
National Center for Science and Engineering Statistics | NCSES 21-201
3
Disclaimer
Working papers are intended to report exploratory results of research and analysis undertaken by the National Center for
Science and Engineering Statistics (NCSES) within the National Science Foundation (NSF). Any opinions, findings,
conclusions or recommendations expressed in this working paper do not necessarily reflect the views of the National
Science Foundation. This working paper has been released to inform interested parties of ongoing research or activities
and to encourage further discussion of the topic.
This working paper represents the work of the authors and is not considered official government statistics.

National Center for Science and Engineering Statistics | NCSES 21-201
4
Abstract
Most studies of innovation that examine determinants of success or failure look exclusively at the factors that make
innovation successful. To better understand causes and patterns of unsuccessful or failed innovation within firms, we
conducted 19 case studies of failure from the past 20 years. These cases draw primarily from coverage by the popular
and trade presses and highlight clear models of failure. This paper examines all aspects of the product or business
process life cycle and found that innovation failure typically revolves around firms’ inadequate pre-launch development
activities, which lead to products that do not function properly or in which the marketplace has no interest. There is a gap
between innovation, market-specific product launch, and successful innovation as typically presented in the literature.
This paper exploits this gap and identifies those products and processes that are introduced but fail to achieve
widespread adoption as unsuccessful innovation or innovation failures.

National Center for Science and Engineering Statistics | NCSES 21-201
5
Introduction
Background
A large volume of research has addressed what makes new product development successful (Ernst 2017; Florén et al.
2018). Such studies have been beneficial to a wide array of actors, including business people and policymakers who hope
to replicate the successes experienced by today’s most valuable corporations and harness the growth they generate. In
contrast to the volume of literature on successful new product development and despite Silicon Valley’s unofficial motto
of “Fail Fast, Fail Often,” little attention has been paid in the empirical literature to innovation failure.
Some of this inattention probably stems from the scholarly view of failure as simply the opposite of commercial success
and scholars’ implicit accommodation of both concepts within empirical and conceptual studies of success. Though the
two concepts are related, the National Center for Science and Engineering Statistics (NCSES) within the National Science
Foundation (NSF) and SRI International (SRI) have identified innovation failure as a gap in current research.
The Oslo Manual 2018: Guidelines for Collecting and Interpreting Innovation Data (OECD/Eurostat 2018) is used in
developing NCSES’s surveys relating to innovation and in this paper. The Oslo Manual definition of innovation does not
require an innovation to be a commercial, financial, or strategic success at the time of measurement (OECD/Eurostat
2018:71). A product innovation can fail commercially or a business process innovation may require more time to meet its
objectives. The Oslo Manual also states that innovation activities can create knowledge or information that is not used to
introduce an innovation during the observation period. This includes knowledge from activities that fail to meet their
primary innovation goals.
Unsuccessful or failed innovations often receive significant attention in the popular press. These failures also often occur
despite early optimism about the ultimately unsuccessful inventions’ potential to positively disrupt current business
practices, markets, and entire industries. Direct understanding of product innovation failure potentially represents an
untapped resource for decisionmakers in the private and public sectors. If policymakers understand the reasons for
innovation failures—and as a result business failures—they may be able to develop policy to prevent those failures.
Research into business process innovation failure also promises to be useful. Most current research on business process
innovation focuses on the impact of such innovation on firm or macroeconomic performance rather than the
determinants of why it succeeds or fails.
The work reported herein relies on case studies to identify instances of innovation failure, the causes of failure, and the
points in the innovation process at which failure is likely to occur. Analysis of these case studies identifies patterns of
innovation failure and common root causes. The case studies reveal a narrative that explains when and why an innovation
failed and highlight when the critical actions and decisions by inventors that led to the failures occurred.
Shedding light on patterns of innovation failure provides new perspective on understanding data from NCSES surveys on
firm innovation. In particular, it suggests closer survey attention be paid to qualitative differences in the reasons firms
abandoned innovation activities. In many cases, companies may be giving up on innovation projects for good reasons,
such as acquiring new data that suggest weak market interest in the planned innovation.
This paper uses the Oslo Manual’s definition of innovation as “a new or improved product or process (or combination
thereof) that differs significantly from the unit’s previous products or processes and that has been made available to
potential users (product) or brought into use by the unit (process)” (OECD 2018:22). The Oslo Manual distinguishes
between two broad types of innovation: product and business process innovation:
Product innovation. A new or improved good or service that differs significantly from the firm’s previous goods or
services and that has been introduced on the market.
National Center for Science and Engineering Statistics | NCSES 21-201
6
Business process innovation. A new or improved business process for one or more business functions that differs
significantly from the firm’s previous business processes and that has been brought into use by the firm. (OECD
2018:23)
The Oslo Manual defines innovation activities as follows:
Innovation activities include “all developmental, financial and commercial activities undertaken by a firm that are intended
to result in an innovation for the firm” (OECD 2018:70). The Oslo Manual further notes that “a business innovation is a new
or improved product or business process (or combination thereof) that differs significantly from the firm’s previous
products or business processes and that has been introduced on the market or brought into use by the firm” (OECD
2018:68).
The terms unsuccessful innovation or innovation failure are used in this paper to facilitate discussion. An unsuccessful
innovation or innovation failure is a product or business process that did not meet a company’s expectations when it was
introduced to the market for a variety of reasons; it is not a comment on the quality of the product or business process.
The Innovation Horizon and the Product or Business Process Life Cycle
To understand how and why a new product or business process innovation fails, the authors examined the timing of
failure, starting with the product or business process life cycle. The product or business process life cycle traces the
product or process from launch, through its growth phase, its maturity phase, and finally to the period during which it
declines and is phased out of the market.
For the purposes of this paper, products were considered eligible to be designated as innovation failures only in the
launch, growth, or maturity phases. The natural decline of a product or business process after a successful marketplace
run was not considered an innovation failure. Failures at launch occur when products are burdened from the start or near-
start of their commercial introduction with challenges that prevent gaining appreciable marketplace acceptance. Failures
during the growth stage are when a product is successfully launched but then faces significant marketplace challenges
when innovators attempt to grow rapidly. Failures in the maturity phase are rare; they occur when a widely adopted
product or business process faces sudden challenges in the marketplace that abruptly and significantly diminish or
destroy its marketplace value.
In addition to references to the product or business process life cycle, this paper defines the innovation horizon as the
period during which innovation activities can occur (see figure 1). Innovation-related activities can happen before and
after a new product is launched. Pre-launch innovation activities include ideation and product (or business process)
development. Post-launch innovation failures often can be traced to development shortcomings that occurred during pre-
launch activities. Innovation activities often continue after new products or processes are launched, as companies refine
their offerings in response to marketplace feedback, competitive pressure, and other factors. Maturity is the final state for
product or business process refinement, and so innovation activities come to a stop, by definition, once a product enters
this phase. For this reason, the innovation horizon includes only the ideation, development, launch, and growth periods.

National Center for Science and Engineering Statistics | NCSES 21-201
7
FIGURE 1
Innovation horizon and product life cycle
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.
This paper uses the same phases of launch, growth, maturity, and decline for our analysis of business process
innovations. In those locations in which the text refers to product innovation, the same concepts refer to business process
innovation.

National Center for Science and Engineering Statistics | NCSES 21-201
8
Approach
To identify failure root causes of innovation, NCSES and SRI conducted a series of case studies focusing on innovations
brought to market after the year 2000. Failure root causes are identified as follows:
These five categories were identified by considering the primary causes of failure across the case studies. Each case
study can be clearly linked to one of these five root causes as the primary reason an innovation failed in the marketplace.
In several cases, more than one factor ultimately contributed to an innovation’s failure.
Information on each case of innovation failure came mainly from press reporting. Case studies of commercially
successful product innovations are common in academic literature, but coverage of product innovation failures is less
prevalent and more likely to be found in the popular press. Following the Oslo Manual definition of innovation, for a new
product or business process to be considered in this analysis, it must have been launched, that is, introduced on the
market or brought into use by the firm prior to its failure. A product or business process that does not exit the
development stage is not considered an innovation.
The failure timing for each innovation failure was identified as the product or business process life cycle phase during
which it failed (launch, growth, or maturity). The five earliest phases of the life cycle (including the two pre-launch
innovation activities) were used to identify when the root cause of the failure took place; this is the failure root cause
timing. For example, a product might fail at launch (its failure timing) due to inadequacies during the product development
phase (its failure root cause timing). Thus, failure timing can occur during launch, growth, or maturity; failure root cause
timing can occur during ideation, development, launch, growth, or maturity.
Failure timing and failure root cause timing were used as the primary case study parameters for analysis. This paper uses
these data to identify common patterns of innovation failure. In addition, for failure root cause, failure timing, and failure
root cause timing, each case study classified a failed product or business process innovation and its sector as radical or
incremental. Radical innovations are new-to-world innovations that fundamentally change the way value is delivered to the
marketplace or that create wholly new markets. Incremental innovation refers to improvements to existing products and
processes that make them better, cheaper, faster but do not change the nature of consumer value.
When examined together, failure root cause, failure timing, and failure root cause timing tell the story of innovation failure.
They provide a narrative that explains when and why an innovation failed and highlight when the critical actions and
decisions by inventors that led to the failures occurred.
No market demand. A product or business process was developed and brought to market, but there was little market
demand for it.
Poor performance. A product or business process was developed and brought to market, but it failed to function as
intended.
Insufficient complementary assets. A product or business process was developed and brought to market, but the
adjacent business inputs required for its successful customer use were not sufficiently available.
Poorly defendable position. A product or business process was developed and brought to market and met with positive
market interest, but the innovator was unable to secure the innovation’s market position, for example, by way of
intellectual property protection.
Regulatory restriction. A product or business process was developed and brought to market, but regulatory restrictions
on its use limited its economic value.
●
●
●
●
●

