
Principles of Heat and
Mass Transfer
UNIT 1 PRINCIPLES OF HEAT AND MASS
TRANSFER
Structure
1.0 Objectives
1.1 Introduction
1.2 Heat Transfer System
Conduction
Convection
Radiation
Overall Heat Transfer Coefficients
Heat Transfer from Condensing Vapours
Heat Transfer to Boiling Liquids
1.3 Type of Food for Heat Processing
1.4 Heat Penetration
1.5 Heat Transfer Characteristics of Food
1.6 Devices for Determination of Heat Penetration
1.7 Determination of Cold Point in a Food Container
1.8 Calculation of Process Time
1.9 Factors Affecting Heat Penetration
1.10 Let Us Sum Up
1.11 Key Words
1.12 Answers to Check Your Progress Exercises
1.13 Some Useful Books
1.0 OBJECTIVES
By the time you have studied this unit, you should be able to:
• define the basic principles and methods of heat transfer;
• explain the role of heat transfer in heat preservation processes;
• identify the type of food for heat processing;
• determine the heat penetration and calculate the process time in a food;
and
• identify the factors affecting heat transfer and apply corrective measures to
enhance the process of heat transfer.
1.1 INTRODUCTION
Heat transfer is an important operation in the food industry. Whether it is
called cooking, baking, drying, sterilizing or freezing, heat transfer is part of
processing of almost every food. Heat transfer is a dynamic process in which
heat is transferred spontaneously from one body to another cooler body. The
rate of heat transfer depends upon the differences in temperature between the
bodies, the greater the difference in temperature, the greater will be the rate of
heat transfer.
Temperature difference between the source of heat and the receiver of heat is,
therefore, the driving force in heat transfer. An increase in the temperature
difference increases the driving force and, thus the rate of heat transfer. The
heat passing from one body to another travels through some medium, which in
general offers resistance to the heat flow. Both these factors, the temperature
difference and the resistance to heat flow, affect the rate of heat transfer.
7

Food Preservation by
Application of Heat
1.2 HEAT TRANSFER SYSTEM
Heat can be transferred from one object to another in three ways: by
conduction, by convection and by radiation.
Conduction is the movement of heat by direct transfer of molecular energy
within solids. The molecules with greater energy communicating some of this
energy to neighbouring molecules with less energy. An example of conduction
is the heat transfer through the solid walls of a refrigerated store.
Convection is the transfer of heat by the movement of groups of molecules in
a fluid. The groups of molecules may be moved by either density changes or
by forced motion of the fluid. An example of convection heating is cooking in
a jacketed pan: without a stirrer, density changes cause heat transfer by natural
convection; while with a stirrer, the convection is forced.
Radiation is the transfer of heat energy by electromagnetic waves, which
transfer heat from one body to another, in the same way as electromagnetic
light waves transfer light energy. An example of radiant heat transfer is when
a foodstuff is passed below a bank of electric resistance heaters that are red-
hot (electric grill).
In general, heat is transferred in solids by conduction and in fluids by
conduction and convection (Figure 1.1). Heat transfer by radiation occurs
through open space, can often be neglected, and is most significant when
temperature differences are substantial. In practice, the three types of heat
transfer may occur simultaneously. For calculations it is often best to consider
the mechanisms separately, and then to combine them where necessary.
(a) (b)
Figure 1.1: Heat transfer in containers by (a) conduction and (b) convection
1.2.1 Conduction
In the case of heat conduction, the equation, heat transfer rate = driving
force/resistance, can be applied directly. The driving force is the temperature
difference per unit length of heat-transfer path, i.e., temperature gradient.
Instead of resistance to heat flow, its reciprocal, conductance, is used. This
changes the form of the general equation to:
Rate of heat transfer = driving force x conductance, which is:
dQ/dt = kA dT/dT x (1.1)
8

Principles of Heat and
Mass Transfer
Where, dQ/dt (JS
−1
)is the rate of heat transfer, the quantity of heat energy
transferred per unit of time, A (m
2
)is the area of cross-section of the heat flow
path, dT/dT x ( Cm )is the temperature gradient, that is the rate of change of
temperature per unit length of path, and
0 −1
k (Jm s K or Wm K )is the
thermal conductivity of the medium. Notice the distinction between thermal
conductance, which relates to the actual thickness of a given material (
−1 −1 0 −1 0 −1
k/x) and
thermal conductivity, which relates only to unit thickness. Eq. (1.1) is known
as the Fourier equation for heat conduction.
Thermal conductivity does change slightly with temperature, but in many
applications it can be regarded as a constant for a given material. Most
foodstuffs contain a high proportion of water and as the thermal conductivity
of water is about 0.7 J m
−1
s
−1
°C
-1
above 0°C, thermal conductivities of foods
are in the range 0.6-0.7 J m
−1
s
-1
°C
−1
. Ice has a substantially higher thermal
conductivity than water, about 2.3 J m
−1
s
−1
°C
−1
. The thermal conductivity of
frozen foods is, therefore, higher than foods at normal temperatures.
1.2.2 Convection
Convection heat transfer is the transfer of energy by the mass movement of
groups of molecules. It is restricted to liquids and gases, as mass molecular
movement does not occur at an appreciable speed in solids. It cannot be
mathematically predicted as easily as can transfer by conduction or radiation
and so its study is largely based on experimental results rather than on theory.
Newton found, experimentally, that the rate of cooling of the surface of a
solid, immersed in a colder fluid, was proportional to the difference between
the temperature of the surface of the solid and the temperature of the cooling
fluid. This is known as Newton's Law of Cooling, and it can be expressed by
the following equation.
q = h
s
A(T – T
a
TT
s
) (1.2)
Where, h
s
is called the surface heat-transfer coefficient, T is the temperature
of the cooling fluid and
T
a
TT
s
is the temperature at the surface of the solid. The
surface heat-transfer coefficient can be regarded as the conductance of a
hypothetical surface film of the cooling medium of thickness
x
f
such that
h
s
= k
f
/x
f
Where, k
f
is the thermal conductivity of the cooling medium. It is useful at
this point, however, to appreciate the magnitude of h
s
under various common
conditions and these are shown in Table 1.1.
Table 1.1: Approximate range of surface heat transfer coefficients
hs (J m
−2
s
−1
°C
−1
)
Boiling liquids 2400-24,000
Condensing liquids 1800-18,000
Still air 6
Moving air (3 m s
−1
)
30
Liquids flowing through pipes 1200-6000
1.2.3 Radiation
9

