
FORMS VERSION F SERIES
Released:October 16, 2020
FELLOWSHIP INSTRUCTIONS FOR NIH
AND OTHER PHS AGENCIES
SF424 (R&R) APPLICATION PACKAGES
Guidance developed and maintained by NIH for preparing and
submitting applications via Grants.gov to NIH and other PHS
agencies using the SF424 (R&R)

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
TABLE OF CONTENTS
TABLE OF CONTENTS
TABLE OF CONTENTS 2
F.100 - How to Use the Application Instructions 3
F.110 - Application Process 6
F.120 - Significant Changes 10
F.130 - Program Overview 16
F.200 - SF 424 (R&R) Form 18
F.220 - R&R Other Project Information Form 33
F.230 - Project/Performance Site Location(s) Form 43
F.240 - R&RSenior/Key Person Profile (Expanded) Form 48
F.430 - PHS Fellowship Supplemental Form 60
F.500 - PHS Human Subjects and Clinical Trials Information 84
F.600 - PHS Assignment Request Form 121
Form Screenshots i
F - 2

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.100 - How to Use the Application Instructions
F.100 - How to Use the Application
Instructions
Use these application instructions to fill out the forms that are posted in your funding
opportunity announcement.
View the How to Apply Video Tutorials.
Quick Links
Step 1. Become familiar with the application process
Step 2. Use these instructions, together with the forms and information in the funding opportunity
announcement, to complete your application
Step 3. Choose an application instruction format
Step 4. Complete the appropriate forms
Step 5. Stay informed of policy changes and updates
Step 6. Understand what data NIH makes public
Helpful Links
The information on the following pages may be useful in the application process
l OER Glossary
l Grants Policy Statement
l Guide to Grants and Contracts
l Frequently Asked Questions
Step 1. Become familiar with the application process.
Understanding the application process is critical to successfully submitting your application.
Use the F.110 - Application Process section of these instructions to learn the importance of
completing required registrations before submission, how to submit and track your application,
where to find page limits and formatting requirements, and more information about the
application process.
F - 3

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.100 - How to Use the Application Instructions
Step 2. Use these instructions, together with the forms and information
found in the funding opportunity announcement, to complete your
application.
The funding opportunity announcement (FOA)will include specific instructions and the forms
needed for your application submission.
Remember that the FOA instructions always supersede these application instructions.
Step 3. Choose an application instruction format.
Do you know your activity code, but don’t know which application instructions to use? Refer to
NIH’s table on Selecting the Correct Application Instructions to determine which set of
application instructions applies to your grant program.
Comprehensive Instructions Program-Specific Instructions
Use the General (G) instructions, available in
both
HTML
and
PDF
format, to complete
the application forms for any type of grant
program.
Take advantage of the filtered PDFs to view
specific application instructions for:
l Research (R)
l Career Development (K)
l Training (T)
l Fellowship (F)
l Multi-project (M)
l SBIR/STTR (B)
Step 4. Complete the appropriate forms.
Unless otherwise specified in the FOA, follow the standard instruction, as well as any additional
program-specific instructions for each form in your application.
Program-specific instructions are presented in gray call-out boxes that are color coded
throughout the application instructions. Consult the F.130 - Program Overview section for
context for program specific instructions.
Step 5. Stay informed of policy changes and updates.
l Refer to the F.120 - Significant Changes section for the most recent changes to these
application instructions.
l Review Notices of NIH Policy Changes since the posting of the Application Guide.
F - 4

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.100 - How to Use the Application Instructions
Step 6. Understand what data NIH makes public.
Information submitted as part of the application will be used by reviewers to evaluate the
scientific merit of the application and by NIH staff to make the grant award and monitor the
grant after award. The exception to this is the F.600 - PHS Assignment Request Form, which is
only seen by staff in the Division of Receipt and Referral (DRR), Center for Scientific Review (CSR).
If the application is funded, the following fields will be made available to the public through the
NIH Research Portfolio Online Reporting Tool (RePORTER) and will become public information:
l Name of Project Director/Principal Investigator (PD/PI), to also include Project Leaders on sub-
projects to multi-project projects
l PD/PI title
l PD/PI email address
l Organizational name
l Institutional address
l Project summary/abstract
l Public health relevance statement
In addition, key elements related to ongoing funded projects will be made available to the public,
including those listed in the data dictionary at ExPORTER. Additional elements may be made
available after announcements through the NIH Guide for Grants and Contracts, a weekly
electronic publication that is available on NIH’s Funding page, or additions to the NIH Grants
Policy Statement, as needed.
F - 5

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.110 - Application Process
F.110 - Application Process
Understanding the application process is critical to successfully submitting your application.
Use this section of this guide to learn the importance of completing required registrations
before submission; how to submit and track your application; where to find information
about page limits, formatting requirements, due dates, and submission policies; and more
information about the application process. This application process information is also
available on our How to Apply – Application Guide page.
Quick Links
Prepare to Apply and Register
Write Application
Submit
Related Resources
Prepare to Apply and Register
Systems and Roles
Learn about the main systems involved in application submission and the role you and your
colleagues play in the submission process. The main systems are Grants.gov, eRA Commons, and
ASSIST.
Register
Determine your registration status. Organizations, organizational representatives, investigators,
and others need to register in multiple federal systems in order to for you to submit a grant
application. Registration can take six weeks or more to complete. Start today! See NIH’s
Registration website.
Understand Funding Opportunities
Identify the right funding opportunity announcement (FOA) for your research and learn about
key information you will find in the FOA.
Types of Applications
Are you submitting a new, renewal, revision, or resubmission application? Learn about the
different types of applications and special submission requirements.
Submission Options
Determine which system is most convenient for your application submission: NIH’s ASSIST web-
based application submission system, Grants.gov Workspace, or, if applicable, your
organization’s own submission system.
F - 6

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.110 - Application Process
Obtain Software
Applicants must have the free Adobe Reader software, a PDF generator, and a web browser to
submit an application. Learn which versions are compatible with our systems.
Write Application
Write Your Application
Read tips for developing a strong application that helps reviewers evaluate its science and merit.
Develop Your Budget
Learn about the kinds of costs you may include in your budget submission, the difference
between modular and detailed budgets, and more about how to develop your budget.
Format Attachments
Follow these requirements for preparing the documents you attach to your application.
Requirements include criteria for the PDF files, fonts, margins, headers and footers, paper size,
citations, formatting pages, etc.
Rules for Text Fields
Learn the rules for form text fields – allowable characters, cutting and pasting, character limits,
and formatting.
Page Limits
Follow the page limits specified in this table for your specific grant program, unless otherwise
specified in the FOA.
Data Tables
Find instructions, blank data tables, and samples to use with institutional research training
applications.
Reference Letters
Some types of programs, such as fellowships and some career development awards, require the
submission of reference letters by the referee. Learn about selecting a referee and find
instructions for submission.
Biosketches
Biosketches are required in both competing applications and progress reports. Find instructions,
blank format pages, and sample biosketches.
Submit
Submit, Track and View
Learn how to submit your application, and about your responsibility for tracking your application
and viewing the application image in the eRA Commons before the application deadline. If you
can’t view your application in eRA Commons, we can’t review it.
F - 7

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.110 - Application Process
How We Check for Completeness
Your application will be checked at Grants.gov, by eRA systems, and by federal staff before it is
referred for review.
Changed/Corrected Applications
You will need to submit a changed/corrected application to correct issues that either you or our
systems find with your application. Learn how and when you may submit a changed/corrected
application.
Related Resources
Due Dates and Policies
Due Dates
View standard due dates for competing applications. The FOA will identify whether to follow
standard due dates or whether to follow an alternative due date.
Submission Policies
Learn the nuances of application submission policies, including when late applications might be
allowed, what to do if due dates fall on a weekend or holiday, whether we allow post-submission
materials, how to document system issues, the rules around resubmission applications, etc.
Dealing with System Issues
Are you experiencing system issues with ASSIST, Grants.gov, System for Award Management
(SAM), or the eRA Commons that you believe threaten your ability to submit on time? NIH will not
penalize applicants who experience confirmed issues with federal systems that are beyond their
control. You must report the problem before the submission deadline.
After Submission
Receipt and Referral
Understand how and when applications are given an application identification number and
assigned to a review group and an NIH Institute or Center (IC) for possible funding.
Peer Review
Learn about our two phase peer review process, including initial peer review, Council review,
review criteria, scoring, and summary statements.
Pre-award Process
Learn what happens between peer review and award for applications that have been deemed
highly meritorious in the scientific peer review process. Be ready: if you received a great score in
peer review, you’ll have to submit Just-in-Time information.
Post award Monitoring andReporting
If you receive a grant from the NIH, you will need a lot of information to be a successful steward
of federal funds. This page provides a brief overview of grantee monitoring and reporting
requirements.
F - 8

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.110 - Application Process
Resources
News - Items of Interest
The NIH eSubmission Items of Interest page provides comprehensive information, in an informal
format, on the changes impacting application development and submission.
Annotated Form Sets
These handy documents are a great visual resource for understanding many of the validation
checks we will run against your submitted application.
Contacting NIH Staff
NIH staff is here to help. We strongly encourage NIH applicants and grantees to communicate
with us throughout the grant life cycle. Understanding the roles of NIH staff can help you contact
the right person at each phase of the application and award process.
Contacting Staff at Other PHS Agencies
Applicants are strongly encouraged to communicate with agency staff throughout the entire
application review and awards process.
Systems
ASSIST
eRA Commons
Grants.gov
Information Collection
Authorization
The PHS Act establishes the authority with which NIH and other PHS agencies award grants and
collect information related to grant awards.
Paperwork Burden
The paperwork burden provides the estimated time for completing a grant application.
Collection of Personal Demographic Data
NIH collects personal data through the eRA Commons Personal Profile. The data is confidential
and is maintained under the Privacy Act record system.
F - 9

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.120 - Significant Changes
F.120 - Significant Changes
The Application Instructions are updated and released 2-3 times per year as needed.
Additionally, minor revisions may be made outside of these releases.
This section details all significant changes and revisions made to the instructions since the
last major release.
Within the instructions, new instructions will be marked with this symbol.
In the web version, use your mouse to hover over the icon to read an explanation of the
change.
In a PDF version, this symbol will be visible but will not display hover text. For more
information, see the explanation in the Significant Changes section below.
Revision Notes - October 16, 2020
PHS 398 Career Development Award Supplemental Form
l Under "12. Description of Candidate's Contribution to Program Goals", added hyperlink to Notice
of NIH's Interest in Diversity (NOT-OD-20-031).
PHS 398 Research Training Program Plan Form
l Under section “C. Recruitment Plan to Enhance Diversity” of the “Program Plan”, added hyperlinks
to the Notice of NIH’s Interest in Diversity (NOT-OD-20-031).
PHS Human Subjects and Clinical Trials Information
l Removed language that indicated to skip the rest of the form if you answered “No” to the “Does
the proposed research involve human specimens and/or data?” question.
l Modified text in instruction to “4.1.d. Study Phase” to match form option “N/A”.
Form Screenshots
l Updated form screenshots
F - 10

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.120 - Significant Changes
Release Notes - February 25, 2020
SF 424 Research and Related (R&R) Form Changes
FORMS-F application packages incorporate the latest versions of the federal-wide forms
managed by Grants.gov (OMB Number: 4040-0001, Expiration Date: 12/31/2022).
SF 424 (R&R) Form
l Clarified instructions regarding Agency Routing Identifier.
l Added instructions for applications proposing the use of human fetal tissue obtained from elective
abortions (HFT):
l Added language under section "21. Cover Letter Attachment," sub-section content (Item
9).
l Updated general and SBIR/STTR instructions for the Type of Applicant question, as “Women
Owned” and “Socially and Economically Disadvantaged” information is now collected though SAM.
l Clarified instruction regarding the content of the "Cover Letter Attachment" to indicate that it
must not be used to communicate application assignment preferences.
R&R Senior/Key Person Profile (Expanded) Form
l Added instructions for Career Development and Fellowship applications for the “Credential, e.g.,
agency login” field under the PD/PI Credential Field of the "Profile- Project Director/Principal
Investigator" section.
l Made minor text edits.
R&R Budget and associated R&R Subaward Budget Attachment(s) Form
l Within section F. Other Direct Costs: “8-10: Other”- removed instruction to list inpatient and
outpatient care costs specifically on lines 8 and 9.
l Within section F. Other Direct Costs: “8-10: Other”- removed note regarding requesting an
exception to the single IRB (sIRB) policy.
l Added instructions for applications proposing the use of human fetal tissue obtained from elective
abortions (HFT):
o
Added special instruction for proposed human fetal tissue research under “Who should use
the R&R Budget Form?”
l Added special instructions under “Additional Instructions for Multi-project”.
l Added special instruction for proposed human fetal tissue research under section “F.8-10.
Other.”
l Added special instruction for proposed human fetal tissue research under section “L. Budget
Justification.”
l Added special instruction for proposed human fetal tissue research under section “F.1. Materials
and Supplies.”
F - 11

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.120 - Significant Changes
l Made minor text edits.
Forms-F Changes
PHS 398 Cover Page Supplement Form
l Updated OMB Expiration Date to 2/28/2023
l Added new "Human Fetal Tissue" Section.
l Added instructions for applications proposing the use of human fetal tissue obtained from
elective abortions (HFT).
l Added new "Does the proposed project involve human fetal tissue obtained from elective
abortions?" field.
l Added new instructions and attachment for "HFT Compliance Assurance."
l Added new instructions and attachment for "HFT Sample IRB Consent Form."
l Renumbered form fields.
PHS 398 Modular Budget Form
l Updated OMB Expiration Date to 2/28/2023.
l Added instructions for applications proposing the use of human fetal tissue obtained from
elective abortions (HFT):
l Added special instruction for proposed human fetal tissue research under "Who
should use the PHS 398 Modular Budget Form?"
l Made minor text edits.
PHS 398 Training Budget Form
l Updated OMB Expiration Date to 2/28/2023
PHS 398 Training Subaward Budget Attachment(s) Form
l Updated OMB Expiration Date to 2/28/2023
PHS Additional Indirect Costs Form
l Updated OMB Expiration date to 2/28/2023
PHS 398 Research Plan Form
Updated OMB Expiration Date to 2/28/2023
Clarified instructions on the content of the "Letters of Support" attachment in the "Other
Research Plan Section."
l Removed previous instructions for applications submitted for due dates on or before January
24, 2019 within section 3, "Research Strategy."
l Added instructions for applications proposing the use of human fetal tissue obtained from
elective abortions (HFT):
F - 12

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.120 - Significant Changes
o
Under the introductory part of section 3, "Research Strategy": Added new section titled
Note for Applications Proposing the Use of Human Fetal Tissue.
n Within section 3 "Research Strategy", subsection 3 "Approach" - added a new
bullet point for this information titled 'Special Instructions for Proposed Human
Fetal Tissue Research."
l Made minor text edits.
PHS 398 Career Development Award Supplemental Form
l Updated OMB Expiration Date to 2/28/2023
l Added instructions about rigor, experimental design, and quantitative approaches to the
“Candidate Information and Goals for Career Development” section.
l Added new “Description of Candidate’s Contribution to Program Goals” attachment.
l Renumbered form fields.
l Removed previous instructions for applications submitted for due dates on or before January
24, 2019 within section 4 "Research Strategy."
l Added instructions for applications proposing the use of human fetal tissue obtained from
elective abortions (HFT):
o
Under the introductory part of Section 4 "Research Strategy:: Added a new section titled Note
for Applications Proposing the Use of Human Fetal Tissue.
n Within Section 4 "Research Strategy" - subsection 3 "Approach" - added a new bullet
point for this information titled Special Instructions for Proposed Human Fetal Tissue
Research.
l Made minor text edits.
PHS 398 Research Training Program Plan Form
l Updated OMB Expiration Date to 2/28/2023
l Removed previous instructions for applications submitted for due dates on or before January
24, 2019 within Section 8 "Letters of Support."
l Added instructions to the “Proposed Training” section of the “Program Plan” about rigor,
experimental design, and quantitative approaches.
l Added instructions for the “Plan for Instruction in Methods for Enhancing Reproducibility”
attachment.
l Updated instructions for the “Progress Report (for Renewal applications)” attachment.
l Made minor text edits.
PHS Fellowship Supplemental Form
l Updated OMB Expiration Date to 2/28/2023
l Added instructions about rigor, experimental design, and quantitative approaches to the
“Training Goals and Objectives” section of the “Applicant’s Background and Goals for
Fellowship Training” attachment.
l Added instructions for the “Authentication of Key Biological and/or Chemical Resources” field.
F - 13

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.120 - Significant Changes
l Added new “Description of Candidate’s Contribution to Program Goals” attachment.
l Renumbered form fields.
l Made minor text edits.
PHS Human Subjects and Clinical Trials Information
l Updated OMB Expiration date to 2/28/2023.
o
Changes were made to the form’s organization in the following sections:
o
Who should use the PHS Human Subjects and Clinical Trials Information form
o
Using the PHS Human Subjects and Clinical Trials Information form
o
Use of Human Specimens and/or Data
l Clarified and updated instructions throughout. Significant changes were made for the
following fields:
o
“Provide the ClinicalTrials.gov Identifier”
o
“Section 2 – Study Population Characteristics” instructions now reflect updated
exceptions for required questions.
o
Study Timeline
o
Section 3.2: "Is this a multi-site study that will use the same protocol to conduct non-
exempt human subjects research at more than one domestic site?" - reflect updated
instructions.
o
“Data and Safety Monitoring Plan” attachment
l Updated instructions for delayed onset studies regarding use of single IRB.
l Added new “Inclusion of Individuals Across the Lifespan” attachment.
l Updated instructions for the “Inclusion of Women and Minorities” attachment to reflect
separate “Inclusion of Individuals Across the Lifespan” attachment.
l Added new “Inclusion Enrollment Report Title” field.
l Removed the “Brief Summary” field.
Changed the “Narrative Study” field to “Detailed Description.”
l Updated instructions to Section 3.2 "Is this a multi-site study that will use the same protocol
to conduct non-exempt human subjects research at more than one domestic site?" and the
single IRB plan attachment.
o
Included instructions specific for AHRQ applicants.
l Included instructions specific for AHRQ applicants to Section 3.3 "Data and Safety Monitoring
Plan."
l Added new "Is this an applicable clinical trial under FDAAA?" field.
l Renumbered form fields.
F - 14

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.120 - Significant Changes
PHS Assignment Request Form
l Updated OMB Expiration Date to 2/28/2023.
l Updated language in the form to clarify that this form is for suggestions.
l Removed the “Do Not Assign to Awarding Component” and “Do Not Assign to Study Section”
fields and instructions.
l Added new “Rationale for assignment suggestions” field.
F - 15

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.130 - Program Overview
F.130 - Program Overview
Quick Links
Individual Fellowship Applications ("F" Series).
Individual Fellowship Applications (“F” Series)
The purpose of individual fellowship awards is to provide individual research training
opportunities to fellows at the graduate and postdoctoral levels. This section contains
information for preparing Kirschstein-NRSA (NRSA) fellowship and non-NRSA fellowship
applications.
Additional Instructions for Fellowship:
Additional fellowship instructions will be denoted by a gray call-out box with
orange color coding and with the heading “Additional Instructions for Fellowship”
throughout these application instructions.
NRSA Programs: The NRSA program helps ensure that a diverse pool of highly trained scientists
is available in adequate numbers and in appropriate research areas to carry out the nation’s
biomedical and behavioral research agenda. NRSA fellowships are awarded as a result of national
competition for research training in specified health-related areas. Certain specialized individual
fellowships, such as the predoctoral fellowships (F31 and F30), postdoctoral fellowships (F32),
senior fellowships (F33), and other institute-specific fellowship programs, are provided under this
authority.
Non-NRSA Programs: Fogarty International Center (FIC) and National Library of Medicine (NLM)
also have unique funding authorities for fellowships that are not under the NRSA authority. Note
that non-NRSA programs may have eligibility requirements, due dates, award provisions, and
review criteria that differ from those of NRSA programs. Applicants should refer to their FOA.
Reference Letters: Instructions for submitting the required reference letters for applicable
programs are not contained in these application instructions. Instead, follow the instructions on
NIH’s Reference Letters page. Referees must submit reference letters through the eRA Commons
by the application due date.
Payback Service Requirement: For NRSA programs that include postdoctoral fellows, the
program director must explain the terms of the payback service requirement to all prospective
postdoctoral fellowship candidates. A complete description of the service payback obligation is
available in the NIH Grants Policy Statement, Section 11.4.2: Implementation.
Before Applying:
1. Become familiar with Activity Code: Applicants should become familiar with the “F”
activity code for which support is being requested. A listing of “F” series activity codes,
with their descriptions, is available on the NIH F Kiosk and the AHRQ-Sponsored
Training Opportunities page.
F - 16

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.130 - Program Overview
2. Refer to your specific FOA: Refer to your specific FOA for specific information
associated with the award mechanism, including the eligibility requirements,
requirements for a mentor, review criteria, award provisions, any special application
instructions, and names of individuals who may be contacted for additional or clarifying
information prior to application submission.
l FOAs and other guidelines are available on the NIH F Kiosk.
l Guidelines for the AHRQ fellowships may be found on AHRQ's Research Training
and Education website.
3. Contact Awarding Component: Applicants are encouraged to consult with the
appropriate NIH IC or AHRQ staff prior to submitting an application, as not all
predoctoral, postdoctoral, and senior fellowships are supported by each IC or AHRQ.
l A list of contacts specifically for extramural training at the NIH ICs can be found
on the NIH Training Advisory Committee Roster.
l For contacts at AHRQ, see AHRQ’s Research Training Staff Contacts website.
The following chart provides a list of fellowship activity codes. Since this information is subject to
change, prospective applicants are encouraged to review the F Kiosk for the most current
program information.
Summary of Individual Fellowship Award Programs
Activity
Code
Program Description NRSA?
F05 International Research Fellowships No
F30
Individual Predoctoral National Research Service Award (NRSA) for
M.D./Ph.D. and Other Dual Degree Fellowships
Yes
F31 Predoctoral Individual National Research Service Award Yes
F32 Postdoctoral Individual National Research Service Award Yes
F33 National Research Service Awards for Senior Fellows Yes
F37 Medical Informatics Fellowships No
F38 Applied Medical Informatics Fellowships No
F99/K00 Individual Predoctoral to Postdoctoral Fellow Transition Award No
F - 17

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.200 - SF 424 (R&R) Form
F.200 - SF 424 (R&R) Form
The SF 424 (R&R) Form is used in all grant applications.
This form collects information including type of
submission, applicant information, type of applicant,
and proposed project dates.
View larger image
Quick Links
1. Type of Submission
2. Date Submitted and Applicant Identifier
3. Date Received by State and State Application Identifier
4a. Federal Identifier
4b. Agency Routing Identifier
4c. Previous Grants.gov Tracking ID
5. Applicant Information
6. Employer Identification
7. Type of Applicant
8. Type of Application
9. Name of Federal Agency
10. Catalog of Federal Domestic Assistance Number and Title
11. Descriptive Title of Applicant's Project
12. Proposed Project
13. Congressional District of Applicant
14. Project Director/Principal Investigator Contact Information
15. Estimated Project Funding
16. Is Application Subject to Review by State Executive Order 12372 Process?
17. Certification
18. SFLLL (Disclosure of Lobbying Activities) or Other Explanatory Documentation
19. Authorized Representative
20. Pre-application
21. Cover Letter Attachment
F - 18

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.200 - SF 424 (R&R) Form
1. Type of Submission
This field is required. Check one of the "Type of Submission" boxes:
Pre-application:
The pre-application option is not used by NIH or other PHS agencies unless specifically noted in a
funding opportunity announcement (FOA).
Application:
An "Application" is a request for financial support of a project or activity submitted on specified
forms and in accordance with NIH instructions. (See NIH Types of Applications for an explanation
of the types of applications).
Changed/Corrected Application:
Check this box if you are correcting either system validation errors or application assembly
problems that occurred during the submission process. Changed/corrected applications must be
submitted before the application due date.
When you submit a changed/corrected application, follow these guidelines:
l After submission of an application, there is a two-day application viewing window. Prior to the
due date, you may submit a changed/corrected application. Submitting a changed/corrected
application will replace the previous submission and remove the previous submission from
consideration.
l If you check the “Changed/Corrected Application” box, then "Field 4.c Previous Grants.gov
Tracking ID” is required.
l Do not use the “Changed/Corrected Application” box to denote a resubmission application.
Resubmission applications will be indicated in “Field 8. Type of Application.” See NIH Glossary
for the definition of Resubmission.
2. Date Submitted and Applicant Identifier
The “Date Submitted” field will auto-populate upon application submission.
Fill in the “Applicant Identifier” field, if applicable. The Applicant Identifier is reserved for applicant
use, not the federal agency to which the application is being submitted.
3. Date Received by State and State Application Identifier
Skip the “Date Received by State” and “State Application Identifier” fields.
4.a. Federal Identifier
New Applications without Pre-application: Leave this field blank.
New Applications following Pre-application: Enter the agency-assigned pre-application
number.
F - 19

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.200 - SF 424 (R&R) Form
Resubmission, Renewal, and Revision Applications: The Federal Identifier is required. Include
only the IC and serial number of the previously assigned application/award number (e.g., use
CA987654 from 1R01CA987654-01A1).
4.b. Agency Routing Identifier
Skip the “Agency Routing Identifier” field unless otherwise specified in the FOA or notice in the
NIH Guide for Grants & Contracts.
Applications in response to a NIH Notice of Special Interest require the notice number (e.g., NOT-
IC-FY-XXX) to be entered into this field in order to assign and track applications and awards for
the described initiative.
4.c. Previous Grants.gov Tracking ID
The “Previous Grants.gov Tracking ID” field is required if you checked the “Changed/Corrected
Application” box in “Field 1. Type of Submission.” A Tracking ID number is of the form, for
example, GRANT12345678.
5. Applicant Information
The "Applicant Information" fields reflect information for the applicant organization, not a
specific individual.
Organizational DUNS:
This field is required.
Enter the DUNS or DUNS+4 number of the applicant organization.
This DUNS or DUNS+4 number must match the number entered in the eRA Commons
Institutional Profile (IPF) for the applicant organization. The applicant’s Authorized Organization
Representative (AOR) is encouraged to confirm that a DUNS has been entered into the eRA
Commons IPF prior to application submission. The same DUNS should be used in the eRA
Commons IPF, Grants.gov, System for Award Management (SAM) registration, and in the DUNS
field in the application.
If your organization does not already have a DUNS number, you will need to go to the Dun &
Bradstreet website to obtain the number.
Legal Name:
Enter the legal name of the organization.
Department:
Enter the name of the primary organizational department, service, laboratory, or equivalent level
within the organization.
Division:
Enter the name of the primary organizational division, office, major subdivision, or equivalent
level within the organization.
Street1:
This field is required. Enter the first line of the street address for the applicant organization.
F - 20

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.200 - SF 424 (R&R) Form
Street2:
Enter the second line of the street address for the applicant organization.
City:
This field is required. Enter the city for the address of the applicant organization.
County/Parish:
Enter the county/parish for the address of the applicant organization.
State:
This field is required if the applicant organization is located in the United States or its territories.
Enter the state or territory where the applicant organization is located.
Province:
If “Country” is Canada, enter the province of the applicant organization; otherwise, skip the
“Province” field.
Country:
This field is required. Select the country for the address of the applicant organization.
ZIP/Postal Code:
The ZIP+4 is required if the applicant organization is located in the United States. Otherwise, the
postal code is optional. Enter the ZIP+4 (nine-digit postal code) or postal code of the applicant
organization.
Person to be contacted on matters involving this application
This information is for the administrative contact (e.g., AOR or business official), not the PD/PI.
This person is the individual to be notified if additional information is needed and/or if an award
is made.
Prefix:
Enter or select the prefix, if applicable, for the name of the person to contact on matters related
to this application.
First Name:
This field is required. Enter the first (given) name of the person to contact on matters related to
this application.
Middle Name:
Enter the middle name of the person to contact on matters related to this application.
Last Name:
This field is required. Enter the last (family) name of the person to contact on matters related to
this application.
Suffix:
Enter or select the suffix, if applicable, for the name of the person to contact on matters related
to this application.
Position/Title:
Enter the position/title for the person to contact on matters related to this application.
F - 21