National Center for Science and Engineering Statistics | NCSES 21-201
9
Results
Case Studies
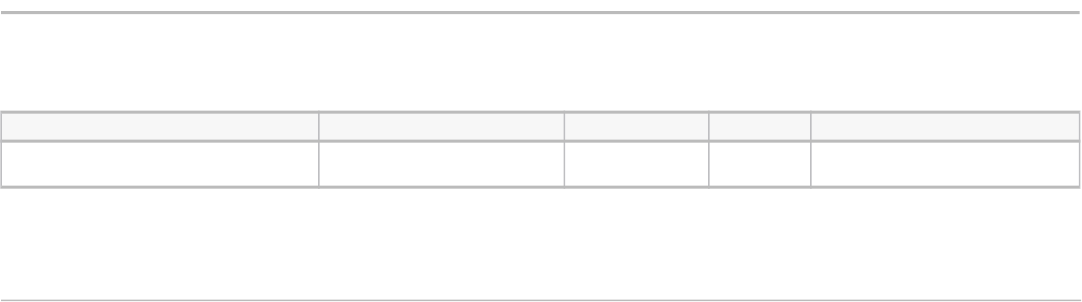
This paper looks at 19 total case studies in 9 industries. Cases studies per sector are reported in table 1. The complete
list of case studies is provided in table 2. Eighteen of the case studies were of product innovations (15 of goods and 3 of
services) and one of new business processes. The radical and incremental case studies were more evenly split, with 10
radical innovations and 9 incremental innovations accounted for within the case studies.
TABLE 1
Case studies per sector
(Number)
Sector Case studies
Consumer electronics 4
Mobile electronics and services 4
Transportation 3
Household appliances 2
Media 2
Clothing 1
Financial technology 1
Medical technology 1
Software 1
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.
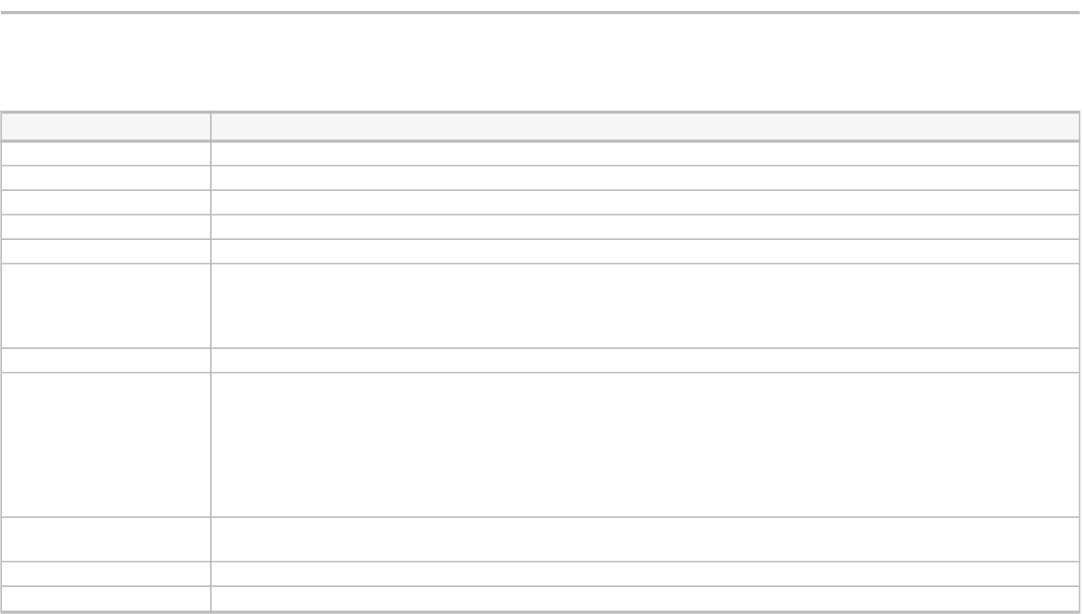
TABLE 2
Case studies by sector, category of innovation, type of innovation, and failure root cause
(Case index)
Case name Sector Innovation type Failure type Failure root cause
Google Glass Mobile electronics and services Product (good) Radical No market demand
Microsoft Windows Vista Software Product (good) Incremental Poor performance
Samsung Galaxy Note 7 Mobile electronics and services Product (good) Incremental Poor performance
iTunes Ping Media Product (service) Incremental Poor performance
DVD-Audio Consumer electronics Product (good) Incremental No market demand
Hoverboards Transportation Product (good) Radical Poorly defendable position
Boeing 737 MAX Transportation Product (good) Incremental Poor performance
Zozosuit Clothing Product (good) Radical Insufficient complementary assets
Juicero Household appliances Product (good) Radical No market demand
Google+ Media Product (service) Incremental No market demand
Ubuntu Phone Mobile electronics and services Product (good) Incremental Poor performance
Sedasys Medical technology Product (good) Radical No market demand
Google Wallet Consumer finance Product (service) Radical Insufficient complementary assets
Segway Transportation Product (good) Radical No market demand
Iridium Satellite Phone Mobile electronics and services Product (good) Radical No market demand
3-D Television Consumer electronics Product (good) Incremental No market demand
Wii U Consumer electronics Product (good) Incremental Insufficient complementary assets
Laundroid Household appliances Product (good) Radical Poor performance
National Center for Science and Engineering Statistics | NCSES 21-201
10
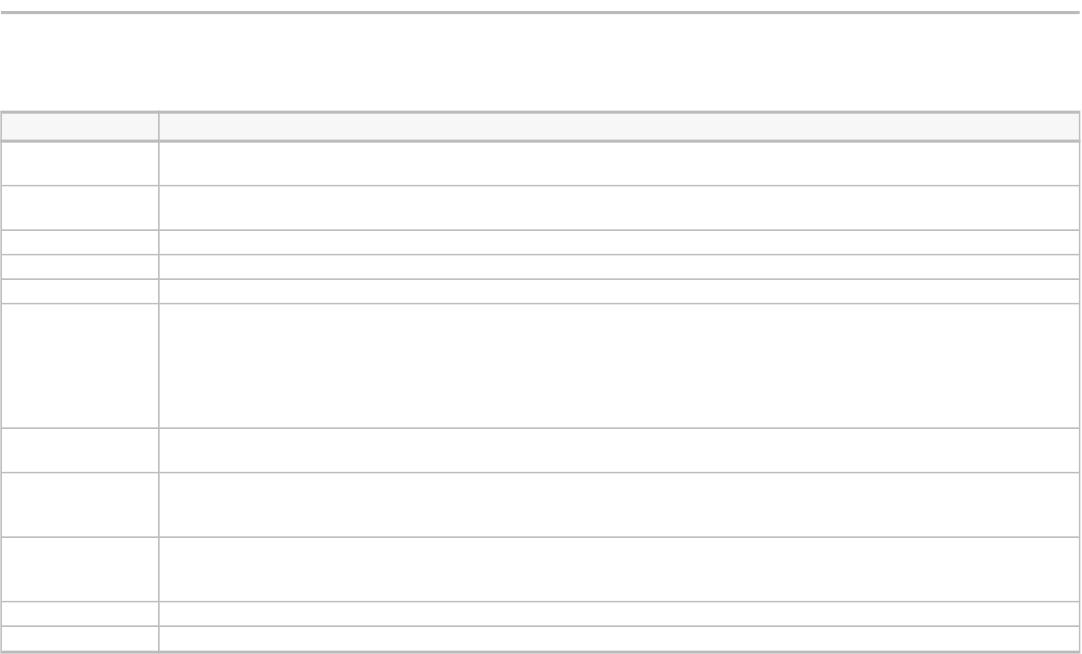
TABLE 2
Case studies by sector, category of innovation, type of innovation, and failure root cause
(Case index)
Case name Sector Innovation type Failure type Failure root cause
Sony BMG Extended Copy Protection
(XCP)
Consumer electronics Business process Radical Regulatory restrictions
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.
Case Study Examples
The "Appendix: Case Studies" section provides summaries for each of the 19 case studies. Four of them are detailed here
by way of example.
Laundroid (Launched 2018)
Created by the Japanese company Seven Dreamers, Laundroid was an artificial intelligence (AI)-driven garment-folding
automated wardrobe. After the user placed unfolded clothes into the “insert box,” multiple robotic arms lifted each
garment, which was then scanned by cameras. Data from the cameras were sent to a server on which the AI would
analyze the item and decide the best way to handle and fold it. During the analysis a companion app would track each
piece of clothing that passed through Laundroid, allowing users to catalog the items by owner or type (Salkin 2017).
Its potential utility as an online wardrobe organizer notwithstanding, the device was beset with technical problems in its
basic function of folding. At its debut at the Consumer Electronics Show (CES) 2018, the device was unable to fold socks,
had difficulty with T-shirts and dark colored garments, and jammed at least once and had to be cleared by technicians
(Barry 2018). Even when functioning up to specification, it took about 5–10 minutes to fold one T-shirt. Laundroid was
also extremely large and bulky, and, with a price point of around $16,000, was not attractive to most consumers.
The device may have seemed even less appealing in comparison to its California competitor Foldimate, which, for a cost
of around $1,000, can fold a T-shirt in 5 seconds (though Foldimate requires the user to feed each garment into the
device) (Summers 2019). Despite backing by Panasonic and Daiwa House, Seven Dreamers went bankrupt after taking
out around $20 million in debt in an effort to push Laundroid to market; however, it appears Seven Dreamers has since
abandoned the project (McDermott 2019).
Juicero (Launched 2016)
A Silicon Valley-based startup, Juicero, raised $120 million from investors to develop a Wi-Fi-connected, app-enabled, at-
home juicer. The product, also called Juicero, intended to ease the at-home juicing process by using Juicero-branded
produce packets that would be squeezed by the machine, reducing the amount of clean-up and manual labor that was
typically required to produce fruit and vegetable juices at home. The countertop machine originally sold for $700 and
consumers were required to purchase product-specific produce packets; the packets could be purchased individually (for
$5–$8 per packet) or through a weekly subscription service. After launching in spring 2016, the price of the machine was
eventually lowered to $400.
To use Juicero, the device had to be connected to a user’s Wi-Fi network. This connectivity was required for the machine’s
QR (quick response) code scanner to scan the produce packets and ensure that the packets had not expired; if the packet
had expired, the machine would refuse to squeeze the packet and produce juice. Additionally, if there was a recall of a
certain fruit or vegetable, the machine would refuse to squeeze packets that contained the recalled produce. These
alleged safety features were referred to as the world’s “first closed loop food safety system,” in which Juicero procured,
National Center for Science and Engineering Statistics | NCSES 21-201
11
packaged, shipped, and produced juices directly for the consumer. This closed-loop system advertised Juicero as more
than just an at-home juicing machine, but also a protector of consumer health (Thompson 2017). To some consumers,
however, it may have been seen as attempt to lock them into purchasing Juicero-branded products so long as they
intended to use the Juicero countertop machine.
In spring 2017, the mechanics of the machine were proven to be over-engineered and unnecessary to produce juices from
Juicero-branded produce packets. In a video uploaded to a popular news site, it was shown that a pair of human hands
was able to produce roughly the same amount of juice from the Juicero produce packets in less time than the supposed
four tons of force produced by Juicero’s metal plates (Huet and Zaleski 2017). The combination of the high price point
and the revelation that the machine was, at most, as effective as human hands caused demand for Juicero to collapse.
Before closing operations, Juicero offered a full refund to anyone who had purchased a machine. Juicero executives cited
