
General Best Practice Guidelines for Immunization: Introduction
Suggested citation: Kroger A, Bahta L, Long S, Sanchez P. General Best Practice Guidelines for Immunization.
[www.cdc.gov/vaccines/hcp/acip-recs/general- recs/downloads/general-recs.pdf]. Accessed on [DATE].
1
–
General Best Practice Guidelines for
Immunization
BEST PRACTICES GUIDANCE
Kroger A, Bahta L, Long S, Sanchez P

General Best Practice Guidelines for Immunization: Introduction
Suggested citation: Kroger A, Bahta L, Long S, Sanchez P. General Best Practice Guidelines for Immunization.
[www.cdc.gov/vaccines/hcp/acip-recs/general- recs/downloads/general-recs.pdf]. Accessed on [DATE].
2
General Best Practice Guidelines for
Immunization
Kroger A, Bahta L, Long S, Sanchez P
1. Introduction
The Centers for Disease Control and Prevention (CDC) recommends routine vaccination
to prevent 17 vaccine-preventable diseases that occur in infants, children, adolescents, or
adults. This report provides information for clinicians and other health care providers
about concerns that commonly arise when vaccinating persons of various ages. Providers
and patients must navigate numerous issues, such as the timing of each dose, screening
for contraindications and precautions, the number of vaccines to be administered, the
educational needs of patients and parents, and interpreting and responding to adverse
events. Vaccination providers help patients understand the substantial body of
(occasionally conflicting) information about vaccination.
This vaccination best practice guidance is intended for clinicians and other health care
providers who vaccinate patients in varied settings, including hospitals, provider offices,
pharmacies, schools, community health centers, and public health clinics. The updated
guidelines include 1) new information on simultaneous vaccination and febrile seizures;
2) enhancement of the definition of a “precaution” to include any condition that might
confuse diagnostic accuracy; 3) confirmation that if a patient is not acutely moderately or
severely ill, vaccination during hospitalization is a best practice; 4) more descriptive
characterization of anaphylactic allergy; 5) incorporation of protocols for management of
anaphylactic allergy; 6) allowances for alternate route (subcutaneous instead of
intramuscular) for hepatitis A vaccination; 7) an age cutoff of 12 years through 17 years of
age for validating a dose of intradermal influenza vaccine; 8) deletion of much of the

General Best Practice Guidelines for Immunization: Introduction
Suggested citation: Kroger A, Bahta L, Long S, Sanchez P. General Best Practice Guidelines for Immunization.
[www.cdc.gov/vaccines/hcp/acip-recs/general- recs/downloads/general-recs.pdf]. Accessed on [DATE].
3
content from storage and handling, including storage units, temperature monitoring, and
expiration dates (because this content is now codified and continually updated in the
CDC’s Vaccine Storage and Handling Toolkit, available at
https://www.cdc.gov/vaccines/hcp/admin/storage/toolkit/index.html.); 9) incorporation
of Infectious Diseases Society of America guidance on vaccination of persons with altered
immunocompetence; 10) timing of intramuscular administration in patients with
bleeding disorders; 11) updated data on vaccination record policy; 12) additional impacts
of the Affordable Care Act (1,2) on adult vaccination; and 13) updated programmatic
contact information on source material for vaccine information.
The guidance is organized in the following 10 documents: 1) Timing and Spacing of
Immunobiologics; 2) Contraindications and Precautions; 3) Preventing and Managing
Adverse Reactions; 4) Vaccine Administration; 5) Storage and Handling of
Immunobiologics; 6) Altered Immunocompetence; 7) Special Situations; 8) Vaccination
Records; 9) Vaccination Programs; and 10) Vaccine Information Sources. A glossary
follows (see Appendix 1: Glossary).
This report will help vaccination providers to assess vaccine benefits and risks, use
recommended administration practices, understand the most effective strategies for
ensuring that vaccination coverage in the population remains high, and communicate the
importance of vaccination to reduce the effects of vaccine-preventable disease. These best
practice guidelines are intended for use in the United States; vaccine availability, use, and
epidemiologic circumstances might differ in other countries and might warrant different
guidance.

General Best Practice Guidelines for Immunization: Introduction
Suggested citation: Kroger A, Bahta L, Long S, Sanchez P. General Best Practice Guidelines for Immunization.
[www.cdc.gov/vaccines/hcp/acip-recs/general- recs/downloads/general-recs.pdf]. Accessed on [DATE].
4
REFERENCES
1.
The Patient Protection and Affordable Care Act, Pub. L. No. 111-148 (2010).
2.
U.S. Department of Health and Human Services. Read the law: the Affordable
Care Act, section by section. 2015; https://www.hhs.gov/healthcare/about- the-
law/read-the-law/index.html. Accessed 9 March, 2017.

General Best Practice Guidelines for Immunization: History
5
2. History
The best practice guidelines in this report update the previous ACIP General
Recommendations on Immunization (1) and are based both on review and analysis of
available scientific evidence and on expert opinion of the diverse group of health-care
providers and public health officials who are members of GRWG. This group includes
professionals from academic medicine (pediatrics, family practice, and pharmacy);
international (Canada), federal, and state public health professionals; and a member
from the nongovernmental Immunization Action Coalition (see Appendix 2:
Membership). This revision involved consensus-building based on new evidence from
the published literature and opinion from subgroups of subject matter experts consulted
on specific topics.
The process by which the guidelines were drafted varied for each document; each
document is therefore discussed individually below. ACIP voted to accept the proposed
guidance in October 2014; for additional information, see
www.cdc.gov/vaccines/acip/meetings/meetings-info.html.

General Best Practice Guidelines for Immunization: History
6
Timing and Spacing of Immunobiologics
GRWG met monthly beginning in January 2011, and formed a subgroup to focus on
review of guidelines around administration of simultaneous vaccination and febrile
seizures. Meetings were held in April, May, and June 2011 to discuss the evidence. Other
issues related to timing and spacing of vaccinations were discussed between February
2012 and September 2014 over 7 meetings (in February 2012, June 2012, August 2012,
November 2012, January 2013, January 2014, May 2014, and September 2014). The
evidence supporting this document is based on expert opinion and arrived at by
consensus. Presentations of the evidence were made to ACIP in June 2011, October
2011, and February 2013. Major changes include 1) guidance for simultaneous
vaccination in the context of a risk for febrile seizures and 2) clarification of the use of
the grace period between doses of the measles, mumps, rubella, and varicella vaccine
(MMRV).
Contraindications and Precautions
GRWG met monthly and focused on revisions to the Contraindications and Precautions
section beginning in January 2012, over 6 meetings (January 2012, February 2012, June
2012, August 2012, November, 2012, December 2012, and January 2013; see
www.cdc.gov/vaccines/acip/meetings/meetings-info.html). The evidence supporting
this document is based on a review of the published literature. Publications about
vaccination during surgery, hospitalization, and anesthesia were obtained from the
databases PubMed and MDConsult, searched from 1973 to 2014 using the MeSH
(medical subject headings) terms “anesthesia” and “immunization”. The search and
selection of studies was limited to English-language and human studies. The search and
selection process yielded 20 publications, including review articles, observational
studies, and letters to the editor. Presentations of proposed best practices were made to
ACIP in February 2013 and a vote from ACIP affirming the language below was made in
October 2014. Major changes include 1) enhancement of the definition of a “precaution”
to include any condition that might confuse diagnostic accuracy and 2) guidance to
vaccinate during a hospitalization if a patient is not acutely moderately or severely ill.

General Best Practice Guidelines for Immunization: History
7
Preventing and Managing Adverse Reactions
GRWG met monthly and focused on revisions to the Preventing and Managing Adverse
Reactions section beginning in April 2013, following revision to the document by the
Allergy Subgroup. Selected members from this subgroup participated in the April 2013
main work group call. GRWG then met again in May 2013. The evidence supporting this
document is based on expert opinion and arrived at by consensus. Presentations of
proposed guidance were made to ACIP in June 2013, and a vote from ACIP affirming the
language below was made in October 2014. Major changes included 1) more descriptive
characterization of anaphylactic allergy and 2) incorporation of protocols for managing
adverse reactions. ACIP voted to accept the proposed statement in October 2014.
Vaccine Administration
An addition to the 2017 Vaccine Administration best practices was presented to ACIP on
October 16, 2018, about health care provider exposure to vaccine components.
Storage and Handling of Immunobiologics
GRWG met in December 2013 to discuss Storage and Handling of Immunobiologics and
met one additional time in January 2014. The evidence supporting this document is
based on expert opinion and arrived at by consensus. A presentation of proposed
language was made to ACIP in February 2014, and a vote from ACIP approving the
language below was made in October 2014. Most of the 2011 language was removed
because this content is now codified and continually updated in the CDC’s Vaccine
Storage and Handling Toolkit, available at
www.cdc.gov/vaccines/hcp/admin/storage/toolkit/index.html. This content included
Storage Units, Monitoring Storage Temperature, Vaccine Inventory, and Vaccine
Transport.
General Best Practice Guidelines for Immunization: History
8
Altered Immunocompetence
GRWG met twice in March and April 2014 to discuss best practices guidance for Altered
Immunocompetence. This section incorporates general content from the Infectious
Diseases Society of America (IDSA) policy statement 2013 IDSA Clinical Practice
Guideline for Vaccination of the Immunocompromised Host (2), to which CDC
provided input in November 2011. The evidence supporting this document is based on
expert opinion and arrived at by consensus. ACIP voted to accept this proposed
statement in June 2015.
Special Situations
GRWG met in April 2012 and then in 4 follow-up meetings in May, August, and
November 2012, and January 2013. A focal point of discussion involved best practices
guidance for intramuscular administration of persons with increased bleeding risk.
Subject matter experts from the National Center for Birth Defects and Developmental
Disabilities (NCBDDD) were invited to a work group meeting, and revisions to the
guidance involving the timing of intramuscular administration were made in
collaboration with these subject matter experts, primarily to ensure that ACIP’s best
practices guidance does not conflict with NCBDDD recommendations regarding the
timing of clotting factor deficiency replacement. The evidence supporting this
document is based on expert opinion and arrived at by consensus.
GRWG presented the Special Situations section to ACIP in February 2013. ACIP voted to
accept the proposed statement in June 2015.
General Best Practice Guidelines for Immunization: History
9
Vaccination Records
GRWG met in August and September 2013, and presented the vaccination records
language to ACIP in October 2013. The evidence supporting this document is based on
expert opinion and arrived at by consensus. ACIP voted to accept this proposed best
practices guidance in June 2015.
Vaccination Programs
GRWG met in April 2014. The major revision to this section is the addition of language
related to Affordable Care Act (3,4) coverage of adult vaccination. The evidence
supporting this document is based on expert opinion and arrived at by consensus.
GRWG presented this section to ACIP in June 2014. ACIP voted to accept this proposed
statement in June 2015.
Vaccination Information Sources
GRWG met in September 2014 and presented this section to ACIP. The evidence
supporting this document is based on expert opinion and arrived at by consensus. ACIP
voted to accept this proposed statement in June 2015.
General Best Practice Guidelines for Immunization: History
10
REFERENCES
1.
Kroger AT, Sumaya CV, Pickering LK, Atkinson WL. General recommendations
on immunization: recommendations of the Advisory Committee on Immunization
Practices (ACIP). MMWR Recomm Rep. 2011:1- 60.
2.
Rubin L, Levin M, Ljungman P, et al. 2013 IDSA clinical practice guideline for
vaccination of the immunocompromised host. Clin Infect Dis.
2014;58(3):e44-100. DOI: 10.1093/cid/cit684
3.
The Patient Protection and Affordable Care Act, Pub. L. No. 111-148 (2010).
4.
U.S. Department of Health and Human Services. Read the law: the Affordable
Care Act, section by section. 2015; https://www.hhs.gov/healthcare/about- the-
law/read-the-law/index.html. Accessed 9 March, 2017.
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
11
3. Timing and Spacing of Immunobiologics
Updates
Major changes to the best practice guidance for timing and spacing of immunobiologics
include 1) guidance for simultaneous vaccination in the context of a risk for febrile seizures
and 2) clarification of the use of the grace period between doses of MMRV.
General Principles for Vaccine Scheduling
Optimal response to a vaccine depends on multiple factors, including the type of vaccine, age
of the recipient, and immune status of the recipient. Recommendations for the age at which
vaccines are administered are influenced by age-specific risks for disease, age-specific risks
for complications, age-specific responses to vaccination, and potential interference with the
immune response by passively transferred maternal antibodies. Vaccines are generally
recommended for members of the youngest age group at risk for experiencing the disease for
which vaccine efficacy and safety have been demonstrated.
Vaccines can be divided into two general categories: live or non-live. Most live vaccines used
in the United States are “live attenuated”, meaning that the microbe in the vaccine is alive but
has been weakened (attenuated) through serial passage in cultures, or produced through
genetic technology. One live vaccine licensed for use in the United States (oral adenovirus
vaccine) is not attenuated. Live vaccines must replicate in order to induce an immune
response. Several factors can interfere with replication, and these are a driving factor in the
principles of timing and spacing of live vaccine. These principles will be discussed later in
this chapter. Non-live vaccines can include whole or fractional vaccines produced by
inactivating (killing) the microbe; or fractional vaccines such as recombinant vaccines,
produced through genetic technologies. Also included under the category of non-live vaccines
are toxoids, which generate an antibody response to toxins produced by a microbe rather
than to the microbe itself. Several recently developed non-live vaccines do not contain
antigen but employ RNA or DNA to instruct the recipient’s own cellular mechanism to
generate antigenic material. Some vaccines described as “live attenuated” (e.g., Jynneos
Smallpox/Monkeypox vaccine) do not replicate and for the purposes of timing and spacing
recommendations behave like non-live vaccines.

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
12
Certain vaccines available outside the U.S. might be categorized differently as to vaccine type
(e.g., live attenuated JE vaccine and live oral polio vaccine) (Table 3-1).
Tetanus and diphtheria toxoids require booster doses to maintain protective antibody
concentrations (1). Unconjugated polysaccharide vaccines do not induce T-cell memory, and
additional doses (although they elicit the same or a lower antibody concentration) might
increase the duration of protection. Conjugation with a protein carrier improves the
effectiveness of polysaccharide vaccines by inducing T-lymphocyte–dependent immunologic
function (2). Many vaccines that stimulate both cell-mediated immunity and neutralizing
antibodies (e.g., live, attenuated virus vaccines) can usually induce prolonged immunity, even
if antibody titers decline over time (3). Subsequent exposure to such viruses usually results in
a rapid anamnestic antibody response without viremia.
Approximately 90%-95% of recipients of a single dose of certain live vaccines administered
by injection at the recommended age (i.e., measles, rubella, and yellow fever vaccines)
develop protective antibodies, generally within 14 days of the dose. For varicella and mumps
vaccines, 80%-85% of vaccines are protected after a single dose. However, because a limited
proportion (5%-20%) of measles, mumps, and rubella (MMR) or varicella vaccinees fail to
respond to 1 dose, a second dose is recommended to provide another opportunity to develop
immunity (4).
Of those who do not respond to the first dose of the measles component of MMR or varicella
vaccine, 97%-99% respond to a second dose (5,6).
The Recommended Immunization Schedules for Persons Aged 0 Through 18 Years and
the Recommended Adult Immunization Schedule are revised annually. Physicians and other
health-care providers should ensure that they are following the most up-to-date schedules, which
are available from CDC at
www.cdc.gov/vaccines/schedules/hcp/index.html.
Spacing of Multiple Doses of the Same Antigen
Vaccination providers should adhere to recommended vaccination schedules (Table 3-2).
Administration at recommended ages and in accordance with recommended intervals
between doses of multidose antigens provides optimal protection.

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
13
Administration of doses of a multidose vaccine using intervals that are shorter than
recommended might be necessary in certain circumstances, such as impending international
travel or when a person is behind schedule on vaccinations but needs rapid protection. In
these situations, an accelerated schedule can be implemented using intervals between doses
that are shorter than intervals recommended for routine vaccination (7). The accelerated or
minimum intervals and ages for scheduling catch-up vaccinations are available at
www.cdc.gov/vaccines/schedules/hcp/index.html. Vaccine doses should not be administered
at intervals less than these minimum intervals or at an age that is younger than the minimum
age.*
(a)
Before administering a vaccine dose, providers might need to verify that all previous doses
were administered after the minimum age and in accordance with minimum intervals (Table
3-2). In clinical practice, vaccine doses occasionally are administered at intervals less than
the minimum interval or at ages younger than the minimum age. Doses administered too
close together or at too young an age can lead to a suboptimal immune response. However,
administering a dose a few days earlier than the minimum interval or age is unlikely to have a
substantially negative effect on the immune response to that dose. Known as the “grace
period”, vaccine doses administered ≤4 days before the minimum interval or age are
considered valid; however, local or state mandates might supersede this 4-day guideline
(7).(b) (Day 1 is the day before the day that marks the minimum age or minimum interval for
a vaccine.) The scenario most applicable to the grace period is a visit to a provider several
days prior to the date indicated by the minimum interval, such as for a mild illness. Follow-
up is unlikely soon after or even for a longer period of time following this mild illness visit;
this therefore raises the question of whether vaccines be administered during the mild illness
visit to avoid missed opportunities to vaccinate. Because of the unique schedule for rabies
vaccine and the accelerated Twinrix schedule the 4-day guideline does not apply to this
vaccine (8, personal communication, Hepatitis A subject matter experts). Doses of any
vaccine administered ≥5 days earlier than the minimum interval or age should not be
counted as valid doses and should be repeated as age appropriate. The repeat dose should be
spaced after the invalid dose by the recommended minimum interval (Table 3-2). For
example, if the first and second doses of Haemophilus influenzae type b (Hib) were
administered only 14 days apart, the second dose would be invalid and need to be repeated
because the minimum interval from dose 1 to dose 2 is 4 weeks.
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
14
The repeat dose should be administered ≥4 weeks after the invalid dose (in this case, the
second) (7). The repeat dose is counted as the valid second dose. If the first and second doses
of hepatitis A vaccine were administered less than 6 months apart, the second dose is invalid
and should be repeated 6 months after the invalid second dose (7). However, if this repeat
dose (the third dose) is administered anytime 6 months or more after the first dose, the
series can be considered complete. If the minimum interval between the second and third
dose of hepatitis B vaccine is violated, or if the minimum age of the third dose is violated, the
third dose of hepatitis B vaccine is invalid. The repeat dose can be administered as early as 8
weeks after the 2
nd
valid dose as long as the dose is also after 24 weeks of age and 16 weeks
after the 1
st
dose. (9)
If the first dose in a series is given ≥5 days before the recommended minimum age, the dose
should be repeated on or after the date when the child reaches at least the minimum age (7).
If the vaccine is a live vaccine, ensuring that a minimum interval of 28 days has elapsed from
the invalid dose is recommended (7). For example, if the first dose of varicella vaccine were
inadvertently administered at age 10 months, the repeat dose would be administered no
earlier than the child’s first birthday (the minimum age for the first dose). If the first dose of
varicella vaccine were administered at age 11 months and 2 weeks, the repeat dose should be
administered no earlier than 4 weeks thereafter, which would occur after the first birthday.
Certain vaccines (e.g., adult tetanus and diphtheria toxoids [Td], pediatric diphtheria and
tetanus toxoids [DT], tetanus toxoid) produce increased rates of local or systemic reactions in
certain recipients when administered more frequently than recommended (10, 11).
Careful record keeping, maintenance of patient histories, use of immunization information
systems (IISs), and adherence to recommended schedules can decrease the incidence of such
reactions without adversely affecting immunity.
Simultaneous Administration
Simultaneous administration of vaccines is defined as administering more than one vaccine
on the same clinic day, at different anatomic sites, and not combined in the same syringe.
Experimental evidence and extensive clinical experience provide the scientific basis for
administering vaccines simultaneously (12).
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
15
Simultaneously administering all vaccines for which a person is eligible at the time of a visit
increases the probability that a child, adolescent, or adult will be vaccinated fully by the
appropriate age (13). A study conducted during a measles outbreak demonstrated that
approximately one-third of measles cases among unvaccinated but vaccine-eligible preschool
children might have been prevented if MMR had been administered at the same visit when
another vaccine was administered (14). Simultaneous administration also is critical when
preparing for foreign travel in the near future and when a health-care provider is uncertain
that a patient will return for additional doses of vaccine.
With some exceptions, simultaneously administering the most widely used live and non-live
vaccines has produced seroconversion rates and rates for adverse reactions similar to those
observed when the vaccines are administered separately (12, 15-17). Routine administration
of all age-appropriate doses of vaccines simultaneously is recommended for children for
whom no specific contraindications exist at the time of the visit (7). MMR and varicella
vaccine can be administered simultaneously (7). Live, attenuated influenza vaccine (LAIV)
does not interfere with the immune response to MMR or varicella vaccines administered at
the same visit (18). No data exist about the immunogenicity of oral Ty21a typhoid vaccine
when administered concurrently or within 30 days of live-virus vaccines. In the absence of
such data, if typhoid vaccination is warranted, administration should not be delayed because
of recent administration of live, attenuated virus vaccines (19).
Simultaneous administration of pneumococcal polysaccharide vaccine (PPSV) and
inactivated influenza vaccine elicits a satisfactory antibody response without increasing the
incidence or severity of adverse reactions (20). Simultaneous administration of PPSV23 and
inactivated influenza vaccine is recommended for all persons for whom both vaccines are
indicated. Tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis vaccine (Tdap)
and inactivated influenza vaccine (IIV) can be administered simultaneously (21). Hepatitis B
vaccine administered with yellow fever vaccine is as safe and immunogenic as when these
vaccines are administered separately (22). Measles and yellow fever vaccines have been
administered safely at the same visit and without reduction of immunogenicity of either
component (23,24).
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
16
During the 2010-2011 influenza season, surveillance systems detected safety signals for
febrile seizures in young children following IIV and PCV13 vaccines (25). CDC studied the
health-care visit records of more than 200,000 vaccinated children ages 6 months through
59 months through the Vaccine Safety Datalink Project during the 2010-2011 influenza
season. The analyses found that febrile seizures following IIV and PCV13 vaccines given to
this age group were rare, but did occur at higher than expected rates. The risk for febrile
seizures peaked in children age 16 months and were more common when the 2 vaccines were
given during the same health-care visit. In this group, about one additional febrile seizure
occurred among every 2,200 children vaccinated. After assessing benefits and risks, ACIP
continues to recommend IIV and PCV13 be given concomitantly if both are recommended
(25,26).
There are 2 exceptions to the recommendation that vaccines should be administered
simultaneously. In persons with anatomic or functional asplenia and/or HIV infection,
quadrivalent meningococcal conjugate vaccine (MCV4)-D (MenACWY-D, Menactra) and
pneumococcal conjugate vaccine (PCV)13 (PCV13, Prevnar 13) should not be administered
simultaneously (27). This is based on immunogenicity studies that showed reduced antibody
concentrations for 3 serotypes of pneumococcus (subtypes 4, 6B, and 18C) when PCV7 was
administered simultaneously with MenACWY-D. For persons with anatomic or functional
asplenia and/or HIV, PCV13 should be administered first and MenACWY-D 4 weeks later.
In patients recommended to receive both PCV13 and PPSV23, the 2 vaccines should not be
administered simultaneously (28). PCV13 should be administered first. If PPSV23 has been
administered first, PCV13 should be administered no earlier than 8 weeks later in children 6-
18 years, and one year later in adults 19 years and older. Immunogenicity studies evaluating
responses to PCV13 and PPSV23 administered in series showed a better immune response
when PCV13 was administered first. An evaluation of immune response after a second
pneumococcal vaccination administered 1 year after the initial study doses showed that
subjects who received PPSV23 as the initial study dose had lower antibody responses after
subsequent administration of PCV13 than those who had received PCV13 as the initial dose
followed by a dose of PPSV23, regardless of the level of the initial response to PPSV23
(29,30).

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
17
Depending on which vaccines are administered during the first year of life, a child might
receive up to 9 injections at the 12- through 15-month visit (MMR, varicella, Hib, PCV13,
pediatric diphtheria and tetanus toxoids and acellular pertussis [DTaP], inactivated
poliovirus [IPV], hepatitis A, hepatitis B [HepB], and influenza vaccines). Although there is
no exact limit on the number of injections, with a little flexibility, a provider can ensure that
the primary series doses are given without administering too many injections at each visit. To
reduce the number of injections at the 12- through 15-month visit, the hepatitis B series and 3
doses of IPV (31) can be administered before the child’s first birthday.
There are many other examples of ways the vaccination schedule provides flexibility. The
majority of children aged 1 year who have received 2 Hib vaccine doses (polyribosylribitol
phosphate-meningococcal outer membrane protein [PRP-OMP]) or 3 Hib vaccine doses
(PRP-tetanus [PRP-T]) and 3 previous doses of DTaP and PCV13 have protection against
Hib, diphtheria, pertussis, tetanus, and pneumococcus, which lasts throughout infancy
(2, 32). The third (PRP-OMP) or fourth (PRP-T) dose of the Hib series and the fourth doses
of DTaP and PCV13 are critical in boosting antibody titer and ensuring continued protection
(2, 33-35). The fourth dose of DTaP is recommended at age 15-18 months but may be
administered as early as age 12 months if 6 months have elapsed since the third dose and if
there is concern that the child might not return by age 18 months (33). For infants at low risk
for infection with hepatitis B virus (i.e., mother tested negative for hepatitis B surface antigen
[HBsAg] at the time of delivery and is not in a high risk group), the hepatitis B series can be
completed at any time for children aged 6-18 months (36). The minimum age for
administration of combination vaccines is the oldest minimum age for any of the individual
components; the minimum interval between doses is equal to the greatest minimum interval
of any of the individual components. Recommended spacing of doses should be
maintained (Table 3-2).
Combination Vaccines
Combination vaccines merge equivalent component vaccines into single products to prevent
more than one disease or to protect against multiple strains of infectious agents causing the
same disease. Licensed combination vaccines can be used whenever any components of the
combination are indicated and its other components are not contraindicated and if licensed
by the Food and Drug Administration (FDA) for that dose in the series.
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
18
Use of combination vaccines can reduce the number of injections patients receive and
alleviate concern associated with the number of injections (29,37,38). Studies have
demonstrated that parents and providers might be uncomfortable with multiple injections
during single visits (39-41). Potential advantages of combination vaccines include 1)
improved vaccine coverage rates (42), 2) timely vaccination coverage for children who are
behind in the schedule (43, 44), 3) reduced shipping and stocking costs, 4) reduced costs for
extra health care visits necessitated by deferral of vaccination, and 5) facilitation of
additional new vaccines into vaccination programs.
Potential disadvantages of combination vaccines include the following: 1) adverse events that
might occur more frequently after administration of a combination vaccine compared with
administration of separate antigens at the same visit, such as fever that occurs with the
combination MMRV vaccine and combination DTaP-HepB-IPV vaccine (45,46); 2) confusion
and uncertainty about selection of vaccine combinations and schedules for subsequent doses,
especially when vaccinations are given by multiple providers who might be using different
products; 3) reduced pathogen coverage if the combination product covers fewer types of one
particular vaccine-preventable disease-causing agent (47); 4) extra doses of certain antigens
in the combination product (e.g., a provider who administers 4 doses of DTaP-HepB-IPV
vaccine will give an extra dose of hepatitis B component); and 5) a shorter shelf-life than the
individual component vaccines. The economic impact of the use of combination vaccines is
unclear because combination products have the potential for either increased or decreased
costs compared with single-antigen component vaccines. The price of a combination vaccine
might exceed the total price of separate vaccines containing the same antigens. However,
combination vaccines might represent a better overall economic value if the direct and
indirect costs of extra injections, delayed or missed vaccinations, and additional handling
and storage are taken into consideration (48).
Licensed Combination Vaccines
In this report, a combination vaccine is defined as a product containing components that can
be divided equally into independently available routine vaccines. A dash ( – ) between
vaccine products indicates that products are supplied in their final form by the manufacturer
and do not require mixing or reconstitution by the user. A slash ( / ) indicates that the
products must be mixed or reconstituted by the user.

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
19
Seven combination vaccines for which separate antigens or antigen combinations exist have
been licensed by FDA since 1996 in the United States (Table 3-3) (49-55). In the future,
combination vaccines might include increasing numbers of components in different arrays to
protect against these and other diseases. The use of a combination vaccine generally is
preferred over separate injections of the equivalent component vaccines (56). Considerations
should include provider assessment,(c) patient preference, and the potential for adverse
events. An exception is the first dose of MMRV. Unless the parent or caregiver expresses a
preference for MMRV vaccine, MMR and varicella vaccine should be administered for the
first dose for children aged 12-47 months (see Contraindications and Precautions) (45).
Situations might arise in which one component of a combination vaccine is specifically
preferred to another component in that same vaccine. Future research considerations for
newly licensed combination vaccines should focus on safety of doses that are not needed
because a patient is already vaccinated against the agents, whether the combination vaccine
will improve the timeliness of vaccination, and potential reduced costs from disease
prevention resulting from timely vaccination.
Combination Vaccines and FDA Licensure
Only combination vaccines licensed by FDA should be used (56). Vaccination providers
should not combine separate vaccines into the same syringe to administer together unless
mixing is indicated for the patient’s age and is explicitly specified on the FDA-approved
product label inserts. Only 2 combination vaccines, (DTaP-IPV/Hib vaccine, marketed as
Pentacel, and Hib-MenCY, marketed as MenHibrix) contain separate antigen components for
which FDA approves mixing by the user. The safety, immunogenicity, and effectiveness of
unlicensed combinations are unknown.
Interchangeability of Formulations
FDA generally licenses a combination vaccine based on studies demonstrating that the
product’s immunogenicity (or efficacy) and safety are comparable or equivalent to
monovalent or combination products licensed previously (38).
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
20
FDA licensure also generally indicates that a combination vaccine may be used
interchangeably with monovalent formulations and other combination products with similar
component antigens produced by the same manufacturer to continue the vaccination series.
For example, DTaP, DtaP-IPV/Hib, DtaP-HepB-IPV, and future DtaP vaccines that contain
similar acellular pertussis antigens from the same manufacturer may be used
interchangeably if licensed and indicated for the patient’s age (35).
Interchangeability of Combination Vaccines from Different
Manufacturers
Licensure of a vaccine by FDA does not necessarily indicate that the vaccine is
interchangeable with products from other manufacturers. Such data are ascertained and
interpreted more readily for diseases with known correlates of protective immunity (e.g.,
specific serologic markers). For diseases without such surrogate laboratory markers,
prelicensure field vaccine efficacy (phase III) trials or postlicensure surveillance generally are
required to determine protection (57). ACIP prefers that doses of vaccine in a series come
from the same manufacturer; however, if this is not possible or if the manufacturer of doses
given previously is unknown, providers should administer the vaccine that they have
available.
Vaccine Supply
Although vaccination providers should stock sufficient quantities of combination and
monovalent vaccines needed to vaccinate children, adolescents, and adults against all
diseases for which vaccines are recommended (29, 38), all available types or brand-name
products need not be stocked. Potential advantages of stocking a limited number of vaccines
include 1) reducing confusion and potential errors when staff members must handle
redundant products and formulations, 2) minimizing waste when less commonly used
products expire, 3) decreasing cold storage capacity requirements, and 4) minimizing
administrative costs related to accounting, purchasing, and handling. The National Pediatric
Vaccine Stockpile exists to offset supply challenges (58).
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
21
Extra Doses of Vaccine Antigens
Administering extra antigens contained in a combination vaccine should be avoided in most
situations (56). Using combination vaccines containing certain antigens not indicated at the
time of administration to a patient might be justified when 1) the extra antigen is not
contraindicated, 2) products that contain only the needed antigens are not readily available,
and 3) potential benefits to the patient outweigh the potential risk for adverse events
associated with the extra antigens. An extra dose of many live-virus vaccines and Hib or
hepatitis B vaccine has not been found to be harmful (59,60). However, the risk for an
adverse event might increase when extra doses are administered at an earlier time than the
recommended interval for certain vaccines (e.g., tetanus toxoid vaccines and PPSV)
(20, 33, 61).
A vaccination provider might not have vaccines available that contain only the antigens
needed as indicated by a child’s vaccination history. Alternatively, although the indicated
vaccines might be available, the provider might prefer to use a combination vaccine to reduce
the required number of injections. In such cases, the benefits and risks of administering the
combination vaccine with an unneeded antigen should be carefully considered and discussed
with the patient or parent.
When non-live vaccines (which are often adsorbed to aluminum-salt adjuvants) are
administered, the reactogenicity of the vaccine must be considered in balancing the benefits
and risks of extra doses (56). Because clinical experience suggests low reactogenicity, an
extra dose of Hib or hepatitis B vaccine may be administered as part of a combination
vaccine to complete a vaccination series for another component of the combination.
Administration of extra doses of tetanus toxoid vaccines earlier than the recommended
intervals can increase the risk for severe local reactions (21, 34). Examples of such vaccines
include DtaP, DT (for children), and Td (for adolescents and adults). Extra doses of tetanus-
toxoid–containing vaccines might be appropriate for certain patients, including for children
who previously received DT or Td vaccine and need protection from pertussis (in DtaP or
Tdap) or for immigrants with uncertain vaccination histories.

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
22
Conjugate Vaccine Carrier Proteins
Protein conjugates used in Hib conjugate vaccines produced in the United States include
tetanus toxoid (in PRP-T) which is also used as a component of DtaP and Tdap vaccines (21).
Simultaneous or sequential vaccination with Hib and these tetanus-toxoid containing
vaccines is recommended when both are indicated (56). MCV4 and PCV13 both contain
diphtheria-toxoid conjugates. There has been concern about simultaneous administration of
vaccines containing like conjugates. One brand of MCV4, MenACWY-D (Menactra),
demonstrates reduced immunogenicity of the antibody response to Streptococcal pneumonia
strains when administered simultaneously with PCV13 compared with separate
administration. It is recommended to space these vaccines by 28 days in a person with
anatomic asplenia (47). Simultaneous or sequential vaccination of MCV4-CRM (Menveo),
PCV13, and Tdap (34, 62), all of which contain diphtheria toxoid, is not associated with
reduced immunogenicity or increase in local adverse events.
Nonsimultaneous Administration
There is no evidence that non-live vaccines interfere with the immune response to other non-
live vaccines or to live vaccines. Any non-live vaccine can be administered either
simultaneously or at any time before or after a different non-live vaccine or live vaccine
(Table 3-4). The 2 exceptions, as mentioned above, are a 4-week interval between PCV13 and
MenACWY-D in a person with anatomic asplenia and the separation of doses between PCV13
and PPSV23 (6-12 months recommended for non-high risk, 8 week minimum) if PCV13 is
given first, 8 weeks in children 6-18 years, and 1 year minimum in adults 19 years and older if
PPSV23 is given first (27).
Limited data are available regarding interference between live vaccines used in the United
States. The immune response to one live-virus vaccine might be impaired if administered
within 28 days (i.e., 4 weeks) of another live-virus vaccine (63,64). In a study conducted in 2
U.S. health maintenance organizations, the risk for varicella vaccine failure (i.e., varicella
disease in a vaccinated person) among persons who received varicella vaccine within 28 days
of MMR vaccination was threefold higher than among persons who received varicella vaccine
>28 days after MMR vaccination (65). Another study determined that the response to yellow
fever vaccine is not affected by monovalent measles vaccine administered 1-27 days earlier
(23).

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
23
The effect of nonsimultaneous administration of rubella, mumps, varicella, and yellow fever
vaccines is unknown.
Two or more injectable or nasally administered live vaccines not administered on the same
day should be separated by at least 4 weeks (Table 3-4), to minimize the potential risk for
interference. If 2 such vaccines are separated by <4 weeks, the second vaccine administered
should not be counted and the dose should be repeated at least 4 weeks later. On the day a
live injectable or intranasal vaccine will be administered, providers should ensure that no live
injectable or intranasal vaccine was given in the previous 28 days.
The 4-day grace period discussed earlier, which may be used to shorten the minimum
interval between doses of the same vaccine, should not be applied to this 4-week interval
between 2 different live vaccines (56). Confusion about this prohibition may arise when 2 live
vaccines whose intervals are identical are administered simultaneously. For example, if MMR
and varicella vaccines are administered on the same day, the second dose of each vaccine
could come due 4 weeks later (depending on the patient’s age). If either vaccine had been
given alone at both time points, the 4-day grace period could be applied to the second dose.
But in this situation the live vaccine rule prevents the grace period from being applied to the
second dose of either vaccine, because Varicella-2, if administered earlier than 4 weeks, could
potentially be affected by MMR1, and likewise MMR2 could be affected by Varicella-1. Note
that this prohibition also applies if the combination MMRV is used rather than individual
MMR and varicella vaccines.
The oral vaccines Ty21a typhoid vaccine and rotavirus can be administered simultaneously
with or at any interval before or after other live vaccines (injectable or intranasal) if indicated
(66).
Spacing of Vaccines and Antibody-Containing Products
Live Vaccines
Ty21a typhoid, yellow fever, LAIV, and rotavirus vaccines may be administered at any time
before, concurrent with, or after administration of any antibody-containing preparation such
as immune globulin, hyperimmune globulin, or intravenous immune globulin (IGIV) (67).

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
24
Blood (e.g., whole blood, packed red blood cells, and plasma) and other antibody-containing
blood products (e.g., immune globulin, hyperimmune globulin, and IGIV) can inhibit the
immune response to measles and rubella vaccines for ≥3 months. The effect of blood and
immune globulin preparations on the response to mumps and varicella vaccines is unknown;
however, commercial immune globulin preparations contain antibodies to these viruses.
Blood products available in the United States are unlikely to contain a substantial amount of
antibody to yellow fever virus. The length of time that interference with injectable live-virus
vaccine (other than yellow fever) can persist after the antibody-containing product is a
function of the amount of antigen-specific antibody contained in the product (68,70).
Therefore, after an antibody-containing product is received, live vaccines (other than Ty21a
typhoid, yellow fever, LAIV, and rotavirus vaccines) should be delayed until the passive
antibody has degraded (Table 3-5). In circumstances where there is high-risk of vaccine-
preventable disease it is acceptable to administer a dose of vaccine prior to completion of this
interval. If a dose of injectable live-virus vaccine (other than yellow fever) is administered
after an antibody-containing product but at an interval shorter than recommended in this
report, the vaccine dose should be repeated. The repeat dose should be administered at the
interval indicated for the antibody-containing product, after the invalid dose of vaccine
(Table 3-6). Immunogenicity and safety of dengue vaccine after administration of
intravenous immunoglobulin (IGIV) and other immunoglobulin containing products has not
been studied. Clinicians considering dengue vaccine for persons who recently received blood
products (including IGIV) or other immunoglobulin containing products should delay pre-
vaccination testing and administration of vaccine doses by 12 months.
Although passively acquired antibodies can interfere with the response to rubella vaccine, the
low dose of anti-Rho(D) globulin or any other blood product administered to postpartum
women have not been demonstrated to reduce the response to the RA27/3 strain rubella
vaccine (71). Congenital rubella syndrome and congenital varicella are conditions with
considerable morbidity and represent a true risk in future pregnancies. Because of the
importance of rubella and varicella immunity among women of child-bearing age (4, 72), the
postpartum vaccination of women without evidence of immunity to rubella or varicella with
MMR, varicella, or MMRV vaccines should not be delayed because of receipt of anti-Rho(D)
globulin or any other blood product during the last trimester of pregnancy or at delivery.

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
25
Any reduction in immunity caused by anti-Rho(D) globulin or other blood products is
outweighed by the opportunity to generate immunity. These women should be vaccinated
immediately after giving birth and, if possible, tested ≥3 months later to ensure immunity to
rubella and, if appropriate, to measles (2). Measles and rubella serologies have a low false-
positive rate and are therefore acceptable for use in this limited postpartum context.
Interference might occur if administration of an antibody-containing product becomes
necessary after administration of MMR or varicella vaccines. Usually, vaccine virus
replication and stimulation of immunity occurs 1-2 weeks after vaccination. If the interval
between administration of any of these vaccines and subsequent administration of an
antibody-containing product is <14 days, vaccination should be repeated after the
recommended interval (Tables 3-5 and 3-6) unless serologic testing indicates a protective
antibody response (7).
A humanized mouse monoclonal antibody product (palivizumab) is available as prophylaxis
for serious lower respiratory tract disease from respiratory syncytial virus among infants and
young children. This product contains only antibody to respiratory syncytial virus and does
not interfere with the immune response to licensed live or non-live vaccines.
Non-live Vaccines
Antibody-containing products interact less with non-live vaccines compared with live
vaccines (73). Therefore, administering non-live vaccines either simultaneously with or at
any interval before or after receipt of an antibody-containing product should not
substantially impair development of a protective antibody response (Table 3-5). The vaccine
or toxoid and antibody preparation should be administered at different sites using the
standard recommended dose.
Interchangeability of Single-Component Vaccines from
Different Manufacturers
Certain vaccines that provide protection from the same diseases are available from different
manufacturers, and these vaccines usually are not identical in antigen content or in amount
or method of formulation.
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
26
Manufacturers use different production processes, and their products might contain different
concentrations of antigen per dose or a different stabilizer or preservative.
Available data indicate that infants who receive sequential doses of different Hib conjugate,
hepatitis B, and hepatitis A vaccines produce a satisfactory antibody response after a
complete primary series (74-77).
All brands of Hib conjugate, hepatitis B,
(d)
hepatitis A, rotavirus,
(e)
and quadrivalent
meningococcal conjugate vaccines are interchangeable within their respective series.
If different brands of a particular vaccine require a different number of doses for series
completion (e.g., Hib and rotavirus vaccines) and a provider mixes brands in the primary
series, the higher number of doses is recommended for series completion (e.g., doses of
either rotavirus or Hib vaccine). For Hib vaccines, any monovalent or combination conjugate
vaccine is acceptable for the booster dose of the series, if only one product was used for the
primary series (56).
Limited data are available about the safety, immunogenicity, and efficacy of using acellular
pertussis (i.e., DTaP) vaccines from different manufacturers for successive doses of the
pertussis series. Data from one study indicate that for the first 3 doses of the DTaP series, 1-2
doses of Tripedia (Sanofi Pasteur) followed by Infanrix (GlaxoSmithKline) for the remaining
dose (or doses) is comparable to 3 doses of Tripedia with regard to immunogenicity, as
measured by antibodies to diphtheria, tetanus, and pertussis toxoids, and filamentous
hemagglutinin (78). However, in the absence of a clear serologic correlate of protection for
pertussis, the relevance of these immunogenicity data for protection against pertussis is
unknown. When feasible, the same brand of DTaP vaccine should be used for all doses of the
vaccination series (56). If vaccination providers do not know or have available the type of
DTaP vaccine previously administered to a child, any DTaP vaccine may be used to continue
or complete the series (56). For a child who needs 2 doses of influenza vaccine (IIV or LAIV),
it is preferable to use the same type of vaccine for both doses. However, if the child is eligible
for either IIV or LAIV, and the type of vaccine used for the first dose is not available, either
vaccine can be used for the second dose (56). In a postlicensure study, meningococcal
conjugate vaccines from different manufacturers were evaluated for successive doses of
meningococcal conjugate vaccine. Persistence of antibodies were studied in recipients of
MCV4-CRM after previous receipt of either MCV4-CRM or MenACWY-D.
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
27
The percentage of persons with protective titers were the same for all serogroups.
No data exist on the use of MenACWY-D after MCV4-CRM. Health-care providers should use
every opportunity to provide a dose when indicated, regardless of the vaccine brand used for
the previous dose or doses. For vaccines in general, vaccination should not be deferred
because the brand used for previous doses is not available or is unknown (29, 79).
Lapsed Vaccination Schedule
Vaccination providers should administer vaccines as close to the recommended intervals as
possible. However, intervals between doses that are longer than recommended typically do
not reduce final antibody concentrations, although protection might not be attained until the
recommended number of doses has been administered. With some exceptions (e.g. oral
typhoid vaccine) an interruption in the vaccination schedule does not require restarting the
entire series of a vaccine or toxoid or addition of extra doses (7).
Unknown or Uncertain Vaccination Status
Vaccination providers frequently encounter persons who do not have adequate
documentation of vaccinations. With the exception of influenza vaccine and PPSV23,
providers should only accept written, dated records as evidence of vaccination; self-reported
doses of influenza vaccine and PPSV23 are acceptable (61,80). The rationale for acceptance
for influenza vaccine is that the time period of recall is one year or less, making it very likely
that correct recall will occur. The rationale for acceptance for PPSV23 is high frequency of
vaccination leads to an increased rate of local reactions due to the reactogenicity of this
vaccine. Although vaccinations should not be postponed if records cannot be found, an
attempt to locate missing records should be made by contacting previous health care
providers, reviewing state or local IISs, and searching for a personally held record. If records
cannot be located within a reasonable time, these persons should be considered susceptible
and started on the age-appropriate vaccination schedule. While serology should generally
NOT be performed to ascertain evidence of immunity, serologic testing for immunity is an
alternative to vaccination for certain antigens (e.g., measles, rubella, hepatitis A, and
tetanus).
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
28
However, commercial serologic testing might not always be sufficiently sensitive or
standardized for detection of vaccine-induced immunity (with the exception of hepatitis B
vaccination at 1-2 months after the final dose), and research laboratory testing might not be
readily available. An exception to the prohibition on serologic testing is dengue. Laboratory
testing to ascertain immunity to dengue (of which serology is one option) must be performed
prior to vaccination, and only those individuals who are positive should be vaccinated.
(a)
During measles outbreaks, if cases are occurring among infants aged <12 months, measles vaccination of infants as young as 6 months
can be used as an outbreak control measure. However, doses administered at ages <12 months should not be counted as part of the
series (81).
(b)
In certain situations, local or state requirements might mandate that doses of selected vaccines be administered on or after specific
ages. For example, a school entry requirement might not accept a dose of MMR or varicella vaccine administered before the child’s first
birthday. ACIP recommends that physicians and other health-care providers comply with local or state vaccination requirements when
scheduling and administering vaccines.
(c)
Provider assessment should include number of injections, vaccine availability, likelihood of improved coverage, likelihood of patient
return, and storage and cost considerations.
(d)
The exception is the 2-dose hepatitis B vaccination series for adolescents aged 11-15 years. Only Recombivax HB (Merck Vaccine
Division) should be used in the schedule. Engerix-B (GlaxoSmithKline) is not approved by FDA for this schedule.
(e)
Based on expert opinion.