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.200 - SF 424 (R&R) Form
Street1:
This field is required. Enter the first line of the street address for the person to contact on
matters related to this application.
Street2:
Enter the second line of the street address for the person to contact on matters related to this
application.
City:
This field is required. Enter the city for the address of the person to contact on matters related to
this application.
County/Parish:
Enter the county/parish for the address of the person to contact on matters related to this
application.
State:
This field is required if the person to contact on matters related to this application is located in
the United States or its Territories. Enter the state or territory where the person to contact on
matters related to this application is located.
Province:
If “Country” is Canada, enter the province for the person to contact on matters related to this
application; otherwise, skip the “Province” field.
Country:
Select the country for the address of the person to contact on matters related to this application.
ZIP/Postal Code:
The ZIP+4 is required if the person to contact on matters related to this application is in the
United States. Otherwise, the postal code is optional. Enter the ZIP+4 (nine-digit postal code) or
postal code of the person to contact on matters related to this application.
Phone Number:
This field is required. Enter the daytime phone number for the person to contact on matters
related to this application.
Fax Number:
Enter the fax number for the person to contact on matters related to this application.
E-mail:
Enter the e-mail address for the person to contact on matters related to this application. Only
one e-mail address is allowed, but it may be a distribution list.
6. Employer Identification
This field is required.
Enter either the organization’s Taxpayer Identification Number (TIN) or Employer Identification
Number (EIN) as assigned by the Internal Revenue Service. If your organization is not in the
United States, enter 44-4444444. Your EIN may be 12 digits, and if this is the case, enter all 12
digits.
F - 22

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.200 - SF 424 (R&R) Form
7. Type of Applicant
This field is required.
In the first field under “7. Type of Applicant,” enter the appropriate applicant type. If your
applicant type is not specified (e.g., for eligible Agencies of the Federal Government), select “X:
Other (specify),” and indicate the name (e.g., the appropriate federal agency) in the space below.
Additional Instructions for Fellowship:
The information in “7. Type of Applicant” is for the applicant organization, not a
specific individual authorized organization representative (AOR) or fellowship
PD/PI.
Other (Specify):
Complete only if “X. Other (specify)” is selected as the "Type of Applicant."
Women Owned:
Do not use the "Women Owned" checkbox.
Socially and Economically Disadvantaged:
Do not use the "Socially and Economically Disadvantaged" checkbox.
Note: NIH, CDC, and FDA use the Business Type information provided in the System for Award
Management entity record for the applicant organization, rather than the “Woman Owned” and
“Socially and Economically Disadvantaged” checkboxes, to determine the small business
organization type. For more information, see the NIH Guide Notice on Small Business
Organization Type Information Pulled from System for Award Management Record Rather than
Grant Application Form.
8. Type of Application
This field is required.
Select the type of application. Check only one application type. Use the following list of existing
definitions to determine what application type you have. For more information, see NIH Types of
Applications.
l New. Check this option when submitting an application for the first time or in
accordance with other submission policies. See the NIH Grants Policy Statement, Section
2.3.7.4: Submission of Resubmission Application.
l Resubmission. Check this option when submitting a revised (altered or corrected) or
amended application. See also the NIH Application Submission Policies. If your
application is both a “New/Revision/Renewal” and a “Resubmission,” check only the
“Resubmission” box.
l Renewal. Check this option if you are requesting additional funding for a period
subsequent to that provided by a current award. A renewal application competes with
all other applications and must be developed as fully as if the applicant were applying for
the first time.
l Continuation. The box for “Continuation” is used only for specific FOAs.
F - 23

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.200 - SF 424 (R&R) Form
l Revision. Check this option for competing revisions and non-competing administrative
supplements. For more information on competing revisions, see NIH Competing
Revisions. For more information on administrative supplements, see NIH Administrative
Supplements.
Additional Instructions for Fellowship:
The applicant should generally check “New” or “Resubmission.” Unless otherwise
specified in the FOA, individual fellowship awards usually cannot be renewed,
supplemented, or revised. Contact the awarding institute or center staff or refer to
the FOA if clarification is needed.
If Revision, mark appropriate box(es).
You may select more than one.
A. Increase Award
B. Decrease Award
C. Increase Duration
D. Decrease Duration
E. Other (specify)
If “E. Other (specify)” is selected, specify in the space provided.
The boxes for options B, C, D, and E will generally not be used and should not be selected unless
specifically addressed in a particular FOA.
Is this application being submitted to other agencies? What Other Agencies?
In the field “Is this application being submitted to other agencies?” check “Yes” if one or more of
the specific aims submitted in your application is also contained in a similar, identical, or
essentially identical application submitted to another federal agency.
Otherwise, check "No."
If you checked "Yes," indicate the agency or agencies to which the application has been
submitted.
9. Name of Federal Agency
The “Name of Federal Agency” field is pre-populated from the opportunity package and reflects
the agency from which assistance is being requested with this application.
10. Catalog of Federal Domestic Assistance Number and Title
This field is pre-populated from the opportunity package and reflects the Catalog of Federal
Domestic Assistance (CFDA) number of the program under which assistance is requested.
This field may be blank if you are applying to an opportunity that references multiple CFDA
numbers. When this field is blank, leave it blank. The appropriate CFDA number will be
automatically assigned by the agency once the application is assigned to the appropriate
awarding component.
F - 24

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.200 - SF 424 (R&R) Form
11. Descriptive Title of Applicant’s Project
This field is required.
Enter a brief descriptive title of the project.
The descriptive title is limited to 200 characters, including spaces and punctuation.
New Applications: You must have a title different than any other NIH or other PHS Agency
project submitted for the same application due date with the same Project Director/Principal
Investigator (PD/PI).
Resubmission or Renewal Applications: You should normally have the same title as the
previous grant or application; however, if the specific aims of the project have significantly
changed, choose a new title.
Revision Applications: You must have the same title as the currently funded grant.
12. Proposed Project
Start Date:
This field is required. Enter the proposed start date of the project. The start date is an estimate,
and is typically at least nine months after application submission. The project period should not
exceed what is allowed in the FOA.
Ending Date:
This field is required. Enter the proposed ending date of the project.
13. Congressional District of Applicant
Enter the Congressional District as follows: a 2-character state abbreviation, a hyphen, and a 3-
character district number. Examples: CA-005 for California’s 5th district, VA-008 for Virginia’s 8th
district.
If outside the United States, enter 00-000.
For States and U.S. Territories with only a single congressional district, enter “001” for the district
number.
For jurisdictions with no representative, enter “099.”
For jurisdictions with a nonvoting delegate, enter “098” for the district number. Example: DC-098
or PR-098.
If you do not know your Congressional District: Go to The United States House of
Representatives website and search for your Congressional District by entering your ZIP+4. If you
do not know your ZIP+4, look it up on the USPS Look Up Zip Code website.
14. Project Director/Principal Investigator Contact Information
This information is for the PD/PI. The PD/PI is the individual responsible for the overall scientific
and technical direction of the project.
In the eRA Commons profile, the person listed here in “14. Project Director/Principal Investigator
Contact Information” must be affiliated with the applicant organization entered in “5. Applicant
F - 25

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.200 - SF 424 (R&R) Form
Information.” If you are proposing research at an institute other than the one you are currently
at, do not create a separate Commons account with the proposed applicant organization. For
additional information on creating affiliations for users in the eRA Commons, see eRA Account
Management System's Online Help.
If submitting an application reflecting multiple PD/PIs, the individual listed here as the Contact
PD/PI in “14. Project Director/Principal Investigator Contact Information” will be the first PD/PI
listed in F.240 - R&RSenior/Key Person Profile (Expanded) Form.
See F.240 - R&R Senior/Key Person Profile (Expanded) Form for additional instructions for
multiple PD/PIs. To avoid potential errors and delays in processing, ensure that the information
provided in this section is identical to the PD/PI profile information contained in the eRA
Commons.
Additional Instructions for Fellowship:
Provide the name of the individual fellowship applicant (considered the PD/PI for
fellowship award programs). If the PD/PI is not located at the applicant
organization at the time the application is submitted, the information should
reflect where the fellowship applicant can be reached prior to the requested award
start date.
If your proposed fellowship is at a different site than your current institution, the
proposed sponsoring institution will be the applicant organization. You must
affiliate your Commons account with the institution so that you have access to
records submitted on your behalf. Do not create a separate Commons account
with the proposed sponsoring institution.
Multiple PD/PIs cannot apply to fellowship applications.
Prefix:
Enter or select the prefix, if applicable, for the name of the PD/PI.
First Name:
This field is required. Enter the first (given) name of the PD/PI.
Middle Name:
Enter the middle name of the PD/PI.
Last Name:
This field is required. Enter the last (family) name of the PD/PI.
Suffix:
Enter or select the suffix, if applicable, for the PD/PI. Do not use this field to record degrees (e.g.,
Ph.D. or M.D.). Degrees for the PD/PI are requested separately in the R&R Senior/Key Person
Profile (Expanded) Form.
Position/Title:
Enter the position/title of the PD/PI.
Organization Name:
This field is required. This field may be pre-populated from the applicant information section in
this form.
F - 26

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.200 - SF 424 (R&R) Form
Department:
Enter the name of primary organizational department, service, laboratory, or equivalent level
within the organization of the PD/PI.
Division:
Enter the name of primary organizational division, office, major subdivision, or equivalent level
within the organization of the PD/PI.
Street1:
This field is required. Enter first line of the street address for the PD/PI.
Street2:
Enter the second line of the street address for the PD/PI.
City:
This field is required. Enter the city for the address of the PD/PI.
County/Parish:
Enter the county/parish for the address of the PD/PI.
State:
This field is required if the PD/PI is located in the United States or its Territories. Enter the state or
territory where the PD/PI is located.
Province:
If “Country” is Canada, enter the province for the PD/PI; otherwise, skip the “Province” field.
Country:
Select the country for the PD/PI.
ZIP/Postal Code:
The ZIP+4 is required if the PD/PI address is in the United States. Otherwise, the postal code is
optional. Enter the ZIP+4 (nine-digit postal code) or postal code of the PD/PI.
Phone Number:
This field is required. Enter the daytime phone number for the PD/PI.
Fax Number:
Enter the fax number for the PD/PI.
E-mail:
This field is required. Enter the e-mail address for the PD/PI.
15. Estimated Project Funding
All four fields in “15. Estimated Project Funding” are required.
a. Total Federal Funds Requested
Enter the total federal funds, including Direct Costs and F&A Costs (Indirect Costs), requested for
the entire project period.
F - 27

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.200 - SF 424 (R&R) Form
Additional Instructions for Fellowship:
Applicants should refer to the NIH Research Training and Career Development
website for current stipend and other budgetary levels. Enter the total amount
requested for the entire period of support. This amount should include the
applicable stipend amount, the actual tuition and fees, and the standard
institutional allowance.
If new stipend or other payment levels for Kirschstein-NRSA fellowships are
announced after the time of application, these amounts will be automatically
adjusted at the time of award.
Extraordinary Costs: Additional funds may be requested by the institution when
the training of a fellow involves extraordinary costs for travel to field sites remote
from the sponsoring institution or accommodations for fellows who are disabled,
as defined by the Americans with Disabilities Act. The funds requested for
extraordinary costs must be reasonable in relationship to the total dollars awarded
under a fellowship and must be directly related to the approved research training
project. Such additional funds shall be provided only in exceptional circumstances
that are fully justified and explained by the institution in the application.
b. Total Non-Federal Funds
For applications to NIH and other PHS agencies, enter “0” in this field unless cost sharing is a
requirement for the specific FOA.
c. Total Federal & Non-Federal Funds
Enter the total federal and non-federal Funds requested. The amount in this field will be the
same as the amount in the “Total Federal Funds Requested” field unless the specific FOA
indicates that cost sharing is a requirement.
d. Estimated Program Income
Indicate any program income estimated for this project, if applicable.
Additional Instructions for Fellowship:
Enter “0," as the “Estimated Program Income” does not apply to fellowship
applications.
16. Is Application Subject to Review by State Executive Order 12372 Process?
Applicants should check “No, Program is not covered by E.O. 12372.”
17. Certification
This field is required.
The list of NIH and other PHS agencies Certifications, Assurances, and other Policies is found in
the NIH Grants Policy Statement, Section 4: Public Policy Requirements and Objectives.
The applicant organization is responsible for verifying its eligibility and the accuracy, validity, and
conformity with the most current institutional guidelines of all the administrative, fiscal, and
scientific information in the application, including the Facilities and Administrative rate.
F - 28

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.200 - SF 424 (R&R) Form
Deliberate withholding, falsification, or misrepresentation of information could result in
administrative actions, such as withdrawal of an application, suspension and/or termination of
an award, debarment of individuals, as well as possible criminal and/or civil penalties. The signer
further certifies that the applicant organization will be accountable both for the appropriate use
of any funds awarded and for the performance of the grant-supported project or activities
resulting from this application. The grantee institution may be liable for the reimbursement of
funds associated with any inappropriate or fraudulent conduct of the project activity.
Check “I agree” to provide the required certifications and assurances.
18. SFLLL (Disclosure of Lobbying Activities) or Other Explanatory Documentation
If applicable, attach the SFLLL or other explanatory document as per FOA instructions.
If unable to certify compliance with the Certification in the “17. Certification” section above,
attach an explanation. Additionally, as applicable, attach the SFLLL (Standard Form LLL, Disclosure
of Lobbying Activities) or other documents in this item.
For more information:
See the NIH Grants Policy Statement, Section 4.1.17: Lobbying Prohibition, and the NIH Lobbying
Guidance for Grantee Activities page.
19. Authorized Representative
The authorized representative is equivalent to the individual with the organizational authority to
sign for an application. This individual is otherwise known as the authorized organization
representative (AOR) in Grants.gov or the signing official (SO) in eRA Commons.
Prefix:
Enter or select the prefix, if applicable, for the name of the AOR/SO.
First Name:
This field is required. Enter the first (given) name of the AOR/SO
Middle Name:
Enter the middle name of the AOR/SO.
Last Name:
This field is required. Enter the last (family) name of the AOR/SO.
Suffix:
Enter or select the suffix, if applicable, for the AOR/SO.
Position/Title:
This field is required. Enter the position/title of the name of the AOR/SO.
Organization Name:
This field is required. Enter the name of the organization for the AOR/SO.
Department:
Enter the name of the primary organizational department, service, laboratory, or equivalent level
within the organization for the AOR/SO.
F - 29

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.200 - SF 424 (R&R) Form
Division:
Enter the name of the primary organizational division, office, major subdivision, or equivalent
level within the organization for the AOR/SO.
Street1:
This field is required. Enter the first line of the street address for the AOR/SO.
Street2:
Enter the second line of the street address for the AOR/SO.
City:
This field is required. Enter the city for the address of the AOR/SO.
County/Parish:
Enter the county/parish for the address of the AOR/SO.
State:
This field is required if the AOR/SO is located in the United States or its Territories. Enter the state
or territory where the AOR/SO is located.
Province:
If “Country” is Canada, enter the province for the AOR/SO; otherwise, skip the “Province” field.
Country:
Select the country for the address of the AOR/SO.
ZIP/Postal Code:
The ZIP+4 is required if the AOR/SO is in the United States. Otherwise, the postal code is optional
Enter the ZIP+4 (nine-digit postal code) or postal code of the AOR/SO.
Phone Number:
This field is required. Enter the daytime phone number for the AOR/SO.
Fax Number:
Enter the fax number for the AOR/SO.
Email:
This field is required. Enter the e-mail address for the AOR/SO.
Signature of Authorized Representative:
Grants.gov will record the electronic signature for the AOR/SO who submits the application.
It is the organization’s responsibility to assure that only properly authorized individuals sign in
this capacity and/or submit the application to Grants.gov.
Date Signed:
Grants.gov will generate this date upon application submission.
20. Pre-application
Unless specifically noted in a FOA, NIH and other PHS agencies do not use pre-applications. The
"Pre-application" attachment field should not be used for any other purpose.
F - 30

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.200 - SF 424 (R&R) Form
If permitted by your FOA, attach this information as a PDF.
21. Cover Letter Attachment
The cover letter is for internal use only and will not be shared with peer reviewers.
Who must complete the “Cover Letter Attachment:”
Refer to the “content” list below for items that are permitted, as well as for specific situations in
which a cover letter must be included.
A cover letter must not be included with post-award submissions, such as administrative
supplements, change of grantee institution, or successor-in-interest.
Format:
Attach the cover letter, addressed to the Division of Receipt and Referral, in accordance with the
FOA and/or these instructions.
Attach the cover letter in the correct location, specifically verifying that the cover letter has
not been uploaded to the “20. Pre-application” field which is directly above the “21. Cover
Letter Attachment” field. This will ensure the cover letter attachment is kept separate from the
assembled application in the eRA Commons and made available only to appropriate staff.
Content:
Do not use the cover letter to communicate application assignment preferences. The
Assignment Request Form is provided for that purpose.
The letter should contain any of the following information, as applicable:
1. Application title.
2. Title of FOA (PA or RFA).
3. For late applications (see Late Application policy on NIH's Application Submission
Policies) include specific information about the timing and nature of the delay.
4. For changed/corrected applications submitted after the due date, a cover letter is
required, and it must explain the reason for late submission of the changed/corrected
applications. If you already submitted a cover letter with a previous submission and are
now submitting a late change/corrected application, you must include all previous cover
letter text in the revised cover letter attachment. The system does not retain any
previously submitted cover letters; therefore, you must repeat all information previously
submitted in the cover letter as well as any additional information.
5. Explanation of any subaward budget components that are not active for all budget
periods of the proposed grant (see F.310 – R&R Subaward Budget Attachment(s) Form).
6. Statement that you have attached any required agency approval documentation for the
type of application submitted. This may include approval for applications that request
$500,000 or more, approval for Conference Grant or Cooperative Agreement (R13 or
U13), etc. It is recommended that you include the official communication from an NIH
official as part of your cover letter attachment.
7. When intending to submit a video as part of the application, the cover letter must
include information about the intent to submit it; if this is not done, the video will not be
F - 31

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.200 - SF 424 (R&R) Form
accepted. See NIH Grants Policy Statement, Section 2.3.7.7: Post Submission Grant
Application Materials for additional information.
8. Include a statement in the cover letter if the proposed studies will generate large-scale
human or non-human genomic data as detailed in the NIH Genomic Data Sharing Policy
(see the NIH Grants Policy Statement, Section 2.3.7.10: NIH Genomic Data Sharing and
Section 8.2.3.3: Genomic Data Sharing (GDS) Policy/Policy for Genome-Wide Association
Studies (GWAS)).
9.
1
Include a statement in the cover letter if the proposed studies involve human fetal
tissue obtained from elective abortions (HFT), regardless of whether or not Human
Subjects are involved and/or there are costs associated with the HFT. For further
information on HFTpolicy refer to the NIHGrants Policy Statement, Section 2.3.7.11
Human Fetal Tissue from Elective Abortions, Section 4.1.14 Human Fetal Tissue Research
and Section 4.1.14.2 Human Fetal Tissue from Elective Abortions.
Additional Instructions for Fellowship:
Individual fellowship applicants must include a cover letter that contains a list of
referees (including name, departmental affiliation, and institution).
1
A statement is required in the cover letter if the studies proposed involve human fetal tis-
sue for any reason. See NIH GPS 2.3.7.11, 4.1.14 and 4.1.14.2
F - 32

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.220 - R&R Other Project Information Form
F.220 - R&R Other Project Information
Form
The R&R Other Project Information Form is used for all
grant applications. This form includes questions on the
use of human subjects, vertebrate animals, and
environmental impact. This form also has fields to
upload an abstract, project narrative, references,
information on facilities, and equipment lists.
View larger image
Quick Links
1. Are Human Subjects Involved?
1a. If YES to Human Subjects
2. Are Vertebrate Animals Used?
2a. If YES to Vertebrate Animals
3. Is proprietary/privileged information included in the application?
4. Environmental Questions
5. Is the research performance site designated, or eligible to be designated, as a historic place?
6. Does this project involve activities outside of the United States or partnerships with inter-
national collaborators?
7. Project Summary/Abstract
8. Project Narrative
9. Bibliography & References Cited
10. Facilities & Other Resources
11. Equipment
12. Other Attachments
Additional Instructions for Fellowship:
This R&R Other Project Information Form should be completed in consultation
with the sponsor and administrative officials at the sponsoring institution.
1. Are Human Subjects Involved?
This field is required.
F - 33

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.220 - R&R Other Project Information Form
If activities involving human subjects are planned at any time during the proposed project at any
performance site, check “Yes.” Check “Yes” even if the proposed project is exempt from
regulations for the Protection of Human Subjects, or if activities involving human subjects are
anticipated within the period of award but plans are indefinite.
If activities involving human subjects are not planned at any time during the proposed project at
any performance site, select “No” and skip the rest of the "Are Human Subjects Involved" section.
Whether you answer “Yes” or “No” to the “Are Human Subjects Involved?” question here, your
answer will populate the relevant field in the F.500 – PHS Human Subjects and Clinical Trials
Information form (see exception for Training Applications in the Training-specific instructions).
Follow the F.500 – PHS Human Subjects and Clinical Trials Information form instructions to
complete the relevant questions in that form.
Need help determining whether your application includes human subjects? Check out the
NIH Research Involving Human Subjects website for information, including an Infopath
Questionnaire designed to walk applicants through the decision process.
Note on the use of human specimens or data: Applications involving the use of human
specimens or data may or may not be considered to be research involving human subjects,
depending on the details of the materials to be used. If you check “No” to “Are Human Subjects
Involved?” but your application proposes using human specimens or data, you will be required to
provide a clear justification about why this use does not constitute human subjects research.
Follow the F.500 – PHS Human Subjects and Clinical Trials Information form instructions.
For more information on human biospecimens or data: Refer to the NIH page on Frequently
Asked Questions on Human Specimens, Cell Lines, or Data and the Research Involving Private
Information or Biological Specimens flowchart.
Additional Instructions for Fellowship:
In many instances, the fellow will be participating in research supported by a
research project grant for which the IRB review of human subjects is already
complete or an exemption has been designated. This review or exemption
designation is sufficient, provided that the IRB determines that participation of the
Fellow does not substantially modify the research.
1.a. If YES to Human Subjects
Your answers here in question “1.a. If YES to Human Subjects” will populate the corresponding
fields in the F.500 – PHS Human Subjects and Clinical Trials Information form.
Is the Project Exempt from Federal regulations? Yes/No
If the project is exempt from federal regulations, check “Yes” and check the appropriate
exemption number.
Human subjects research should only be designated as exempt if all of the proposed research
projects in an application meet the criteria for exemption.
If the project is not exempt from federal regulations, check “No.”
For more information, see the NIH’s Exempt Human Subjects Research infographic.
If yes, check appropriate exemption number 1, 2, 3, 4, 5, 6, 7, 8:
If you selected “Yes” to “Is the Project Exempt from Federal Regulations,” select the appropriate
exemption number.
F - 34

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.220 - R&R Other Project Information Form
The categories of research that qualify for exemption are defined in the Common Rule for the
Protection of Human Subjects. These regulations can be found at 45 CFR 46.
Need help determining the appropriate exemption number? Refer to NIH's Research Involving
Human Subjects Frequently Asked Questions.
The Office of Human Research Protections (OHRP) guidance states that appropriate use of
exemptions described in 45 CFR 46 should be determined by an authority independent from the
investigators (for more information, see OHRP's Frequently Asked Questions). Institutions often
designate their Institutional Review Board (IRB) to make this determination. Because NIH does
not require IRB approval at the time of application, the exemptions designated often represent
the opinion of the PD/PI, and the justification provided for the exemption by the PD/PI is
evaluated during peer review.
If no, is the IRB review Pending? Yes/No
If IRB review is pending, check “Yes.”
Applicants should check “Yes” to the question “Is the IRB review Pending?” even if the IRB
review/approval process has not started by the time of submission.
If IRB review is not pending (e.g., if the review is complete), check “No.”
IRB Approval Date:
Enter the latest IRB approval date (if available). Leave blank if IRBapproval is pending.
An IRB approval date is not required at the time of submission when IRB review is pending. This
may be requested later in the pre-award cycle as a Just-In-Time requirement. See the NIH Grants
Policy Statement, Section 2.5.1: Just-in-Time Procedures for more information.
Human Subject Assurance Number:
Enter the approved Federalwide Assurance (FWA) number that the applicant has on file with
OHRP. Enter the 8-digit number. Do not enter “FWA” before the number.
Enter “None” if the applicant organization does not have an approved FWA on file with OHRP. In
this case, the applicant organization, by the signature in the Certification section on the F.200 -
SF424 (R&R) Form, is declaring that it will comply with 45 CFR 46 and proceed to obtain a FWA
(see Office for Human Research Protections website). Do not enter the FWA number of any
collaborating institution.
Additional Instructions for Fellowship:
If research proposed in the fellowship application has been previously reviewed
and approved by an IRB and is covered by an approved FWA, provide the FWA
number and the latest IRB approval date for the proposed activities. The latest IRB
approval date must be within one year of the application due date.
2. Are Vertebrate Animals Used?
This field is required.
If activities involving vertebrate animals are planned at any time during the proposed project at
any performance site, check “Yes.” Otherwise, check “No” and skip the rest of the “2. Are
Vertebrate Animals Used?” section.
F - 35

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.220 - R&R Other Project Information Form
Note that the generation of custom antibodies constitutes an activity involving vertebrate
animals.
If animal involvement is anticipated within the period of award but plans are indefinite, check
"Yes."
Additional Instructions for Fellowship:
In many instances, the fellow will be participating in research supported by a
research project grant for which the IACUC review has been completed and
approval obtained. This review is sufficient, provided that participation of the fellow
does not substantially modify the research. The appropriate grant(s) must be
identified along with the IACUC approval date(s).
The sponsoring institution must ensure that the fellow is enrolled in the
institution’s animal welfare training and safety programs for personnel who have
contact with animals, as appropriate. It is also the sponsoring institution’s
responsibility to ensure that the fellow is properly supervised when working with
live vertebrate animals.
If you have answered “Yes” to the “Are Vertebrate Animals Used?” question, you
must also provide an explanation and anticipated timing of animal use in F.430 -
PHS Fellowship Supplemental Form, Vertebrate Animals. This attachment must be
submitted and reviewed prior to the involvement of animals in any research
studies.
2.a. If YES to Vertebrate Animals
Is the IACUC review Pending?
If an Institutional Animal Care and Use Committee (IACUC) review is pending, check "Yes."
Applicants should check “Yes” to the "Is the IACUC review Pending?" question even if the IACUC
review/approval process has not started by the time of submission.
If IACUC review is not pending (e.g. if the review is complete), check “No."
IACUC Approval Date:
Enter the latest IACUC approval date (if available). Leave blank if IACUC approval is pending.
IACUC approval must have been granted within three years of the application submission date to
be valid.
An IACUC approval date is not required at the time of submission. NIH does not require
verification of review and approval of the proposed research by the IACUC before peer review of
the application. However, this information is required under the NIH Grants Policy Statement
Section 2.5.1: Just-in-Time Procedures.
Animal Welfare Assurance Number
Enter the federally approved assurance number, if available.
Enter “None” if the applicant organization does not have an Office of Laboratory Animal Welfare
(OLAW)-approved Animal Welfare Assurance.
To determine whether the applicant organization holds an Animal Welfare Assurance with an
associated number, see the lists of Domestic and Foreign Assured institutions. Do not enter the
Animal Welfare Assurance number for a Project/Performance Site of a collaborating
institution.
F - 36