Food Preservation by
Application of Heat
Radiation heat transfer is the transfer of heat energy by electromagnetic
radiation. Radiation operates independently of the medium through which it
occurs and depends upon the relative temperatures, geometric arrangements
and surface structures of the materials that are emitting or absorbing heat.
Radiation of wavelength 0.8-400 µm (infrared) is referred to as thermal
radiation or heat rays since electro magnetic radiation with this wavelength is
most readily absorbed and converted to heat energy. The infrared radiation is
used primarily for surface heating as it is transmitted rapidly to the surface. It
is used for dehydration of fruits and vegetables, freeze drying, baking, etc.
Radiation can be significant with small temperature differences as, for
example, in freeze-drying and in cold stores, but it is generally more important
where the temperature differences are greater. Under these circumstances, it is
often the most significant mode of heat transfer, for example in bakers' ovens
and in radiant driers.
The basic formula for radiant-heat transfer is the Stefan-Boltzmann Law
q = A σT
4
(1.3)
Where, T is the absolute temperature (measured from the absolute
zero of temperature at −273°C, and indicated in Bold type) in degrees
Kelvin (K) in the SI system, and (sigma) is the Stefan-Boltzmann constant
= 5.73 × 10
−8
J m
−2
s
−1
K
−4
The absolute temperatures are calculated by the
formula K = (°C+273).
This law gives the radiation emitted by a perfect radiator (a black body as this
is called though it could be a red-hot wire in actuality). A black body gives the
maximum amount of emitted radiation possible at its particular temperature.
Real surfaces at a temperature T do not emit as much energy as predicted by
Eq. (1.3), but it has been found that many emit a constant fraction of it. For
these real bodies, including foods and equipment surfaces, that emit a constant
fraction of the radiation from a black body, the equation can be rewritten
q
= εA σT
4
(1.4)
Where, ε (epsilon) is called the emissivity of the particular body and is a
number between 0 and 1. Bodies obeying this equation are called grey bodies.
Emissivities vary with the temperature T and with the wavelength of the
radiation emitted. For many purposes, it is sufficient to assume that for:
* dull black surfaces (lamp-black or burnt toast, for example), emissivity is
approximately 1;
* surfaces such as paper/painted metal/wood and most foods, emissivities
are about 0.9;
* rough un-polished metal surfaces, emissivities vary from 0.7 to 0.25;
* polished metal surfaces, emissivities are about or below 0.05.
These values apply at the low and moderate temperatures, which are those
encountered in food processing. Just as a black body emits radiation, it also
absorbs it and according to the same law, Eq. (1.3). Again grey bodies absorb
a fraction of the quantity that a black body would absorb, corresponding this
time to their absorptivity α (alpha). For grey bodies it can be shown that α =
ε. The fraction of the incident radiation that is not absorbed is reflected, and
thus, there is a further term used, the reflectivity, which is equal to (1 – α).
The radiant energy transferred between two surfaces depends upon their
temperatures, the geometric arrangement, and their emissivities. For two
10

Principles of Heat and
Mass Transfer
parallel surfaces, facing each other and neglecting edge effects, each must
intercept the total energy emitted by the other, either absorbing or reflecting it.
In this case, the net heat transferred from the hotter to the cooler surface is
given by:
q = ACσ (T − T
1
4
TT
2
4
) (1.5)
where 1/C = 1/ε
1
+ 1/ε
2
− 1, ε
1
is the emissivity of the surface at temperature
T and ε is the emissivity of the surface at temperature T
1 2
TT
2
.
1.2.4 Overall Heat Transfer Coefficients
It is most convenient to use overall heat transfer coefficients in heat transfer
calculations as these combine all of the constituent factors into one, and are
based on the overall temperature drop. Radiation coefficients, subject to the
limitations discussed in the section on radiation, can be incorporated in the
overall coefficient. The radiation coefficients should be combined with the
convection coefficient to give a total surface coefficient, as they are in series,
and so:
h
s
= (h
r
+ h
c
) (1.6)
The overall coefficient U for a composite system, consisting of surface film,
composite wall, surface film, in series, can then be calculated as:
1/U = 1/(h
r
+ h
c
)
1
+ x
1
/k
1
+ x
2
/k
2
+ …+ 1/(h
r
+ h
c
)
2
(1.7)
In Eq. (1.7) often one or two terms are much more important than other terms
because of their numerical values. In such a case, the important terms,
signifying the low thermal conductance are said to be the controlling terms.
1.2.5 Heat Transfer from Condensing Vapours
The rate of heat transfer obtained when a vapour is condensing to a liquid is
very often important. In particular, it occurs in the food industry in steam-
heated vessels where the steam condenses and gives up its heat; and in
distillation and evaporation where the vapours produced must be condensed.
In condensation, the latent heat of vaporization is given up at constant
temperature, the boiling temperature of the liquid. Two generalized equations
have been obtained:
1) For condensation on vertical tubes or plane surfaces
h
v
= 0.94 [(k
3
ρ
2
g/μ) × (λ/LΔT)]
0.25
(1.8)
Where, λ (lambda) is the latent heat of the condensing liquid in J kg
−1
, L is
the height of the plate or tube and the other symbols have their usual
meanings.
2) For condensation on a horizontal tube
h
h
= 0.72 [(k
3
ρ
2
g/μ) × (λ/DΔT)]
0.25
(1.9)
1.2.6 Heat Transfer to Boiling Liquids
When the presence of a heated surface causes a liquid near it to boil, the
intense agitation gives rise to high local coefficients of heat transfer. A
considerable amount of experimental work has been carried out on this, but
generalized correlations are still not very adequate. It has been found that the
11