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
29
TABLE 3-1 Types of Vaccines
Vaccine Category
Examples
Live
Oral adenovirus vaccine
*
Live attenuated
ACAM2000 smallpox vaccine
Bacille Calmette Guerin (BCG) vaccine
Dengue vaccine
Ebola vector vaccine
Live attenuated influenza vaccine (LAIV)
Live oral typhoid vaccine (Ty21a)
Measles-mumps-rubella – containing (MMR,
MMRV)
Oral cholera vaccine
Rotavirus vaccines (RV1, RV5)
Varicella (Var) vaccine
Yellow Fever vaccine
Non-live
Anthrax vaccine
COVID-19 vaccines (Pfizer, Moderna,
Novavax)
Haemophilus influenzae type b (Hib)
vaccines
Hepatitis A (HepA) vaccines
Hepatitis B (HepB) vaccines
Human papillomavirus (HPV) vaccines
Inactivated poliovirus vaccine (IPV)
Inactivated typhoid vaccine (Typhim Vi)
Influenza vaccines (IIV4, RIV4)
Japanese Encephalitis Vaccine (JEV)
Meningococcal conjugate (MenACWY)
vaccine
Pneumococcal conjugate vaccines (PCV13,
PCV20, PCV15)
Pneumococcal polysaccharide vaccine
(PPSV23)
Rabies vaccine
Recombinant zoster vaccine (RZV)
Respiratory syncytial virus vaccine (RSV)
Serogroup B meningococcal (MenB) vaccines
(MenB-FHbp, MenB-4C)
Tetanus-toxoid, diphtheria-toxoid, or
pertussis-containing vaccines (DTaP, Tdap,
DT, Td, DTaP-HepB-IPV, DTaP-IPV/Hib,
DTaP-IPV, DTaP-IPV-Hib-HepB)
†
Non-replicating
§
COVID-19 vaccine (Janssen)
Jynneos smallpox/monkeypox vaccine
*
Oral adenovirus vaccine is used primarily in the military for prevention of adenovirus
infection. It should not be confused with Janssen COVID-19 vaccine which is used for the
prevention of SARS-CoV-2 infection
†
The tetanus-toxoid components of these vaccines are toxoids, not vaccines.
§
These vaccines do not replicate and therefore behave like non-live vaccines.

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
30
TABLE 3-2. Recommended and minimum ages and intervals between
vaccine
doses
(a),(b),(c),(d)
Known as the “grace period”, vaccine doses administered ≤4 days before the minimum
interval or age are considered valid; however, local or state mandates might supersede
this 4-day guideline
“3 calendar months” (or fewer) can be converted into weeks per the formula “1 month = 4
weeks”
Vaccine and
dose
number
Recommended
age for this dose
Minimum
age for this
dose
Recommended
interval to next
dose
Minimum
interval to
next dose
DEN4CYD-1 9—16 years 9 years 6 months
5 months after
age at 1
st
dose
DEN4CYD-2 9—16 years 9 years + 5
months
6 months
5 months after
age at second
dose
DEN4CYD-3 9—16 years 9 years +10
months
— —
DTaP-1
(e)
2 months 6 weeks 8 weeks 4 weeks
DTaP-2 4 months 10 weeks 8 weeks 4 weeks
DTaP-3 6 months 14 weeks 6-12 months
(f)
6 months
(f)
DTaP-4 15-18 months 15 months
(f)
3 years 6 months
DTaP-5
(g)
4-6 years 4 years — —
HepA-1
(e)
12-23 months 12 months 6-18 months 6 months
HepA-2 ≥18 months 18 months — —
HepB-1
(h)
Birth Birth 4 weeks-4 months 4 weeks
HepB-2 1-2 months 4 weeks 8 weeks-17 months 8 weeks
HepB-3
(i)
6-18 months 24 weeks — —
Hib-1
(j)
2 months 6 weeks 8 weeks 4 weeks
Hib-2 4 months 10 weeks 8 weeks 4 weeks
Hib-3
(k)
6 months 14 weeks 6-9 months 8 weeks
Hib-4 12-15 months 12 months — —
HPV Two Dose Series
(l)

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
31
HPV-1 11-12 years 9 years 6 months 5 months
HPV-2
11-12 years (+6
months)
9 years + 5
months
(m)
— —
HPV Three Dose Series
HPV-1
(n)
11-12 years 9 years 1-2 months 4 weeks
HPV-2
11-12 years (+1-2
months)
9 years (+4
weeks)
4 months 12 weeks
(n)
HPV-3
(n)
11-12 years (+6
months)
9 years (+5
months)
— —
Influenza,
inactivated
(o)
≥6 months 6 months
(p)
4 weeks 4 weeks
IPV-1
(e)
2 months 6 weeks 8 weeks 4 weeks
IPV-2 4 months 10 weeks 8 weeks-14 months 4 weeks
IPV-3 6-18 months 14 weeks 3-5 years 6 months
IPV-4
(q)
4-6 years 4 years — —
LAIV
(o)
2-49 years
(r)
2 years 4 weeks 4 weeks
MenACWY-1
(s)
11-12 years 2 months
(t)
4-5 years 8 weeks
MenACWY-2 16 years
11 years (+8
weeks)
(u)
— —
MenB-1
Healthy adolescents:
16-23 years
16 years Bexsero: 4 weeks
Trumenba: 6
months
(c)
Bexsero: 4
weeks
Trumenba: 6
months
(c)
Persons at increased
risk: ≥10 years
10 years Bexsero: 4 weeks
Trumenba: 1-2
months
(c)
Bexsero: 4
weeks
Trumenba: 1
month
MenB-2 Healthy adolescents:
16-23 years (+1
month)
16 years (+1
month)
— —
Persons at increased
risk: ≥10 years (+1
month)
10 years (+1
month)
Bexsero: —
Trumenba: 4-5
months
(c)
Bexsero: —
Trumenba: 4
months
(c)

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
32
MenB-3
(v)
Persons at increased
risk: ≥ 10 years (+ 6
months
(c)
)
10 years (+ 6
months
(c))
— —
MMR-1
(w)
12-15 months 12 months 3-5 years 4 weeks
MMR-2
(w)
4-6 years 13 months — —
PCV13-1
(j, x)
2 months 6 weeks 8 weeks 4 weeks
PCV13-2 4 months 10 weeks 8 weeks 4 weeks
PCV13-3 6 months 14 weeks 6 months 8 weeks
PCV13-4 12-15 months 12 months — —
PCV15 See footnote x below
PCV20 See footnote x below
PPSV-1 — 2 years 5 years 5 years
PPSV-2
(y)
— 7 years — —
Rotavirus-1
(z)
2 months 6 weeks 8 weeks 4 weeks
Rotavirus-2 4 months 10 weeks 8 weeks 4 weeks
Rotavirus-3
(z)
6 months 14 weeks — —
Td 11-12 years 7 years 10 years 5 years
Tdap
(aa)
≥11 years 7 years — —
Varicella-1
(w)
12-15 months 12 months 3-5 years 12 weeks
(bb)
Varicella-2
(w)
4-6 years 15 months
(cc)
— —
RZV - 1 ≥50 years 50 years
(dd)
2-6 months 4 weeks
RZV - 2 ≥50 years (+ 2-6
months)
50 years — —

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
33
Abbreviations: DEN4CYD = dengue vaccine; DTaP = diphtheria and tetanus toxoids and acellular pertussis; HepA =
hepatitis A; HepB = hepatitis B; Hib = Haemophilus influenzae type b; HPV = human papillomavirus; IPV =
inactivated poliovirus; LAIV = live, attenuated influenza vaccine; MenACWY = quadrivalent meningococcal conjugate
vaccine; MenB = serogroup B meningococcal vaccine; MenB-4C = Bexsero; MenB-FHbp = Trumenba; MMR =
measles, mumps, and rubella; MMRV
= measles, mumps, rubella, and varicella; MPSV4 = quadrivalent meningococcal polysaccharide vaccine; PCV13 =
pneumococcal conjugate vaccine; PPSV23= pneumococcal polysaccharide vaccine; PRP-OMP = polyribosylribitol
phosphate-meningococcal outer membrane protein conjugate; RZV = recombinant zoster vaccine; Td = tetanus and
diphtheria toxoids; Tdap = tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis.
(a)
Combination vaccines are available. Use of licensed combination vaccines is generally preferred to separate
injections of their equivalent component vaccines. When administering combination vaccines, the minimum age for
administration is the oldest age for any of the individual components. The minimum interval between doses is equal to
the greatest interval of any of the individual components.
(b)
Information on travel vaccines, including typhoid, Japanese encephalitis, and yellow fever, is available at
https://www.cdc.gov/travel.
Information on other vaccines that are licensed in the United States but not distributed,
including anthrax and smallpox, is available at http://emergency.cdc.gov/bioterrorism/ .
(c)
“Months” refers to calendar months.
(d)
Within a number range, a hyphen (-) should be read as “through.”
(e)
Combination vaccines containing the hepatitis B component are available (see Table 3-3). These vaccines should not
be administered to infants aged <6 weeks because of the other vaccine components (i.e., Hib, DTaP, HepA, and IPV).
(f)
The minimum recommended age for DTaP-4 is 15 months, with a recommended 6 months from DTaP-3 (the
recommended interval between DTaP-3 and DTaP-4 is 6 months). However, DTaP4 need not be repeated if given on
or after 12 months of age and at least 4 months after DTaP-3. The 4-day grace period can be applied when validating
past doses and can be applied to the minimum age of 12 months and the minimum interval of 4 months between
DTaP-3 and DTaP-4. The 4-day grace period can be used when planning doses ahead of time, but should be applied to
the minimum age of 15 months and the minimum interval between DTaP-3 and DTaP-4 of 6 months.
(g)
If a fourth dose of DTaP is given on or after the fourth birthday, a fifth dose is not needed if the interval between the
third dose and fourth dose is at least 6 months.
(h)
Adjuvanted Hepatitis B vaccine (HepB-CgG) can be administered to adults 18 years old and older on a two dose
schedule, the first and second dose separated by 4 weeks.
(i)
HepB-3 should be administered at least 8 weeks after HepB-2 and at least 16 weeks after HepB-1 and should not be
administered before age 24 weeks.
(j)
For Hib and PCV13, children receiving the first dose of vaccine at age ≥7 months require fewer doses to complete the
series.
(k)
If PRP-OMP (Pedvax-Hib, Merck Vaccine Division) was administered at ages 2 and 4 months, a dose at age 6
months is not necessary. The final dose has a minimum age of 12 months.
(l)
A two-dose schedule of HPV vaccine is recommended for most persons beginning the series between 9 through 14
years of age. See HPV vaccine-specific recommendations for details.
www.cdc.gov/mmwr/volumes/65/wr/pdfs/mm6549a5.pdf
(m)
If a patient is eligible for a 2-dose HPV series, and the second dose is given less than four weeks after the first dose,
it is an invalid dose. Administer another dose 6-12 months after the first dose. If the second dose is given less than five
months after the first dose, but more than four weeks after the first dose, the next dose should be administered at least
12 weeks after the second dose, and at least 6-12 months after the first dose. The 4-day grace period may be used. If
the third dose was administered before December 16, 2016, and was administered 12 weeks after the 2nd dose, and 16
weeks after the first dose, it is a valid dose. The 4-day grace period may be used. If the third dose was administered on
or after December 16, 2016, and was administered 12 weeks after the 2nd dose and 5 months after the first dose, it is a
valid dose. The 4-day grace period may be used.
(n)
The minimum age for HPV-3 is based on the baseline minimum age for the first dose (i.e., 9 years) and the
minimum interval of 5 months between the first and third dose. If the third dose was administered before December
16, 2016, and was administered 12 weeks after the 2nd dose, and 16 weeks after the first dose, it is a valid dose. The 4-
day grace period may be used. If the third dose was administered on or after December 16, 2016, and was
administered.
(o)
One dose of influenza vaccine per season is recommended for most persons. To determine which children younger
than 9 years should receive 2 doses in a single season, please see influenza vaccine-specific recommendations (82). If a
dose of LAIV is administered to a child 6 months to 2 years of age in error, the dose does not need to be repeated.

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
34
(p)
The minimum age for inactivated influenza vaccine varies by vaccine manufacturer. See package insert for vaccine-
specific minimum ages.
(q)
A fourth dose is not needed if the third dose was administered at ≥4 years and at least 6 months after the previous
dose.
(r) If a dose of LAIV is administered to someone 6 months to 2 years of age, the dose does NOT need to be repeated. If
someone is still indicated for dose 2 of the prime-boost, and younger than 2 years of age, the second dose should be
IIV.
(s)
Revaccination with meningococcal vaccine is recommended for previously vaccinated persons who remain at high
risk for meningococcal disease (47).
(t)
MenACWY-D (Menactra) can be given as young as 9 months for high-risk persons. MenACWY-CRM (Menveo) can
be given as young as 2 months for high-risk persons. Hib-MenCY can be given as young as 6 weeks for high-risk
persons. Hib-MenCY is given as a 4-dose series at 2 months, 4 months, 6 months and 12-18 months. MenACWY-TT
(MenQuadfi) can be given as young as 2 years for high-risk persons.
(u)
For routine non-high risk adolescent vaccination, the minimum age for the booster dose is 16 years.
(v)
This dose is not necessary if Bexsero is correctly administered, or if Trumenba is correctly administered to healthy
adolescents.
(w)
Combination MMRV vaccine can be used for children aged 12 months-12 years. See text for details.
(x)
Infants and children can optionally receive PCV15 instead of PCV13. High-risk adults 19 years through 64 years
who are known to have not received any pneumococcal vaccine or whose PCV13 history is unknown should receive
either PCV20 or PCV15. Adults 65 years old and older who are known to have not received any pneumococcal
vaccine or whose PCV13 history is unknown should receive either PCV20 or PCV15. If PCV15 is administered and if
PPSV23 naïve, a dose of PPSV23 should be administered a year later. On the basis of shared clinical decision-
making, adults 65 years old and older who have received a complete series of pneumococcal vaccine can receive a
dose of PCV20 at least five years after the most recent dose. For adults who have started the recommended
pneumococcal vaccine series with PCV13 but have not received all recommended doses, look at
ACIP Vaccine
Recommendations and Schedules for details.
(y)
A second dose of PPSV23 5 years after the first dose is recommended for persons aged ≤65 years at highest risk for
serious pneumococcal infection and those who are likely to have a rapid decline in pneumococcal antibody
concentration (61).
(z)
The first dose of rotavirus must be administered at age 6 weeks through 14 weeks and 6 days. The vaccine series
should not be started for infants aged ≥15 weeks, 0 days. Rotavirus should not be administered to children older than 8
months, 0 days of age regardless of the number of doses received between 6 weeks and 8 months, 0 days of age. If 2
doses of Rotarix (GlaxoSmithKline) are administered as age appropriate, a third dose is not necessary.
(aa)
Only 1 dose of Tdap is recommended. Subsequent doses should be given as Td or Tdap. For management of a
tetanus-prone wound in persons who have received a primary series of tetanus-toxoid–containing vaccine, the
minimum interval after a previous dose of any tetanus-containing vaccine is 5 years.
(bb)
A special grace period of 2 months, based on expert opinion, can be applied to the minimum interval of 3 months,
when evaluating records retrospectively, which results in an acceptable minimum interval of 4 weeks. An additional 4
days should not be added on to this grace period.
(cc)
A special grace period of 2 months, based on expert opinion, can be applied to the minimum age of 15 months when
evaluating records retrospectively, which results in an acceptable minimum age of 13 months. An additional 4 days
should not be added on to this grace period.
(dd)
If a 1
st
dose of recombinant zoster vaccine is administered to someone 18 – 49 years of age, the dose does not need
to be repeated. A 4 day grace period can be added to the absolute minimum age of 18 years when evaluating records
retrospectively.

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
35
TABLE 3-3. FDA-licensed combination vaccines
(a)
Vaccine
(b)
Trade name
(year licensed)
Age range
Routinely recommended
ages
HepA-HepB
Twinrix (2001)
≥18 years
Three doses on a schedule of
0, 1, and 6 months
DTaP-HepB-IPV
Pediarix (2002)
6 weeks-6 years
Three-dose series at 2, 4,
and 6 months of age
MMRV
ProQuad (2005)
12 months-12
years
Two doses, the first at 12-15
months, the second at 4-6
years
DTaP-IPV
Kinrix (2008)
4-6 years
Fifth dose of DTaP and
fourth dose of IPV
DTaP-IPV/Hib
Pentacel (2008)
6 weeks-4 years
Four-dose schedule at 2, 4,
6, and 15-18 months of age
Hib-MenCY
MenHibrix (2012)
6 weeks-18
months
Four-dose schedule at 2, 4,
6, and 12-15 months of age
(c)
DTaP-IPV
Quadracel (2015)
4-6 years
Fifth dose of DTaP and
fourth or fifth dose of IPV
DTaP-IPV-Hib-
HepB
Vaxelis (2018)
6 weeks – 4
years
Three-dose series at 2, 4,
and 6 months of age
Abbreviations: DT = diphtheria and tetanus toxoids; DTaP = diphtheria and tetanus toxoids and acellular pertussis;
FDA = Food and Drug Administration; HepA = hepatitis A; HepB = hepatitis B; Hib = Haemophilus influenzae type b;
IPV = inactivated poliovirus; MMR = measles, mumps, and rubella; MMRV = measles, mumps, rubella, and varicella;
Td = tetanus and diphtheria toxoids; Tdap = tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis.
Source: (82).
(a)
Although MMR, DTaP, DT, Td, and Tdap are combination vaccines, they are not included on this list because they
are not available in the United States as single-antigen products.
(b)
In descriptions of combination vaccines, dash (-) indicates products in which the active components are supplied in
their final (combined) form by the manufacturer; slash ( / ) indicates products in which active components must be
mixed by the user.
(c)
Hib-MenCY can be used for routine dosing of Hib vaccine but is recommended only for meningococcal vaccination
in persons at high-risk of meningococcal disease.

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
36
TABLE 3-4. Guidelines for spacing of live and non-live antigens
Antigen combination Recommended minimum interval between doses
Two or more non-live
(a),(b),(c)
May be administered simultaneously or at any interval
between doses
Non-live and live
(d)
May be administered simultaneously or at any interval
between doses
Two or more live injectable
(d)
28 days minimum interval, if not administered
simultaneously
Source: (83).
(a)
Certain experts suggest a 28-day interval between tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis
(Tdap) vaccine and tetravalent meningococcal conjugate vaccine if they are not administered simultaneously.
(b)
In persons with functional or anatomic asplenia, MCV-D and PCV13 should not be administered simultaneously and
should be spaced by 4 weeks. Likewise for persons with immunosuppressive high-risk conditions indicated for PCV13
and PPSV23, PCV13 should be administered first, and PPSV23 should be administered no earlier than 8 weeks later.
For persons 65 years old or older indicated for PCV13 and PPSV23, PCV13 should be administered first and PPSV23
should be administered 6-12 months later. For persons with high-risk conditions indicated for PCV15 and PPSV23, the
interval between PCV15 and PPSV23 should be one year, for those with immunosuppressive high-risk conditions the
interval may be shortened to 8 weeks.
(c)
For providers who wish to use Menactra in patients indicated for DTaP, the two vaccines should be administered
simultaneously. If they cannot be administered simultaneously, administer Menactra first, and then administer
DTaP 6 months later.
(d)
The live oral vaccines Ty21a typhoid vaccine and rotavirus vaccine may be administered simultaneously with or
at any interval before or after non-live or live injectable vaccines.

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
37
TABLE 3-5. Guidelines for administering antibody-containing products
(a)
and vaccines
Type of
administration
Products administered Recommended minimum
interval between doses
Simultaneous
(during the same
clinic day)
Antibody-containing products and
non-live antigen
Can be administered
simultaneously at different
anatomic sites or at any time
interval between doses
Antibody-containing products and
live antigen
Should not be administered
simultaneously.
(b)
If
simultaneous administration of
measles-containing vaccine or
varicella vaccine is unavoidable,
administer at different sites and
revaccinate or test for
seroconversion after the
recommended interval (see
Table 3-6)
Nonsimultaneous
Administered
first
Administered
second
Antibody-
containing
products
Non-live
antigen
No interval necessary
Non-live
antigen
Antibody-
containing
products
No interval necessary
Antibody-
containing
products
measles,
mumps, rubella
vaccine,
varicella
vaccine, and
combined
measles,
mumps, rubella,
varicella vaccine
antigens
Dose related
(b),(c)

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
38
MMR vaccine,
varicella vaccine,
and combined
measles, mumps,
rubella, varicella
vaccine antigens
Antibody-
containing
products
2 weeks
(b)
(a)
Blood products containing substantial amounts of immune globulin include intramuscular, subcutaneous, and
intravenous immune globulin, specific hyperimmune globulin (e.g., hepatitis B immune globulin, tetanus immune
globulin, varicella zoster immune globulin, and rabies immune globulin), whole blood, packed red blood cells, plasma,
and platelet products.
(b)
Yellow fever vaccine; rotavirus vaccine; oral Ty21a typhoid vaccine; and live, attenuated influenza vaccine are
exceptions to these recommendations. These live, attenuated vaccines can be administered at any time before or after
or simultaneously with an antibody-containing product.
(c)
The duration of interference of antibody-containing products with the immune response to the measles component
of measles-containing vaccine, and possibly varicella vaccine, is dose related (see Table 3-6).

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
39
TABLE 3-6. Recommended intervals between administration of antibody-
containing products and measles- or varicella-containing vaccine, by
product and indication for vaccination
Product/Indication Dose (mg IgG/kg)
and route
(a)
Recommended interval
before measles- or live
varicella-containing
vaccine
(b)
administration
(months)
(c)
Blood transfusion
RBCs, washed 10 mL/kg, negligible
IgG/kg IV
None
RBCs, adenine-saline added 10 mL/kg (10 mg
IgG/kg) IV
3
Packed RBCs (hematocrit 65%)
(d)
10 mL/kg (60 mg
IgG/kg) IV
6
Whole blood (hematocrit 35%-
50%)
(d)
10 mL/kg (80-100
mg IgG/kg) IV
6
Plasma/platelet products 10 mL/kg (160 mg
IgG/kg) IV
7
Botulinum Immune Globulin
Intravenous (Human)
1.0 mL/kg (50 mg
IgG/kg) IV
6
Cytomegalovirus IGIV 150 mg/kg maximum 6
Hepatitis A IG
Contact prophylaxis 0.1 mL/kg (16.5
mg IgG/kg) IM
6
(e)
International travel, <1 month stay 0.1 mL/kg (16.5
mg IgG/kg) IM
6
(e)
International travel, ≥1 month stay 0.2 mL/kg (33 mg
IgG/kg) IM
6
(e)
Hepatitis B IG 0.06 mL/kg (10 mg
IgG/kg) IM
3
IGIV
Replacement therapy for immune
deficiencies
(f)
300-400 mg/kg IV
(e)
8
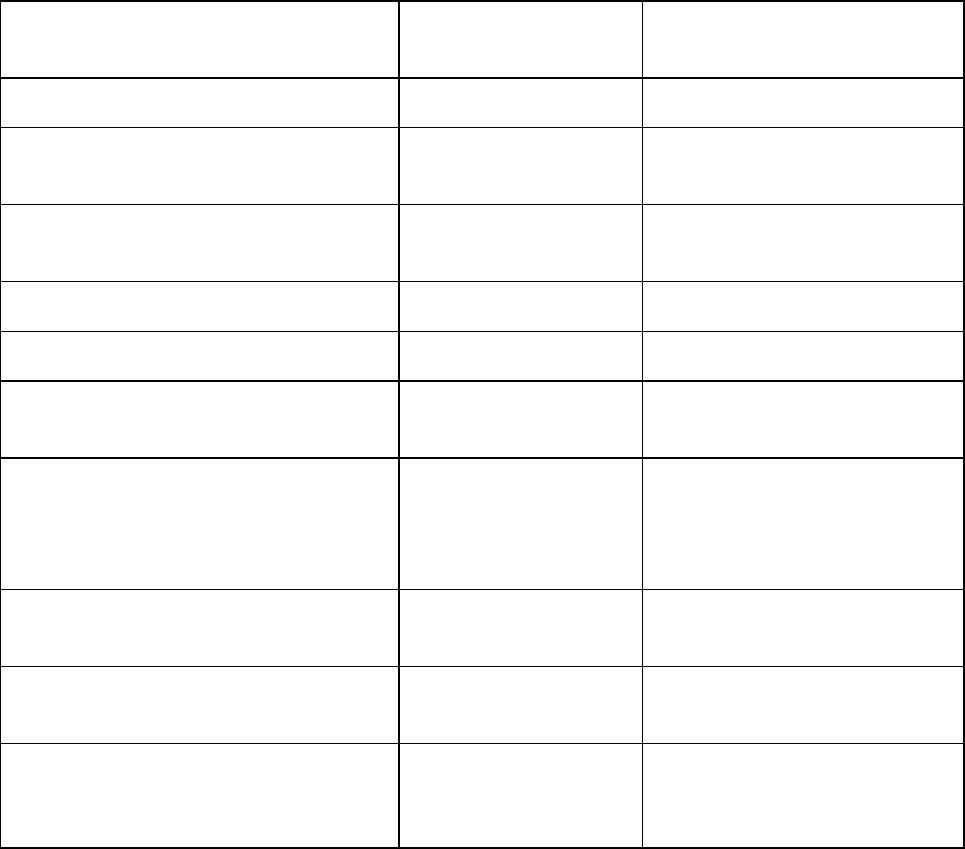
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
40
Immune thrombocytopenic
purpura treatment
400 mg/kg IV
8
Postexposure varicella prophylaxis 400 mg/kg IV
8
Postexposure measles prophylaxis
for immunocompromised contacts
400 mg/kg IV
8
Immune thrombocytopenic
purpura treatment
1000 mg/kg IV 10
Kawasaki disease 2 g/kg IV 11
Measles prophylaxis IG
Standard (i.e.,
nonimmunocompromised) contact
0.50 mL/kg (80 mg
IgG/kg) IM
6
Monoclonal antibody to
respiratory syncytial virus F
protein (e.g., Synagis
[MedImmune])
(g)
15 mg/kg IM None
Rabies IG
20 IU/kg (22 mg
IgG/kg) IM
4
Tetanus IG
250 units (10 mg
IgG/kg) IM
3
Varicella IG
125 units/10 kg (60-
200 mg IgG/kg) IM,
maximum 625 units
5

General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
41
Abbreviations: HIV = human immunodeficiency virus; IG = immune globulin; IgG = immune globulin G; IGIV =
intravenous immune globulin; mg IgG/kg = milligrams of immune globulin G per kilogram of body weight; IM =
intramuscular; IV = intravenous; RBCs = red blood cells.
(a)
This table is not intended for determining the correct indications and dosages for using antibody-
containing products.
Unvaccinated persons might not be protected fully against measles during the entire recommended interval, and
additional doses of IG or measles vaccine might be indicated after measles exposure. Concentrations of measles
antibody in an IG preparation can vary by manufacturer’s lot. Rates of antibody clearance after receipt of an IG
preparation also might vary. Recommended intervals are extrapolated from an estimated half-life of 30 days for
passively acquired antibody and an observed interference with the immune response to measles vaccine for 5 months
after a dose of 80 mg IgG/kg.
Sources: Mason W, Takahashi M, Schneider T. Persisting passively acquired measles
antibody following gamma globulin therapy for Kawasaki disease and response to live virus vaccination [Abstract 311].
Presented at the 32 meeting of the Interscience Conference on Antimicrobial Agents and Chemotherapy, Los Angeles,
California, October, 1992, AND Siber GR, Werner BG, Halsey NA, et al. Interference of immune globulin with measles
and rubella immunization. J Pediatr. 1993;122(2):204-211. DOI: 10.1016/S0022-3476(06)80114-9, AND Mason WH,
Schneider TL, Takahashi M. Duration of passively acquired measles antibody and response to live virus vaccination
allowing gamma globulin therapy for Kawasaki syndrome. Prog Pediatr Cardiol. 1992;1(1):82. DOI: 10.1016/S1058-
9813(06)80067-6. The extrapolation is performed by counting months from 80 mg down to (1-3 mg) (e.g. 80 >>> 40
>> >20 >> >10 >>> 5>>>2.5….equal to FIVE intervals) and adding a grace month, so 80 mg values take a “6 month”
interval)
(b)
Does not include recombinant zoster vaccine, since this vaccine is non-live.
(c)
The interval between blood/passive antibody products and testing for history of dengue infection (which is mandatory
prior to administration of dengue vaccine) should be 12 months
(d)
Assumes a serum IgG concentration of 16 mg/mL.
(e)
The reason the interval is 6 months (and not 4 months) is that the quantity of 16.5 IgG/kg does not reflect the upper
ceiling of the quantity of measles IgG in the product.
(f)
Measles vaccination is recommended for children with mild or moderate immunosuppression from HIV infection,
and varicella vaccination may be considered for children with mild or moderate immunosuppression from HIV
infection, but both are contraindicated for persons with severe immunosuppression from HIV or any other
immunosuppressive disorder.
(g)
Contains antibody only to respiratory syncytial virus.
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
42
REFERENCES
1.
CDC. Diphtheria, tetanus, and pertussis: recommendations for vaccine use and
other preventive measures. Recommendations of the Immunization Practices
Advisory Committee (ACIP). MMWR Recomm Rep. 1991;40(RR-10):1-28.
2.
CDC. Haemophilus b conjugate vaccines for prevention of Haemophilus
influenzae type b disease among infants and children two months of age and
older. Recommendations of the Immunization Practices Advisory Committee
(ACIP). MMWR Recomm Rep. 1991;40(RR-1):1-7.
3.
Plotkin SA. Immunologic correlates of protection induced by vaccination.
Pediatr Infect Dis J. 2001;20(1):63-75. DOI: 10.1097/00006454-200101000-
00013
4.
Watson JC, Hadler SC, Dykewicz CA, Reef S, Phillips L. Measles, mumps, and
rubella—vaccine use and strategies for elimination of measles, rubella, and
congenital rubella syndrome and control of mumps: recommendations of the
Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep.
1998;47(RR-8):1-57.
5.
Watson JC, Pearson JA, Markowitz LE, et al. An evaluation of measles
revaccination among school-entry-aged children. Pediatrics. 1996;97(5):613-
618.
6.
Marin M, Guris D, Chaves SS, Schmid S, Seward JF. Prevention of varicella:
recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR Recomm Rep. 2007;56(RR-4):1-40.
7.
Atkinson WL, Pickering LK, Schwartz B, Weniger BG, Iskander JK, Watson JC.
General recommendations on immunization. Recommendations of the Advisory
Committee on Immunization Practices (ACIP) and the American Academy of
Family Physicians (AAFP). MMWR Recomm Rep. 2002;51(RR-2):1-35.
8.
Manning SE, Rupprecht CE, Fishbein D, et al. Human rabies prevention—United
States, 2008: recommendations of the Advisory Committee on Immunization
Practices. MMWR Recomm Rep. 2008;57(RR-3):1-28.
9.
https://www.cdc.gov/vaccines/programs/iis/cdsi.html
10.
Levine L, Edsall G. Tetanus toxoid: what determines reaction proneness? J
Infect Dis. 1981;144(4):376. DOI: 10.1093/infdis/144.4.376
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
43
11.
Edsall G, Elliott MW, Peebles TC, Eldred MC. Excessive use of tetanus toxoid
boosters. JAMA. 1967;202(1):111-113. DOI: 10.1001/jama.1967.03130140075009
12.
King GE, Hadler SC. Simultaneous administration of childhood vaccines: an
important public health policy that is safe and efficacious. Pediatr Infect Dis J.
1994;13(5):394-407.
13.
National Vaccine Advisory Committee. Standards for child and adolescent
immunization practices. Pediatrics. 2003;112(4):958-963.
14.
Hutchins SS, Escolan J, Markowitz LE, et al. Measles outbreak among
unvaccinated preschool-aged children: opportunities missed by health care
providers to administer measles vaccine. Pediatrics. 1989;83(3):369-374.
15.
Deforest A, Long SS, Lischner HW, et al. Simultaneous administration of
measles-mumps-rubella vaccine with booster doses of diphtheria-tetanus-
pertussis and poliovirus vaccines. Pediatrics. 1988;81(2):237-246.
16.
Dashefsky B, Wald E, Guerra N, Byers C. Safety, tolerability, and
immunogenicity of concurrent administration of Haemophilus influenzae type b
conjugate vaccine (meningococcal protein conjugate) with either measles-
mumps-rubella vaccine or diphtheria-tetanus-pertussis and oral poliovirus
vaccines in 14- to 23-month-old infants. Pediatrics. 1990;85(4 Pt 2):682-689.
17.
Giammanco G, Li Volti S, Mauro L, et al. Immune response to simultaneous
administration of a recombinant DNA hepatitis B vaccine and multiple
compulsory vaccines in infancy. Vaccine. 1991;9(10):747-750. DOI:
10.1016/0264-410X(91)90291-D
18.
Grohskopf LA, Sokolow LZ, Olsen SJ, Bresee JS, Broder KR, Karron RA.
Prevention and control of influenza with vaccines: recommendations of the
Advisory Committee on Immunization Practices, United States, 2015-16
influenza season. MMWR Morb Mortal Wkly Rep. 2015;64(30):818-825.
19.
CDC. Typhoid immunization: recommendations of the Immunization Practices
Advisory Committee (ACIP). MMWR Recomm Rep. 1990;39(RR-10):1-5.
20.
DeStefano F, Goodman RA, Noble GR, McClary GD, Smith S, Broome CV.
Simultaneous administration of influenza and pneumococcal vaccines. JAMA.
1982;247(18):2551-2554. DOI: 10.1001/jama.1982.03320430055032
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
44
21.
Kretsinger K, Broder KR, Cortese MM, et al. Preventing tetanus, diphtheria, and
pertussis among adults: use of tetanus toxoid, reduced diphtheria toxoid and
acellular pertussis vaccine recommendations of the Advisory Committee on
Immunization Practices (ACIP) and recommendation of ACIP, supported by the
Healthcare Infection Control Practices Advisory Committee (HICPAC), for use of
Tdap among health-care personnel. MMWR Recomm Rep. 2006;55(RR-17):1-
37.
22.
Yvonnet B, Coursaget P, Deubel V, Diop-Mar I, Digoutte JP, Chiron JP.
Simultaneous administration of hepatitis B and yellow fever vaccines. J Med
Virol. 1986;19(4):307-311. DOI: 10.1002/jmv.1890190403
23.
Stefano I, Sato HK, Pannuti CS, et al. Recent immunization against measles does
not interfere with the sero-response to yellow fever vaccine. Vaccine. 1999;17(9-
10):1042-1046. DOI: 10.1016/S0264-410X(98)00320-X
24.
Staples JE, Gershman M, Fischer M. Yellow fever vaccine: recommendations of
the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm
Rep. 2010;59(RR-7):1-27.
25.
Leroy Z, Broder K, Menschik D, Shimabukuro T, Martin D. Febrile seizures after
2010-2011 influenza vaccine in young children, United States: a vaccine safety
signal from the vaccine adverse event reporting system. Vaccine.
2012;30(11):2020-2023. DOI: 10.1016/j.vaccine.2011.12.042
26.
Tse A, Tseng HF, Greene SK, Vellozzi C, Lee GM. Signal identification and
evaluation for risk of febrile seizures in children following trivalent inactivated
influenza vaccine in the Vaccine Safety Datalink Project, 2010-2011. Vaccine.
2012;30(11):2024-2031. DOI: 10.1016/j.vaccine.2012.01.027
27.
CDC. Recommendation of the Advisory Committee on Immunization Practices
(ACIP) for use of quadrivalent meningococcal conjugate vaccine (MenACWY-D)
among children aged 9 through 23 months at increased risk for invasive
meningococcal disease. MMWR Morb Mortal Wkly Rep. 2011;60(40):1391-
1392.
28.
CDC. Use of 13-valent pneumococcal conjugate vaccine and 23-valent
pneumococcal polysaccharide vaccine for adults with immunocompromising
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
45
conditions: recommendations of the Advisory Committee on Immunization
Practices (ACIP). MMWR Morb Mortal Wkly Rep. 2012;61(40):816-819.
29.
CDC. Use of 13-valent pneumococcal conjugate vaccine and 23-valent
pneumococcal polysaccharide vaccine among children aged 6-18 years with
immunocompromising conditions: recommendations of the Advisory Committee
on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep.
2013;62(25):521-524.
30.
Tomczyk S, Bennett NM, Stoecker C, et al. Use of 13-valent pneumococcal
conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine among
adults aged >/=65 years: recommendations of the Advisory Committee on
Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep.
2014;63(37):822-825.
31.
CDC. Recommended immunization schedules for persons aged 0 through 18
years—United States, 2011. MMWR Morb Mortal Wkly Rep. 2011;60(5):1-4.
32.
Shinefield HR, Black S, Ray P, et al. Safety and immunogenicity of heptavalent
pneumococcal CRM197 conjugate vaccine in infants and toddlers. Pediatr Infect
Dis J. 1999;18(9):757-763. DOI: 10.1097/00006454-199909000-00004
33.
CDC. Pertussis vaccination: use of acellular pertussis vaccines among infants
and young children. Recommendations of the Advisory Committee on
Immunization Practices (ACIP). MMWR Recomm Rep. 1997;46(RR-7):1-25.
34.
Broder KR, Cortese MM, Iskander JK, et al. Preventing tetanus, diphtheria, and
pertussis among adolescents: use of tetanus toxoid, reduced diphtheria toxoid
and acellular pertussis vaccines recommendations of the Advisory Committee on
Immunization Practices (ACIP). MMWR Recomm Rep. 2006;55(RR-3):1-34.
35.
Nuorti JP, Whitney CG. Prevention of pneumococcal disease among infants and
children - use of 13-valent pneumococcal conjugate vaccine and 23-valent
pneumococcal polysaccharide vaccine - recommendations of the Advisory
Committee on Immunization Practices (ACIP). MMWR Recomm Rep.
2010;59(RR-11):1-18.
36.
Mast EE, Margolis HS, Fiore AE, et al. A comprehensive immunization strategy
to eliminate transmission of hepatitis B virus infection in the United States:
recommendations of the Advisory Committee on Immunization Practices (ACIP)
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
46
part 1: immunization of infants, children, and adolescents. MMWR Recomm
Rep. 2005;54(RR-16):1-31.
37.
Committee on Infectious Diseases. Recommended childhood and adolescent
immunization schedules—United States, 2010. Pediatrics. 2010;125(1):195-
196. DOI: 10.1542/peds.2009-3194
38.
CDC. Recommended adult immunization schedule—United States, 2011.
MMWR Morb Mortal Wkly Rep. 2011;60(4):1-4.
39.
Woodin KA, Rodewald LE, Humiston SG, Carges MS, Schaffer SJ, Szilagyi
PG. Physician and parent opinions. Are children becoming pincushions from
immunizations? Arch Pediatr Adolesc Med. 1995;149(8):845-849. DOI:
10.1001/archpedi.1995.02170210019003
40.
Kuppermann M, Nease RF, Jr., Ackerson LM, Black SB, Shinefield HR, Lieu TA.
Parents' preferences for outcomes associated with childhood vaccinations.
Pediatr Infect Dis J. 2000;19(2):129-133. DOI: 10.1097/00006454-200002000-
00010
41.
Meyerhoff A, Jacobs RJ, Greenberg DP, Yagoda B, Castles CG. Clinician
satisfaction with vaccination visits and the role of multiple injections,
results from the COVISE Study (Combination Vaccines Impact on
Satisfaction and Epidemiology). Clin Pediatr (Phila). 2004;43(1):87-93.
42.
Marshall GS, Happe LE, Lunacsek OE, et al. Use of combination vaccines is
associated with improved coverage rates. Pediatr Infect Dis J.
2007;26(6):496- 500. DOI: 10.1097/INF.0b013e31805d7f17
43.
Kalies H, Grote V, Verstraeten T, Hessel L, Schmitt HJ, von Kries R. The use
of combination vaccines has improved timeliness of vaccination in children.
Pediatr Infect Dis J. 2006;25(6):507-512. DOI:
10.1097/01.inf.0000222413.47344.23
44.
Happe LE, Lunacsek OE, Kruzikas DT, Marshall GS. Impact of a pentavalent
combination vaccine on immunization timeliness in a state Medicaid
population. Pediatr Infect Dis J. 2009;28(2):98-101. DOI:
10.1097/INF.0b013e318187d047
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
47
45.
Marin M, Broder KR, Temte JL, Snider DE, Seward JF. Use of combination
measles, mumps, rubella, and varicella vaccine: recommendations of the
Advisory Committee on Immunization Practices (ACIP). MMWR Recomm
Rep. 2010;59(RR-3):1-12.
46.
Thompson LA, Irigoyen M, Matiz LA, LaRussa PS, Chen S, Chimkin F. The
impact of DTaP-IPV-HB vaccine on use of health services for young infants.
Pediatr Infect Dis J. 2006;25(9):826-831. DOI:
10.1097/01.inf.0000232635.81312.06
47.
Meningococcal Vaccination: Recommendations of the Advisory Committee on
Immunization Practices, United States, 2020. Recommendations and Reports /
September 25, 2020 / 69(9);1-41.
48.
Weniger BG, Chen RT, Jacobson SH, et al. Addressing the challenges to
immunization practice with an economic algorithm for vaccine selection.
Vaccine. 1998;16(19):1885-1897. DOI: 10.1016/S0264-410X(98)00170-4
49.
Liang J, Wallace G, Mootrey G. Licensure of a diphtheria and tetanus toxoids
and acellular pertussis adsorbed and inactivated poliovirus vaccine and guidance
for use as a booster dose. MMWR Morb Mortal Wkly Report 2015;64: 948-9.
50.
CDC. FDA approval of a Haemophilus b Conjugate Vaccine combined by
reconstitution with an acellular pertussis vaccine. MMWR Morb Mortal Wkly
Rep. 1996;45(45):993-995.
51.
CDC. FDA approval for a combined hepatitis A and B vaccine. MMWR Morb
Mortal Wkly Rep. 2001;50(37):806-807.
52.
CDC. FDA licensure of diphtheria and tetanus toxoids and acellular pertussis
adsorbed, hepatitis B (recombinant), and poliovirus vaccine combined,
(PEDIARIX) for use in infants. MMWR Morb Mortal Wkly Rep.
2003;52(10):203-204.
53.
CDC. Licensure of a combined live attenuated measles, mumps, rubella, and
varicella vaccine. MMWR Morb Mortal Wkly Rep. 2005;54(47):1212-1214.
54.
CDC. Licensure of a diphtheria and tetanus toxoids and acellular pertussis
adsorbed and inactivated poliovirus vaccine and guidance for use as a booster
dose. MMWR Morb Mortal Wkly Rep. 2008;57(39):1078-1079.
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
48
55.
CDC. Licensure of a diphtheria and tetanus toxoids and acellular pertussis
adsorbed, inactivated poliovirus, and haemophilus B conjugate vaccine and
guidance for use in infants and children. MMWR Morb Mortal Wkly Rep.
2008;57(39):1079-1080.
56.
Kroger AT, Sumaya CV, Pickering LK, Atkinson WL. General recommendations
on immunization: recommendations of the Advisory Committee on
Immunization Practices (ACIP). MMWR Recomm Rep. 2011:1-60.
57.
Plotkin SA. Vaccines: correlates of vaccine-induced immunity. Clin Infect Dis.
2008;47(3):401-409. DOI: 10.1086/589862
58.
Lane KS, Chu SY, Santoli JM. The United States pediatric vaccine stockpile
program. Clinical infectious diseases : an official publication of the Infectious
Diseases Society of America. 2006;42 Suppl 3:S125-129. DOI: 10.1086/499591
59.
Midthun K, Horne AD, Goldenthal KL. Clinical safety evaluation of combination
vaccines. Dev Biol Stand. 1998;95:245-249.
60.
Pichichero ME, Blatter MM, Reisinger KS, et al. Impact of a birth dose of
hepatitis B vaccine on the reactogenicity and immunogenicity of diphtheria-
tetanus-acellular pertussis-hepatitis B-inactivated poliovirus-Haemophilus
influenzae type b combination vaccination. Pediatr Infect Dis J.
2002;21(9):854- 859. DOI: 10.1097/01.inf.0000027669.37444.24
61.
CDC. Prevention of pneumococcal disease: recommendations of the
Advisory Committee on Immunization Practices (ACIP). MMWR Recomm
Rep. 1997;46(RR-8):1-24.
62.
Bilukha OO, Rosenstein N. Prevention and control of meningococcal disease.
Recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR Recomm Rep. 2005;54(RR-7):1-21.
63.
Petralli JK, Merigan TC, Wilbur JR. Action of endogenous interferon against
vaccinia infection in children. Lancet. 1965;2(7409):401-405. DOI:
10.1016/S0140-6736(65)90755-5
64.
Petralli JK, Merigan TC, Wilbur JR. Circulating interferon after
measles vaccination. N Engl J Med. 1965;273:198-201. DOI:
10.1056/nejm196507222730405
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
49
65.
Verstraeten T, Jumaan AO, Mullooly JP, et al. A retrospective cohort study of
the association of varicella vaccine failure with asthma, steroid use, age at
vaccination, and measles-mumps-rubella vaccination. Pediatrics.
2003;112(2):e98-103. DOI: 10.1542/peds.112.2.e98
66.
CDC. General recommendations on immunization. Recommendations of the
Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep.
1994;43(RR-1):1-38.
67.
Harpaz R, Ortega-Sanchez IR, Seward JF. Prevention of herpes zoster:
recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR Recomm Rep. 2008;57(RR-5):1-30; quiz CE32-34.
68.
Siber GR, Werner BG, Halsey NA, et al. Interference of immune globulin with
measles and rubella immunization. J Pediatr. 1993;122(2):204-211. DOI:
10.1016/S0022-3476(06)80114-9
69.
Mason WH, Schneider TL, Takahashi M. Duration of passively acquired measles
antibody and response to live virus vaccination allowing gamma globulin therapy
for Kawasaki syndrome. Prog Pediatr Cardiol. 1992;1(1):82. DOI:
10.1016/S1058-9813(06)80067-6
70.
Kaplan JE, Nelson DB, Schonberger LB, et al. The effect of immune globulin on
the response to trivalent oral poliovirus and yellow fever vaccinations. Bull
World Health Organ. 1984;62(4):585-590.
71.
Black NA, Parsons A, Kurtz JB, McWhinney N, Lacey A, Mayon-White RT. Post-
partum rubella immunisation: a controlled trial of two vaccines. Lancet.
1983;2(8357):990-992. DOI: 10.1016/S0140-6736(83)90979-0
72.
CDC. Control and prevention of rubella: evaluation and management of
suspected outbreaks, rubella in pregnant women, and surveillance for congenital
rubella syndrome. MMWR Recomm Rep. 2001;50(RR-12):1-23.
73.
Siber GR, Snydman DR. Use of immune globulin in the prevention and
treatment of infections. In: Remington J, Swartz M, eds. Current clinical topics
in infectious diseases. Vol 12. Malden, MA: Blackwell Science; 1992.
74.
Greenberg DP, Lieberman JM, Marcy SM, et al. Enhanced antibody responses in
infants given different sequences of heterogeneous Haemophilus influenzae type
b conjugate vaccines. J Pediatr. 1995;126(2):206-211. DOI: 10.1016/S0022-
3476(95)70546-5
General Best Practice Guidelines for Immunization: Timing and Spacing of Immunobiologics
50
75.
Anderson EL, Decker MD, Englund JA, et al. Interchangeability of conjugated
Haemophilus influenzae type b vaccines in infants. JAMA. 1995;273(11):849-
853. DOI: 10.1001/jama.1995.03520350031024
76.
Piazza M, Abrescia N, Picciotto L, et al. [Demonstration of the interchangeability
of 2 types of recombinant anti-hepatitis-B vaccine]. Boll Soc Ital Biol Sper.
1993;69(4):273-280.
77.
Bryan JP, Henry CH, Hoffman AG, et al. Randomized, cross-over, controlled
comparison of two inactivated hepatitis A vaccines. Vaccine. 2000;19(7-8):743-
750. DOI: 10.1016/S0264-410X(00)00301-7
78.
Greenberg DP, Pickering LK, Senders SD, et al. Interchangeability of 2
diphtheria-tetanus-acellular pertussis vaccines in infancy. Pediatrics.
2002;109(4):666-672. DOI: 10.1542/peds.109.4.666
79.
CDC. Use of diphtheria toxoid-tetanus toxoid-acellular pertussis vaccine as a
five-dose series. Supplemental recommendations of the Advisory Committee on
Immunization Practices (ACIP). MMWR Recomm Rep. 2000;49(RR-13):1-8.
80.
Fiore AE, Uyeki TM, Broder K, et al. Prevention and control of influenza with
vaccines: recommendations of the Advisory Committee on Immunization
Practices (ACIP), 2010. MMWR Recomm Rep. 2010;59(RR-8):1-62.
81.
McLean HQ, Fiebelkorn AP, Temte JL, Wallace GS. Prevention of measles,
rubella, congenital rubella syndrome, and mumps, 2013: summary
recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR Recomm Rep. 2013;62(RR-4):1-34.
82.
Grohskopf LA, Alyanak E, Broder KR. Prevention and Control of Seasonal
Influenza with Vaccines: Recommendations of the Advisory Committee on
Immunization Practices — United States, 2020―21 Influenza Season. MMWR
Recomm Rep 2020;69(No. RR-8):1-26.
83.
American Academy of Pediatrics. Active Immunization. In: Pickering L, Baker C,
Kimberlin D, Long S, eds. Red Book: 2012 Report of the Committee on
Infectious Diseases. 28th ed. Elk Grove Village, IL: American Academy of
Pediatric; 2012.