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.220 - R&R Other Project Information Form
When an applicant organization does not have an Animal Welfare Assurance number, the
authorized organization representative’s signature on the application constitutes declaration
that the applicant organization will submit an Animal Welfare Assurance when requested by
OLAW.
If the animal work will be conducted at an institution with an Animal Welfare Assurance and the
applicant organization does not have the following:
l an animal care and use program;
l facilities to house animals and conduct research on site; and
l IACUC;
then, the applicant must obtain an Inter-institutional Assurance from OLAW prior to an award.
3. Is proprietary/privileged information included in the application?
This field is required.
Patentable ideas; trade secrets; or privileged, confidential commercial, or financial information
should be included in applications only when such information is necessary to convey an
understanding of the proposed project.
If the application includes such information, check “Yes” and clearly mark each line or paragraph
on the pages containing the proprietary/privileged information with a statement similar to: “The
following contains proprietary/privileged information that (name of applicant) requests not be
released to persons outside the government, except for purposes of review and evaluation.” This
statement can be included at the top of each page as applicable.
If a grant is awarded as a result of or in connection with the submission of this application, the
government shall have the right to use or disclose the information to the extent authorized by
law. Although the grantee institution and the PD/PI will be consulted about any such disclosure,
the NIH and other PHS agencies will make the final determination. Any indication by the applicant
that the application contains proprietary or privileged information does not automatically shield
the information from release in response to a Freedom of Information Act (FOIA) request should
the application result in an award (see 45 CFR 5). Additionally, if an applicant fails to identify
proprietary information at the time of submission as instructed here, a significant substantive
justification will be required to withhold the information if requested under FOIA.
4. Environmental Questions
Question 4 pertains to the environmental impact of the proposed research.
4.a. Does this Project Have an Actual or Potential Impact - positive or negative - on the envir-
onment?
This field is required.
Indicate whether or not this project has an actual or potential impact on the environment.
Most NIH research grants are not expected to individually or cumulatively have a significant effect
on the environment, and NIH has established several categorical exclusions allowing most
applicants to answer “No” unless a specific FOA indicates that the National Environmental Policy
Act (NEPA) applies. However, if an applicant expects that the proposed project will have an actual
F - 37

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.220 - R&R Other Project Information Form
or potential impact on the environment, or if any part of the proposed research and/or project
includes one or more of the following scenarios, check “Yes.”
1. The potential environmental impacts of the proposed research may be of greater scope
or size than other actions included within a category.
2. The proposed research threatens to violate a federal, state, or local law established for
the protection of the environment or for public health and safety.
3. Potential effects of the proposed research are unique or highly uncertain.
4. Use of especially hazardous substances or processes is proposed for which adequate
and accepted controls and safeguards are unknown or not available.
5. The proposed research may overload existing waste treatment plants due to new loads
(volume, chemicals, toxicity, additional hazardous wasted, etc.).
6. The proposed research may have a possible impact on endangered or threatened
species.
7. The proposed research may introduce new sources of hazardous/toxic wastes or require
storage of wastes pending new technology for safe disposal.
8. The proposed research may introduce new sources of radiation or radioactive materials.
9. Substantial and reasonable controversy exists about the environmental effects of the
proposed research.
4.b. If yes, please explain:
If you answered “Yes” to Question 4.a., you must provide an explanation here as to the actual or
potential impact of the proposed research on the environment. Your entry is limited to 55
characters.
4.c. If this project has an actual or potential impact on the environment, has an exemption been
authorized or an environmental assessment (EA) or environmental impact statement (EIS) been
performed? Yes/No.
This field is required if you answered “Yes” to Question 4.a. Check “Yes” or “No.”
4.d. If yes, please explain:
Enter additional details about the EA or EIS here. Your entry is limited to 55 characters.
5. Is the research performance site designated, or eligible to be designated, as a
historic place?
This field is required.
If any research performance site is designated, or eligible to be designated, as a historic place,
check the “Yes” box. Otherwise, check “No.”
5.a. If yes, please explain:
If you checked “Yes” to indicate that any performance site is designated, or eligible to be
designated, as a historic place, provide the explanation here. Your entry is limited to 55
characters.
F - 38

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.220 - R&R Other Project Information Form
6. Does this project involve activities outside of the United States or partnerships
with international collaborators?
This field is required.
Indicate whether this project involves activities outside of the United States or partnerships with
international collaborators. Check “Yes” or “No.”
Applicants to NIH and other PHS agencies must check “Yes” if the applicant organization is a
foreign institution or if the project includes a foreign component. See NIH Glossary for a
definition of a foreign component.
If you have checked “Yes” to Question 6, you must include a “Foreign Justification” attachment in
Field 12, Other Attachments. Describe special resources or characteristics of the research project
(e.g., human subjects, animals, disease, equipment, and techniques), including the reasons why
the facilities or other aspects of the proposed project are more appropriate than a domestic
setting. In the body of the text, begin the section with a heading indicating “Foreign
Justification” and name the file “Foreign Justification.”
Additional Instructions for Fellowship:
If you have checked “Yes” to Question 6, your justification must include a
description of how the foreign training is more appropriate than in a domestic
setting. Include reasons why the facilities, the sponsor, and/or other aspects of the
proposed experience are more appropriate in a foreign setting. The justification is
evaluated in terms of the scientific advantages of the foreign training as compared
to the training available domestically. Foreign training will be considered for
funding only when the scientific advantages are clear. The foreign justification
should be provided as a separate attachment in the "12. Other Attachments"
section in F.220 - R&R Other Project Information Form.
6.a. If yes, identify countries:
This field is required if you answered “Yes” to Question 6. Enter the countries with which
international cooperative activities are planned.
You may use abbreviations. Your entry is limited to 55 characters.
6.b. Optional Explanation:
This field is optional. Enter an explanation for involvement with outside entities. Your entry is
limited to 55 characters.
7. Project Summary/Abstract
The "Project Summary/Abstract" attachment is required.
The project summary is a succinct and accurate description of the proposed work and should be
able to stand on its own (separate from the application). This section should be informative to
other persons working in the same or related fields and understandable to a scientifically literate
reader. Avoid both descriptions of past accomplishments and the use of the first person. Please
be concise.
F - 39

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.220 - R&R Other Project Information Form
Format:
This section is limited to 30 lines of text, and must follow the required font and margin
specifications. A summary that exceeds the 30-line limit will be flagged as an error by the Agency
upon submission.
Attach this information as a PDF file. See the Format Attachments page.
Content:
State the application's broad, long-term objectives and specific aims, making reference to the
health relatedness of the project (i.e., relevance to the mission of the agency). Describe the
research design and methods for achieving the stated goals. Be sure that the project summary
reflects the key focus of the proposed project so that the application can be appropriately
categorized.
Do not include proprietary, confidential information or trade secrets in the project summary. If
the application is funded, the project summary will be entered into an NIH database and made
available on the NIH Research Portfolio Online Reporting Tool (RePORT) and will become public
information.
Note that the "Project Summary/Abstract" attachment is not same as the "Research Strategy"
attachment.
Additional Instructions for Fellowship:
In addition to summarizing the research project to be conducted under the
fellowship award, describe the fellowship training plan and the environment in
which the research training will take place. The entire “Project Summary/Abstract”
attachment is limited to 30 lines of text.
8. Project Narrative
The "Project Narrative" attachment is required.
Content:
Describe the relevance of this research to public health in, at most, three sentences. For example,
NIH applicants can describe how, in the short or long term, the research would contribute to
fundamental knowledge about the nature and behavior of living systems and/or the application
of that knowledge to enhance health, lengthen life, and reduce illness and disability. If the
application is funded, this public health relevance statement will be combined with the project
summary (above) and will become public information.
9. Bibliography & References Cited
Who must complete the “Bibliography & References Cited” attachment:
The “Bibliography & References Cited” attachment is required unless otherwise noted in the
FOA.
Format:
Attach this information as a PDF file. See the Format Attachments page.
F - 40

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.220 - R&R Other Project Information Form
Content:
See the following instructions for which references to include in the “Bibliography and
References Cited” attachment.
Additional Instructions for Fellowship:
The “Bibliography & References Cited” attachment should include any references
cited in F.430 - PHS Fellowship Supplemental Form and in the F.500 - PHS Human
Subjects and Clinical Trials Information form.
When citing articles that fall under the Public Access Policy, were authored or co-authored by the
applicant, and arose from NIH support, provide the NIH Manuscript Submission reference
number (e.g., NIHMS97531) or the PubMed Central (PMC) reference number (e.g., PMCID234567)
for each article. If the PMCID is not yet available because the Journal submits articles directly to
PMC on behalf of their authors, indicate “PMC Journal – In Process.” NIHmaintains a list of such
journals.
Citations that are not covered by the Public Access Policy, but are publicly available in a free,
online format may include URLs or PubMed ID (PMID) numbers along with the full reference. The
references should be limited to relevant and current literature. While there is not a page
limitation, it is important to be concise and to select only those literature references pertinent to
the proposed research.
You are allowed to cite interim research products. Note: interim research products have specific
citation requirements. See related Frequently Asked Questions for more information.
10. Facilities & Other Resources
Format:
The “Facilities & Other Resources” attachment is required unless otherwise specified in the FOA.
Content:
Describe how the scientific environment in which the research will be done contributes to the
probability of success (e.g., institutional support, physical resources, and intellectual rapport). In
describing the scientific environment in which the work will be done, discuss ways in which the
proposed studies will benefit from unique features of the scientific environment or from unique
subject populations or how studies will employ useful collaborative arrangements.
If there are multiple performance sites, describe the resources available at each site.
Describe any special facilities used for working with biohazards and any other potentially
dangerous substances. Note: Information about select agents must be described in the
Research Plan, Select Agent Research.
For early stage investigators (ESIs), describe institutional investment in the success of the
investigator. See NIH's New and Early Stage Investigator Policies. Your description may include
the following elements:
l resources for classes, travel, or training;
l collegial support, such as career enrichment programs, assistance and guidance in the
supervision of trainees involved with the ESI’s project, and availability of organized peer
groups;
F - 41

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.220 - R&R Other Project Information Form
l logistical support, such as administrative management and oversight and best practices
training;
l financial support, such as protected time for research with salary support.
Additional Instructions for Fellowship:
Include a detailed description of the institutional facilities and resources available
to the fellowship applicant. The information provided is of major importance in
establishing the feasibility of the goals of the fellowship training plan.
11. Equipment
The “Equipment” attachment is required.
Format:
Attach this information as a PDF file.
Content:
List major items of equipment already available for this project and, if appropriate, identify the
equipment's location and pertinent capabilities.
12. Other Attachments
Attach a file to provide additional information only in accordance with the FOA and/or agency-
specific instructions.
If applicable, attach a “Foreign Justification” here. (See Question 6 above).
F - 42
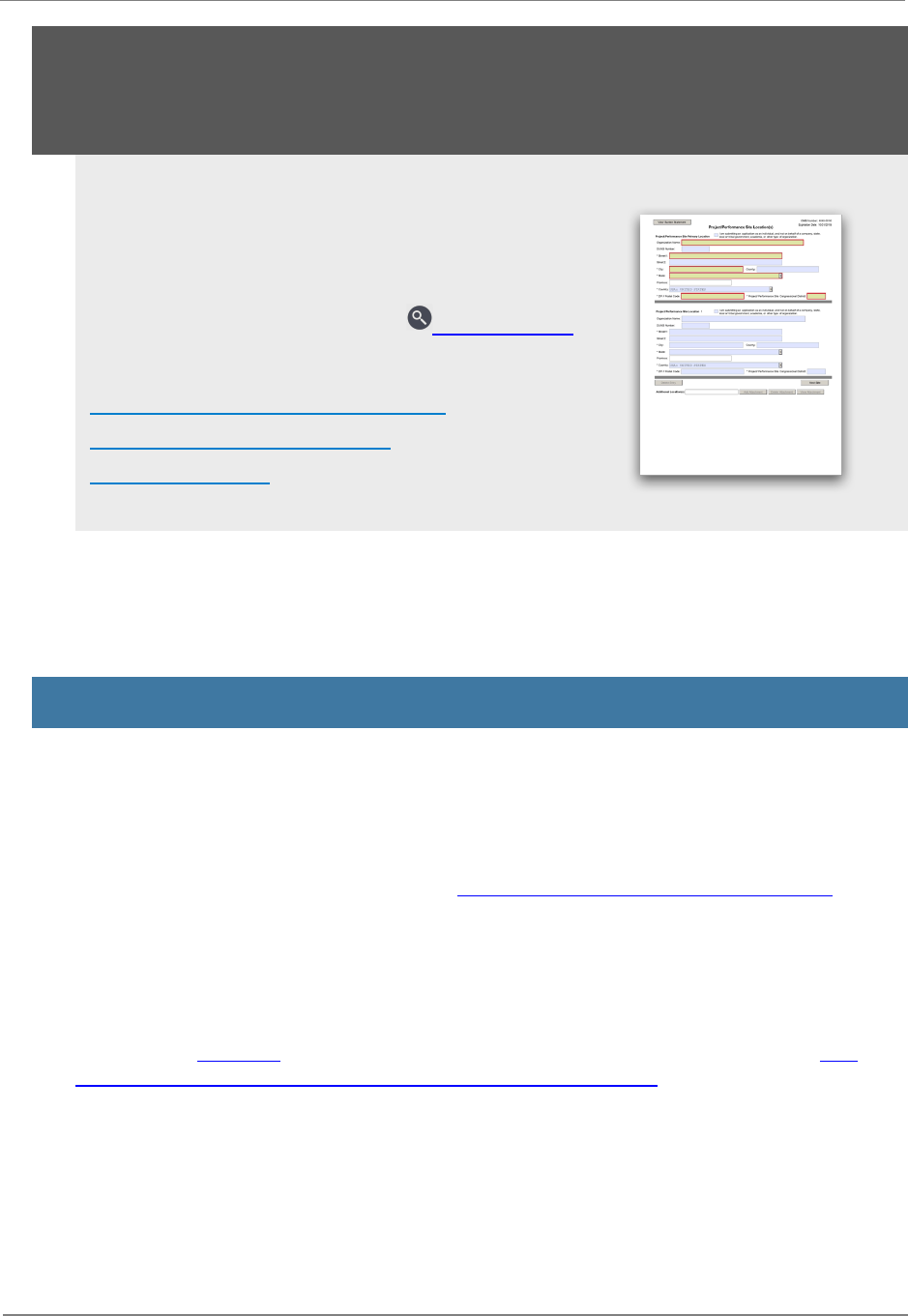
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.230 - Project/Performance Site Location(s) Form
F.230 - Project/Performance Site
Location(s) Form
The Project/Performance Site Location(s) Form is used
for all grant applications. It is used to report the primary
location and any other locations at which the project
will be performed.
View larger image
Quick Links
Project/Performance Site Primary Location
Project/Performance Site Location 1
Additional Location(s)
Using the Project/Performance Site Location(s) Form:
This form allows for the collection of multiple performance sites. If you need to add more
project/performance site locations than the form allows, enter the information in a separate file and
add it to the “Additional Locations” section.
Project/Performance Site Primary Location
Generally, the primary location should be that of the applicant organization or identified as off-
site in accordance with the conditions of the applicant organization’s negotiated Facilities and
Administrative (F&A) agreement. This information must agree with the F&A information on the
budget form of the application.
Provide an explanation of resources available from each project/performance site on the
"Facilities and Resources" attachment of the F.220 - R&R Other Project Information Form.
If the proposed project involves human subjects or live vertebrate animals, it is up to the
applicant organization to ensure that all sites meet certain criteria:
Human Subjects: If a project/performance site is engaged in research involving human subjects,
the applicant organization is responsible for ensuring that the project/performance site operates
under an appropriate Federal Wide Assurance for the protection of human subjects and
complies with 45 CFR 46 and other NIH human subject related policies described in the NIH
Grants Policy Statement, Section 4.1.15: Human Subjects Protections.
Vertebrate Animals: For research involving live vertebrate animals, the applicant organization
must ensure that all project/performance sites hold an Office of Laboratory Animal Welfare
(OLAW)-approved Animal Welfare Assurance. If the animal work will be conducted at an
institution with an Animal Welfare Assurance and the applicant organization does not have the
following:
F - 43

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.230 - Project/Performance Site Location(s) Form
l an animal care and use program;
l facilities to house animals and conduct research on site; and
l an IACUC;
then applicant must obtain an Inter-institutional Assurance from OLAW prior to an award.
Additional Instructions for Fellowship:
One of the sites indicated must be the sponsoring organization, and generally, the
sponsoring organization is the primary location. Indicate where the training
described in the Research Training Plan will be conducted. If there is more than one
training site, including any Department of Veterans Affairs (VA) facilities or foreign
sites, list them all in the fields provided for Location 1, and additional locations, as
necessary.
If there are unusual circumstances involved in the research training proposed, such
as fieldwork or a degree sought from an institution other than the one in which
the research training will take place, describe these circumstances in F.220 - R&R
Other Project Information Form, Facilities and Resources.
“I am submitting an application as an individual, and not on behalf of a company, state, local or
tribal government, academia, or other type of organization”:
Do not check the box for “I am submitting an application as an individual, and not on behalf of a
company, state, local or tribal government, academia, or other type of organization” unless
otherwise specified by the FOA.
Organization Name:
This field is required. Enter the organization name of the primary site where the work will be
performed.
DUNS Number:
This field is required for the primary performance site.
Enter the DUNS or DUNS+4 number associated with the organization where the project will be
performed.
Street1:
This field is required. Enter the first line of the street address of the primary performance site
location.
Street2:
Enter the second line of the street address of the primary performance site location.
City:
This field is required. Enter the city for the address of the primary performance site location.
County:
Enter the county of the primary performance site location.
State:
This field is required if the site is located in the United States or its Territories. Enter the state or
territory where the primary performance site is located.
F - 44

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.230 - Project/Performance Site Location(s) Form
Province:
If “Country" is Canada, enter the province for the primary performance site; otherwise, skip the
“Province” field.
Country:
This field is required. Select the country of the address for the primary performance site location.
ZIP/Postal Code:
The ZIP+4 is required if the primary performance site location is in the United States. Otherwise,
the postal code is optional. Enter the ZIP+4 (nine-digit postal code) or postal code of the primary
performance site.
Project/Performance Site Congressional District:
Enter the Congressional District as follows: a 2-character state abbreviation, a hyphen, and a 3-
character district number. Examples: CA-005 for California’s 5th district, VA-008 for Virginia’s 8th
district.
It is likely this field will be identical to the “Congressional District of Applicant” field provided
elsewhere in the application.
If the program/project is outside the United States, enter 00-000.
For States and U.S. territories with only a single congressional district, enter “001” for the district
number.
For jurisdictions with no representative, enter “099.”
For jurisdictions with a nonvoting delegate, enter “098” for the district number. Example: DC-098
or PR-098.
If all districts in a state are affected, enter “all” for the district number. Example: "MD-all" for all
congressional districts in Maryland.
If nationwide (all districts in all states), enter "US-all."
If you do not know the Congressional District: Go to the United States House of
Representatives website and search for the Congressional District by entering the ZIP+4. If you
do not know the ZIP+4, look it up on the USPS Look Up Zip Code website.
Project/Performance Site Location 1
Use this “Project/Performance Site Location 1” block to provide information on performance sites
in addition to the Primary Performance Site listed above, if applicable. Include any VA facilities and
foreign sites.
Organization Name:
Enter the organization name of the performance site location.
DUNS Number:
Enter the DUNS or DUNS+4 number associated with the performance site.
Street1:
This field is required. Enter first line of the street address of the performance site location.
F - 45

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.230 - Project/Performance Site Location(s) Form
Street2:
Enter the second line of the street address of the performance site location.
City:
This field is required. Enter the city for the address of the performance site location.
County:
Enter the county of the performance site location.
State:
This field is required if the project performance site is located in the United States or its
Territories. Enter the state or territory where the performance site is located.
Province:
If “Country” is Canada, enter the province for the performance site; otherwise, skip the “Province”
field.
Country:
This field is required. Select the country of the performance site location.
ZIP/Postal Code:
The ZIP+4 is required if the performance site location is in the United States. Otherwise, the
postal code is optional. Enter the ZIP+4 (nine-digit postal code) of the performance site location.
Project/Performance Site Congressional District:
Enter the Congressional District as follows: a 2-character state abbreviation, a hyphen, and a 3-
character district number. Examples: CA-005 for California’s 5th district, VA-008 for Virginia’s 8th
district.
If the program/project is outside the United States, enter 00-000.
For States and U.S. territories with only a single congressional district enter “001” for the district
number.
For jurisdictions with no representative, enter “099.”
For jurisdictions with a nonvoting delegate, enter “098” for the district number. Example: DC-098
or PR-098.
If all districts in a state are affected, enter “all” for the district number. Example: "MD-all" (for all
congressional districts in Maryland).
If nationwide (all districts in all states), enter "US-all."
If you do not know the Congressional District: Go to the United States House of
Representatives website and search for your Congressional District by entering your ZIP+4. If you
do not know the ZIP+4 look it up on the USPS Look Up Zip Code website.
Additional Location(s)
If you need to add more project/performance site locations than the form allows, enter the
information in a separate file and add it to the “Additional Locations” section.
F - 46

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.240 - R&RSenior/Key Person Profile (Expanded) Form
F.240 - R&R Senior/Key Person Profile
(Expanded) Form
The R&R Senior/Key Person Profile (Expanded) Form is
used for all grant applications, and allows the collection
of data for all senior/key persons associated with the
project. Some information for the PD/PI may be pre-
populated from the SF424 (R&R) form. See instructions in
F.200 - SF 424 (R&R) Form if these fields are empty.
View larger image
Quick Links
Profile - Project Director/Principal Investigator
Instructions for a Biographical Sketch
Profile - Senior/Key Person
Additional Senior/Key Person Profile(s)
Using the R&R Senior/Key Person Profile (Expanded) Form
This form allows for the data collection for a PD/PI and up to 99 other senior/key individuals (including
any multi-PD/PIs). After the first 100 individuals have been entered, use the “Additional Senior/Key
Person Profiles Format Page” to attach any remaining data.
To ensure proper performance of this form, save your work frequently.
Who qualifies as a Senior/Key Person?
Unless otherwise specified in a FOA, senior/key personnel are defined as all individuals who contribute
in a substantive, meaningful way to the scientific development or execution of the project, whether or
not salaries are requested. Consultants should be included in this “Senior/Key Person Profile
(Expanded)” Form if they meet this definition.
List individuals that meet the definition of senior/key regardless of what organization they work for.
Profile - Project Director/Principal Investigator
Enter data in this “Profile – Project Director/Principal Investigator” section for the Project
Director/Principal Investigator (PD/PI).
The PD/PI must have an eRA Commons account with the PI role, and the account must be
affiliated with the applicant organization. If you are proposing research at an institute other than
the one you are currently at, do not create a separate Commons account with the proposed
applicant organization. For information on eRA Commons account administration, see the eRA
Account Management System’s Online Help.
F - 48

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.240 - R&RSenior/Key Person Profile (Expanded) Form
Special Instructions for Multiple PD/PIs: When submitting an application involving multiple
PD/PIs, list the “Contact” PD/PI in this field. List all additional PD/PIs in the Senior/Key Person
section(s) below.
Additional Instructions for Fellowship:
For all fellowship applications, the applicant is considered the PD/PI. Therefore, the
applicant must have an eRA Commons account with the PI role, and the account
must be affiliated with the applicant organization. For additional information on
eRA Commons account administration, see the eRA Account Management
System’s Online Help.
If your proposed fellowship is at a different site than your current institution, the
proposed sponsoring institution will be the applicant organization. You must
affiliate your Commons account with the institution so that you have access to
records submitted on your behalf. Do not create a separate Commons account
with the proposed sponsoring institution.
Note that “multiple PD/PIs” are not applicable to fellowship applications, so do not
use the PD/PI role for any other senior/key personnel.
Prefix:
This field may be pre-populated from the SF 424 (R&R) and reflects the prefix, if applicable, for the
name of the PD/PI.
First Name:
This field is required. This field may be pre-populated from the SF 424 (R&R) and reflects the first
(given) name of the PD/PI.
Middle Name:
This field may be pre-populated from the SF 424 (R&R) and reflects the middle name of the PD/PI.
Last Name:
This field is required. This field may be pre-populated from the SF 424 (R&R) and reflects the last
(family) name of the PD/PI.
Suffix:
This field may be pre-populated from the SF 424 (R&R) and reflects the suffix for the name of the
PD/PI.
Position/Title:
This field may be pre-populated from the SF 424 (R&R) and reflects the position/title of the PD/PI.
Department:
This field may be pre-populated from the SF 424 (R&R) and reflects the name of the primary
organizational department, service, laboratory, or equivalent level within the organization of the
PD/PI.
Organization Name:
This field is required. This field may be pre-populated from the SF 424 (R&R) and reflects the
name of the organization of the PD/PI.
F - 49

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.240 - R&RSenior/Key Person Profile (Expanded) Form
Division:
This field may be pre-populated from the SF 424 (R&R) and reflects the name of the primary
organizational division, office, major subdivision, or equivalent level within the organization of
the PD/PI.
Street1:
This field is required. This field may be pre-populated from the SF 424 (R&R) and reflects the first
line of the street address for the PD/PI.
Street2:
This field may be pre-populated from the SF 424 (R&R) and reflects the second line of the street
address for the PD/PI.
City:
This field is required. This field may be pre-populated from the SF 424 (R&R) and reflects the city
for the address of the PD/PI.
County/Parish:
This field may be pre-populated from the SF 424 (R&R) and reflects the county/parish for the
address of the PD/PI.
State:
This field is required if the PD/PI is located in the United States or its Territories. This field may be
pre-populated from the SF 424 (R&R) and reflects the state or territory in which the PD/PI is
located.
Province:
If “Country” is Canada, enter the province for the PD/PI; otherwise, skip the “Province” field. This
field may be pre-populated from the SF 424 (R&R) and reflects the province in which the PD/PI is
located.
Country:
This field may be pre-populated from the SF 424 (R&R) and reflects the country for the address of
the PD/PI.
ZIP/Postal Code:
The ZIP+4 is required if the PD/PI address is in the United States. Otherwise, the postal code is
optional. This field may be pre-populated from the SF 424 (R&R) and reflects the postal code of
the address of the PD/PI.
Phone Number:
This field is required. This field may be pre-populated from the SF 424 (R&R) and reflects the
daytime phone number for the PD/PI.
Fax Number:
This field may be pre-populated from the SF 424 (R&R) and reflects the fax number for the PD/PI.
E-mail:
This field is required. This field may be pre-populated from the SF 424 (R&R) and reflects the e-
mail address for the PD/PI.
F - 50

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.240 - R&RSenior/Key Person Profile (Expanded) Form
Credential, e.g., agency login:
This field is required. Enter the assigned eRA Commons username for the project’s PD/PI. The eRA
Commons username must hold the PI role and be affiliated with the applicant organization.
Applications will not pass agency validation requirements without a valid eRA Commons
username.
Special Instructions for Multiple PD/PI: The Commons username must be provided for
all individuals assigned the Project Role of PD/PI on the application.
Additional Instructions for Fellowship:
Enter the eRA Commons username for the PD/PI (Fellowship candidate). The
eRA Commons Personal Profile associated with the username entered in the
Credential field must include an ORCID ID. For more information on linking an
ORCID ID to an eRA Commons Personal Profile, see the ORCID ID topic in the eRA
Commons online help.
Project Role:
Enter "PD/PI" for the Project Role for the PD/PI.
Other Project Role Category:
Skip the “Other Project Role Category” field, as no other role can be added to the PD/PI role.
Degree Type:
Enter the highest academic or professional degree or other credentials (e.g., R.N.).
Degree Year:
Enter the year the highest degree or other credential was obtained.
Attach Biographical Sketch
Provide a biographical sketch for each PD/PI. See instructions below on how to complete a
biographical sketch.
Attach Current & Pending Support:
Do not use this attachment upload for NIH and other PHS agency submissions unless otherwise
specified in the FOA.
While this information is not required at the time of application submission, it may be requested
later in the pre-award cycle. If and when this occurs, refer to the NIH Grants Policy Statement,
Section 2.5.1: Just-in-Time Procedures.
Instructions for a Biographical Sketch
These instructions apply to Research (R), Career Development (K), Training (T), Fellowship (F),
Multi-project (M), and SBIR/STTR (B).
Who must complete the “Biographical Sketch” section:
All senior/key personnel and other significant contributors (OSCs) must include biographical
sketches (biosketches).
F - 51