the reason for closure to be that “creating an effective manufacturing and distribution system for a nationwide customer
base requires infrastructure that [Juicero] cannot achieve on [its] own as a standalone business” (Levin 2017).
Sedasys (Launched 2015)
Launched in 2015, Sedasys was developed by Johnson & Johnson to assist in administering anesthesia to patients at U.S.
hospitals. The purpose of Sedasys was to automate the sedation of patients undergoing certain low-risk procedures,
reducing the need for anesthesiologists. The removal of anesthesiologists was intended to significantly reduce the cost of
routine, low-risk procedures conducted at hospitals, such as colonoscopies; anesthesiologists cost hospitals anywhere
from $600 to $2,000 per procedure, whereas Sedasys cost roughly $150 per procedure (Tobe 2013). Sedasys was able to
administer the popular anesthetic propofol at rates specific to each patient, allowing for the same quick recovery times as
when anesthesiologists administered the drug (Frankel 2015).
To sell Sedasys to health care providers, Johnson & Johnson was required to secure approval from the U.S. Food and
Drug Administration (FDA). Due to the machine’s anticipated displacement of highly paid anesthesiologists, Johnson &
Johnson faced strong opposition from the American Society of Anesthesiologists (ASA). To appease both the FDA and
the ASA, Johnson & Johnson agreed to limit the use case of Sedasys to endoscopic procedures, which are inherently low-
risk procedures with relatively short recovery times. When using Sedasys for these procedures, a registered nurse would
initiate the sedation process with the press of a button, which would begin the flow of propofol to the patient via IV. The
Sedasys machine would monitor patient breathing, blood oxygen level, and heart rate, ensuring that the patient was not
having an adverse reaction to the anesthetic; if Sedasys detected any abnormalities, it could slow the flow of propofol or
stop it altogether (Frankel 2015).
Though Johnson & Johnson was able to secure FDA and ASA approval for the more limited use case of endoscopic
procedures, Sedasys did not find commercial success in the medical technologies market. A primary cause of Sedasys’s
underperformance was the limited use case; while endoscopic procedures are among the most common services
conducted in U.S. hospitals, the inability of hospitals to use Sedasys for more intensive procedures limited the potential
cost savings. Similarly, though ASA approved of the limited use case, anesthesiologists were still needed by hospitals for
more complicated procedures. It became easier for hospitals not to purchase Sedasys at all rather than use them only
under certain circumstances. Due to the limited sales, Johnson & Johnson stopped selling Sedasys in 2016.
Google Glass (Launched 2013)
Launched to a limited group of pre-selected individuals in 2013 at $1,500, Google Glass (Glass) was advertised as a voice-
controlled head-mounted computer that could be worn similarly to a pair of glasses.
1
Glass was web-connected, allowing
the device to do many of the things a smartphone could do, such as make phone calls, browse the Internet, and take
photos (Dashevsky and Hachman 2014). With a battery life of 2 to 3 hours, Glass was primarily a means of
complementing smartphones, rather than replacing them, which it did by integrating smartphone capabilities into a
wearable device that did not require manually accessing a smartphone every time a call needed to be made or answered.
National Center for Science and Engineering Statistics | NCSES 21-201
12
While Glass was marketed as a device to be used by the public, it was initially offered only to a select group of individuals.
Termed “Glass Explorers,” individuals in this group were invited by Google to purchase, at full price, a Glass set to test
before its full release. Glass was advertised for use by the public, though Google did not identify any specific use cases for
the device for this group. Throughout the development phase, Glass developers debated the product’s intended audience,
especially its fashion appeal versus its utilitarian/functional appeal (Weider 2020). This resulted in no clear use cases for
Glass that set it apart from smartphones, which had much longer battery lives and millions of third-party applications that
could be downloaded and used. In addition to the hardware and software obstacles, Glass could be exceptionally
awkward in many social contexts. People found it unsettling that they could be photographed or video-recorded without
their knowledge or consent, which led to some restaurants and businesses banning Glass from their premises.
The hardware, software, and social considerations prevented Glass from scaling to a full-audience release. The company
cancelled any plans to further produce Glass for public purchase in January 2015. Since then, several commercial and
industrial sectors have identified specific use cases in which Glass offers functionality to specialized workers. This pivot
from general to specialized use applications in certain sectors was largely possible due to Glass’s core functionality,
which allowed a worker to retrieve and analyze data in real time without needing to use their hands.
An example of a specialized application is Boeing’s use of Glass in the assembly of wire harnesses for airplanes. In 2014,
Boeing purchased several Glass headsets and launched an internal company project to identify a use case for Glass.
Boeing was interested in a hands-free device that would allow workers to view instructions for assembling wire harnesses
in airplanes in real time, accelerating the process and minimizing the opportunity for error. To complement the hardware
functionality of Glass, Boeing developed an application that relied on a network of QR codes that could be scanned by
Glass. When an assembly worker scanned a code, instructions would appear on Glass that directed the user to the correct
parts. Assembly workers could also live stream their view to experts at other Boeing locations who could provide
instructions for issues in real time (Sacco 2016).
Analysis
Analysis of the 19 cases revealed five general categories of innovation failure, which we have called failure root causes:
no market demand, poor performance, poorly defendable position, insufficient complementary assets, and regulatory
restrictions.
Considering the three possible values for innovation failure timing and five possible values for root cause timing, there are
15 (3 x 5) possible combinations of the two. Because the innovation horizon has been defined to exclude the maturity
phase of the product or business process life cycle, none of the three combinations involving this phase are relevant to
our analysis space. In addition, failure during launch cannot be caused by something that happened during the growth
phase, so that combination is also not relevant to our analysis. This leaves 11 relevant combinations of failure timing and
failure root cause timing. Any of the five innovation failure root causes can apply to any timing scenario, for a total of 55
(11 x 5) possible combinations of failure root cause, failure timing, and failure root cause timing. Of these possible
innovation failure narratives, only 9 were represented in the 19 project case studies. These 9 narratives are presented in
figure 2.