General Best Practice Guidelines for Immunization: Contraindications and Precautions
51
4. Contraindications and Precautions
Updates
Major changes to the best practice guidance in this section include 1) enhancement of the
definition of a “precaution” to include any condition that might confuse diagnostic
accuracy and 2) recommendation to vaccinate during a hospitalization if a patient is not
acutely moderately or severely ill.
General Principles
National standards for pediatric vaccination practices have been established and include
descriptions of valid contraindications and precautions to vaccination (2). Persons who
administer vaccines should screen patients for contraindications and precautions to the
vaccine before each dose of vaccine is administered (Table 4-1). Screening is facilitated by
consistent use of screening questionnaires, which are available from certain state
vaccination programs and other sources (e.g., the Immunization Action Coalition,
http://www.immunize.org).
Contraindications
Contraindications (conditions in a recipient that increases the risk for a serious adverse
reaction) to vaccination are conditions under which vaccines should not be administered.
Because the majority of contraindications are temporary, vaccinations often can be
administered later when the condition leading to a contraindication no longer exists. A
vaccine should not be administered when a contraindication is present; for example,
MMR vaccine should not be administered to severely immunocompromised persons (1).
However, certain conditions are commonly misperceived as contraindications (i.e., are
not valid reasons to defer vaccination).
Severely immunocompromised persons generally should not receive live vaccines (3).
Because of the theoretical risk to the fetus, women known to be pregnant generally should
not receive live, attenuated virus vaccines (4). Persons who experienced encephalopathy
within 7 days after administration of a previous dose of pertussis- containing vaccine not
attributable to another identifiable cause should not receive additional doses of a vaccine
that contains pertussis (4,5).

General Best Practice Guidelines for Immunization: Contraindications and Precautions
52
Severe Combined Immunodeficiency (SCID) disease and a history of intussusception are
both contraindications to the receipt of rotavirus vaccines (6).
Precautions
A precaution is a condition in a recipient that might increase the risk for a serious adverse
reaction, might cause diagnostic confusion, or might compromise the ability of the
vaccine to produce immunity (e.g., administering measles vaccine to a person with
passive immunity to measles from a blood transfusion administered up to 7 months
prior) (7). A person might experience a more severe reaction to the vaccine than would
have otherwise been expected; however, the risk for this happening is less than the risk
expected with a contraindication. In general, vaccinations should be deferred when a
precaution is present. However, a vaccination might be indicated in the presence of a
precaution if the benefit of protection from the vaccine outweighs the risk for an adverse
reaction.
The presence of a moderate or severe acute illness with or without a fever is a
precaution to administration of all vaccines (Table 4-1). The decision to administer or
delay vaccination because of a current or recent acute illness depends on the severity of
symptoms and etiology of the condition. The safety and efficacy of vaccinating persons
who have mild illnesses have been documented (8-11). Vaccination should be deferred
for persons with a moderate or severe acute illness. This precaution avoids causing
diagnostic confusion between manifestations of the underlying illness and possible
adverse effects of vaccination or superimposing adverse effects of the vaccine on the
underlying illness. After they are screened for contraindications, persons with
moderate or severe acute illness should be vaccinated as soon as the acute illness has
improved. Studies indicate that failure to vaccinate children with minor illnesses can
impede vaccination efforts (12–14). Among persons whose compliance with medical
care cannot be ensured, use of every opportunity to administer appropriate vaccines is
critical.
Hospitalization should be used as an opportunity to provide recommended vaccinations.
Health-care facilities are held to standards of offering influenza vaccine for hospitalized
patients, so providers are incentivized to vaccinate these patients at some point during
hospitalization (15). Likewise, patients admitted for elective procedures will not be
acutely ill during all times during their hospitalization. Most studies that have explored

General Best Practice Guidelines for Immunization: Contraindications and Precautions
53
the effect of surgery or anesthesia on the immune system were observational, included
only infants and children, and were small and indirect, in that they did not look at the
immune effect on the response to vaccination specifically (16-35). They do not provide
convincing evidence that recent anesthesia or surgery significantly affect response to
vaccines. Current, recent, or upcoming anesthesia/surgery/hospitalization is not a
contraindication to vaccination, but certain factors might lead a provider to consider
current, recent, or upcoming anesthesia/surgery/hospitalization as a precaution (16-35).
Efforts should be made to ensure vaccine administration during the hospitalization or at
discharge. For patients who are deemed moderately or severely ill throughout the
hospitalization, vaccination should occur at the earliest opportunity (i.e., during
immediate post-hospitalization follow-up care, including home or office visits) when
patients’ clinical symptoms have improved.
A personal or family history of seizures is a precaution for MMRV vaccination; this is
because a recent study found an increased risk for febrile seizures in children 12-23
months who receive MMRV compared with MMR and varicella vaccine (36).
Neither Contraindications Nor Precautions
Clinicians or other health-care providers might misperceive certain conditions or
circumstances as valid contraindications or precautions to vaccination when they
actually do not preclude vaccination (2) (Table 4-2). These misperceptions result in
missed opportunities to administer recommended vaccines (37).
Routine physical examinations and procedures (e.g., measuring temperatures) are not
prerequisites for vaccinating persons who appear to be healthy. The provider should ask
the parent or guardian if the child is ill. If the child has a moderate or severe illness, the
vaccination should be postponed.
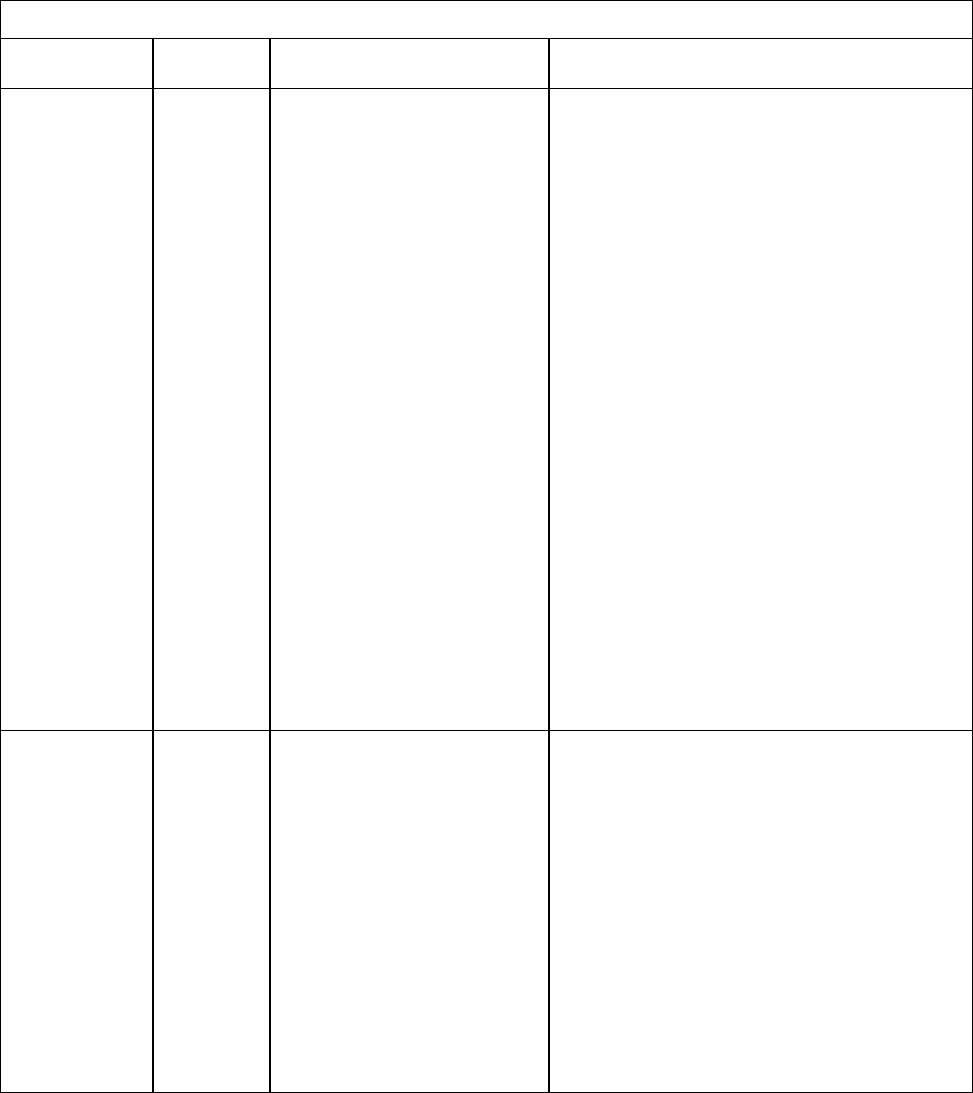
General Best Practice Guidelines for Immunization: Contraindications and Precautions
54
TABLE 4-1. Contraindications and precautions
(a)
to commonly used vaccines
Vaccine Citation Contraindications Precautions
Dengue–
ONLY use in
persons who
have
laboratory
confirmation
of previous
dengue
infection
AND reside
in endemic
dengue areas
(b)
(38) Lack of laboratory
evidence of previous
dengue infection
Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component
Severe
immunodeficiency (e.g.,
hematologic and solid
tumors, receipt of
chemotherapy,
congenital
immunodeficiency,
long-term
immunosuppressive
therapy
(c)
or patients
with HIV infection who
are severely
immunocompromised)
Pregnancy
HIV infection without evidence of
severe immunosuppression
Moderate or severe acute illness
with or without fever
DT, Td (4) Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component
GBS <6 weeks after previous dose of
tetanus-toxoid–containing vaccine
History of Arthus-type
hypersensitivity reactions after a
previous dose of diphtheria-toxoid—
containing or tetanus-toxoid–
containing vaccine; defer vaccination
until at least 10 years have elapsed
since the last tetanus-toxoid-
containing vaccine
Moderate or severe acute illness with
or without fever

General Best Practice Guidelines for Immunization: Contraindications and Precautions
55
DTaP (39) Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component
Encephalopathy (e.g.,
coma, decreased level of
consciousness,
prolonged seizures), not
attributable to another
identifiable cause,
within 7 days of
administration of
previous dose of DTP or
DTaP
Progressive neurologic disorder,
including infantile spasms,
uncontrolled epilepsy, progressive
encephalopathy; defer DTaP until
neurologic status clarified and
stabilized
GBS <6 weeks after previous dose of
tetanus-toxoid–containing vaccine
History of Arthus-type
hypersensitivity reactions after a
previous dose of diphtheria-toxoid–
containing or tetanus-toxoid–
containing vaccine; defer vaccination
until at least 10 years have elapsed
since the last tetanus-toxoid–
containing vaccine
Moderate or severe acute illness with
or without fever
Hepatitis A (40) Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component
Moderate or severe acute illness with
or without fever
Hepatitis B (41) Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component
Hypersensitivity to yeast
Moderate or severe acute illness with
or without fever
Hib (42) Severe allergic reaction
(e.g., anaphylaxis) after
Moderate or severe acute illness with
or without fever

General Best Practice Guidelines for Immunization: Contraindications and Precautions
56
a previous dose or to a
vaccine component
Age <6 weeks
HPV
(d)
(
43) Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component,
including yeast
Moderate or severe acute illness with
or without fever
IIV
(e)
(
44)
Severe allergic reaction
(e.g., anaphylaxis) after
previous dose of
influenza vaccine or to
vaccine component
GBS <6 weeks after a previous dose of
influenza vaccine
Moderate or severe acute illness with
or without fever
IPV
(
45)
Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component
Pregnancy
Moderate or severe acute illness with
or without fever

General Best Practice Guidelines for Immunization: Contraindications and Precautions
57
LAIV
(f)
(
44)
Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component
Concomitant use of
aspirin or salicylate-
containing medication
in children and
adolescents
LAIV4 should not be
administered to persons
who have taken
oseltamivir or zanamivir
within the previous 48
hours, peramivir within
the previous 5 days, or
baloxavir within the
previous 17 days.
(h)
Pregnancy
Children aged 2 through
4 years who have
received a diagnosis of
asthma or whose
parents or caregivers
report that a health care
provider has told them
during the preceding 12
months that their child
had wheezing or asthma
or whose medical record
indicates a wheezing
episode has occurred
during the preceding 12
months.
Persons with active
cerebrospinal
fluid/oropharyngeal
communications/leaks.
GBS <6 weeks after a previous dose of
influenza vaccine
Asthma in persons aged 5 years old or
older
Medical conditions which might
predispose to higher risk of
complications attributable to
influenza
(g)
Moderate or severe acute illness with
or without fever
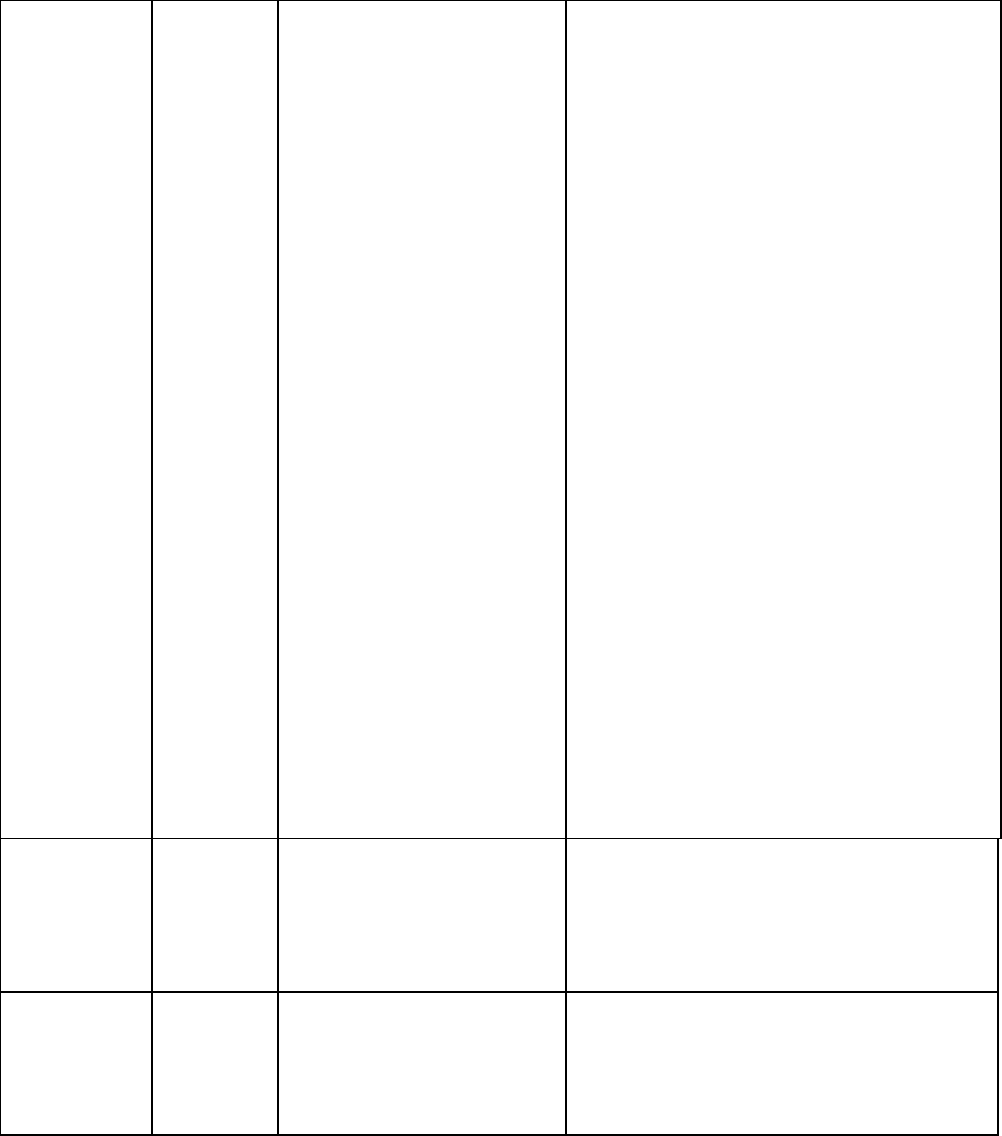
General Best Practice Guidelines for Immunization: Contraindications and Precautions
58
Close contacts and
caregivers of severely
immunosuppressed
persons who require a
protected environment.
Persons with cochlear
implants (due to the
potential for CSF leak,
which might exist for
some period of time
after implantation.
Providers might
consider consultation
with a specialist
concerning risk of
persistent CSF leak if an
age-appropriate
inactivated or
recombinant vaccine
cannot be used).
Altered
Immunocompetence
Anatomic or functional
asplenia (e.g. sickle cell
disease
MenACWY
(
46) Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component,
including yeast
Moderate or severe acute illness with
or without fever
Preterm birth (MenACWY-CRM)
(i)
MenB
(46,4
8) Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component
Moderate or severe acute illness with
or without fever
Pregnancy
Latex sensitivity (MenB-4c)

General Best Practice Guidelines for Immunization: Contraindications and Precautions
59
MMR
(j),(k)
(
1) Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component
Pregnancy
Known severe
immunodeficiency (e.g.,
from hematologic and
solid tumors, receipt of
chemotherapy,
congenital
immunodeficiency,
long-term
immunosuppressive
therapy
(c)
or patients
with HIV infection who
are severely
immunocompromised)
Family history of altered
immunocompetence
(m)
Recent (≤11 months) receipt of
antibody-containing blood product
(specific interval depends on product)
History of thrombocytopenia or
thrombocytopenic purpura
Need for tuberculin skin testing or
interferon-gamma release assay
(IGRA) testing
(l)
Moderate or severe acute illness with
or without fever
MPSV4
(
49) Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component
Moderate or severe acute illness with
or without fever
PCV13,
PCV15,
PCV20
(
50) Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose of PCV
or any diphtheria-
toxoid– containing
vaccine or to a
component of a vaccine
(PCV or any diphtheria-
toxoid– containing
vaccine)
Moderate or severe acute illness with
or without fever
PPSV23
(
51)
Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component
Moderate or severe acute illness with
or without fever
RIV
(
44)
Severe allergic reaction
(e.g., anaphylaxis) to
any component of the
vaccine
GBS <6 weeks after a previous dose of
influenza vaccine
Moderate or severe acute illness with
or without fever

General Best Practice Guidelines for Immunization: Contraindications and Precautions
60
Rotavirus
(
6) Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component
SCID
History of
intussusception
Altered immunocompetence other
than SCID
Chronic gastrointestinal disease
Spina bifida or bladder exstrophy
(n)
Moderate or severe acute illness with
or without fever
Tdap
(
52)
Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component
Encephalopathy (e.g.,
coma, decreased level of
consciousness,
prolonged seizures), not
attributable to another
identifiable cause, within
7 days of administration
of previous dose of DTP,
DTaP, or Tdap
GBS <6 weeks after a previous dose of
tetanus-toxoid–containing vaccine
Progressive or unstable neurological
disorder, uncontrolled seizures, or
progressive encephalopathy until a
treatment regimen has been
established and the condition has
stabilized
History of Arthus-type
hypersensitivity reactions after a
previous dose of diphtheria-toxoid—
containing or tetanus-toxoid–
containing vaccine; defer vaccination
until at least 10 years have elapsed
since the last tetanus-toxoid–
containing vaccine
Moderate or severe acute illness with
or without fever
Varicella
(j),(k
)
(
53) Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component
Known severe
immunodeficiency (e.g.,
from hematologic and
solid tumors, receipt of
chemotherapy,
congenital
immunodeficiency,
long-term
immunosuppressive
therapy
(c)
or patients
with HIV infection who
are severely
immunocompromised)
(j)
Pregnancy
Family history of altered
immunocompetence
(m)
Recent (≤11 months) receipt of
antibody-containing blood product
(specific interval depends on product)
Moderate or severe acute illness with
or without fever
Receipt of specific antiviral drugs
(acyclovir, famciclovir, or
valacyclovir) 24 hours before
vaccination (avoid use of these
antiviral drugs for 14 days after
vaccination)
Use of aspirin or aspirin-containing
products
(o)

General Best Practice Guidelines for Immunization: Contraindications and Precautions
61
Zoster
(
54)
Severe allergic reaction
(e.g., anaphylaxis) after
a previous dose or to a
vaccine component
Moderate or severe acute illness with
or without fever
Abbreviations: DT = diphtheria and tetanus toxoids; DTaP = diphtheria and tetanus toxoids and acellular pertussis;
DTP = diphtheria toxoid, tetanus toxoid, and pertussis; GBS = Guillain-Barré syndrome; Hib = Haemophilus influenzae
type b; HIV = human immunodeficiency virus; HPV = human papillomavirus; IIV = inactivated influenza vaccine; IPV =
inactivated poliovirus; LAIV = live, attenuated influenza vaccine; MenACWY = quadrivalent meningococcal conjugate
vaccine; MMR = measles, mumps, and rubella; MPSV4 = quadrivalent meningococcal polysaccharide vaccine; PCV13 =
pneumococcal conjugate vaccine; PPSV23= pneumococcal polysaccharide vaccine; SCID = severe combined
immunodeficiency; RIV=recombinant influenza vaccine; Td = tetanus and diphtheria toxoids; Tdap = tetanus toxoid,
reduced diphtheria toxoid, and acellular pertussis.
(a)
Events or conditions listed as precautions should be reviewed carefully. Benefits of and risks for administering a
specific vaccine to a person under these circumstances should be considered. If the risk from the vaccine is believed to
outweigh the benefit, the vaccine should not be administered. If the benefit of vaccination is believed to outweigh the
risk, the vaccine should be administered. Whether and when to administer DTaP to children with proven or suspected
underlying neurologic disorders should be decided on a case-by-case basis.
(b)
Only persons with laboratory confirmation of immunity according to strict guidance at
https://www.cdc.gov/dengue/vaccine/hcp/testing.html s
hould receive dengue vaccination.
(c)
Substantially immunosuppressive steroid dose is considered to be ≥2 weeks of daily receipt of 20 mg or 2 mg/kg body
weight of prednisone or equivalent.
(d)
HPV vaccine is not recommended during pregnancy
(e)
When applying this contraindication to ccIIV, the history of severe allergic reaction (e.g., anaphylaxis) must be specific
to the event occurring following a dose of ccIIV. Likewise, when applying this contraindication to RIV, the history of
severe allergic reaction (e.g., anaphylaxis) must be specific to the event occurring following a dose of RIV. A history of
severe allergic reaction (e.g., anaphylaxis) to a non-ccIIV vaccine or to a component specific to components not
contained in ccIIV, is a precaution to ccIIV. A history of severe allergic reaction (e.g., anaphylaxis) to a non-RIV vaccine
or to a component specific to components not contained in RIV is a precaution to RIV.
(f)
In addition, ACIP recommends LAIV not be used for pregnant women, immunosuppressed persons, and children aged
2-4 years who have asthma or who have had a wheezing episode noted in the medical record within the past 12 months,
or for whom parents report that a health care provider stated that they had wheezing or asthma within the last 12 months.
LAIV should not be administered to persons who have taken influenza antiviral medications within the previous 48
hours. Persons who care for severely immunosuppressed persons who require a protective environment should not
receive LAIV, or should avoid contact with such persons for 7 days after receipt.
(g)
See reference: Grohskopf LA, Alyanak E, Ferdinands JM, et al. Prevention and Control of Seasonal Influenza with
Vaccines: Recommendations of the Advisory Committee on Immunization Practices, United States, 2021-2022 Influenza
Season. MMWR Recomm Rep 2021;70(No. RR-5):1-30.

General Best Practice Guidelines for Immunization: Contraindications and Precautions
62
(h)
These values are based on the clearance of the particular antiviral. LAIV4 should not be administered to persons who
have taken oseltamivir or zanamivir within the previous 48 hours, peramivir within the previous 5 days, or baloxavir
within the previous 17 days. This “contraindication” is due to concern with reduced effectiveness of the vaccine. To obtain
specific information, please refer to Grohskopf LA, Alyanak, E, Broder KR, et. al. Prevention and Control of Seasonal
Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices — United States,
2020–21 Influenza Season. MMWR Recomm Rep 2020;69 (No. RR-8:1-26. Also at
https://www.cdc.gov/mmwr/volumes/69/rr/pdfs/rr6908a1-H.pdf
(i)
This precaution applies to infants younger than 9 months old
(j)
HIV-infected children may receive varicella vaccine if CD4+ T-lymphocyte count is ≥15% and should receive MMR
vaccine if they are aged ≥12 months and do not have evidence of current severe immunosuppression (i.e., individuals
aged ≤5 years must have CD4+T lymphocyte [CD4] percentages ≥15% for ≥6 months; and individuals aged >5 years must
have CD4+percentages ≥15% and CD4+≥200 lymphocytes/mm
3
for ≥6 months) or other current evidence of measles,
rubella, and mumps immunity. In cases when only CD4+cell counts or only CD4+percentages are available for those
older than age 5 years, the assessment of severe immunosuppression can be based on the CD4+values (count or
percentage) that are available. In cases when CD4+percentages are not available for those aged ≤5 years, the assessment
of severe immunosuppression can be based on age-specific CD4+counts at the time CD4+counts were measured; i.e.,
absence of severe immunosuppression is defined as ≥6 months above age-specific CD4+count criteria: CD4+count >750
lymphocytes/mm
3
while aged ≤12 months and CD4+count ≥500 lymphocytes/mm
3
while aged 1 through 5 years.
Sources: (1,50).
(k)
MMR and varicella-containing vaccines can be administered on the same day. If not administered on the same
day, these vaccines should be separated by at least 28 days.
(l)
If active tuberculosis is suspected, MMR should be delayed. Measles vaccination might suppress tuberculin reactivity
temporarily. Measles-containing vaccine can be administered on the same day as tuberculin skin or IGRA testing. If
testing cannot be performed until after the day of MMR vaccination, the test should be postponed for ≥4 weeks after the
vaccination. If an urgent need exists to skin test or IGRA, do so with the understanding that reactivity might be reduced
by the vaccine.
(m)
family history of congenital or hereditary immunodeficiency in first-degree relatives (e.g., parents and siblings),
unless the immune competence of the potential vaccine recipient has been substantiated clinically or verified by a
laboratory
(n)
For RV1 only, based on latex in product/packaging. Note that anaphylactic allergy to latex is covered in the
contraindication, and would also be isolated to RV 1 in the case of latex. For more details, see (55).
(o)
No adverse events associated with the use of aspirin or aspirin-containing products after varicella vaccination have
been reported; however, the vaccine manufacturer recommends that vaccine recipients avoid using aspirin or aspirin-
containing products for 6 weeks after receiving varicella vaccines because of the association between aspirin use and
Reye syndrome after varicella. Vaccination with subsequent close monitoring should be considered for children who have
rheumatoid arthritis or other conditions requiring therapeutic aspirin. The risk for serious complications associated with
aspirin is likely to be greater in children in whom natural varicella develops than it is in children who receive the vaccine
containing attenuated VZV. No association has been documented between Reye syndrome and analgesics or antipyretics
that do not contain aspirin.”

General Best Practice Guidelines for Immunization: Contraindications and Precautions
63
TABLE 4-2. Conditions incorrectly perceived as contraindications or
precautions to vaccination (i.e., vaccines may be given under these
conditions)
Vaccine
Conditions commonly misperceived as
contraindications or precautions
General for all
vaccines, including
DTaP, pediatric
DT, adult Td,
adolescent-adult
Tdap, IPV, MMR,
Hib, hepatitis A,
hepatitis B,
varicella, rotavirus,
PCV13, IIV, LAIV,
PPSV23,
MenACWY, MPSV4,
HPV, and herpes
zoster
Mild acute illness with or without fever
Lack of previous physical examination in well-appearing
person
Current antimicrobial therapy
(a)
Convalescent phase of illness
Preterm birth (hepatitis B vaccine is an exception in certain
circumstances)
(b)
Recent exposure to an infectious disease
History of penicillin allergy, other nonvaccine allergies,
relatives with allergies, or receiving allergen extract
immunotherapy
History of GBS
(c)
DTaP
Fever within 48 hours after vaccination with a previous dose of
DTP or DTaP
Collapse or shock-like state (i.e., hypotonic hyporesponsive
episode) within 48 hours after receiving a previous dose of
DTP/DTaP
Seizure ≤3 days after receiving a previous dose of DTP/DTaP
Persistent, inconsolable crying lasting ≥3 hours within 48
hours after receiving a previous dose of DTP/DTaP
Family history of seizures
Family history of sudden infant death syndrome
Family history of an adverse event after DTP or DTaP
administration
Stable neurologic conditions (e.g., cerebral palsy, well-
controlled seizures, or developmental delay)
Hepatitis B
Pregnancy
Autoimmune disease (e.g., systemic lupus erythematosus or
rheumatoid arthritis)
HPV
Immunosuppression
Previous equivocal or abnormal Papanicolaou test
Known HPV infection
Breastfeeding
History of genital warts
IIV
Nonsevere (e.g., contact) allergy to latex, thimerosal, or egg
Concurrent administration of Coumadin (generic: warfarin) or
aminophylline
IPV
Previous receipt of ≥1 dose of oral polio vaccine

General Best Practice Guidelines for Immunization: Contraindications and Precautions
64
LAIV
Health-care providers that see patients with chronic diseases
or altered immunocompetence (an exception is providers for
severely immunocompromised patients requiring care in a
protected environment)
Breastfeeding
Contacts of persons with chronic disease or altered
immunocompetence (an exception is contacts of severely
immunocompromised patients requiring care in a protected
environment)
MMR
(d),(e)
Positive tuberculin skin test
Simultaneous tuberculin skin or interferon-gamma release
assay (IGRA) testing
(f)
Breastfeeding
Pregnancy of recipient’s mother or other close or household
contact
Recipient is female of child-bearing age
Immunodeficient family member or household contact
Asymptomatic or mildly symptomatic HIV infection
Allergy to eggs
PPSV23
History of invasive pneumococcal disease or pneumonia
Rotavirus
Prematurity
Immunosuppressed household contacts
Pregnant household contacts
Tdap
History of fever of ≥40.5°C (≥105°F) for <48 hours after
vaccination with a previous dose of DTP or DTaP
History of collapse or shock-like state (i.e., hypotonic
hyporesponsive episode) within 48 hours after receiving a
previous dose of DTP/DTaP
History of seizure <3 days after receiving a previous dose of
DTP/DTaP
History of persistent, inconsolable crying lasting >3 hours
within 48 hours after receiving a previous dose of DTP/DTaP
History of extensive limb swelling after DTP/DTaP/Td that is
not an Arthus-type reaction
History of stable neurologic disorder
History of brachial neuritis
Latex allergy that is not anaphylactic
Breastfeeding
Immunosuppression
Varicella
Pregnancy of recipient’s mother or other close or household
contact
Immunodeficient family member or household contact
(g)
Asymptomatic or mildly symptomatic HIV infection
Humoral immunodeficiency (e.g., agammaglobulinemia)

General Best Practice Guidelines for Immunization: Contraindications and Precautions
65
Zoster
Therapy with low-dose methotrexate (≤0.4 mg/kg/week),
azathioprine (≤3.0 mg/kg/day), or 6-mercaptopurine (≤1.5
mg/kg/day) for treatment of rheumatoid arthritis, psoriasis,
polymyositis, sarcoidosis, inflammatory bowel disease, or other
conditions
Health-care providers of patients with chronic diseases or
altered immunocompetence
Contacts of patients with chronic diseases or altered
immunocompetence
Unknown or uncertain history of varicella in a U.S.-born
person
Abbreviations: DT = diphtheria and tetanus toxoids; DTP = diphtheria toxoid, tetanus toxoid, and pertussis;
DTaP = diphtheria and tetanus toxoids and acellular pertussis; GBS = Guillain-Barré syndrome; HBsAg = hepatitis
B surface antigen; Hib = Haemophilus influenzae type b; HIV = human immunodeficiency virus; HPV = human
papillomavirus; IIV = inactivated influenza vaccine; IPV = inactivated poliovirus; LAIV = live, attenuated influenza
vaccine; MenACWY = quadrivalent meningococcal conjugate vaccine; MMR = measles, mumps, and rubella;
MPSV4 = quadrivalent meningococcal polysaccharide vaccine; PCV = pneumococcal conjugate vaccine; PPSV23=
pneumococcal polysaccharide vaccine; Td = tetanus and diphtheria toxoids; Tdap = tetanus toxoid, reduced
diphtheria toxoid, and acellular pertussis.
(a)
Antibacterial drugs might interfere with Ty21a oral typhoid vaccine, and certain antiviral drugs might interfere
with varicella-containing vaccines and LAIV4.
(b)
Hepatitis B vaccination should be deferred for infants weighing <2,000 g if the mother is documented to be
HBsAg negative. Vaccination should commence at chronological age 1 month or at hospital discharge. For infants
born to HBsAg-positive women, hepatitis B immune globulin and hepatitis B vaccine should be administered
within 12 hours after birth, regardless of weight.
(c)
An exception is Guillain-Barré syndrome within 6 weeks of a dose of influenza vaccine or tetanus-toxoid–
containing vaccine, which are precautions for influenza vaccines and tetanus-toxoid containing vaccines,
respectively.
(d)
MMR and varicella vaccines can be administered on the same day. If not administered on the same day, these
vaccines should be separated by at least 28 days.
(e)
HIV-infected children should receive immune globulin after exposure to measles. HIV-infected children can
receive varicella and measles vaccine if CD4+ T-lymphocyte count is >15%. (55).
(f)
Measles vaccination might suppress tuberculin reactivity temporarily. Measles-containing vaccine can be
administered on the same day as tuberculin skin or IGRA testing. If testing cannot be performed until after the day
of MMR vaccination, the test should be postponed for at least 4 weeks after the vaccination. If an urgent need
exists to skin test or IGRA, do so with the understanding that reactivity might be reduced by the vaccine.
(g)
If a vaccinee experiences a presumed vaccine-related rash 7-25 days after vaccination, the person should avoid
direct contact with immunocompromised persons for the duration of the rash.
General Best Practice Guidelines for Immunization: Contraindications and Precautions
66
REFERENCES
1. McLean HQ, Fiebelkorn AP, Temte JL, Wallace GS. Prevention of measles,
rubella, congenital rubella syndrome, and mumps, 2013: summary
recommendations of the Advisory Committee on Immunization Practices (ACIP).
MMWR Recomm Rep. 2013;62(RR-4):1-34.
2. National Vaccine Advisory Committee. Standards for child and adolescent
immunization practices. Pediatrics. 2003;112(4):958-963.
3. Rubin L, Levin M, Ljungman P, et al. 2013 IDSA clinical practice guideline for
vaccination of the immunocompromised host. Clin Infect Dis. 2014;58(3):e44-
100. DOI: 10.1093/cid/cit684
4. Kroger A, Atkinson W, Pickering L. General immunization practices. In: Plotkin
S, Orenstein W, Offit P, eds. Vaccines. 6
th
ed. China: Elsevier Saunders; 2013:88-
111.
5. CDC. Diphtheria, tetanus, and pertussis: recommendations for vaccine use and
other preventive measures. Recommendations of the Immunization Practices
Advisory Committee (ACIP). MMWR Recomm Rep. 1991;40(RR-10):1-28.
6. CDC. Addition of history of intussusception as a contraindication for rotavirus
vaccination. MMWR Morb Mortal Wkly Rep. 2011;60(41):1427.
7. Siber GR, Werner BG, Halsey NA, et al. Interference of immune globulin with
measles and rubella immunization. J Pediatr. 1993;122(2):204-211. DOI:
10.1016/S0022-3476(06)80114-9
8. Halsey NA, Boulos R, Mode F, et al. Response to measles vaccine in Haitian
infants 6 to 12 months old. Influence of maternal antibodies, malnutrition, and
concurrent illnesses. N Engl J Med. 1985;313(9):544-549. DOI:
10.1056/nejm198508293130904
9. Ndikuyeze A, Munoz A, Stewart J, et al. Immunogenicity and safety of measles
vaccine in ill African children. Int J Epidemiol. 1988;17(2):448-455. DOI:
10.1093/ije/17.2.448
10. Lindegren ML, Atkinson WL, Farizo KM, Stehr-Green PA. Measles vaccination in
pediatric emergency departments during a measles outbreak. JAMA.
1993;270(18):2185-2189. DOI: 10.1001/jama.1993.03510180055033
11. Atkinson W, Markowitz L, Baughman A, et al. Serologic response to measles
vaccination among ill children [Abstract 422]. 32nd Interscience Conference on
Antimicrobial Agents and Chemotherapy; 1992; Anaheim, CA.
General Best Practice Guidelines for Immunization: Contraindications and Precautions
67
12. Orenstein W, Rodewald L, Hinman A, Schuchat A. Immunization in the United
States. In: Plotkin S, Orenstein W, Offit P, eds. Vaccines. 5th ed. China:
Saunders/Elsevier; 2008:1479-1510.
13. Lewis T, Osborn LM, Lewis K, Brockert J, Jacobsen J, Cherry JD. Influence of
parental knowledge and opinions on 12-month diphtheria, tetanus, and
pertussis vaccination rates. Am J Dis Child. 1988;142(3):283-286. DOI:
10.1001/archpedi.1988.02150030053018
14. Farizo KM, Stehr-Green PA, Markowitz LE, Patriarca PA. Vaccination levels and
missed opportunities for measles vaccination: a record audit in a public pediatric
clinic. Pediatrics. 1992;89(4 Pt 1):589-592.
15. Centers for Medicare & Medicaid Services. Overview of specifications of measures
displayed on hospital compare as of December 14, 2006. 2006;
http://www.cms.hhs.gov/HospitalQualityInits/downloads/HospitalOverviewOfS
pecs200512.pdf. Accessed 9 March, 2017.
16. Donovan R, Soothill JF. Immunological studies in children
undergoing tonsillectomy. Clin Exp Immunol. 1973;14(3):347-357.
17. Puri P, Reen DJ, Browne O, Blake P, Guiney EJ. Lymphocyte response after
surgery in the neonate. Arch Dis Child. 1979;54(8):599-603. DOI:
10.1136/adc.54.8.599
18. Mollitt DL, Steele RW, Marmer DJ, Stevers Golladay E, Costas S. Surgically
induced immunologic alterations in the child. J Pediatr Surg.
1984;19(6):818- 822. DOI: 10.1016/S0022-3468(84)80376-0
19. Mollitt DL, Marmer DJ, Steele RW. Age-dependent variation of lymphocyte
function in the postoperative child. J Pediatr Surg. 1986;21(7):633-635.
DOI: 10.1016/S0022-3468(86)80420-1
20. Kurz R, Pfeiffer KP, Sauer H. Immunologic status in infants and children
following surgery. Infection. 1983;11(2):104-113. DOI: 10.1007/BF01641075
21. Merry C, Puri P, Reen DJ. Effect of major surgery on neutrophil chemotaxis and
actin polymerization in neonates and children. J Pediatr Surg. 1997;32(6):813-
817. DOI: 10.1016/S0022-3468(97)90626-6
22. Platt MP, Lovat PE, Watson JG, Aynsley-Green A. The effects of anesthesia
and surgery on lymphocyte populations and function in infants and children. J
Pediatr Surg. 1989;24(9):884-887. DOI: 10.1016/S0022-3468(89)80588-3
General Best Practice Guidelines for Immunization: Contraindications and Precautions
68
23. Mattila-Vuori A, Salo M, Iisalo E. Immune response in infants undergoing
application of cast: comparison of halothane and balanced anesthesia. Can J
Anaesth. 1999;46(11):1036-1042. DOI: 10.1007/bf03013198
24. Espanol T, Todd GB, Soothill JF. The effect of anaesthesia on the lymphocyte
response to phytohaemagglutinin. Clin Exp Immunol. 1974;18(1):73-79.
25. Hauser GJ, Chan MM, Casey WF, Midgley FM, Holbrook PR. Immune
dysfunction in children after corrective surgery for congenital heart disease. Crit
Care Med. 1991;19(7):874-881.
26. Puri P, Lee A, Reen DJ. Differential susceptibility of neonatal lymphocytes to the
immunosuppressive effects of anesthesia and surgery. Pediatr Surg Int.
1992;7(1):47-50. DOI: 10.1007/bf00181002
27. Hansen TG, Tonnesen E, Andersen JB, Toft P, Bendtzen K. The peri-operative
cytokine response in infants and young children following major surgery. Eur J
Anaesthesiol. 1998;15(1):56-60. DOI: 10.1046/j.1365-2346.1998.00230.x
28. Mattila-Vuori A, Salo M, Iisalo E, Pajulo O, Viljanto J. Local and systemic
immune response to surgery under balanced anaesthesia in children. Paediatr
Anaesth. 2000;10(4):381-388. DOI: 10.1046/j.1460-9592.2000.00505.x
29. Romeo C, Cruccetti A, Turiaco A, et al. Monocyte and neutrophil activity after
minor surgical stress. J Pediatr Surg. 2002;37(5):741-744. DOI:
10.1053/jpsu.2002.32268
30. Vuori A, Salo M, Viljanto J, Pajulo O, Pulkki K, Nevalainen T. Effects of post-
operative pain treatment using non-steroidal anti-inflammatory analgesics,
opioids or epidural blockade on systemic and local immune responses in
children. Acta Anaesthesiol Scand. 2004;48(6):738-749. DOI: 10.1111/j.1399-
6576.2004.00404.x
31. Siebert JN, Posfay-Barbe KM, Habre W, Siegrist CA. Influence of anesthesia on
immune responses and its effect on vaccination in children: review of evidence.
Paediatr Anaesth. 2007;17(5):410-420. DOI: 10.1111/j.1460-9592.2006.02120.x
32. Currie J. Vaccination: is it a real problem for anesthesia and surgery? Paediatr
Anaesth. 2006;16(5):501-503. DOI: 10.1111/j.1460-9592.2006.01898.x
33. Siebert J, Posfay-Barbe KM, Habre W, Siegrist C-A. Author's reply. Paediatr
Anaesth. 2007;17(12):1218-1220. DOI: 10.1111/j.1460-9592.2007.02369.x
General Best Practice Guidelines for Immunization: Contraindications and Precautions
69
34. Nafiu OO, Lewis I. Vaccination and anesthesia: more questions than answers.
Paediatr Anaesth. 2007;17(12):1215-1215. DOI: 10.1111/j.1460-
9592.2007.02318.x
35. Short JA, Van Der Walt JH, Zoanetti DC. Author's reply. Paediatr Anaesth.
2007;17(12):1215-1216. DOI: 10.1111/j.1460-9592.2007.02321.x
36. Marin M, Broder KR, Temte JL, Snider DE, Seward JF. Use of combination
measles, mumps, rubella, and varicella vaccine: recommendations of the
Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep.
2010;59(RR-3):1-12.
37. Szilagyi PG, Rodewald LE. Missed opportunities for immunizations: a review of
the evidence. J Public Health Manag Pract. 1996;2(1):18-25. DOI:
10.1097/00124784-199600210-00005
38. Paz-Bailey G, Adams L, Wong JM, et al. Dengue Vaccine: Recommendations of the
Advisory Committee on Immunization Practices, United States, 2021. MMWR
Recomm Rep 2021;70(No. RR-6):1-18.
39. CDC. Use of diphtheria toxoid-tetanus toxoid-acellular pertussis vaccine as a
five- dose series. Supplemental recommendations of the Advisory Committee on
Immunization Practices (ACIP). MMWR Recomm Rep. 2000;49(RR-13):1-8.
40. Fiore AE, Wasley A, Bell BP. Prevention of hepatitis A through active or passive
immunization: recommendations of the Advisory Committee on Immunization
Practices (ACIP). MMWR Recomm Rep. 2006;55(RR-7):1-23.
41. Mast EE, Margolis HS, Fiore AE, et al. A comprehensive immunization strategy
to eliminate transmission of hepatitis B virus infection in the United States:
recommendations of the Advisory Committee on Immunization Practices (ACIP)
part 1: immunization of infants, children, and adolescents. MMWR Recomm
Rep. 2005;54(RR-16):1-31.
42. Briere EC, Rubin L, Moro PL, Cohn A, Clark T, Messonnier N. Prevention and
control of Haemophilus influenzae type b disease: recommendations of the
advisory committee on immunization practices (ACIP). MMWR Recomm
Rep. 2014;63(RR-1):1-14.
43. Markowitz L, Dunne E, Saraiya M, et al. Human papillomavirus vaccination:
recommendations of the Advisory Committee on Immunization Practices (ACIP).
MMWR Recomm Rep. 2014;63(RR-05):1-30.