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.240 - R&RSenior/Key Person Profile (Expanded) Form
Format:
Use the sample format on the Biographical Sketch Format Page to prepare this section for all
grant applications.
Figures, tables (other than those included in the provided format pages), or graphics are not
allowed in the biosketch. Do not embed or attach files (e.g. video, graphics, sound, data).
The biosketch may not exceed five pages per person. This five-page limit includes the table at the
top of the first page.
Attach this information as a PDFfile. See the Format Attachments page.
Content:
Note that the instructions here follow the format of Biographical Sketch Format Page.
Name:
Fill in the name of the senior/key person or other significant contributor in the “Name” field of
the Biosketch Format Page.
eRA Commons User Name:
If the individual is registered in the eRA Commons, fill in the eRA Commons User Name in the
“eRA Commons User Name” field of the Biosketch Format Page.
The “eRA Commons User Name” field is required for the PD/PI (including career development and
fellowship applicants), primary sponsors of fellowship applicants, all mentors of candidates for
mentored career development awards, and candidates for diversity and reentry research
supplements.
The “eRA Commons User Name” field is optional for other project personnel.
The eRA Commons User Name should match the information provided in the Credential
field of the R&R Senior/Key Person Profile (Expanded) Form in your grant application.
Position Title:
Fill in the position title of the senior/key person or other significant contributor in the “Position
Title” field of the Biosketch Format Page.
Education/Training
Complete the education block. Begin with the baccalaureate or other initial professional
education, such as nursing. Include postdoctoral, residency, and clinical fellowship training, as
applicable, listing each separately.
For each entry provide:
l the name and location of the institution
l the degree received (if applicable)
l the month and year of end date (or expected end date). For fellowship applicants only,
also include the month and year of start date.
l the field of study (for residency entries, the field of study should reflect the area of
residency training)
Following the education block, complete Sections A-D of the biographical sketch.
F - 52

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.240 - R&RSenior/Key Person Profile (Expanded) Form
A. Personal Statement
Briefly describe why you are well-suited for your role(s) in this project. Relevant factors may
include: aspects of your training; your previous experimental work on this specific topic or related
topics; your technical expertise; your collaborators or scientific environment; and/or your past
performance in this or related fields.
You may cite up to four publications or research products that highlight your experience and
qualifications for this project. Research products can include, but are not limited to, audio or
video products; conference proceedings such as meeting abstracts, posters, or other
presentations; patents; data and research materials; databases; educational aids or curricula;
instruments or equipment; models; protocols; and software or netware.
You are allowed to cite interim research products. Note: interim research products have specific
citation requirements. See related Frequently Asked Questions for more information.
Note the following additional instructions for ALL applicants/candidates:
l If you wish to explain factors that affected your past productivity, such as family care
responsibilities, illness, disability, or military service, you may address them in this “A.
Personal Statement” section.
l Indicate whether you have published or created research products under another name.
l You may mention specific contributions to science that are not included in Section C. Do
not present or expand on materials that should be described in other sections of this
Biosketch or application.
l Figures, tables, or graphics are not allowed.
Note the following instructions for specific subsets of applicants/candidates:
l For institutional research training, institutional career development, or research
education grant applications, faculty who are not senior/key persons are encouraged,
but not required, to complete the "A. Personal Statement" section.
l Applicants for dissertation research awards (e.g., R36) should, in addition to addressing
the points noted above, also include a description of their career goals, their intended
career trajectory, and their interest in the specific areas of research designated in the
FOA.
l Candidates for research supplements to promote diversity in health-related research
should, in addition to addressing the points noted above, also include a description of
their general scientific achievements and/or interests, specific research objectives, and
career goals. Indicate any current source(s) of educational funding.
B. Positions and Honors
List in chronological order the positions you’ve held that are relevant to this application,
concluding with your present position. High school students and undergraduates may include
any previous positions. For individuals who are not currently located at the applicant
organization, include the expected position at the applicant organization and the expected start
date.
List any relevant academic and professional achievements and honors. In particular:
F - 53

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.240 - R&RSenior/Key Person Profile (Expanded) Form
l Students, postdoctorates, and junior faculty should include scholarships, traineeships,
fellowships, and development awards, as applicable.
l Clinicians should include information on any clinical licensures and specialty board
certifications that they have achieved.
C. Contributions to Science
Who should complete the “Contributions to Science” section:
All senior/key persons should complete the “Contributions to Science” section except candidates
for research supplements to promote diversity in health-related research who are high school
students, undergraduates, and post-baccalaureates.
Format:
Briefly describe up to five of your most significant contributions to science. The description of
each contribution should be no longer than one half page, including citations.
While all applicants may describe up to five contributions, graduate students and postdoctorates
may wish to consider highlighting two or three they consider most significant.
Content:
For each contribution, indicate the following:
l the historical background that frames the scientific problem;
l the central finding(s);
l the influence of the finding(s) on the progress of science or the application of those
finding(s) to health or technology; and
l your specific role in the described work.
For each contribution, you may cite up to four publications or research products that are
relevant to the contribution. If you are not the author of the product, indicate what your role or
contribution was. Note that while you may mention manuscripts that have not yet been accepted
for publication as part of your contribution, you may cite only published papers to support each
contribution. Research products can include audio or video products (see the NIH Grants Policy
Statement, Section 2.3.7.7: Post-Submission Grant Application Materials); conference
proceedings such as meeting abstracts, posters, or other presentations; patents; data and
research materials; databases; educational aids or curricula; instruments or equipment; models;
protocols; and software or netware.
You are allowed to cite interim research products. Note: interim research products have specific
citation requirements. See related Frequently Asked Questions for more information.
You may provide a URL to a full list of your published work. This URL must be to a Federal
Government website (a .gov suffix). NIH recommends using My Bibliography. Providing a URL to a
list of published work is not required.
Descriptions of contributions may include a mention of research products under development,
such as manuscripts that have not yet been accepted for publication. These contributions do not
have to be related to the project proposed in this application.
F - 54

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.240 - R&RSenior/Key Person Profile (Expanded) Form
D. Additional Information: Research Support and/or Scholastic Performance
Note the following instructions for specific subsets of applicants/candidates:
l High school students are not required to complete Section D. Additional Information:
Research Support and/or Scholastic Performance.
l Career development award applicants should complete the "Research Support" section
but skip the “Scholastic Performance” section.
l Generally, the following types of applicants can skip the “Research Support” section and
must complete only the “Scholastic Performance” section. However, when these
applicants also have Research Support, they may complete both sections.
o
applicants for predoctoral and postdoctoral fellowships
o
applicants to dissertation research grants (e.g., R36)
o
candidates for research supplements to promote diversity in health-related research
from the undergraduate through postdoctoral levels
Research Support
These instructions apply to all applicants who are completing the “Research Support” section.
List ongoing and completed research projects from the past three years that you want to draw
attention to. Briefly indicate the overall goals of the projects and your responsibilities. Do not
include the number of person months or direct costs.
Do not confuse “Research Support” with “Other Support.” Other Support information is not
collected at the time of application submission.
l Research Support: As part of the Biosketch section of the application, “Research
Support” highlights your accomplishments, and those of your colleagues, as scientists.
This information will be used by the reviewers in the assessment of each your
qualifications for a specific role in the proposed project, as well as to evaluate the overall
qualifications of the research team.
l Other Support: NIH staff may request complete and up-to-date “other support”
information from you as part of Just-in-Time information collection.
Scholastic Performance
Predoctoral applicants/candidates (including undergraduates and post-baccalaureates): List
by institution and year all undergraduate and graduate courses, with grades. In addition, explain
any grading system used if it differs from a 1-100 scale; an A, B, C, D, F system; or a 0-4.0 scale.
Also indicate the levels required for a passing grade.
Postdoctoral applicants: List by institution and year all graduate scientific and/or professional
courses with grades. In addition, explain any grading system used if it differs from a 1-100 scale;
an A, B, C, D, F system; or a 0-4.0 scale. Also indicate the levels required for a passing grade.
Profile – Senior/Key Person 1
Enter data in this “Profile – Senior/Key Person 1” section to provide information on a senior/key
person (other than the PD/PI listed above), if applicable.
F - 55

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.240 - R&RSenior/Key Person Profile (Expanded) Form
Format:
List all senior/key person profiles, followed by other significant contributors (OSC) profiles.
Content – Who to include in the “Profile – Senior/Key Person” section:
Senior/Key Persons: Fill in a separate “Profile – Senior/Key Person” block for each senior/key
personnel. Those with a postdoctoral role should be included if they meet the NIH Glossary
definition of senior/key personnel. A biosketch is required for all senior/key persons.
Other Significant Contributors: Also use the “Profile – Senior/Key Person” section to list any
other significant contributors (OSCs). Consultants should be included if they meet the NIH
Glossary definition of OSC. OSCs should be listed after all other senior/key persons.
A biosketch is required for all OSCs. The biosketch should highlight the OSC’s accomplishments
as a scientist. Reviewers assess these pages during peer review. For more information on review
criteria, see the Review Criteria at a Glance document. Although Other Support information is
required as a just-in-time submission, Other Support information will NOT be required or
accepted for OSCs since considerations of overlap do not apply to these individuals.
Should the level of involvement increase for an individual listed as an OSC, thus requiring
measurable effort on the award, the individual must be redesignated as “senior/key personnel.”
This change must be made before any compensation is charged to the project.
For more information:
For more information, refer to these NIHSenior/Key Personnel Frequently Asked Questions.
Additional Instructions for Fellowship:
Who to include in the “Profile – Senior/Key Person” information section:
Fellowship awards require a primary sponsor, and there may also be co-sponsor(s).
Sponsors and co-sponsors should be identified as senior/key personnel, even if
they are not committing any specified measurable effort to the proposed project,
and they must provide an eRA Commons username.
In addition to involving sponsors and co-sponsors, fellowship applications may
also involve collaborators, consultants, advisory committee members, and
contributors. These individuals are usually not considered senior/key personnel
unless they contribute in a substantive, meaningful way to the project. In
determining which individuals to identify as “Senior/Key,” applicants may wish to
keep in mind that those listed as senior/key personnel on the application should
not be asked to provide reference letters, as such letters are expected to be from
individuals not directly involved in the application. For more information, see NIH's
Reference Letters page.
Prefix:
Enter or select the prefix, if applicable, for the name of the senior/key person.
First Name:
This field is required. Enter the first (given) name of the senior/key person.
Middle Name:
Enter the middle name of the senior/key person.
F - 56

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.240 - R&RSenior/Key Person Profile (Expanded) Form
Last Name:
This field is required. Enter the last (family) name of the senior/key person.
Suffix:
Enter or select the suffix, if applicable, for the senior/key person.
Position/Title:
Enter the position/title of the senior/key person.
Department:
Enter the name of the primary organizational department, service, laboratory, or equivalent level
within the organization of the senior/key person.
Organization Name:
This field is required. Enter the name of the organization of the senior/key person.
Division:
Enter the name of the primary organizational division, office, major subdivision, or equivalent
level within the organization of the senior/key person.
Street1:
This field is required. Enter the first line of the street address for the senior/key person.
Street2:
Enter the second line of the street address for the senior/key person.
City:
This field is required. Enter the city for the address of the senior/key person.
County/Parish:
Enter the county/parish for the address of the senior/key person.
State:
This field is required if the Senior/Key person is located in the United States or its Territories.
Enter the state or territory where the senior/key person is located.
Province:
If “Country” is Canada, enter the province for the senior/key person; otherwise, skip the
“Province” field.
Country:
This field is required. Select the country for the address of the senior/key Person.
ZIP/Postal Code:
The ZIP+4 is required if the Senior/Key Person is in the United States. Otherwise, the postal code
is optional. Enter the ZIP+4 (nine-digit postal code) or postal code of the senior/key person.
Phone Number:
This field is required. Enter the daytime phone number for the senior/key person.
Fax Number:
Enter the fax number for the senior/key person.
F - 57
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.240 - R&RSenior/Key Person Profile (Expanded) Form
E-mail:
This field is required. Enter the e-mail address for the senior/key person.
Credential, e.g., agency login:
If you have an established eRA Commons personal profile, enter the senior/key person's
username. If you do not have an eRA Commons personal profile, skip the “Credential” field.
Additional Instructions for Fellowship:
For senior/key person who are the primary sponsor, an eRA Commons username
must be provided in the “Credential” field. For more information, see the NIH Guide
Notice on eRA Commons Username Requirements for Sponsors of Fellowship
Applications.
Project Role:
Select a project role. Use "Other (Specify)" if the desired category is not available.
Special Instructions for Multiple PD/PIs:All PD/PIs must be assigned the “PD/PI” role,
even those at organizations other than the applicant organization. The role of “Co-PD/PI”
is not currently used by NIH or other PHS agencies to designate a multiple PD/PI
application. In order to avoid confusion, do not use the role of “Co-PD/PI.”
Note on OSCs: For OSCs, enter “Other (Specify)” for the “Project Role” field, and enter “Other
Significant Contributor” in the “Other Project Role Category” field.
Additional Instructions for Fellowship:
For sponsors and co-sponsors, enter “Other (Specify)” for the “Project Role” field,
and enter the appropriate role (e.g., Sponsor) in the “Other Project Role Category”
field.
“Multiple PD/PIs” are not applicable to fellowship applications. The PD/PI role must
be used only for the applicant and not for any other senior/key personnel.
Other Project Role Category:
Complete this field (e.g., Engineer, Chemist, Sponsor, Mentor) if you selected “Other Professional”
or “Other (Specify)” in the “Project Role” field.
Degree Type:
Enter the highest academic or professional degree or other credentials (e.g., R.N.).
Degree Year:
Enter the year the highest degree or other credential was obtained.
Attach Biographical Sketch:
Provide a biographical sketch for each senior/key person and each OSC. See instructions above
on how to complete a biographical sketch.
Attach Current & Pending Support:
Note: The terms “current and pending support,” “other support,” and “active and pending
support” are used interchangeably.
F - 58

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.240 - R&RSenior/Key Person Profile (Expanded) Form
Do not use the "Current &Pending Support" attachment upload for NIH or other PHS agency
submissions unless otherwise specified in the FOA.
While this information is not required at the time of application submission, it may be requested
later in the pre-award cycle. If and when this occurs, refer to the NIH Grants Policy Statement,
Section 2.5.1: Just-in-Time Procedures for instructions and use the Current and Pending Support
Format Page.
Additional Senior/Key Person Profile(s)
If you need to add more Senior/Key Person Profiles than the form allows, enter the information in
a separate file and attach it as a PDF.
A format page for Additional Senior/Key Person Profiles can be found at NIH's Additional
Senior/Key Person Form page.
F - 59

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
F.430 - PHS Fellowship Supplemental
Form
The PHS Fellowship Supplemental Form is used only for
fellowship applications.
This form includes fields to upload several attachments
including the Specific Aims, Research Strategy, and
Applicant Background and Goals.
The attachments in this form, together with the rest of
your application, should include sufficient information
needed for evaluation of the project and fellow,
independent of any other documents (e.g., previous
application). Be specific and informative, and avoid
redundancies.
View larger image
Quick Links
Introduction
1. Introduction to Application (for Resubmission applications)
Fellowship Applicant Section
2. Applicant's Background and Goals for Fellowship Training
Research Training Plan Section
3. Specific Aims
4. Research Strategy
5. Respective Contributions
6. Selection of Sponsor and Institution
7. Progress Report Publication List (for Renewal applications)
8. Training in the Responsible Conduct of Research
Sponsor(s), Collaborator(s), and Consultant(s) Section
9. Sponsor and Co-Sponsor Statements
10. Letters of Support from Collaborators, Contributors, and Consultants
Institutional Environment and Commitment to Training Section
11. Description of Institutional Environment and Commitment to Training
12. Description of Candidate’s Contribution to Program Goals
Other Research Training Plan Section
Vertebrate Animals
F - 60

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
13. Are vertebrate animals euthanized?
14. Vertebrate Animals
15. Select Agent Research
16. Resource Sharing Plan
17. Authentication of Key Biological and/or Chemical Resources
Additional Information Section
18. Human Embryonic Stem Cells
19. Alternate Phone Number
20. Degree Sought During Proposed Award
21. Field of Training for Current Proposal
22. Current or Prior Kirschstein-NRSA Support?
23. Applications for Concurrent Support?
24. Citizenship
25. Change of Sponsoring Institution
Budget Section
26. Tuition and Fees
27. Present Institutional Base Salary
28. Stipends/Salary During First Year of Proposed Fellowship
Appendix
29. Appendix
Who should use the PHS FellowshipSupplemental Form:
Use the PHS Fellowship Supplemental Form only if you are submitting a fellowship application.
Fellowship applicants and sponsors are strongly encouraged to speak with a PHS Program Official for
Institute- or Center (IC)-specific guidance before preparing this application. Refer to the Table of IC-
specific Information, Requirements, and Staff Contacts in your FOA. In addition, a list of contacts
specifically for extramural training at the NIH ICs can be found at NIH Training Advisory Committee
Roster. For AHRQ, see Research Training Staff Contacts. You are encouraged to check these websites
for the most current contact information.
It is important that the attachments in this form be developed in collaboration with your sponsor, but
they should be written by you, the fellowship applicant.
Read all the instructions in the FOA before completing this section to ensure that your application
meets all IC-specific criteria.
F - 61

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
Applicants must follow all policies and requirements related to formatting, page limits, and
proprietary information. See the following pages for more information:
l Format Attachments
l Page Limits
l NIH Grants Policy Statement, Section 2.3.11.2: Confidentiality of Information
l NIH Grants Policy Statement, Section 2.3.11.2.2: The Freedom of Information Act
Introduction
1. Introduction to Application (for Resubmission applications)
Who must complete the “Introduction to Application” attachment:
An "Introduction" attachment is required only if the type of application is resubmission or if the
FOA specifies that one is needed. An introduction is not allowed for new or renewal applications.
Descriptions of different types of applications are listed here: NIH Types of Applications.
Format:
Follow the page limits for the Introduction in the NIH Table of Page Limits unless otherwise
specified in the FOA.
Attach this information as a PDF file. See NIH's Format Attachments page.
Content:
Resubmission applications: See specific instructions on the content of the Introduction on the
NIH's Resubmission Applications page.
Fellowship Applicant Section
2. Applicant’s Background and Goals for Fellowship Training
Who must complete the "Applicant's Background and Goals for Fellowship Training"
attachment:
The “Applicant’s Background and Goals for Fellowship Training” attachment is required.
Format:
Follow the page limits for Applicant’s Background and Goals for Fellowship Training in the NIH
Table of Page Limits unless otherwise specified in the FOA.
Attach this information as a PDF file. See NIH's Format Attachments page.
Content:
Organize the Applicant’s Background and Goals for Fellowship Training attachment in the
specified order and use the instructions provided below unless otherwise specified in the FOA.
F - 62

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
Start each section with the appropriate heading - Doctoral Dissertation and Research Experience,
Training Goals and Objectives, and Activities Planned Under this Award.
A. Doctoral Dissertation and Research Experience
Briefly summarize your past research experience, results, and conclusions, and describe how
that experience relates to the proposed fellowship. In some cases, a proposed fellowship
may build directly on previous research experiences, results, and conclusions. In other
situations, past research experiences may lead a candidate to apply for a fellowship in a new
or different area of research. Do not list academic courses in this section.
Applicants with no research experience: Describe any other scientific experiences.
Advanced graduate students (i.e., those who have or will have completed their
comprehensive examinations by the time of award): Include a narrative of your planned
doctoral dissertation (may be preliminary).
Postdoctoral fellowship applicants: Specify which areas of your proposed research were
part of your predoctoral thesis or dissertation and which, if any, were part of a previous
postdoctoral project.
B. Training Goals and Objectives
l Describe your overall training goals for the duration of the fellowship and how the
proposed fellowship will enable the attainment of these goals.
l Identify the skills, theories, conceptual approaches, etc., to be learned or enhanced
during the award, including, as applicable, expertise in rigorous research design,
experimental methods, quantitative approaches, and data analysis and interpretation, as
applicable.
l Discuss how the proposed research will facilitate your transition to the next career stage.
C. Activities Planned Under this Award
The activities planned under this award should be individually tailored and well-integrated
with your research project.
l Describe, by year, the activities (research, coursework, professional development, clinical
activities, etc.) you will be involved in during the proposed award. Estimate the
percentage of time to be devoted to each activity. The percentage should total 100 for
each year.
l Describe the research skills and techniques that you intend to learn during the award
period.
l Provide a timeline detailing the proposed research training, professional development,
and clinical activities for the duration of the fellowship award. Detailed timelines of
research activities involving animals, human subjects, or clinical trials are requested in
other sections of the fellowship application and should not be included here. The
timeline you provide here should be distinct from the Study Timeline in the PHS Human
Subjects and Clinical Trials Information form.
F - 63

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
Research Training Plan Section
A Research Training Plan is required for all types of fellowship awards and is a major part of the
fellowship application. It is important to relate the proposed research to the applicant's scientific
career goals. Explain the relationship between the applicant’s research on the fellowship award
and the sponsor’s ongoing research program.
The information in these introductory paragraphs to the Research Training Plan Section applies
to all Research Training Plan Section attachments: Specific Aims, Research Strategy, Respective
Contributions, Selection of Sponsor and Institution, Progress Report Publication List, and
Training in the Responsible Conduct of Research.
For most types of research, the plan should include:
l a specific hypothesis,
l a list of the specific aims and objectives that will be used to examine the hypothesis,
l a description of the methods/approaches/techniques to be used in each aim,
l a discussion of possible problems and how they will be managed, and
l alternative approaches that might be tried if the initial approaches do not work.
The Research Training Plan is expected to be tailored to the experience level of the applicant and
to allow him/her to develop the necessary skills for further career advancement. Reviewers will
evaluate the plan accordingly. The plan should be achievable within the requested time period.
Pilot or preliminary studies and routine data gathering are generally not appropriate as the focus
of the Research Training Plan.
Although applicants for fellowship awards are expected to write the Research Training Plan, the
sponsor should review a draft of the plan and discuss it in detail with the applicant. Review by
other knowledgeable colleagues is also helpful. Although it is understood that fellowship
applications do not require the extensive experimental detail usually incorporated into regular
research grant applications, a fundamentally sound Research Training Plan should be provided.
3. Specific Aims
Who must complete the "Specific Aims" attachment:
The “Specific Aims” attachment is required unless otherwise specified in the FOA.
Format:
Follow the page limits for Specific Aims in the NIH Table of Page Limits, unless otherwise specified
in the FOA. A “Specific Aims” attachment that includes graphics will generate a warning by the
Agency upon submission.
Attach this information as a PDF. See NIH's Format Attachments page.
Content:
State concisely the goals of the proposed research and summarize the expected outcome(s),
including the impact that the results of the proposed research will have on the research field(s)
involved.
F - 64

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
List succinctly the specific objectives of the research proposed (e.g., to test a stated hypothesis,
create a novel design, solve a specific problem, challenge an existing paradigm or clinical practice,
address a critical barrier to progress in the field, or develop new technology).
4. Research Strategy
Who must complete the "Research Strategy" attachment:
The “Research Strategy” attachment is required.
Format:
Follow the page limits for the Research Strategy in the NIH Table of Page Limits unless otherwise
specified in the FOA. Although multiple sections of information are required in the Research
Strategy as detailed below, the page limit applies to the entirety of the single Research Strategy
attachment.
Attach this information as a PDF file. See NIH's Format Attachments page.
Content:
Organize the Research Strategy in the specified order and use the instructions provided below,
unless otherwise specified in the FOA. Start each section with the appropriate section heading –
Significance, Approach, etc. Cite published experimental details in the Research Strategy and
provide the full reference in F.220 - R&R Other Project Information Form, Bibliography and
References Cited.
Note for Applications Proposing the Involvement of Human Subjects and/or Clinical Trials:
l Use the Research Strategy section to discuss the overall strategy, methodology, and analyses
of your proposed research, but do not duplicate information collected in the PHS Human
Subjects and Clinical Trials Information form.
l The PHS Human Subjects and Clinical Trials Information form will capture detailed study
information, including eligibility criteria; inclusion of women, minorities, and children; and
protection and monitoring plans.
l You are encouraged to refer to information in the PHS Human Subjects and Clinical Trials
Information form as appropriate in your discussion (e.g., see Question 2.4 Inclusion of Women
and Minorities).
Note for Applicants with Multiple Specific Aims: you may address the Significance and
Approach either for each Specific Aim individually or for all of the Specific Aims collectively.
1. Significance
l Explain the importance of the problem or critical barrier to progress that the proposed
project addresses.
l Describe the strengths and weaknesses in the rigor of the prior research (both
published and unpublished) that serves as the key support for the proposed project.
l Explain how the proposed project will improve scientific knowledge, technical capability,
and/or clinical practice in one or more broad fields.
l Describe how the concepts, methods, technologies, treatments, services, or preventative
interventions that drive this field will be changed if the proposed aims are achieved.
F - 65

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
2. Approach
l Describe the overall strategy, methodology, and analyses to be used to accomplish the
specific aims of the project. Describe plans to address weaknesses in the rigor of the
prior research that serves as the key support for the proposed project. Describe the
experimental design and methods proposed and how they will achieve robust and
unbiased results. Unless addressed separately in the Resource Sharing Plan attachment,
include how the data will be collected, analyzed, and interpreted, as well as any resource
sharing plans, as appropriate. Resources and tools for rigorous experimental design can
be found at the Enhancing Reproducibility through Rigor and Transparency website.
l For trials that randomize groups or deliver interventions to groups, describe how your
methods for analysis and sample size are appropriate for your plans for participant
assignment and intervention delivery. These methods can include a group- or cluster-
randomized trial or an individually randomized group-treatment trial. Additional
information is available at the Research Methods Resources webpage.
l Discuss potential problems, alternative strategies, and benchmarks for success
anticipated to achieve the aims.
l If the project is in the early stages of development, describe any strategy to establish
feasibility, and address the management of any high risk aspects of the proposed work.
l Explain how relevant biological variables, such as sex, are factored into research designs
and analyses for studies in vertebrate animals and humans. For example, strong
justification from the scientific literature, preliminary data, or other relevant
considerations, must be provided for applications proposing to study only one sex. Refer
to NIH Guide Notice on Sex as a Biological Variable in NIH-funded Research for
additional information.
l Point out any procedures, situations, or materials that may be hazardous to personnel
and the precautions to be exercised. If applicable, a full discussion on the use of select
agents should appear in the Select Agent Research attachment below.
l If research on Human Embryonic Stem Cells (hESCs) is proposed, but an approved cell
line from the NIH hESC Registry cannot be chosen, provide a strong justification for why
an appropriate cell line cannot be chosen from the registry at this time.
l If you are proposing to gain clinical trial research experience (i.e., you will not be leading
an independent clinical trial), briefly describe your role on the clinical trial.
As applicable, also include the following information as part of the Research Strategy,
keeping within the two sections (Significance and Approach) listed above.
Preliminary Studies for New Applications:
For new applications, include information on preliminary studies (including data collected by
others in the lab), if any. Discuss the applicant's preliminary studies, data, and/or experience
pertinent to this application.
Progress Report for Renewal Applications:
Renewal applications for individual Fellowships are rare. You should consult with your
program official before preparing such an application.
Note that the Progress Report falls within the Research Strategy and is therefore included in
the page limits for the Research Strategy. If you are submitting a renewal application,
F - 66