Food Preservation by
Application of Heat
apparent coefficient varies considerably with the temperature difference
between the heating surface and the liquid. For temperature differences greater
than about 20°C, values of h decrease, apparently because of blanketing of the
heating surface by vapours. Over the range of temperature differences from 1
to 20°C, values of h for boiling water increase from 1200 to about 60,000 J
m
−2
s
−1
°C
−1
. For boiling water under atmospheric pressure, the following
equation is approximately true:
h = 50(ΔT)
2.5
(1.10)
Where, ΔT is the difference between the surface temperature and the
temperature of the boiling liquid and it lies between 2 and 20°C. In many
applications the high boiling film coefficients are not of much consequence, as
resistance in the heat source controls the overall coefficients.
#
Check Your Progress Exercise 1
Note: a) Use the space below for your answer.
b) Compare your answers with those given at the end of the unit.
1. List the different methods of heat transfer.
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
2. What is the relationship of conduction heat transfer rate with temperature
difference? What is the name of the equation used for determining the
conduction heat transfer rate?
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
3. What is the name of the equation used for expressing the radiative flux
from an object? How is it related to temperature and properties of the
material?
………………………………………………………………………………
………………………………………………………………………………
12

Principles of Heat and
Mass Transfer
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
4. While estimating the rate of heat transfer between two objects, the
temperature of one of the objects is doubled. If convection and radiation
are the two modes of heat transfer between the two objects, which mode
would increase more and why?
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
1.3 TYPE OF FOOD FOR HEAT PROCESSING
There are essentially two types of food when we talk of thermal processing:
1. Acid foods
2. Low acid foods
These two categories of foods differ significantly in their behaviour when
thermally processed. The acidity of the food, using the pH scale as a measure
of acidity, where 1 = very high acid and 4 = very low acid; the dividing line
for acid
foods and low-acid foods is pH 4.6. Acid foods can be canned at a
temperature of 100°C, while low-acid
foods must be pressure canned (to a
temperature of 115°C). The reason for this is that the toxin-producing,
potentially lethal organism, Clostridium botulinum, will not grow and produce
toxins at a pH below 4.6. Many spoilage microorganisms will not grow
between pH 1 and 4.6 either. The most common spoilage microorganisms
associated with acid foods are yeasts and moulds that can tolerate acid
environments.
1. Acid foods
High acid foods contain more natural acids. Many fruits are high acid
foods and the presence of these natural acids helps prevent growth of some
spoilage microorganisms. If the food product has a high enough acid level,
boiling-water temperatures are high enough to destroy spoilage organisms.
This is a prevention method for the deadly Clostridium botulinum bacteria.
2. Low acid foods
Low acid foods, such as vegetables and meat products, contain very little
natural acid. They must be processed at higher temperatures than boiling-
water to destroy any Clostridium botulinum bacteria. Water boils at
100°C, at sea level, and at a lower temperature at higher elevations.
Turning up the temperature under the pot or letting the water boil for a
13

Food Preservation by
Application of Heat
longer time does not raise the temperature of the water above its boiling
point. To make water boil at a higher temperature, it has to be put under
pressure, such as in a pressure canner. When a food is processed at 1.0
kg/cm
2
pressure, the water boils when it gets to 115°C, rather than at
100°C. This is high enough to kill the bacteria that causes botulism
poisoning.
Adjust for Altitude to Ensure Safety
The above values of temperatures have been determined for mean sea level.
As we move up the mountains, the atmospheric pressure goes down and water
boils at lower temperatures as altitude increases. Lower boiling temperatures
are less effective for killing bacteria. You must increase either the process
time or canner pressure to make up for lower boiling temperatures.
Because altitude affects pressure and the boiling point of liquid, adjustments
must be made when canning foods at altitudes of 300 m above sea level or
higher. When using the boiling water bath method, processing time must be
increased. Add 5 minutes to processing time for altitudes between 300 m and
1500 m above sea level. When using the pressure canner method, pressure
must be increased. If using a dial-gauge pressure canner, process foods at 0.8
bar pressure for altitudes between 600 m and 1200 m and at 0.9 bar pressure
for altitudes between 1200 m and 1800 m. If using the weight-gauge pressure
canner, use 1.0 bar of pressure.
When you mix low-acid and acid foods, assume that the mixture remains low-
acid. Although tomatoes used to be considered an acid food, some are now
known to have pH values slightly above 4.6, which means they are low-acid.
To safely can them as acid foods in a boiling-water canner, you must add
lemon juice or citric acid.
#
Check Your Progress Exercise 2
Note: a) Use the space below for your answer.
b) Compare your answers with those given at the end of the unit.
1. How are acid and low-acid foods distinguished?
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
2. How is a thermal process for an acid food different than that for a low-acid
food?
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
14

Principles of Heat and
Mass Transfer
3. What is the method used to raise the boiling point of water in food
processing?
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
4. What changes are required for thermal processing of foods at high
altitudes?
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
1.4 HEAT PENETRATION
Heat penetration studies are required to be conducted for verifying the
sterilizing temperature of a load (food) meant for moist heat sterilization.
These studies are conducted to ensure that the coolest unit within a pre-
defined loading pattern (including minimum and maximum loads) will
consistently be exposed to sufficient heat lethality (minimum “F”).
Heat penetration curve can be drawn by plotting the logarithmic difference
between either retort temperature and product temperature (heating curve) or
product temperature and cooling medium temperature (cooling curve) versus
time. The purpose of a heat-penetration study is to determine the heating and
cooling behaviour of a product/package combination in a specific retort
system for the establishment of safe thermal processes and evaluating process
deviations. The study is designed to adequately and accurately examine all
critical factors associated with the product, package and process, which affect
heating rates. A goal in conducting these studies is to identify the worst-case
temperature response expected to occur in commercial production as
influenced by the product, package and process.
Several product, process, package and measurement-related factors can
contribute to variations in the time-temperature data gathered during a heat-
penetration test. Establishment of a process requires expert judgment and
sound experimental data for determining which factors are critical and the
effect of changing those factors both within and beyond established critical
limits.
A typical heat penetration curve is shown in Figure 1.2. A broken heating
curve occurs when a food is initially heated by convective heating but then
undergoes a rapid transition to conductive heating (for example in foods,
which contain high concentration of starch, which undergoes a sol-to-gel
transition).
15