General Best Practice Guidelines for Immunization: Contraindications and Precautions
70
44. Grohskopf LA, Sokolow LZ, Olsen SJ, et al. Prevention and Control of Seasonal
Influenza with Vaccines Recommendations of the Advisory Committee on
Immunization Practices — United States, 2016–17 Influenza Season. MMWR
Recomm Rep 2016;65(No. RR-5):1-54.
45. Prevots DR, Burr RK, Sutter RW, Murphy TV. Poliomyelitis prevention in
the United States. Updated recommendations of the Advisory Committee on
Immunization Practices (ACIP). MMWR Recomm Rep. 2000;49(RR-5):1-
22; quiz CE21-27.
46. Cohn AC, MacNeil JR, Clark TA, et al. Prevention and control of meningococcal
disease: recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR Recomm Rep. 2013;62(RR-2):1-28.
47. Bexsero Package Insert. Available at
www.fda.gov/BiologicsBloodVaccines/Vaccines/ApprovedProducts/ucm431374.
htm (accessed 05/04/17)
48. Trumenba Package Insert. Available at
www.fda.gov/downloads/BiologicsBloodVaccines/Vaccines/ApprovedProducts/
UCM421139.pdf (accessed 05/04/17)
49. Bilukha OO, Rosenstein N. Prevention and control of meningococcal disease.
Recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR Recomm Rep. 2005;54(RR-7):1-21.
50. Advisory Committee on Immunization Practices. Preventing pneumococcal
disease among infants and young children. Recommendations of the Advisory
Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly
Rep. 2000;49(RR-9):1-35.
51. CDC. Prevention of pneumococcal disease: recommendations of the Advisory
Committee on Immunization Practices (ACIP). MMWR Recomm Rep.
1997;46(RR-8):1-24.
52. Broder KR, Cortese MM, Iskander JK, et al. Preventing tetanus, diphtheria, and
pertussis among adolescents: use of tetanus toxoid, reduced diphtheria toxoid
and acellular pertussis vaccines recommendations of the Advisory Committee on
Immunization Practices (ACIP). MMWR Recomm Rep. 2006;55(RR-3):1-34.
53. Marin M, Guris D, Chaves SS, Schmid S, Seward JF. Prevention of varicella:
recommendations of the Advisory Committee on Immunization Practices (ACIP).
MMWR Recomm Rep. 2007;56(RR-4):1-40.
General Best Practice Guidelines for Immunization: Contraindications and Precautions
71
54. Harpaz R, Ortega-Sanchez IR, Seward JF. Prevention of herpes zoster:
recommendations of the Advisory Committee on Immunization Practices (ACIP).
MMWR Recomm Rep. 2008;57(RR-5):1-30; quiz CE32-34.
55. Grohskopf LA, Olsen SJ, Sokolow LZ, et al. Prevention and control of seasonal
influenza with vaccines: recommendations of the Advisory Committee on
Immunization Practices (ACIP)—United States, 2014-15 influenza season.
MMWR Morb Mortal Wkly Rep. 2014;63(32):691-697.
56. Cortese MM, Parashar UD. Prevention of rotavirus gastroenteritis among infants
and children: recommendations of the Advisory Committee on Immunization
Practices (ACIP). MMWR Recomm Rep. 2009;58(RR-2):1-25.
57. American Academy of Pediatrics. Passive immunization. In: Pickering L, Baker C,
Kimberlin D, Long S, eds. Red Book: 2012 Report of the Committee on Infectious
Diseases. 28th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2012.

General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
72
5. Preventing and Managing Adverse Reactions
Updates
Major changes to the best practice guidance include 1) more descriptive characterization
of anaphylactic allergy and 2) incorporation of protocols for managing adverse reactions.
Benefit and Risk Communication
Parents, guardians, legal representatives, and adolescent and adult patients should be
informed about the benefits of and risks from vaccines in language that is culturally
sensitive and at an appropriate educational level. Opportunity for questions should be
provided before each vaccination. Discussion of the benefits of and risks from vaccination
is sound medical practice and is required by law.
The National Childhood Vaccine Injury Act of 1986 (1) requires that vaccine information
materials be developed for each vaccine covered by the Act (uscode.house.gov). These
materials, known as vaccine information statements (VISs), must be provided by all public
and private vaccination providers each time a vaccine is administered. Copies of VISs are
available from state health authorities responsible for vaccination and from CDC
(www.cdc.gov/vaccines/hcp/vis/index.html). Translations of VISs into languages other
than English are available from certain state vaccination programs and from the
Immunization Action Coalition website (http://www.immunize.org). The act does not
require that a signature be obtained; however, documentation of consent might be
recommended or required by certain state or local health authorities or school authorities.
Some parents or patients question the need for or safety of vaccinations and want to
discuss the risks from and benefits of certain vaccines. Some refuse certain vaccines or
reject all vaccinations for personal or religious reasons. Having a basic understanding of
how patients and parents of patients view vaccine risk and developing effective
approaches to address vaccine safety concerns are imperative for vaccination providers.

General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
73
Each person understands and reacts to vaccine information on the basis of different
factors, including previous experience, education, personal values, method of data
presentation, perceptions of the risk for disease and perceived ability to control these
risks, and risk tolerance. In some circumstances, decisions about vaccination are based on
inaccurate information about risk provided by the media and certain websites. Websites
and other sources of vaccine information may be inaccurate or incomplete. Health care
providers can be a pivotal source of science-based credible information by discussing with
parents and patients the risks from and benefits of vaccines, which helps patients make
informed decisions.
When a parent or patient initiates a discussion about a perceived vaccine adverse reaction,
the health care provider should discuss the specific concerns and provide factual
information, using appropriate language. Effective, empathetic vaccine risk
communication is essential in responding to misinformation and concerns, with health
care providers recognizing that risk assessment and decision-making can be difficult and
confusing. Certain vaccines might be acceptable to a parent who is resistant to other
vaccines. This partial acceptance can be used to facilitate additional communication. Their
concerns can be addressed using the VIS and offering other resource materials (e.g.,
vaccination information from CDC: www.cdc.gov/vaccines/hcp/vis/index.html).
The American Academy of Pediatrics (AAP) does not recommend that providers exclude
from their practice patients whose parents or guardians question or refuse vaccination.
However, an effective public health strategy is to identify common ground and discuss
measures that need to be followed if the decision is to defer vaccination (2). Health care
providers should reinforce key points about each vaccine, including safety, and emphasize
risks for disease among unvaccinated children. Parents should be advised of state laws
regarding entry to schools or child-care facilities, which might require that unvaccinated
children be excluded from the facility during outbreaks (www.cdc.gov/vaccines/imz-
managers/coverage/schoolvaxview/requirements/index.html). These discussions should
be documented in the patient’s medical record, including the refusal to receive certain
vaccines (i.e., informed refusal). When a vaccine is refused when first offered the provider
should take the opportunity to offer the vaccine again at the next visit.

General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
74
Preventing Adverse Reactions
Vaccines are intended to produce active immunity to specific antigens. An adverse
reaction is an undesirable side effect that occurs after a vaccination. Vaccine adverse
reactions are classified as 1) local, 2) systemic, or 3) allergic (additional information is
available at
https://www.fda.gov/BiologicsBloodVaccines/Vaccines/ApprovedProducts/ucm09383
3.htm. Local reactions (e.g., redness) are usually the least severe and most frequent.
Systemic reactions (e.g., fever) occur less frequently than local reactions, and severe
allergic reactions (e.g., anaphylaxis) are the least frequent reactions. Severe adverse
reactions are rare (3).
Some of the systemic reactions may be complicated by the onset of syncope. Syncope
(vasovagal or vasodepressor reaction) can occur after vaccination and is most common
among adolescents and young adults. In 2005, the Vaccine Adverse Event Reporting
System (VAERS) began detecting a trend of increasing syncope reports that coincided with
the licensure of 3 vaccines for adolescents: human papillomavirus (HPV), MenACWY, and
Tdap (4). Of particular concern among adolescents has been the risk for serious secondary
injuries, including skull fracture and cerebral hemorrhage. Of 463 VAERS reports of
syncope during January 1, 2005, to July 31, 2007, a total of 41 listed syncope with
secondary injury with information on the timing after vaccination, and the majority of
these syncope reports (76%) occurred among adolescents. Among all age groups, 80% of
reported syncope episodes occur within 15 minutes of vaccine administration (additional
information is available at www.cdc.gov/vaccinesafety/concerns/fainting.html). Providers
should take appropriate measures to prevent injuries if a patient becomes weak or dizzy or
loses consciousness. Adolescents and adults should be seated or lying down during
vaccination.Vaccine providers, particularly when vaccinating adolescents, should consider
observing patients (with patients seated or lying down) for 15 minutes after vaccination to
decrease the risk for injury should they faint (4). If syncope develops, patients should be
observed until the symptoms resolve.
Although allergic reactions are a common concern for vaccine providers, these reactions
are uncommon and anaphylaxis following vaccines is rare, occurring at a rate of
approximately one per million doses for many vaccines (5).

General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
75
Epinephrine and equipment for managing an airway should be available for immediate
use (6). The best practice to prevent allergic reactions is to identify individuals at
increased risk by obtaining a history of allergy to previous vaccinations and vaccine
components that might indicate an underlying hypersensitivity. Acute allergic reactions
following vaccinations might be caused by the vaccine antigen, residual animal protein,
antimicrobial agents, preservatives, stabilizers, or other vaccine components (7).
Components of each vaccine are listed in the respective package insert. An extensive list of
vaccine components and their use, as well as the vaccines that contain each component,
has been published (8) and also is available from CDC
(www.cdc.gov/vaccines/pubs/pinkbook/downloads/appendices/b/excipient-table- 2.pdf).
Additional information and tables of potential allergens in different vaccines are available
at (http://www.vaccinesafety.edu/components-Allergens.htm). The allergens identified in
the history can be cross-checked against the allergens identified in package inserts.
Managing Acute Vaccine Reactions
Vaccine providers should be familiar with identifying immediate-type allergic reactions,
including anaphylaxis, and be competent in treating these events at the time of vaccine
administration. Providers should also have a plan in place to contact emergency medical
services immediately in the event of a severe acute vaccine reaction.
Allergic reactions can include: local or generalized urticaria (hives) or angioedema;
respiratory compromise due to wheezing or swelling of the throat; hypotension; and
shock. Immediate-immunoglobulin E (IgE)–mediated (type 1) immune reactions, such as
anaphylaxis, usually occur within minutes of parenteral administration and involve
specific IgE interactions with discrete antigens (9,10). Rapid recognition and initiation of
treatment are required to prevent possible progression to respiratory failure or
cardiovascular collapse. It is important to note that urticaria may not be present in all
cases of anaphylaxis. For respiratory or cardiovascular symptoms, or other signs or
symptoms of anaphylaxis, immediate intramuscular epinephrine is the treatment of
choice (11,12). Additional doses of epinephrine as well as other drugs also might be
indicated (Tables 5-1 and 5-2) (12). Considering the availability of emergency services,
providers should plan for the availability of at least 3 doses of epinephrine (in dosing
formulations appropriate to each and every age/group served by the provider) for a

General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
76
hypothetical emergency situation. If hypotension is present, the patient should be placed
in a recumbent position with the legs elevated. Maintenance of the airway, oxygen
administration, and intravenous normal saline might be necessary.
After the patient is stabilized, arrangements should be made for immediate transfer to an
emergency facility for additional evaluation and treatment. Because anaphylaxis may
recur after patients begin to recover, monitoring in a medical facility for several hours is
advised, even after complete resolution of symptoms and signs. Additional information on
management of patients with anaphylaxis has been published (9).
Persons Who Have Had an Allergic Reaction Following a
Previous Immunization
For an individual patient who has experienced an immediate reaction to immunization, it
is important to identify the type of reaction that occurred, obtain a history of prior allergic
reactions, and try to identify the particular agent responsible. An algorithm approach to
these patients has been published (13) and additional advice is available for allergists on
the evaluation of these adverse events (10). In general, a history of a severe allergic
reaction to a vaccine should be considered a contraindication to additional doses of the
same vaccine (13). Referral of the individual to an allergist for evaluation is usually
indicated to possibly determine the component responsible, before making decisions
regarding administration of the additional doses of the same vaccine or other vaccines that
have the same components. Patients who have not had a severe allergic reaction following
a vaccine, but who have a history of possible allergy to a vaccine component can often be
vaccinated safely after careful evaluation (6).
Influenza Vaccination of Persons with a History of Egg
Allergy
Severe allergic and anaphylactic reactions can occur in response to a number of influenza
vaccine components, but such reactions are rare (6). All but the recombinant inactivated
influenza vaccine may have come into contact with egg protein. The use of influenza
vaccines for persons with a history of egg allergy has been reviewed recently by ACIP (14).

General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
77
VAERS data mining did not identify a higher than expected proportion of serious allergic
events after influenza vaccination during the 2011-2012 season, relative to all other
reported vaccines and adverse events in the database. Persons with a history of egg allergy
should receive recombinant inactivated vaccine (if 18 years or older), or IIV.
Other measures, such as dividing and administering the vaccine by a 2-step approach and
skin testing with vaccine, are not recommended (10).
All vaccines should be administered in settings in which personnel and equipment for
rapid recognition and treatment of anaphylaxis are available. ACIP recommends that all
vaccination providers be certified in cardiopulmonary resuscitation (CPR), have an office
emergency plan, and ensure that all staff are familiar with the plan (6). Some persons who
report allergy to egg might not be egg-allergic. Those who are able to eat lightly cooked egg
(e.g., scrambled egg) without reaction are unlikely to be allergic.
Egg-allergic persons might tolerate egg in baked products (e.g., bread or cake). Tolerance
to egg-containing foods does not exclude the possibility of egg allergy (15). Egg allergy can
be confirmed by a consistent medical history of adverse reactions to eggs and egg-
containing foods, plus skin and/or blood testing for IgE antibodies to egg proteins.
A previous severe allergic reaction to influenza vaccine, regardless of the component
suspected to be responsible for the reaction, is a contraindication to future receipt of the
vaccine (14).
Yellow Fever Vaccination of Persons with a History of Egg
Allergy
Yellow fever vaccine contains egg protein. There have been insufficient studies to
determine which patients with egg allergy may be able to receive yellow fever vaccine, but
there are reports of patients with true egg allergy safely receiving yellow fever vaccine after
evaluation by specialists with expertise in the management of allergic reactions (16,17).
According to the manufacturer, persons who are able to eat eggs or egg products may
receive the vaccine (18). However, potential hypersensitivity reactions might occur in
persons with a history of minor reactions to eggs.

General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
78
For egg-sensitive persons, a scratch test or intradermal test can be performed before
administering the vaccine to check for reactivity.
If a person has a severe egg-sensitivity or has a positive skin test to the vaccine, but the
vaccination is recommended because of their travel destination-specific risk,
desensitization can be performed under direct supervision of a physician experienced in
the management of anaphylaxis.
The desensitization procedure is detailed in the product insert (see yellow fever
recommendations at
https://www.fda.gov/BiologicsBloodVaccines/Vaccines/ApprovedProducts/ucm09407
4.htm).
Vaccines with MMR or Varicella Components and Persons
with a History of Egg Allergy
Varicella vaccine is grown in human diploid cell cultures and can safely be administered to
persons with a severe allergy to eggs or egg proteins (19). Measles and mumps vaccine
viruses are grown in chick embryo fibroblast tissue culture. However, persons with a
severe egg allergy can receive measles- or mumps-containing vaccines in the usual manner
because the content of these proteins is extremely low (20). The rare severe allergic
reactions after measles- or mumps-containing vaccines or varicella are thought to be
caused by other components of the vaccine (e.g., gelatin) (21-24). MMR, MMRV, varicella
and other vaccines contain hydrolyzed gelatin as a stabilizer.
Vaccines and Persons with a History of Allergy to
Substances Other than Eggs
Persons who have had an anaphylactic reaction to gelatin or gelatin-containing products
should be evaluated by an allergist prior to receiving gelatin-containing vaccines (6).
Certain vaccines contain trace amounts of antimicrobial agents or other preservatives
(e.g., neomycin or thimerosal), although allergies to these are rare. No licensed vaccine
contains penicillin or penicillin derivatives.

General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
79
Most often, neomycin hypersensitivity manifests as contact dermatitis, a delayed-type
(cell-mediated) immune response rather than immediate-hypersensitivity (IgE-mediated
allergy)–type response (25,26). A history of delayed-type reactions to neomycin is not a
contraindication for administration of neomycin-containing vaccines. There has only been
1 reported case of immediate hypersensitivity reaction following a neomycin-containing
vaccine (27). Persons who have had anaphylactic reactions to neomycin should be
evaluated by an allergist prior to receiving vaccines containing neomycin (6).
Thimerosal, an organic mercurial compound in use since the 1930s, is added to certain
immunobiologics as a preservative. Since mid-2001, vaccines routinely recommended for
infants younger than 6 months of age have been manufactured without thimerosal as a
preservative (14). Live, attenuated vaccines have never contained thimerosal. Thimerosal-
free formulations of inactivated influenza vaccine are available. Inactivated influenza
vaccine also is available in formulations with only trace amounts of thimerosal, which
remains as a manufacturing residual but is not added at the higher concentration that
would be necessary for it to function as a preservative. Thimerosal at a preservative
concentration is present in certain other vaccines that can be administered to children
(e.g., Td and DT). Information about the thimerosal content of vaccines is available from
FDA at
https://www.fda.gov/BiologicsBloodVaccines/SafetyAvailability/VaccineSafety/UCM096
22 8.
Reactions to thimerosal have been described as local delayed-type hypersensitivity
reactions with only rare reports of immediate reactions (28-31). Thimerosal elicits positive
delayed-type hypersensitivity patch tests in 1%-18% of persons tested; however, these tests
have no relevance to acute allergic reactions that might occur within minutes or hours
after immunization (32,33). The majority of persons do not experience reactions to
thimerosal administered as a component of vaccines even when patch or intradermal tests
for thimerosal indicate hypersensitivity (31). A local or delayed-type hypersensitivity
reaction to thimerosal is not a contraindication to receipt of a vaccine that contains
thimerosal (34).

General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
80
Latex is sap from the rubber tree. Latex contains naturally occurring plant proteins that
can be responsible for immediate-type allergic reactions. Latex is processed to form either
natural rubber latex products such as gloves or dry, natural rubber products such as
syringe plunger tips and vial stoppers. Synthetic rubber is also used in gloves, syringe
plungers, and vial stoppers but does not contain the latex proteins linked to immediate-
type allergic reactions. Natural rubber latex or dry, natural rubber used in vaccine
packaging generally is noted in the manufacturers’ package inserts.
Immediate-type allergic reactions due to latex allergy have been described after
vaccination, but such reactions are rare (35).
If a person reports a severe anaphylactic allergy to latex, vaccines supplied in vials or
syringes that contain natural rubber latex should be avoided if possible (6). If not, if the
decision is made to vaccinate, providers should be prepared to treat immediate allergic
reactions due to latex, including anaphylaxis. The most common type of latex
hypersensitivity is a delayed-type (type 4, cell-mediated) allergic contact dermatitis (36).
For patients with a history of contact allergy to latex, vaccines supplied in vials or syringes
that contain dry natural rubber or natural rubber latex may be administered.
Reporting Adverse Events After Vaccination
Modern vaccines are safe and effective; however, adverse events have been reported after
administration of all vaccines (3). More complete information about adverse reactions to a
specific vaccine is available in the package insert for each vaccine and from CDC at
www.cdc.gov/vaccines/vac-gen/side-effects.htm. An adverse event is an untoward event
that occurs after a vaccination that might be caused by the vaccine product or vaccination
process. These events range from common, minor, local reactions to rare, severe, allergic
reactions (e.g., anaphylaxis). Reporting to VAERS helps establish trends, identify clusters
of adverse events, or generate hypotheses. However, establishing evidence for cause and
effect on the basis of case reports and case series alone is usually not possible, because
health problems that have a temporal association with vaccination do not necessarily
indicate causality.

General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
81
Many adverse events require more detailed epidemiologic studies to compare the
incidence of the event among vaccinees with the incidence among unvaccinated persons.
Potential causal associations between reported adverse events after vaccination can be
assessed through epidemiologic or clinical studies.
The National Childhood Vaccine Injury Act of 1986 (1) requires health care personnel and
vaccine manufacturers to report to VAERS specific adverse events that occur after
vaccination. The reporting requirements are different for manufacturers and health care
personnel. Manufacturers are required to report all adverse events that occur after
vaccination to VAERS, whereas health-care providers are required to report events that
appear in the reportable events table on the VAERS website at
https://vaers.hhs.gov/docs/VAERS_Table_of_Reportable_Events_Following_Vaccinat
ion.pdf.
In addition to the mandated reporting of events listed on the reportable events table,
health care personnel should report to VAERS all events listed in product inserts as
contraindications, as well as all clinically significant adverse events, even if they are
uncertain that the adverse event is related causally to vaccination (6). Persons other than
health care personnel also can report adverse events to VAERS.
General information on VAERS is available at https://vaers.hhs.gov/index.html. Specific
information for healthcare providers is available
at https://vaers.hhs.gov/resources/infoproviders.html. Reporting to VAERS is fully
electronic and can be done using an online reporting tool or a writable PDF; instructions
are available at https://vaers.hhs.gov/reportevent.html. Further assistance on VAERS
reporting is available through email at [email protected] and the VAERS toll free number
1-800-822-7967.
National Vaccine Injury Compensation Program
The National Vaccine Injury Compensation Program, established by the National
Childhood Vaccine Injury Act of 1986 (1), is a no-fault system in which persons thought to
have experienced an injury or to have died as a result of administration of a covered
vaccine can seek compensation.

General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
82
The program became operational on October 1, 1988, and is intended as an alternative to
civil litigation under the traditional tort system in that negligence need not be proven.
Claims arising from covered vaccines must first be adjudicated through the program
before civil litigation can be pursued.
The program relies on the Vaccine Injury Table, which lists the vaccines covered by the
program and the injuries (including death), disabilities, illnesses, and conditions for which
compensation might be awarded. The table defines the time during which the first
symptom or substantial aggravation of an injury must appear after vaccination to be
eligible. Successful claimants receive a legal presumption of causation if a condition listed
in the table is proven, thus avoiding the need to prove actual causation in an individual
case. Claimants also can prevail for conditions not listed in the reportable events table if
they prove causation for covered vaccines.
Additional information is available from the Health Resources and Services
Administration (HRSA at https://www.hrsa.gov/vaccine-compensation/index.html or by
telephone at 800-338- 2382). Persons who would like to file a claim for vaccine injury
should contact the U.S. Court of Federal Claims (717 Madison Place, N.W., Washington,
DC 20005; telephone: 202-357-6400).

General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
83
TABLE 5-1: Rapid overview: Emergent management of anaphylaxis in
infants and children
(a)
Diagnosis is
made
clinically:
The most common signs and symptoms are cutaneous (e.g., sudden onset of generalized
urticaria, angioedema, flushing, pruritus). However, 10 to 20% of patients have no skin
findings.
Danger signs: Rapid progression of symptoms, evidence of respiratory distress (e.g., stridor,
wheezing, dyspnea, increased work of breathing, retractions, persistent cough, cyanosis), signs
of poor perfusion, abdominal pain, vomiting, dysrhythmia, hypotension, collapse.
Acute
management:
The first and most important therapy in anaphylaxis is epinephrine. There are NO absolute
contraindications to epinephrine in the setting of anaphylaxis.
Airway: Immediate intubation if evidence of impending airway obstruction from angioedema.
Delay may lead to complete obstruction. Intubation can be difficult and should be performed
by the most experienced clinician available. Cricothyrotomy may be necessary.
IM epinephrine (1 mg/mL preparation): Epinephrine 0.01 mg/kg should be injected
intramuscularly in the mid-outer thigh. For large children (>50 kg), the maximum is 0.5 mg
per dose. If there is no response or the response is inadequate, the injection can be repeated in
5 to 15 minutes (or more frequently). If epinephrine is injected promptly IM, patients respond
to one, two, or at most, three injections. If signs of poor perfusion are present or symptoms are
not responding to epinephrine injections, prepare IV epinephrine for infusion (see below).
Place patient in recumbent position, if tolerated, and elevate lower extremities.
Oxygen: Give 8 to 10 L/minute via facemask or up to 100% oxygen, as needed.
Normal saline rapid bolus: Treat poor perfusion with rapid infusion of 20 mL/kg. Reevaluate
and repeat fluid boluses (20 mL/kg), as needed. Massive fluid shifts with severe loss of
intravascular volume can occur. Monitor urine output.
Albuterol: For bronchospasm resistant to IM epinephrine, give albuterol 0.15 mg/kg
(minimum dose: 2.5 mg) in 3 mL saline inhaled via nebulizer. Repeat, as needed.
H1 antihistamine: Consider giving diphenhydramine 1 mg/kg (max 50 mg) IV given over 5
minutes
, or cetirizine (children age 6 months to 5 years can receive 2.5 mg IV, those 6 to 11 years
of age can receive 5 or 10 mg IV, over 2 minutes).
H2 antihistamine: Consider giving famotidine 0.25 mg/kg (max 20 mg) IV, over at least 2
minutes.
Glucocorticoid: Consider giving methylprednisolone 1 mg/kg (max 125 mg) IV.
Monitoring: Continuous noninvasive hemodynamic monitoring and pulse oximetry monitoring
should be performed. Urine output should be monitored in patients receiving IV fluid
resuscitation for severe hypotension or shock.
Treatment of
refractory
symptoms:
Epinephrine infusion:
(b)
In patients with inadequate response to IM epinephrine and IV saline,
give epinephrine continuous infusion at 0.1 to 1 mcg/kg/minute, titrated to effect.
Vasopressors:
(b)
Patients may require large amounts of IV crystalloid to maintain blood
pressure. Some patients may require a second vasopressor (in addition to epinephrine). All
vasopressors should be given by infusion pump, with the doses titrated continuously according
to blood pressure and cardiac rate/function monitored continuously and oxygenation
monitored by pulse oximetry
IM: intramuscular; IV: intravenous.
(a)
A child is defined as a prepubertal patient weighing less than 40 kg.
(b)
All patients receiving an infusion of epinephrine and/or another vasopressor require continuous noninvasive
monitoring of blood pressure, heart rate and function, and oxygen saturation. We suggest that pediatric centers
provide instructions for preparation of standard concentrations and also provide charts for established infusion
rate for epinephrine and other vasopressors in infants and children.
Source: Campbell RL, Kelso JM. Anaphylaxis: Emergency treatment. In: UpToDate,
Post TW (Ed), UpToDate, Waltham, MA. (Accessed on December 9, 2021)
Source: (37).
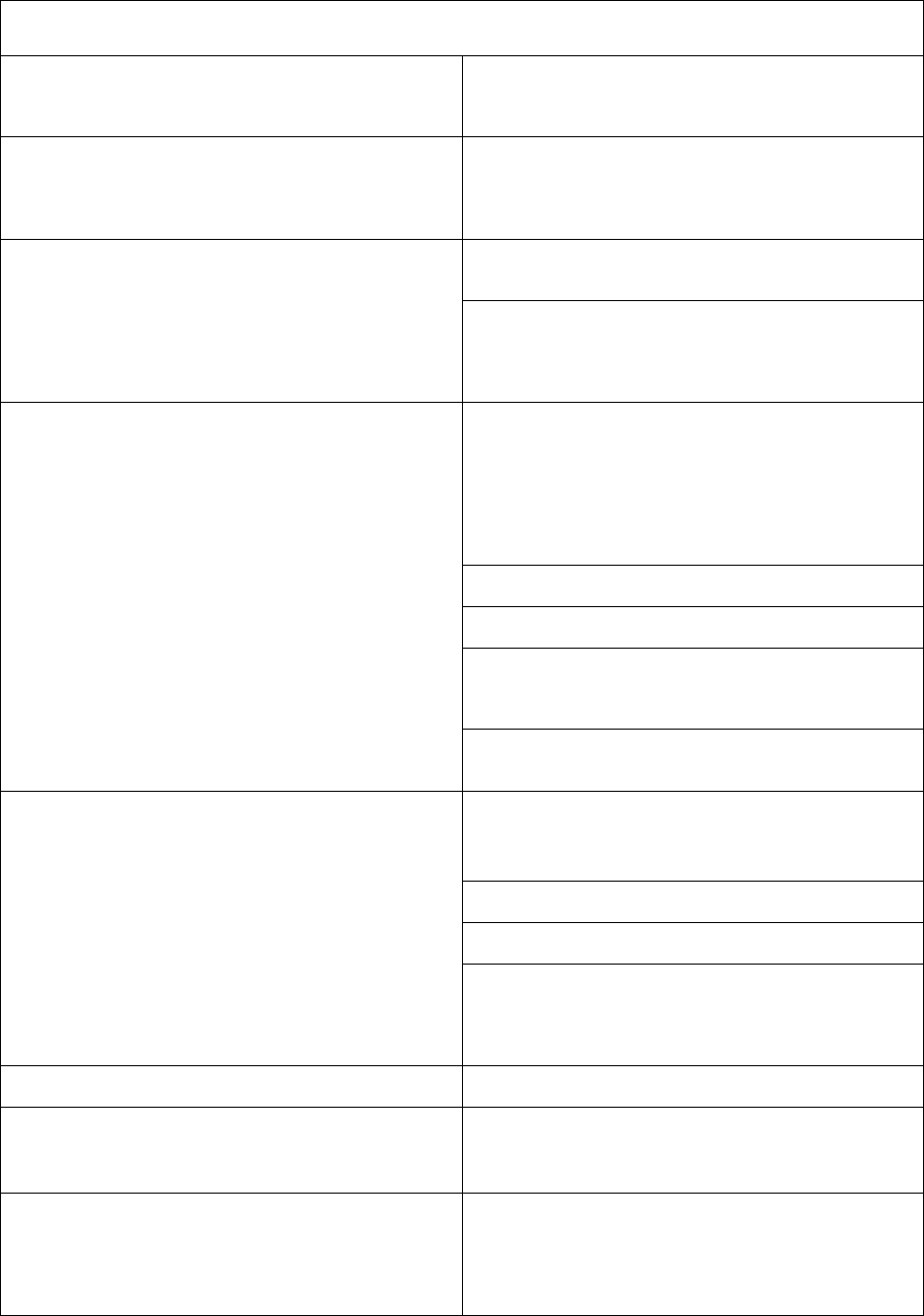
General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
84
Table 5-2: Rapid overview: Emergency management of anaphylaxis in
adults
Diagnosis is made clinically:
The most common signs and symptoms are cutaneous
(e.g., sudden onset of generalized urticaria,
angioedema, flushing, pruritus). However, 10 to 20% of
patients have no skin findings.
Danger signs: Rapid progression of symptoms,
respiratory distress (e.g., stridor, wheezing, dyspnea,
increased work of breathing, persistent cough,
cyanosis), vomiting, abdominal pain, hypotension,
dysrhythmia, chest pain, collapse.
Acute management:
The first and most important treatment in anaphylaxis
is epinephrine. There are NO absolute contraindications
to epinephrine in the setting of anaphylaxis.
Airway: Immediate intubation if evidence of impending
airway obstruction from angioedema. Delay may lead to
complete obstruction. Intubation can be difficult and
should be performed by the most experienced clinician
available. Cricothyrotomy may be necessary.
Promptly and simultaneously, give:
IM epinephrine (1 mg/mL preparation): Give
epinephrine 0.3 to 0.5 mg intramuscularly, preferably
in the mid-outer thigh. Can repeat every 5 to 15
minutes (or more frequently), as needed. If
epinephrine is injected promptly IM, most patients
respond to one, two, or at most, three doses. If
symptoms are not responding to epinephrine
injections, prepare IV epinephrine for infusion.
Place patient in recumbent position, if tolerated, and
elevate lower extremities.
Oxygen: Give 8 to 10 L/minute via facemask or up to
100% oxygen, as needed.
Normal saline rapid bolus: Treat hypotension with
rapid infusion of 1 to 2 liters IV. Repeat, as needed.
Massive fluid shifts with severe loss of intravascular
volume can occur.
Albuterol (salbutamol): For bronchospasm resistant to
IM epinephrine, give 2.5 to 5 mg in 3 mL saline via
nebulizer. Repeat, as needed.
Adjunctive therapies:
H1 antihistamine:
(a)
Consider giving cetirizine 10 mg
IV (given over 2 minutes) diphenhydramine 25 to 50
mg IV (given over 5 minutes) (for relief of urticaria
and itching only)
H2 antihistamine:
(a)
Consider giving famotidine 20
mg IV (given over 2 minutes).
Glucocorticoid:
(a)
Consider giving methylprednisolone
125 mg IV.
Monitoring: Continuous noninvasive hemodynamic
monitoring and pulse oximetry monitoring should be
performed. Urine output should be monitored in
patients receiving IV fluid resuscitation for severe
hypotension or shock.
Treatment of refractory symptoms:
Epinephrine infusion
(b),
: For patients with inadequate
response to IM epinephrine and IV saline, give
epinephrine continuous infusion, beginning at
0.1
mcg/kg/minute by infusion pump
(c)
. Titrate the dose
continuously according to blood pressure, cardiac rate
and function, and oxygenation.
Vasopressors
(b
: Some patients may require a second
vasopressor (in addition to epinephrine). All
vasopressors should be given by infusion pump, with
the doses titrated continuously according to blood
pressure and cardiac rate/function and oxygenation
monitored by pulse oximetry.

General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
85
Glucagon: Patients on beta blockers may not respond to
epinephrine and can be given glucagon 1 to 5 mg IV
over 5 minutes, followed by infusion of 5 to 15
mcg/minute. Rapid administration of glucagon can
cause vomiting.
Instructions on how to prepare and administer epinephrine for IV continuous infusions are available as
separate tables in UpToDate.
IM: intramuscular; IV: intravenous.
(a)
These medications should not be used as initial or sole treatment.
(b)
All patients receiving an infusion of epinephrine and another vasopressor require continuous noninvasive
monitoring of blood pressure, heart rate and function, and oxygen saturation.
(c)
For example, the initial infusion rate for a 70 kg patient would be 7 mcg/minute. This is consistent with the
recommended range for non–weight-based dosing for adults, which is 2 to 10 mcg/minute. Non–weight-based
dosing can be used if the patient's weight is not known and cannot be estimated.
Source: Campbell RL, Kelso JM. Anaphylaxis: Emergency treatment. In: UpToDate, Post
TW (Ed), UpToDate, Waltham, MA. (Accessed on December 9, 2021)
Source: (37).
General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
86
REFERENCES
1.
National Childhood Vaccine Injury Act, 42 U.S.C. Sect. 300aa-1 to 300aa-34
(1986).
2.
Opel DJ, Heritage J, Taylor JA, et al. The architecture of provider-parent
vaccine discussions at health supervision visits. Pediatrics. 2013:2013-2037.
DOI: 10.1542/peds.2013-2037
3.
CDC. Update: vaccine side effects, adverse reactions, contraindications, and
precautions. Recommendations of the Advisory Committee on Immunization
Practices (ACIP). MMWR Recomm Rep. 1996;45(RR-12):1-35.
4.
CDC. Syncope after vaccination—United States, January 2005-July 2007.
MMWR Morb Mortal Wkly Rep. 2008;57(17):457-460.
5.
Bohlke K, Davis RL, Marcy SM, et al. Risk of anaphylaxis after vaccination of
children and adolescents. Pediatrics. 2003;112(4):815-820. DOI:
10.1542/peds.112.4.815
6.
Kroger A, Atkinson W, Pickering L. General immunization practices. In:
Plotkin S, Orenstein W, Offit P, eds. Vaccines. 6th ed. China: Elsevier
Saunders; 2013:88-111.
7.
Grabenstein JD. Clinical management of hypersensitivities to vaccine
components. Hospital Pharmacy. 1997;32:77-87.
8.
Grabenstein JD. ImmunoFacts: Vaccines and Immunologic Drugs—2013.
38th ed. St. Louis, MO: Lippincott Williams & Wilkins; 2012.
9.
Lieberman P, Nicklas RA, Oppenheimer J, et al. The diagnosis and
management of anaphylaxis practice parameter: 2010 update. J Allergy Clin
Immunol. 2010;126(3):477-480.e471-442. DOI: 10.1016/j.jaci.2010.06.022
10.
Kelso JM, Greenhawt MJ, Li JT, et al. Adverse reactions to vaccines practice
parameter 2012 update. J Allergy Clin Immunol. 2012;130(1):25-43. DOI:
10.1016/j.jaci.2012.04.003
11.
Ruggeberg JU, Gold MS, Bayas JM, et al. Anaphylaxis: case definition and
guidelines for data collection, analysis, and presentation of immunization
safety data. Vaccine. 2007;25(31):5675-5684. DOI:
10.1016/j.vaccine.2007.02.064
General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
87
12.
Sampson HA, Munoz-Furlong A, Bock SA, et al. Symposium on the definition
and management of anaphylaxis: summary report. J Allergy Clin Immunol.
2005;115(3):584-591. DOI: 10.1016/j.jaci.2005.01.009
13.
Wood RA, Berger M, Dreskin SC, et al. An algorithm for treatment of patients
with hypersensitivity reactions after vaccines. Pediatrics. 2008;122(3):e771-
777. DOI: 10.1542/peds.2008-1002
14.
Grohskopf LA, Sokolow LZ, Olsen SJ, Bresee JS, Broder KR, Karron RA.
Prevention and control of influenza with vaccines: recommendations of the
Advisory Committee on Immunization Practices, United States, 2015-16
influenza season. MMWR Morb Mortal Wkly Rep. 2015;64(30):818-825.
15.
Erlewyn-Lajeunesse M, Brathwaite N, Lucas JS, Warner JO.
Recommendations for the administration of influenza vaccine in children
allergic to egg. BMJ. 2009;339:b3680. DOI: 10.1136/bmj.b3680
16.
Mosimann B, Stoll B, Francillon C, Pecoud A. Yellow fever vaccine and egg
allergy. J Allergy Clin Immunol. 1995;95(5 Pt 1):1064. DOI: 10.1016/S0091-
6749(95)70118-4
17.
Munoz-Cano R, Sanchez-Lopez J, Bartra J, Valero A. Yellow fever vaccine and
egg allergy: really a problem? Allergy. 2010;65(4):533-534. DOI:
10.1111/j.1398-9995.2009.02205.x
18.
Sanofi Pasteur Inc. Yellow fever vaccine: YF-VAX®[Package insert].
Swiftwater, PA: Sanofi Pasteur Inc.; 2015. Available at:
http://www.fda.gov/downloads/BiologicsBloodVaccines/Vaccines/Approved
Products/UCM142831.pdf. Accessed 02 Feb 2017.
19.
Marin M, Guris D, Chaves SS, Schmid S, Seward JF. Prevention of varicella:
recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR Recomm Rep. 2007;56(RR-4):1-40.
20.
Watson JC, Hadler SC, Dykewicz CA, Reef S, Phillips L. Measles, mumps, and
rubella—vaccine use and strategies for elimination of measles, rubella, and
congenital rubella syndrome and control of mumps: recommendations of the
Advisory Committee on Immunization Practices (ACIP). MMWR Recomm
Rep. 1998;47(RR-8):1-57.
General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
88
21.
Kelso JM, Jones RT, Yunginger JW. Anaphylaxis to measles, mumps, and
rubella vaccine mediated by IgE to gelatin. J Allergy Clin Immunol.
1993;91(4):867-872. DOI: 10.1016/0091-6749(93)90344-F
22.
Sakaguchi M, Nakayama T, Inouye S. Food allergy to gelatin in children with
systemic immediate-type reactions, including anaphylaxis, to vaccines. J
Allergy Clin Immunol. 1996;98(6 Pt 1):1058-1061. DOI: 10.1016/S0091-
6749(96)80191-6
23.
Sakaguchi M, Ogura H, Inouye S. IgE antibody to gelatin in children with
immediate-type reactions to measles and mumps vaccines. J Allergy Clin
Immunol. 1995;96(4):563-565. DOI: 10.1016/S0091-6749(95)70304-7
24.
Sakaguchi M, Yamanaka T, Ikeda K, et al. IgE-mediated systemic reactions to
gelatin included in the varicella vaccine. J Allergy Clin Immunol.
1997;99(2):263-264. DOI: 10.1016/S0091-6749(97)70108-8
25.
Rietschel RL, Bernier R. Neomycin sensitivity and the MMR vaccine. JAMA.
1981;245(6):571. DOI: 10.1001/jama.1981.03310310017008
26.
Elliman D, Dhanraj B. Safe MMR vaccination despite neomycin allergy.
Lancet. 1991;337(8737):365. DOI: 10.1016/0140-6736(91)90995-2
27.
Kwittken PL, Rosen S, Sweinberg SK. MMR vaccine and neomycin allergy.
Am J Dis Child. 1993;147(2):128-129. DOI:
10.1001/archpedi.1993.02160260018005
28.
Aberer W. Vaccination despite thimerosal sensitivity. Contact Dermatitis.
1991;24(1):6-10. DOI: 10.1111/j.1600-0536.1991.tb01621.x
29.
Cox NH, Forsyth A. Thiomersal allergy and vaccination reactions. Contact
Dermatitis. 1988;18(4):229-233. DOI: 10.1111/j.1600-0536.1988.tb02809.x
30.
Kirkland LR. Ocular sensitivity to thimerosal: a problem with hepatitis B
vaccine? South Med J. 1990;83(5):497-499.
31.
Zheng W, Dreskin SC. Thimerosal in influenza vaccine: an immediate
hypersensitivity reaction. Ann Allergy Asthma Immunol. 2007;99(6):574-
575. DOI: 10.1016/s1081-1206(10)60391-2
32.
Wantke F, Demmer CM, Götz M, Jarisch R. Contact dermatitis from
thimerosal. Contact Dermatitis. 1994;30(2):115. DOI: 10.1111/j.1600-
0536.1994.tb00580.x
General Best Practice Guidelines for Immunization: Preventing and Managing Adverse Reactions
89
33.
Moller H. All these positive tests to thimerosal. Contact Dermatitis.
1994;31(4):209-213. DOI: 10.1111/j.1600-0536.1994.tb01989.x
34.
Russell M, Pool V, Kelso JM, Tomazic-Jezic VJ. Vaccination of persons
allergic to latex: a review of safety data in the Vaccine Adverse Event
Reporting System (VAERS). Vaccine. 2004;23(5):664-667. DOI:
10.1016/j.vaccine.2004.06.042
35.
Lear JT, English JS. Anaphylaxis after hepatitis B vaccination. Lancet.
1995;345(8959):1249. DOI: 10.1016/S0140-6736(95)92039-0
36.
Slater JE. Latex allergy. J Allergy Clin Immunol. 1994;94(2 Pt 1):139-149;
quiz 150. DOI: 10.1053/ai.1994.v94.a55437
37.
Adapted from: Simons FER. Anaphylaxis. J Allergy Clin Immunol.
2010;125:S161.

General Best Practice Guidelines for Immunization: Vaccine Administration
90
6. Vaccine Administration
Infection Control and Sterile Technique
General Precautions
Persons administering vaccinations should follow appropriate precautions to minimize
risk for disease exposure and spread. Hands should be cleansed with an alcohol-based
waterless antiseptic hand rub or washed with soap and water before preparing vaccines for
administration and between each patient contact (1). Occupational Safety and Health
Administration (OSHA) regulations do not require gloves to be worn when administering
vaccinations, unless persons administering vaccinations have open lesions on their hands
or are likely to come into contact with a patient’s body fluids (2). If worn, gloves should be
changed between patients.
Vaccine Administration: Preparation and Timely Disposal
Vaccines should be drawn up in a designated clean medication area that is not adjacent to
areas where potentially contaminated items are placed. Multi-dose vials to be used for
more than one patient should not be kept or accessed in the immediate patient treatment
area. This is to prevent inadvertent contamination of the vial through direct or indirect
contact with potentially contaminated surfaces or equipment that could then lead to
infections in subsequent patients (3). Smallpox vaccine is accessed by dipping a
bifurcated needle directly into the vaccine vial. The vaccine adheres to the sides of the
bifurcated needle, and is administered via skin puncture. The vial must be accessed in the
immediate patient area to reduce environmental contamination by vaccine virus. To
prevent contamination of the vial, make sure the patient area is clean and free of
potentially contaminated equipment.
Different single-components of combination vaccines should never be mixed in the same
syringe by an end-user unless specifically licensed for such use (4). Single-dose vials and
manufacturer-filled syringes are designed for single-dose administration and should be
discarded if vaccine has been withdrawn or reconstituted and subsequently not used
within the time frame specified by the manufacturer. Syringes that are prefilled by the
manufacturer and activated (i.e., syringe cap removed or needle attached) but unused
should be discarded at the end of the clinic day. For non-live vaccines, manufacturers
General Best Practice Guidelines for Immunization: Vaccine Administration
91
typically recommend use within the same day that a vaccine is withdrawn or reconstituted.
For live vaccines that require reconstitution, manufacturers typically recommend the
vaccine be used as soon as possible after reconstitution and be discarded if not used within
30 minutes after reconstitution. For example, varicella vaccine should be discarded if not
used within 30 minutes after reconstitution, whereas MMR vaccine, once reconstituted,
must be kept in a dark place at 36°F to 46°F (2°C to 8°C) and should be discarded within 8
hours if not used. When in doubt about the appropriate handling of a vaccine, vaccination
providers should contact that vaccine’s manufacturer.
ACIP discourages the routine practice of providers’ prefilling syringes for several reasons.
Because the majority of vaccines have a similar appearance after being drawn into a
syringe, prefilling might result in administration errors. Because unused prefilled syringes
also typically must be discarded if not used within the same day that they are filled,
vaccine wastage might occur. The FDA does not license administration syringes for
vaccine storage.
In certain circumstances in which a single vaccine type is being used (e.g., in preparation
for a community influenza vaccination campaign), filling a small number (10 or fewer) of
syringes may be considered (5). The doses should be administered as soon as possible
after filling, by the same person who filled the syringes. Unused syringes that are prefilled
by the manufacturer and activated (i.e., syringe cap removed or needle attached) should be
discarded at the end of the clinic day. Vaccine from two or more vials should never be
combined to make one or more doses. This can lead to violation of expiration dates and
product contamination (6,7).
Health Care Provider Exposure to Vaccine Components
Providers are sometimes concerned when they have the same contraindications or
precautions as their patients from whom they withhold or defer vaccine. For
administration of routinely recommended vaccines, there is no evidence of risk of
exposure of vaccine components to the health care provider, so conditions in the provider
labeled as contraindications and precautions to a vaccine components are not a reason to
withdraw from this function of administering the vaccine to someone else. Historic
concerns about exposure to vaccine components are limited to non-parenteral vaccines in
which some degree of environmental exposure is unavoidable (5, 8), or situations in which
General Best Practice Guidelines for Immunization: Vaccine Administration
92
self-inoculation is likely due to the nature of the vaccine microbe [e.g. reduced attenuation
of smallpox vaccine virus (9)]. Persons administering ACAM 2000 smallpox vaccine to
laboratory and health care personnel at risk for occupational exposure to orthopoxviruses
can decrease the risk for inadvertent infection through recommended infection prevention
measures. However, because of a theoretical risk for infection, vaccination with
ACAM2000 can be offered to health care personnel administering this vaccine, provided
individual persons have no specified contraindications to vaccination (10).
Safe Use of Needles and Syringes
Needles and syringes used for vaccine injections must be sterile and disposable. A separate
needle and syringe should be used for each injection. Changing needles between drawing
vaccine from a vial and injecting it into a recipient is not necessary unless the needle has
been damaged or contaminated (11).
Bloodborne diseases (e.g., hepatitis B, hepatitis C, human immunodeficiency virus [HIV])
are occupational hazards for clinicians and other health-care providers. The Needlestick
Safety and Prevention Act (2) was enacted in 2000 to reduce the incidence of needlestick
injury and the consequent risk for bloodborne diseases acquired from patients. The act
directed OSHA to strengthen its existing bloodborne pathogen standards. The revised
standards became effective in 2001 (2). These federal regulations require the use of
engineering and work practice controls to eliminate or minimize employee exposure to
bloodborne pathogens. Engineering controls means controls (e.g., sharps disposal
containers, self-sheathing needles, safer medical devices, such as sharps with engineered
sharps injury protections and needleless systems) that isolate or remove the bloodborne
pathogens hazard from the workplace). Needle-shielding or needle-free devices that might
satisfy the occupational safety regulations for administering injectable vaccines are
available in the United States (12-13). The regulations also require maintenance of records
documenting injuries caused by needles and other medical sharp objects and that
nonmanagerial employees be involved in the evaluation and selection of safety-engineered
devices before they are procured. Additional information about implementation and
enforcement of these regulations is available from OSHA.