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
provide a Progress Report. Provide the beginning and ending dates for the period covered
since the last competitive review. In the Progress Report, you should:
l Summarize the specific aims of the previous project period and the importance of the
findings, and emphasize the progress made toward their achievement.
l Explain any significant changes to the specific aims and any new directions, including
changes resulting from significant budget reductions.
l Discuss previous participant enrollment (e.g., recruitment, retention, inclusion of
women, minorities, children, etc.) for any studies meeting the NIH Glossary definition
for clinical research. Use the Progress Report section to discuss, but do not duplicate
information collected elsewhere in the application.
Do not include a list of publications, manuscripts accepted for publication, patents, or other
printed materials in the Progress Report. That information will be included in the "Progress
Report Publication List" attachment.
5. Respective Contributions
Who must complete the "Respective Contributions" attachment:
The “Respective Contributions” attachment is required.
Format:
Follow the page limits for Respective Contributions in the NIH Table of Page Limits unless
otherwise specified in the FOA.
Attach this information as a PDF file. See NIH's Format Attachments page.
Content:
Describe the collaborative process between you and your sponsor/co-sponsor(s) in the
development, review, and editing of this Research Training Plan. Also discuss your respective
roles in accomplishing the proposed research.
6. Selection of Sponsor and Institution
Who must complete the "Selection of Sponsor and Institution" attachment:
The “Selection of Sponsor and Institution” attachment is required.
Format:
Follow the page limits for Selection of Sponsor and Institution in the NIH Table of Page Limits
unless otherwise specified in the FOA.
Attach this information as a PDF file. See NIH's Format Attachments page.
Content:
Describe the rationale/justification for the selection of both the sponsor and the institution.
1. Explain why the sponsor, co-sponsor (if any), and institution were selected to accomplish
the research training goals. If the proposed research training is to take place at a site
other than the applicant organization, provide an explanation here.
F - 67

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
2. Foreign Institution: If you are proposing a research training experience at a foreign
institution, describe how the foreign institution and sponsor offer special opportunities
for training that are not currently available in the United States. Key factors in the
selection of a foreign institution should be described. The need for and level of
proficiency in reading, speaking, and comprehending the foreign language should be
addressed.
3. Postdoctoral and Senior Fellowship Applicants requesting training at their
Doctorate or Current Institution: Training is expected to broaden a fellow's perspective.
Therefore, if you are requesting training at either your doctorate institution or any
institution where you have been training for more than a year, you must explain why
further training at that institution would be valuable. Individuals applying for senior
fellowships who are requesting training at the institution at which they are employed
should provide a similar explanation.
7. Progress Report Publication List (for Renewal applications)
Who must complete the “Progress Report Publication List” attachment:
A “Progress Report Publication List” is required only if the type of application is renewal.
Descriptions of different types of applications are listed here: NIH Types of Applications.
Format:
Attach this information as a PDF file. See NIH's Format Attachments page.
Content:
In the rare instance that you are submitting a renewal application, list the titles and complete
references to all appropriate publications, manuscripts accepted for publication, patents, and
other printed materials that have resulted from the project since it was last reviewed
competitively.
You are allowed to cite interim research products. Note: Interim research products have specific
rules and citation requirements. See related Frequently Asked Questions on citing interim
research products and claiming them as products of your NIH award.
Provide the NIH Manuscript Submission reference number (e.g., NIHMS97531) or the PubMed
Central (PMC) reference number (e.g., PMCID234567) for the following types of articles:
l Articles that fall under the Public Access Policy;
l Articles that were authored or co-authored by the fellowship applicant and arose from
NIH support;
l Articles that were authored or co-authored by the fellowship applicant and arose from
AHRQ funding provided after February 19, 2016 (see Guide Notice on Policy for Public
Access to AHRQ-Funded Scientific Publications).
If the PMCID is not yet available because the Journal submits articles directly to PMC on behalf of
their authors, indicate “PMC Journal – In Process.” NIHmaintains a list of such journals.
Citations that are not covered by the NIH Public Access Policy, but are publicly available in a free,
online format may include URLs or PMCID numbers along with the full reference. Note that copies
of these publications are not accepted as appendix material.
F - 68

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
8. Training in the Responsible Conduct of Research
Who must complete the "Training in the Responsible Conduct of Research" attachment:
The “Training in the Responsible Conduct of Research” attachment is required.
Format:
Follow the page limits for Training in the Responsible Conduct of Research in the NIH Table of
Page Limits unless otherwise specified in the FOA.
Attach this information as a PDF file. See NIH's Format Attachments page.
Content:
The plan must address the five required instructional components outlined in the NIH Policy on
Instruction in the Responsible Conduct of Research (RCR), as more fully described in the NIH
Grants Policy Statement, Section 11.2.3.4: Responsible Conduct of Research:
1. Format: Describe the required format of instruction (i.e., face-to-face lectures,
coursework, and/or real-time discussion groups). A plan with only on-line instruction is
not acceptable.
2. Subject Matter: Describe the breadth of subject matter (e.g., conflict of interest,
authorship, data management, human subjects and animal use, laboratory safety,
research misconduct, and research ethics).
3. Faculty Participation: Describe the role of the sponsor/mentor(s) and other faculty
involvement in the instruction.
4. Duration of Instruction: Describe the total number of contact hours of instruction,
taking into consideration the duration of the program.
5. Frequency of Instruction: Instruction must occur during each career stage and at least
once every four years. Document any prior instruction during the applicant’s current
career stage, including the inclusive dates instruction was last completed.
Senior fellows may fulfill the requirement for instruction in RCR by participating as lecturers and
discussion leaders.
For more information:
See the NIH Grants Policy Statement, Section 11.2.3.4: Responsible Conduct of Research.
Sponsor(s), Collaborator(s), and Consultant(s) Section
9. Sponsor and Co-Sponsor Statements
Who must complete the “Sponsor and Co-Sponsor Statement” attachment:
The “Sponsor and Co-Sponsor Statement” attachment is required. The sponsor and each co-
sponsor must provide statements as described below.
F - 69

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
Format:
Follow the page limits for Sponsor and Co-Sponsor Statements in the NIH Table of Page Limits
unless otherwise specified otherwise in the FOA.
The Sponsor and Co-Sponsor Statements must be appended together and uploaded as a single
PDF file. See NIH's Format Attachments page.
Content:
Complete these items as comprehensively as possible so that a meaningful evaluation of the
training environment can be made by the reviewers.
Create a heading at the top of the first page titled “Sponsor and Co-Sponsor Statements.”
Organize each statement in the specified order and use the instructions below, unless otherwise
specified in the FOA. Start each section with the appropriate section heading – Research Support
Available; Sponsor’s/Co-Sponsor’s Previous Fellows/Trainees; Training Plan, Environment,
Research Facilities; Number of Fellows/Trainees to be Supervised During the Fellowship; and
Applicant’s Qualifications and Potential for a Research Career.
Each sponsor and co-sponsor statement must address all of the following sections (A-E).
A. Research Support Available
In a table, list all current and pending research and research training support specifically
available to the applicant for this particular training experience. Include funding source,
complete identifying number, title of the research or training program, name of the PD/PI,
start and end dates, and the amount of the award. If the sponsor’s research support will
end prior to the end of the proposed training period, the sponsor should describe a
contingency plan for how the fellow’s research will be supported.
The role of the sponsor/co-sponsor in the Research Training Plan should be described. If
one or more co-sponsors is proposed, this plan should describe the role of each sponsor
and how they will communicate and coordinate their efforts to mentor the applicant
effectively.
B. Sponsor's/Co-Sponsor’s Previous Fellows/Trainees
State the total number of predoctoral and postdoctoral individuals previously sponsored.
Select up to five that are representative, and for those five, provide information on their time
spent in the lab, their present employing organizations, and their present position titles or
occupations.
C. Training Plan, Environment, Research Facilities
The applicant’s Research Training Plan should be individualized for the applicant, keeping in
mind the candidate’s strengths and any gaps in needed skills. The Research Training Plan
should be designed to enhance both research and clinical training (if applicable).
Describe the Research Training Plan that you have developed specifically for the fellowship
applicant. Be sure to include the following points:
l Include items such as classes, seminars, opportunities for interaction with other
groups and scientists, and any professional skills development opportunities.
l Describe the research environment and available research facilities and equipment.
l Indicate the relationship of the proposed research training to the applicant's career
goals.
F - 70

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
l Describe the skills and techniques that the applicant will learn. Relate these to the
applicant's career goals.
The information contained in the “Training Plan, Environment, Research Facilities” section of
the Sponsor’s and Co-sponsors’ Statements should be coordinated with information
provided under the Description of Institutional Environment and Commitment to Training
attachment below.
F30 Applications: The Research Training Plan should provide opportunities to integrate
clinical experiences during the research training component; a plan for a smooth transition
to the clinical training component; and should have the potential to facilitate the applicant’s
transition to a residency or other program appropriate for his/her career goals. Sponsors
and co-sponsors should discuss these clinical aspects of the applicant’s training as well.
F31, F32, F33 Applications: The Research Training Plan should facilitate the applicant's
transition to the next stage of his/her career. Sponsors and co-sponsors should discuss this
aspect of the applicant's training as well.
D. Number of Fellows/Trainees to be Supervised During the Fellowship
Indicate how many pre- and/or post- doctoral fellows/trainees the Sponsor/Co-sponsor is
expected to supervise during the award period. Co-sponsor statements must also include
this information.
E. Applicant's Qualifications and Potential for a Research Career
Describe how the fellowship applicant is suited for this research training opportunity based
on his/her academic record and research experience level. Include information about how
the Research Training Plan, and your own expertise as the sponsor or co-sponsor, will assist
in producing an independent researcher.
Note: If the applicant is proposing to gain experience in a clinical trial as part of his or her
research training, then the sponsor or co-sponsor should include information in the statement
to document leadership of the clinical trial (in addition to the information above). Include the
following:
l Source of funding;
l ClinicalTrials.gov Identifier (e.g., NCT87654321), if applicable;
l A description of how your expertise is appropriate to guide the applicant in any
proposed clinical trials research experience; and
l A statement/attestation that the sponsor will be responsible for the clinical trial.
l The sponsor must have primary responsibility for leading and overseeing the trial and
must describe how he/she will provide this oversight (be careful to not overstate the
fellow’s responsibilities).
l Include details on the specific roles/responsibilities of the fellow and sponsor, keeping in
mind that the terms of a fellowship award do not permit the fellow to lead a clinical trial.
10. Letters of Support from Collaborators, Contributors, and Consultants
Note that Letters of Support are not the same as Reference Letters, which are required for some
fellowship award applications. For more information about Reference Letters see the NIH
Reference Letters page.
F - 71

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
Format:
Follow the page limits for Letters of Support from Collaborators, Contributors, and Consultants
in the NIH Table of Page Limits unless otherwise specified in the FOA.
Letters of support must be appended together and uploaded as a single PDF file. See NIH's
Format Attachments page.
Content:
If any collaborators, consultants, or advisors are expected to contribute to the scientific
development or execution of the fellow’s planned project and research training, attach letters of
support from those individuals here, describing their anticipated role and contributions.
Institutional Environment and Commitment to Training Section
11. Description of Institutional Environment and Commitment to Training
Who must complete the “Description of Institutional Environment and Commitment to
Training” attachment:
The “Description of Institutional Environment and Commitment to Training” attachment is
required, and includes “Educational Information” for F30 and F31 applications.
Format:
Follow the page limits for the Description of Institutional Environment and Commitment to
Training in the NIH Table of Page Limits unless otherwise specified in the FOA.
Attach this information as a PDF file. See NIH's Format Attachments page.
Content:
Document a strong, well-established research program related to the candidate's area of
interest. Describe opportunities for intellectual interactions with other individuals in training and
other investigators, including courses offered, journal clubs, seminars, and presentations.
Indicate the facilities and other resources that will be made available for both career
enhancement and the research proposed in this application. Refer to the resources description
in F.220 - R&R Other Project Information Form, Facilities and Other Resources, and information
provided in the Sponsor and Co-sponsor Statements attachment.
F30 and F31 applications: Educational Information
Describe the institution’s dual-degree (F30) or graduate (F31) program in which the
applicant is enrolled. This description should include the structure of the program, the
required milestones and their usual timing, the number of courses, any teaching
commitments, clinical requirements, qualifying exams, and the average time to degree over
the past 10 years. Describe the progress/status of the applicant in relation to the program’s
timeline, and the frequency and method by which the program formally monitors and
evaluates a student’s progress.
For F30 applications specifically, describe any clinical tutorials during the graduate research
years and any activities to ease transition from the graduate to the clinical years of the dual-
degree program. Describe any research-associated activities during the clinical years of the
dual-degree program.
F - 72

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
Include the name of the individual providing this information at the end of the description.
This information is typically provided by the director of the graduate program or the
department chair.
12. Description of Candidate’s Contribution to Program Goals
Who must complete the "Description of Candidate’s Contribution to Program Goals"
attachment:
Applicants to diversity-related FOAs (e.g., diversity-related F31): The “Description of
Candidate’s Contribution to Program Goals” attachment is required.
All other Fellowship applicants: Skip the “Description of Candidate’s Contribution to Program
Goals” attachment, as it is not required.
Format:
Attach this information as a PDF file. See NIH's Format Attachments page.
Content:
The sponsoring institution must provide a document on institutional letterhead that explains
how the candidate’s participation will further the goals of the fellowship program to promote
diversity in health-related research.
For NIH’s Interest in Diversity, see the Notice of NIH's Interest in Diversity.
Signatures:
The “Description of Candidate’s Contribution to Program Goals” attachment must be dated
and signed by an institutional official. In most cases, this will be the dean or the chairman of
the department. The signature must appear over the signer's name and title at the end of
the statement.
Other Research Training Plan Section
Vertebrate Animals
Are Vertebrate Animals Used?
This field is pre-populated from the F.220 - R&R Other Project Information Form.
If you have answered “No” for activities involving vertebrate animals and activities
involving vertebrate animals are not planned at any time during the proposed project at any
performance site: Skip Questions 13 and 14 below.
If you have answered “Yes” for activities involving vertebrate animals: Answer Questions 13
and 14 below in consultation with both your Sponsor and AO.
13. Are vertebrate animals euthanized?
An answer is required if you answered “Yes” to “Are Vertebrate Animals Used?” above.
Check "Yes" or "No" to indicate whether animals in the project are euthanized.
F - 73

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
If “Yes” to euthanasia, is method consistent with AVMA guidelines?
An answer is required if you answered “Yes” to “Are Vertebrate Animals Euthanized?”
Check “Yes” or “No” to indicate whether the method of euthanasia is consistent with the
American Veterinary Medical Association (AVMA) Guidelines for the Euthanasia of Animals.
For more information: See AVMA Guidelines for the Euthanasia of Animals.
If “No” to AVMA guidelines, describe method and provide scientific justification:
If you answered “No” to “Is method consistent with AVMA guidelines?,” you must describe (in
1000 characters or fewer) the method of euthanasia and provide a scientific justification for its
use.
If you answered “Yes” to “Is method consistent with AVMA guidelines?” skip this question and
scientific justification.
14. Vertebrate Animals
Who must complete the “Vertebrate Animals” attachment:
Include a “Vertebrate Animals” attachment if you answered “Yes” to the question “Are Vertebrate
Animals Used?” on the F.220 - R&R Other Project Information Form.
Format:
Attach this information as a PDF file. See NIH's Format Attachments page.
Do not use the Vertebrate Animals attachment to circumvent the page limits of the Research
Strategy.
Content:
If live vertebrate animals are involved in the project, address each of the following criteria:
1. Description of Procedures: Provide a concise description of the proposed procedures to
be used that involve live vertebrate animals in the work outlined in the “Research
Strategy” attachment. The description must include sufficient detail to allow evaluation
of the procedures. Identify the species, strains, ages, sex, and total numbers of animals
by species, to be used in the proposed work. If dogs or cats are proposed provide the
source of the animals.
2. Justifications: Provide justification that the species are appropriate for the proposed
research. Explain why the research goals cannot be accomplished using an alternative
model (e.g., computational, human, invertebrate, in vitro).
3. Minimization of Pain and Distress: Describe the interventions including analgesia,
anesthesia, sedation, palliative care, and humane endpoints that will be used to minimize
discomfort, distress, pain, and injury.
Each of the criteria must be addressed. Failure to adequately address the criteria may negatively
affect the application's impact score. In addition to the 3 criteria above, you should also:
l Identify all project performance (or collaborating) sites and describe the proposed
research activities with vertebrate animals that will be conducted at those sites.
l Explain when and how animals are expected to be used if plans for the use of animals
have not been finalized.
F - 74

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
See the following pages for more information:
l NIH’s Office of Laboratory Animal Welfare website
l NIH's Vertebrate Animals Section Worksheet
l NIH Grants Policy Statement, Section 4.1.1.1: Animal Welfare Assurance Requirements (an
applicable Animal Welfare Assurance will be required if the grantee institution does not have
one)
15. Select Agent Research
Who must complete the “Select Agent Research” attachment:
Include a “Select Agent Research” attachment if your proposed activities involve the use of select
agents at any time during the proposed project period, either at the applicant organization or at
any performance site.
Format:
Attach this information as a PDF file. See NIH's Format Attachments page.
For more information:
Select agents are hazardous biological agents and toxins that have been identified by HHS or the
U.S. Department of Agriculture (USDA) as having the potential to pose a severe threat to public
health and safety, to animal and plant health, or to animal and plant products. The Centers for
Disease Control and Prevention (CDC) and the Animal APHIS Select Agent Programs jointly
maintain a list of these agents. See the Federal Select Agent Program website.
See also the NIH Grants Policy Statement, Section 4.1.24.1: Public Health Security and Bioterrorism
Preparedness and Response Act (Select Agents).
Content:
Excluded select agents: If the activities proposed in the application involve only the use of a
strain(s) of select agents which has been excluded from the list of select agents and toxins as per
42 CFR 73.3, the select agent requirements do not apply. Use this “Select Agent Research”
attachment to identify the strain(s) of the select agent that will be used and note that it has been
excluded from this list. The CDC maintains a list of exclusions, which is available on the Select
Agents and Toxins Exclusions website.
Applying for a select agent to be excluded: If the strain(s) is not currently excluded from the list
of select agents and toxins but you have applied or intend to apply to HHS for an exclusion from
the list, use this section to indicate the status of your request or your intent to apply for an
exclusion and provide a brief justification for the exclusion.
All applicants proposing to use select agents: Address the following three points for each site
at which select agent research will take place. Although no specific page limitation applies to this
section, be succinct.
1. Identify the select agent(s) to be used in the proposed research.
2. Provide the registration status of all entities* where select agent(s) will be used.
l If the performance site(s) is a foreign institution, provide the name(s) of the country
or countries where select agent research will be performed.
F - 75

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
l *An “entity” is defined in 42 CFR 73.1 as “any government agency (federal, state, or
local), academic institution, corporation, company, partnership, society, association,
firm, sole proprietorship, or other legal entity.”
3. Provide a description of all facilities where the select agent(s) will be used.
l Describe the procedures that will be used to monitor possession, use, and transfer
of select agent(s).
l Describe plans for appropriate biosafety, biocontainment, and security of the select
agent(s).
l Describe the biocontainment resources available at all performance sites.
16. Resource Sharing Plan
Format:
Attach this information as a PDF file. See NIH's Format Attachments page.
Content:
Data Sharing Plan: Investigators seeking $500,000 or more in direct costs (exclusive of
consortium F&A) in any budget period are expected to include a brief 1-paragraph description of
how final research data will be shared, or explain why data-sharing is not possible (for example
human subject concerns, the Small Business Innovation Development Act provisions, etc.).
Specific FOAs may require that all applications include this information regardless of the dollar
level. Applicants are encouraged to read the FOA carefully and discuss their data-sharing plan
with their program contact at the time they negotiate an agreement with the Institute/Center
(IC) staff to accept assignment of their application. For more information, see the NIH Data
Sharing Policy or the NIH Grants Policy Statement, Section 8.2.3.1: Data Sharing Policy.
Sharing Model Organisms: Regardless of the amount requested, all applications where the
development of model organisms is anticipated are expected to include a description of a specific
plan for sharing and distributing unique model organisms or state why such sharing is restricted
or not possible. For more information, see the NIH Grants Policy Statement, Section 8.2.3.2:
Sharing Model Organisms.
Genomic Data Sharing (GDS): Applicants seeking funding for research that generates large-
scale human or non-human genomic data are expected to provide a plan for sharing of these
data. Examples of large-scale genomic data include genome-wide association studies (GWAS),
single nucleotide polymorphisms (SNP) arrays, and genome sequence, transcriptomic,
epigenomic, and gene expression data. Supplemental Information to the NIH GDS provides
examples of genomic research projects that are subject to the Policy. For more information, see
the NIH GDS Policy, the NIH Grants Policy Statement, Section 8.2.3.3: Genomic Data Sharing (GDS)
Policy/Policy for Genome-Wide Association Studies (GWAS), and the GDS website.
Note on GDS: For proposed studies generating human genomic data under the scope of the
GDS Policy, an Institutional Certification may be submitted at the time of application submission,
but it is not required at that time. The Institutional Certification, however, will be requested as
Just-in-Time (JIT) information prior to award. The Institutional Certification, or in some cases, a
Provisional Institutional Certification, must be submitted and accepted before the award can be
issued.
F - 76

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
For more information:
NIH considers the sharing of unique research resources developed through NIH-sponsored
research an important means to enhance the value and further the advancement of the research.
When resources have been developed with NIH funds and the associated research findings
published or provided to NIH, it is important that they be made readily available for research
purposes to qualified individuals within the scientific community. See NIH Grants Policy
Statement, Section 8.2.3: Sharing Research Resources.
17. Authentication of Key Biological and/or Chemical Resources
Format:
Attach this information as a PDF file. See NIH's Format Attachments page.
Content:
If applicable to the proposed science, briefly describe methods to ensure the identity and validity
of key biological and/or chemical resources used in the proposed studies. A maximum of one
page is suggested.
More information:
Key biological and/or chemical resources are characterized as follows:
l Key biological and/or chemical resources may or may not have been generated with NIH
funds and: 1) may differ from laboratory to laboratory or over time; 2) may have qualities
and/or qualifications that could influence the research data; and 3) are integral to the
proposed research. These include, but are not limited to, cell lines, specialty chemicals,
antibodies, and other biologics.
l Standard laboratory reagents that are not expected to vary do not need to be included in the
plan. Examples are buffers and other common biologicals or chemicals.
l See NIH's page on Rigor and Reproducibility for more information.
Additional Information Section
18. Human Embryonic Stem Cells
Use the following instructions to complete the fields in this section.
For additional guidance, see the NIH Grants Policy Statement, Section 4.1.13: Human Stem Cell
Research.
Does the proposed project involve human embryonic stem cells (hESC)?
An answer to this question is required.
If the proposed project involves hESC, check “Yes” and complete the rest of the fields in the
Human Embryonic Stem Cells section.
If the proposed project does not involve hESC, check “No” and skip the rest of fields in the
Human Embryonic Stem Cells section.
F - 77

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
Specific stem cell line cannot be referenced at this time. One from the registry will be used.
If you will use hESC but a specific line from the NIH hESC Registry cannot be chosen at the time of
application submission, check this box. Additionally, provide a strong justification (in the
Research Strategy) for why an appropriate cell line cannot be chosen from the registry at this
time.
If you cannot specify which cell lines will be used at the time of application submission, specific
cell line information will be required as Just-in-Time information prior to award.
Cell Line(s):
List the 4-digit registration number of the specific cell line(s) from the NIH hESC Registry (e.g.
0123). Up to 200 lines can be added.
19. Alternate Phone Number
Enter an alternate phone number (e.g., cell phone) for the fellowship applicant. This should be a
different number than the one provided in the PD/PI contact information in the F.200 - SF424
(R&R) Form.
20. Degree Sought During Proposed Award
Complete the following fields if you will be working toward a degree while receiving fellowship
support.
Degree:
Select the type of degree you will be working toward during the proposed award. If the degree is
not on the drop down menu, please select “OTH: Other.”
If “other,” indicate degree type:
If you selected “OTH: Other” for the “Degree,” indicate the type of degree you will be working
toward during the proposed award.
Expected Completion Date (MM/YYYY):
Enter the expected completion date of the degree sought during the proposed award.
21. Field of Training for Current Proposal
An answer to this field required.
Select a single "Field of Training" code that best describes the proposed area of research training.
This information is used for reporting purposes only and is not used for study section
assignments.
22. Current or Prior Kirschstein-NRSA Support?
Current or Prior Kirschstein-NRSA Support? Yes/No
An answer to this question is required. Check the appropriate box to indicate whether you
currently have or have had prior Kirschstein-NRSA support.
F - 78

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
If yes, identify current and prior Kirschstein-NRSA support below:
Select the appropriate “Level” and “Type” of Kirschstein-NRSA support. “Level” indicates either
predoctoral or postdoctoral level (not the level of experience). “Type” indicates either individual
fellowship or institutional research training grant.
If known, enter the start and end dates (month, day, and year) of the support and the grant
number (e.g., T32 GM123456 or F31 HL345678) of the current and/or prior support.
You may enter up to four separate listings for current and/or prior support.
Note on Kirschstein-NRSA time limits: An individual cannot receive more than five years of
cumulative predoctoral Kirschstein-NRSA support and three years cumulative postdoctoral
Kirschstein-NRSA support (the total of institutional grants and individual fellowships) without a
waiver from the awarding component. The awarding components have different policies on
waiving the statutory limits on support. Therefore, the fellowship applicant must request a waiver
from the probable awarding IC before requesting a period of support that would exceed these
limits. Refer to the Table of IC-specific Information, Requirements, and Staff Contacts in your FOA.
The fellow’s sponsor and AOR must endorse the request. The request must include justification
and specify the amount of additional support for which approval is sought. Individuals seeking
additional support beyond the third year of postdoctoral support are strongly advised to consult
with their awarding IC Program Officer before submitting a waiver request. It is important to read
carefully the applicable FOA that may have an overall approval to exceed these limits (e.g., the F30
programs allow for up to six years of predoctoral support).
If you receive additional Kirschstein-NRSA support while this application is pending, you must
promptly report such information to the awarding component to which this application has been
assigned.
23. Applications for Concurrent Support?
Applications for Concurrent Support? Yes/No
An answer to this question is required. Check the appropriate box to indicate whether the
fellowship applicant has applied or will be applying for other support that would run
concurrently with the period covered by this application.
If yes, describe in an attached file:
Attach this information as a PDF file. See NIH's Format Attachments page.
If you answered “Yes” to the “Applications for Concurrent Support?” question, you must provide
a description of the concurrent support. Include the type, dates, source(s), and amount in the
attachment.
If you receive any support from these other applications while this application is pending, you
must promptly report such information to the awarding component to which this application has
been assigned.
24. Citizenship
Information on Citizenship Requirements for Fellowship Applicants:
Individual Kirschstein-NRSA Fellowship Requirements: To be eligible for a Kirschstein-NRSA
individual fellowship (F30, F31, F32, F33), the fellowship applicant must be a citizen or non-citizen
national of the United States or of its possessions or territories, or must have been lawfully
F - 79