Food Preservation by
Application of Heat
Figure 1.2: Heat penetration curve
There are a number of ways of estimating how effective heat sterilization can
be. The thermal death time is the time microbes must be exposed to a
particular temperature before they are all dead. Similarly, the thermal death
point is the temperature at which all microbes in a sample are killed. Both are
very unsatisfactory, since they depend on many factors such as number of
microbes present in a sample, analytical conditions and techniques, etc.
1.5 HEAT TRANSFER CHARACTERISTICS OF FOOD
It is necessary to have information on both the heat resistance of
microorganisms or enzymes and the rate of heat penetration into the food for
determination of process time of a food.
Heat is transferred from steam or pressurized water through the container and
into the food. Generally the surface heat transfer coefficient is very high and is
not a limiting factor in heat transfer. The following factors influence the rate
of heat penetration into a food:
i) Type of product
ii) Size of the container
iii) Agitation of the container
iv) Temperature of the retort
v) Shape of the container. Tall containers promote convection currents in
convective heating foods.
vi) Type of container. Heat penetration is faster through metal than through
glass or plastics owing to differences in thermal conductivity.
In this section, the objective is to learn as to how the thermal properties of
food products affect the heat penetration and the quantity of heat. Two thermal
properties of importance are thermal conductivity and thermal diffusivity in
determining heat penetration. Specific heat and latent heat are important in
determining the quantity of heat required for the process
.
I. Thermal conductivity is the property indicating the rate at which heat
flows through a food product. A product with high thermal conductivity
lets the heat flow easily, whereas a material with low thermal conductivity,
also known as an insulator, puts resistance to the flow of heat. Fourier’s
heat conduction equation could be used to derive the units of thermal
conductivity, i.e., W/(m°C). It does change slightly with temperature, but
16

Principles of Heat and
Mass Transfer
in many applications it can be regarded as a constant for a given material.
Most foodstuffs contain a high proportion of water and as the thermal
conductivity of water is about 0.7 J m
−1
s
−1
°C
−1
above 0°C, thermal
conductivities of foods are in the range of 0.6-0.7 J m
−1
s
−1
°C
−1
. Ice has a
substantially higher thermal conductivity than water, about 2.3 J m
−1
s
−1
°C
−1
. The thermal conductivity of frozen foods is, therefore, higher than
foods at normal temperatures.
Typical thermal conductivities
Metals: k = 50-400 W/m°C
Water: k = 0.597 W/m°C
Air: k = 0.0251 W/m°C
Insulating materials: k = 0.035 - 0.173 W/m°C
For foods it is represented as
k = 0.25 m
c
+ 0.155 m
p
+ 0.16 m
f
+ 0.135 m
a
+ 0.58 m
m
Where m is mass fraction and subscripts c: carbohydrate, p: protein, f: fat,
a: ash and m: moisture.
or
k = 0.55p/100 + 0.26(100−p)/l00 J m
−1
s
−1
°C
−1
above freezing
= 2.4p/100 + 0.26(100 -p)/l00 J m
−1
s
-1
°C
−1
below freezing.
Where p is the percentage of water in the foodstuff.
II. Thermal diffusivity is the actual ability of a food to conduct heat to
adjacent molecules. Thermal diffusivity is a derived property that is the
ratio of thermal conductivity and the product of density and specific heat.
The units of thermal diffusivity, therefore, work out to be m
2
/s. Higher
value of thermal diffusivity means faster heat penetration and vice versa.
III. Specific heat: The specific heat is an important quantity that determines
the amount of energy that must be supplied or withdrawn from a unit mass
of material in order to increase or decrease its temperature by one degree.
Knowledge of the specific heat of a material is, therefore, important in the
design of processes such as chilling, freezing, warming, sterilization and
cooking. Specific heat has the units of kJ/(kg.K) in SI system of units.
Specific heat = 4.19p/100 + 0.84(100 − p)/100 kJ kg
−1
°C
−1
above freezing
= 2.1 p/100 + 0.84(100 − p)/100 kJ kg
−1
°C
−1
below freezing.
p is percentage of water in food stuff
IV. Phase transitions: It is important to determine the temperature at which
transitions occur, the enthalpy change associated with a transition, the type
of transition involved (exothermic or endothermic), and the quantity of
material that undergoes a transition. As an example, we will consider the
melting and crystallization of food components. When a material changes
its physical state from solid-to-liquid (melting) or from liquid-to-solid
(crystallization) it absorbs or gives out heat, respectively. A process that
absorbs heat is an endothermic process, whereas a process that evolves
heat is an exothermic process. Pure substances usually have very sharp
melting or crystallization points and, therefore, all the heat is absorbed or
evolved over a narrow range of temperature. Most foods are complex
17
Food Preservation by
Application of Heat
materials and, therefore, do not exhibit sharp transitions from one phase to
another. The amount of heat required for the phase change is called the
latent heat and has the units of kJ/kg.
Latent heat = 335p/100 kJ kg
−1
This equation and the ones given earlier for thermal conductivity and
specific heat represent a considerable over-simplification so they should be
used with caution, particularly in the region between −18°C to 0°C.
Freezing of foodstuffs occur over a range of temperatures and not at any
fixed point.
Some properties of liquids and thermal data for food products are depicted in
Tables 1.2 and 1.3, respectively.
Table 1.2: Some properties of liquids
Thermal
conductivity
Specific
heat
Density Viscosity Temperature
(J m
−1
s
−1
°C
−1
)
(kJ kg
−1
°C
−1
)
(kg m
−3
) (N s m
−2
)
(°C)
Water 0.57 4.21 1000
1.87 × 10
−3
0
4.21 987
0.56 × 10
−3
50
0.68 4.18 958
0.28 × 10
−3
100
Sucrose
20% soln.
0.54 3.8 1070
1.92 × 10
−3
20
0.59 × 10
−3
80
60% soln.
6.2 × 10
−3
20
5.4 × 10
−3
80
Sodium
chloride
22% soln.
0.54 3.4 1240
2.7 × 10
−3
2
Acetic
acid
0.17 2.2 1050
1.2 × 10
−3
20
Ethyl
alcohol
0.18 2.3 790
1.2 × 10
−3
20
Glycerine 0.28 2.4 1250
830 × 10
−3
20
Olive oil 0.17 2.0 910
84 × 10
−3
20
Rape-seed
oil
900
118 × 10
−3
20
Soya-bean
oil
910
40 × 10
−3
30
Tallow 900
18 × 10
−3
65
18