General Best Practice Guidelines for Immunization: Vaccine Administration
93
To prevent inadvertent needlestick injury or reuse, safety mechanisms should be deployed
after use and needles and syringes should be discarded immediately in labeled, puncture-
proof containers located in the same room where the vaccine is administered (5). Used
needles should never be recapped.
Route of Administration
Injectable Route
Routes of administration are recommended by the manufacturer for each immunobiologic
(Table 6-1). With the exceptions of bacille Calmette-Guérin (BCG) vaccine and smallpox
vaccine [ACAM2000] (both administered by the percutaneous route), injectable vaccines
are administered by the intramuscular or subcutaneous route. The smallpox/monkeypox
vaccine (Jynneos) is primarily administered by the subcutaneous route but in some
circumstances is administered by the intradermal route. Deviation from the recommended
route of administration might reduce vaccine efficacy (14-15) or increase the risk for local
adverse reactions (16-18).
The method of administration of injectable vaccines is determined, in part, by the
inclusion of adjuvants in some vaccines. An adjuvant is a vaccine component distinct from
the antigen that enhances the immune response to the antigen, but might also increase
risk of adverse reactions. To decrease risk of local adverse events, non-live vaccines
containing an adjuvant should be injected into a muscle. Administering a vaccine
containing an adjuvant either subcutaneously or intradermally can cause local irritation,
induration, skin discoloration, inflammation, and granuloma formation.
Intramuscular Injections
Needle Length
Injectable immunobiologics should be administered where local, neural, vascular, or tissue
injury is unlikely. Use of longer needles has been associated with less redness or swelling
than occurs with shorter needles because of injection into deeper muscle mass (16).
Appropriate needle length depends on age and body mass. Injection technique is the most
important parameter to ensure efficient intramuscular vaccine delivery.

General Best Practice Guidelines for Immunization: Vaccine Administration
94
For all intramuscular injections, the needle should be long enough to reach the muscle
mass and prevent vaccine from seeping into subcutaneous tissue, but not so long as to
involve underlying nerves, blood vessels, or bone (15,19-22). Vaccinators should be
familiar with the anatomy of the area into which they are injecting vaccine.
Intramuscular injections are administered at a 90-degree angle to the skin, preferably into
the anterolateral aspect of the thigh or the deltoid muscle of the upper arm, depending on
the age of the patient (Table 6-2).
The needle gauge for intramuscular injection is 22-25 gauge. A decision on needle length
and site of injection must be made for each person on the basis of the size of the muscle,
the thickness of adipose tissue at the injection site, the volume of the material to be
administered, injection technique, and the depth below the muscle surface into which the
material is to be injected (Figure 1). Some experts allow intramuscular injection with a ⅝-
inch needle but ONLY if the skin is stretched flat (21). If the subcutaneous and muscle
tissue are bunched to minimize the chance of striking bone (19), a 1-inch needle or larger
is required to ensure intramuscular administration. Aspiration before injection of vaccines
or toxoids (i.e., pulling back on the syringe plunger after needle insertion but before
injection) is not necessary because no large blood vessels are present at the recommended
injection sites, and a process that includes aspiration might be more painful for infants
(22).
Infants (Aged <12 Months)
For the majority of infants, the anterolateral aspect of the thigh is the recommended site
for injection because it provides comparatively larger muscle mass than the deltoid
(Figure 2) (23). In certain circumstances (e.g., physical obstruction to other sites and no
reasonable indication to defer doses), the gluteal muscle can be used. If the gluteal muscle
must be used, care should be taken to define the anatomic landmarks.
(a)
For the majority of
infants, a 1-inch needle is sufficient to penetrate the thigh muscle.
Toddlers (Aged 12 Months-2 Years)
For toddlers, the anterolateral thigh muscle is preferred, and when this site is used, the
needle should be at least 1 inch long. The deltoid muscle can be used if the muscle mass is
adequate. If 2 vaccines are to be administered in a single limb, they should be spaced an
inch apart (4, 24).

General Best Practice Guidelines for Immunization: Vaccine Administration
95
Children (Aged 3-10 Years)
The deltoid muscle is preferred for children aged 3-10 years (25); the needle length for
deltoid site injections can range from ⅝ to 1 inch on the basis of technique. The
anterolateral thigh can also be used (25). In this case the needle length should be 1 inch to
1.25 inches. Knowledge of body mass can be useful for estimating the appropriate needle
length (26).
Young Adolescents (Aged 11-18 years)
The deltoid muscle is preferred for adolescents 11-18 years of age. The anterolateral thigh
can also be used. For injection into the anterolateral thigh, most adolescents will require a
1-1.5-inch needle to ensure intramuscular administration (27).
Adults (Aged ≥19 Years)
For adults, the deltoid muscle is recommended for routine intramuscular vaccinations
(23) (Figure 3). The anterolateral thigh also can be used. When injecting into the deltoid
muscle, for adults a measurement of body mass/weight is allowable prior to vaccination,
understanding that resources to measure body mass/weight are not available in all clinical
settings. For men and women who weigh <130 lbs (<60 kg), a ⅝-inch needle is sufficient
to ensure intramuscular injection in the deltoid muscle if the injection is made at a 90-
degree angle and the tissue is not bunched. For men and women who weigh 130-152 lbs
(60-70 kg), a 1-inch needle is sufficient. For women who weigh 152-200 lbs (70-90 kg) and
men who weigh 152-260 lbs (70-118 kg), a 1- to 1.5-inch needle is recommended. For
women who weigh >200 lbs (>90 kg) or men who weigh >260 lbs (>118 kg), a 1.5-inch
needle is recommended (table 6-2) (20). For injection into the anterolateral thigh muscle,
a 1.5-inch needle should be used, although a 1-inch needle may be used if the skin is
stretched tightly and subcutaneous tissues are not bunched.
Subcutaneous Injections
Subcutaneous injections are administered at a 45-degree angle, usually into the thigh for
infants aged <12 months and in the upper-outer triceps area of persons aged ≥12 months.
Subcutaneous injections may be administered into the upper-outer triceps area of an
infant if necessary. A ⅝-inch, 23- to 25-gauge needle should be inserted into the
subcutaneous tissue (Figures 4 and 5) (4).
General Best Practice Guidelines for Immunization: Vaccine Administration
96
Oral Route
Rotavirus, adenovirus, cholera vaccine, and oral typhoid vaccines are the only vaccines
administered orally in the United States. Oral typhoid capsules should be administered as
directed by the manufacturer. The capsules should not be opened or mixed with any other
substance. Rotavirus vaccines are licensed for infants. There are 2 brands of rotavirus
vaccine, and they have different types of applicators. Providers should consult package
inserts for details.
Intranasal Route
Live attenuated influenza vaccine is approved for healthy nonpregnant persons aged 2-49
years and is the only vaccine administered by the intranasal route. The administration
device is a nasal sprayer with a dose-divider clip that allows introduction of one 0.1-mL
spray into each naris. The tip should be inserted slightly into the naris before
administration. Even if the person coughs or sneezes immediately after administration or
the dose is expelled any other way, the vaccine dose need not be repeated (5).
Severely immunosuppressed persons (i.e., those who require care in a protected
environment, e.g., bone marrow transplant recipients, individuals with severe combined
immunodeficiency diseases) should not administer LAIV. It would be uncommon for
persons with these conditions to be in a role administering vaccines. Other persons at
increased risk for influenza complications can administer LAIV. These include persons
with underlying medical conditions placing them at higher risk or who are likely to be at
risk, including pregnant women, persons with asthma, and persons aged ≥50 years (2).
Multiple Injections
If multiple vaccines are administered at a single visit, administer each preparation at a
different anatomic site (28). The location of all injection sites with the corresponding
vaccine injected should be documented in each patient’s medical record. Health-care
practices should consider using a vaccination site map so that all persons administering
vaccines routinely use a particular anatomic site for each particular vaccine.
For infants and younger children, if more than 2 vaccines are injected in a single limb, the
thigh is the preferred site because of the greater muscle mass; the injections should be
sufficiently separated (separate anatomic sites [i.e. ≥1 inch] if possible) so that any local
General Best Practice Guidelines for Immunization: Vaccine Administration
97
reactions can be differentiated (13,29). For older children and adults, the deltoid muscle
can be used for more than one intramuscular injection.
If a vaccine and an immune globulin preparation are administered simultaneously (e.g.,
Td/Tdap and tetanus immune globulin [TIG], hepatitis B and hepatitis B immunoglobulin
[HBIG]), separate limbs should be used for each injection (29-30).
Jet Injections
Jet injectors are needle-free devices that pressurize liquid medication, forcing it through a
nozzle orifice into a narrow stream capable of penetrating skin to deliver a drug or vaccine
into intradermal, subcutaneous, or intramuscular tissues (32-33). Immune responses
generated by jet injectors against both attenuated and non-live viral and bacterial antigens
are usually equivalent to, and occasionally greater than, immune responses induced by
needle injection. However, local reactions or injuries (e.g., skin laceration, transient
neuropathy, hematoma) are sometimes more frequent on delivery of vaccine by jet
injectors compared with needle injection, depending on the inherent irritability of the
vaccine and operator technique (33).
Multiple use jet injectors using the same nozzle for consecutive injections without
intervening sterilization were used in mass vaccination campaigns from the 1950s through
the 1990s (33); however, these were found to be unsafe because of the possibility of
bloodborne pathogen transmission (34-37) and should not be used. A new generation of
jet injectors with disposable cartridges and syringes has been developed since the 1990s.
With a new, sterile dose chamber and nozzle for each patient and correct use, these
devices do not have the same safety concerns as multiple-use nozzle jet injectors. Several
of the newer devices have been approved by FDA for use with specific vaccines (33). Jet
injectors prevent needlestick injuries to health-care providers (2) and can overcome
improper, unsterile reuse and other drawbacks of needles and syringes in developing
countries (9, 38-39).

General Best Practice Guidelines for Immunization: Vaccine Administration
98
Methods for Alleviating Discomfort and Pain Associated
with Vaccination
Comfort measures, such as distraction (e.g., playing music or pretending to blow away the
pain), cooling of the injection site(s), topical analgesia, ingestion of sweet liquids,
breastfeeding, swaddling, and slow, lateral swaying can help infants or children cope with
the discomfort associated with vaccination (40-42). Pretreatment (30-60 minutes before
injection) with a 5% topical lidocaine-prilocaine emulsion might decrease the pain of
vaccination by causing superficial anesthesia (43-44). Evidence indicates that this cream
does not interfere with the immune response to MMR (45). There is no evidence the cream
interferes with other vaccines (46-49).
Topical lidocaine-prilocaine emulsion should not be used on infants aged <12 months who
are receiving treatment with methemoglobin-inducing agents (e.g., acetaminophen, amyl
nitrate, nitroprusside, dapsone) because of the possible development of
methemoglobinemia (50). Use of a topical refrigerant (vapocoolant) spray immediately
before vaccination can reduce the short-term pain associated with injections and can be as
effective as lidocaine-prilocaine cream (51).
Evidence does not support use of antipyretics before or at the time of vaccination;
however, they can be used for the treatment of fever and local discomfort that might occur
following vaccination. Studies of children with previous febrile seizures have not
demonstrated antipyretics to be effective in the prevention of febrile seizures (48).
Clinical Implications of Nonstandard Vaccination
Practices
Best practice guidance for route, site, and dosage of immunobiologics is derived from data
from clinical trials, practical experience, normal intervals of health care visits, and
theoretical considerations. ACIP discourages variations from the recommended route, site,
volume, or number of doses of any vaccine.
Variation from the recommended route and site can result in inadequate protection. In
adults (but not in infants) (52), the immunogenicity of hepatitis B is substantially lower
when the gluteal rather than the deltoid site is used for administration (8). Hepatitis B
administered intradermally might result in a lower seroconversion rate and final titer of
General Best Practice Guidelines for Immunization: Vaccine Administration
99
hepatitis B surface antibody than when administered by the deltoid intramuscular route
(53-54). Hepatitis B administered by any route other than intramuscular, or in adults at
any site other than the deltoid or anterolateral thigh, should not be counted as valid and
should be repeated (9). Similarly, doses of rabies vaccine administered in the gluteal site
should not be counted as valid doses and should be repeated (54). Hepatitis A vaccine and
meningococcal conjugate vaccine do not need to be repeated if administered by the
subcutaneous route (55-56). However, for DTaP, Hib, and PCV13, there is no evidence
related to immunogenicity of these 3 vaccines given subcutaneously. Providers should
address circumstances in which dose(s) of these vaccines have been administered
subcutaneously on a case-by-case basis.
Inactivated influenza vaccine is immunogenic when administered in a lower-than-
standard dose by the intradermal route to healthy adult volunteers. Intradermal injection
produced antibody responses similar to intramuscular injection in vaccinees aged 18-60
years (57). However, the immunogenicity for persons aged ≥65 years is inadequate, and
varying the recommended route and dose either with the intradermal product licensed
through 64 years of age or with other influenza vaccines is not recommended (24).
Live, attenuated injectable vaccines (e.g., MMR, varicella, yellow fever) and certain non-
live vaccines (e.g., meningococcal polysaccharide) are recommended by the manufacturers
to be administered by subcutaneous injection. PPSV23and IPV are recommended by the
manufacturer to be administered by the subcutaneous or intramuscular route. Response
to vaccines recommended by the subcutaneous route is unlikely to be affected if the
vaccines are administered by the intramuscular rather than subcutaneous route.
Repeating doses of vaccine administered by the intramuscular route when recommended
to be by the subcutaneous route is not necessary (10).
Administering volumes smaller than recommended (e.g., inappropriately divided doses)
might result in inadequate protection. Using reduced doses administered at multiple
vaccination visits that equal a full dose or using smaller divided doses is not recommended
(4). Any vaccination using less than the standard dose should not be counted, and the
person should be revaccinated according to age unless serologic testing indicates that an
adequate response has developed. However, if 2 half-volume formulations of vaccine have
already been administered on the same clinic day to a patient recommended for the full
volume formulation, these 2 doses can count as one full dose. If less than a full
General Best Practice Guidelines for Immunization: Vaccine Administration
100
recommended dose of a vaccine is administered because of syringe, applicator, or needle
leakage, the dose should be repeated (5). Using larger-than-recommended dosages can be
hazardous because of excessive local or systemic concentrations of antigens or other
vaccine constituents.
(a)
If the gluteal muscle is chosen, injection should be administered lateral and superior to a line between the posterior superior iliac
spine and the greater trochanter or in the ventrogluteal site, the center of a triangle bounded by the anterior superior iliac spine, the
tubercle of the iliac crest, and the upper border of the greater trochanter.

General Best Practice Guidelines for Immunization: Vaccine Administration
101
TABLE 6-1. Dose and route of administration for selected vaccines
Vaccine
Dose
Route
DEN4CYD
0.5 mL
Subcut
DTaP, DT, Td, Tdap
0.5 mL
IM
DTaP-HepB-IPV
0.5 mL
IM
DTaP/Hib
0.5 mL
IM
DTaP-IPV/Hib
0.5 mL
IM
DTaP-IPV
0.5 mL
IM
Hib
0.5 mL
IM
Hib-MenCY
0.5 mL
IM
HepA ≤18 years: 0.5 mL
≥19 years: 1.0 mL
IM
HepB ≤19 years: 0.5 mL(a)
≥20 years: 1.0 mL
IM
HepA-HepB
≥18 years: 1.0 mL
IM
LAIV 0.2 mL divided dose between
nares
Intranasal spray
IIV 6-35 months: 0.25 mL or 0.5 mL
≥3 years: 0.5 mL(b)
IM
MenB
0.5 mL
IM
MMR
0.5 mL
Subcut
MMRV
0.5 mL
Subcut
MenACWY
0.5 mL
IM
MPSV4
0.5 mL
Subcut
PCV13
0.5 mL
IM
PCV15
0.5 mL
IM
PCV20
0.5 mL
IM
PPSV23
0.5 mL
IM or Subcut
HPV
0.5 mL
IM
IPV
0.5 mL
IM or Subcut
Rotavirus (RV1 or RV5)
(1.0 mL or 2.0 mL)
Oral
Varicella
0.5 mL
Subcut
RZV
0.5 mL(c)
IM
Abbreviations: DEN4CYD = dengue vaccine; DT = diphtheria and tetanus toxoids; DTaP = diphtheria and tetanus
toxoids and acellular pertussis; HepA = hepatitis A; HepB = hepatitis B; Hib = Haemophilus influenzae type b; HPV =
human papillomavirus; IIV = inactivated influenza vaccine; IM = intramuscular; IPV = inactivated poliovirus; LAIV =

General Best Practice Guidelines for Immunization: Vaccine Administration
102
TABLE 6-1. Dose and route of administration for selected vaccines
Vaccine
Dose
Route
live, attenuated influenza vaccine; MenACWY = quadrivalent meningococcal conjugate vaccine; MenCY = bivalent
meningococcal conjugate vaccine component; MMR = measles, mumps, and rubella; MMRV = measles, mumps,
rubella, and varicella; MPSV4 = quadrivalent meningococcal polysaccharide vaccine; PCV13 = pneumococcal
conjugate vaccine; PPSV23= pneumococcal polysaccharide vaccine; RV1 = live, attenuated monovalent rotavirus
vaccine; RV5 = live, reassortment pentavalent rotavirus vaccine; RZV = recombinant adjuvanted zoster vaccine;
Subcut = subcutaneous; Td = tetanus and diphtheria toxoids; Tdap = tetanus toxoid, reduced diphtheria toxoid, and
acellular pertussis.
Source: Adapted from Immunization Action Coalition: http://www.immunize.org.
(a)
Persons aged 11-15 years may be administered Recombivax HB (Merck), 1.0 mL (adult formulation) on a 2-dose schedule.
(b)
Note that prefilled syringes of High-Dose Fluzone have a volume of 0.7 cc a
nd the recommended volume of administration is
0.7 ccs.
Do not withdraw more than 0.5 mL from the reconstituted product, even if some product is left in the vial.

General Best Practice Guidelines for Immunization: Vaccine Administration
103
TABLE 6-2. Needle length and injection site of IM injections for children
aged ≤18 years (by age) and adults aged ≥19 years (by sex and weight)
Age group Needle length Injection site
Children (birth-18 years)
Neonates
(a)
5/8 inch (16 mm)
(b)
Anterolateral thigh
Infants, 1-12 months 1 inch (25 mm) Anterolateral thigh
Toddlers, 1-2 years 1-1.25 inch (25-32
mm)
Anterolateral thigh
(c)
5/8
(b)
-1 inch (16-25
mm)
Deltoid muscle of arm
Children, 3-10 years 5/8
(b)
-1 inch (16-25
mm)
Deltoid muscle of arm
(c)
1-1.25 inches (25-32
mm)
Anterolateral thigh
Children, 11-18 years
5/8
(b)
-1 inch (16-25
mm)
Deltoid muscle of arm
(c)
1-1.5 inches (25-38
mm)
Anterolateral thigh
Adults (≥19 years)
Men and women, <60 kg (130 lbs) 1 inch (25 mm)
(d)
Deltoid muscle of arm
Men and women, 60-70 kg (130-
152 lbs)
1 inch (25 mm)
Men, 70-118 kg (152-260 lbs)
1-1.5 inches (25-38
mm)
Women, 70-90 kg (152-200 lbs)
Men, >118 kg (260 lbs) 1.5 inches (38 mm)
Women, >90 kg (200 lbs)
Men and women, any weight
1.5 inches (38 mm)
(e)
Anterolateral thigh

General Best Practice Guidelines for Immunization: Vaccine Administration
104
Abbreviation: IM = intramuscular.
Source: (14).
(a)
First 28 days of life.
(b)
If skin is stretched tightly and subcutaneous tissues are not bunched.
(c)
Preferred site.
(d)
Some experts recommend a 5/8-inch needle for men and women who weigh <60 kg, if used, skin must be
stretched tightly (do not bunch subcutaneous tissue).
(e)
Some experts recommend a 1-inch needle if the skin is stretched tightly and subcutaneous tissues are not
bunched.

General Best Practice Guidelines for Immunization: Vaccine Administration
105
Figure 1. Intramuscular needle insertion
Source: Adapted from California Immunization Branch.
Alternate Text: This drawing shows intramuscular needle insertion into a cross-
section of skin. The needle is inserted at a 90-degree angle and penetrates the dermis,
fatty tissue (subcutaneous), and muscle tissue.

General Best Practice Guidelines for Immunization: Vaccine Administration
106
Figure 2. Intramuscular/subcutaneous site of administration: anterolateral
thigh
Source: Adapted from Minnesota Department of Health.
Alternate Text: This drawing shows a mother holding an infant. The anterolateral
aspect of the infant’s thigh is shaded, showing the proper site for
intramuscular/subcutaneous vaccine administration.

General Best Practice Guidelines for Immunization: Vaccine Administration
107
Figure 3. Intramuscular site of administration: deltoid
Source: Adapted from Minnesota Department of Health and Immunize.org.
Alternate Text: This line drawing is a side view of an adult. The deltoid muscle of the
arm is shaded, showing the proper site for intramuscular vaccine administration.

General Best Practice Guidelines for Immunization: Vaccine Administration
108
Figure 4. Subcutaneous site of administration: triceps
Source: Adapted from the Minnesota Department of Health.
Alternate Text: This line drawing is a rear/dorsal view of an adult. The triceps muscle
of the arm is shaded, showing the proper site for subcutaneous vaccine administration.

General Best Practice Guidelines for Immunization: Vaccine Administration
109
Figure 5. Subcutaneous needle insertion
Source: Adapted from California Immunization Branch.
Alternate Text: This drawing shows subcutaneous needle insertion into a cross-
section of skin. The needle is inserted at a 45-degree angle and penetrates the dermis
and fatty tissue (subcutaneous) but not the muscle tissue.
General Best Practice Guidelines for Immunization: Vaccine Administration
110
REFERENCES
1.
Boyce JM, Pittet D. Guideline for hand hygiene in health-care settings.
Recommendations of the Healthcare Infection Control Practices
Advisory Committee and the HICPAC/SHEA/APIC/IDSA Hand Hygiene
Task Force. MMWR Recomm Rep. 2002;51(RR-16):1-45, quiz CE41-44.
2.
Occupational Health and Safety Administration. Occupational exposure
to bloodborne pathogens; needlesticks and other sharps injuries; Final
Rule (29 CFR Part 1910). Fed Regist. 2001;66(12):5318-5325.
3.
Siegel J, Rhinehart E, Jackson M, Chiarello L, the Healthcare Infection
Control Practices Advisory Committee. 2007 guideline for isolation
precautions: preventing transmission of infectious agents in healthcare
settings
Atlanta, GA: CDC;2007.
4.
Centers for Disease Control and Prevention. General recommendations
on immunization. Recommendations of the Advisory Committee on
Immunization Practices (ACIP). MMWR Recomm Rep. 1994;43(RR-1):1-
38.
5.
Centers for Disease Control and Prevention. Prevention and Control of
Seasonal Influenza with Vaccines: Recommendations of the Advisory
Committee on Immunization Practices (ACIP), 2009). MMWR
2009;58(No. RR-8):1-54
6.
Centers for Disease Control and Prevention. Recommendations for
preventing transmission of infections among chronic hemodialysis
patients. MMWR 2001;50(No. RR-5):1-46.
7.
Atkinson WL, Pickering LK, Schwartz B, Weniger BG, Iskander JK,
Watson JC. General recommendations on immunization.
Recommendations of the Advisory Committee on Immunization
Practices (ACIP) and the American Academy of Family Physicians
(AAFP). MMWR Recomm Rep. 2002;51(RR- 2):1-35.
8.
Centers for Disease Control and Prevention. Prevention of Rotavirus
Gastroenteritis Among Infants and Children: Recommendations of the
Advisory Committee on Immunization Practices (ACIP). MMWR
2009;58(No. RR-2):1-26.
General Best Practice Guidelines for Immunization: Vaccine Administration
111
9.
Centers for Disease Control and Prevention. Recommendations for using
smallpox vaccine in a prevent vaccination program: supplemental
recommendations of the Advisory Committee on Immunization
Practices (ACIP) and the Healthcare Infection Control Practices
Advisory Committee (HICPAC). MMWR 2003;52(No. RR-7):1-18).
10.
https://www.cdc.gov/mmwr/volumes/65/wr/pdfs/mm6510a2.pdfpdf
icon
(accessed February 2, 2019).
11.
Kroger AT, Sumaya CV, Pickering LK, Atkinson WL. General
recommendations on immunization: recommendations of the Advisory
Committee on Immunization Practices (ACIP). MMWR Recomm
Rep.
2011:1-60.
12.
Atkinson WL, Pickering LK, Schwartz B, Weniger BG, Iskander JK,
Watson JC. General recommendations on immunization.
Recommendations of the Advisory Committee on Immunization
Practices (ACIP) and the American Academy of Family Physicians
(AAFP). MMWR Recomm Rep. 2002;51(RR-2):1-35.
13.
Drucker E, Alcabes PG, Marx PA. The injection century: massive
unsterile injections and the emergence of human
pathogens. Lancet. 2001;358(9297):1989-1992. DOI: 10.1016/s0140-
6736(01)06967-7
14.
International Health Care Worker Safety Center. List of safety-engineered
sharp devices and other products designed to prevent occupational exposures
to bloodborne pathogensexternal icon
. 2003. Accessed 07 Feb 2017.
15.
Shaw FE, Jr., Guess HA, Roets JM, et al. Effect of anatomic injection
site, age and smoking on the immune response to hepatitis B
vaccination. Vaccine. 1989;7(5):425-430. DOI: 10.1016/0264-
410X(89)90157-6
16.
Zuckerman JN. The importance of injecting vaccines into muscle.
Different patients need different needle
sizes. BMJ. 2000;321(7271):1237-1238. DOI: 10.1136/bmj.321.7271.1237
17.
Ipp MM, Gold R, Goldbach M, et al. Adverse reactions to diphtheria,
tetanus, pertussis-polio vaccination at 18 months of age: effect of
injection site and needle length. Pediatrics. 1989;83(5):679-682.
General Best Practice Guidelines for Immunization: Vaccine Administration
112
18.
Michaels L, Poole RW. Injection granuloma of the buttock. Can Med
Assoc J.
1970;102(6):626-628.
19.
Haramati N, Lorans R, Lutwin M, Kaleya RN. Injection granulomas.
Intramuscle or intrafat? Arch Fam Med. 1994;3(2):146-148.
20.
Bergeson PS, Singer SA, Kaplan AM. Intramuscular injections in
children. Pediatrics. 1982;70(6):944-948.
21.
Poland GA, Borrud A, Jacobson RM, et al. Determination of deltoid fat
pad thickness. Implications for needle length in adult
immunization. JAMA. 1997;277(21):1709-1711. DOI:
10.1001/jama.1997.03540450065037
22.
Groswasser J, Kahn A, Bouche B, Hanquinet S, Perlmuter N, Hessel L.
Needle length and injection technique for efficient intramuscular vaccine
delivery in infants and children evaluated through an ultrasonographic
determination of subcutaneous and muscle layer
thickness. Pediatrics. 1997;100(3 Pt 1):400-403. DOI:
10.1542/peds.100.3.400
23.
Ipp M, Taddio A, Sam J, Gladbach M, Parkin PC. Vaccine-related pain:
randomised controlled trial of two injection techniques. Arch Dis
Child.
2007;92(12):1105-1108. DOI: 10.1136/adc.2007.118695
24.
CDC. Recommendation of the Immunization Practices Advisory
Committee: general recommendations on immunization MMWR Morb
Mortal Wkly Rep.
1983;32(1):1-16.
25.
Kroger AT, Atkinson WL, Marcuse EK, Pickering LK. General
recommendations on immunization: recommendations of the Advisory
Committee on Immunization Practices (ACIP). MMWR Recomm
Rep.
2006;55(RR-15):1-48.
26.
Jackson LA, Yu O, Nelson JC, et al. Injection site and risk of medically
attended local reactions to acellular pertussis
vaccine. Pediatrics. 2011;127(3):e581-587. DOI: 10.1542/peds.2010-1886
27.
Middleman AB, Anding R, Tung C. Effect of needle length when
immunizing obese adolescents with hepatitis B
vaccine. Pediatrics. 2010;125(3):e508-512. DOI: 10.1542/peds.2009-1592
General Best Practice Guidelines for Immunization: Vaccine Administration
113
28.
Fiore AE, Uyeki TM, Broder K, et al. Prevention and control of influenza
with vaccines: recommendations of the Advisory Committee on
Immunization Practices (ACIP), 2010. MMWR Recomm
Rep.
2010;59(RR-8):1-62.
29.
CDC. General recommendations on immunization: recommendations of
the Public Health Service Advisory Committee on Immunization
Practices. MMWR Morb Mortal Wkly Rep. 1976;25(44):1-3.
30.
Scheifele D, Bjornson G, Barreto L, Meekison W, Guasparini R.
Controlled trial of Haemophilus influenzae type B diphtheria toxoid
conjugate combined with diphtheria, tetanus and pertussis vaccines, in
18-month-old children, including comparison of arm versus thigh
injection. Vaccine. 1992;10(7):455-460. DOI: 10.1016/0264-
410X(92)90394-Y
31.
CDC. Diphtheria, tetanus, and pertussis: recommendations for vaccine
use and other preventive measures. Recommendations of the
Immunization Practices Advisory Committee (ACIP). MMWR Recomm
Rep.
1991;40(RR-10):1-28.
32.
Mast EE, Margolis HS, Fiore AE, et al. A comprehensive immunization
strategy to eliminate transmission of hepatitis B virus infection in the
United States: recommendations of the Advisory Committee on
Immunization Practices (ACIP) part 1: immunization of infants,
children, and adolescents. MMWR Recomm Rep. 2005;54(RR-16):1-31.
33.
Hingson RA, Davis HS, Rosen M. Historical development of jet injection
and envisioned uses in mass immunization and mass therapy based
upon 2 decades experience. Mil Med. 1963;128(6):516-524.
34.
Weniger B, Papania M. Alternative vaccine delivery methods. In: Plotkin
S, Orenstein W, Offit P, eds. Vaccines. 5th ed. China: Saunders/Elsevier;
2008:1357-1392.
35.
CDC. Hepatitis B associated with jet gun injection—California. MMWR
Morb Mortal Wkly Rep.
1986;35(23):373-376.
General Best Practice Guidelines for Immunization: Vaccine Administration
114
36.
Canter J, Mackey K, Good LS, et al. An outbreak of hepatitis B associated
with jet injections in a weight reduction clinic. Arch Intern
Med.
1990;150(9):1923-1927. DOI:
10.1001/archinte.1990.00390200105020
37.
Hoffman PN, Abuknesha RA, Andrews NJ, Samuel D, Lloyd JS. A model
to assess the infection potential of jet injectors used in mass
immunisation. Vaccine. 2001;19(28-29):4020-4027. DOI:
10.1016/S0264-410X(01)00106-2
38.
Kelly K, Loskutov A, Zehrung D, et al. Preventing contamination
between injections with multiple-use nozzle needle-free injectors: a
safety trial. Vaccine. 2008;26(10):1344-1352. DOI:
10.1016/j.vaccine.2007.12.041
39.
Simonsen L, Kane A, Lloyd J, Zaffran M, Kane M. Unsafe injections in
the developing world and transmission of bloodborne pathogens: a
review. Bull World Health Organ. 1999;77(10):789-800.
40.
Kane A, Lloyd J, Zaffran M, Simonsen L, Kane M. Transmission of
hepatitis B, hepatitis C and human immunodeficiency viruses through
unsafe injections in the developing world: model-based regional
estimates. Bull World Health Organ. 1999;77(10):801-807.
41.
Gray L, Watt L, Blass EM. Skin-to-skin contact is analgesic in healthy
newborns. Pediatrics. 2000;105(1):e14. DOI: 10.1542/peds.105.1.e14
42.
Gray L, Miller LW, Philipp BL, Blass EM. Breastfeeding is analgesic in
healthy newborns. Pediatrics. 2002;109(4):590-593. DOI:
10.1542/peds.109.4.590
43.
Harrington JW, Logan S, Harwell C, et al. Effective analgesia using
physical interventions for infant
immunizations. Pediatrics. 2012;129(5):815-822. DOI:
10.1542/peds.2011-1607
44.
Taddio A, Nulman I, Goldbach M, Ipp M, Koren G. Use of lidocaine-
prilocaine cream for vaccination pain in infants. J
Pediatr.
1994;124(4):643-648. DOI: 10.1016/S0022-3476(05)83150-6
General Best Practice Guidelines for Immunization: Vaccine Administration
115
45.
Uhari M. A eutectic mixture of lidocaine and prilocaine for alleviating
vaccination pain in infants. Pediatrics. 1993;92(5):719-721.
46.
Gupta NK, Upadhyay A, Dwivedi AK, Agarwal A, Jaiswal V, Singh A.
Randomized controlled trial of topical EMLA and vapocoolant spray for
reducing pain during wDPT vaccination. World J Pediatr.
2017;13(3):236-241.
47.
Gupta NK, Upadhyay A, Agarwal A, Goswami G, Kumar J, Sreenivas V.
Randomized controlled trial of topical EMLA and breastfeeding for
reducing pain during wDPT vaccination. Eur J Pediatr. 2013;172:1527-
1533.
48.
Reis EC, Holubkov R. Vapocoolant spray is equally effective as EMLA
cream in reducing immunization pain in school-aged
children. Pediatrics. 1997;100(6):E5. DOI: 10.1542/peds.100.6.e5
49.
Jacobsen RM, Swan A, Adegbenro A, et. al. Making vaccines more
acceptable – methods to prevent and minimize pain and other common
adverse events associated with vaccines. Vaccine. 2001;19:2418-2427.
50.
Halperin SA, McGrath P, Smith B, Houston T. Lidocaine-prilocaine
patch decreases the pain associated with the subcutaneous
administration of measles-mumps-rubella vaccine but does not
adversely affect the antibody response. J Pediatr. 2000;136(6):789-794.
DOI: 10.1016/S0022-3476(00)64169-0
51.
Frayling IM, Addison GM, Chattergee K, Meakin G.
Methaemoglobinaemia in children treated with prilocaine-lignocaine
cream. BMJ. 1990;301(6744):153-154. DOI: 10.1136/bmj.301.6744.153
52.
American Academy of Pediatrics Steering Committee on Quality
Improvement and Management, Subcommittee on Febrile Seizures.
Febrile seizures: clinical practice guideline for the long-term
management of the child with simple febrile
seizures. Pediatrics. 2008;121(6):1281-1286. DOI: 10.1542/peds.2008-
0939
General Best Practice Guidelines for Immunization: Vaccine Administration
116
53.
Cook IF, Murtagh J. Comparative immunogenicity of hepatitis B vaccine
administered into the ventrogluteal area and anterolateral thigh in
infants. J Paediatr Child Health. 2002;38(4):393-396. DOI:
10.1046/j.1440-1754.2002.00013.x
54.
Redfield RR, Innis BL, Scott RM, Cannon HG, Bancroft WH. Clinical
evaluation of low-dose intradermally administered hepatitis B virus
vaccine. A cost reduction strategy. JAMA. 1985;254(22):3203-3206.
DOI: 10.1001/jama.1985.03360220069031
55.
Coleman PJ, Shaw FE, Jr., Serovich J, Hadler SC, Margolis HS.
Intradermal hepatitis B vaccination in a large hospital employee
population. Vaccine. 1991;9(10):723-727. DOI: 10.1016/0264-
410X(91)90287-G
56.
Fishbein DB, Sawyer LA, Reid-Sanden FL, Weir EH. Administration of
human diploid-cell rabies vaccine in the gluteal area. N Engl J
Med.
1988;318(2):124-125. DOI: 10.1056/nejm198801143180219
57.
CDC. Inadvertent misadministration of meningococcal conjugate
vaccine—United States, June-August 2005. MMWR Morb Mortal Wkly
Rep.
2006;55(37):1016-1017.
58.
Ragni MV, Lusher JM, Koerper MA, Manco-Johnson M, Krause DS.
Safety and immunogenicity of subcutaneous hepatitis A vaccine in
children with haemophilia. Haemophilia. 2000;6(2):98-103. DOI:
10.1046/j.1365-2516.2000.00386.x
59.
Belshe RB, Newman FK, Cannon J, et al. Serum antibody responses after
intradermal vaccination against influenza. N Engl J
Med.
2004;351(22):2286-2294. DOI: 10.1056/NEJMoa043555

General Best Practice Guidelines for Immunization: Storage and Handling of Immunobiologics
117
7. Storage and Handling of Immunobiologics
Updates
Most of the 2011 language was removed because this content is now codified and
continually updated in the CDC’s Vaccine Storage and Handling Toolkit, available at
www.cdc.gov/vaccines/hcp/admin/storage/toolkit/index.html. This content included
Storage Units, Monitoring Storage Temperature, Vaccine Inventory, and Vaccine
Transport.
General Principles
Failure to adhere to recommended specifications for storage and handling of
immunobiologics can reduce or destroy their potency, resulting in inadequate or no
immune response in the recipient
(www.cdc.gov/vaccines/hcp/admin/storage/toolkit/index.html). Recommendations in the
product package inserts, including methods for reconstitution of the vaccine, should be
followed carefully. Maintenance of vaccine quality is the shared responsibility of all
handlers of vaccines from the time a vaccine is manufactured until administration. All
vaccines should be inspected on delivery and monitored during storage to ensure that the
recommended storage temperatures are maintained. Vaccines should continue to be
stored at recommended temperatures immediately upon receipt until use. Inadequate
vaccine storage also can result in significant costs to replace vaccine inventory
(www.cdc.gov/vaccines/hcp/admin/storage/toolkit/index.html).
Storage Temperature
Vaccines licensed for refrigerator storage should be stored at 2°C-8°C (36°F-46°F). Liquid
vaccines containing an aluminum adjuvant permanently lose potency when exposed to
freezing temperatures. Non-live vaccines that are stored in a liquid state (i.e., non-
lyophilized [freeze-dried]) but that do not contain aluminum adjuvants should also
generally be kept at refrigerator temperature, although whether or not they lose potency
when frozen is not known.

General Best Practice Guidelines for Immunization: Storage and Handling of Immunobiologics
118
Non-live lyophilized vaccines generally do not need to be frozen, but lyophilized varicella-
containing vaccines that are recommended to be stored frozen lose potency when exposed
to higher temperatures because the viruses degrade more quickly at storage temperatures
that are warmer than recommended (Table 7-1). These varicella-containing vaccines also
can be prone to losses in sterility if kept too cold, due to increased gas permeability of the
rubber vaccine vial (observed with use of dry ice at temperatures below -50°C or -58°F
[personal communication, manufacturer]).
Expiration Dates and Windows
All vaccines have an expiration date determined by the manufacturer that must be
observed. Providers should record the vaccine expiration dates and lot numbers on a stock
or inventory record for each vaccine vial when a shipment is received. When vaccines are
removed from storage, clinicians and other health-care providers should note whether an
expiration window exists for vaccine stored at room temperature or at an intermediate
temperature. For example, single-component varicella vaccine that is stored frozen must
be discarded after 72 hours of storage at refrigerator temperature. Vaccine transport
between the storage site and the administration clinic is discouraged unless the cold chain
is maintained, and vaccine transport by the patient is particularly discouraged. An
expiration window also applies to vaccines that have been reconstituted. For example,
after reconstitution, MMR vaccine should be kept at refrigerator temperature and must be
administered within 8 hours. Doses of expired vaccines that are administered
inadvertently generally should not be counted as valid and should be repeated. Non-live
vaccines should generally be repeated as soon as possible. Recombinant zoster vaccine
(RZV) should be administered 28 days after the invalid dose, to reduce the burden of
adverse reactions which occurs with this vaccine. Live vaccines should be repeated after a
28-day interval from the invalid dose to reduce the risk for interference from interferon on
the subsequent doses. For rotavirus vaccines, the repeat dose should be administered after
a 28-day interval from the invalid dose or at the maximum age for the vaccine dose
(whichever is earlier). Additional information about expiration dates is available at
https://www.cdc.gov/vaccines/hcp/admin/storage/toolkit/index.html.
General Best Practice Guidelines for Immunization: Storage and Handling of Immunobiologics
119
Response to Out-of-Range Temperature Reading
An out-of-range temperature reading should prompt immediate action. A plan should be
developed ahead of time to address various types of emergencies that might require
removal of vaccine from the original storage unit. Transfer of vaccines to a predesignated
alternative emergency storage site might be necessary if a temperature problem cannot be
resolved immediately (e.g., plugging in an unplugged unit or closing a door that has been
left open). It is critical to avoid freezing vaccine during transport (improperly packing
vaccine with ice can damage vaccines). Vaccine should be marked “do not use” and moved
to the alternate site after verifying that the alternate unit is at the proper temperature.
Determinations of vaccine viability in practice include consideration of both time and
magnitude of temperature excursions and should be made in consultation with state/local
public health departments or the vaccine manufacturer, as one or both of these groups
may have additional information based on a broad international perspective. Damage to
the immunogenicity of a vaccine exposed to temperatures outside of the recommended
range might not be apparent visually. As a general rule, vaccines that have been stored at
inappropriate temperatures should not be administered unless public health authorities or
the manufacturer determine it is safe and effective to do so. If such vaccines already have
been administered, vaccine exposed to inappropriate temperatures that is inadvertently
administered should generally be repeated. Non-live vaccines should generally be repeated
as soon as possible. Recombinant zoster vaccine (RZV) is neither live nor inactivated: the
repeat dose of RZV should be administered 28 days after the invalid dose, to reduce the
burden of adverse reactions which occurs with this vaccine. Live vaccines should be
repeated after a 28-day interval from the invalid dose to reduce the risk for interference
from interferon on the subsequent doses. For rotavirus vaccines, the repeat dose should be
administered after a 28-day interval from the invalid dose or at the maximum age for the
vaccine dose (whichever is earlier). Clinicians should consult promptly with state or local
health departments in these situations. Consultation with CDC is available when
necessary.

General Best Practice Guidelines for Immunization: Storage and Handling of Immunobiologics
120
TABLE 7-1. Vaccine storage temperature recommendations
Nonlyophilized, aluminum-adjuvanted vaccines
Vaccines Vaccine storage
temperature
Diluent storage temperature
Diphtheria-tetanus-
containing vaccines
(DT, Td) or pertussis-
containing vaccines
(DTaP, Tdap)
2°C-8°C (36°F-46°F)
Do not freeze
No diluent
(a)
HepA and HepB
2°C-8°C (36°F-46°F)
Do not freeze
No diluent
MenB
(b)
2°C-8°C (36°F-46°F)
Do not freeze
No diluent
PCV13
2°C-8°C (36°F-46°F)
Do not freeze
No diluent
PCV15
2°C-8°C (36°F-46°F)
Do not freeze
No diluent
PCV20 2°C-8°C (36°F-46°F)
Do not freeze
No diluent
HPV
(b)
2°C-8°C (36°F-46°F)
Do not freeze
No diluent
Nonlyophilized, nonaluminum-adjuvanted vaccines
Vaccines Vaccine storage
temperature
Diluent storage temperature
PRP-OMP Hib 2°C-8°C (36°F-46°F) No diluent
IPV
(b)
2°C-8°C (36°F-46°F) No diluent
MenACWY
(b),(c)
2°C-8°C (36°F-46°F) No diluent
PPSV 2°C-8°C (36°F-46°F) No diluent
IIV
(b)
2°C-8°C (36°F-46°F) No diluent
RZV
(b)
2°C-8°C (36°F-46°F)
Do not freeze
2°C-8°C (36°F-46°F)
Do not freeze

General Best Practice Guidelines for Immunization: Storage and Handling of Immunobiologics
121
Lyophilized (non-varicella) vaccines
Vaccines
Vaccine storage
temperature
Diluent storage temperature
PRP-T Hib
(b)
2°C-8°C (36°F-46°F)
(d)
2°C-8°C (35°F-46°F) Do not freeze
MMR
(b)
-50°C-8°C (-58°F-
46°F) (d)
2°C-25°C (35°F-77°F)
Can be refrigerated or stored at
room temperature
DEN4CYD 2°C-8°C (36°F-46°F)
Do not freeze
2°C-8°C (36°F-46 °F) Do not freeze
Varicella-containing vaccines
Vaccines
Vaccine storage
temperature
Diluent storage temperature
MMRV
(b)
-50°C to -15°C (-58°F-
5°F)
2°C-25°C (35°F-77°F)
Can be refrigerated or stored at
room temperature
Varicella
(b)
-50°C to -15°C (-58°F-
5°F)
2°C-25°C (35°F-77°F)
Can be refrigerated or stored at
room temperature
Noninjectable vaccines
Vaccines Vaccine storage
temperature
Diluent storage temperature
RV5 vaccine
(b)
2°C-8°C (36°F-46°F)
Do not freeze
No diluent
RV1 vaccine
(b)
2°C-8°C (36°F-46°F)
Do not freeze
The diluent may be stored at a
controlled room temperature 20°C-
25°C (68°F-77°F).
Do not freeze
LAIV
(b)
2°C-8°C (36°F-46°F) No diluent
Abbreviations: DEN4CYD = dengue vaccine; DT = diphtheria and tetanus toxoids; DTaP = diphtheria and
tetanus toxoids and acellular pertussis; HepA = hepatitis A; HepB = hepatitis B; Hib = Haemophilus influenzae
type b; HPV = human papillomavirus; IIV = inactivated influenza vaccine; IPV = inactivated poliovirus; LAIV =
live, attenuated influenza vaccine; MenACWY = quadrivalent meningococcal conjugate vaccine; MenB = Serogroup
B meningococcal vaccine; MMR = measles, mumps, and rubella; MMRV = measles, mumps, rubella, and varicella;
MPSV4 q adri alent

General Best Practice Guidelines for Immunization: Storage and Handling of Immunobiologics
122
REFERENCES
1.
Kroger A, Atkinson W, Pickering L. General immunization practices. In:
Plotkin S, Orenstein W, Offit P, eds. Vaccines. 6th ed. China: Elsevier
Saunders; 2013:88-111.
2.
CDC. Guidelines for maintaining and managing the vaccine cold chain.
MMWR Morb Mortal Wkly Rep. 2003;52(42):1023-1025.
meningococcal polysaccharide vaccine; PCV13 = pneumococcal conjugate vaccine; PPSV23= pneumococcal
polysaccharide vaccine; PRP-OMP = polyribosylribitol phosphate-meningococcal outer membrane protein
conjugate; PRP-T = polyribosylribitol phosphate polysaccharide conjugated to a tetanus toxoid; PRP-T Hib =
polyribosylphosphate tetanus-toxoid conjugate Hib vaccine; PRP-T Hib-MenCY = polyribosylphosphate-tetanus-
toxoid Hib vaccine with a bivalent Meningococcal vaccine; RV = rotavirus; RV1 = live, attenuated monovalent
rotavirus vaccine; RV5 = live, reassortment pentavalent rotavirus vaccine; Td = tetanus and diphtheria toxoids;
Tdap = tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis.
Sources: (1,2).
(a)
DTaP-Daptacel is sometimes used as a diluent for ActHib.
(b)
Protect from light.
(c)
There are 2 meningococcal conjugate vaccines; Menactra is nonlyophilized, and Menveo is lyophilized. Both
powder and diluent should be stored at 35°F-46°F.
(d)
The lyophilized pellet may be stored at freezer temperature; the reconstituted vaccine should be stored at
refrigerator temperature.