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
admitted to the United States for permanent residence by the time the award is issued.
Individuals on temporary student visas are not eligible for NRSA support unless otherwise
specified in the FOA.
Non-NRSA Requirements: If you are applying for a non-NRSA fellowship program supported by
the NIH for which citizenship or permanent residency is not required (e.g., Fogarty International
Center programs, F99/K00), you must have a valid visa in your possession that allows you to
remain in the United States (or in a foreign research training setting, if applicable) long enough
to be productive on the proposed fellowship project. It is the responsibility of the sponsoring
institution to determine and document in the application that the individual fellowship
applicant’s visa will allow him or her to remain in the proposed research training setting for the
period of time necessary to complete the proposed fellowship. Information may be requested by
the NIH or another PHS Agency prior to issuance of an award.
All Fellowship Applicants:
Check the applicable boxes for the following questions:
U.S. Citizen: U.S. Citizen or Non-Citizen National? Yes/No
Check “Yes” if the candidate is a U.S. Citizen or Non-Citizen national; otherwise check “No.”
Non-Citizen nationals are people who, although not citizens of the United States, owe
permanent allegiance to the United States. They generally are people born in outlying
possessions of the United States (e.g., American Samoa and Swains Island).
If you answered “Yes,” skip the rest of “Question 31. Citizenship” and you can continue with
“Question 32. Change of Sponsoring Institution.”
If you answered “No,” please continue to fill out the rest of “Question 31. Citizenship” following
the instructions below.
If “No” to U.S. Citizen or Non-Citizen National, please select the most appropriate response
from the options provided:
Non-U.S. Citizen With a Permanent U.S. Resident Visa:
Check this box if the fellowship applicant has been lawfully admitted for permanent
residence (i.e., is in the possession of a current and valid Permanent Resident Card or other
legal verification of such status).
A notarized statement will be required before an award is issued. The statement must show
that a licensed notary has seen the fellowship applicant’s valid Permanent Resident Card
(USCIS Form I-551) or other valid verification from the U.S. Immigration and Naturalization
Service of legal admission to the U.S.
Non-U.S. Citizen With a Temporary U.S. Visa:
Check this box if the fellowship applicant currently holds a temporary U.S. visa.
If you are a non-U.S. citizen with a temporary visa applying for an award that requires
permanent residency status, and expect to be granted a permanent resident visa by the start
date of the award, check here:
If the fellowship applicant has applied for permanent residence and expects to hold a
permanent resident visa by the earliest possible start date of the award, please check this
box to indicate that permanent residence status is pending. A notarized statement will be
required as a part of the pre-award process.
F - 80

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
25. Change of Sponsoring Institution
Check this box if you are submitting your application with a change of sponsoring institution. If
the box is checked, you must also provide the name of the former sponsoring institution.
Budget Section
26. Tuition and Fees
Who must complete the “Tuition and Fees” section:
All fellowship applicants must complete this “Tuition and Fees” section.
Content:
Indicate whether funds are being requested for tuition and fees by checking the appropriate box
(“None Requested” or “Funds Requested”).
Predoctoral Fellowship Applicants: List, by year, the estimated costs of tuition and fees.
Postdoctoral and Senior Fellowship Applicants: List, by year, the costs associated with specific
course work (or a degree-granting program, if applicable) that supports the research training
experience and that are identified and described in the “Activities Planned Under this Award”
section of the Applicant’s Background and Goals for Fellowship Training attachment.
For more information:
In accordance with the NIH Grants Policy Statement, Section 11.2.9.4: Institutional Allowance,
funds to offset the costs of health insurance are included in the standard Institutional Allowance,
and are not to be requested as part of Tuition and Fees.
Refer to the NIH Research Training and Career Development website for helpful resources and
FAQs about tuition and fees.
27. Present Institutional Base Salary
Who must complete the “Institutional Base Salary” section:
Only senior fellowship applicants should complete the “Institutional Base Salary” section.
Amount:
Provide your present base salary. The value must be in U.S. dollars.
Academic Period:
Indicate the period of time on which the salary is determined (e.g., academic year of 9 months,
full-time 12 months, etc.).
Number of Months:
Indicate the number of months per year you receive your base salary. The number may not be
more than 12, but may include a decimal to indicate partial months (e.g., 9.5).
F - 81

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
28. Stipends/Salary During First Year of Proposed Fellowship
Who must complete the “Stipends/Salary During First Year of Proposed Fellowship” section:
Only senior fellowship applicants should complete the “Stipends/Salary During First Year of
Proposed Fellowship” section.
a. Federal Stipend Requested: Amount and Number of Months
Enter the amount of the stipend being requested for the initial period of support (i.e., the first
year of proposed fellowship) and the number of months requested.
b. Supplementation from Other Sources: Amount, Number of Months, Type, and Source
Enter the anticipated amount and the number of months (during the first year of the proposed
fellowship) for any stipend/salary supplementation. Also enter the type of supplementation
expected (e.g., sabbatical leave, salary, etc.) and the source of such funding.
Appendix
29. Appendix
Refer to the FOA to determine whether there are any special appendix instructions for your
application. See the updated NIH Guide Notice on the Appendix Policy.
Format:
A maximum of 10 PDF attachments is allowed in the Appendix. If more than 10 allowable
appendix attachments are needed, combine the remaining information into attachment #10.
Use filenames for attachments that are descriptive of the content.
A summary sheet listing all of the items included in the Appendix is encouraged but not required.
When including a summary sheet, it should be included in the first appendix attachment.
Content:
The only allowable appendix materials are:
l Blank data collection forms, blank survey forms, and blank questionnaire forms - or
screenshots thereof
l Simple lists of interview questions
Note: In your blank forms and lists, do not include items such as: data, data
compilations, lists of variables or acronyms, data analyses, publications, manuals,
instructions, descriptions or drawings/figures/diagrams of data collection methods or
machines/devices.
l Blank informed consent/assent forms
l Other items only if they are specified in the FOA as allowable appendix materials
No other items are allowed in the Appendix. Simply relocating disallowed materials to other parts
of the application will result in a noncompliant application.
F - 82

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.430 - PHS Fellowship Supplemental Form
Some FOAs may have different instructions for the Appendix. Always follow the instructions in
your FOA if they conflict with these instructions.
Note: Applications will be withdrawn and not reviewed if they do not follow the appendix
requirements in these instructions or in your FOA.
Information that expands upon or complements information provided in any section of the
application - even if it is not required for the review - is not allowed in the Appendix unless it is
listed in the allowed appendix materials above or in your FOA. For example, do not include
material transfer agreements (MTA) in the Appendix unless otherwise specified in the FOA.
For more information:
l The NIH Guide Notice on Reminder: NIH Applications Must Be Complete and Compliant
With NIH Policy and Application Instructions At Time of Submission.
l Failure of reviewers to address non-required appendix materials in their reviews is not an
acceptable basis for an appeal of initial peer review. For more information, see the NIH
Grants Policy Statement, Section 2.4.2: Appeals of Initial Scientific Review.
l Appendix Policy Frequently Asked Questions
F - 83

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
F.500 - PHS Human Subjects and Clinical
Trials Information
The PHS Human Subjects and Clinical Trials Information
form is used to collect information on human subjects
research, clinical research, and/or clinical trials, including
study population characteristics, protection and
monitoring plans, and a protocol synopsis.
This form accommodates the full spectrum of all types of
clinical trials, including, but not limited to, behavioral,
exploratory/development, mechanistic, pilot/feasibility,
early phase, efficacy, effectiveness, group-randomized,
and others.
Read all the instructions in the Funding Opportunity
Announcement (FOA) before completing this form to
ensure your application meets all IC-specific criteria.
"Section II. Award Information" of the FOA will indicate
whether clinical trials are or are not allowed and
whether clinical trial research experience is or is not
allowed. The designation of your FOA will determine how to use these instructions, and
subsequently, how to fill out this form.
The PHS Human Subjects and Clinical Trials Information form, together with the rest of your
application, should include sufficient information for the evaluation of the project,
independent of any other documents (e.g., previous application). Be specific, describe each
study clearly, and avoid redundancies. Be especially careful to avoid redundancies with your
research strategy.
View larger image
Quick Links
PHS Human Subjects and Clinical Trials Information
Use of Human Specimens and/or Data
If No to Human Subjects
If Yes to Human Subjects
Other Requested Information
Study Record(s)
Delayed Onset Study(ies)
Study Record: PHS Human Subjects and Clinical Trials Information
Section 1 - Basic Information
F - 84

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
1.1 Study Title (each study title must be unique)
1.2 Is this Study Exempt from Federal Regulations?
1.3 Exemption Number
1.4 Clinical Trial Questionnaire
1.5 Provide the ClinicalTrials.gov Identifier (e.g. NCT87654321) for this trial, if applicable.
Section 2 - Study Population Characteristics
2.1 Conditions or Focus of Study
2.2 Eligibility Criteria
2.3 Age Limits
2.3.a Inclusion of Individuals Across the Lifespan
2.4 Inclusion of Women and Minorities
2.5 Recruitment and Retention Plan
2.6 Recruitment Status
2.7 Study Timeline
2.8 Enrollment of First Participant
2.9 Inclusion Enrollment Report(s)
Section 3 - Protection and Monitoring Plans
3.1 Protection of Human Subjects
3.2 Is this a multi-site study that will use the same protocol to conduct non-exempt human
subjects research at more than one domestic site?
3.3 Data and Safety Monitoring Plan
3.4 Will a Data and Safety Monitoring Board be appointed for this study?
3.5 Overall Structure of the Study Team
Section 4 - Protocol Synopsis
4.1 Study Design
4.2 Outcome Measures
4.3 Statistical Design and Power
4.4 Subject Participation Duration
4.5 Will the study use an FDA-regulated intervention?
4.6 Is this an applicable clinical trial under FDAAA?
4.7 Dissemination Plan
Section 5 - Other Clinical Trial-related Attachments
5.1 Other Clinical Trial-related Attachments
F - 85

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
Complete the PHS Human Subjects and Clinical Trials Information form after you have completed the
F.220 - R&R Other Project Information Form.
This form accommodates the full spectrum of all types of clinical trials, including, but not limited to,
exploratory/development, mechanistic, pilot/feasibility, early phase, efficacy, effectiveness, group-
randomized, and others.
Who should use the PHS Human Subjects and Clinical Trials Information form:
The designation of your FOA will determine how to use these instructions, and subsequently, how to
fill out this form.
All applicants must use the PHS Human Subjects and Clinical Trials Information form regardless of your
answer to the question “Are human subjects involved?” on the F.220 - R&R Other Project Information
Form.
Note for studies involving only the secondary use of identifiable biospecimens or data: For
studies where the only involvement of human subjects is the use of identifiable biospecimens or data
originally collected for another purpose, complete the PHS Human Subjects and Clinical Trials
Information form with information specific to the current study and not the original collection unless
the information associated with the original collection is pertinent to the proposed study. If
information about the original collection is necessary, provide context and clearly distinguish between
the current study and historical information.
Using the PHS Human Subjects and Clinical Trials Information form:
Everyone must complete the "Use of Human Specimens and/or Data" section of the PHS Human
Subjects and Clinical Trials Information form. However, your answer to the “Are human subjects
involved?” question will determine which other sections of the PHS Human Subjects and Clinical Trials
Information form you must complete. Once you have completed the "Use of Human Specimens and/or
Data" section, follow instructions on the form that are specific to your answer to the "Are human
subjects involved?" question on the F.220 - R&R Other Project Information Form:
l if you answered "Yes" to the question "Are human subjects involved?" on the F.220 - R&R
Other Project Information Form, see the "If Yes to Human Subjects" section for instructions.
l if you answered "No" to the question "Are human subjects involved?" on the F.220 - R&R
Other Project Information Form, see the "If No to Human Subjects" section for instructions.
The PHS Human Subjects and Clinical Trials Information form allows you to add Study Record(s) and/or
Delayed Onset Study(ies), as applicable.
Within each Study Record, you will add detailed information at the study level. Do not duplicate
studies within your application. Each study within the application should be unique and should have a
unique study title. Each Study Record is divided into numbered sections:
l Section 1 - Basic Information
l Section 2 – Study Population Characteristics (includes Inclusion Enrollment Report)
l Section 3 – Protection and Monitoring Plans
l Section 4 – Protocol Synopsis
l Section 5 – Other Clinical Trial-related Attachments
Note: The PHS Human Subjects and Clinical Trials Information form will capture detailed information at
the study level. Although you are encouraged to refer to information in the PHS Human Subjects and
Clinical Trials Information form in your discussion of the Research Strategy, do not duplicate
F - 86

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
information between the Research Strategy attachment and the PHS Human Subjects and Clinical
Trials Information form.
For more information on what a “study” is for the purposes of the PHS Human Subjects and Clinical
Trials Information form, see the relevant FAQ on the Applying Electronically FAQ page.
The PHS Human Subjects and Clinical Trials Information form is dynamic and may eliminate sections
that are not relevant to your application. The dynamic form behavior may not be enabled on all
submission methods.
Note: Some fields in this form match fields within ClinicalTrials.gov and are identified as such within
these instructions. Additional information about the fields can be found on the ClinicalTrials.gov
Protocol Registration Data Element Definitions website.
Additional Instructions for Fellowship:
Fellowship applicants are permitted to conduct research involving human subjects;
however, they are NOT permitted to lead an independent clinical trial.
For more information, see:
l FAQs on Clinical Trial-specific FOAs, especially the items related to Fellowship
awards:
o
FAQ about whether F awards allow clinical trials
o
FAQ about why Fellows are not allowed to lead an independent clinical trial
o
FAQ about whether there is a list of responsibilities that a Fellow must
assume with a clinical trial research experience
o
FAQ about who is responsible for the conduct of clinical trials proposed in a
Fellowship application
Fellowship applicants who are not proposing a clinical trial: Follow the
standard instructions to complete the PHS Human Subjects and Clinical Trials
Information form.
Fellowship applicants who are proposing to gain clinical trial research
experience under a sponsor’s supervision (i.e., you will not be leading an
independent clinical trial): You will generally follow the standard instructions to
complete the PHS Human Subjects and Clinical Trials Information form, but follow
relevant Fellowship instructions where they are given. Make sure you are applying
to a FOA that allows Clinical Trial Research Experience (this is noted in “Section II.
Award Information” of the FOA). Additionally, the sponsor or co-sponsor is
required to include a statement to document leadership of the clinical trial. The
statement must include the following:
l Source of funding;
l ClinicalTrials.gov identifier (e.g., NCT87654321), if applicable;
l A description of how the sponsor or co-sponsor’s expertise is appropriate to
guide the applicant in any proposed clinical trials research experience; and
l A statement/attestation that the sponsor will be responsible for the clinical trial
F - 87

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
o
The sponsor must have primary responsibility for leading and overseeing
the trial and must describe how she/he will provide this oversight (be
careful not to overstate the fellow’s responsibilities).
o
Include details on the specific roles/responsibilities of the fellow and
sponsor, keeping in mind that the terms of a fellowship award do not
permit the fellow to lead a clinical trial.
This statement must be included in the “Sponsor and Co-Sponsor Statements”
attachment of the F.430 - PHS Fellowship Supplemental Form.
Applicants must follow all policies and requirements related to formatting, proprietary
information, human subjects, and clinical trials. See the following pages for more information:
l Format Attachments
l Rules for Text Fields
l NIH Grants Policy Statement, Section 2.3.11.2: Confidentiality of Information
l NIH Grants Policy Statement, Section 2.3.11.2.2: The Freedom of Information Act
l NIH's Human Subjects Research website
l NIH's Clinical Trials website
Note: There are no page limits for any attachments in the PHS Human Subjects and Clinical Trials
Information form.
PHS Human Subjects and Clinical Trials
Information
Applicants must complete the human subjects questions on the F.220 - R&R Other Project
Information Form prior to completing this form.
Use of Human Specimens and/or Data
Regardless of your answer to the question “Are Human Subjects Involved?” on the F.220
- R&R Other Project Information Form, answer the following question(s) about the use of
human specimens and/or human data.
Does any of the proposed research in the application involve human specimens and/or data?
Select “Yes” or “No” to indicate whether the proposed research involves human specimens
and/or data.
Note: Applications involving the use of human specimens or data may not be considered to be
research involving human subjects, depending on the details of the materials to be used.
Provide an explanation for any use of human specimens and/or data not considered to be human
subjects research.
If you answered “No” to the “Does any of the proposed research in the application involve human
specimens and/or data?” question, you do not need to attach an explanation here.
F - 88

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
If you answered “Yes” to the “Does any of the proposed research in the application involve
human specimens and/or data?” question, you must provide an explanation for any use of
human specimens and/or data not considered to be human subjects research. To help determine
whether your research is classified as human subjects research, refer to the Research Involving
Private Information or Biological Specimens flowchart. For any human specimens and/or data
that is considered human subjects research, you will add a Study Record. Do not duplicate the
information in your explanation in any of your Study Records.
Attach the explanation as a PDF file. See NIH’s Format Attachments page.
This explanation should include:
l information on who is providing the data/biological specimens and their role in the
proposed research;
l a description of the identifiers that will be associated with the human specimens and
data;
l a list of who has access to subjects’ identities; and
l information about the manner in which the privacy of research participants and
confidentiality of data will be protected.
Please complete the human subjects section of the Research & Related Other
Project Information form prior to completing this form.
Are Human Subjects Involved? Yes/No
This field is pre-populated from the F.220 - R&R Other Project Information Form. If the value in
this field appears to be incorrect, you may correct it by adjusting it on the F.220 - R&R Other
Project Information Form.
Is the Project Exempt from Federal regulations? Yes/No
This field is pre-populated from the F.220 - R&R Other Project Information Form. If the value in
this field appears to be incorrect, you may correct it by adjusting it on the F.220 - R&R Other
Project Information Form.
Exemption number: 1, 2, 3, 4, 5, 6, 7, 8
This field is pre-populated from the F.220 - R&R Other Project Information Form. If the value in
this field appears to be incorrect, you may correct it by adjusting it on the F.220 – R&R Other
Project Information Form.
Note: If you change your answer to the “Are Human Subjects Involved” question on the F.220 - R&R
Other Project Information Form after you have started entering information into the PHS Human
Subjects and Clinical Trials Information form, your data in the PHS Human Subjects and Clinical Trials
Information form may be lost.
F - 89

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
If No to Human Subjects
If you answered "No" to the question “Are Human Subjects Involved?” on the F.220 - R&R
Other Project Information Form, skip the rest of the PHS Human Subjects Clinical Trials
Information form unless otherwise directed by your FOA.
If Yes to Human Subjects
If you answered “Yes” to the question “Are Human Subjects Involved?” on the F.220 - R&R
Other Project Information Form, add a Study Record for each proposed study involving
human subjects by selecting “Add New Study” or “Add New Delayed Onset Study,” as
appropriate.
Other Requested Information
Who may provide Other Requested Information:
Follow the instructions below and any instructions in your FOA to determine whether you are
permitted to include the “Other Requested Information” attachment.
Format:
Attach this information as a PDF file. See NIH’s Format Attachments page.
Content:
Content is limited to what is described in your FOA or in these instructions. Do not use the
“Other Requested Information” attachment to include any other information.
Renewal applications: When preparing a renewal (or resubmission of a renewal), you can
provide a list of ongoing studies or ClinicalTrials.gov identifiers (e.g., NCT87654321).
Study Record(s)
Adding Study Record Attachment(s):
Add a study record for each proposed study involving human subjects. If specific plans for your
study involving human subjects can be described in the application but will not begin
immediately (i.e., your study has a delayed start), you must add a Study Record for that study. If
your study anticipates involving human subjects within the period of award but specific plans
cannot be described in the application (i.e., delayed onset), see the instructions for Delayed
Onset Study(ies).
For all submission methods, the Study Record is used to collect human subjects study data.
Note: The steps to add a Study Record attachment(s) may vary with the submission method. For
example, from the ASSIST Human Subjects and Clinical Trials tab, use the ‘Add New Study’ button
to access the data entry screens to enter Study Record information directly into ASSIST. With
other submission methods, you may have to extract a blank copy of the Study Record, complete it
offline, and then attach it to your application.
Note on Grouping Studies into Study Records: While there may be more than one way to split
or group studies into Study Records, you are encouraged to group studies that use the same
human subjects population and same research protocols into a single Study Record, to the
F - 90

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
extent that the information you provide is accurate and understandable to NIH staff and
reviewers.
If information in any attachment is identical across studies, include the complete information only
in the first Study Record for which the information is relevant. In the subsequent Study Records
for which the identical information is needed, upload an attachment that says, “See information
for attachment X in Study Record entitled [include study title]." No other information is needed in
the attachment. Do not submit attachments that are duplicated from one Study Record to
another. Note that you should not name Study Records by number. Examples of attachments
that may be identical across studies include, but are not limited to, the 3.1 Protection of Human
Subjects and 3.5 Overall Structure of the Study Team attachments.
See the NIH Glossary definitions of Study and Study Record.
The PHS Human Subjects and Clinical Trials Information form accommodates up to 150 separate
Study Records.
Format:
All attachments must be PDF files. If you extract a Study Record, it will already be in a fillable PDF
format. Please use this PDF file and do not alter the format of the Study Record file. Use unique
filenames for each human subject study record. The filename for each attachment within a study
must be unique within the application (i.e., do not use the same filename in multiple Study
Records).
Content:
Follow the instructions in the “Study Record: PHS Human Subjects and Clinical Trials Information”
section below.
Delayed Onset Study(ies)
If you anticipate conducting research involving human subjects but cannot describe the study at
the time of application (i.e., your study is a delayed onset human subject study), enter a Delayed
Onset Study Record as instructed below.
Generally, for any study that you include as a delayed onset study in this section, you will provide
a study title, indicate whether the study is anticipated to include a clinical trial, and include a
justification attachment. Since by definition, information for a delayed onset study is not available
at the time of application, you will not be given the option to complete a full Study Record for a
delayed onset study. For delayed onset studies, the Delayed Onset Study Record is sufficient.
Notes on delayed onset studies:
l Delayed onset does NOT apply to a study that can be described but will not start
immediately (i.e., delayed start). Refer to the NIH Glossary definition of Delayed Onset
Study and Delayed Start.
l If you anticipate multiple delayed onset studies, you can include them together in a
single Delayed Onset Study Record.
Study Title
This field is required.
The Study Title can have a maximum of 600 characters.
F - 91

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
Enter a brief, unique title that describes the study the participants will be involved in. Each study
within your application must have a unique Study Title. The first 150 characters will display in the
application image bookmarks.
Note on multiple delayed onset studies: If you are including multiple delayed onset studies in
one delayed onset study entry, you may enter “Multiple Delayed Onset Studies” as the title of this
record.
Anticipated Clinical Trial?
This field is required.
Check this box if you anticipate that this study will be a clinical trial. For help determining whether
your study meets the definition of clinical trial, see the Clinical Trial Questionnaire below.
Read your FOA carefully to determine whether clinical trials are allowed in your application.
Note on multiple delayed onset studies: If you are including multiple delayed onset studies in
one delayed onset study entry, and you anticipate that any of these studies will be a clinical trial,
check the “Anticipated Clinical Trial?” checkbox.
Additional Instructions for Fellowship:
Do not check the “Anticipated Clinical Trial?” box. Fellowship FOAs do not allow
independent clinical trials.
Justification Attachment
This attachment is required.
Attach the justification as a PDF file. See NIH’s Format Attachments page.
l All delayed onset studies must provide a justification explaining why human subjects
study information is not available at the time of application.
l
If NIH’s Single Institutional Review Board (sIRB) policy will apply to your study, this
justification must also include information regarding how the study will comply with the
policy. The applicant must provide a statement naming the sIRB of record in the Just-in-
Time submission prior to award.
l If NIH’s Policy on the Dissemination of NIH-Funded Clinical Trial Information will apply to
your study, this justification must also include the dissemination plan.
Note on multiple delayed onset studies: If you are including more than one delayed onset
study in any given delayed onset study entry, address all the included studies in a single
justification attachment.
F - 92

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
Study Record: PHS Human Subjects and Clinical
Trials Information
Section 1 - Basic Information
Who must complete “Section 1 – Basic Information:”
“Section 1 – Basic Information” is required for all studies involving human subjects.
1.1 Study Title (each study title must be unique)
The “Study Title” field is required.
The Study Title can have a maximum of 600 characters.
Enter a brief title that describes the study the participants will be involved in. If there is more than
one study (i.e., you are including more than one Study Record and/or delayed onset study in your
application), each one must have a unique study title. The first 150 characters will display in the
bookmarks of the application image.
Note: When registering a clinical trial in ClinicalTrials.gov, all study titles across your organization
must be unique.
Note: This field matches a ClinicalTrials.gov field (Official Title).
1.2 Is this Study Exempt from Federal Regulations?
An answer to the “Is this Study Exempt from Federal Regulations?” question is required.
Indicate whether the study is exempt from Federal regulations for the Protection of Human
Subjects.
For more information, see the NIH's Definition of Human Subjects Research website.
1.3 Exemption Number
The “Exemption Number” field is required if you selected “Yes” to the “Is this Study Exempt from
Federal Regulations?” question.
Select the appropriate exemption number(s) for this particular study. Multiple selections are
permitted. Regardless of whether these exemptions may apply to you in the future, you must fill
out your application following the instructions below.
For more information:
The categories of research that qualify for exemption are defined in the Common Rule for the
Protection of Human Subjects. These regulations can be found at 45 CFR 46.
Need help determining the appropriate exemption number?
l Refer to NIH's Human Subjects FAQs.
l See the NIH's Human Subjects Frequently Asked Questions section on Exemptions.
F - 93

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
The Office of Human Research Protections (OHRP) guidance states that appropriate use of
exemptions described in 45 CFR 46 should be determined by an authority independent from the
investigators (for more information, see OHRP's Frequently Asked Questions). Institutions often
designate their Institutional Review Board (IRB) to make this determination. Because NIH does
not require IRB approval at the time of application, the exemptions designated often represent
the opinion of the PD/PI, and the justification provided for the exemption by the PD/PI is
evaluated during peer review.
1.4 Clinical Trial Questionnaire
The Clinical Trial Questionnaire is required.
Note for basic and mechanistic studies involving human participants: The NIH definition of a
clinical trial encompasses a broad range of studies, including studies using human participants
that aim to understand fundamental aspects of phenomena, the pathophysiology of a disease,
or the mechanism of action of an intervention. This includes many mechanistic studies and
studies submitted to Basic Experimental Studies with Humans FOAs.
Answer “Yes” or “No” to the following questions to determine whether this study involves a
clinical trial. Answer the following questions based only on the study you are describing in this
Study Record.
Note: The answer to question “1.4.a Does the study involve human participants?” will be pre-
populated with “Yes” for all study records. You will not be able to change this answer.
1.4.a. Does the study involve human participants? Yes/No
1.4.b. Are the participants prospectively assigned to an intervention? Yes/No
1.4.c. Is the study designed to evaluate the effect of the intervention on the participants?
Yes/No
1.4.d. Is the effect that will be evaluated a health-related biomedical or behavioral out-
come? Yes/No
If you answered “Yes” to all the questions in the Clinical Trial Questionnaire, this study meets the
definition of a clinical trial.
Refer to the table below for information about what sections of this form are required, based on
your answers to Question 1.4 "Clinical Trial Questionnaire."
Form Section
If you answered "yes"
to all the questions in
the Clinical Trial
Questionnaire
If you answered "no"
to any of the questions
in the Clinical Trial
Questionnaire
Section 2 - Study Population
Characteristics
Required
Required
Section 3 - Protection and
Monitoring Plans
Required
Required
F - 94

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
Form Section
If you answered "yes"
to all the questions in
the Clinical Trial
Questionnaire
If you answered "no"
to any of the questions
in the Clinical Trial
Questionnaire
Section 4 - Protocol Synopsis
Required
Do not complete
Section 5 - Other Clinical Trial-
related Attachments
Required if specified in the
FOA
Do not complete
Additional Instructions for Fellowship:
Fellowship applicants who are proposing to gain clinical trial research
experience under a sponsor’s supervision (i.e., you will not be leading an
independent clinical trial): Even if you answered “Yes” to all the questions in the
Clinical Trial Questionnaire, only certain fields of the PHS Human Subjects and
Clinical Trials Information form are required (and other fields are not allowed)
because the study is not an independent clinical trial. Do not provide information in
“Section 4 – Protocol Synopsis” or in “Section 5 – Other Clinical Trial-related
Attachments” of the Study Record. Inputting information into these sections will
result in errors and will prevent your application from being accepted.
You will generally follow the standard instructions to complete the PHS Human
Subjects and Clinical Trials Information form, but follow relevant Fellowship
instructions where they are given.
For more information:
l NIH Glossary’s definition of an NIH-defined clinical trial
l NIH's Definition of a Clinical Trial page
l NIH Definition of Clinical Trials Case Studies page
l FAQs on the NIH Clinical Trial Definition
l NIH’s decision tool will help determine whether your human subjects research study is
an NIH-defined clinical trial
l Your study may also be subject to additional regulations. Read NIH’s Requirements for
Registering & Reporting NIH-funded Clinical Trials in ClinicalTrials.gov.
1.5. Provide the ClinicalTrials.gov Identifier (e.g., NCT87654321) for this trial, if
applicable
If a clinical trial has already been entered into ClinicalTrials.gov, enter the ClinicalTrials.gov
identifier (e.g., NCT87654321) for this trial. Enter the identifier only if you are proposing to work
on that specific clinical trial. If you are only getting samples and/or data from a clinical trial that
has already been entered into ClinicalTrials.gov, do NOT enter the identifier.
F - 95