National Center for Science and Engineering Statistics | NCSES 21-201
13
FIGURE 2
Possible unsuccessful innovation and innovation failure narratives
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.
The slight majority (13 out of 19, or 68%) of cases examined failed during launch and the root cause(s) frequently
occurred in the product or business process development phase (figure 3). However, some innovations made it beyond
the launch stage before failing (32%), with 21% failing in the growth stage and 11% in the maturity stage. Overall, the most
common failure narrative was the failure of an innovation during launch due to the lack of market demand (37%) (figure
4).
2
While the root causes of innovation failure overwhelmingly occurred during product development, innovations in the case
group of 19 also failed because of factors arising during ideation or during the launch or growth phases. Figure 4
examines the relationship between when a product failed and the root cause of its failure. “No market demand” (42%) and
“poor performance” (32%) account for almost three-fourths of the cases examined, with these categories of failure
occurring mostly during the launch and growth stages.

National Center for Science and Engineering Statistics | NCSES 21-201
14
FIGURE 3
Heat map of common unsuccessful innovation and innovation failure stages
na = not applicable.
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.
FIGURE 4
Heat map of unsuccessful innovation and common innovation failures
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.
The most common failure narrative centers on firms whose new products fail at launch due to lack of demand (see top
row of figure 2). This, in many ways, is the archetype of the new technology failure story. Companies become enamored
by their technology and push to develop a product without adequately investigating whether the marketplace would have
any interest in it. Google Glass is an example here. The product was an impressive piece of new technology with many
novel capabilities but had virtually no demand from the general population, who had no interest in using Glass as a mobile
phone replacement or even complement.

National Center for Science and Engineering Statistics | NCSES 21-201
15
Following closely behind this narrative in frequency is one in which shortcomings during the development phase led to
failure at launch because new innovations underperform. Laundroid, for example, was a $16,000 device for folding
laundry. It was physically enormous (about the size of an extra-wide kitchen refrigerator) and often failed to fold clothes,
getting stuck on common garment types like dark T-shirts. In principle, many consumers would be very interested in a
machine that effectively folded clothes, but the Laundroid did not deliver on this value proposition. Taken together, lack of
market demand and poor performance accounted for three-fourths of the failures we analyzed. Insufficient
complementary assets accounted for another 16%. By far, the root cause for innovation failure most often occurred in the
development phase, which accounted for 79% of all failures.
The results of these case studies raise the question of why so many firms bring innovations to market when fundamental
commercial success factors, such as market demand or the availability of complementary assets, are unverified. Such
shortsightedness is not unique to small companies or startups; several of the cases examined here came out of large
market leaders, such as Google and Apple. Consideration of the reasons that firms do or do not continue innovation
activities despite evidence that they may not result in commercially successful innovations has implications for the
structure and interpretation of surveys of firm innovation practices, detailed in the following section.
Implications for Firm Innovation Surveys
The Annual Business Survey (ABS), conducted by the Census Bureau in partnership with NCSES, collects data on research
and development (R&D), innovation, technology, intellectual property (IP), and business owner characteristics. The ABS
includes questions related to methods (activities) used by firms for innovation, barriers to innovation, and expectations of
innovation. This research will be used to inform future iterations of the ABS.
Regarding innovation activities, the ABS asks firms if they have engaged in any of the following:
Item b, engineering and design activities, includes “planning of technical specifications, testing, evaluation, setup and pre-
production for goods, services, processes or systems; installing equipment, tooling-up, testing, trials and user
demonstrations; and activities to extract knowledge or design information from existing products or process equipment.”
Engineering and design activities also “include activities to develop a new or modified function, form, or appearance for
goods, services, or processes.” Item c, marketing and brand equity activities, includes “market research, market testing,
methods for pricing, product placement and product promotion; product advertising, the promotion of products at trade
fairs or exhibitions, and the development of marketing strategies.”
The innovation failure case studies included in this analysis indicate lack of market demand as the single most common
root cause of failure. The activities underlined above (emphasis added) are those focused on estimating the potential
market demand of an innovation. The underlined activities are currently included alongside many other engineering and
marketing activities that are unrelated to measuring market demand. Given how important understanding market demand
is to innovation success, it may be worth paying specific attention to these market-measuring activities in future cycles of
the ABS.
a.
b.
c.
d.
e.
f.
g.
h.
R&D
Engineering and design activities
Marketing and brand equity activities
IP–related activities
Employee training
Software development and database activities
Acquisition of machinery, equipment, and other tangible assets
Management related to innovation
National Center for Science and Engineering Statistics | NCSES 21-201
16
The ABS asks how important the following factors are in discouraging innovation activities:
The 19 case studies of innovation failure point to firms continuing innovation activities despite what hindsight suggests
was ample reason to believe new innovations would not be commercially successful. Although the case studies do not
explicitly address firms’ decisions to cease innovation activity, firms often make the decision to not continue with projects
because they acquire information during the development phase (pre-launch innovation activities phase) that indicates
planned innovations likely will not succeed. They may, for example, conduct market research and customer testing as part
of their development process and determine that there is too little market demand for their anticipated new product, or
that the value proposition of competitive products will be markedly superior to that of their own. In such cases, it makes
sense for firms to cease innovation activities related to the product or business process in question.
Ceasing innovation activities for a given project because data acquired during development indicate that the innovation
has low odds of commercial success is qualitatively different from ending (or failing to start) innovation activities
because of exogenous barriers, such as lack of funds or lack of skills. The list of innovation-interfering factors above does
not explicitly make this distinction. In future surveys, it may be beneficial to ask firms separately about (a) exogenous
barriers to innovation (e.g., lack of funds, lack of skills, lack of collaboration partners), and (b) their informed decisions to
discontinue innovation activities because those activities suggest low odds for commercial success. Another ABS
question asks firms if they have abandoned innovation activities within the survey reporting period. Future cycles of the
ABS may separately assess whether firms have abandoned innovation activities due to exogenous barriers or because of
internal evaluations of success likelihood.
Finally, the ABS asks firm respondents whether new or improved goods or services introduced during the reporting period
met their business’s expectations, such as market share, sales, or profits. Options for response include:
Where expectations have not been met, future cycles of the ABS may ask firms why and align the answers to root causes
for innovation such as those identified in the case studies.
a.
b.
c.
d.
e.
f.
g.
h.
i.
j.
k.
Lack of internal finance for innovation
Lack of credit or private equity
Difficulties in obtaining public grants or subsidies
Costs too high
Lack of skilled employees within this business
Lack of collaboration partners
Lack of access to external knowledge
Uncertain market demand for your ideas
Too much competition in your market
Different priorities within this business
Government regulations
Yes, expectations were exceeded
Yes, expectations were met
No, expectations were not met
Too early to tell
●
●
●
●