Principles of Heat and
Mass Transfer
Milk
(whole)
0.56 3.9 1030
2.12 × 10
−3
20
Milk
(skim)
1040
1.4 × 10
−3
25
Cream
20% fat
1010
6.2 × 10
−3
3
30% fat 1000
13.8 × 10
−3
3
Table 1.3: Thermal data for some food products
Specific heat
Above
freezing
Below
freezing
Freezing
point (°C)
Percent
water
(kJ kg
-1
°C
-1
)
Latent heat of
fusion
(kJ kg
−1
)
Fruit
Apples
−2
84 3.60 1.88 280
Bananas
−2
75 3.35 1.76 255
Grapefruit
−2
89 3.81 1.93 293
Peaches
−2
87 3.78 1.93 289
Pineapples
−2
85 3.68 1.88 285
Watermelons
−2
92 4.06 2.01 306
Vegetables
Asparagus
−1
93 3.93 2.01 310
Beans (green)
−1
89 3.81 1.97 297
Cabbage
−1
92 3.93 1.97 306
Carrots
−1
88 3.60 1.88 293
Corn
−1
76 3.35 1.80 251
Peas
−1
74 3.31 1.76 247
Tomatoes
−1
95 3.98 2.01 310
Water 0 100 4.19 2.05 335
1.6 DEVICES FOR DETERMINATION OF HEAT
PENETRATION
There are different types of thermometers available for measuring the
temperature in a thermal process and, thereby, permitting the determination of
heat penetration.
I. Mercury-in-glass (MIG) thermometer
Each retort system used for the thermal processing is equipped with a MIG
thermometer. Aseptic processing systems may have a temperature
indicating device other than MIG thermometer as the sole temperature
indicator. The MIG thermometer is the reference instrument for all
temperature readings, including vent temperature, come-up temperature
and process temperature during processing.
It is important that the MIG thermometer be tested/calibrated at the
operating temperature of the retort system (i.e., 115C, 120C, 125C
19

Food Preservation by
Application of Heat
etc.) and if possible in the heating medium used in the retort. If the retort is
operated at more than one processing temperature or over a wide range of
temperatures the MIG thermometer should be checked at all of the
temperatures normally used for processing. The MIG thermometers should
be tested against a thermometer that can be traced back to a BIS Standard
thermometer. The accuracy of the standard thermometer should be
checked at least once every 3 years depending upon how it is handled and
stored.
II. Temperature recording device
Each retort system is equipped with an accurate temperature-recording
device. The recording device provides a continuous record of the
temperature in the retort system during thermal processing. Common
systems in use are circular or strip charts, which are marked with ink pens,
electrical sparks, pressure pins, or which are created by graph plotters at
the time temperature readings are received. Electronic temperature
monitors and recorders are now available for the purpose and should be
utilized for greater accuracy and precision avoiding human errors. A band
or ribbon type surface pyrometer is used by processors to monitor
container surface temperatures.
III. Temperature sensors
Temperature measurement can be accomplished by essentially five basic
methods: (1) liquid-in-glass, (2) resistance thermometry, (3)
thermoelectric thermometry, (4) optical/radiation pyrometry, and (5) bi-
metal. Investigators are most familiar with the liquid (mercury or alcohol
usually) -in-glass and the bi-metal (dial gauge) types. It is possible now
that investigators will encounter the use of the optical/radiation pyrometers
as well.
i) Resistance thermometry
A resistance thermometer is a temperature-measuring instrument
consisting of a sensor (an electrical circuit element whose resistance
varies with temperature), a framework on which to support the sensor,
a sheath by which the sensor is protected, and wires by which the
sensor is connected to a measuring instrument, which is used to
indicate the effect of variations in the sensor resistance. Resistance
thermometers provide absolute calibration of temperatures in that no
reference junctions are involved, and no special extension wires are
needed between the sensor and the measuring instrument (as with
thermocouples).
The sensors can be of two types: resistance temperature detectors
(RTD’s) and thermistors. The RTD sensing element is formed of solid
conductors (usually in wire form) wound upon an insulating core. The
insulating core is usually made of mica or ceramic. The conductors,
which are wound in a helical coil to prevent mechanical restraints
during thermal expansion, are generally made of platinum; however
nickel and copper have been used. Platinum best meets the
requirements because being a noble metal, it can be highly refined, it
resists contamination, it is mechanically and electrically stable, and the
relationship between temperature and resistance is quite linear.
Thermistors (a contraction for “thermally sensitive resistors”) are
electrical circuit elements formed of solid semi conducting materials
20

Principles of Heat and
Mass Transfer
such as oxides of nickel, manganese, iron, cobalt, copper, magnesium,
titanium, and other metals. The powdered metal is formed under
pressure into the desired shape, usually a flat disc. The disc is sintered,
leads are attached, and encapsulated in epoxy. The finished thermistor
can also be encased in a sheath of plastic, stainless steel, copper or
aluminum. Both the RTD and thermistor can be obtained in various
configurations.
ii) Thermoelectric thermometry
The thermoelectric thermometer is a temperature measuring instrument
consisting of two continuous, dissimilar thermocouple wires extending
from a measuring junction to a reference junction with copper
connecting wires to a potentiometer. Unlike the resistance types, where
power must be supplied to the circuit, the thermocouple circuit
generates a measurable low voltage output that is almost directly
proportional to the temperature difference between the “hot” junction
and the “cold” junction. A unit change in this temperature difference
will produce some net change in electromotive force (emf or voltage).
Thermoelectric thermometry makes use of the known relationship
between a difference in junction temperatures and the resulting emf
developed by a thermocouple circuit. The temperature of one junction
(reference junction, T1) is held at a constant known value. This is
usually accomplished with an ice water (0°C) bath. The temperature of
the other junction (measuring junction, T2) is determined by
measuring the thermocouple circuit emf and referring to calibration
tables for the particular thermocouple materials. The thermocouple
junction usually is formed by twisting and fusing the two wires
together or they may be butt-welded. The finished element may be
used bare or enclosed in a sheath.
Check Your Progress Exercise 3
#
Note: a) Use the space below for your answer.
b) Compare your answers with those given at the end of the unit.
1. Write the full forms of the following abbreviations:
a) MIG thermometer
b) RTD sensor
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
2. How is a thermocouple used for temperature measurement?
21

