Obstetrician/gynecologists Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Blood pressure, Internists Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Blood pressure, Family physians Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Cholesterol level, Nurse practitioners Not applicable Toward
21.6 Provision of recommended services, adults 18 years
and over: Cholesterol level,
Obstetricians/gynecologists Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Cholesterol level, Internists Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Cholesterol level, Family physicians Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Breast exam, Nurse practitioners Not applicable Away
21.6 Provision of recommended services, adults 18 years
and over: Breast exam,
Obstetricians/gynecologists Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Breast exam, Internists Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Breast exam, Family physicians Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Pap smear, Nurse practitioners Not applicable Away
21.6 Provision of recommended services, adults 18 years
and over: Pap smear, Obstetricians/gynecologists Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Pap smear, Internists Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Pap smear, Family physicians Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Mammogram, Nurse practitioners Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Mammogram, Obstetricians/gynecologists Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Mammogram, Internists Not applicable Cannot assess
21.6 Provision of recommended services, adults 18 years
and over: Mammogram, Family physicians Not applicable Cannot assess
21.7 Local health department assurance of access to
essential clinical preventive services: Proportion of
people served Not applicable Cannot assess
21.7 Local health department assurance of access to
essential clinical preventive services: Assess
services provided to the local population Not applicable Cannot assess
21.7 Local health department assurance of access to
essential clinical preventive services: Collect data Not applicable Cannot assess
21.7 Local health department assurance of access to
essential clinical preventive services: Evaluate
whether a gap exists Not applicable Cannot assess
Healthy People 2000 Final Review 377

Table VII. Special population objectives included in Healthy People 2000—Con.
Objective
Number
Duplicate
objective Short objective text
Disparity
assessment in
relation to
reference
population*
Objective
status in
relation to
target
Adolescents and Young adults—Con.
21.7 Local health department assurance of access to
essential clinical preventive services: Provide Not applicable Cannot assess
22.2 Objectives with national data sources Not applicable Toward
22.2a State level data for at least two-thirds of State
objectives Not applicable
Toward
* For method of calculation, see Appendix Technical Notes.
Special population objectives included in Healthy People 2000: Adolescents and Young adults
Objective status
Disparity Measure
Eliminated
Narrowing
gap
No
change
Widening
gap
Cannot
assess
Disparity
total
Not a disparity
measure
Overall
total
Met..................... 0 2 0 0 3 5 32 37
Toward .................. 0 5 0 5 0 10 87 97
Mixed/No change .......... 0 0 0 0 0 0 4 4
Away ................... 0 0 0 0 1 1 42 43
Cannot assess ............ 0 0 0 0 0 0 62 62
TOTAL.................. 0 7 0 5 4 16 227 243
378 Healthy People 2000 Final Review