National Center for Science and Engineering Statistics | NCSES 21-201
17
Future Research
The current research included 19 cases of innovation failure from the 21st century. Obvious areas of expansion include
increasing the number of cases and examining the historical record of innovation failure. Expanding the number of cases
will allow for verification and expansion of the current and preliminary five categories of innovation failure and possibly for
creation of a category hierarchy in which main categories of failure are subdivided into variants or subcategories.
Figure 5 illustrates example subcategories for the “no market demand” failure root cause. Although Juicero, Sedasys, and
Google Glass failed due to a lack of market demand, there were significant differences across these innovations with
respect to how demand played into their failure. In the case of Juicero, once the product’s value proposition became clear
to the public, its demand disappeared completely. Consumers were unwilling to spend $400 for a machine to squeeze
juice from a bag. Genuine demand existed for Sedasys—replacing expensive anesthesiologists with a machine is a
compelling proposition. Its narrow use case made it uneconomical, however, and so demand could not be sustained.
Finally, Google Glass found no demand among its original consumer market, but its capabilities allowed for a pivot into
new uses. Similar subcategorization is likely with other failure categories and would help with understanding the nature,
patterns, and causes of innovation failure.
FIGURE 5
Possible unsuccessful innovation and innovation failure narratives
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.
Examining historical cases of innovation failure would expand the sample and also help identify any trends in the causes
or patterns of innovation failure over time. In addition to exploring whether the raw root causes for failure have shifted
over time, it would also be interesting to know if there have been other changes to the patterns of innovation failure. For
example, has the length of time from market introduction (launch) to when an innovation is removed from the market
changed? It may have shortened as firms embrace the virtues of failing fast. On the other hand, anecdotal arguments
indicate that many startups spend their venture capital even after their innovations seem destined to fail. More cases, and
more cases over a longer period, would help answer such questions.

National Center for Science and Engineering Statistics | NCSES 21-201
18
Conclusion
Nineteen case studies of innovation failure indicate that if innovations are to fail, they are most likely to do so during their
launch phase. Innovations fail for a variety of reasons, but three-fourths of the cases examined failed either because of
insufficient demand in the marketplace or because the innovations failed to function well enough. Coincidently, though
not surprisingly, more than three-fourths of the 19 failures can be traced to shortcomings in the new product development
process. These results are preliminary and reflect analysis of only 19 cases of innovation failure. Refinement of these
findings requires additional cases and inclusion of cases of innovation failure prior to the year 2000. In addition, this
research will be used to inform future iterations of the ABS, which contains a module on innovation including questions on
methods (activities) used by firms for innovation, barriers to innovation, and expectations of innovation.

National Center for Science and Engineering Statistics | NCSES 21-201
19
Notes
1 The Oslo Manual defines an innovation as being introduced in one market (consumer). When the product is introduced
in another market (business), that is considered another innovation. Google Glass did not succeed as intended in the
consumer market.
2 Whenever using percentages, these figures apply only to the population of the 19 case studies and are not
generalizable to the larger population.

National Center for Science and Engineering Statistics | NCSES 21-201
20
References
Barry K. 2018. Robots Will Soon Be Folding Your Laundry—But Will They Really? Available at https://www.reviewed.com/
laundry/news/no-a-robot-wont-be-folding-your-laundry-any-time-soon. Accessed 31 March 2021.
Dashevsky E, Hachman M. 2014. 16 Cool Things You Can Do with Google Glass. Available at https://www.pcmag.com/
news/16-cool-things-you-can-do-with-google-glass. Accessed 31 March 2021.
Ernst H. 2017. Success Factors of New Product Development: A Review of the Empirical Literature. International Journal
of Management Reviews, 4(1).
Florén H, Frishammar J, Parida V, Wincent J. 2018. Critical Success Factors in Early New Product Development: a Review
and a Conceptual Model. International Entrepreneurial Management Journal, 14:411-427.
Frankel TC. 2015. New Machine Could One Day Replace Anesthesiologists. The Washington Post May 11. Available at
https://www.washingtonpost.com/business/economy/new-machine-could-one-day-replace-anesthesiologists/
2015/05/11/92e8a42c-f424-11e4-b2f3-af5479e6bbdd_story.html. Accessed 31 March 2021.
Huet E, Zaleski O. 2017. Silicon Valley’s $400 Juicer May Be Feeling the Squeeze. Available at https://
www.bloomberg.com/news/features/2017-04-19/silicon-valley-s-400-juicer-may-be-feeling-the-squeeze. Accessed 31
March 2021.
Levin S. 2017. Squeezed out: Widely Mocked Startup Juicero Is Shutting down. The Guardian September 1. Available at
https://www.theguardian.com/technology/2017/sep/01/juicero-silicon-valley-shutting-down. Accessed 31 March 2021.
McDermott S. 2019. Laundry-Folding Robot Laundroid Folds under $20M Worth of Pressure. Available at https://
www.cnet.com/news/laundry-folding-robot-laundroid-folds-under-20m-worth-of-pressure/. Accessed 31 March 2021.
OECD/Eurostat. 2018. Oslo Manual 2018: Guidelines for Collecting and Interpreting Innovation Data, 4th ed. Paris: OECD
Publishing/Luxembourg: Eurostat. Available at: https://doi.org/10.1787/9789264304604-en. Accessed 31 March 2021.
Sacco A. 2016. Google Glass Takes Flight at Boeing. Available at https://www.cio.com/article/3095132/google-glass-
takes-flight-at-boeing.html. Accessed 31 March 2021.
Salkin A. 2017. Take a Load Off. The Robots That Fold Laundry Are Coming. New York Times May 17. Available at https://
www.nytimes.com/2017/05/24/business/entrepreneurship-laundroid-self-folding-laundry-machine.html. Accessed 31
March 2021.
Summers N. 2019. Laundroid Company Folds before Its Giant Robot Does. Available at https://www.engadget.com/
2019-04-23-laundroid-robot-seven-dreamers-bankruptcy.html. Accessed 31 March 2021.
Thompson D. 2017. How Juicero's Story Set the Company Up for Humiliation. The Atlantic April 21. Available at https://
www.theatlantic.com/business/archive/2017/04/juicero-lessons/523896/. Accessed 31 March 2021.
Tobe F. 2013. SEDASYS Robotic Anesthesiologist Approved by FDA. Available at https://www.therobotreport.com/
sedasys-a-robotic-anesthesiologist/. Accessed 31 March 2021.
Weider JB. 2020. How and Why Google Glass Failed. Available at https://www.investopedia.com/articles/investing/
052115/how-why-google-glass-failed.asp. Accessed 31 March 2021.

National Center for Science and Engineering Statistics | NCSES 21-201
21
Suggested Citation
Benskin J, Jankowski J, Kindlon A, Randazzese L, Sullivan J; National Center for Science and Engineering Statistics. 2021.
Understanding Unsuccessful Innovation. NCSES 21-201. Alexandria, VA: National Science Foundation. Available at https://
www.nsf.gov/statistics/2021/ncses21201/.

National Center for Science and Engineering Statistics | NCSES 21-201
22
Contact Us
Authors
John Benskin
Project Leader, Center for Innovation Strategy and Policy
SRI International, under contract to NCSES
John E. Jankowski
Program Director
Research and Development Statistics Program, NCSES
E-mail: [email protected]
Tel: (703) 292-7781
Audrey E. Kindlon
Survey Manager
Research and Development Statistics Program, NCSES
E-mail: [email protected]
Tel: (703) 292-2332
Lucien Randazzese
Director, Center for Innovation Strategy and Policy
SRI International, under contract to NCSES
J.R. Sullivan
Senior Research Analyst, Center for Innovation Strategy and Policy
SRI International, under contract to NCSES
NCSES
National Center for Science and Engineering Statistics
Directorate for Social, Behavioral and Economic Sciences
National Science Foundation
2415 Eisenhower Avenue, Suite W14200
Alexandria, VA 22314
Tel: (703) 292-8780
FIRS: (800) 877-8339

National Center for Science and Engineering Statistics | NCSES 21-201
23
Appendix: Case Studies
Table Title
A-1 Case study definitions
A-2 Case study: Google Glass
A-3 Case study: Microsoft Windows Vista
A-4 Case study: Samsung Galaxy Note 7
A-5 Case study: iTunes Ping
A-6 Case study: DVD-Audio
A-7 Case study: Hoverboards
A-8 Case study: Boeing 737 Max
A-9 Case study: Zozosuit
A-10 Case study: Juicero
A-11 Case study: Google+
A-12 Case study: Ubuntu Touch
A-13 Case study: Sedasys
A-14 Case study: Google Wallet
A-15 Case study: Segway
A-16 Case study: Iridium Satellite Phone
A-17 Case study: 3D Television
A-18 Case study: Wii U
A-19 Case study: Laundroid
A-20 Case study: Sony BMG Extended Copy Protection (XCP)