Food Preservation by
Application of Heat
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
3. What is a thermistor? How does it differ from a RTD sensor?
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
4. How is temperature in Kelvin scale related to temperature in Celsius
scale?
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
1.7 DETERMINATION OF COLD POINT IN A FOOD
CONTAINER
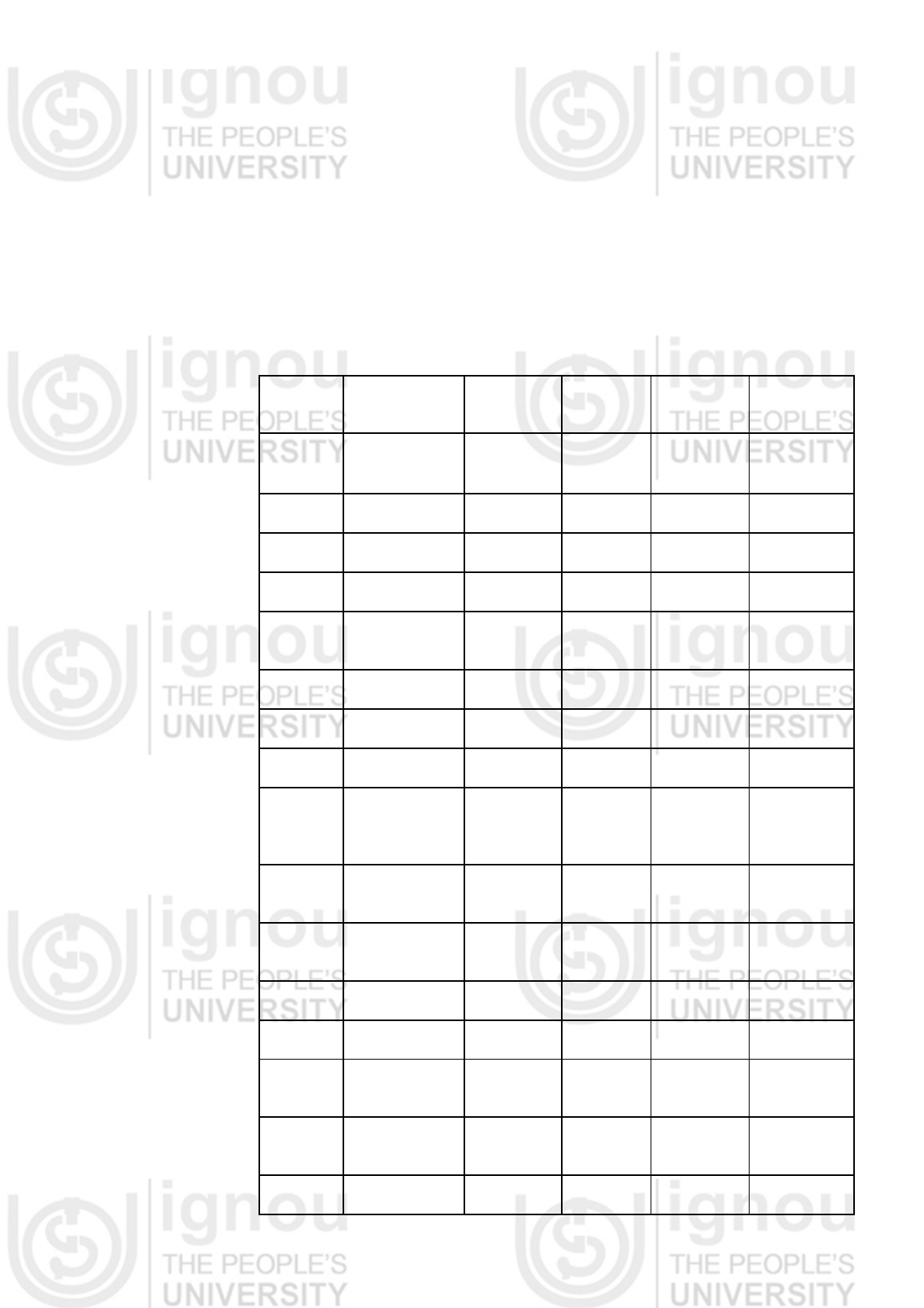
In a study, determination of the cold spot was made using data collected for
heat penetration curves at 5 potential cold spot locations in the jars in 18
canner loads (Table 1.4). Two levels of two procedural variations were used in
testing for process calculations. Temperature profiles were compared for two
fill weights (450g, 480g) and two fill temperatures (direct-fill, and after a 10
minute wait, which had means of 84.4°C and 80.4°C, respectively). Process
calculation was accomplished by using thermocouples in each of six jars in
different canner loads of each of the three fill methods (standard, low initial
temperature, and high-fill weight). These jars were processed to 90.5°C plus
an additional 5 minutes. Processing was done in a boiling water canner using
the stovetop burners of a household gas range. Data were recorded using
Type T copper-constantan thermocouples.
Cold Spot Location
• The cold spot for this product and jar combination was located at the
geometric centre of the jar, Table 1.4.
22