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
If you are building on an existing study (e.g., ancillary study), enter the ClinicalTrials.gov identifier
only for the ancillary study (if registered separately), not the parent study.
Note: The number you enter in this field should match the ClinicalTrials.gov identifier assigned by
ClinicalTrials.gov.
Section 2 - Study Population Characteristics
Who must complete “Section 2 - Study Population Characteristics:”
All of “Section 2 – Study Population Characteristics” is required (see exceptions for Question 2.7 Study
Timeline and for Question 2.8 Enrollment of First Subject) for all human subjects studies unless the
following applies to you:
l If you selected only Exemption 4 and no other exemptions on the "1.3 Exemption Number"
question, then “Section 2 – Study Population Characteristics” is not required.
2.1 Conditions or Focus of Study
At least 1 entry is required, and up to 20 entries are allowed (enter each entry on its own line).
Each entry is limited to 255 characters.
Identify the name(s) of the disease(s) or condition(s) you are studying, or the focus of the study. If
available, use appropriate descriptors from NLM's Medical Subject Headings (MeSH) so the
application can be categorized. Include an entry for each condition.
Note: This field matches a ClinicalTrials.gov field (Primary Disease or Condition Being Studied in
the Trial, or the Focus of the Study).
2.2 Eligibility Criteria
List the study’s inclusion and exclusion criteria. To provide a bulleted list, use a dash (or other
character) followed by a space (“- “) at the start of each bullet. Be sure to check the formatting in
the assembled application image. Further explanation or justification should be included in the
Recruitment and Retention plan.
Your text entry is limited to 15,000 characters (but typically needs only 500 characters).
Note: This field matches a ClinicalTrials.gov field (Eligibility Criteria).
For more information about formatting text entry fields, see NIH's Rules for Text Fields page and
the ClinicalTrials.gov's Protocol Registration and Results System User's Guide.
2.3 Age Limits
Minimum Age
Enter the numerical value for the minimum age a potential participant can be to be eligible for
the study. Provide the relevant units of time (i.e., years, months, weeks, days, hours, or minutes). If
there is no lower limit or no lower limit is known, enter “N/A (No Limit)” and do not enter a unit of
time.
F - 96

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
Maximum Age
Enter the numerical value for the maximum age a potential participant can be to be eligible for
the study. Provide the relevant units of time (i.e., years, months, weeks, days, hours, or minutes). If
there is no upper limit or no upper limit is known, enter “N/A (No Limit)” and do not enter a unit
of time.
Note: This field matches a ClinicalTrials.gov field (Age Limits).
2.3.a Inclusion of Individuals Across the Lifespan
Format:
Attach this information as a PDF file. See NIH’s Format Attachments page.
Content:
Discuss each of the points listed below. Also include any additional information requested in the
FOA.
You will also have to complete an Inclusion Enrollment Report (IER). Note that you may need to
include multiple IERs for each study. Refer to the instructions for the IER below for more
information.
Inclusion of Individuals Across the Lifespan
For the purposes of the Inclusion of Individuals Across the Lifespan, exclusion of any specific age
or age range group (e.g., children or older adults) should be justified in this section. In addition,
address the following points:
l Individuals of all ages are expected to be included in all NIH-defined clinical research
unless there are scientific or ethical reasons not to include them. Discuss whether
individuals will be excluded based on age and provide a rationale for the minimum and
maximum age of study participants, if applicable. Additionally, if individuals will be
excluded based on age, provide a scientific or ethical rationale for their exclusion. See the
NIH Policy and Guidelines on the Inclusion of Individuals Across the Lifespan as
Participants in Research Involving Human Subjects for additional information about
circumstances that may justify the exclusion of individuals based on age.
l Include a description of the expertise of the investigative team for working with
individuals of the ages included, the appropriateness of the available facilities to
accommodate individuals in the included age range, and how the age distribution of
participants will contribute to a meaningful analysis relative to the purpose of the study.
When children are involved in research, the policies under HHS’ 45 CFR 46, Subpart D - Additional
Protections for Children Involved as Subjects in Research apply and must be addressed in the
Protection of Human Subjects attachment.
Existing Datasets or Resources. If you will use an existing dataset, resource, or samples that may
have been collected as part of a different study, you must address inclusion, following the
instructions above. Generally, you must provide details about the sex/gender, race, and ethnicity
of the existing dataset/resource and justify the details as appropriate to the scientific goals of the
proposed study.
For more information about what is considered an existing dataset or resource for inclusion
policy, see the NIH FAQs on Monitoring Inclusion When Working with Existing Datasets and/or
Resources.
F - 97

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
For more information, see:
l NIH Policy Implementation Page on Inclusion Across the Lifespan
l Inclusion Across the Lifespan: Guidance for Applying the Policy infographic
l NIH FAQs on Inclusion Across the Lifespan
l HHS’ 45 CFR 46 Subpart D – Additional Protections for Children
l NIH Grants Policy Statement, Section 4.1.15.7: Inclusion of Individuals Across the Lifespan
as Participants in Research Involving Human Subjects
2.4 Inclusion of Women and Minorities
Format:
Attach this information as a PDF file. See NIH’s Format Attachments page.
Content:
Discuss each of the points listed below and include any additional information requested in the
FOA.
You will also have to complete an Inclusion Enrollment Report (IER). Note that you may need to
include multiple IERs for each study. Refer to the instructions for the IER below for more
information.
Inclusion of Women and Minorities
Address the following points:
l Describe the planned distribution of subjects by sex/gender, race, and ethnicity.
l Describe the rationale for selection of sex/gender, racial, and ethnic group members in
terms of the scientific objectives and proposed study design. The description may
include, but is not limited to, information on the population characteristics of the
disease or condition under study.
l Describe proposed outreach programs for recruiting sex/gender, racial, and ethnic
group members.
l Inclusion and Excluded Groups: Provide a reason for limiting inclusion of any group by
sex/gender, race, and/or ethnicity. In general, the cost of recruiting certain groups
and/or geographic location alone are not acceptable reasons for exclusion of particular
groups. See the Inclusion of Women and Minorities as Participants in Research Involving
Human Subjects - Policy Implementation Page for more information.
Existing Datasets or Resources. If you will use an existing dataset, resource, or samples that may
have been collected as part of a different study, you must address inclusion, following the
instructions above. Generally, you must provide details about the sex/gender, race, and ethnicity
of the existing dataset/resource and justify the details as appropriate to the scientific goals of the
proposed study.
For more information about what is considered an existing dataset or resource for inclusion
policy, see the NIH FAQs on Monitoring Inclusion When Working with Existing Datasets and/or
Resources.
F - 98

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
NIH-Defined Phase III Clinical Trials. If the proposed research includes an NIH-Defined Phase
III Clinical Trial, the “Inclusion of Women and Minorities” attachment MUST address plans for how
sex/gender, race, and ethnicity will be taken into consideration in the design and valid analysis of
the trial. See the instructions for “Valid Analysis” and “Plans to test for Differences in Effect among
Sex/gender, Racial, and/or Ethnic Groups” below.
Additional information about valid analysis is available on the NIH Policy and Guidelines on The
Inclusion of Women and Minorities as Subjects in Clinical Research page.
Valid Analysis (for NIH-Defined Phase III Clinical Trials only):
Address the following issues for ensuring valid analyses:
l Inclusive eligibility criteria – in general, the cost of recruiting certain groups and/or
geographic location alone are not acceptable reasons for exclusion of particular
groups;
l Allocation of study participants of both sexes/genders and from different racial
and/or ethnic groups to the intervention and control groups by an unbiased process
such as randomization;
l Unbiased evaluation of the outcome(s) of study participants; and
l Use of unbiased statistical analyses and proper methods of inference to estimate and
compare the intervention effects by sex/gender, race, and/or ethnicity, particularly if
prior evidence strongly suggests that such differences exist.
Plan to Test for Differences in Effect among Sex/gender, Racial, and/or Ethnic Groups (for NIH-
Defined Phase III Clinical Trials only):
Applicants also should address whether they plan to test for differences in effect among
sex/gender, racial, and/or ethnic groups and why such testing is or is not appropriate.
This plan must include selection and discussion of one of the following analysis plans:
l Plans to conduct analyses to detect significant differences in intervention effect
among sex/gender, racial, and/or ethnic subgroups when prior studies strongly
support these significant differences among one or more subgroups, or
l Plans to include and analyze sex/gender, racial, and/or ethnic subgroups when prior
studies strongly support no significant differences in intervention effect between
subgroups. (Representation of sex/gender, racial, and ethnic groups is not required
as subject selection criteria, but inclusion is encouraged.), or
l Plans to conduct valid analyses of the intervention effect in sex/gender, racial, and/or
ethnic subgroups (without requiring high statistical power for each subgroup) when
the prior studies neither support nor negate significant differences in intervention
effect among subgroups.
For more information, see:
l NIH's Policy Implementation Page on the Inclusion of Women and Minorities
l HHS’ 45 CFR 46 Subpart B – Additional Protections for Pregnant Women, Fetuses, and
Neonates
l NIH Grants Policy Statement, Section 4.1.15.8: Inclusion of Women and Minorities as
Subjects in Clinical Research and Reporting Sex/Gender, Racial, and Ethnic Participation
F - 99

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
2.5 Recruitment and Retention Plan
Who must complete the "Recruitment and Retention Plan" attachment:
The “Recruitment and Retention Plan” attachment is required unless the following applies to you:
l You selected only Exemption 4 and no other exemptions on the “1.3 Exemption
Number” question.
Format:
Attach this information as a PDF file. See NIH’s Format Attachments page.
Content:
Describe how you will recruit and retain participants in your study. You should address both
planned recruitment activities as well as proposed engagement strategies for retention.
2.6. Recruitment Status
Who must complete the "Recruitment Status" question:
The “Recruitment Status” question is required unless the following applies to you:
l You selected only Exemption 4 and no other exemptions on the “1.3 Exemption
Number” question.
Content:
From the dropdown menu, select the "Recruitment Status" that best describes the proposed
study, based upon the status of the individual sites. If any facility in a multi-site study has an
individual site status of “recruiting,” then choose “recruiting” for this question. Only one selection
is allowed. Choose from the following options:
l Not yet recruiting
l Recruiting
l Enrolling by invitation
l Active, not recruiting
l Completed
l Suspended
l Terminated (Halted Prematurely)
l Withdrawn (No Participants Enrolled)
Note: This field matches a ClinicalTrials.gov field (Overall Recruitment Status).
2.7. Study Timeline
Who must complete the "Study Timeline" attachment:
The “Study Timeline” attachment is required if you answered "Yes" to all the questions in the
"Clinical Trial Questionnaire" (i.e., your study is a clinical trial).
F - 100

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
The "Study Timeline" attachment is optional if either of the following apply to you:
l You selected only Exemption 4 and no other exemptions on the “1.3 Exemption
Number” question.
l You answered "No" to any of the questions in the "Clinical Trial Questionnaire" (i.e., your
study is not a clinical trial).
Format:
Attach this information as a PDF file. See NIH's Format Attachments page.
Content:
Provide a description or diagram describing the study timeline. The timeline should be general
(e.g., "one year after notice of award"), and should not include specific dates.
Note: Additional milestones or timelines may be requested as just-in-time information or post-
award.
2.8. Enrollment of First Participant
Who must complete the "Enrollment of First Participant" question:
Do not complete this field if you will answer "Yes" to the question "Using an Existing Dataset or
Resource" in the Inclusion Enrollment Report.
The “Enrollment of First Participant” question is otherwise required unless the following applies
to you:
l You selected only Exemption 4 and no other exemptions on the “1.3 Exemption
Number” question.
Content:
Enter the date (MM/DD/YYYY) of the enrollment of the first participant into the study. From the
dropdown menu, select whether this date is anticipated or actual.
2.9. Inclusion Enrollment Report(s)
Who must complete the Inclusion Enrollment Report(s):
An Inclusion Enrollment Report is required for all human subjects studies unless, on Question 1.3
“Exemption Number,” you selected only Exemption 4 and no other exemptions.
Using the Inclusion Enrollment Report:
Each proposed study, unless it falls under Exemption 4, must contain at least one Inclusion Enrollment
Report (IER). However, more than one IER per study is allowed.
Once you have added an IER for a given study, you may edit, remove, or view it.
Note: You can add a maximum of 20 IERs per Study Record. These can be a combination of planned
and cumulative reports.
Multi-site studies: Generally, if the application includes a study recruiting subjects at more than one
site/location, investigators may create one IER or separate, multiple IERs to enable reporting by study
F - 101

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
or by site, depending on the scientific goals of the study and whether monitoring of inclusion
enrollment would benefit from being combined or separated. At a minimum, participants enrolled at
non-U.S. sites must be reported separately from participants enrolled at U.S. sites, even if they are part
of the same study. Please review the FOA to determine whether there are any other specific
requirements about how to complete the IER.
Duplicative Inclusion Reports: It is important that the IER for a given study be associated with only
one application and be provided only once in a given application (e.g., do not submit the same IER on
both the data coordinating center and the research site). If submitting individual application(s) as part
of a network or set of linked applications, please provide the IER with the individual site applications
unless otherwise directed by the FOA.
Renewal applications: When preparing a renewal (or resubmission of a renewal), investigators should
provide a narrative description regarding the cumulative enrollment from the previous funding period
(s) as part of the progress report section of the research strategy attachment in the application. The
IER should NOT be used for this purpose. If a given study will continue with the same enrollment or
additional enrollment, or if new studies are proposed, provide a new IER for each as described in the
instructions below.
Resubmission applications: If IERs were provided in the initial submission application, and if those
studies will be part of the resubmission application, complete the IER and submit again with the
resubmission application, regardless of whether the enrollment has changed or not. Also, provide any
new (additional) IERs.
Revision applications: Provide an IER if new studies are planned as part of the Revision and they meet
the NIH definition for clinical research.
For more information:
Refer to the Inclusion of Women and Minorities as Participants in Research Involving Human Subjects -
Policy Implementation Page.
1. Inclusion Enrollment Report Title
The “Inclusion Enrollment Report Title” field is required.
The “Inclusion Enrollment Report title can have a maximum of 600 characters.
Enter a unique title for each IER. The title should indicate specific criteria that uniquely identify
each report. If the Project Title is pre-populated, you may edit it so that each IER title is unique.
2. Using an Existing Dataset or Resource?
The “Using an Existing Dataset or Resource” question is required.
If the study involves analysis of an existing dataset or resource (e.g., biospecimens) only, answer
“Yes” to this question. If the study involves prospective recruitment or new contact with
participants answer “No” to this question. Use separate IERs for studies involving use of existing
datasets or resources only and for studies that involve prospective recruitment or new contact
with study participants.
For additional guidance on what is considered an existing dataset, refer to the NIH FAQs on
Monitoring Inclusion When Working with Existing Datasets and/or Resources.
3. Enrollment Location Type (Domestic/Foreign)
The “Enrollment Location Type” field is required.
F - 102

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
Select whether the participants described in the IER are based at a U.S. (Domestic) or at a non-
U.S. (Foreign) site. Participants at U.S. and non-U.S. sites must be reported separately (i.e., on
separate IERs), even if it is for the same study.
For additional guidance on how to complete the IER if you will be working with non-U.S.
populations, refer to these FAQs on Inclusion on the Basis of Sex/Gender and Race/Ethnicity.
4. Enrollment Country(ies)
The “Enrollment Country(ies)” field is optional.
Indicate the country or countries in which participants will be enrolled. Multiple U.S. sites can be
reported together in one IER. Foreign countries can be reported together in one IER. However,
you must use separate IERs for U.S. and non-U.S. sites. You can add up to 200 countries per IER.
5. Enrollment Location(s)
The “Enrollment Location(s)” field is optional.
Indicate the type of enrollment location (e.g., hospital, university, or research center), not the
name of the enrollment location.
Enrollment locations are typically where the research is conducted, and can be different from the
recruitment site.
6. Comments
Your comments are limited to 500 characters.
Enter information you wish to provide about this IER. This includes, but is not limited to,
addressing information about distinctive subpopulations if relevant to the scientific hypotheses
being studied. If inclusion monitoring is conducted on another study or NIH grant (e.g., data
coordinating center or research site), please indicate here.
Revision applications: If there are no updates to the IER(s) in your original grant application, do
not include an IER in your Revision application. Instead, provide a comment in this field to the
effect that previous IER(s) are still applicable. If you are revising the IER(s) in your original grant
application, provide a comment here to that effect.
Planned
Who must complete planned enrollment tables:
All studies must enter planned enrollment counts unless your proposed study will use only an
existing dataset or resource. Planned enrollment generally means that individuals will be
recruited into the study and/or that individuals have already been recruited and continue to be
part of the study.
For more information about what is considered an existing dataset or resource for inclusion
policy, see the NIH FAQs on Inclusion on the Basis of Sex/Gender and Race/Ethnicity.
For more information on racial categories, see the NIH Glossary definition of Racial Categories.
For more information on ethnic categories, see the NIH Glossary definition of Ethnic Categories.
Racial Categories
American Indian/Alaska Native:
These fields are required.
F - 103

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
Enter the expected number of females and males (in the respective fields) who are both American
Indian/Alaska Native and Not Hispanic or Latino. Enter the expected number of females and
males (in the respective fields) who are both American Indian/Alaska Native and Hispanic or
Latino.
Asian:
These fields are required.
Enter the expected number of females and males (in the respective fields) who are both Asian
and Not Hispanic or Latino. Enter the expected number of females and males (in the respective
fields) who are both Asian and Hispanic or Latino.
Native Hawaiian or Other Pacific Islander:
These fields are required.
Enter the expected number of females and males (in the respective fields) who are both Native
Hawaiian or Other Pacific Islander and Not Hispanic or Latino. Enter the expected number of
females and males (in the respective fields) who are both Native Hawaiian or Other Pacific
Islander and Hispanic or Latino.
Black or African American:
These fields are required.
Enter the expected number of females and males (in the respective fields) who are both Black or
African American and Not Hispanic or Latino. Enter the expected number of females and males (in
the respective fields) who are both Black or African American and Hispanic or Latino.
White:
These fields are required.
Enter the expected number of females and males (in the respective fields) who are both White
and Not Hispanic or Latino. Enter the expected number of females and males (in the respective
fields) who are both White and Hispanic or Latino.
More than One Race:
These fields are required.
Enter the expected number of females and males (in the respective fields) who both identify with
more than one racial category and are Not Hispanic or Latino. Enter the expected number of
females and males (in the respective fields) who both identify with more than one racial category
and are Hispanic or Latino.
Total:
The total fields at the bottom will be automatically calculated and reflect the totals of all racial
categories for females, males, and individuals of unknown/not reported sex/gender who are Not
Hispanic or Latino and of all racial categories for females, males, and individuals of unknown/not
reported sex/gender who are Hispanic or Latino. The “Total” fields in the right column will be
automatically calculated to total all individuals.
Cumulative (Actual)
Who must complete cumulative (actual) enrollment tables:
You must enter cumulative enrollment counts if your proposed study will use an existing dataset
or resource.
F - 104

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
For more information about what is considered an existing dataset or resource for inclusion
policy, see the NIH FAQs on Inclusion on the Basis of Sex/Gender and Race/Ethnicity.
For more information on racial categories, see the NIH Glossary definition of Racial Categories.
For more information on ethnic categories, see the NIH Glossary definition of Ethnic Categories.
Racial Categories
American Indian/Alaska Native:
These fields are required.
Enter the number of females and males (in the respective fields) who are both American
Indian/Alaska Native and Not Hispanic or Latino. Enter the number of females and males (in the
respective fields) who are both American Indian/Alaska Native and Hispanic or Latino. Use the
“Unknown/Not Reported” fields as needed (i.e., race and/or ethnicity is unknown).
Asian:
These fields are required.
Enter the number of females and males (in the respective fields) who are both Asian and Not
Hispanic or Latino. Enter the expected number of females and males (in the respective fields) who
are both Asian and Hispanic or Latino. Use the “Unknown/Not Reported” fields as needed (i.e.,
race and/or ethnicity is unknown).
Native Hawaiian or Other Pacific Islander:
These fields are required.
Enter the number of females and males (in the respective fields) who are both Native Hawaiian or
Other Pacific Islander and Not Hispanic or Latino. Enter the expected number of females and
males (in the respective fields) who are both Native Hawaiian or Other Pacific Islander and
Hispanic or Latino. Use the “Unknown/Not Reported” fields as needed (i.e., race and/or ethnicity
is unknown).
Black or African American:
These fields are required.
Enter the number of females and males (in the respective fields) who are both Black or African
American and Not Hispanic or Latino. Enter the expected number of females and males (in the
respective fields) who are both Black or African American and Hispanic or Latino. Use the
“Unknown/Not Reported” fields as needed (i.e., race and/or ethnicity is unknown).
White:
These fields are required.
Enter the number of females and males (in the respective fields) who are both White and Not
Hispanic or Latino. Enter the expected number of females and males (in the respective fields) who
are both White and Hispanic or Latino. Use the “Unknown/Not Reported” fields as needed (i.e.,
race and/or ethnicity is unknown).
More than One Race:
These fields are required.
Enter the number of females and males (in the respective fields) who both identify with more
than one racial category and are Not Hispanic or Latino. Enter the expected number of females
and males (in the respective fields) who both identify with more than one racial category and are
F - 105

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
Hispanic or Latino. Use the “Unknown/Not Reported” fields as needed (i.e., race and/or ethnicity
is unknown).
Unknown or Not Reported:
These fields are required.
Enter the number of females, males, and individuals of unknown/not reported sex/gender (in the
respective fields) whose race is unknown/not reported and who are Not Hispanic or Latino. Enter
the number of females, males, and individuals of unknown/not reported sex/gender (in the
respective fields) whose race is unknown/not reported and who are Hispanic or Latino. Enter the
number of females, males, and individuals of unknown/not reported sex/gender (in the
respective fields) who are both of unknown/not reported race and of unknown/not reported
ethnicity. Use the “Unknown/Not Reported” fields as needed (i.e., race and/or ethnicity is
unknown).
Total:
The total fields at the bottom will be automatically calculated and reflect the totals of all racial
categories for females, males, and individuals of unknown/not reported sex/gender who are Not
Hispanic or Latino and of all racial categories for females, males, and individuals of unknown/not
reported sex/gender who are Hispanic or Latino. Use the “Unknown/Not Reported” fields as
needed (i.e., race and/or ethnicity is unknown). The “Total” fields in the right column will be
automatically calculated to total all individuals.
Section 3 – Protection And Monitoring Plans
Who must complete “Section 3 – Protection and Monitoring Plans:”
All of “Section 3 – Protection and Monitoring Plans” is required for all studies involving human
subjects, unless otherwise noted.
3.1 Protection of Human Subjects
The “Protection of Human Subjects” attachment is required.
Format:
Attach this information as a PDF file. See NIH’s Format Attachments page.
Do not use the “Protection of Human Subjects” attachment to circumvent the page limits of the
Research Strategy.
For Human Subjects Research Claiming Exemptions: If you are claiming that your human
subjects research falls under any exemptions, justify why the research meets the criteria for the
exemption(s) that you have claimed. This justification should explain how the proposed research
meets the criteria for the exemption claimed. Do not merely repeat the criteria or definitions
themselves.
For Studies that involve Non-Exempt Human Subjects Research: For any proposed non-
exempt study involving human subjects, NIH requires a Protection of Human Subjects
attachment that is commensurate with the risks of the study, its size, and its complexity. Organize
your attachment into four sections, following the headings and specified order below, and
discuss each of the points listed below. Start each section with the appropriate section heading –
F - 106

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
Risks to Human Subjects, Adequacy of Protection Against Risks, Potential Benefits of the
Proposed Research to Research Participants and Others, and Importance of the Knowledge to be
Gained. Also include any additional information requested in the FOA.
1. Risks to Human Subjects
a. Human Subjects Involvement, Characteristics, and Design
l Briefly describe the overall study design.
l Describe the subject population(s) to be included in the study; the procedures for assignment
to a study group, if relevant; and the anticipated numbers of subjects for each study group.
l List any collaborating sites where human subjects research will be performed, and describe
the role of those sites and collaborating investigators in performing the proposed research.
b. Study Procedures, Materials, and Potential Risks
l Describe all planned research procedures (interventions and interactions) involving study
subjects; how research material, including biospecimens, data, and/or records, will be
obtained; and whether any private identifiable information will be collected in the proposed
research project.
l For studies that will include the use of previously collected biospecimens, data or records,
describe the source of these materials, whether these can be linked with living individuals,
and who will be able to link the materials.
l Describe all the potential risks to subjects associated with each study intervention, procedure
or interaction, including physical, psychological, social, cultural, financial, and legal risks; risks
to privacy and/or confidentiality; or other risks. Discuss the risk level and the likely impact to
subjects.
l Where appropriate, describe alternative treatments and procedures, including their risks and
potential benefits. When alternative treatments or procedures are possible, make the
rationale for the proposed approach clear.
2. Adequacy of Protection Against Risks
a. Informed Consent and Assent
l Describe the process for obtaining informed consent. Include a description of the
circumstances under which consent will be sought and obtained, who will seek it, the nature
of the information to be provided to prospective subjects, and the method of documenting
consent. When appropriate, describe how potential adult subjects’ capacity to consent will be
determined and the plans for obtaining consent from a legally authorized representative for
adult subjects not able to consent.
o
For research involving children: If the proposed studies will include children, describe
the process for meeting HHS regulatory requirements for parental permission and child
assent (45 CFR 46.408). See the HHS page on Research with Children FAQs and the NIH
page on Requirements for Child Assent and Parent/Guardian Permission.
l If a waiver of some or all of the elements of informed consent will be sought, provide
justification for the waiver. Do not submit informed consent document(s) with your
application unless you are requested to do so.
F - 107

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
b. Protections Against Risk
l Describe planned strategies for protecting against or minimizing all potential risks identified,
including strategies to manage and protect the privacy of participants and confidentiality of
research data.
l Where appropriate, discuss plans for ensuring necessary medical or professional intervention
in the event of adverse effects on participants.
l Describe plans for handling incidental findings, such as those from research imaging,
screening tests, or paternity tests.
c. Vulnerable Subjects, if relevant to your study
Explain the rationale for the involvement of special vulnerable populations, such as fetuses, neonates,
pregnant women, children, prisoners, institutionalized individuals, or others who may be considered
vulnerable populations. 'Prisoners' includes all subjects involuntarily incarcerated (for example, in
detention centers).
Pregnant Women, Fetuses, and Neonates or Children
If the study involves vulnerable subjects subject to additional protections under Subparts B and D
(pregnant women, fetuses, and neonates or children), provide a clear description of the risk level and
additional protections necessary to meet the HHS regulatory requirements.
l HHS’ Subpart B - Additional Protections for Pregnant Women, Fetuses, and Neonates
l HHS’ Subpart D - Additional Protections for Children
l OHRP Guidance on Subpart D Special Protections for Children as Research Subjects and the
HHS 407 Review Process
Prisoners
If the study involves vulnerable subjects subject to additional protections under Subpart C (prisoners),
describe how proposed research meets the additional regulatory requirements, protections, and
plans to obtain OHRP certification for the involvement of prisoners in research.
Refer to HHS regulations, and OHRP guidance:
l HHS’ Subpart C - Additional Protections Pertaining to Prisoners as Subjects
l OHRP Subpart C Guidance on Involvement of Prisoners in Research
3. Potential Benefits of the Proposed Research to Research Participants and Others
l Discuss the potential benefits of the research to research participants and others.
l Discuss why the risks to subjects are reasonable in relation to the anticipated benefits to
research participants and others.
l Note: Financial compensation of subjects should not be presented as a benefit of
participation in research.
4. Importance of the Knowledge to be Gained
l Discuss the importance of the knowledge to be gained as a result of the proposed research.
l Discuss why the risks to subjects are reasonable in relation to the importance of the
knowledge that reasonably may be expected to result.
F - 108