National Center for Science and Engineering Statistics | NCSES 21-201
24
TABLE A-1
Case study definitions
(Definitions)
Innovation Brief description of the innovation
Radical or incremental
Radical innovations shape big changes in the world. Incremental innovations fill in the process of change
continuously.
Category Product or business process
Sector Industry
Failure timing Product life cycle period during which the innovation failed: launch, growth, or maturity
Failure root cause
Reasons for the failure: no market demand, poor performance, insufficient complementary assets, poorly defendable
position, or regulatory restriction
Failure root cause timing This can occur before or after product launch. It is the phase at which the root cause of innovation failure took place.
Outcomes Results of the innovation
Business insight into the
innovation process
Lessons learned and impact on the company
Pivot Steps taken by the company following the innovation's initial failure
Pivot enabler Reasons company was able to pivot the innovation
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
25
TABLE A-2
Case study: Google Glass
(Innovation failure summary)
Variable Description
Innovation Head-mounted wearable mobile device, launched 15 April 2013
Radical or incremental Radical
Category Product
Sector Mobile electronics and services
Failure timing Launch stage, failure in January 2015
Failure root cause
No market demand
• Produced offered relatively little significant new functionality for users
• Developers did not adequately (or at all) consider privacy concerns, security concerns, or social awkwardness
Failure root cause timing Product development
Outcomes
• Low consumer penetration
• Widespread derision for product with (short-lived) affect on Google's reputation as innovator
Business insight into the
innovation process
• Insufficient customer perspective in the ideation state of innovation
• Earlier customer perspective may have led to earlier pivot in use case
Pivot • Yes, in commercial, health, and science applications
Pivot enabler
• Product functionality, specifically the ability to retrieve data on and analyze an object or person while looking
at the object or person
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
26
TABLE A-3
Case study: Microsoft Windows Vista
(Innovation failure summary)
Variable Description
Innovation Windows operating system, launched 30 January 2007
Radical or incremental Incremental
Category Product
Sector Software
Failure timing Launch stage, failure in 2007
Failure root cause
Poor performance
• Drift in top-level product objective led to long delay in launch, resulting in a product ill-suited to the evolving
technology environment into which it was launched
• Project management failure led to poorly coordinated feature plan, leading to features that worked poorly or were
not well received
Failure root cause timing Product development
Outcomes
• Despite some professional praise for new features, Windows Vista was a commercial flop
• Public relations debacle
• Many consumers demanded new machines "downgraded" to Windows XP
• Did not dent and may have increased Apple market share
Business insight into the
innovation process
• Need to use strategic planning to focus on what problems are to be solved
• Project management must ensure product feature development matched to market conditions
Pivot na
Pivot enabler na
na = not applicable.
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
27
TABLE A-4
Case study: Samsung Galaxy Note 7
(Innovation failure summary)
Variable Description
Innovation "Phablet"-style mobile phone, launched 19 August 2016
Radical or incremental Incremental
Category Product
Sector Mobile electronics and services
Failure timing Launch stage, failure in October 2016
Failure root cause
Poor performance
• Rushed too many new features
• Flawed design
• Poor testing and quality control
Failure root cause timing Product development
Outcomes
• Fires and explosions
• U.S. wireless carriers stopped selling device; devices banned on aircraft
• Public lost confidence in Samsung quality control
• Product-specific financial losses
Business insight into the innovation process • Improve testing—Samsung instituted eight-point battery testing
Pivot na (2017 released Fan Edition)
Pivot enabler na
na = not applicable.
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
28
TABLE A-5
Case study: iTunes Ping
(Innovation failure summary)
Variable Description
Innovation Music-oriented social networking system, launched 1 September 2010
Radical or incremental Incremental
Category Service
Sector Media
Failure timing Launch stage, failure on 30 September 2012
Failure root cause
Poor performance
• Product launched without agreement in place to access critical component (Facebook)
Failure root cause timing Product launch—Apple launched and accessed Facebook without data-sharing agreement in place
Outcomes
• Usage remained very low
• Unsuccessful as both social network and driver for music sales through iTunes
Business insight into the innovation process • Do not launch without critical component
Pivot na (Some elements ended up in Apple Music, launched in 2015)
Pivot enabler na (Some elements ended up in Apple Music, launched in 2015)
na = not applicable.
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
29
TABLE A-6
Case study: DVD-Audio
(Innovation failure summary)
Variable Description
Innovation
Digital audio format for use with DVD media, developed by the DVD Forum (led by Sony and other electronics and media
companies), launched in summer 2000
Radical or incremental Incremental
Category Product
Sector Consumer electronics
Failure timing
Launch stage, failure within first 12 months (continued to be produced until 2007 despite virtually zero market
penetration)
Failure root cause
No market demand
• Developers did not test their assumption that enhanced sampling rates and bit depth (bits-per-sample) would translate
to better listener experience
• Developers did not foresee changing consumer preference for mobility in music listening (Apple iPod released 2001)
Failure root cause
timing
Product development
Outcomes
• Format never caught on with consumers and is now virtually dead
• As of June 2019, Amazon listed only 155 audio titles in DVD-Audio format
Business insight into
the innovation process
• Consumer demand for new product features always needs to be tested
• Better consumer testing during the development phase would likely have revealed lack of demand
Pivot na
Pivot enabler na
na = not applicable.
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.
National Center for Science and Engineering Statistics | NCSES 21-201
30
TABLE A-7
Case study: Hoverboards
(Innovation failure summary)
Variable Description
Innovation Simplified version of Segway that was easier to transport and cheaper to buy, launched in May 2013
Radical or incremental Radical
Category Product
Sector Transportation
Failure timing Maturity stage, failure in June 2016
Failure root cause
Poorly defendable position
• Difficulty in identifying and enforcing relevant patents, leading to market saturation with unlicensed knockoffs
• Copycats used subpar production and safety standards, leading to reports of severe injuries and fires and,
ultimately, the product's banning
Failure root cause timing Growth
Outcomes
• Hoverboards were banned on all major airlines
• Most large retailers, including Amazon, Walmart, and Target, pulled hoverboards from their physical and online
stores
• CPSC introduced new safety standards that effectively banned most hoverboards from being sold in the United
States
• Give market conditions, it was no longer economically viable for the original producer to produce a CPSC-compliant
product
Business insight into the
innovation process
na
Pivot na
Pivot enabler na
na = not applicable.
CPSC = Consumer Product Safety Commission.
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
31
TABLE A-8
Case study: Boeing 737 Max
(Innovation failure summary)
Variable Description
Innovation New iteration of popular airliner to boost efficiency, launched 29 January 2016
Radical or incremental Incremental
Category Product
Sector Transportation
Failure timing Maturity stage, failure in March 2019
Failure root cause
Poor performance
• A new software system was not adequately described to pilots and was given a significant amount of behind-the-
scenes power that overrode pilot commands
• Boeing claimed the new aircraft was functionally equivalent to prior models when, in reality, it had significant
performance differences
• Federal regulators overdevolved regulatory oversight power and authority to Boeing
Failure root cause timing Product development
Outcomes
• Two Boeing 737 Max jets crashed, the first in October 2018 and the second in March 2019, resulting in hundreds of
deaths
• As a result of these crashes, the jet was grounded in virtually all countries in which they operated
• Many airlines that rely on the Max had to cancel a significant number of flights through at least December 2019,
resulting in lost revenue
• Boeing has continued to produce Max jets but is unable to deliver them to customers; some airlines have cancelled
their future orders of the Max jet
Business insight into the
innovation process
• Need to ensure changes to existing systems are made apparent to system users
Pivot na
Pivot enabler na
na = not applicable.
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
32
TABLE A-9
Case study: Zozosuit
(Innovation failure summary)
Variable Description
Innovation
Sensor-covered bodysuit works with an app to uploaded customers body measurements to Zozo, allowing creation of
customized casual clothing, launched in 2017
Radical or
incremental
Radical
Category Process
Sector Clothing
Failure timing Growth stage, failure in 2019
Failure root cause
Insufficient complementary assets
• Production bottlenecks prevented Zozo from fulfilling large number of custom clothes orders in a timely fashion
• Company shipped non-customized off-the-rack items to customers, many of which were often ill-fitting
Failure root cause
timing
Product development
Outcomes
• Zozo overseas expansion, which relied on the Zozosuit, was largely a failure
• Zozo stock lost 20% in value March 2018–March 2019
• Some customers used suit only to keep track of their measurements and never bought any clothes
Business insight into
the innovation
process
• Changes to one part of the customer delivery process must be examined for their impact on other parts of the process
• "Perfect fit" is not as critical to casual clothes as to formalwear
• Enthusiasm about a free technology platform does not necessarily predict sales
Pivot
• Zozosuit repurposed as personal measurement system for custom formalwear (phased out for casualwear)
• New sizing database based on data collected by Zozosuit created for Japanese market to correlate small number of
body measurements (e.g., height and weight) with off-the-rack sizes for casual clothes
Pivot enabler
• Formalwear volume is lower than casualwear, enabling timely order fulfillment
• Formalwear is more expensive than casual wear, thus increasing profit margins per Zozosuit
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
33
TABLE A-10
Case study: Juicero
(Innovation failure summary)
Variable Description
Innovation
Juicero was a start-up that sold an expensive, Wi-Fi-connected home juicer that squeezed Juicero-branded produce packs
to make organic juices, launched in March 2016
Radical or
incremental
Radical
Category Product
Sector Household appliances
Failure timing Launch stage, failure in September 2017
Failure root cause
No market demand
• Bloomberg discovered consumers could squeeze the packets by hand and get most (7.5 ounces) of the juice out in about
1.5 minutes, without the use of the machine; the machine could get 8 ounces out in 2 minutes
• Juicero originally cost $700 (later reduced to $400), which was significantly more expensive than other juicing techniques
• Only Juicero-branded fruit packs ($5–$8 each) could be used with the machine, requiring consumers to purchase monthly
subscription packages to get continued use out of their machine, a transparent way to force consumer loyalty to Juicero
Failure root cause
timing
Product development
Outcomes
• High price point and knowledge of the uselessness of the machine caused demand to collapse
• After announcing they would no longer be selling the machine or fruit pouches, the company offered full refunds to
anyone who purchased a machine
• The official cause of closure was cited as an inability to create an “effective manufacturing and distribution system for a
nationwide customer base”
Business insight
into the innovation
process
• Innovation must address real market needs with compelling value proposition
Pivot na
Pivot enabler na
na = not applicable.
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
34
TABLE A-11
Case study: Google+
(Innovation failure summary)
Variable Description
Innovation
A social networking site developed by Google that integrated many of Google's other services, to be in competition with
Facebook and Twitter, launched 28 June 2011
Radical or
incremental
Incremental
Category Service
Sector Media
Failure timing Growth stage, failure in April 2019
Failure root cause
No market demand
• Developed as a result of a corporate boardroom decision to compete in the social network space, rather than in
response to an identified need or gap in existing social networking platforms
• In search of a marking focus, Google+ was marketed as a platform for engaging with strangers on a variety of topics/