Principles of Heat and
Mass Transfer
• The D value is the number of minutes it takes the straight line portion of
the heat penetration plot to pass through one logarithmic cycle.
• A larger D represents a slower rate of heat penetration.
Table 1.4: Cold spot determination of cranberry salsa in pint jars
Thermocouple
height in pint jar
Average D value
n = 18
Range Standard
deviation
Centre 54.86¹ 48.5-73.4 5.3
½" Below Center 53.89 48.6-64.7 3.9
1" Below Center 51.94 45.8-64.9 4.8
1½" Below Center 48.98 43.0-60.8 4.7
2" Below Center 47.00 41.4-58.0 4.5
¹Location of cold spot, as determined by largest individual D value (worst-case
scenario)
Heat penetration measures the rate at which a product heats during a thermal
process. A temperature sensor or thermocouple measures temperature changes
in the slowest heating region of the product or container and temperature is
monitored on a recording device. The time/temperature data, and heat resistant
data for the target microorganism, are used to calculate the scheduled process.
1.8 CALCULATION OF PROCESS TIME
The time/temperature relationship required for desired reduction of microbial
population is based on thermal resistance characteristics of the
microorganisms. The translation of this information into a form for use by the
operator of a commercial system requires integration with the heating and
cooling characteristics of the food product within the container. The methods
to be presented lead to the establishment of a processing system operator time
to ensure that the impact of the thermal process is equivalent to the desired
time/temperature for a given microbial population.
One of the first concepts to be understood when establishing process times is
lethality. The term lethality can be defined as the integrated influence of time
and temperature on a microbial population. Lethality is expressed as time at a
reference temperature.
Time, min
Lethal rate
0
0
0.1
0.2
10
20 30
40
Figure 1.4: Lethal rate curve for typical process in retort
23

Food Preservation by
Application of Heat
The lethal rate increases gradually with time as the temperature of the product
increases. As the product temperature begins to plateau at a magnitude near
the heating medium temperature, the lethal rate also plateaus and eventually
decreases as the product temperature decreases during cooling. Lethality is
expressed in time units for the process accomplished at the heating medium
temperature.
The time/temperature relationship representing the process is compared to the
process requirement needed to achieve product safety or an established
spoilage rate. For example, if the process under consideration is being used to
ensure the elimination of Clostridium botulinum as a health risk, the lethality
for the process must be equal to or greater than the thermal death time for the
microbial population.
The D-value or Decimal Reduction Time may be used as a measure. This is
defined as the time taken under specified conditions and at a particular
temperature to kill 90 per cent of the microbes in a sample. Only 10 per cent
or 1/10 of the original number of microbes survive the decimal reduction time:
hence its name. D-values can be determined from survivor curves when the
log of population is plotted against time.
D
reference temperature
= Time/(Log
a
-Log
b
)
Where a = the initial population, and b = the survivors after a time interval. F
value is a mathematically calculated number that describes the total lethal
effect of the process at the slowest heating point in a food container. It is the
equivalent, in minutes at a given temperature, of all heat considered with
respect to its capacity to destroy spores or vegetative cells of a particular
microorganism.
The effectiveness of a canning process is determined from a combination of
experimentation and calculation. Processing parameters are expressed in terms
of a series of symbols of which D, z, and F are key. When bacterial spores are
heated to a lethal temperature as during retorting of canned foods, the death of
most species approximates a first order chemical reaction that can be
described by a straight line on a semi-logarithmic graph paper. Figure 1.4
hows a hypothetical result from heating a species of spore at 115°C (240°F). s
In Figure 1.5, one minute is required to reduce the survivors from 10,000 to
1,000 or a 90 per cent reduction (one log reduction). Similarly, one minute is
required to reduce the survivors from 1,000 to 100 per gram of food and so on
until only 0.01 of a spore is present in 1 gram of food-which really means that
there remains only one living spore for each 100 grams of food. This time to
reduce the survivors by 90% is the Decimal reduction (D) value or in Figure 1,
D = 1 min. The subscript after the D indicates temperature at which the D
value was determined. Many factors affect the D value, such as the species of
spore, and the kind of food the spore is suspended in.
115
24

Principles of Heat and
Mass Transfer
Figure 1.5: Thermal death time curve for Clostridium botulinum
Check Your Progress Exercise 4
#
Note: a) Use the space below for your answer.
b) Compare your answers with those given at the end of the unit.
1. What is Decimal Reduction Time (D)? How is it determined?
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
2. What is F value and how is it related to D value?
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
3. Name the microorganism that is considered in the determination of
thermal processing.
25

Food Preservation by
Application of Heat
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
4. What is meant by lethality in food processing? How is it related to various
process parameters?
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
………………………………………………………………………………
1.9 FACTORS AFFECTING HEAT PENETRATION
There are many factors, which affect the heat transfer into the food. Generally
the surface heat transfer coefficient is very high and is not a limiting factor in
heat transfer. The following factors are important which influences the rate of
heat penetration into a food:
1. Type of product: Liquid or particulate food (for example peas in brine),
where natural convection current is established, heat faster than solid food
(for example meat pastes and corned beef), where heat is transferred by
conduction. The low thermal conductivity of food is a major limitation to
heat transfer in conduction heating.
2. Size of the container: Heat penetration to the centre is faster in small
containers than in large containers.
3. Agitation of the container: End-over-end agitation (Figure 1.6) and, to a
lesser extent, axial agitation increases the effectiveness of natural
convection curie: and thereby increases the rate of heat penetration in
viscous or semi-solid foods (for example beans in tomato sauce).
26

Principles of Heat and
Mass Transfer
Figure 1.6: End-over-end agitation of containers
4. Temperature of the retort: A higher temperature difference between the
food and the heating medium causes faster heat penetration.
5. Shape of the container: Tall containers promote convection currents in
convective heating foods.
6. Type of container: Heat penetration is faster through metal than through
glass or plastics owing to differences in thermal conductivity.
1.10 LET US SUM UP
We have in this unit learnt the basic concept of heat transfer. Heat is
transferred by conduction, convection or radiation modes in a given situation.
The methods of temperature measurement include mercury-in-glass (MIG)
thermometers, resistance temperature detectors, thermistors, thermocouples
and radiation pyrometers. Temperature measurements permit us to evaluate
heat penetration rates in the thermal processes so as to determine the process
durations to achieve acceptable sterilization levels. These levels differ for
acid and less acid foods. If the pH level is below 4.6 the food is classified as
an acid food. However, if the pH is equal to or more than 4.6, the foods are
low-acid and the process temperatures would have to be more than 100°C.
Temperatures more than 100°C are achievable through raising process
pressure above that of the ambient. It is important to identify the cold spot in
the sterilization process because the heat penetration to that spot will control
the overall effectiveness of the process. Decimal reduction time at a given
reference temperature is used to fix the process time. Usually, 12 logarithmic
cycles are allowed for the microbial population reduction and, thus, the
process time F is equal to 12 D. The factors responsible for affecting the
temperature distribution and heat penetration rate need to be given due
consideration for finalizing the process durations.
1.11 KEY WORDS
27