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
For more information:
l Refer to the NIH’s Human Subjects Research website.
3.2 Is this a multi-site study that will use the same protocol to conduct non-
exempt human subjects research at more than one domestic site?
Select "Yes" or "No" to indicate whether this is a multi-site study that will use the same
protocol to conduct non-exempt human subjects research at more than one domestic site.
Select “N/A” only if any of the following apply (do not select “N/A” if none of the following apply):
l You answered “Yes” to “Question 1.2 Is this Study Exempt from Federal Regulations?
(Yes/No)”
l You are a training grant applicant.
Applicants who check “Yes” are expected to use a single Institutional Review Board (sIRB) to
conduct the ethical review required by HHS regulations for the Protections of Human Subjects
Research unless review by a sIRB would be prohibited by a federal, tribal, or state law, regulation,
or policy.
Note: The NIH sIRB policy applies to participating domestic sites. Foreign sites
participating in NIH-funded, multi-site studies are not expected to follow this
policy.
For more information:
l HHS regulations and requirements for the Protections of Human Subjects can be found
at 45 CFR 46.
l See NIH’s Single IRB Policy for Multi-site Research for more information.
l See the FAQ about answering “No” for this question on the Applying Electronically FAQ
page.
If yes, describe the single IRB plan
For NIHApplicants, the single IRBplan is no longer required. See additional information in
the content section below.
For AHRQ applicants, if this is a research project that involves more than one institution and
that will be conducted in the United States, Applicants are expected to use a single Institutional
Review Board (sIRB) to conduct the ethical review required by HHS regulations for the
Protections of Human Subjects Research, and include a single IRB plan as instructed below,
unless review by a sIRB would be prohibited by a federal, tribal, or state law, regulation, or policy.
Note: The sIRB requirement applies to participating sites in the United States. Foreign sites
participating in AHRQ-funded, cooperative research studies are not expected to follow this
requirement.
Format:
Attach this information as a PDF file. See NIH’s Format Attachments page.
F - 109

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
Although one sIRB attachment per application is sufficient, you must include a file for each study
within your application. All filenames within your application must be unique. You may either
attach the same sIRB plan (with different filenames) to different studies or attach a file that refers
to the sIRB plan in another study within your application. For example, you may attach a file that
says “See sIRB plan in the 'My Unique Study Name' study.”
Content:
For NIHapplicants, the single IRB plan is no longer required. Do not provide an attachment.
The applicant must provide a statement naming the sIRB of record in the Just-in-Time
submission prior to award.
For Studies with Legal-, Regulatory-, or Policy-based Claims for Exception as described by
the sIRB Policy: As part of the Just-in-Time submission prior to award, indicate that review by an
sIRB will not be possible for all or some sites (specify which sites) because local IRB review is
required by an existing federal/state/tribal law or policy. Include a specific citation to the relevant
law, policy, or regulation.
For sites requesting an exception based on compelling justification: Indicate which site(s) is
requesting an exception to the use of the sIRB and provide compelling justification based on
ethical or human subjects protection issues or other well-justified reasons. NIH will determine
whether to grant an exception following an assessment of the need. Note: If you intend to
request an exception to the sIRB policy based on compelling justification, do not account for this
exception in your proposed budget. The proposed budget must reflect any necessary sIRB costs
without an exception (i.e., applicants should not assume that an exception will be granted when
considering what sIRB costs to include in the budget).
For more information:
l NIH's Single IRB Policy for Multi-site Research page
l NIH's FAQs on Single IRB Policy for Multi-site Research
l NIH's Office of Science Policy's FAQs on NIH Policy on the Use of a Single IRB for Multi-
Site Research Costs
l NIH's Office of Science Policy's FAQs on Implementation of the sIRB policy
l NIH Guide Notice on the Revised NIHPolicy on SIRB.
For AHRQ applicants, the single IRB plan should include the following elements:
l Describe how you will comply with the single IRB review requirement under the Revised
Common Rule at 45 CFR 46.114 (b) (cooperative research). If available, provide the name of the
IRB that you anticipate will serve as the sIRB of record.
l Indicate that all identified participating sites will agree to rely on the proposed sIRB and that
any sites added after award will rely on the sIRB.
l Briefly describe how communication between sites and the sIRB will be handled.
l Indicate that all participating sites will, prior to initiating the study, sign an
authorization/reliance agreement that will clarify the roles and responsibilities of the sIRB and
participating sites.
l Indicate which institution or entity will maintain records of the authorization/reliance
agreements and of the communication plan.
F - 110

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
l Note: Do not include the authorization/reliance agreement(s) or the communication plan(s)
documents in your application.
l Note: If you anticipate research involving human subjects but cannot describe the study at
the time of application, include information regarding how the study will comply with the
single Institutional Review Board (sIRB) requirement prior to initiating any multi-site study in
the delayed onset study justification.
For Studies with Legal-, Regulatory-, or Policy-based Claims for Exception as described by
the sIRB Policy: Indicate that review by a sIRB will not be possible for all or some sites (specify
which sites) because local IRB review is required by an existing federal/state/tribal law or policy.
Include a specific citation to the relevant law, policy, or regulation.
For more information:
• AHRQ Guide Notice on Single IRB
• AHRQ Protection of Human Subjects page
3.3 Data and Safety Monitoring Plan
A “Data and Safety Monitoring Plan” attachment is required if you answered “Yes” to all the
questions in the “Clinical Trial Questionnaire.” The “Data and Safety Monitoring Plan” attachment
is optional for all other human subjects research.
For human subjects research that does not involve a clinical trial: Your study, although it is
not a clinical trial, may have significant risks to participants, and it may be appropriate to include
a data and safety monitoring plan. If you choose to include a data and safety monitoring plan,
you may follow the content criteria listed below, as appropriate.
For AHRQ Applicants, Data and Safety Monitoring (DSM) plans are required in all non-exempt
research applications when support is sought to study the effect of a health-related intervention
on outcomes in human subjects where there is greater than minimal risk.
If you seek AHRQ support to conduct non-exempt research to study the effect of a health-
related intervention on outcomes in human subjects where there is greater than minimal risk, a
“Data and Safety Monitoring Plan” attachment is required.
Refer to AHRQ Data and Safety Monitoring Policy
Format:
Attach this information as a PDF file. See NIH’s Format Attachments page.
Content:
Additional Instructions for Fellowship:
Fellowship applicants who are proposing to gain clinical trial research
experience under a sponsor’s supervision (i.e., you will not be leading an
independent clinical trial): Include only the following information in your data and
safety monitoring plan (i.e., do not follow the standard instructions for the data and
safety monitoring plan):
F - 111

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
l The names of the individual(s) or group that will be responsible for trial
monitoring (i.e., the lead investigator of clinical trial)
l If applicable, the name of an independent safety monitor or a data and
safety monitoring board
For any proposed clinical trial, NIH requires a data and safety monitoring plan (DSMP) that is
commensurate with the risks of the trial, its size, and its complexity. Provide a description of the
DSMP, including:
l Indicate how many people and what type of entity will provide the monitoring. Include
such details as whether a single person, multiple people, or a data safety monitoring
board will provide monitoring. Also indicate what type of entity will provide the
monitoring (e.g., PD/PI, Independent Safety Monitor/Designated Medical Monitor,
Independent Monitoring Committee, Safety Monitoring Committee, Data and Safety
Monitoring Board, etc.).
l The overall framework for safety monitoring and what information will be monitored.
l The frequency of monitoring, including any plans for interim analysis and stopping rules
(if applicable).
l The process by which Adverse Events (AEs), including Serious Adverse Events (SAEs) such
as deaths, hospitalizations, and life threatening events and Unanticipated Problems
(UPs), will be managed and reported, as required, to the IRB, the person or group
responsible for monitoring, the awarding IC, the NIH Office of Biotechnology Activities,
and the Food and Drug Administration.
l The individual(s) or group that will be responsible for trial monitoring and advising the
appointing entity. Because the DSMP will depend on potential risks, complexity, and the
nature of the trial, a number of options for monitoring are possible. These include, but
are not limited to, monitoring by a:
o
PD/PI: While the PD/PI must ensure that the trial is conducted according to the
approved protocol, in some cases (e.g., low risk trials, not blinded), it may be
acceptable for the PD/PI to also be responsible for carrying out the DSMP.
o
Independent safety monitor/designated medical monitor: a physician or other expert
who is independent of the study.
o
Independent Monitoring Committee or Safety Monitoring Committee: a small group
of independent experts.
o
Data and Safety Monitoring Board (DSMB): a formal independent board of experts
including investigators and biostatisticians. NIH requires the establishment of DSMBs
for multi-site clinical trials involving interventions that entail potential risk to the
participants, and generally, for all Phase III clinical trials, although Phase I and Phase II
clinical trials may also need DSMBs. If a DSMB is used, please describe the general
composition of the Board without naming specific individuals.
For more information:
l NIH Grants Policy Statement, Section 4.1.15.6: Data and Safety Monitoring
l NIH Policies and IC Guidance for Data and Safety Monitoring of Clinical Trials
F - 112

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
3.4 Will a Data and Safety Monitoring Board be appointed for this study?
The “Data Safety and Monitoring Board” question is required if you answered “Yes” to all the
questions in the “Clinical Trial Questionnaire.” This question is optional for all other human
subjects research.
Check the appropriate box to indicate whether a Data Safety and Monitoring Board (DSMB) will
be appointed for this study.
3.5 Overall Structure of the Study Team
The “Overall Structure of the Study Team” attachment is optional. Refer to your specific FOA for
specific instructions on the "Overall Structure of the Study Team" attachment.
Format:
Attach this information as a PDF file. See NIH’s Format Attachments page.
Content:
Provide a brief overview of the organizational/administrative structure and function of the study
team, particularly the administrative sites, data coordinating sites, enrollment/participating sites,
and any separate laboratory or testing centers. The attachment may include information on
study team composition and key roles (e.g., medical monitor, data coordinating center), the
governance of the study, and a description of how study decisions and progress are
communicated and reported.
Note: Do not include study team members’ individual professional experiences (i.e., biosketch
information).
Section 4 – Protocol Synopsis
Who must complete “Section 4 – Protocol Synopsis:"
If you answered "Yes" to all the questions in the "Clinical Trial Questionnaire:" All the questions
in the “Protocol Synopsis” section are required.
If you answered “No” to any question in the “Clinical Trial Questionnaire:” Do not provide
information in this section. Inputting information in this section will result in errors and will prevent
your application from being accepted.
Additional Instructions for Fellowship:
Fellowship applicants proposing to gain clinical trial research experience under
a sponsor’s supervision (i.e., you will not be leading an independent clinical
trial): Do not provide information in “Section 4 - Protocol Synopsis.” Inputting
information in this section will result in errors and will prevent your application
from being accepted.
F - 113

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
4.1. Study Design
4.1.a. Detailed Description
Enter a narrative description of the protocol. Studies differ considerably in the methods used to
assign participants and deliver interventions. Describe your plans for assignment of participants
and delivery of interventions. You will also need to show that your methods for sample size and
data analysis are appropriate given those plans. For trials that randomize groups or deliver
interventions to groups, special methods are required; additional information is available at the
Research Methods Resources webpage. The Narrative Study Description is not meant to be a
repeat of the Research Strategy.
The narrative description is limited to 32,000 characters (but typically needs only 5,000
characters), should be written in layperson’s terms, and may repeat some of the information in
the Research Strategy.
Note: This field matches a ClinicalTrials.gov field (Detailed Description).
For more information about formatting text entry fields, see NIH's Rules for Text Fields page.
4.1.b. Primary Purpose
Enter or select from the dropdown menu a single "Primary Purpose" that best describes the
clinical trial. Choose from the following options:
l Treatment
l Prevention
l Diagnostics
l Supportive Care
l Screening
l Health Services Research
l Basic Science
l Device Feasibility
l Other (If you select “Other,” provide a description in the space provided. Your response
is limited to 255 characters.)
Note: This field matches a ClinicalTrials.gov field (Primary Purpose).
4.1.c. Interventions
Complete the “Interventions” fields for each intervention to be used in your proposed protocol. If
an arm of the study to which subjects will be assigned (as discussed in 4.1.a. Detailed Description)
includes more than one intervention (e.g., drug plus educational intervention), complete this
section for each intervention. You can add up to 20 interventions.
Intervention Type: Enter or select from the dropdown menu the intervention type the clinical
trial will administer during the proposed award. Choose from the following options:
l Drug (including placebo)
l Device (including sham)
l Biological/Vaccine
l Procedure/Surgery
F - 114

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
l Radiation
l Behavioral (e.g., Psychotherapy, Lifestyle Counseling)
l Genetic (including gene transfer, stem cell, and recombinant DNA)
l Dietary Supplement (e.g., vitamins, minerals)
l Combination Product
l Diagnostic Test
l Other
Name: Enter the name of the intervention. The name is limited to 200 characters.
Description: Enter a description of the intervention. The description is limited to 1,000
characters.
Note: This field matches a ClinicalTrials.gov field. (Interventions, including Intervention Type and
Intervention Name(s)).
For more information on how to answer this question for behavioral research trials, refer to the
relevant FAQ on the Applying Electronically FAQ page.
4.1.d. Study Phase
Enter or select from the dropdown menu a "Study Phase" that best describes the clinical trial. If
your study involves a device or behavioral intervention, choose “N/A”.
Choose from the following options:
l Early Phase 1 (or Phase 0)
l Phase 1
l Phase 1/2
l Phase 2
l Phase 2/3
l Phase 3
l Phase 4
l N/A
Is this an NIH-defined Phase III clinical trial? Yes/No
Select "Yes" or "No" to indicate whether the study includes an NIH-defined Phase III clinical trial.
Device and behavioral intervention studies may select “Yes” here even if the answer above is
“Other”.
For more information on how to answer this question for devices or behavioral interventions,
refer to the relevant FAQ on the Applying Electronically FAQ page.
4.1.e. Intervention Model
Enter or select from the dropdown menu a single "Intervention Model" that best describes the
clinical trial. If you select “Other,” provide a description in the space provided. Choose from the
following options:
l Single Group
l Parallel
F - 115

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
l Cross-Over
l Factorial
l Sequential
l Other (If you select “Other,” provide a description in the space provided. Your response
is limited to 255 characters.)
Note: This field matches a ClinicalTrials.gov field (Interventional Study Model).
For more information: Definitions of intervention models may be found in ClinicalTrials.gov’s
Glossary of Common Site Terms or in the ClinicalTrials.gov’s description of Study Design.
4.1.f. Masking
Select "Yes" or "No" to indicate whether the protocol uses masking. Note that masking is also
referred to as “blinding.”
If you answered “Yes” to the “Masking” question, select one or more types of masking that best
describes the protocol. Choose from the following options:
l Participant
l Care Provider
l Investigator
l Outcomes Assessor
Note: This field matches a ClinicalTrials.gov field (Masking).
4.1.g. Allocation
Enter or select from the dropdown menu a single "Allocation" that best describes how subjects
will be assigned in your protocol. If allocation is not applicable to your clinical trial, select “N/A”
(e.g., for a single-arm trial). Choose from the following options:
l N/A
l Randomized
l Non-randomized
Note: This field matches a ClinicalTrials.gov field (Allocation).
4.2. Outcome Measures
Complete the “Outcome Measures” fields for each primary, secondary, and other important
measures to be collected during your proposed clinical trial. You may have more than one
primary outcome measure, and you can add up to 50 outcome measures.
Name: Enter the name of the individual outcome measure. The outcome measure must be
unique within each Study Record.
Type: Enter or select from the dropdown menu the type of the outcome measure. Choose from
the following options:
l Primary – select this option for the outcome measures specified in your protocol that are
of greatest importance to your study
l Secondary – select this option for outcome measures specified in your protocol that are
of lesser importance to your study than your primary outcomes
F - 116

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
l Other – select this option for additional key outcome measures used to evaluate the
intervention.
Time Frame: Indicate when a measure will be collected for analysis (e.g., baseline, post-
treatment).
Brief Description: Describe the metric used to characterize the outcome measure if the metric is
not already included in the outcome measure name. Your description is limited to 999 characters.
NIH-Defined Phase III Clinical Trials: If the proposed research includes an NIH-Defined Phase
III Clinical Trial, then outcomes for required analyses by sex/gender, race, and ethnicity should be
entered.
Additional information about valid analysis is available on the NIH Policy and Guidelines on The
Inclusion of Women and Minorities as Subjects in Clinical Research page.
Note: This field matches a ClinicalTrials.gov field (e.g., Primary Outcome Measure Information,
which includes Title, Description, and Time Frame).
For more information:
l Refer to the relevant FAQ for question 4.2 Outcome Measures on the Applying
Electronically FAQ page.
4.3. Statistical Design and Power
Format:
Attach this information as a PDF file. See NIH’s Format Attachments page.
Content:
Specify the number of subjects you expect to enroll, the expected effect size, the power, and the
statistical methods you will use with respect to each outcome measure you listed in 4.2 Outcome
Measures.
You will need to show that your methods for sample size and data analysis are appropriate given
your plans for assignment of participants and delivery of interventions. For trials that randomize
groups or deliver interventions to groups, special methods are required; additional information is
available at the Research Methods Resources webpage.
4.4 Subject Participation Duration
Enter the time (e.g., in months) it will take for each individual participant to complete all study
visits. If the participation duration is unknown or not applicable, write “unknown” or “not
applicable.” The subject participation duration is limited to 255 characters.
4.5 Will the study use an FDA-regulated intervention?
Select "Yes" or "No" to indicate whether the study will use an FDA-regulated intervention (see the
definition of “FDA Regulated Intervention” under the Oversight section of the ClinicalTrials.gov
Protocol Registration Data Element Definitions for Interventional and Observational Studies
page).
F - 117

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
4.5.a. If yes, describe the availability of Investigational Product (IP) and Investigational New
Drug (IND)/Investigational Device Exemption (IDE) status:
This attachment is required if you answered “Yes” to the “Will the study use an FDA-regulated
intervention?” question.
Format:
Attach this information as a PDF file. See NIH’s Format Attachments page.
This attachment’s typical length is approximately 3,000 characters.
Content:
Provide a summary describing the availability of study agents and support for the acquisition and
administration of the study agent(s).
Please indicate, if applicable, the IND/IDE status of the study agent, including whether a clinical
investigation is exempt from the IND/IDE requirement. Also indicate whether the investigators
have had any interactions with the FDA (e.g., indicate if the FDA has stated that research may
proceed). If the study agent currently has an IND/IDE number, provide that information.
Do not include the IND/IDE application, manufacturer’s product specifications, study protocol, or
protocol amendments in this attachment.
Additional information such as FDA letters or correspondence with the FDA may be requested in
the FOA.
Note: The awarding component may request consultation with the FDA and the IND/IDE
sponsor about the proposed clinical trial after peer review and prior to award.
4.6 Is this an applicable clinical trial under FDAAA?
Select "Yes" or "No" to indicate whether the study is an applicable clinical trial (ACT) under the
Food and Drug Administration Amendments Act (FDAAA).
For more information:
l NIH Glossary’s definition of an applicable clinical trial
l FAQs on the ClinicalTrials.gov & FDAAA
4.7 Dissemination Plan
Format:
Attach this information as a PDF file. See NIH’s Format Attachments page.
Although one Dissemination Plan per application is sufficient, you must include a file for each
study within your application. All filenames within your application must be unique. You may
either attach the same Dissemination Plan to different studies or attach a file that refers to the
Dissemination Plan in another study within your application. For example, you may attach a file
that says “See Dissemination Plan in the 'My Unique Study Name' study.”
Content:
Explain briefly your plan for the dissemination of NIH-funded clinical trial information and
address how the expectations of the policy will be met. The plan must contain sufficient
F - 118

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
information to assure the following:
l the applicant will ensure that clinical trial(s) under the award are registered and results
information is submitted to ClinicalTrials.gov as outlined in the policy and according to
the specific timelines stated in the policy;
l informed consent documents for the clinical trial(s) will include a specific statement
relating to posting of clinical trial information at ClinicalTrials.gov; and
l the recipient institution has an internal policy in place to ensure that clinical trials
registration and results reporting occur in compliance with policy requirements.
Note: Do not include informed consent documents in the Dissemination Plan attachment.
Note: If your human subjects study meets the definition of “Delayed Onset,” include the
Dissemination Plan attachment in the delayed onset study justification.
For more information:
l See the NIH Policy on the Dissemination of NIH-Funded Clinical Trial Information
l See the NIH Guide Notice on the Delayed Enforcement and Short-Term Flexibilities for
Some Requirements Affecting Prospective Basic Science Studies Involving Human
Participants
l See the NIH Grants Policy Statement, Section 4.1.3.1 NIH Policy on Dissemination of NIH-
Funded Clinical Trial Information.
Section 5 – Other Clinical Trial-related Attachments
Who must complete “Section 5 – Other Clinical Trial-related Attachments:"
If you answered “Yes” to all the questions in the “Clinical Trial Questionnaire:” Include an
attachment only if your FOA specifies that an attachment(s) is required or permitted; otherwise, do
not include any Other Clinical Trial-related attachments.
If you answered “No” to any question in the “Clinical Trial Questionnaire:” Do not provide
information in this section. Inputting information in this section will result in errors and will prevent
your application from being accepted.
Additional Instructions for Fellowship:
Fellowship applicants proposing to gain clinical trial research experience under
a sponsor’s supervision (i.e., you will not be leading an independent clinical
trial): Do not provide information in “Section 5 – Other Clinical Trial-related
Attachments.” Inputting information in this section will result in errors and will
prevent your application from being accepted.
5.1 Other Clinical Trial-related Attachments
Format:
Attach this information as a PDF file. See NIH’s Format Attachments page.
F - 119

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.500 - PHS Human Subjects and Clinical Trials Information
A maximum of 10 PDF attachments is allowed in the “Other Clinical Trial-related Attachments”
section.
Content:
Provide additional trial-related information only if your FOA specifically requests it. Include only
attachments requested in the FOA, and use requested filenames. If a specific filename is not
given in the FOA, use a meaningful filename since it will become a bookmark in the assembled
application image.
F - 120

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.600 - PHS Assignment Request Form
F.600 - PHS Assignment Request Form
The PHS Assignment Request Form may be used to
communicate specific application assignment and
review preferences to the Division of Receipt and
Referral (DRR) and to Scientific Review Officers (SROs).
This information will not be part of your assembled
application, and it will neither be made available to
program staff nor provided to reviewers. It is used
specifically to convey additional, optional information
about your preference(s) for assignment and review of
your application to DRR and SROs.
View larger image
Completing the PHS Assignment Request Form:
This form is optional. Use it only if you wish to communicate
specific awarding component assignments or review preferences. There is no requirement that all
fields or all sections be completed. You have the flexibility to make a single entry or to provide
extensive information using this form.
Note on Application Assignments: The Division of Receipt and Referral (DRR), Center for Scientific
Review (CSR) is responsible for assigning applications to awarding components such as NIH
Institutes/Centers (ICs) and other PHS agencies for funding consideration. DRR also assigns
applications to NIH Scientific Review Groups (SRGs) and Special Emphasis Panels (SEPs).
Awarding Component Assignment Suggestions (optional)
To facilitate accurate communication of any assignment preferences to NIH referral and review
staff, use the short abbreviation (e.g., NCI for the National Cancer Institute).
All assignment suggestions will be considered; however, not all assignment suggestions can be
honored. Applications are assigned based on relevance of your application to an individual
awarding component mission and scientific interests in addition to administrative requirements
such as IC participation in the funding opportunity announcement used to submit your
application.
Descriptions of the scientific areas covered by all NIH ICs and links to other PHS agency
information can be found on the PHS Assignment Information website.
You do not need to make entries in all three boxes of the “Awarding Component Assignment
Suggestions” section.
Suggested Awarding Component(s):
You may enter up to three preferences for primary assignment in the boxes in the “Suggested
Awarding Component(s)” row. Note: Your application will be assigned based on the most
F - 121

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.600 - PHS Assignment Request Form
appropriate match between it, the terms of the FOA, and the mission of each possible awarding
component, with your preference(s) taken into consideration when possible.
Study Section Assignment Suggestions (optional)
To facilitate accurate communication of any review assignment preferences to NIH referral and
review staff, use the short abbreviation of the SRG/SEP you would prefer. For example, enter
“CAMP” for the NIH Cancer Molecular Pathobiology study section or enter “ZRG1HDMR” for the
NIH Healthcare Delivery and Methodologies SBIR/STTR panel for informatics. Be careful to
remove all hyphens, parentheses, and spaces when you type in the suggestion. Freeform text
(such as "special emphasis panel" or "member conflict SEP") should not be entered.
All suggestions will be considered; however, not all assignment suggestions can be honored.
More information about how to identify CSR and NIH SRGs and SEPs, including their short
abbreviations, can be found on CSR Study Sections and Special Emphasis Panel. A list of all NIH
SRGs and SEPs is also available.
While the majority of NIH research grant and fellowship applications are reviewed by CSR, some
are assigned to individual IC review groups and some are clustered for review in SRGs/SEPs,
depending on existing locus of review agreements within NIH and other PHS agencies. This limits
flexibility for honoring assignment preferences.
You do not need to make an entry in all three boxes of the "Study Section Assignment
Suggestions" section.
Suggested Study Sections:
You may enter up to three preferences for SRGs/SEPs in the boxes in the “Suggested Study
Sections” row. Use one box per individual SRG/SEP preference suggestion. All review preferences
will be considered. Note: Your application will be assigned based on the most appropriate match
between it, the terms of the FOA, and the guidelines for each SRG/SEP, with your preference(s)
taken into consideration when possible.
Note: This information is not applicable if you are submitting an application to an RFA.
Rationale for assignment suggestions (optional)
Enter the rationale (i.e., why you think the assignment is appropriate) for your Awarding
Component and Study Section suggestions.
Your answer can have a maximum of 1000 characters.
List individuals who should not review your application and why (optional)
You may list specific individuals, if any, who should not review your application and why they
should not review your application. Provide sufficient information (e.g., name, organizational
affiliation) so that the SRO can correctly identify the individual. Be prepared to provide additional
information to the SRO if needed. Simply stating “Dr. John Smith is in conflict with my application”
is not helpful.
Your answer can have a maximum of 1000 characters.
F - 122

Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series
F.600 - PHS Assignment Request Form
Identify scientific areas of expertise needed to review your application (optional)
You may list up to five general or specific types of expertise needed for the review of your
application. Limit your answers to areas of expertise – do not enter names of individuals you
would like to review your application.
Each field can have a maximum of 40 characters.
F - 123
Form Screenshots
F.- i
Form Screenshots
Quick Links
SF 424 (R&R) Form
R&R Other Project Information Form
Project/Performance Site Location(s) Form
R&R Senior/Key Person Profile (Expanded) Form
PHS Fellowship Supplemental Form
PHS Human Subjects and Clinical Trials Information
PHS Assignment Request Form
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

Form Screenshots
F.- ii
SF 424 (R&R)Form
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

Form Screenshots
F.- iii
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

Form Screenshots
F.- iv
R&R Other Project Information Form
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

Form Screenshots
F.- v
Project/Performance Site Location(s) Form
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

Form Screenshots
F.- vi
R&R Senior/Key Person Profile (Expanded) Form
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

Form Screenshots
F.- vii
PHSFellowship Supplemental Form
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

Form Screenshots
F.- viii
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

Form Screenshots
F.- ix
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

Form Screenshots
F.- x
PHS Human Subjects and Clinical Trials Information
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

Form Screenshots
F.- xi
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

Form Screenshots
F.- xii
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

Form Screenshots
F.- xiii
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

Form Screenshots
F.- xiv
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

Form Screenshots
F.- xv
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

Form Screenshots
F.- xvi
PHS Assignment Request Form
Fellowship Instructions for NIH and Other PHS Agencies - Forms Version F Series