General Best Practice Guidelines for Immunization: Altered Immunocompetence
123
8. Altered Immunocompetence
Updates
This section incorporates general content from the Infectious Diseases Society of America
policy statement, 2013 IDSA Clinical Practice Guideline for Vaccination of the
Immunocompromised Host (1), to which CDC provided input in November 2011. The
evidence supporting this guidance is based on expert opinion and arrived at by consensus.
General Principles
Altered immunocompetence, a term often used synonymously with immunosuppression,
immunodeficiency, and immunocompromise, can be classified as primary or secondary.
Primary immunodeficiencies generally are inherited and include conditions defined by an
inherent absence or quantitative deficiency of cellular, humoral, or both components that
provide immunity. Examples include congenital immunodeficiency diseases such as X-
linked agammaglobulinemia, SCID, and chronic granulomatous disease. Secondary
immunodeficiency is acquired and is defined by loss or qualitative deficiency in cellular or
humoral immune components that occurs as a result of a disease process or its therapy.
Examples of secondary immunodeficiency include HIV infection, hematopoietic
malignancies, treatment with radiation, and treatment with immunosuppressive drugs.
The degree to which immunosuppressive drugs cause clinically significant
immunodeficiency generally is dose related and varies by drug. Primary and secondary
immunodeficiencies might include a combination of deficits in both cellular and humoral
immunity. Certain conditions like asplenia and chronic renal disease also can cause
altered immunocompetence.
Determination of altered immunocompetence is important to the vaccine provider
because incidence or severity of some vaccine-preventable diseases is higher in persons
with altered immunocompetence; therefore, certain vaccines (e.g., inactivated influenza
vaccine, pneumococcal vaccines) are recommended specifically for persons with these
diseases (2,3). Administration of live vaccines might need to be deferred until immune
function has improved. This is primarily a safety concern, because persons who have
altered immunocompetence and receive live vaccines might be at increased risk for an

General Best Practice Guidelines for Immunization: Altered Immunocompetence
124
adverse reaction because of uninhibited growth of the attenuated live virus or bacteria.
Vaccines might be less effective during the period of altered immunocompetence. Non-live
vaccines might best be deferred during a period of altered immunocompetence; in this
circumstance, the concern is with effectiveness and not safety. Additionally, if a non-live
vaccine is administered during the period of altered immunocompetence, it might need to
be repeated after immune function has improved.
The degree of altered immunocompetence in a patient should be determined by a
physician. The challenge for clinicians and other health-care providers is assessing the
safety and effectiveness of vaccines for conditions associated with primary or secondary
immunodeficiency, especially when new therapeutic modalities are being used and
information about the safety and effectiveness of vaccines has not been characterized fully
in persons receiving these drugs (Table 8-1). Laboratory studies can be useful for assessing
the effects of a disease or drug on the immune system. Tests useful to assess humoral
immunity include immunoglobulin (and immunoglobulin subset) levels and specific
antibody levels (e.g., tetanus and diphtheria). Tests that demonstrate the status of cellular
immunity include lymphocyte numbers (i.e., a complete blood count with differential), a
test that delineates concentrations and proportions of lymphocyte subsets (i.e., B and T
lymphocytes, CD4+ B lymphocytes versus CD8+ T lymphocytes), and tests that measure
T-cell proliferation or function in response to specific or nonspecific stimuli (e.g.,
lymphocyte proliferation assays) (4,5). The ability to characterize a drug or disease
condition as affecting cellular or humoral immunity is only the first step; using this
information to draw inferences about whether particular vaccines are indicated or whether
caution is advised with use of live or non-live vaccines is more complicated and might
require consultation with an infectious diseases or immunology specialist.
Altered Immunocompetence as an Indication to Receive a
Vaccine Outside of Routinely Recommended Age Groups
This section describes situations in which vaccines are recommended outside of the
routine-age-based recommendation because the risk for vaccine-preventable disease is
increased due to altered immunocompetence.

General Best Practice Guidelines for Immunization: Altered Immunocompetence
125
Persons with altered immunocompetence generally are recommended to receive
polysaccharide-based vaccines (PCV13, PPSV23, and Hib), on the basis of increased risk
for disease if the vaccine is withheld. For certain specific categories of altered
immunocompetence, patients are also recommended to receive polysaccharide based
vaccines (MenACWY, Hib-MenCY, and MPSV4).
Pneumococcal Vaccines
Two types of vaccine against invasive pneumococcal disease are available in the United
States: PCV13 and PPSV23. PCV13 is recommended routinely for all children beginning at
age 2 months through age 59 months and for adults aged 65 years or older. PCV13 is also
recommended for children, adolescents, and adults with conditions that place them at
high risk for invasive disease from Streptococcus pneumoniae. PCV13 is recommended for
persons aged 6-64 years who have not previously received PCV13 and have congenital
immunodeficiency disorders (including B- or T-lymphocyte deficiency, complement
deficiencies, and phagocytic disorders), anatomic or functional asplenia (including sickle
cell disease and other hemoglobinopathies), HIV infection, cochlear implant,
cerebrospinal fluid leak, chronic renal failure, nephrotic syndrome, iatrogenic
immunosuppression, or other immunocompromising conditions.
PPSV23 is licensed for use in persons aged ≥2 years and recommended routinely for
adults aged 65 years and older. PPSV23 is also recommended for persons age 2 through 64
years with congenital immunodeficiency disorders, anatomical and functional asplenia,
HIV infection, cochlear implant, cerebrospinal fluid leak, and iatrogenic
immunosuppression. Complete recommendations on use of PCV13 and PPSV23 are
available in the Recommended Immunization Schedules for Persons Aged 0 Through 18
Years and the Recommended Adult Immunization Schedule (2,6).
Meningococcal Vaccines
Three types of meningococcal vaccines are licensed in the United States: meningococcal
conjugate (MenACWY and Hib-MenCY), meningococcal polysaccharide (MPSV4), and
serogroup B meningococcal (MenB) vaccines.
General Best Practice Guidelines for Immunization: Altered Immunocompetence
126
Persons with functional or anatomic asplenia (including sickle cell disease), HIV infection
and persistent complement component deficiency (including persons taking eculizumab
[Soliris]) (7) are at increased risk for meningococcal disease and should receive
MenACWY vaccine. Persons with functional or anatomic asplenia (including sickle cell
disease) and persistent complement component deficiency (including persons taking
eculizumab [Soliris]) should receive MenB vaccine. For children 2 months through 23
months of age, an age-appropriate series of meningococcal conjugate vaccine should be
administered. If MenACWY-D (Menactra) is administered to a child with asplenia or HIV,
it should be after 2 years of age and at least 4 weeks after the completion of all PCV13
doses. A 2-dose primary series of either MenACWY-CRM (Menveo) or MenACWY-D
(Menactra) should be administered to persons 2 years of age or older with asplenia or
complement deficiency. Following the primary series of vaccine, a 3-year interval to the
next dose is recommended for persons who received their previous dose at younger than 7
years. A 5-year interval is recommended for persons who received their previous dose at
age 7 years or older. Although MPSV4 is the only meningococcal vaccine licensed for
persons older than 55 years of age, adults 56 years and older with asplenia or complement
deficiency should be vaccinated with MenACWY-CRM or MenACWY-D rather than
MPSV4 (8). Meningococcal serogroup B vaccines are licensed for persons 10-25 years of
age and are recommended for persons 10 years of age or older for persons with high-risk
conditions like functional or anatomic asplenia or persistent complement component
deficiency. There are presently no recommendations for booster doses of either MenB
vaccine (9,10). Complete recommendations for use of meningococcal vaccines are
available in the Recommended Immunization Schedules for Persons Aged 0 Through 18
Years and the Recommended Adult Schedule (2,6).
Hib Vaccines
Hib conjugate vaccines are available in single or combined antigen preparations. Hib
vaccine is recommended routinely for all children through age 59 months.

General Best Practice Guidelines for Immunization: Altered Immunocompetence
127
Children 12 through 59 months who are at high risk for invasive Hib disease (i.e.,
recipients of chemotherapy or radiation for malignant neoplasms, or those with functional
or anatomic asplenia, HIV infection, immunoglobulin deficiency, or early complement
component deficiency) and who are unvaccinated or received only one dose of Hib vaccine
before 12 months of age should receive 2 additional doses of Hib vaccine; those who
received 2 or more doses of Hib before 12 months of age should receive one additional
dose.
A child younger than 5 years of age receiving chemotherapy or radiation therapy should
have Hib doses repeated if the doses were received during therapy or within 14 days of
starting therapy; repeat doses should be started at least 3 months after completion of
therapy. Recipients of hematopoietic cell transplants should be revaccinated with 3 doses
of Hib vaccine, starting 6-12 months after successful transplant, regardless of vaccination
history or age.
Children 5-18 years of age with HIV who are unimmunized(a) should receive a dose of Hib
vaccine; Hib vaccination is not recommended in HIV-infected adults. Unimmunized(a)
asplenic patients older than 59 months of age or adults should receive a dose of Hib
vaccine. Anyone 15 months of age or older who is undergoing a splenectomy and is
unimmunized(a) should receive a dose of Hib vaccine (11). Complete recommendations for
use of Hib vaccine are available in the Recommended Immunization Schedules for
Persons Aged 0 Through 18 Years and the Recommended Adult Immunization
Schedule (2,6).
Zoster (Shingrix) Vaccine
Zoster (Shingrix) vaccine is recommended for persons 19 years old and older who have
altered immunocompetence.
Vaccination of Contacts of Persons with Altered
Immunocompetence
Household contacts and other close contacts of persons with altered immunocompetence
should receive all age- and exposure-appropriate vaccines, with the exception of smallpox
vaccine (12,13).
General Best Practice Guidelines for Immunization: Altered Immunocompetence
128
Receipt of vaccines will prevent the vaccine-preventable disease, so there can be no
potential transmission to the contact with altered immunocompetence. The live MMR,
varicella, and rotavirus vaccines should be administered to susceptible household contacts
and other close contacts of immunocompromised patients when indicated. No specific
precautions are needed unless the varicella vaccine recipient has a rash after vaccination,
in which case direct contact with susceptible household contacts with altered
immunocompetence should be avoided until the rash resolves (14,15). All members of the
household should wash their hands after changing the diaper of an infant who received
rotavirus vaccine.
This minimizes rotavirus transmission, as shedding may occur up to one month after the
last dose (16,17). Household and other close contacts of persons with altered
immunocompetence should receive annual influenza vaccination. Introduction of low
levels of vaccine viruses into the environment likely is unavoidable when administering
LAIV. LAIV vaccine viruses are cold-adapted, so they can replicate in the nose and
generate an immune response without entering the lungs (i.e., they are temperature
sensitive and replicate poorly at core body temperatures). No instances have been
reported of illness caused by attenuated vaccine virus infections among health-care
providers or immunocompromised patients. LAIV may be administered to healthy
household and other close contacts of persons with altered immunocompetence unless the
person with altered immunocompetence is in a protective environment, typically defined
as a specialized patient-care area with a positive airflow relative to the corridor, high-
efficiency particulate air filtration, and frequent air changes (3). No preference exists for
inactivated influenza vaccine use by health-care workers or other persons who have close
contact with persons with lesser degrees of immunosuppression (e.g., persons with
diabetes, persons with asthma taking high-dose corticosteroids, or persons infected with
HIV), and no preference exists for inactivated influenza vaccine use by health-care
workers or other healthy persons aged 5-49 years in close contact with all other groups at
high risk.

General Best Practice Guidelines for Immunization: Altered Immunocompetence
129
Non-live Vaccines: Safety
All non-live vaccines can be administered safely to persons with altered
immunocompetence, whether the vaccine is a killed whole-organism or a recombinant,
subunit, split-virus, toxoid, polysaccharide, or polysaccharide protein-conjugate vaccine.
Non-live Vaccines: Effectiveness
Except for inactivated influenza vaccine, vaccination during chemotherapy or radiation
therapy should be avoided if possible because antibody response might be suboptimal.
Patients vaccinated within a 14-day period before starting immunosuppressive therapy or
while receiving immunosuppressive therapy should be considered unimmunized and
should be revaccinated at least 3 months after therapy is discontinued if immune
competence has been restored. Patients who have quantitative B-cell deficiencies and are
receiving immunoglobulin therapy should not receive either non-live or live vaccines while
receiving the immunoglobulin therapy because of concerns about effectiveness of the
vaccines. Patients on chemotherapy with anti-B cell antibodies (e.g., rituximab) should
wait at least 6 months after therapy before being vaccinated with non-live vaccines. Some
experts recommended longer than 6 months for some anti-B cell antibodies. For other
forms of altered immunocompetence, if non-live vaccines are indicated, the usual
schedules are recommended. However, the effectiveness of such vaccinations might be
suboptimal (1).
Live, Attenuated Viral and Bacterial Vaccines:
Effectiveness
The same rationale regarding effectiveness that exists with non-live vaccines also exists
with live vaccines.
Live, Attenuated Viral and Bacterial Vaccines: Safety
Severe complications have followed vaccination with certain live, attenuated viral and live,
attenuated bacterial vaccines among persons with altered immunocompetence (18-26).
General Best Practice Guidelines for Immunization: Altered Immunocompetence
130
Persons with most forms of altered immunocompetence should not receive live vaccines
(MMR, varicella, MMRV, LAIV, yellow fever, Ty21a oral typhoid, BCG, smallpox, and
rotavirus). However, exceptions exist, and are discussed in this section.
Patients with any defect in phagocytic function (e.g., chronic granulomatous disease,
leukocyte adhesion deficiency, myeloperoxidase deficiency, Chediak-Higashi syndrome)
should NOT receive live bacterial vaccines. Patients with a specific type of defect in
phagocytic function—chronic granulomatous disease—should receive recommended live
attenuated viral vaccines in addition to non-live vaccines but should NOT receive live
bacterial vaccines.
Patients with defects in phagocytic function that are undefined or known to be
accompanied by defects in T-cell and natural killer cell function (e.g., leukocyte adhesion
deficiency, myeloperoxidase deficiency, Chediak-Higashi syndrome) should NOT receive
live attenuated viral or bacterial vaccines. These conditions include specific deficits in T-
cell and natural killer cell function, reducing the response to live viral vaccine antigens to
an extent not seen in chronic granulomatous disease (1). Children with deficiencies in
complement should receive recommended live, attenuated viral and live, attenuated
bacterial vaccines. Children with asplenia should not receive LAIV, but can receive
recommended live, attenuated viral and live, attenuated bacterial vaccines.
Persons with severe cell-mediated immunodeficiency should not receive live, attenuated
viral or bacterial vaccines. Patients with defects of the interferon-gamma/interleukin-12
axis should not receive live bacterial vaccines. Patients with deficiencies of interferon-
gamma or interferon-alpha should not receive live viral or live bacterial vaccine. These
defects involve a deficiency in cytokine production which affects the immune response to a
wide scope of antigens, both bacterial and viral (1). Two factors support vaccination of
HIV-exposed or HIV-infected infants with rotavirus vaccines: 1) the HIV diagnosis might
not be established in infants born to HIV-infected mothers before the age of the first
rotavirus vaccine dose (only 1.5%-3% of HIV-exposed infants in the United States will be
determined to be HIV-infected), and 2) the vaccine strains of rotavirus are considerably
attenuated. Patients taking exogenous interferon as therapy should not receive live
bacterial or live viral vaccines.
General Best Practice Guidelines for Immunization: Altered Immunocompetence
131
Children with HIV infection are at increased risk for complications from varicella and
herpes zoster infection compared with immunocompetent children (27, 28). Limited data
among HIV-infected children younger than 8 years (specifically, those individuals with
CDC class N, A, or B with age-specific CD4+ T-lymphocyte percentages of ≥15%) indicate
that single-component varicella vaccine is immunogenic, effective, and safe (14,28). Data
on use of varicella vaccine in HIV-infected adolescents and adults are lacking. However,
on the basis of expert opinion, the safety of varicella vaccine in HIV-infected persons older
than 8 years with comparable levels of immune function (CD4+T-lymphocyte count
greater than 200 cells/mm3) is likely to be similar to that of children aged younger than 8
years (14).
Varicella vaccine should be considered for persons who meet these criteria. Eligible HIV-
infected persons 12 months of age or older should receive 2 doses of single-component
varicella vaccine with a 3-month interval between doses (14,28). MMRV vaccine should
not be administered to any HIV-infected person.
Persons with HIV infection are at increased risk for severe complications if infected with
measles. No severe or unusual adverse events have been reported after measles
vaccination among HIV-infected persons who did not have evidence of severe
immunosuppression (29-32). Two doses of MMR vaccine are recommended for all HIV-
infected individuals aged ≥12 months who do not have evidence of current severe
immunosuppression (i.e., individuals aged ≤5 years must have CD4+T lymphocyte
[CD4+] percentages ≥15% for ≥6 months, and individuals aged >5 years must have
CD4+percentages ≥15% and CD4+≥200 lymphocytes/mm3 for ≥6 months) and do not
have current evidence of measles, rubella, and mumps immunity. In cases when only
CD4+cell counts or only CD4+percentages are available for those >5 years, the assessment
of severe immunosuppression can be based on the CD4+values (count or percentage) that
are available. In cases when CD4+percentages are not available for those aged ≤5 years,
the assessment of severe immunosuppression can be based on age-specific CD4+counts at
the time CD4+counts were measured; i.e., absence of severe immunosuppression is
defined as ≥6 months above age-specific CD4+count criteria: CD4+count >750
lymphocytes/mm3 while aged ≤12 months and CD4+count ≥500 lymphocytes/mm3 while
aged 1 through 5 years (33).

General Best Practice Guidelines for Immunization: Altered Immunocompetence
132
Similarly, repeat doses of MMR vaccination are recommended for individuals with
perinatal HIV infection who were vaccinated prior to establishment of effective
combination antiretroviral therapy (cART). They should receive 2 appropriately spaced
doses of MMR vaccine once effective cART has been established (individuals aged ≤5
years must have CD4+percentages ≥15% for ≥6 months; individuals aged >5 years must
have CD4+percentages ≥15% and CD4+≥200 lymphocytes/mm3 for ≥6 months) unless
they have other acceptable current evidence of measles, rubella, and mumps immunity.
HIV-infected persons who are receiving regular doses of IGIV are unlikely to respond to
varicella vaccine or MMR vaccine because of the continued presence of passively acquired
antibody.
However, because of the potential benefit, MMR and varicella vaccines should be
considered approximately 14 days before the next scheduled dose of IGIV (if not otherwise
contraindicated), although an optimal immune response might not occur depending on
the presence of neutralizing antibodies against the vaccine virus. Vaccination should be
repeated (if not otherwise contraindicated) after the recommended interval (see Table 3-6
in the Timing and Spacing of Immunobiologics of this document). In most cases, this is
after the therapy has been discontinued.
Patients with leukemia, lymphoma, or other malignancies whose disease is in remission,
who have restored immunocompetence, and whose chemotherapy has been discontinued
for at least 3 months can receive live-virus vaccines. Persons with impaired humoral
immunity (e.g., hypogammaglobulinemia or dysgammaglobulinemia) may be vaccinated
with varicella vaccine (14). However, most persons with these disorders also receive
periodic doses of IGIV. Appropriate spacing should be maintained between administration
of IGIV and varicella vaccine in an attempt to prevent an inadequate response to
vaccination caused by the presence of neutralizing antibodies from the IGIV.
Zoster incidence is higher in persons with altered immunocompetence (34). Adults with
most types of altered immunocompetence are expected to maintain residual immunity to
varicella-zoster virus because of chronic latent infection that protects against primary
varicella but provides incomplete protection against zoster. Zoster vaccine is not
recommended in persons with primary or acquired immunodeficiency (e.g., lymphoma,
leukemia, tumors involving bone marrow, and patients receiving chemotherapy) and some
General Best Practice Guidelines for Immunization: Altered Immunocompetence
133
HIV infected patients (34). Zoster vaccine may be administered to certain persons age 50
or older with altered immunocompetence, such as persons receiving low dosages of
immunosuppressive medications, those with isolated B-cell deficiencies (i.e., impaired
humoral immunity), or those with HIV infection who have CD4+ T-lymphocyte counts
>200 cells/mm3.
Recipients of Hematopoietic Cell Transplants
A hematopoietic stem cell transplant (HSCT) results in immunosuppression because of
the hematopoietic ablative therapy administered before the transplant, drugs used to
prevent or treat graft-versus-host disease, and, in some cases, from the underlying disease
process necessitating transplantation (35-37). HSCT involves ablation of the bone marrow
followed by reimplantation of the person’s own stem cells or stem cells from a donor. The
ablation caused by the HSCT will also gradually remove immune memory from previous
vaccination. Antibody titers to vaccine-preventable diseases (e.g., tetanus, poliovirus,
measles, mumps, rubella, and encapsulated bacteria) decrease 1-4 years after autologous
or allogeneic HSCT if the recipient is not revaccinated. HSCT recipients of all ages are at
increased risk for certain vaccine-preventable diseases, including diseases caused by
encapsulated bacteria (e.g., pneumococcal and Hib infections). As a result, HSCT
recipients who received vaccines prior to their HSCT should receive repeat doses routinely
after HSCT, regardless of the source of the transplanted stem cells (35-37). Revaccination
doses following HSCT are indicated with pneumococcal vaccines, DTaP vaccine, Tdap
vaccine, Hib vaccine, hepatitis A vaccine, hepatitis B vaccine, IPV, inactivated influenza
vaccines, meningococcal conjugate vaccine (for individuals 11 through 18 years or at high-
risk), serogroup B meningococcal vaccine (for individuals 16 through 23 years or at high-
risk), and human papillomavirus (HPV) vaccines (for individuals aged 9-26 years, or 27
through 45 years of age based on shared clinical decision-making) (1, 35). Recombinant
zoster vaccine may be re-administered 6-12 months after an allogeneic HSCT, and may be
re-administered 3-12 months after an autologous HSCT. For more specificity on the
precise timing of re-administration of RZV, note that this should optimally occur 2
months prior to cessation of prophylactic antiviral therapy (acyclovir, famciclovir, and
valacyclovir). Varicella and MMR vaccines may be re-administered after HSCT if 24
months have passed since HSCT, the patient does NOT have graft-vs-host disease, and is
considered immunocompetent.
General Best Practice Guidelines for Immunization: Altered Immunocompetence
134
Yellow fever vaccine, rabies vaccine, tick-borne encephalitis vaccine, and Japanese
encephalitis vaccine are not routinely administered vaccines, so their use post-HSCT will
be driven by a disease-specific risk such as exposure or travel. If someone has received
yellow fever vaccine prior to an HSCT, another dose should be re-administered post-HSCT
(38). BCG, LAIV, typhoid vaccine, and rotavirus vaccine are not recommended after
HSCT. Most non-live vaccines should be re-initiated 6 months after the HSCT (37).
Inactivated influenza vaccine should be administered beginning at least 6 months after
HSCT and annually thereafter for the life of the patient. A dose of inactivated influenza
vaccine can be given as early as 4 months after HSCT, but a second dose should be
considered in this situation (37). A second dose is recommended routinely for all children
younger than 9 years receiving influenza vaccine for the first time. Revaccination of HCST
patients with pneumococcal vaccines is recommended regardless of whether doses were
administered prior to HSCT. For those HSCT recipients that were vaccinated prior to
HSCT with PCV7, PCV13, PCV15, or PCV20, sequential administration of 4 doses of
PCV20 is recommended, beginning 3-6 months after the transplant. The first three doses
should be separated by 4 weeks and the fourth dose of PCV20 should be administered 6
months after the third dose or 1 year after the HSCT, whichever is later. As an alternative
to PCV20, three doses of PCV15 separated by 4 weeks, followed by PPSV23 one year after
the last dose of PCV15 (and at least 4 weeks after the third dose of PCV15) may be
administered. If the patient has graft-versus-host disease, substitute a fourth dose of
PCV15 for PPSV23. For doses of pneumococcal conjugate vaccine administered after
HSCT consisting of PCV13 or PCV15, these doses count as post-HSCT doses, so one always
has the option to transition FROM PCV15 to PCV20 for the post-HSCT dosing regimen. A
3-dose regimen of Hib vaccine should be administered beginning 6 months after
transplant; at least 1 month should separate the doses (37). The revaccination schedule for
pertussis-containing vaccines includes 3 doses of DTaP for patients <7 years (14). For
patients ≥7 years, providers have 3 options for revaccination: 1) 3 doses of DTaP; 2) one
dose of Tdap and 2 doses of DT; or 3) one dose of Tdap and 2 doses of Td (1).
Providers need to make a clinical judgment whether they will follow the revaccination
schedules described above in circumstances where particular vaccines were NOT
administered prior to the HSCT. In this circumstance, providers may just vaccinate
according to routine recommendations for post-HSCT vaccination. For instance, there are
General Best Practice Guidelines for Immunization: Altered Immunocompetence
135
specific recommendations for Hib and pertussis-containing vaccines. Use of the 3-dose
Hib schedule following HSCT is supported for both patients that received Hib prior to
HSCT and those who did not receive Hib prior to HSCT (6, 11). For children >6 years who
did not receive previous doses of pertussis-containing vaccine prior to the HSCT, the
preferred schedule following HSCT is a dose of Tdap followed by 2 doses of Td (personal
communication, subject matter experts).
This is identical to one of the alternative regimens for revaccination doses, described
above.
Conditions or Drugs that Might Cause
Immunodeficiencies
Asplenia and use of corticosteroids or certain drugs have the potential to be
immunosuppressive and are presumed to cause some degree of altered
immunocompetence.
Anatomic or Functional Asplenia
Persons with anatomic asplenia (e.g., surgical removal or congenital absence of the spleen)
or functional asplenia (as occurs in persons with sickle cell disease) are at increased risk
for infection by encapsulated bacteria, especially S. pneumoniae (pneumococcus), N.
meningitidis (meningococcus), and Hib (7-8, 39).
Children should receive an age-appropriate series of PCV13. Unvaccinated children 2-5
years should receive 2 doses of PCV13. Children ≥6 years should receive a dose of PCV13 if
they have not previously received a dose of PCV13. Persons aged ≥2 years should receive 2
doses of PPSV23 separated by 5 years, beginning 8 or more weeks after completing all
recommended doses of PCV13 (6-7, 40-41). In circumstances where both PCV13 and
PPSV23 are indicated, doses of PCV13 should be administered first followed by PPSV23 8
weeks after the last dose of PCV13.
Meningococcal conjugate (MenACWY) and serogroup B (MenB) vaccines are
recommended for persons with anatomic or functional asplenia (including sickle cell
disease). For children 2-23 months of age, a series of MenACWY-CRM (Menveo) or Hib-
MenCY (MenHibrix) should be administered. For persons ≥2 years of age, a 2-dose

General Best Practice Guidelines for Immunization: Altered Immunocompetence
136
primary series of either MenACWY-CRM or MenACWY-D (Menactra) should be
administered. If a person with functional or anatomic asplenia is catching up on
pneumococcal conjugate vaccine (PCV13), and the provider only carries MenACWY-D,
indicated doses of PCV13 should be completed first and MenACWY-D should be given 4
weeks after the PCV13 series is completed. Following the primary series of vaccine, a 3-
year interval to the next dose is recommended for asplenic children who received their last
previous dose at age younger than 7 years. A 5-year interval for asplenic persons is
recommended for persons who received their last previous dose at age 7 years or older.
Meningococcal B (MenB) vaccine should be administered as either a 2-dose series of
MenB-4C (Bexsero) or a 3-dose series of MenB-FHbp (Trumenba). The same vaccine
product must be used for all doses. Based on available data and expert opinion, MenB-4C
or MenB-FHbp may be administered concomitantly with MenACWY vaccines, but at a
different anatomic site, if feasible. There are presently no recommendations for booster
doses of either MenB vaccine.
Hib vaccine is recommended routinely for all children through age 59 months. Children
12-59 months with functional or anatomic asplenia and who are unvaccinated or who
received only one dose of Hib disease before 12 months of age should receive 2 doses of
Hib vaccine; those who received 2 or more doses of Hib before 12 months of age should
receive one additional dose. Unimmunized(a) asplenic patients older than 59 months of
age should receive one dose of Hib vaccine. Anyone ≥15 months of age who is undergoing
a splenectomy and is unimmunized(a) should receive one dose of Hib vaccine.
Pneumococcal, meningococcal, and Hib vaccinations should be administered at least 14
days before elective splenectomy, if possible. If the vaccinations are not administered
before surgery, they should be administered after the procedure as soon as the patient’s
condition is stable.
Corticosteroids
The amount of systemically absorbed corticosteroids and the duration of administration
needed to suppress the immune system of an otherwise immunocompetent person are not
well defined. Although the immunosuppressive effects of steroid treatment vary, the
majority of clinicians consider a dose equivalent to either ≥2 mg/kg of body weight or ≥20
General Best Practice Guidelines for Immunization: Altered Immunocompetence
137
mg/day of prednisone or equivalent for persons who weigh >10 kg when administered for
≥14 consecutive days as sufficiently immunosuppressive to raise concern about the safety
of vaccination with live-virus vaccines (37). This dosage is referred to as “high-dose
corticosteroids”. Corticosteroids used in greater than physiologic doses also can reduce the
immune response to vaccines.
Vaccination providers should defer live-virus vaccination for at least 1 month after
discontinuation of high-dose systemically absorbed corticosteroid therapy administered
for ≥14 days. Following vaccination, the decision needs to be made when to restart
immunosuppressive therapy. There are no specific recommendations about when to
restart immunosuppressive medicines. However, when initiating immunosuppressive
therapy, providers should wait 4 weeks after a live vaccine and 2 weeks after a non-live
vaccine. However, if patients require therapy for chronic inflammatory conditions, this
therapy should not be delayed because of past administration of vaccines (1).
Corticosteroid therapy usually is not a contraindication to administering live-virus vaccine
when administration is 1) short term (i.e., <14 days); 2) a low to moderate dose (i.e., <20
mg of prednisone or equivalent per day or <2mg/kg body weight per day for a young
child); 3) long-term, alternate-day treatment with short-acting preparations; 4)
maintenance physiologic doses (replacement therapy); or 5) topical (skin or eyes), inhaled,
or by intra-articular, bursal, or tendon injection (37). No evidence of an increased risk for
more severe reactions to live, attenuated viral vaccines has been reported among persons
receiving corticosteroid therapy by aerosol, and such therapy is not a reason to delay
vaccination.
Other Immunosuppressive Drugs
When feasible, clinicians should administer all indicated vaccines before initiation of
chemotherapy, before treatment with other immunosuppressive drugs, and before
radiation or splenectomy. Persons receiving chemotherapy or radiation for leukemia and
other hematopoietic malignancies, or for solid tumors, should be assumed to have altered
immunocompetence. Live, attenuated vaccines should not be administered for at least 3
months after such immunosuppressive therapy. Non-live vaccines administered during
chemotherapy should be readministered after immune competence is regained. Children

General Best Practice Guidelines for Immunization: Altered Immunocompetence
138
vaccinated before receiving chemotherapy for leukemia, lymphoma, other malignancies,
or radiation generally are thought to retain immune memory after treatment, although
revaccination with the common childhood vaccines after chemotherapy for acute
lymphoblastic leukemia might be indicated (42).
In general, revaccination of a person after chemotherapy or radiation therapy is
considered unnecessary if the previous vaccination occurred before therapy and not
during therapy, with the exception of recipients of HSCT, who should be revaccinated as
recommended previously. Determination of the level of immune memory and the need for
revaccination should be made by the treating physician.
Certain immunosuppressive medications are administered to prevent solid organ
transplant rejection. Live vaccines should be withheld for 2 months following
discontinuation of anti-rejection therapies in patients with a solid organ transplant.
Other immunosuppressive medications include human immune mediators like
interleukins and colony-stimulating factors, immune modulators, and medicines like
tumor necrosis factor-alpha inhibitors and anti-B cell agents. Non-live and live vaccines
should be administered 2 or more weeks before initiating such therapies. Live vaccines
should be withheld 3 months following such therapies, and both non-live and live vaccines
should be withheld at least 6 months following therapy with anti-B cell agents. Some
experts recommend longer than 6 months following anti-B cell agents.
Anti-B cell agents suppress antibody-producing cells for a prolonged duration, hence the
longer interval recommended before administering vaccines (17).
Zoster vaccine is an exception and should be withheld 1 month following anti-B cell
agents. After vaccination, anti-B cell agents can be resumed 1 month later.
(a)
Patients who have not received a primary series and booster dose or at least 1 dose of Hib vaccine after 14 months of age are
considered unimmunized.

General Best Practice Guidelines for Immunization: Altered Immunocompetence
139
Table 8-1. Vaccination of persons with primary and secondary immunodeficiencies
Category Specific types Contraindications
(a)
Risk-specific
recommended
vaccines
(a)
Effectiveness
and
comments
Primary: B-
lymphocyte
(humoral)
Severe antibody
deficiencies (e.g., X-
linked
agammaglobulinemia
and common variable
immunodeficiency)
OPV
(b)
Smallpox
©
LAIV
BCG
Ty21a (live typhoid)
Yellow fever
MMR
MMRV
DEN4CYD
Pneumococcal
Hib (children
12–59 months of
age)
(d)
Recombinant
zoster vaccine
(e)
The
effectiveness
of any
vaccine is
uncertain if
it depends
only on the
humoral
response
(e.g.,
PPSV23)
IGIV
interferes
with the
immune
response to
measles
vaccine and
possibly
varicella
vaccine
Less severe antibody
deficiencies (e.g.,
selective IgA
deficiency and IgG
subclass deficiency)
OPV
(b)
BCG
DEN4CYD
Yellow fever
(f)
Other live vaccines
appear to be safe
Pneumococcal
Hib (children
12–59 months of
age)
(d)
Recombinant
zoster
(e)
All vaccines
likely
effective;
immune
response
might be
attenuated
Primary: T-
lymphocyte
(cell-
mediated
Complete defects
(e.g., SCID disease,
complete DiGeorge
syndrome)
All live vaccines
(g), (h),
(i)
Pneumococcal
Hib (children
12–59 months of
age)
(d)
Vaccines
unlikely to
be effective

General Best Practice Guidelines for Immunization: Altered Immunocompetence
140
and
humoral)
Recombinant
zoster
(e)
Partial defects (e.g.,
most patients with
DiGeorge syndrome,
Wiskott-Aldrich
syndrome, ataxia-
telangiectasia)
All live
vaccines
(g),(h),(i)
Pneumococcal
Meningococcal
Hib (children 12-
59 months of
age)
(d)
Recombinant
zoster
(e)
Effectiveness
of any
vaccine
depends on
degree of
immune
suppression
Interferon-alpha
deficiencies,
interferon-gamma
deficiencies
All live vaccines
(g), (h)
Recombinant
zoster
(e)
Interferon-
gamma/interleukin-
12 axis deficiencies
Live bacterial
vaccines
(g)
Recombinant
zoster
(e)
Primary:
phagocytic
function
Chronic
granulomatous
disease
Live bacterial
vaccines
(g)
Recombinant
zoster
(e)
Live viral
vaccines
likely safe
and effective
Phagocytic
deficiencies that are
undefined or
accompanied by
defects in T-cell and
NK cell dysfunction
(such as a Chediak-
Higashi syndrome,
Leukocyte Adhesion
Deficiency [LAD],
and myeloperoxidase
deficiency)
Live
vaccines
(g),(h)
Pneumococcal
Recombinant
zoster
(e)
All non-live
vaccines safe
and likely
effective
Primary:
complement
Persistent
complement,
properdin, or Factor
B deficiency
None Pneumococcal
Meningococcal
Hib (children 12-
59 months of
age)
(d)
All routine
vaccines
likely
effective

General Best Practice Guidelines for Immunization: Altered Immunocompetence
141
Secondary HIV/AIDS OPV
(b)
Smallpox BCG
LAIV
Ty21a
MMRV
DEN4-CYD
Withhold MMR, and
varicella in severely
immunocompro
mised persons
Yellow fever vaccine
might have a
contraindication or a
precaution depending
on clinical parameters
of immune function
(j)
Pneumococcal
Hib
(d),(k)
HepB
MenACWY
Recombinant
zoster
(e)
MMR and
Varicella
vaccine in
those with
mild
immunosup
pression,
rotavirus,
and all non-
live vaccines,
including
inactivated
influenza as
per routine
vaccination
schedule,
might be
effective
(l)
Generalized
malignant neoplasm,
transplantation,
immunosuppressive
or radiation therapy
Live viral and
bacterial, depending
on immune
status
(g),(h),(m)
Pneumococcal
Hib
(m)
Recombinant
zoster
(e)
Effectiveness
of any
vaccine
depends on
degree of
immune
suppression
Taking eculizumab
(Solaris), and/or
ravulizumab
(Ultomiris)
None Meningococcal
Asplenia LAIV Pneumococcal
Meningococcal
Hib
(d),(o)
Chronic renal disease None Pneumococcal
HepB
(p)
All routine
vaccines
likely
effective

General Best Practice Guidelines for Immunization: Altered Immunocompetence
142
Abbreviations: AIDS = acquired immunodeficiency syndrome; BCG = bacille Calmette-Guérin; HepB = hepatitis B; Hib =
Haemophilus influenzae type b; HIV = human immunodeficiency virus; IG = immunoglobulin; IGIV = immune globulin
intravenous; IgA = immune globulin A; IgG = immune globulin G; LAIV = live, attenuated influenza vaccine; MenACWY =
quadrivalent meningococcal conjugate vaccine; MMR = measles, mumps, and rubella; MMRV = measles, mumps, rubella,
and varicella; OPV = oral poliovirus vaccine (live); PPSV23= pneumococcal polysaccharide vaccine; SCID = severe combined
immunodeficiency; Ty21a = live oral typhoid vaccine.
Source: (43).
(a)
Other vaccines that are universally or routinely recommended should be given if not contraindicated. An exception is
patients with B-cell deficiencies receiving immunoglobulins, who should not receive either live or non-live vaccines, due
to safety (live vaccines) and efficacy (live and non-live vaccines) concerns.
(b)
OPV is no longer available in the United States.
(c)
This table refers to contraindications for nonemergency vaccination (i.e., the ACIP recommendations); emergency
response recommendations are addressed in the clinical guidance for smallpox vaccine use in an emergency.
(d)
Children 12-59 months: if unimmunized or received zero or only 1 dose, and that dose was administered before 12 months
of age, should receive 2 Hib doses, 8 weeks apart; if received 2 or more doses before age 12 months, and none after 12
months, should receive 1 Hib dose 8 weeks after the last dose; if completed a primary series and received a booster dose at
age 12 months or older, no additional Hib doses are recommended.
(e)
19 years and older only.
(f)
There are no data to support IgA deficiency as a contraindication for yellow fever vaccine.
(g)
Live bacterial vaccines: BCG and oral Ty21a Salmonella Typhi vaccine.
(h)
Live viral vaccines: MMR, MMRV, OPV, LAIV, yellow fever, rotavirus, varicella, and vaccinia (smallpox). Nonemergency
smallpox vaccination is not recommended for children younger than 18 years or the general public.
(i)
Regarding T-lymphocyte immunodeficiency as a contraindication for rotavirus vaccine, data exist only for SCID.
(j)
Symptomatic HIV infection or CD4+ T-lymphocyte count of <200/mm
3
or <15% of total lymphocytes for children aged
<6 years is a contraindication to yellow fever vaccine administration. Asymptomatic HIV infection with CD4+ T-
lymphocyte count of 200-499/mm
3
for persons aged ≥6 years or 15%-24% of total lymphocytes for children aged <6 years
is a precaution for yellow fever vaccine administration. Details of yellow fever vaccine recommendations are available from
CDC (44)
(k)
Patients 5-18 years of age who have not received a Hib primary series and a booster dose or at least one Hib dose after 14
months of age.
(l)
HIV-infected children should be considered for varicella vaccine if CD4+ T-lymphocyte count is ≥15% and should receive
MMR vaccine if they are aged ≥12 months and do not have 1) evidence of current severe immunosuppression (i.e.,

General Best Practice Guidelines for Immunization: Altered Immunocompetence
143
individuals aged ≤5 years must have CD4+T lymphocyte [CD4] percentages ≥15% for ≥6 months; and individuals aged >5
years must have CD4+percentages ≥15% and CD4+≥200 lymphocytes/mm
3
for
≥6 months) and 2) other current evidence of measles, rubella, and mumps immunity. In cases when only CD4+cell counts
or only CD4+percentages are available for those older than age 5 years, the assessment of severe immunosuppression can
be based on the CD4+values (count or percentage) that are available. In cases when CD4+percentages are not available for
those aged ≤5 years, the assessment of severe immunosuppression can be based on age-specific CD4+counts at the time
CD4+counts were measured; i.e., absence of severe immunosuppression is defined as ≥6 months above age-specific
CD4+count criteria: CD4+count >750 lymphocytes/mm
3
while aged ≤12 months and CD4+count ≥500 lymphocytes/mm
3
while aged 1 through 5 years (33).
(m)
Withholding non-live vaccines also is recommended with some forms of immunosuppressive therapy, like anti-CD20
antibodies, induction or consolidation chemotherapy, or patients with major antibody deficiencies receiving
immunoglobulins. Inactivated influenza vaccine is an exception, but consideration should be given to repeating doses of
any non-live vaccine administered during these therapies.
(n)
Persons younger than 60 months undergoing chemotherapy or radiation therapy who have not received a Hib primary
series and a booster dose or at least one Hib dose after 14 months of age; HSCT patients of any ages, regardless of Hib
vaccine history.
(o)
Persons older than 59 months who are asplenic and persons 15 months or older who are undergoing elective
splenectomy who have not received a Hib primary series and a booster dose or at least one Hib dose after 14 months of
age.
(p)
Hepatitis B vaccine is recommended for all persons birth through 59 years of age. Hepatitis B vaccine is also recommended
for patients with chronic renal disease in all age groups (including 60 years old and older) based on the risk from dialysis-
based bloodborne transmission.
General Best Practice Guidelines for Immunization: Altered Immunocompetence
144
REFERENCES
1. Rubin L, Levin M, Ljungman P, et al. 2013 IDSA clinical practice guideline for
vaccination of the immunocompromised host. Clin Infect Dis. 2014;58(3):e44-
100. DOI: 10.1093/cid/cit684
2. Kim DK, Bridges CB, Harriman KH. Advisory committee on immunization
practices recommended immunization schedule for adults aged 19 years or
older—United States, 2015. MMWR Morb Mortal Wkly Rep. 2015;64(4):91-92.
3. Grohskopf LA, Olsen SJ, Sokolow LZ, et al. Prevention and control of seasonal
influenza with vaccines: recommendations of the Advisory Committee on
Immunization Practices (ACIP)—United States, 2014-15 influenza season.
MMWR Morb Mortal Wkly Rep. 2014;63(32):691-697.
4. Markert ML, Hummell DS, Rosenblatt HM, et al. Complete DiGeorge syndrome:
persistence of profound immunodeficiency. J Pediatr. 1998;132(1):15-21. DOI:
10.1016/S0022-3476(98)70478-0
5. Jeffrey Modell Foundation Medical Advisory Board. 10 warning signs of primary
immunodeficiency [Poster]. 2009; http:// www.info4pi.org/library/educational-
materials/10-warning-signs. Accessed 9 March, 2017.
6. Strikas RA. Advisory committee on immunization practices recommended
immunization schedules for persons aged 0 through 18 years—United States,
2015. MMWR Morb Mortal Wkly Rep. 2015;64(4):93-94.
7. Bilukha OO, Rosenstein N. Prevention and control of meningococcal disease.
Recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR Recomm Rep. 2005;54(RR-7):1-21.
8. Cohn AC, MacNeil JR, Clark TA, et al. Prevention and control of meningococcal
disease: recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR Recomm Rep. 2013;62(RR-2):1-28.
9. Folaranmi T, Rubin L, Martin SW, Patel M, MacNeil JR. Use of serogroup B
meningococcal vaccines in persons aged >/=10 years at increased risk for
serogroup B meningococcal disease: recommendations of the Advisory
Committee on Immunization Practices, 2015. MMWR Morb Mortal Wkly Rep.
2015;64(22):608-612.
General Best Practice Guidelines for Immunization: Altered Immunocompetence
145
10. MacNeil JR, Rubin L, Folaranmi T, Ortega-Sanchez IR, Patel M, Martin SW. Use
of serogroup B meningococcal vaccines in adolescents and young adults:
recommendations of the Advisory Committee on Immunization Practices, 2015.
MMWR Morb Mortal Wkly Rep. 2015;64(41):1171-1176. DOI:
10.15585/mmwr.mm6441a3
11. Briere EC, Rubin L, Moro PL, Cohn A, Clark T, Messonnier N. Prevention and
control of Haemophilus influenzae type b disease: recommendations of the
advisory committee on immunization practices (ACIP). MMWR Recomm Rep.
2014;63(RR-1):1-14.
12. Petersen BW, Harms TJ, Reynolds MG, Harrison LH. Use of vaccinia virus
smallpox vaccine in laboratory and health care personnel at risk for occupational
exposure to orthopoxviruses - recommendations of the Advisory Committee on
Immunization Practices (ACIP), 2015. MMWR Morb Mortal Wkly Rep.
2016;65(10):257-262. DOI: 10.15585/mmwr.mm6510a2
13. Petersen BW, Damon IK, Pertowski CA, et al. Clinical guidance for smallpox
vaccine use in a postevent vaccination program. MMWR Recomm Rep.
2015;64(RR-2):1-26.
14. Marin M, Guris D, Chaves SS, Schmid S, Seward JF. Prevention of varicella:
recommendations of the Advisory Committee on Immunization Practices (ACIP).
MMWR Recomm Rep. 2007;56(RR-4):1-40.
15. Grossberg R, Harpaz R, Rubtcova E, Loparev V, Seward JF, Schmid DS.
Secondary transmission of varicella vaccine virus in a chronic care facility for
children. J Pediatr. 2006;148(6):842-844. DOI: 10.1016/j.jpeds.2006.01.038
16. Cortese MM, Parashar UD. Prevention of rotavirus gastroenteritis among infants
and children: recommendations of the Advisory Committee on Immunization
Practices (ACIP). MMWR Recomm Rep. 2009;58(RR-2):1-25.
17. Anderson EJ. Rotavirus vaccines: viral shedding and risk of transmission. Lancet
Infect Dis. 2008;8(10):642-649. DOI: 10.1016/s1473-3099(08)70231-7
18. Sixbey JW. Routine immunizations and the immunosuppressed child. Adv
Pediatr Infect Dis. 1987;2:79-114.
General Best Practice Guidelines for Immunization: Altered Immunocompetence
146
19. Wright PF, Hatch MH, Kasselberg AG, Lowry SP, Wadlington WB, Karzon DT.
Vaccine-associated poliomyelitis in a child with sex-linked agammaglobulinemia.
J Pediatr. 1977;91(3):408-412. DOI: 10.1016/S0022-3476(77)81309-7
20. Wyatt HV. Poliomyelitis in hypogammaglobulinemics. J Infect Dis.
1973;128(6):802-806. DOI: 10.1093/infdis/128.6.802
21. Davis LE, Bodian D, Price D, Butler IJ, Vickers JH. Chronic progressive
poliomyelitis secondary to vaccination of an immunodeficient child. N Engl J
Med. 1977;297(5):241-245. DOI: 10.1056/nejm197708042970503
22. CDC. Disseminated Mycobacterium bovis infection from BCG vaccination of a
patient with acquired immunodeficiency syndrome. MMWR Morb Mortal Wkly
Rep. 1985;34(16):227-228.
23. Ninane J, Grymonprez A, Burtonboy G, Francois A, Cornu G. Disseminated BCG
in HIV infection. Arch Dis Child. 1988;63(10):1268-1269. DOI:
10.1136/adc.63.10.1268
24. Redfield RR, Wright DC, James WD, Jones TS, Brown C, Burke DS.
Disseminated vaccinia in a military recruit with human immunodeficiency virus
(HIV) disease. N Engl J Med. 1987;316(11):673-676. DOI:
10.1056/nejm198703123161106
25. CDC. Measles pneumonitis following measles-mumps-rubella vaccination of a
patient with HIV infection, 1993. MMWR Morb Mortal Wkly Rep.
1996;45(28):603-606.
26. Cono J, Casey CG, Bell DM. Smallpox vaccination and adverse reactions.
Guidance for clinicians. MMWR Recomm Rep. 2003;52(RR-4):1-28.
27. Derryck A, LaRussa P, Steinberg S, Capasso M, Pitt J, Gershon AA. Varicella and
zoster in children with human immunodeficiency virus infection. Pediatr Infect
Dis J. 1998;17(10):931-933. DOI: 10.1097/00006454-199810000-00023
28. Levin MJ, Gershon AA, Weinberg A, Song LY, Fentin T, Nowak B. Administration
of live varicella vaccine to HIV-infected children with current or past significant
depression of CD4(+) T cells. J Infect Dis. 2006;194(2):247-255. DOI:
10.1086/505149
General Best Practice Guidelines for Immunization: Altered Immunocompetence
147
29. Sprauer MA, Markowitz LE, Nicholson JK, et al. Response of human
immunodeficiency virus-infected adults to measles-rubella vaccination. J Acquir
Immune Defic Syndr. 1993;6(9):1013-1016.
30. McLaughlin M, Thomas P, Onorato I, et al. Live virus vaccines in human
immunodeficiency virus-infected children: a retrospective survey. Pediatrics.
1988;82(2):229-233.
31. Onorato IM, Markowitz LE, Oxtoby MJ. Childhood immunization, vaccine-
preventable diseases and infection with human immunodeficiency virus. Pediatr
Infect Dis J. 1988;7(8):588-595.
32. Palumbo P, Hoyt L, Demasio K, Oleske J, Connor E. Population-based study of
measles and measles immunization in human immunodeficiency virus-infected
children. Pediatr Infect Dis J. 1992;11(12):1008-1014.
33. McLean HQ, Fiebelkorn AP, Temte JL, Wallace GS. Prevention of measles,
rubella, congenital rubella syndrome, and mumps, 2013: summary
recommendations of the Advisory Committee on Immunization Practices (ACIP).
MMWR Recomm Rep. 2013;62(RR-4):1-34.
34. Harpaz R, Ortega-Sanchez IR, Seward JF. Prevention of herpes zoster:
recommendations of the Advisory Committee on Immunization Practices (ACIP).
MMWR Recomm Rep. 2008;57(RR-5):1-30; quiz CE32-34.
35. Tomblyn M, Chiller T, Einsele H, et al. Guidelines for preventing infectious
complications among hematopoietic cell transplantation recipients: a global
perspective. Biol Blood Marrow Transplant. 2009;15(10):1143-1238. DOI:
10.1016/j.bbmt.2009.06.019
36. Ljungman P, Cordonnier C, Einsele H, et al. Vaccination of hematopoietic cell
transplant recipients. Bone Marrow Transplant. 2009;44(8):521-526. DOI:
10.1038/bmt.2009.263
37. American Academy of Pediatrics. Immunization in special clinical circumstances.
In: Pickering L, Baker C, Kimberlin D, Long S, eds. Red Book: 2009 Report of the
Committee on Infectious Diseases. 28th ed. Elk Grove Village, IL: American
Academy of Pediatrics; 2009.
General Best Practice Guidelines for Immunization: Altered Immunocompetence
148
38. Staples JE, Bocchini JA, Jr., Rubin L, Fischer M. Yellow fever vaccine booster
doses: Recommendations of the Advisory Committee on Immunization Practices,
2015. MMWR Morb Mortal Wkly Rep. 2015;64(23):647-650.
39. CDC. Haemophilus b conjugate vaccines for prevention of Haemophilus
influenzae type b disease among infants and children two months of age and
older. Recommendations of the Immunization Practices Advisory Committee
(ACIP). MMWR Recomm Rep. 1991;40(RR-1):1-7.
40. CDC. Prevention of pneumococcal disease: recommendations of the Advisory
Committee on Immunization Practices (ACIP). MMWR Recomm Rep.
1997;46(RR-8):1-24.
41. Nuorti JP, Whitney CG. Prevention of pneumococcal disease among infants and
children - use of 13-valent pneumococcal conjugate vaccine and 23-valent
pneumococcal polysaccharide vaccine - recommendations of the Advisory
Committee on Immunization Practices (ACIP). MMWR Recomm Rep.
2010;59(RR-11):1-18.
42. Brodtman DH, Rosenthal DW, Redner A, Lanzkowsky P, Bonagura VR.
Immunodeficiency in children with acute lymphoblastic leukemia after
completion of modern aggressive chemotherapeutic regimens. J Pediatr.
2005;146(5):654-661. DOI: 10.1016/j.jpeds.2004.12.043
43. American Academy of Pediatrics. Passive immunization. In: Pickering L,
Baker C, Kimberlin D, Long S, eds. Red Book: 2012 Report of the
Committee on Infectious Diseases. 28th ed. Elk Grove Village, IL:
American Academy of Pediatrics; 2012.
44. Staples JE, Gershman M, Fischer M. Yellow fever vaccine:
recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR Recomm Rep. 2010;59(RR-7):1-27.