Food Preservation by
Application of Heat
Conduction : Exchange of molecular energy directly
exchanged, from the hotter to the cooler regions.
Convection : Transfer of heat by the movement of groups of
molecules in a fluid.
Radiation : Transfer of heat energy by electromagnetic
waves.
Black body : It is a body which absorbs all incident light on it.
Grey body : Body which partially absorbs and partially
reflects incident light falling on it.
Fourier equation : It is the general equation guiding conduction
heat transfer.
Newton’s law of
cooling : It is the guiding principle behind convective heat
transfer.
Radiation
pyrometers : Measures temperature of a distant / hot object
without coming into contact with it.
Decimal reduction
time : Time required for reducing the microbial
population to one tenth of its initial number.
Microbial lethality : Time temperature combination to kill all
microorganisms including its spores.
28

Principles of Heat and
Mass Transfer
1.12 ANSWERS TO CHECK YOUR PROGRESS
EXERCISES
"
Check Your Progress Exercise 1
Your answers should include the following points:
1. The different methods of heat transfer are: Conduction, Convection and
Radiation.
2. The rate of conduction heat transfer increases as the temperature gradient
increases.
The equation, dQ/dt = kA dT/dT x is known as Fourier equation of heat
conduction.
3. The basic formula for radiant-heat transfer is the Stefan-Boltzmann
Law, q = ∈ σT
4
.
As indicated in the equation, the radiative heat flux q, is proportional to
the fourth power of temperature. That means for any increase in
temperature the flux increases much faster. The emissivity of the object,
∈, indicates its capacity in relation to a black body to emit thermal
radiation. The value of ∈ is in the range of 0-1; a black body has ∈ = 1
and a perfectly reflective body has ∈ = 0. The σ is Stefan-Boltzmann
constant.
4. If the temperature of one of the objects is doubled, it means the
temperature difference between the two objects has increased. Since
convection is directly proportional to the temperature difference, it will
increase in proportion to the temperature difference. On the other hand the
radiation heat transfer is proportional to the difference in the fourth power
of the temperatures of the two objects, the radiation heat transfer will
increase much more steeply. You can therefore, appreciate that the
radiation heat transfer increases much faster than the conduction or
convection when the temperature difference between two objects
increases.
Check Your Progress Exercise 2
Your answers should include the following points:
1. The acid and low-acid foods are distinguished on the basis of pH. The
foods with pH less than 4.6 are called acid and the foods with pH more
than 4.6 are called low-acid foods.
2. The thermal process for an acid food consists of treating it in a 100°C
boiling water bath, whereas the low-acid food must be pressure treated to a
temperature of 115°C or higher to kill the spoilage causing
microorganisms.
3. To make water boil at a temperature higher than 100°C in food processing,
it has to be put under pressure, such as in a pressure canner. When a food
29

Food Preservation by
Application of Heat
is processed at 1.0 kg/cm
2
pressure, the water boils when it gets to 115°C,
rather than at 100°C.
4. At higher altitudes the atmospheric pressure goes down and water boils at
lower temperatures. Thus, to make the thermal processing effective, either
the process time or canner pressure must be increased to make up for
lower boiling temperatures. That means pressure treatment may be
required even for acid foods.
Check Your Progress Exercise 3
Your answers should include the following points:
1. a) Mercury-in-Glass (MIG) Thermometer
b) Resistance temperature detector sensors
2. A thermocouple is made by joining two dissimilar metals. When one of
the junctions is at a different temperature than the surrounding
temperature, then a small voltage is developed which can then be
measured across the two leads at the other junction. When provision is
made in the circuit to take care of the reference point such as the freezing
point of water, then the resultant voltage is calibrated in terms of
temperature difference between the reference point and the temperature of
the junction.
3. A thermistor is normally a thermally sensitive material whose electrical
resistance changes with temperature. This change in resistance is
calibrated in terms of temperature. It differs from a resistance temperature
detection (RTD) sensor in terms of its sensitivity. As a result a thermistor
is able to sense very small changes in temperature as compared to a RTD
sensor.
4. K = (°C + 273), where K and C are units of temperature in Kelvin scale
and Celsius scale.
Check Your Progress Exercise 4
Your answers should include the following points:
1. The Decimal Reduction Time or D-value is defined as the time taken to
kill 90% of the microbes in a sample under specified conditions and at a
particular temperature. D-values are determined from survivor curves
when the log of population is plotted against time.
D
reference temperature
= Time/(Log
a
-Log
b
)
Where a = the initial population, and b = the surviving population after a
time interval.
2. The F value for a process is the number of minutes required to kill a
known population of microorganisms in a given food under specified
conditions. This F value is usually set at 12 D values and the resultant
microbial population is extremely low such as one microbe in 10,000 cans
(say).
30

Principles of Heat and
Mass Transfer
3. Clostridium botulinum is the reference microorganism, which is used in
determining the different parameters related to thermal processing.
4. Lethality is defined as the integrated influence of time and temperature on
a microbial population. It is expressed in time units for the process ac-
complished at the heating medium temperature. For e.g., a thermal process
may require 65 min at 115°C of steam temperature for a given food
product to achieve full lethality.
1.13 SOME USEFUL BOOKS
1. Henderson, S.M. and Perry, R.L. (1976) Agricultural Process Engineering.
AVI Publishing Co. West Port, Connecticut.
2. McCabe, W.L., Smith, J.C. and Harriott, P. (1993) Unit Operations of
Chemical Engineering. McGraw Hill, New York.
3. Nielsen, S.S. (1998) Introduction to Food Analysis. Aspen Publications
Inc., Maryland.
31