interests; most people want to connect with their friends, not strangers
• Policy of signing up all Google account users of other Google services (such as Gmail) led to very large number of “ghost
accounts” that people had not requested and did not use
Failure root cause
timing
Product development
Outcomes
• Average and shorter user sessions (90% lasted less than 5 seconds; Facebook sessions last, on average, 20 minutes)
• The “ghost town” of inactive accounts encouraged more active users to engage with people they knew on established
networks, like Facebook or Twitter, leading to growth in those competing networks
• Google+ was shut down as a public social network and transferred to an enterprise-only suite of products
• Google spun off many Google+ features into other Google programs (e.g., Photos, Hangouts)
Business insight into
the innovation
process
• Forcing customers to use a project they do not want does not drive commercially meaningful use
Pivot
• Split the useful non-networking features of Google+ into Streams and Photos; Hangouts
• G+ Enterprise retains the core networking features for corporate use
Pivot enabler • Some adoption of Google+ into corporate environment
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
35
TABLE A-12
Case study: Ubuntu Touch
(Innovation failure summary)
Variable Description
Innovation
Developed as an alternative mobile OS to Android and iOS that would not lose support, especially security support, for older
versions on older phones, launched in October 2013
Radical or
incremental
Incremental
Category Product
Sector Mobile electronics and services
Failure timing Launch stage, failure in April 2017
Failure root cause
Poor performance
• Running Ubuntu on mobile devices required hardware specifications that most phones did not ship with, meaning Ubuntu
could not be installed
• Ubuntu was shipped predominantly on a relatively unknown hardware brand, BQ, at a non-subsidized cost; most
consumers prefer to stick with recognized brands (e.g., Apple and Samsung) that can be bought at subsidized costs
through carrier contracts
• Android and iOS had already captured the mobile market and provided consumers a wide offering of third-party apps to
add desired functionality
• Ubuntu phones shipped without the core features being ready, resulting in customers being exposed to a number of bugs
and without the ability to perform a number of basic tasks (e.g., running WhatsApp messaging service; making and
receiving phone calls in some cases)
Failure root cause
timing
Product development
Outcomes
• Sales of phones loaded with Ubuntu Phone OS remained low
• Development of an Ubuntu-based mobile OS was scrapped by Canonical Ltd., the company originally responsible for
pioneering Ubuntu software, and picked up by the UBPorts Foundation as an open source community project comprised of
volunteer developers
Business insight
into the innovation
process
• Addressing market need (older phones losing security updates) cannot be addressed at the cost of other needs (e.g.,
phone functionality)
Pivot • Ubuntu Touch became a niche mobile OS in predominantly European mobile markets
Pivot enabler
• The transfer of the Ubuntu mobile OS from Canonical to UBPorts enabled a larger community of volunteers to work on its
development and better support the narrow market for which it is not used
OS = operating system.
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.
National Center for Science and Engineering Statistics | NCSES 21-201
36
TABLE A-13
Case study: Sedasys
(Innovation failure summary)
Variable Description
Innovation
Machine that administers propofol anesthetic to patients undergoing certain procedures under the supervision of a nurse,
negating the need for more highly paid anesthesiologists, launched 13 October 2014
Radical or
incremental
Radical
Category Product
Sector Medical technology
Failure timing Launch stage, failure in March 2016
Failure root cause
No market demand
• Opposition from the American Society of Anesthesiologists claiming a machine would not be capable of exercising the
same level of care and diligence as a trained professional; limiting it to too few routine procedures (e.g., colonoscopies,
endoscopies)
• As a safety measure added to gain approval by regulators, the machine could only decrease the level of anesthetic—
increases in dosage required intervention of a clinician—further limiting the machine's operability
Failure root cause
timing
Product development
Outcomes
• As a result of the limitations of the machines, it was not financially beneficial to most hospitals
• The machine did not sell well, with only a handful of hospitals in the United States purchasing the equipment
• Johnson & Johnson stopped selling the product
Business insight
into the innovation
process
• Products that require formal or informal approval from expert groups or regulators should not be developed absent their
input or support
Pivot na
Pivot enabler na
na = not applicable.
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
37
TABLE A-14
Case study: Google Wallet
(Innovation failure summary)
Variable Description
Innovation
Google Wallet enabled consumers to load partner bank cards onto their phone, allowing them to pay at registers with their
phone rather than with a bank card, launched 26 May 2011
Radical or
incremental
Radical
Category Service
Sector Financial technology
Failure timing Growth stage, failure in February 2018
Failure root cause
Insufficient complementary assets
• Google failed to negotiate profitable agreements with card issuers, resulting in Google losing money on each Google
Wallet transaction
• Google failed to form partnerships with most banks because it wanted banks to share consumer spending habits and
data to develop targeted ads
• At the time of launch, most mobile devices and merchants did not have the NFC technology required to use Google
Wallet
• Security holes appeared shortly after launch, some of which made it possible for hackers to easily steal any balances
loaded on cards in Google Wallet
Failure root cause
timing
Product development
Outcomes
• Limited opportunities for use led to a suppressed user base of less than 10 million people
• Google shifted Google Wallet to Google Pay Send, allowing users to send money to other users, with plans to fully
integrate it within the Google Pay ecosystem
Business insight into
the innovation
process
• Work with those in the product's operating context to ensure there is an enabling environment for its successful
deployment
Pivot • P2P payment feature of Google Wallet integrated into Google Pay, which replaced Google Wallet and Android Pay
Pivot enabler • Functionality of the original app allowed for easy conversion to P2P payment capability
NFC = near-field communication; P2P = peer to peer.
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
38
TABLE A-15
Case study: Segway
(Innovation failure summary)
Variable Description
Innovation
A two-wheeled personal transportation device that used innovative technology to keep riders balanced and promised to
shorten travel time for certain trip types, launched in December 2001
Radical or
incremental
Radical
Category Product
Sector Transportation
Failure timing Launch stage, failure in 2001
Failure root cause
No market demand
• More expensive than a used car in fair condition (~$5,000)
• Lack of supporting environment, especially regarding urban infrastructure (Where to park? Charge? Use?)
• It was banned in some countries because it was too fast for sidewalks but too slow for roadways
Failure root cause
timing
Product development
Outcomes
• It was projected that 10,000 units would sell per week after launch; after 6 years, only 30,000 units had sold, less than 1%
of projection
Business insight into
the innovation
process
• “Cool” technology alone does not lead to market acceptance; products need to address consumer needs
• Products that make use of public infrastructure (e.g., transportation equipment) cannot be developed without attention
to how it works with the enabling environment
Pivot
• Limited enterprise demand for police departments, tour groups, warehouse workers, mall security guards, airport
maintenance staff
Pivot enabler
• Enterprises have constrained environments that enable the deployment of supporting infrastructure on a smaller, more
achievable scale (e.g., in a warehouse or a shopping mall)
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
39
TABLE A-16
Case study: Iridium Satellite Phone
(Innovation failure summary)
Variable Description
Innovation Iridium satellite phone, launched 1 November 1998
Radical or incremental Radical
Category Product
Sector Mobile electronics and services
Failure timing Launch stage, failure in August 1999
Failure root cause
No market demand
• Development began in 1991 when terrestrial cellular networks were limited
• The subsequent spread of terrestrial cellular networks would limit the appeal of satellite phones to remote areas
• Vast majority of traffic is concentrated in areas where cellular networks were rapidly penetrating
• Company leadership did not reevaluate customer acquisition targets in light of growing cellular coverage
• Equipment ($3,000) and service ($3–$8 per minute) were very expensive, based on assumption of monopoly and
required financial returns
Failure root cause timing Product development
Outcomes
• Dramatically low customer adoption vs. forecasts and targets required by loan covenants
• Company filed for bankruptcy 9 months after launch (August 1999)
• Motorola announced that the Iridium satellites would have to be de-orbited in August 2000
• Government agreed to 2-year contact for services and $72 million in funding, and approved sale of assets to
Iridium Communications Inc. for $25 million
• Original investors who paid for $5 billion plus investment to launch 66 geostationary satellites received less than
1% of their investment back
Business insight into the
innovation process
• Projects with long development cycles need to reevaluate the technology value proposition as new potential
competition emerges
Pivot
• Iridium Communications Inc. operates global satellite communications network mostly for government and
commercial applications
Pivot enabler
• Paying one-half of 1 cent on the dollar for network assets
• Initial government contract guarantee
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
40
TABLE A-17
Case study: 3D Television
(Innovation failure summary)
Variable Description
Innovation
Home televisions equipped with 3D technology, enabling people to engage in 3D television entertainment in their homes,
launched in March 2010
Radical or
incremental
Incremental
Category Product
Sector Consumer electronics
Failure timing Launch stage, failure in early 2017
Failure root cause
No market demand
• Special glasses (some up to $100) had to be worn to view the 3D effect, and there were competing standards for which
glasses had to be used for which TVs; glasses were not interchangeable between different TV brands
• To view movies and cable/ satellite in 3D, consumers had to buy 3D-enabled Blu-ray players and cable/ satellite boxes
• Limited content available for at-home 3D consumption (not many movies were sold in 3D DVD format)
Failure root cause
timing
Product development
Outcomes
• Television makers slowly phased out the 3D option on TV models, beginning in 2013 with Vizio and ending in 2017 with
LG and Sony
Business insight into
the innovation
process
• Make better use of the consumer adoption history of similar products (e.g., the limited success of 3D movies)
Pivot na
Pivot enabler na
na = not applicable.
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
41
TABLE A-18
Case study: Wii U
(Innovation failure summary)
Variable Description
Innovation
A redevelopment of Nintendo's landmark Wii gaming system, including a new GamePad tablet that allowed players to play in
the same game but on different screens, launched 18 November 2012
Radical or
incremental
Incremental
Category Product
Sector Consumer electronics
Failure timing Growth stage, failure in November 2017
Failure root cause
Insufficient complementary assets
• The GamePad made it difficult for game publishers to develop content that could be played on the Wii U as well as other
consoles, so most publishers either refrained from making Wii U games or they made Wii U games that did not make use of
the GamePad
• Weak consumer demand disincentivized publishers from developing Wii U content, leading to vicious cycle
• The GamePad was the signature component of the Wii U, but it was heavy and difficult to use and had to be very close to
the console to work
Failure root cause
timing
Product development
Outcomes
• Wii U production was halted after only 4 years on the market, which is a relatively short life span for a game console (e.g.,
Microsoft's Xbox One was released in 2013 and continues to be the company's flagship gaming device; the original Wii was
launched in November 2006 and continued production until late 2012)
Business insight
into the innovation
process
• New technological approaches need to provide value proposition improvement over existing products and incentivize
consumers to purchase the newer product
Pivot
• The core concept of the GamePad (e.g., a larger handheld screen than most portable gaming device) laid the foundation for
the launch of the Nintendo Switch, which has been immensely successful
Pivot enabler • Nintendo used the experience to understand what users wanted from GamePad-like devices
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.