General Best Practice Guidelines for Immunization: Special Situations
149
9. Special Situations
Updates
Major revisions to this section of the best practices guidance include the timing of
intramuscular administration and the timing of clotting factor deficiency replacement.
Concurrent Administration of Antimicrobial Agents and
Vaccines
With a few exceptions, use of an antimicrobial agent does not interfere with the
effectiveness of vaccination. Antibacterial agents have no effect on non-live vaccines. They
also have no effect on response to live, attenuated vaccines, except live oral Ty21a typhoid
vaccine and BCG vaccines. Ty21a typhoid vaccine should not be administered to persons
receiving antimicrobial agents until 72 hours after the last dose of antimicrobial (1). If
feasible, to avoid a possible reduction in vaccine effectiveness, antibacterial drugs should
not be started or resumed until 1 week after the last dose of Ty21a. Antimicrobial or
immunosuppressive agents may interfere with the immune response to BCG and should
only be used under medical supervision (for additional information, see
www.merck.com/product/usa/pi_circulars/b/bcg/bcg_pi.pdf).
Antiviral drugs used for treatment or prophylaxis of influenza virus infections have no
effect on the response to inactivated influenza vaccine (2). However, live, attenuated
influenza vaccine should not be administered until 48 hours after cessation of therapy
with antiviral influenza drugs. If feasible, to avoid possible reduction in vaccine
effectiveness, antiviral medication should not be administered for 14 days after LAIV
administration (2). If influenza antiviral medications are administered within 2 weeks
after receipt of LAIV, the LAIV dose should be repeated 48 or more hours after the last
dose of zanamivir or oseltamivir. The LAIV dose should be repeated 5 days after peramivir
and 17 days after baloxavir. Alternatively, persons receiving antiviral drugs within the
period 2 days before to 14 days after vaccination with LAIV may be revaccinated with
another approved vaccine formulation (e.g., IIV or recombinant influenza vaccine).
Antiviral drugs active against herpesviruses (e.g., acyclovir or valacyclovir) might reduce
the efficacy of vaccines containing live, attenuated varicella zoster virus (i.e., Varivax and
General Best Practice Guidelines for Immunization: Special Situations
150
ProQuad) (3,4). These drugs should be discontinued at least 24 hours before
administration, if possible. If clinically appropriate, delay use or resumption of antiviral
therapy for 14 days after vaccination. No data exist to suggest that commonly used
antiviral drugs have an effect on rotavirus vaccine or MMR.
Administration of Live Vaccines and Tuberculin Skin
Tests (TSTs) and Interferon-gamma Release Assays
(IGRAs)
Measles illness, severe acute or chronic infections, HIV infection, and malnutrition can
create a relatively anergic state during which the TST might have a false-negative reaction
(5-7). Although live, attenuated measles vaccine theoretically can suppress TST reactivity,
the degree of suppression is likely less than that occurring from acute infection from wild-
type measles virus. Screening children for tuberculosis exposure is accomplished by
medical history rather than TST testing; universal TST screening of all children is no
longer recommended, though TST screening is sometimes indicated (e.g., for persons at
increased risk for tuberculosis exposure based on medical history, or for employees for
occupational health reasons).
In a general screening situation, a provider may find that: both TST and live vaccine are
recommended; live vaccine is recommended, and TST has already been administered
more than one day previous; or TST is indicated, and the live vaccine has already been
administered more than one day previous. TST may be administered simultaneously with
live vaccines, and this is the preferred scenario. If live vaccine is indicated, and TST has
been administered more than one day previous, the live vaccine can be administered at
any interval after the TST. If TST is recommended, and the live vaccine has already been
administered more than one day previous, providers should ensure a 28 day interval, in
other words defer the TST for 28 days after vaccination. A delay in performing the TST
removes the concern of any theoretical transient suppression of TST reactivity. Some
providers choose to perform TST screening and then delay the vaccine until the patient
returns to have the TST read. This option is less favored compared with simultaneous
TST/live vaccination, because it delays receipt of the measles-containing vaccine.
While the above rationale is based on measles suppression of TST response, because of
similar concerns about smallpox vaccine and TST suppression, a TST should not be
performed until 4 weeks after smallpox vaccination (8). No data exist regarding the
General Best Practice Guidelines for Immunization: Special Situations
151
potential degree of TST suppression that might be associated with other live, attenuated
virus vaccines (e.g., varicella or yellow fever). However, in the absence of data, following
guidelines for measles-containing vaccine when scheduling TST screening and
administering other live, attenuated virus vaccines is prudent.
If the opportunity to vaccinate might be missed, vaccination should not be delayed only
because of these theoretical considerations. TST can be repeated 4 weeks after vaccination
if it is negative and concern for TB infection persists.
Interferon gamma release assays (IGRAs), such as the QuantiFERON-TB Gold In-Tube
test and the T-Spot TB test, are blood-test alternatives to the TST for
detecting Mycobacterium tuberculosis infection. The IGRA requires only a single visit to
complete and may be less effected by previous BCG vaccination (9). The same timing
guidelines that apply to the interval between a live vaccine and TST apply to IGRA (i.e., 28
days between live vaccine and IGRA if they do not occur on the same day), because IGRA
(like TST) might be suppressed through immunologic mechanisms. The potential for a
previous TST to cause boosting of future TST results should be considered in adults who
have a negative initial TST (9). Two-step testing, in which TST is repeated in a short time
frame (e.g., 1 to 3 weeks) after an initial negative TST, can illicit boosting and identify
persons whose immune response may have waned with time since infection or BCG
vaccination. For people undergoing serial screening for infection, for instance health care
personnel who are tested yearly, differentiation of positive tests due boosting versus new
infection is important (9). The 2-step test, in which the test is given twice in a short time
frame, reduces the chance of these false negatives, which are important to identify among
adults who may have had or plan to have repeat testing anyway—for example, healthcare
personnel who are tested yearly (9). Because this test consists of 2 TSTs separated by an
interval of 1-3 weeks, there is a greater window of time during which live vaccine
replication could suppress reactivity. If a live vaccine is administered, the first dose of a 2-
step TST should be delayed for 4 weeks, and if additional doses of live vaccines are
indicated thereafter, they should be delayed until the second TST is measured.
TST or IGRA reactivity in the absence of active tuberculosis is not a contraindication to
administration of any vaccine, including live, attenuated virus vaccines.

General Best Practice Guidelines for Immunization: Special Situations
152
Note that TST screening of an asymptomatic individual is clinically different than testing a
person suspected to have active tuberculosis. If a person is suspected to have active
tuberculosis, MMR vaccine is typically not administered. Active tuberculosis should be
considered severe acute illness, and moderate or severe acute illness is a precaution for
vaccination.
Although no studies have reported on the effects of MMR vaccine on persons with active
untreated tuberculosis, a theoretical basis exists for concern that measles vaccine might
exacerbate active tuberculosis (10). As a result, before administering MMR to persons with
untreated active tuberculosis, initiating antituberculosis therapy is advisable (10).
Considering whether concurrent immunosuppression (e.g., immunosuppression caused by
HIV infection) is present before administering live, attenuated vaccines also is necessary,
because immunosuppression is a contraindication to MMR vaccine.
Vaccination of Preterm Infants
In the majority of cases, preterm infants (infants born before 37 weeks’ gestation),
regardless of birth weight, should be vaccinated at the same chronological age and
according to the same schedule and using the same precautions as for full-term infants
and children. Birth weight and size are not factors in deciding whether to vaccinate a
clinically stable preterm infant (11-15), except for hepatitis B vaccination. The full
recommended dose of each vaccine should be used. Divided or reduced doses are not
recommended.
Decreased seroconversion rates might occur among certain preterm infants (i.e., those
with low birth weights [<2,000 g]) after administration of hepatitis B vaccine at birth (16).
However, by the chronological age of 1 month, all preterm infants, regardless of initial
birth weight, are likely to respond as adequately as larger infants (17-19). Infants weighing
<2,000 g born to HBsAg-negative mothers should receive the first dose of the hepatitis B
vaccine series at chronological age 1 month or hospital discharge, if hospital discharge
occurs when the infant is younger than one month of age. Preterm low-birth-weight–
infants born to HBsAg-positive mothers should receive immunoprophylaxis with hepatitis
B vaccine within 12 hours after birth. The initial vaccine dose should not be counted
toward completion of the hepatitis B series, and 3 additional doses of hepatitis B vaccine

General Best Practice Guidelines for Immunization: Special Situations
153
should be administered, beginning when the infant is aged 1 month. For mothers with
unknown HBsAg status, hepatitis B vaccine is recommended within 12 hours of birth
regardless of low-birth-weight status.
In addition to hepatitis B vaccines, hepatitis B Immunoglobulin (HBIG) is recommended
for infants whose mothers are HBsAg positive or unknown.
If the mother is HBsAg positive, HBIG must be given within 12 hours of birth. If the
mother’s HBsAg status is unknown, providers should first attempt to determine the
mother’s status. Regardless, if the infant is preterm or low birth weight, HBIG must be
given within 12 hours of birth. If the infant is neither preterm nor low birth weight,
providers have up to 7 days from birth to determine if the mother is HBsAg negative;
because the protective efficacy of HBIG declines the longer that administration is delayed,
if results are unlikely to be known by day 7 of life, HBIG should be given no later than day
7 if not earlier. If the mother is determined to be HBsAg positive, HBIG should be
administered as soon as possible (20).
If a child aged at least 6 weeks has been in the hospital since birth, deferral of rotavirus
vaccine is recommended until the time of discharge. If an infant were to be vaccinated
with rotavirus vaccine while still needing care in the NICU or nursery, at least a theoretic
risk exists for vaccine virus being transmitted to infants in the same unit who are acutely
ill and to preterm infants who are not age-eligible for vaccine (21). The rotavirus vaccine
series should not be initiated for infants aged ≥15 weeks, 0 days.
Breastfeeding and Vaccination
With 2 exceptions, neither non-live nor live-virus vaccines administered to a lactating
woman affect the safety of breastfeeding for women or their infants. Although live viruses
in vaccines can replicate in the mother, the majority of live viruses in vaccines have been
demonstrated not to be excreted in human milk. Varicella vaccine virus has not been
found in human milk (22). Although rubella vaccine virus has been excreted in human
milk, the virus usually does not infect the infant. If infection does occur, it is well tolerated
because the virus is attenuated (23). Non-live vaccines pose no risk for mothers who are
breastfeeding or for their infants. Breastfeeding is a contraindication for smallpox
vaccination of the mother because of the theoretical risk for contact transmission from

General Best Practice Guidelines for Immunization: Special Situations
154
mother to infant. Yellow fever vaccine should be avoided in breastfeeding women, because
2 cases (one confirmed, one probable) of yellow-fever vaccine associated acute neurotropic
disease (YEL-AND) have been detected in infants whose mothers were vaccinated but
were not vaccinated themselves. In both infants, vaccine virus was recovered from the
cerebrospinal fluid of the infant, but the exact mode of transmission was not precisely
determined because vaccine virus was not recovered from breast milk (24).
However, when nursing mothers cannot avoid or postpone travel to areas endemic for
yellow fever in which risk for acquisition is high, these women should be vaccinated.
Limited data indicate that breastfeeding can enhance the response to certain vaccine
antigens (25). There are no data to suggest that passive transfer of antibodies in human
milk can affect the efficacy of live-virus vaccines. Breastfed infants should be vaccinated
according to the recommended schedule (26-28).
Vaccination During Pregnancy
No evidence exists of risk to the fetus from vaccinating pregnant women with non-live
virus or bacterial vaccines (29,30). In spite of the lack of evidence of risk, HPV vaccine, a
non-live vaccine, is not recommended during pregnancy. Live vaccines administered to a
pregnant woman pose a theoretical risk to the fetus; therefore, live, attenuated virus and
live bacterial vaccines generally are contraindicated during pregnancy. Women should
avoid conception for 4 weeks after vaccination with live vaccines. However, benefits of
vaccinating pregnant women usually outweigh potential risks when the likelihood of
disease exposure is high, when infection would pose a risk to the mother or fetus, and
when the vaccine is unlikely to cause harm. Recommendations for vaccination during
pregnancy are developed using ACIP’s Guiding Principles for Development of ACIP
Recommendations for Vaccination During Pregnancy and Breastfeeding (31).
Women who are pregnant should receive a dose of Tdap for the prevention of infant
pertussis whether or not they have previously received Tdap. Vaccination of the mother
generates antibodies that pass transplacentally to the fetus (32). Vaccination in the third
trimester optimizes the duration of this antibody protection until after birth. Additionally,
preventing pertussis in the mother reduces the risk that the infant is exposed to pertussis
General Best Practice Guidelines for Immunization: Special Situations
155
after birth (33). Health care personnel should administer Tdap during pregnancy,
preferably during the third trimester. If Tdap is not administered during pregnancy to
women who have never received it, it should be administered immediately postpartum.
Pregnant women who are not vaccinated or are only partially vaccinated against tetanus
should complete the primary series (34). Women for whom Td is indicated but who did
not complete the recommended 3-dose series during pregnancy should receive follow-up
after delivery to ensure the series is completed.
One dose of the tetanus vaccine series should be Tdap, if Tdap has not already been
received.
Pregnant and postpartum women are at higher risk for severe illness and complications
from influenza than women who are not pregnant (2, 35). Pregnant women have
protective levels of anti-influenza antibodies after vaccination (36,37). Passive transfer of
anti-influenza antibodies that might provide protection from vaccinated women to
neonates has been reported (36, 38-41). Routine vaccination with inactivated influenza
vaccine is recommended for all women who are or will be pregnant (in any trimester)
during influenza season.
IPV can be administered to pregnant women who are at risk for exposure to wild-type
poliovirus. This includes travelers to areas or countries where polio is epidemic or
endemic; members of communities or specific population groups with disease caused by
wild polioviruses; laboratory workers who handle specimens that might contain
polioviruses; healthcare personnel who have close contact with patients who might be
excreting wild polioviruses; and unvaccinated persons whose children will be receiving
oral poliovirus vaccine (42). Hepatitis A, pneumococcal polysaccharide, meningococcal
conjugate, and meningococcal polysaccharide vaccines should be considered for women at
increased risk for those infections (43-45). Pregnant women who must travel to areas
where there is a risk for acquiring yellow fever should receive yellow fever vaccine, because
the limited theoretical risk from vaccination is outweighed substantially by the risk for
yellow fever infection (24, 46). Hepatitis B vaccine is not contraindicated in pregnancy and
should be given to a pregnant woman for whom it is indicated (20, 47).
Pregnancy is a contraindication for smallpox (vaccinia) vaccine and measles-, mumps-,
General Best Practice Guidelines for Immunization: Special Situations
156
rubella-, and varicella-containing vaccines. Smallpox vaccine is the only vaccine known to
harm a fetus when administered to a pregnant woman. In addition, smallpox vaccine
should not be administered to a household contact of a pregnant woman (8). Women who
are pregnant should not have close contact with anyone who has recently (within the last
28 days) received the smallpox vaccine. Data from studies of children born to mothers
inadvertently vaccinated with rubella vaccine during pregnancy demonstrate rubella
antibody in unvaccinated infants. This could represent passive transfer of maternal
antibody or a fetal antibody response to vaccine virus infection in the fetus.
No cases of congenital rubella or varicella syndrome or abnormalities attributable to fetal
infection have been observed among infants born to susceptible women who inadvertently
received rubella or varicella vaccines during pregnancy (48-50). Because of the importance
of protecting women of childbearing age against rubella and varicella, reasonable practices
in any vaccination program include asking women if they are pregnant or might become
pregnant in the next 4 weeks; not vaccinating women who state that they are or plan to
become pregnant within that interval; explaining the theoretical risk for the fetus if MMR,
varicella, or MMRV vaccine were administered to a woman who is pregnant; and
counseling women who are vaccinated not to become pregnant during the 4 weeks after
MMR, varicella, or MMRV vaccination (10, 48-51). MMRV is an unlikely option for a
pregnant woman because the vaccine is only licensed through 12 years of age. Routine
pregnancy testing of women of childbearing age before administering a live-virus vaccine
is not recommended (3, 10). If a pregnant woman is inadvertently vaccinated or becomes
pregnant within 4 weeks after MMR or varicella vaccination, she should be counseled
about the theoretical basis of concern for the fetus; however, MMR or varicella vaccination
during pregnancy should not be considered a reason to terminate pregnancy (3, 10, 50).
Persons who receive MMR vaccine do not transmit the vaccine viruses to contacts (10).
Transmission of varicella vaccine virus to contacts is exceedingly rare (3). MMR and
varicella vaccines should be administered when indicated to children and other household
contacts of pregnant women (10). Infants living in households with pregnant women
should be vaccinated with rotavirus vaccine according to the same schedule as infants in
households without pregnant women.
Pregnant women should be evaluated for evidence of immunity to rubella and varicella

General Best Practice Guidelines for Immunization: Special Situations
157
and be tested for the presence of HBsAg during every pregnancy (10, 20, 52). Women
without evidence of immunity to rubella and varicella should be vaccinated immediately
after delivery. A second dose of varicella vaccine should be administered 4-8 weeks later. A
woman found to be HBsAg positive should be followed-up carefully to ensure that the
infant receives HBIG and begins the hepatitis B vaccine series no later than 12 hours after
birth and that the infant completes the recommended hepatitis B vaccine series on
schedule (20). No known risk exists for the fetus from passive immunization of pregnant
women with immune globulin preparations.
Persons Vaccinated Outside the United States
Clinicians have a limited ability to determine whether persons are protected on the basis of
their country of origin and their vaccination records alone. Vaccines administered outside
the United States generally can be accepted as valid if the schedule (i.e., minimum ages
and intervals) is similar to that recommended in the United States. There are exceptions
(providers should not accept documentation of these vaccines): Aimmugen, a hepatitis A
vaccine; and Twinrix Jr., a combination vaccine containing hepatitis A and hepatitis B
components (providers can accept the hepatitis B component but NOT the hepatitis A
component).
Only written documentation should be accepted as evidence of previous vaccination. There
is one exception, influenza vaccine, for which self-report can be accepted. Written records
are more likely to predict protection if the vaccines, dates of administration, intervals
between doses, and age at the time of vaccination are comparable to U.S.
recommendations. Although vaccines with inadequate potency have been produced in
other countries (53,54), the majority of vaccines used worldwide are produced with
adequate quality control standards and are potent. Medscape has a guide to pediatric
vaccines used in other countries, available at
https://emedicine.medscape.com/article/2500038-overview. Persons vaccinated outside
of the United States can enter the country through a number of different mechanisms.
Those seeking to immigrate to the United States may be vaccinated under the authority of
a civil surgeon or a panel physician. Some enter the United States as refugees and are
vaccinated under the authority of the Office of Refugee Resettlement, part of the
Administration for Children and Families, in the Department of Health and Human
Services.

General Best Practice Guidelines for Immunization: Special Situations
158
Adopted children’s birth countries often have vaccination schedules that differ from the
recommended childhood vaccination schedule in the United States. Differences in the U.S.
schedule and those used in other countries include the vaccines administered, the
recommended ages of administration, and the number and timing of doses.
Data are inconclusive regarding the extent to which an internationally adopted child’s
vaccination record reflects the child’s protection.
A child’s record might indicate administration of MMR vaccine when only single-antigen
measles vaccine was administered. A study of children adopted from orphanages in the
People’s Republic of China, Russia, and countries in Eastern Europe determined that 67%
of children with documentation of >3 doses of DTP before adoption had nonprotective
titers to these antigens (54).
In contrast, children adopted from these countries who received vaccination in the
community (not only from orphanages) and had documentation of ≥1 doses of DTP
exhibited protective titers 67% of the time (54). However, antibody testing was performed
by using a hemagglutination assay, which tends to underestimate protection and cannot
directly be compared with antibody concentration (55). Data are likely to remain limited
for areas other than the People’s Republic of China, Russia, and Eastern Europe.
Healthcare providers should ensure that household contacts of international adoptees are
vaccinated adequately, particularly for measles, hepatitis A, and hepatitis B (56).
Healthcare providers may use one of multiple approaches if the immunogenicity of
vaccines or the completeness of series administered to persons outside the United States is
in question. Repeating the vaccinations is an acceptable option that usually is safe and
prevents the need to obtain and interpret serologic tests. If avoiding unnecessary
injections is desired, judicious use of serologic testing might help determine which
vaccinations are needed. For some vaccines, the most readily available serologic tests
cannot document protection against infection. This best practices document provides
guidance on possible approaches to evaluation and revaccination for each vaccine
recommended in the United States (Table 9-1).
DTaP Vaccine
General Best Practice Guidelines for Immunization: Special Situations
159
Vaccination providers can revaccinate children younger than 7 years of age with DTaP
vaccine without regard to recorded doses; however, data indicate increased rates of local
adverse reactions after the fourth and fifth doses of DTaP (57). If a revaccination approach
is adopted and a severe local reaction occurs, serologic testing for specific IgG antibody to
tetanus and diphtheria toxins can be measured before administering additional doses.
Protective concentration
(a)
indicates that additional doses are unnecessary and subsequent
vaccination should occur as age appropriate. No established serologic correlates exist for
protection against pertussis.
For a child whose record indicates receipt of ≥3 doses of DTP or DTaP, serologic testing
for specific IgG antibody to both diphtheria and tetanus toxin before additional doses is a
reasonable approach. If a protective concentration is present, recorded doses are
considered valid, and the vaccination series should be completed as age appropriate.
An indeterminate antibody concentration might indicate immunologic memory but
waning antibody; serologic testing can be repeated after a booster dose if vaccination
providers or parents want to avoid revaccination with a complete series.
Alternately, for a child whose records indicate receipt of ≥3 doses, a single booster dose
can be administered followed by serologic testing after 1 month for specific IgG antibody
to both diphtheria and tetanus toxins. If the child has a protective concentration, the
recorded doses are considered valid, and the vaccination series should be completed as age
appropriate. Children with an indeterminate concentration after a booster dose should be
revaccinated with a complete series.
Hepatitis A Vaccine
Children aged 12-23 months without documentation of hepatitis A vaccination or serologic
evidence of immunity should be vaccinated on arrival in the United States (45). Persons
who have received 1 dose should receive the second dose if 6-18 months have passed since
the first dose was administered.
Hepatitis B Vaccine
Persons not known to be vaccinated for hepatitis B should receive an age-appropriate
series of hepatitis B vaccine. A person whose records indicate receipt of a complete series

General Best Practice Guidelines for Immunization: Special Situations
160
of vaccine is considered protected
(b)
, and additional doses are not needed if ≥1 dose was
administered at age ≥24 weeks. Persons who received their last hepatitis B vaccine dose at
an age <24 weeks should receive an additional dose at age ≥24 weeks. People who have
received no doses or a partial series of vaccine should complete the series at the
recommended intervals and ages.
All foreign-born persons and immigrants, refugees, and internationally adopted children
born in Asia, the Pacific Islands, Africa, and other regions of high or intermediate hepatitis
B endemicity should be tested for HBsAg, regardless of vaccination status (58). Those
determined to be HBsAg positive should be monitored for development of liver disease.
Household members of HBsAg-positive children or adults should be vaccinated if they are
not already immune.
Hib Vaccine
Interpretation of a serologic test to verify whether children who were vaccinated >2
months previously are protected against Hib bacteria can be difficult. Because the number
of vaccinations needed for protection decreases with age and because adverse events are
rare (59), age-appropriate vaccination should be provided. Hib vaccination is not
recommended routinely for persons aged ≥5 years (59).
Meningococcal Vaccine
Quadrivalent meningococcal conjugate vaccines are not routinely used in other countries
in adolescents (the United Kingdom is the exception). Unless patients have documented
receipt they should be considered unvaccinated and receive the age-appropriate doses.
MMR Vaccine
The simplest approach to resolving concerns about MMR vaccination is to revaccinate
with 1 or 2 doses of MMR vaccine, depending on age. Serious adverse events after MMR
vaccinations are rare (10). No evidence indicates that administering MMR vaccine
General Best Practice Guidelines for Immunization: Special Situations
161
increases the risk for adverse reactions among persons who are already immune to
measles, mumps, or rubella as a result of previous vaccination or natural disease. Doses of
measles-containing vaccine administered before the first birthday should not be counted
as part of the series (10). Alternatively, serologic testing for IgG antibody to vaccine viruses
indicated on the vaccination record can be considered. Serologic testing is widely available
for measles and rubella IgG antibody.
A person whose record indicates receipt of monovalent measles or measles-rubella vaccine
on or after the first birthday and who has protective antibody against measles and rubella
should receive 1 or 2 doses of MMR or MMRV as age appropriate to ensure protection
against mumps and varicella (and rubella if measles vaccine alone had been
administered). If a person whose record indicates receipt of MMR at age ≥12 months has a
protective concentration of antibody to measles, no additional vaccination is needed
unless a second dose is required for school entry.
Pneumococcal Vaccines
Many industrialized countries now routinely use pneumococcal vaccines. Although
recommendations for pneumococcal polysaccharide vaccine also exist in many countries,
the pneumococcal conjugate vaccine might not be routinely administered. PCV13 and
PPSV23 should be administered according to age-appropriate vaccination schedules or as
indicated by the presence of underlying medical conditions (43, 60).
Poliovirus Vaccine
The simplest approach to vaccinating with poliovirus vaccine is to revaccinate persons
aged <18 years with IPV according to the U.S. schedule. Adverse events after IPV are rare
(42). Children appropriately vaccinated with 3 doses of OPV in economically developing
countries might have suboptimal seroconversion, including to type 3 poliovirus (42).
Rotavirus Vaccine
General Best Practice Guidelines for Immunization: Special Situations
162
Rotavirus vaccination should not be initiated for infants aged ≥15 weeks, 0 days. Infants
who began the rotavirus vaccine series outside the United States but who did not complete
the series and who are still aged ≤8 months, 0 days, should follow the routine schedule
and receive doses to complete the series. If the brand of a previously administered dose is
live, reassortment pentavalent rotavirus vaccine or is unknown, a complete series of
rotavirus vaccine should be documented for series completion
(b)
. All doses should be
administered by age 8 months, 0 days.
Td and Tdap Vaccines
Children aged ≥7 years who are not considered fully vaccinated for pertussis should
receive Tdap vaccine. “Fully vaccinated” means at least 5 doses of DTaP before the seventh
birthday or at least 4 doses of DTaP before the seventh birthday if the fourth dose is given
after the fourth birthday. One dose of Tdap is recommended after the seventh birthday. If
additional doses of vaccine are needed, Td should be administered as age appropriate.
Varicella Vaccine
Varicella vaccine is not available in most countries. A person who lacks evidence of
varicella immunity should be vaccinated as age appropriate (3, 59).
Zoster Vaccine
In the United States, zoster vaccination is recommended for all persons aged ≥50 years
who have no contraindications, including persons who report a previous episode of zoster
or who have chronic medical conditions. For persons who do not have documentation of
receipt of zoster vaccine, the vaccine should be offered at the patient’s first clinical
encounter with the healthcare provider. Two doses of vaccine are recommended 2-6
months apart, each dose a 0.5-mL intramuscular dose. Zoster vaccination is not indicated
to treat acute zoster, to prevent persons with acute zoster from developing postherpetic
neuralgia, or to treat ongoing postherpetic neuralgia. Patients do not need to be asked
about their history of varicella or to have serologic testing conducted to determine zoster
immunity prior to administration of zoster vaccine.
General Best Practice Guidelines for Immunization: Special Situations
163
Vaccinating Persons with Increased Bleeding Risk
Providers often avoid giving intramuscular injections or choose alternative routes for
persons with bleeding disorders because of the risk for hematoma formation after
injections. In one study, hepatitis B vaccine was administered intramuscularly to 153
persons with hemophilia. The vaccination was administered with a 23-gauge or smaller
caliber needle, followed by application of steady pressure to the site for 1-2 minutes. The
vaccinations resulted in a low (4%) bruising rate, and no patients required factor
supplementation (61). Whether antigens that produce more local reactions (e.g., pertussis)
would produce an equally low rate of bruising is unknown.
When hepatitis B or any other intramuscularly administered vaccine is indicated for a
patient with a bleeding disorder, the vaccine should be administered intramuscularly if a
physician familiar with the patient’s bleeding risk determines that the vaccine can be
administered by this route with reasonable safety. If the patient receives antihemophilia or
similar therapy, intramuscularly administered vaccinations can be scheduled shortly after
such therapy is administered.
A fine-gauge needle (23-gauge or smaller caliber) should be used for the vaccination,
followed by firm pressure on the site, without rubbing, for at least 2 minutes. The patient
or family should be given information on the risk for hematoma from the injection.
Patients receiving anticoagulation therapy presumably have the same bleeding risk as
patients with clotting factor disorders and should follow the same guidelines for
intramuscular administration. If possible, vaccination could be scheduled prior to the use
of these medications, so that the patients’ risk of bleeding is not increased by their
therapeutic action.
(a) Enzyme immunoassay tests are available. Physicians should contact the laboratory performing the test for interpretive
standards and limitations. Protective concentrations for antibody to diphtheria and tetanus toxins are defined as >0.1 IU/mL.
(b) A complete series may consist of two or three doses, depending on the brand of vaccine used in the country of origin.
General Best Practice Guidelines for Immunization: Special Situations
164
TABLE 9-1. Approaches to evaluation and vaccination of persons
vaccinated outside the United States who have no (or questionable)
vaccination records
Vaccine Recommended
approach
Alternative approach
(a)
DTaP Revaccination with DTaP,
with serologic testing for
specific IgG antibody to
tetanus and diphtheria
toxins in the event of a
severe local reaction
Persons whose records indicate
receipt of ≥3 doses: serologic testing
for specific IgG antibody to
diphtheria and tetanus toxins before
administering additional doses (see
text), or administer a single booster
dose of DTaP, followed by
serological testing after 1 month for
specific IgG antibody to diphtheria
and tetanus toxins with
revaccination as appropriate (see
text)
HepA Age-appropriate
revaccination
Serologic testing for IgG antibodies
to hepatitis A
HepB Age-appropriate
revaccination and serologic
testing for HBsAg
(b)
—
Hib Age-appropriate
revaccination
—
HPV Age-appropriate
revaccination
—
Meningococcal
conjugate
(MenACWY)
Age-appropriate
revaccination
—
MMR Revaccination with MMR
Serologic testing for IgG antibodies
to measles, mumps, and rubella
Pneumococcal
conjugate (or in
some cases, both
PCV13 and
PPSV23)
Age-appropriate
revaccination
—

General Best Practice Guidelines for Immunization: Special Situations
165
Poliovirus
Revaccination with
inactivated poliovirus
vaccine
—
Rotavirus Age-appropriate
revaccination
—
Tdap Age-appropriate
revaccination of persons
who are candidates for
Tdap vaccine
—
Varicella Age-appropriate
revaccination of persons
who lack evidence of
varicella immunity
—
Zoster
Age-appropriate
revaccination
—
Abbreviations: DTaP = diphtheria and tetanus toxoids and acellular pertussis; HBsAg = hepatitis B surface
antigen; HepA = hepatitis A; HepB = hepatitis B; Hib = Haemophilus influenzae type b; HPV = human
papillomavirus; IgG = immune globulin G; MMR = measles, mumps, and rubella; PCV13 = pneumococcal conjugate
vaccine; PPSV23= pneumococcal polysaccharide vaccine; Tdap = tetanus toxoid, reduced diphtheria toxoid, and
acellular pertussis.
(a)
There is a recommended approach for all vaccines and an alternative approach for some vaccines.
(b)
In rare instances, hepatitis B vaccine can give a false-positive HBsAg result up to 18 days after vaccination;
therefore, blood should be drawn to test for HBsAg before vaccinating (20).
General Best Practice Guidelines for Immunization: Special Situations
166
REFERENCES
1.
Jackson BR, Iqbal S, Mahon B. Updated recommendations for the use of
typhoid vaccine—Advisory Committee on Immunization Practices, United
States, 2015. MMWR Morb Mortal Wkly Rep. 2015;64(11):305-308.
2.
Grohskopf LA, Shay DK, Shimabukuro TT, et al. Prevention and control of
seasonal influenza with vaccines. Recommendations of the Advisory
Committee on Immunization Practices—United States, 2013-2014. MMWR
Recomm Rep. 2013;62(RR-7):1-43.
3.
Marin M, Guris D, Chaves SS, Schmid S, Seward JF. Prevention of varicella:
recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR Recomm Rep. 2007;56(RR-4):1-40.
4.
Harpaz R, Ortega-Sanchez IR, Seward JF. Prevention of herpes zoster:
recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR Recomm Rep. 2008;57(RR-5):1-30; quiz CE32-34.
5.
Starr S, Berkovich S. Effects of measles, gamma-globulin-modified measles
and vaccine measles on the tuberculin test. N Engl J Med. 1964;270:386-391.
DOI: 10.1056/nejm196402202700802
6.
Brickman HF, Beaudry PH, Marks MI. The timing of tuberculin tests in
relation to immunization with live viral vaccines. Pediatrics. 1975;55(3):392-
396.
7.
Berkovich S, Starr S. Effects of live type 1 poliovirus vaccine and other viruses
on the tuberculin test. N Engl J Med. 1966;274(2):67-72. DOI:
10.1056/nejm196601132740203
8.
Wharton M, Strikas RA, Harpaz R, et al. Recommendations for using
smallpox vaccine in a pre-event vaccination program. Supplemental
recommendations of the Advisory Committee on Immunization Practices
(ACIP) and the Healthcare Infection Control Practices Advisory Committee
(HICPAC). MMWR Recomm Rep. 2003;52(RR-7):1-16.
General Best Practice Guidelines for Immunization: Special Situations
167
9.
Mazurek GH, Jereb J, Vernon A, LoBue P, Goldberg S, Castro K.
Updated guidelines for using interferon gamma release assays to
detect Mycobacterium tuberculosis infection - United States, 2010.
MMWR Recomm Rep. 2010;59(RR-5):1-25.
10.
Watson JC, Hadler SC, Dykewicz CA, Reef S, Phillips L. Measles, mumps, and
rubella—vaccine use and strategies for elimination of measles, rubella, and
congenital rubella syndrome and control of mumps: recommendations of the
Advisory Committee on Immunization Practices (ACIP). MMWR Recomm
Rep. 1998;47(RR-8):1-57.
11.
Bernbaum JC, Daft A, Anolik R, et al. Response of preterm infants to
diphtheria-tetanus-pertussis immunizations. J Pediatr. 1985;107(2):184-188.
DOI: 10.1016/S0022-3476(85)80122-0
12.
Koblin BA, Townsend TR, Munoz A, Onorato I, Wilson M, Polk BF. Response
of preterm infants to diphtheria-tetanus-pertussis vaccine. Pediatr Infect Dis
J. 1988;7(10):704-711.
13.
Smolen P, Bland R, Heiligenstein E, Lawless MR, Dillard R, Abramson J.
Antibody response to oral polio vaccine in premature infants. J Pediatr.
1983;103(6):917-919. DOI: 10.1016/S0022-3476(83)80714-8
14.
Omenaca F, Garcia-Sicilia J, Garcia-Corbeira P, et al. Response of preterm
newborns to immunization with a hexavalent diphtheria-tetanus-acellular
pertussis-hepatitis B virus-inactivated polio and Haemophilus influenzae type
b vaccine: first experiences and solutions to a serious and sensitive issue.
Pediatrics. 2005;116(6):1292-1298. DOI: 10.1542/peds.2004-2336
15.
Shinefield H, Black S, Ray P, Fireman B, Schwalbe J, Lewis E. Efficacy,
immunogenicity and safety of heptavalent pneumococcal conjugate vaccine in
low birth weight and preterm infants. Pediatr Infect Dis J. 2002;21(3):182-
186. DOI: 10.1097/00006454-200203000-00003
16.
Lau YL, Tam AY, Ng KW, et al. Response of preterm infants to hepatitis B
vaccine. J Pediatr. 1992;121(6):962-965. DOI: 10.1016/S0022-
3476(05)80352-X
17.
Patel DM, Butler J, Feldman S, Graves GR, Rhodes PG. Immunogenicity of
hepatitis B vaccine in healthy very low birth weight infants. J Pediatr.
1997;131(4):641-643. DOI: 10.1016/S0022-3476(97)70078-7
General Best Practice Guidelines for Immunization: Special Situations
168
18.
Kim SC, Chung EK, Hodinka RL, et al. Immunogenicity of hepatitis B vaccine
in preterm infants. Pediatrics. 1997;99(4):534-536. DOI:
10.1542/peds.99.4.534
19.
Losonsky GA, Wasserman SS, Stephens I, et al. Hepatitis B vaccination of
premature infants: a reassessment of current recommendations for delayed
immunization. Pediatrics. 1999;103(2):E14. DOI: 10.1542/peds.103.2.e14
20.
Mast EE, Margolis HS, Fiore AE, et al. A comprehensive immunization
strategy to eliminate transmission of hepatitis B virus infection in the United
States: recommendations of the Advisory Committee on Immunization
Practices (ACIP) part 1: immunization of infants, children, and adolescents.
MMWR Recomm Rep. 2005;54(RR-16):1-31.
21.
Cortese MM, Parashar UD. Prevention of rotavirus gastroenteritis among
infants and children: recommendations of the Advisory Committee on
Immunization Practices (ACIP). MMWR Recomm Rep. 2009;58(RR-2):1-25.
22.
Bohlke K, Galil K, Jackson LA, et al. Postpartum varicella vaccination: is the
vaccine virus excreted in breast milk? Obstet Gynecol. 2003;102(5 Pt 1):970-
977. DOI: 10.1016/S0029-7844(03)00860-3
23.
Krogh V, Duffy LC, Wong D, Rosenband M, Riddlesberger KR, Ogra PL.
Postpartum immunization with rubella virus vaccine and antibody response
in breast-feeding infants. J Lab Clin Med. 1989;113(6):695-699.
24.
Staples JE, Gershman M, Fischer M. Yellow fever vaccine: recommendations
of the Advisory Committee on Immunization Practices (ACIP). MMWR
Recomm Rep. 2010;59(RR-7):1-27.
25.
Pickering LK, Granoff DM, Erickson JR, et al. Modulation of the immune
system by human milk and infant formula containing nucleotides. Pediatrics.
1998;101(2):242-249. DOI: 10.1542/peds.101.2.242
26.
Kim-Farley R, Brink E, Orenstein W, Bart K. Vaccination and breast feeding.
JAMA. 1982;248(19):2451-2452. DOI: 10.1001/jama.1982.03330190021019
27.
Patriarca PA, Wright PF, John TJ. Factors affecting the immunogenicity of
oral poliovirus vaccine in developing countries: review. Rev Infect Dis.
1991;13(5):926-939.
General Best Practice Guidelines for Immunization: Special Situations
169
28.
Hahn-Zoric M, Fulconis F, Minoli I, et al. Antibody responses to parenteral
and oral vaccines are impaired by conventional and low protein formulas as
compared to breast-feeding. Acta Paediatr Scand. 1990;79(12):1137-1142.
DOI: 10.1111/j.1651-2227.1990.tb11401.x
29.
Koren G, Pastuszak A, Ito S. Drugs in pregnancy. N Engl J Med.
1998;338(16):1128-1137. DOI: 10.1056/nejm199804163381607
30.
Grabenstein JD. Vaccines and antibodies in relation to pregnancy and
lactation. Hospital Pharmacy. 1999;34:949-960.
31.
CDC. Guiding principles for development of ACIP recommendations for
vaccination during pregnancy and breastfeeding. MMWR Morb Mortal Wkly
Rep. 2008;57(21):580.
32.
Munoz FM, Bond NH, Maccato M, et al. Safety and immunogenicity of
tetanus diphtheria and acellular pertussis (Tdap) immunization during
pregnancy in mothers and infants: a randomized clinical trial. JAMA.
2014;311(17):1760-1769. DOI: 10.1001/jama.2014.3633
33.
Wendelboe AM, Njamkepo E, Bourillon A, et al. Transmission of Bordetella
pertussis to young infants. Pediatr Infect Dis J. 2007;26(4):293-299. DOI:
10.1097/01.inf.0000258699.64164.6d
34.
Kretsinger K, Broder KR, Cortese MM, et al. Preventing tetanus, diphtheria,
and pertussis among adults: use of tetanus toxoid, reduced diphtheria toxoid
and acellular pertussis vaccine recommendations of the Advisory Committee
on Immunization Practices (ACIP) and recommendation of ACIP, supported
by the Healthcare Infection Control Practices Advisory Committee (HICPAC),
for use of Tdap among health-care personnel. MMWR Recomm Rep.
2006;55(RR-17):1-37.
35.
Neuzil KM, Reed GW, Mitchel EF, Simonsen L, Griffin MR. Impact of
influenza on acute cardiopulmonary hospitalizations in pregnant women. Am
J Epidemiol. 1998;148(11):1094-1102. DOI:
10.1093/oxfordjournals.aje.a009587
36.
Sumaya CV, Gibbs RS. Immunization of pregnant women with influenza
A/New Jersey/76 virus vaccine: reactogenicity and immunogenicity in mother
and infant. J Infect Dis. 1979;140(2):141-146. DOI: 10.1093/infdis/140.2.141
General Best Practice Guidelines for Immunization: Special Situations
170
37.
Munoz FM, Greisinger AJ, Wehmanen OA, et al. Safety of influenza
vaccination during pregnancy. Am J Obstet Gynecol. 2005;192(4):1098-1106.
DOI: 10.1016/j.ajog.2004.12.019
38.
Englund JA, Mbawuike IN, Hammill H, Holleman MC, Baxter BD, Glezen
WP. Maternal immunization with influenza or tetanus toxoid vaccine for
passive antibody protection in young infants. J Infect Dis. 1993;168(3):647-
656. DOI: 10.1093/infdis/168.3.647
39.
Puck JM, Glezen WP, Frank AL, Six HR. Protection of infants from infection
with influenza A virus by transplacentally acquired antibody. J Infect Dis.
1980;142(6):844-849. DOI: 10.1093/infdis/142.6.844
40.
Reuman PD, Ayoub EM, Small PA. Effect of passive maternal antibody on
influenza illness in children: a prospective study of influenza A in mother-
infant pairs. Pediatr Infect Dis J. 1987;6(4):398-403. DOI:
10.1097/00006454-198704000-00011
41.
Steinhoff MC, Omer SB, Roy E, et al. Influenza immunization in pregnancy—
antibody responses in mothers and infants. N Engl J Med.
2010;362(17):1644-1646. DOI: 10.1056/NEJMc0912599
42.
Prevots DR, Burr RK, Sutter RW, Murphy TV. Poliomyelitis prevention in the
United States. Updated recommendations of the Advisory Committee on
Immunization Practices (ACIP). MMWR Recomm Rep. 2000;49(RR-5):1-22;
quiz CE21-27.
43.
CDC. Prevention of pneumococcal disease: recommendations of the Advisory
Committee on Immunization Practices (ACIP). MMWR Recomm Rep.
1997;46(RR-8):1-24.
44.
Bilukha OO, Rosenstein N. Prevention and control of meningococcal disease.
Recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR Recomm Rep. 2005;54(RR-7):1-21.
45.
Fiore AE, Wasley A, Bell BP. Prevention of hepatitis A through active or
passive immunization: recommendations of the Advisory Committee on
Immunization Practices (ACIP). MMWR Recomm Rep. 2006;55(RR-7):1-23.
General Best Practice Guidelines for Immunization: Special Situations
171
46.
Tsai TF, Paul R, Lynberg MC, Letson GW. Congenital yellow fever virus
infection after immunization in pregnancy. J Infect Dis. 1993;168(6):1520-
1523. DOI: 10.1093/infdis/168.6.1520
47.
Mast EE, Weinbaum CM, Fiore AE, et al. A comprehensive immunization
strategy to eliminate transmission of hepatitis B virus infection in the United
States: recommendations of the Advisory Committee on Immunization
Practices (ACIP) Part II: immunization of adults. MMWR Recomm Rep.
2006;55(RR-16):1-33; quiz CE31-34.
48.
Wilson E, Goss MA, Marin M, et al. Varicella vaccine exposure during
pregnancy: data from 10 years of the pregnancy registry. J Infect Dis.
2008;197 Suppl 2:S178-184. DOI: 10.1086/522136
49.
CDC. Revised ACIP recommendation for avoiding pregnancy after receiving a
rubella-containing vaccine. MMWR Morb Mortal Wkly Rep.
2001;50(49):1117.
50.
CDC. Rubella vaccination during pregnancy—United States, 1971-1988.
MMWR Morb Mortal Wkly Rep. 1989;38(17):289-293.
51.
Black NA, Parsons A, Kurtz JB, McWhinney N, Lacey A, Mayon-White RT.
Post-partum rubella immunisation: a controlled trial of two vaccines. Lancet.
1983;2(8357):990-992. DOI: 10.1016/S0140-6736(83)90979-0
52.
CDC. Control and prevention of rubella: evaluation and management of
suspected outbreaks, rubella in pregnant women, and surveillance for
congenital rubella syndrome. MMWR Recomm Rep. 2001;50(RR-12):1-23.
53.
Murray TS, Groth ME, Weitzman C, Cappello M. Epidemiology and
management of infectious diseases in international adoptees. Clin Microbiol
Rev. 2005;18(3):510-520. DOI: 10.1128/cmr.18.3.510-520.2005
54.
Hostetter MK. Infectious diseases in internationally adopted children:
findings in children from China, Russia, and eastern Europe. Adv Pediatr
Infect Dis. 1999;14:147-161.
55.
Kriz B, Burian V, Sladky K, Burianova B, Mottlova O, Roth Z. Comparison of
titration results of diphtheric antitoxic antibodies obtained by means of
Jensen's method and the methods of tissue cultures and haemagglutination. J
Hyg Epidemiol Microbiol Immunol. 1978;22(4):485-493.
General Best Practice Guidelines for Immunization: Special Situations
172
56.
CDC. Updated recommendations from the Advisory Committee on
Immunization Practices (ACIP) for use of hepatitis A vaccine in close contacts
of newly arriving international adoptees. MMWR Morb Mortal Wkly Rep.
2009;58(36):1006-1007.
57.
CDC. Use of diphtheria toxoid-tetanus toxoid-acellular pertussis vaccine as a
five-dose series. Supplemental recommendations of the Advisory Committee
on Immunization Practices (ACIP). MMWR Recomm Rep. 2000;49(RR-13):1-
8.
58.
Weinbaum CM, Williams I, Mast EE, et al. Recommendations for
identification and public health management of persons with chronic
hepatitis B virus infection. MMWR Recomm Rep. 2008;57(RR-8):1-20.
59.
CDC. Recommended immunization schedules for persons aged 0 through 18
years—United States, 2011. MMWR Morb Mortal Wkly Rep. 2011;60(5):1-4.
60.
Nuorti JP, Whitney CG. Prevention of pneumococcal disease among infants
and children - use of 13-valent pneumococcal conjugate vaccine and 23-valent
pneumococcal polysaccharide vaccine - recommendations of the Advisory
Committee on Immunization Practices (ACIP). MMWR Recomm Rep.
2010;59(RR-11):1-18.
61.
Evans DI, Shaw A. Safety of intramuscular injection of hepatitis B vaccine in
haemophiliacs. BMJ. 1990;300(6741):1694-1695. DOI:
10.1136/bmj.300.6741.1694-a

General Best Practice Guidelines for Immunization: Vaccination Records
173
10. Vaccination Records
Records of Healthcare Providers
Appropriate and timely vaccination documentation helps ensure not only that persons in
need of recommended vaccine doses receive them but also that adequately vaccinated
patients do not receive excess doses. Curtailing the number of excess doses administered
to patients controls costs incurred by patients, providers, insurers, vaccination programs,
and other stakeholders. In addition, avoidance of excess doses of vaccines should decrease
the number of adverse reactions to vaccines. Health care providers who administer
vaccines covered by the National Vaccine Injury Compensation Program (VICP) are
required under the National Childhood Vaccine Injury Act (1) to ensure that the
permanent medical record of the recipient (or a permanent office log or file) indicates the
date the vaccine was administered, the vaccine manufacturer, the vaccine lot number, and
the name, address, and title of the person administering the vaccine. This Act applies to
any vaccine for which there is a routine recommendation for childhood vaccination, even
if many or most doses of the vaccine are administered to adults (e.g., influenza vaccine). In
addition, the provider is required to record the edition date of the VIS distributed and the
date those materials were provided. The Act considers a health-care provider to be any
licensed health care professional, organization, or institution, whether private or public
(including federal, state, and local departments and agencies), under whose authority a
specified vaccine is administered. This information should be kept for all vaccines, not just
for those required by the Act. Providers and staff members also should systematically
update patients’ permanent medical records to reflect any documented episodes of
adverse events after vaccination and any serologic test results related to vaccine-
preventable diseases (e.g., those for rubella screening and anti-HBs).
Personal Records of Patients
Official childhood vaccination records have been adopted by every state and territory and
the District of Columbia to encourage uniformity of records and to facilitate assessment of
vaccination status by schools and child-care centers.

General Best Practice Guidelines for Immunization: Vaccination Records
174
The records also are key tools in vaccination education programs aimed at increasing
parental and patient awareness of the need for vaccines. This record can exist in electronic
file format or in hardcopy format. A permanent vaccination record should be established
for each newborn infant and maintained by the parent or guardian. The parent or
guardian should be educated about the importance of keeping the record up-to-date and
instructed to keep the record indefinitely. These records should be distributed to new
parents and/or guardians before discharge from the hospital or birthing center. Using
vaccination records for adolescents and adults also is encouraged. Standardized adult
vaccination records are available at http://www.immunize.org.
Immunization Information Systems (IISs)
IISs (formerly referred to as immunization registries) are confidential, population-based,
computerized information systems that collect and consolidate vaccination data from
multiple health care providers within a geographic area. IISs are a critical tool that can
increase and sustain vaccination coverage by consolidating vaccination records from
multiple providers, generating reminder and recall vaccination notices for each person,
and providing official vaccination forms and vaccination coverage assessments (2).
Providers should be aware of state and/or regional IISs and requirements for reporting.
Changing vaccination providers during the course of an individual’s vaccination series is
common in the United States. In addition to changes in providers, the vaccination records
of persons who have changed vaccination providers often are unavailable or incomplete or
might not have been entered into an IIS (2). Missing or inaccurate information regarding
vaccines received previously might preclude accurate determination of which vaccines are
indicated at the time of a visit, resulting in administration of extra doses.
A fully operational IIS also can prevent duplicate vaccinations, forecast when the next
dose is due, limit missed appointments, allow recall for those who missed appointments,
determine when vaccines need to be repeated (the technical IIS term for this is
“evaluation”), reduce vaccine waste, and reduce staff time required to produce or locate
vaccination records or certificates.
General Best Practice Guidelines for Immunization: Vaccination Records
175
Most IISs have additional capabilities, such as measurement of vaccination update and
coverage, aid in tracking vaccine inventory and placing vaccine orders, recall of vaccine by
lot number, maintenance of lifetime vaccination histories, and interoperability with other
health information systems. The National Vaccine Advisory Committee recommends that
vaccination providers participate in these systems when possible. Electronic health
records should maintain interoperability with IISs as part of an effort to improve the
quality of care, reduce health disparities, engage patients and families in their health,
improve the coordination of care, improve population health, and ensure adequate privacy
and security protection for personal health information (see
www.cdc.gov/ehrmeaningfuluse/introduction.html)
One of the national Healthy People objectives for 2020 is 95% participation of children
aged <6 years in a fully operational population-based IIS (objective 20.1) (3,4).
Participating in an IIS means having two or more vaccinations recorded in the IIS. 2012
IIS data indicate that approximately 86% of children aged <6 years with two or more
vaccinations were participating in IISs (4,5).
The National Vaccine Advisory Committee recommends that public health departments
work toward including adults in all state IISs, reduce barriers to including adult
vaccination records in IISs, and ensure that IISs meet new standards of EHR
interoperability to track and maintain adult vaccination records (6).
Nationally, 57.8 million U.S. adults aged 19 years or older participated in an IIS in 2012
(4). This number reflects adults who may have had childhood vaccines entered during
childhood and now have aged to adults. In 2013, 32% of U.S. adults had a record in the IIS
and at least one vaccination administered during adulthood.
General Best Practice Guidelines for Immunization: Vaccination Records
176
REFERENCES
1.
National Childhood Vaccine Injury Act, 42 U.S.C. Sect. 300aa-1 to 300aa-34
(1986).
2.
CDC. Immunization information systems progress—United States, 2006. MMWR
Morb Mortal Wkly Rep. 2008;57(11):289-291.
3.
US Department of Health and Human Services. Immunization and infectious
diseases. Healthy people 2010. Vol 1 (conference edition). 1st ed. Washington,
DC: US Government Printing Office; 2000.
4.
CDC. Progress in immunization information systems - United States, 2012.
MMWR Morb Mortal Wkly Rep. 2013;62(49):1005-1008.
5.
Mangione-Smith R, DeCristofaro AH, Setodji CM, et al. The quality of
ambulatory care delivered to children in the United States. N Engl J Med.
2007;357(15):1515-1523. DOI: 10.1056/NEJMsa064637
6.
National Vaccine Advisory Committee. Recommendations from the National
Vaccine Advisory committee: standards for adult immunization practice. Public
Health Rep. 2014;129(2):115-123.

General Best Practice Guidelines for Immunization: Vaccination Programs
177
11. Vaccination Programs
Updates
The major revision to this section is the addition of language related to Affordable Care
Act (1) coverage of adult vaccination.
General Principles
Universal vaccination is a critical part of quality health care and should be accomplished
through routine and catch-up vaccination provided in physicians’ offices, public health
clinics, and other appropriate settings. In the United States, vaccination is considered
primarily the responsibility of individual health care providers and health care systems
serving patients.
Certain programs and other efforts attempt to ensure all patients receive the full schedule
of appropriate vaccinations by removing barriers posed by access to immunizations, cost,
or other factors. Such efforts may include school-located clinics, school-based health
centers, back-to-school immunization clinics, public health clinics for schoolchildren,
periodic influenza vaccination clinics, public health nurse tracking of childhood
immunizations, and government-sponsored financing of vaccines through the Vaccines for
Children and Section 317 program (www.cdc.gov/vaccines/hcp/admin/vfc.html).
In the United States, vaccination programs have eliminated many vaccine-preventable
diseases and markedly reduced the incidence of others (2). Because infants and young
children were the principal recipients of most vaccines developed during the twentieth
century (e.g., poliovirus vaccine), many persons in the United States might believe that
vaccinations are solely for the young; however, vaccinations are recommended for persons
of all ages (3,4). Improved vaccination coverage can result in additional reductions in the
incidence of vaccine-preventable diseases that affect persons throughout the life span, and
decrease associated morbidity and mortality.

General Best Practice Guidelines for Immunization: Vaccination Programs
178
Vaccination of Children and Adolescents
Physicians and other pediatric vaccination providers should adhere to the standards for
child and adolescent vaccination practices (5). These standards are published by the
National Vaccine Advisory Committee and define appropriate vaccination practices for
both public and private sectors. The standards provide guidance on practices that
eliminate barriers to vaccination, including eliminating unnecessary prerequisites for
receiving vaccinations, eliminating missed opportunities to vaccinate, improving
procedures to assess vaccination needs, enhancing knowledge about vaccinations among
parents and providers, and improving management and reporting of adverse events. In
addition, the standards address the importance of recall and reminder systems and using
assessments to monitor clinic or office vaccination coverage levels. Health-care providers
should simultaneously administer as many vaccine doses as possible as indicated on
the Recommended Immunization Schedules for Persons Aged 0 Through 18 Years (3).
While rates of childhood vaccination are generally higher than rates of adult vaccination,
for some doses coverage rates are still low, like the birth dose of hepatitis B vaccine.
Community health-care providers, as well as state and local public health vaccination
programs, should coordinate with partners to identify and maximize outreach to
populations at risk for undervaccination and vaccine-preventable diseases. For example,
the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) is a
categorical federal grant program administered by the U.S. Department of Agriculture
through state health departments. The program provides supplemental foods, health-care
referrals, and nutrition education to low-income pregnant, breastfeeding, or postpartum
women, as well as to infants and children aged
<5 years. Between 8.5 and 8.9 million people participated in this program in 2013
(www.fns.usda.gov/pd/wic-program). In collaboration, WIC and state vaccination
programs assess regularly the vaccination coverage levels of WIC participants and develop
new strategies and aggressive outreach procedures in sites with coverage levels
<90%.

General Best Practice Guidelines for Immunization: Vaccination Programs
179
Vaccination programs and private providers are encouraged to refer eligible
children to obtain WIC nutritional services, at www.fns.usda.gov/wic/immunization-
screening-and-referral-wic (6).
Adolescent-Specific Issues
Vaccinations are recommended throughout life, including during adolescence. The age
range for adolescence is defined as 11-21 years by many professional associations,
including the American Academy of Pediatrics and the American Medical Association
(7,8). Definitions of these age cutoffs differ depending on the source of the definition and
the source’s purpose for creating a definition. Vaccination of adolescents is critical for
preventing diseases for which adolescents are at particularly high or increasing risk, such
as meningococcal disease and human papillomavirus infection. Three vaccines
recommended for adolescents have been licensed since 2005: MenACWY and Tdap were
licensed in 2005, and HPV was licensed in 2006. A second dose of varicella vaccine is
recommended for persons who received 1 dose of varicella vaccine after age 12 months. In
addition, annual seasonal influenza vaccination is recommended for persons aged >6
months who have no contraindications. To ensure vaccine coverage, clinicians and other
health-care providers who treat adolescents must review vaccination history on every
occasion that an adolescent has an office visit.
National goals for vaccination coverage for adolescents aged 13-15 years were included in
Healthy People 2020, at www.healthypeople.gov/2020/topics-
objectives/topic/immunization-and-infectious-diseases. Targets of 80% coverage were
specified for one dose of Tdap, one dose of meningococcal conjugate vaccine, and 3 doses
of HPV vaccine. Results of the published 2014 National Immunization Survey—Teen
indicate that coverage rates for 13-17 years olds is 87.6% for one dose of Tdap and 79.3%
for one dose of meningococcal vaccine. Coverage rates for 13-17 years olds for HPV vaccine
are considerably lower—39.7% for females and 21.6% for males (9,10).
Ensuring adolescents receive routine and catch-up vaccination and achieving high levels
of vaccination coverage present challenges. In general, adolescents do not visit health care
providers frequently. Health care providers should promote annual preventive visits (11),
including one specifically for adolescents aged 11 and 12 years.

General Best Practice Guidelines for Immunization: Vaccination Programs
180
The annual visits should be used as opportunities to provide routinely recommended
vaccine doses, additional catch-up doses needed for lapsed vaccine series, vaccines
recommended for high-risk groups, additional doses that might have been recently
recommended, and other recommended health-care services. Additional strategies include
adolescent immunizations at community-based venues such as pharmacies and schools.
All vaccine doses should be administered according to ACIP vaccine-specific statements
and with the most recent schedules for both routine and catch-up vaccination. Before
leaving any visit for medical care, adolescents should be encouraged to schedule return
visits for any additional vaccine doses needed. During visits that occur outside of influenza
season, providers should discuss and recommend seasonal influenza vaccination and
make explicit plans for vaccination, including timing and anticipated setting (e.g., health
care provider’s office, school, or pharmacy). Catch-up vaccination with multidose
adolescent vaccines generally can occur according to the routine dosing schedule for these
vaccines, although in some circumstances the clinician or health care provider might use
minimum intervals for vaccine doses. These circumstances include an outbreak that
increases risk for disease or the likelihood that doses will be missed in the future (e.g.,
because of transportation challenges). Because of lack of efficacy data for HPV vaccine
administration using minimum intervals, providers are encouraged, when possible, to use
routine dosing intervals for females aged 11-26 years and males aged 11-21 years who have
not yet received 3 HPV vaccine doses as recommended (3,4).
One of the challenges of adolescent vaccination is ensuring that current, complete
vaccination histories are available. Insurers, covered services, or reimbursement levels can
change, and these changes might affect reimbursement for vaccine doses and vaccination
services directly while also causing disruptions in an adolescent’s access to vaccination
providers or venues. In circumstances in which a vaccination record is unavailable,
vaccination providers should attempt to obtain this information from various sources
(e.g., parent, previous providers, or school records). More detail about how to obtain these
records is available from CDC at www.cdc.gov/vaccines/hcp/admin/immuniz-
records.html. With the exception of influenza and pneumococcal polysaccharide vaccines,
if documentation of a vaccine dose is not available, the adolescent should be considered
unvaccinated for that dose.

General Best Practice Guidelines for Immunization: Vaccination Programs
181
Regardless of the venue in which an adolescent receives a dose of vaccine, that vaccine
dose should be documented in the patient’s chart or in an office log, and the information
should be entered into an IIS. The adolescent also should be provided with a record that
documents the vaccination history.
Adult Vaccination
In 2013, the National Vaccine Advisory Committee published updated standards for adult
vaccination (12). These standards are targeted to distinct groups involved in adult
vaccination, including immunizing providers, non-immunizing providers, professional
health-care organizations, and public health departments. All health-care providers,
whether they provide immunizations or not, should incorporate immunization needs
assessment into every clinical encounter, strongly recommend needed vaccine(s) and
either administer vaccine(s) or refer patients to a provider who can immunize, stay up-to-
date on, and educate patients about vaccine recommendations, implement systems to
incorporate vaccine assessment into routine clinical care, and understand how to access
immunization information systems (i.e., immunization registries) (12).
Vaccination rates in adults are considered suboptimal (13,14). New Healthy People
2020
goals include specific subsets of adults, including institutionalized adults aged ≥18
years (for pneumococcal vaccines) and noninstitutionalized adults at high risk aged >18
years (for pneumococcal vaccines) (9).
The most substantial barrier to vaccination coverage is lack of knowledge about these
vaccines among adult patients and adult providers. Other barriers are cost (incomplete
Medicare coverage for recommended vaccines) (15) and the lack of financing mechanisms
for newly licensed and recommended vaccines. Effective for private health insurance plans
drafted or updated after September 2010, coverage for all immunizations that are included
on the immunization schedule(s) must be covered without deductibles or co-pays, when
delivered by an in-network provider. For this reason, cost may present less of a barrier to
adult vaccination as time passes.

General Best Practice Guidelines for Immunization: Vaccination Programs
182
A common challenge for health-care providers is vaccinating adults with unknown
vaccination records. In general (except for influenza and pneumococcal polysaccharide
vaccines), adults should receive a vaccine dose if the dose is recommended and no record
of previous administration exists. If an adult has a record of military service and does not
have records available, providers can assume that the person has received all vaccines
recommended by the military at the time of service entry. Serologic testing might be
helpful in clarifying immune status if questions remain, because at different times and
depending on military assignments, there might be inter-service and individual
differences.
Evidence-based Interventions to Increase Vaccination
Coverage
The independent, nonfederal Task Force on Community Preventive Services, whose
membership is appointed by CDC, provides public health decision-makers with
recommendations on population-based interventions to promote health and prevent
disease, injury, disability, and premature death. The recommendations are based on
systematic reviews of the scientific literature about effectiveness and cost-effectiveness of
these interventions. In addition, the task force identifies critical information about the
other effects of these interventions, the applicability to specific populations and settings,
and the potential barriers to implementation. Additional information, including updates
of published reviews, is available from The Community Guide at
http://www.thecommunityguide.org.
Beginning in 1996, the task force systematically reviewed published evidence on the
effectiveness and cost-effectiveness of population-based interventions to increase coverage
of vaccines recommended for routine use among children, adolescents, and adults. A total
of 197 articles were identified that evaluated a relevant intervention, met inclusion
criteria, and were published during 1980-1997. Reviews of 17 specific interventions were
published in 1999 (13,14,16,17). Using the results of their review, the task force made
recommendations about the use of these interventions (15). Several interventions were
identified and recommended on the basis of published evidence. Follow-up reviews were
published in 2000, and a review of interventions to improve the coverage of adults at high
risk was conducted in 2005 (15,17).

General Best Practice Guidelines for Immunization: Vaccination Programs
183
The interventions and the recommendations are summarized in this section of this report
(Table 11-1). Interventions designated for adults younger than 65 years at high risk for
influenza, invasive pneumococcal disease, and hepatitis B, include provider reminder
systems or a menu of items (combinations of strategies ) (Table 11-2). In 1997, the task
force categorized vaccination requirements for child care, school, and college as a
recommended strategy (14).
A 2008 update of the original task force systematic review of the evidence on the
effectiveness of provider assessment and feedback for increasing coverage rates found that
this strategy remains an effective intervention (18). This later update reviewed 19 new
studies published during 1997-2007. The updated review supports the original task force
recommendation for use of assessment and feedback based on strong evidence of
effectiveness. The task force reviewed studies of assessment and feedback as a strategy
that were conducted in a range of settings, including private practice, managed care,
public health, community health settings, and academic centers. Studies have assessed the
effectiveness of this intervention to improve coverage with MMR, DTP, DTaP, Hib,
influenza, pneumococcal, and Td vaccines (16). The most updated information on this
review is available at www.thecommunityguide.org/findings/vaccination-programs-
provider-assessment-and-feedback. As recognized by the task force, routine assessment
and feedback of vaccination rates obtained at the provider site is one of the most effective
strategies for achieving high, sustainable vaccine coverage. Since 1995, all states receiving
federal funds for vaccination programs have been required to conduct annual assessments
of vaccination rates both in public health clinics and in private provider offices. Primarily
to aid local and state health departments in their efforts to conduct assessments and assist
providers, CDC has developed numerous software applications to measure vaccination
rates in provider practices.
Other General Programmatic Issues
Programmatic challenges, evolving issues, and effective interventions related to adult and
adolescent vaccination programs have been described by other advisory groups and expert
groups. Additional evidence-based approaches are being developed for certain issues (e.g.,
settings for adolescent vaccination delivery) through ongoing research and evaluation.

General Best Practice Guidelines for Immunization: Vaccination Programs
184
Among current programmatic challenges, vaccine financing is especially difficult because
certain problems and solutions differ markedly from one state to another. Practitioners
interested in beginning or continuing to provide vaccinations to patients are encouraged
to consult with local and state public health vaccination programs to learn about publicly
funded programs that might be available in their areas for patients who need vaccination
but have insufficient health insurance coverage and no financial resources. If not already
participating, providers who care for adolescents and children aged <19 years should
enroll in the Vaccines for Children Program
(www.cdc.gov/vaccines/hcp/admin/vfc.html). Through this program’s provision of ACIP-
recommended, federally purchased vaccines, participating providers are able to fully
vaccinate eligible children whose parents might not otherwise be able to afford the
vaccinations. Interested providers are encouraged to work with insurers, state and
specialty-specific medical organizations, vaccine manufacturers, and other stakeholders to
address financial barriers to achieving high vaccination coverage. With availability of safe
and effective vaccines for 18 vaccine-preventable diseases, the capacity for realizing the
potential benefits of these products in the United States depends on reaching children,
adolescents, and adults through dedicated, knowledgeable vaccination providers and
efficient, strong vaccination programs at local, state, and federal levels.

General Best Practice Guidelines for Immunization: Vaccination Programs
185
TABLE 11-1. Recommendations regarding interventions to improve
coverage of vaccines recommended for routine use among children,
adolescents, and adults
Intervention Recommendation
Increase community demand for vaccination
Client reminder or recall systems Recommended
Requirements for entry to schools, child- care
facilities, and colleges
Recommended
Community education alone Insufficient evidence
Community-based interventions implemented in
combination
Recommended
Clinic-based education Insufficient evidence
Patient or family incentives Recommended
Patient or family monetary sanctions Insufficient evidence
Client-held medical records Insufficient evidence
Enhance access to vaccination services
Reducing out-of-pocket costs Recommended
Enhancing access through the U.S. Department of
Agriculture’s Women, Infants, and Children (WIC)
program
Recommended
Home visits, outreach, and case management
targeted to particularly hard-to-reach populations
to increase vaccination rates
Recommended
Enhancing access at schools Recommended
Expanding access in health-care settings Recommended as part of
multicomponent interventions
only
Enhancing access at organized child care centers Recommended
Focus on providers
Provider reminder or recall systems Recommended

General Best Practice Guidelines for Immunization: Vaccination Programs
186
Provider assessment and feedback Recommended
Standing orders Recommended
Provider education alone Insufficient evidence
Health-care systems-based interventions
integrated in combination
Recommended
Immunization information systems Recommended
Source: www.thecommunityguide.org/topic/vaccination.

General Best Practice Guidelines for Immunization: Vaccination Programs
187
TABLE 11-2. Strategies to improve influenza, pneumococcal
polysaccharide, and hepatitis B vaccine coverage among high-risk adults
younger than 65 years
One or both of these interventions to
improve access to vaccination services
1. Expanded access in health-care settings
2. Reducing client out-of-pocket costs
PLUS:
One or more of these provider or system
based interventions
1. Standing orders
2. Provider reminder systems
3. Provider assessment or feedback
AND/OR:
One or both of these interventions to
increase client demand for vaccination
services
1. Client reminder systems
2. Client education
Source (15)
General Best Practice Guidelines for Immunization: Vaccination Programs
188
REFERENCES
1.
The Patient Protection and Affordable Care Act, Pub. L. No. 111-148 (2010).
2.
Roush SW, Murphy TV. Historical comparisons of morbidity and mortality for
vaccine-preventable diseases in the United States. JAMA. 2007;298(18):2155-
2163. DOI: 10.1001/jama.298.18.2155
3.
Strikas RA. Advisory committee on immunization practices recommended
immunization schedules for persons aged 0 through 18 years—United States,
2015. MMWR Morb Mortal Wkly Rep. 2015;64(4):93-94.
4.
Kim DK, Bridges CB, Harriman KH. Advisory committee on immunization
practices recommended immunization schedule for adults aged 19 years or
older—United States, 2015. MMWR Morb Mortal Wkly Rep. 2015;64(4):91-
92.
5.
National Vaccine Advisory Committee. Standards for child and adolescent
immunization practices. Pediatrics. 2003;112(4):958-963.
6.
CDC. Recommendations of the Advisory Committee on Immunization
Practices: programmatic strategies to increase vaccination coverage by age 2
years—linkage of vaccination and WIC services. MMWR Morb Mortal Wkly
Rep. 1996;45(10):217-218.
7.
Hagan J, Shaw J, Duncan P, eds. Bright futures: guidelines for health
supervision on infants, children and adolescents. 3rd ed. Elk Grove Village,
IL: American Academy of Pediatrics; 2008.
8.
CDC. Immunization of adolescents. Recommendations of the Advisory
Committee on Immunization Practices, the American Academy of Pediatrics,
the American Academy of Family Physicians, and the American Medical
Association. MMWR Recomm Rep. 1996;45(RR-13):1-16.
9.
US Department of Health and Human Services. Immunization and infectious
diseases. Healthy People 2020 website.
https://www.healthypeople.gov/2020/topics-objectives/topic/immunization-
and-infectious-diseases. Accessed 09 March, 2017.
General Best Practice Guidelines for Immunization: Vaccination Programs
189
10.
CDC. U.S. vaccination coverage reported via NIS-Teen. 2016; https://
www.cdc.gov/vaccines/imz-managers/coverage/teenvaxview/index.html.
Accessed 09 March 2017.
11.
Mangione-Smith R, DeCristofaro AH, Setodji CM, et al. The quality of
ambulatory care delivered to children in the United States. N Engl J Med.
2007;357(15):1515-1523. DOI: 10.1056/NEJMsa064637
12.
National Vaccine Advisory Committee. Recommendations from the National
Vaccine Advisory committee: standards for adult immunization practice.
Public Health Rep. 2014;129(2):115-123.
13.
Shefer A, Briss P, Rodewald L, et al. Improving immunization coverage rates:
an evidence-based review of the literature. Epidemiol Rev. 1999;21(1):96-142.
14.
CDC. Vaccine-preventable diseases: improving vaccination coverage in
children, adolescents, and adults. A report on recommendations from the
Task Force on Community Preventive Services. MMWR Recomm Rep.
1999;48(RR-8):1-15.
15.
Ndiaye SM, Hopkins DP, Shefer AM, et al. Interventions to improve influenza,
pneumococcal polysaccharide, and hepatitis B vaccination coverage among
high-risk adults: a systematic review. Am J Prev Med. 2005;28(5 Suppl):248-
279. DOI: 10.1016/j.amepre.2005.02.016
16.
Briss PA, Rodewald LE, Hinman AR, et al. Reviews of evidence regarding
interventions to improve vaccination coverage in children, adolescents, and
adults. The Task Force on Community Preventive Services. Am J Prev Med.
2000;18(1 Suppl):97-140. DOI: 10.1016/S0749-3797(99)00118-X
17.
Task Force on Community Preventive S. Recommendations regarding
interventions to improve vaccination coverage in children, adolescents, and
adults12. Am J Prev Med. 2000;18(1, Supplement 1):92-96. DOI:
10.1016/S0749-3797(99)00121-X
18.
CDC. Vaccination programs: provider assessment and feedback. The
Community Guide website. 2015;
https://www.thecommunityguide.org/findings/vaccination-programs-
provider-assessment-and-feedback. Accessed 09 March 2017.

General Best Practice Guidelines for Immunization: Vaccination Programs
190
12. Vaccine Information Sources
In addition to these general recommendations, the following sources contain specific and
updated vaccine information.
• CDC-INFO Contact Center
The CDC-INFO contact center is supported by CDC and provides public health-
related information, including vaccination information, for health-care providers
and the public, 24 hours a day, 7 days a week. To contact CDC-INFO online at any
time, visit https://wwwn.cdc.gov/dcs/ContactUs/Form. To contact CDC-INFO by
telephone, call between 8 am to 8 pm Eastern Time Monday through Friday at
[English and Spanish]: 800-232-4636; telephone [TTY]: 800-232-6348.
• CDC’s National Center for Immunization and Respiratory Diseases
CDC’s National Center for Immunization and Respiratory Diseases website
provides direct access to ACIP’s best practices for vaccination guidance, vaccination
schedules, automated child schedulers, an adult immunization scheduler, vaccine
safety information, publications, provider education and training, and links to other
vaccination-related websites (www.cdc.gov/vaccines/hcp/admin/immuniz-
records.html).
• Morbidity and Mortality Weekly Report (MMWR)
Some ACIP guidance regarding vaccine use, statements of vaccine policy as they are
developed, and reports of specific disease activity are published by CDC in the
MMWR series and can be found at www.cdc.gov/vaccines/hcp/acip-
recs/index.html. Electronic subscriptions are free
(www.cdc.gov/mmwr/index.html). Subscriptions to print versions also are
available from the Superintendent of Documents, U.S. Government Printing Office,
Washington, D.C. 20402-9235 (telephone: 202-512-1800).
• American Academy of Family Physicians (AAFP)
Information from the professional organization of family physicians is available at
www.aafp.org/home.html.

General Best Practice Guidelines for Immunization: Vaccination Programs
191
• American Academy of Pediatrics (AAP)
Every 3 years, AAP issues the Red Book: Report of the Committee on Infectious
Diseases, which contains a composite summary of AAP and ACIP recommendations
concerning infectious diseases and vaccinations for infants, children, and
adolescents (telephone: 888-227-1770; website: www.aap.org/en-
us/Pages/Default.aspx).
• American College of Physicians (ACP)
Produced by faculty of ACP’s Quality Improvement Programs and members of the
ACP Adult Immunization Advisory Board, the ACP Guide to Adult Immunization
helps internists develop systematic processes for incorporating immunization in
their day-to- day practice (see https://www.acponline.org/).
• American Congress of Obstetricians and Gynecologists (ACOG)
The American Congress of Obstetricians and Gynecologists (ACOG), formerly the
American College of Obstetricians and Gynecologists, is a professional association
of physicians specializing in obstetrics and gynecology in the United States.
Information about ACOG can be found at https://www.acog.org/.
• American Pharmacists Association (APhA)
Founded in 1852, APhA is the largest association of pharmacists in the United
States, with more than 62,000 practicing pharmacists, pharmaceutical scientists,
student pharmacists, pharmacy technicians as members. Information about APhA
educational activities can be found at www.pharmacist.com/immunization-center.
• Group on Immunization Education of the Society of Teachers of Family
Medicine
The Group on Immunization Education of the Society of Teachers of Family
Medicine provides information for clinicians, including the free program Shots.
Shots includes the childhood, adolescent, and adult schedules for iPhone, Palm, and
Windows devices, as well as online versions (http://www.immunizationed.org/).

General Best Practice Guidelines for Immunization: Vaccination Programs
192
• Immunization Action Coalition (IAC)
IAC provides child, teen, and adult immunization information for health care
professionals and their patients at http://www.immunize.org. Free materials
include CDC-reviewed technical pieces, patient handouts, VISs in multiple
languages, and the weekly immunization news and information service “IAC
Express,” available at http://www.immunize.org/express. Information for the
general public about vaccines and vaccine-preventable diseases is available at
http://www.vaccineinformation.org.
• Institute for Vaccine Safety
Located at the Johns Hopkins University School of Public Health, the Institute for
Vaccine Safety provides information about vaccine safety concerns and objective
and timely information to physicians and health-care providers and parents. The
Institute for Vaccine Safety also includes links to tables that include all vaccine
components (http://www.vaccinesafety.edu).
• State and Local Health Departments
State and local health departments provide technical advice through hotlines, e-
mail, and websites, including printed information regarding vaccines and
immunization schedules, posters, and other educational materials (see
www.cdc.gov/vaccines/imz- managers/awardee-imz-websites.html and
www.cdc.gov/mmwr/international/relres.html).
• Vaccine Education Center
Located at the Children’s Hospital of Philadelphia, the Vaccine Education Center
provides patient and provider vaccine information (www.chop.edu/centers-
programs/vaccine-education-center).
General Best Practice Guidelines for Immunization: Appendix 1: Glossary
193
Appendix 1: Glossary
Adverse event. An undesirable medical condition that occurs following vaccination
which might be truly caused by a vaccine, or it might be pure coincidence.
Adverse reaction. An undesirable medical condition that has been demonstrated to
be caused by a vaccine. Evidence for the causal relation is usually obtained through
randomized clinical trials, controlled epidemiologic studies, isolation of the vaccine
strain from the pathogenic site, or recurrence of the condition with repeated
vaccination (i.e., rechallenge); synonyms include side effect and adverse effect.
Adjuvant. A vaccine component distinct from the antigen that enhances the immune
response to the antigen.
Antitoxin. A solution of antibodies against a toxin. Antitoxin can be derived from
either human (e.g., tetanus immune globulin) or animal (usually equine) sources (e.g.,
diphtheria and botulism antitoxin). Antitoxins are used to confer passive immunity
and for treatment.
Hyperimmune globulin (specific). Special preparations obtained from blood
plasma from donor pools preselected for a high antibody content against a specific
antigen (e.g., hepatitis B immune globulin, varicella-zoster immune globulin, rabies
immune globulin, tetanus immune globulin, vaccinia immune globulin,
cytomegalovirus immune globulin, botulism immune globulin).
Immune globulin. A sterile solution containing antibodies, which are usually
obtained from human blood. It is obtained by cold ethanol fractionation of large pools
of blood plasma and contains 15%-18% protein. Intended for intramuscular
administration, immune globulin is primarily indicated for routine maintenance of
immunity among certain immunodeficient persons and for passive protection against
measles and hepatitis A.

General Best Practice Guidelines for Immunization: Appendix 1: Glossary
194
Immunization. The process of inducing or providing immunity by administering an
immunobiologic. Immunization can be active or passive. Active immunization is the
production of antibody or other immune responses through administration of a
vaccine or toxoid. Passive immunization means the provision of temporary immunity
by the administration of preformed antibodies.
Immunobiologic. Antigenic substances (e.g., vaccines and toxoids) or antibody-
containing preparations (e.g., globulins and antitoxins) from human or animal donors.
These products are used for active or passive immunization or therapy. Examples of
immunobiologics include antitoxin, immune globulin and hyperimmune globulin,
monoclonal antibodies, toxoids, and vaccines.
Intravenous immune globulin. A product derived from blood plasma from a donor
pool similar to the immune globulin pool, but prepared so that it is suitable for
intravenous use. Intravenous immune globulin is used primarily for replacement
therapy in primary antibody-deficiency disorders, for treatment of Kawasaki disease,
immune thrombocytopenic purpura, hypogammaglobulinemia in chronic lymphocytic
leukemia, and certain cases of human immunodeficiency virus infection (Table 3-6).
Monoclonal antibody. An antibody product prepared from a single
lymphocyte clone, which contains only antibody against a single antigen.
Simultaneous. In the context of vaccine timing and spacing, occurring on the
same clinic day, at different anatomic sites, and not combined in the same syringe.
Toxoid. A modified bacterial toxin that has been made nontoxic, but retains the ability
to stimulate the formation of antibodies to the toxin.
Vaccination. The physical act of administering any vaccine or toxoid.
Vaccine. A suspension of live (usually attenuated) or inactivated microorganisms (e.g.,
bacteria or viruses) or fractions thereof administered to induce immunity and prevent
infectious disease or its sequelae. Some vaccines contain highly defined antigens (e.g.,
the polysaccharide of Haemophilus influenzae type b or the surface antigen of hepatitis
B); others have antigens that are complex or incompletely defined (e.g., Bordetella
pertussis antigens or live, attenuated viruses).
General Best Practice Guidelines for Immunization: Appendix 2: Membership
195
Appendix 2: Membership
Advisory Committee on Immunization Practices
Membership List, October 2018
Chair: ROMERO, José R., MD, FAAP, University of Arkansas for Medical Sciences and
Arkansas Children’s Hospital Little Rock, AR
Executive Secretary: Amanda Cohn, MD, National Center for Immunization
and Respiratory Diseases, CDC, Atlanta, Georgia.
Members: ATMAR, Robert L., MD, Baylor College of Medicine, Houston, TX
BERNSTEIN, Henry, DO, MHCM, FAAP, Zucker School of Medicine at
Hofstra/Northwell Cohen Children’s Medical Center, New Hyde Park, NY
EZEANOLUE, Echezona, MD, MPH, University of Nevada, Las Vegas, NV
FREY, Sharon E., MD, Saint Louis University Medical School, Saint Louis, MO
HUNTER, Paul, MD, City of Milwaukee Health Department Milwaukee, WI
LEE, Grace M., MD, MPH, Lucile Packard Children’s Hospital, Stanford
University School of Medicine, Stanford, CA
MOORE, Kelly, MD, MPH, Vanderbilt University School of Medicine, Nashville,
TN STEPHENS, David, MD, Emory University, Atlanta, GA
SZILAGYI, Peter, MD, MPH, University of California, Los Angeles (UCLA), Los Angeles,
CA
WALTER, Emmanuel (Chip), Jr., MD, MPH, Duke University School of
Medicine, Durham, NC
General Best Practice Guidelines for Immunization: Appendix 2: Membership
196
Ex Officio Members: Eric Deussing, MD, MPH, Department of Defense, DoD; Lori
Hoffman-Hogg, Department of Veterans Affairs, Durham, North Carolina; Doran
Fink, MD, PhD, Food and Drug Administration, Bethesda, Maryland; Narayan Nair,
MD, Health Resources and Services Administration, Rockville, Maryland; Thomas
Weiser, MD, MPH, Indian Health Service, Albuquerque, New Mexico; Tammy
Beckham, National Vaccine Program Office, Washington, District of Columbia; John
Beigel, MD, National Institutes of Health, Bethesda, Maryland.
Liaison Representatives: American Academy of Family Physicians, Pamela G.
Rockwell, DO, Ann Arbor, Michigan; American Academy of Pediatrics, Yvonne
Maldonado, MD, Stanford, California; David Kimberlin, MD, Birmingham, Alabama;
American Academy of Physician Assistants, Marie-Michèle Léger MPH, PA-C,
Alexandria, Virginia; American College Health Association, Susan Even, MD, Columbia,
Missouri; American College of Nurse Midwives, Carol E. Hayes, CNM, MN, MPH,
Atlanta, Georgia; American College of Physicians, Jason M. Goldman, MD, FACP, Boca
Raton, FL; American Geriatrics Society, Kenneth Schmader, MD, Durham, North
Carolina; American Medical Association, Sandra Adamson Fryhofer, MD, Atlanta,
Georgia; American Nurses Association, Chad Rittle, DNP, Pittsburgh, Pennsylvania;
American Osteopathic Association, Stanley Grogg, DO, Tulsa, Oklahoma; American
Pharmacists Association, Stephan L. Foster, PharmD, Memphis, Tennessee; American
Immunization Registry Association, Rebecca Coyle, MSEd, Washington D.C.; America’s
Health Insurance Plans, Mark Netoskie, MD, MBA, Houston, Texas; Association of
Immunization Managers, Chris Finley, RN, MPH, Burlington, VT; Council of State and
Territorial Epidemiologists, Christine Hahn, MD, Boise, ID; Infectious Diseases Society
of America, Kathleen Neuzil, MD, Seattle, Washington; National Association of City and
County Health Officials, Jeff Duchin, MD, Seattle, WA; National Association of Pediatric
Nurse Practitioners, Patricia Stinchfield, MPH, St Paul, Minnesota; National Foundation
for Infectious Diseases, William Schaffner, MD, Nashville, Tennessee; National Medical
Association, Patricia Whitley-Williams, MD, New Brunswick, New Jersey; Pediatric
Infectious Diseases Society, Sean O’Leary, MD, MPH, Colorado; Pharmaceutical
Research and Manufacturers of America, Cory Robertson;
General Best Practice Guidelines for Immunization: Appendix 2: Membership
197
Society for Adolescent Medicine, Amy Middleman, MD, Oklahoma City, OK; Society for
Healthcare Epidemiology of America, David Weber, MD, Chapel Hill, North Carolina
Houston, Texas.
Members of the General Best Practices Work Group, January, 2023
Advisory Committee on Immunization Practices (ACIP), Lynn Bahta, RN; Sarah Long,
MD; Pablo Sanchez, MD; ACIP Liaison and Ex-Officio Members, Marcie Drees, MD,
Society for Health Care Epidemiology of America; Susan Farrall, Office of Infectious
Disease and HIV/AIDS Policy; Mary-Margaret Fill, MD, Council of State and Territorial
Epidemiologists; Michelle Fiscus, MD, Association of Immunization Managers; David
Kim, MD, Office of Infectious Disease and HIV/AIDS Policy; Marie-Michele Leger, MPH,
American Academy of Physician Associates; Anuja Rastogi, Food and Drug
Administration; Charles (Chad) Rittle, American Nurses Association; Pam Rockwell,
DO, American Academy of Family Physicians; Mark Sawyer, MD, American Academy of
Pediatrics; CDC Staff Members, Andrew Kroger, MD, Cynthia Jorgensen, Alison Lale,
MD, Eloisa Llata, MD, Elaine Miller, Sarah Morales, Sarah Schillie, MD, Akpobome
Wodi, MD, Joellen Wolicki, RN; other members and consultants, Kenneth Bromberg,
MD, SUNY Downstate Medical School, Paul Hunter, MD, University of Wisconsin,
Susan Lett, MD, Kelly Moore, MD, Immunize.org, Walter Orenstein, MD, Emory
University School of Medicine, Lynn Trefren, Skip Wolfe.