National Center for Science and Engineering Statistics | NCSES 21-201
42
TABLE A-19
Case study: Laundroid
(Innovation failure summary)
Variable Description
Innovation Robotic AI-driven wardrobe that folds clothes, launched in 2018
Radical or incremental Radical
Category Product
Sector Household appliances
Failure timing Launch stage, failure in 2019
Failure root cause
Poor performance
• Very large
• Unrealistically high price point
Failure root cause timing Product development
Outcomes
• Virtually zero sales
• Seven Dreamers bankrupt in 2019 (company believed to have taken on $20 million or more in debt while trying
to bring product to market)
Business insight into the
innovation process
• Even if “time is right” for a new technology, performance must be acceptable and price must be aligned with
value
Pivot na
Pivot enabler na
na = not applicable.
AI = artificial intelligence.
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.
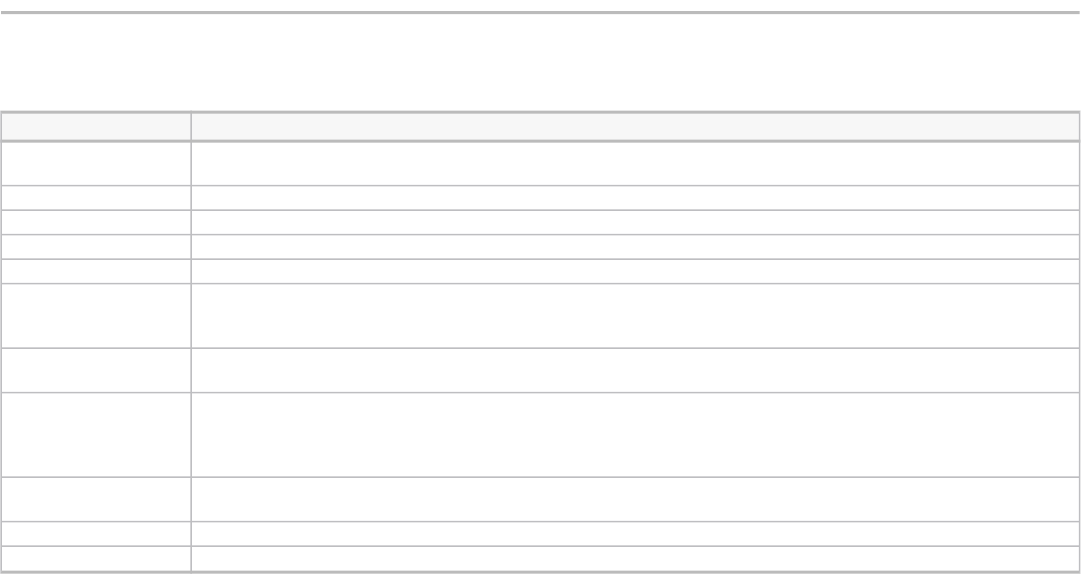
National Center for Science and Engineering Statistics | NCSES 21-201
43
TABLE A-20
Case study: Sony BMG Extended Copy Protection (XCP)
(Innovation failure summary)
Variable Description
Innovation
Software copy protection measures added to CDs as a form of DRM. XCP modified users' computers to interfere with
CD copying, launched in 2005
Radical or incremental Radical
Category Process
Sector Consumer electronics
Failure timing Launch stage, failure in 2005
Failure root cause
Regulatory restrictions
• Software functioned as rootkit
• Implementation was deceptive, illegal, and harmful
Failure root cause
timing
Ideation
Outcomes
• Widespread public outcry
• Sony BMG sued by Texas, New York, and California under spyware laws
• 2005 recall of affected CDs
• Sony BMG was required to compensate consumers who had been negatively impacted
Business insight into
the innovation process
• Avoid introducing new technology into products in areas outside of core expertise, especially without understanding
the technical and legal ramifications
Pivot na
Pivot enabler na
na = not applicable.
DRM = digital rights management.
Source(s):
National Center for Science and Engineering Statistics and SRI International, special research (2020) of 2010–20 open-access articles, including
MIT Technology Review, New York Times, Fast Company, U.S. General Accountability Office, and Defense News.


